Feasibility of Golden Angle Spiral Real-Time Phase Contrast MRI at 0.55T: A Single-Center Prospective Study
Abstract
1. Introduction
2. Methods
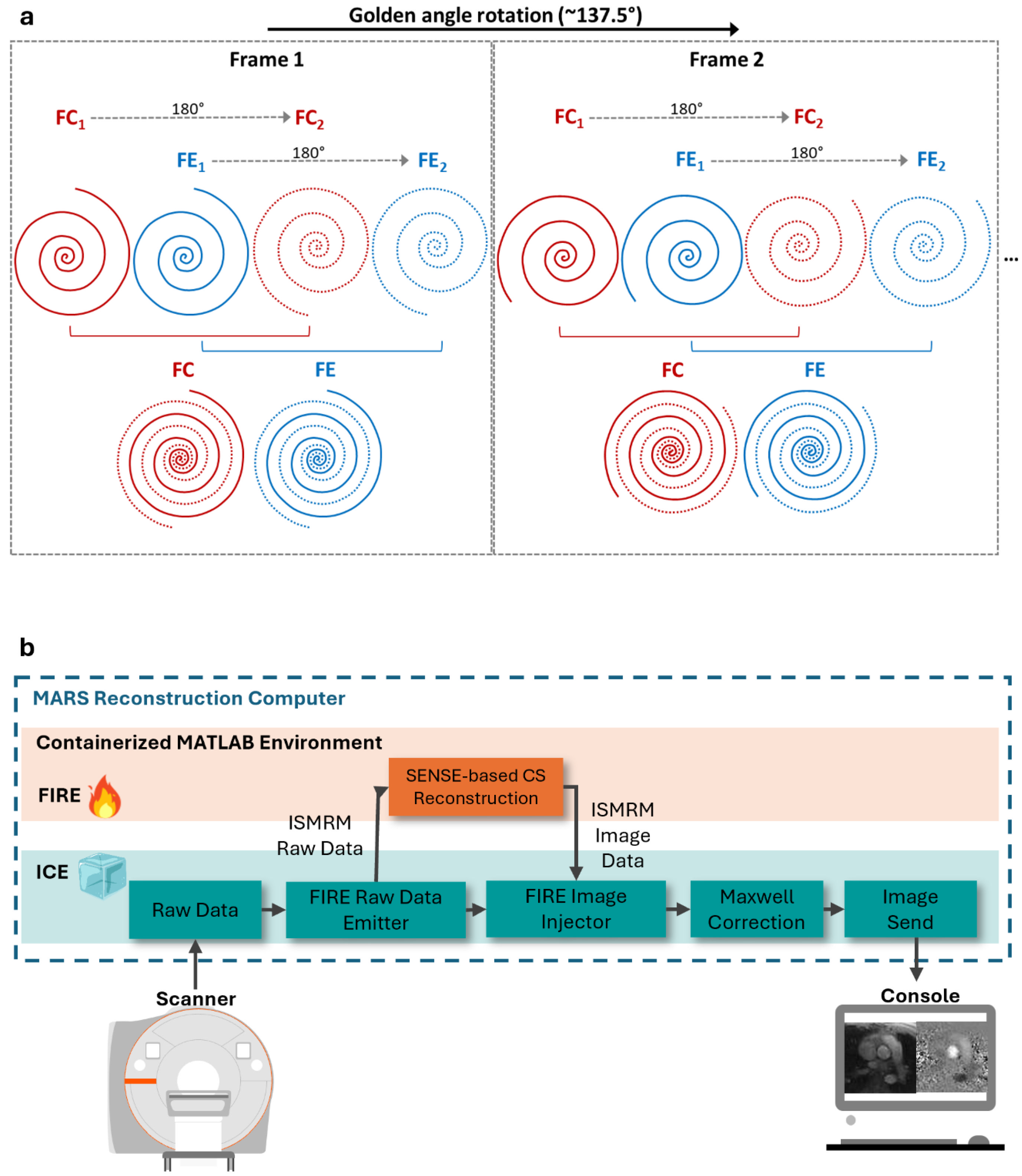
2.1. RT-PCMR Acquisition
2.2. RT-PCMR Reconstruction
2.3. Data Acquisition
2.4. Study Population
2.5. Data Analysis
2.6. Statistical Analysis
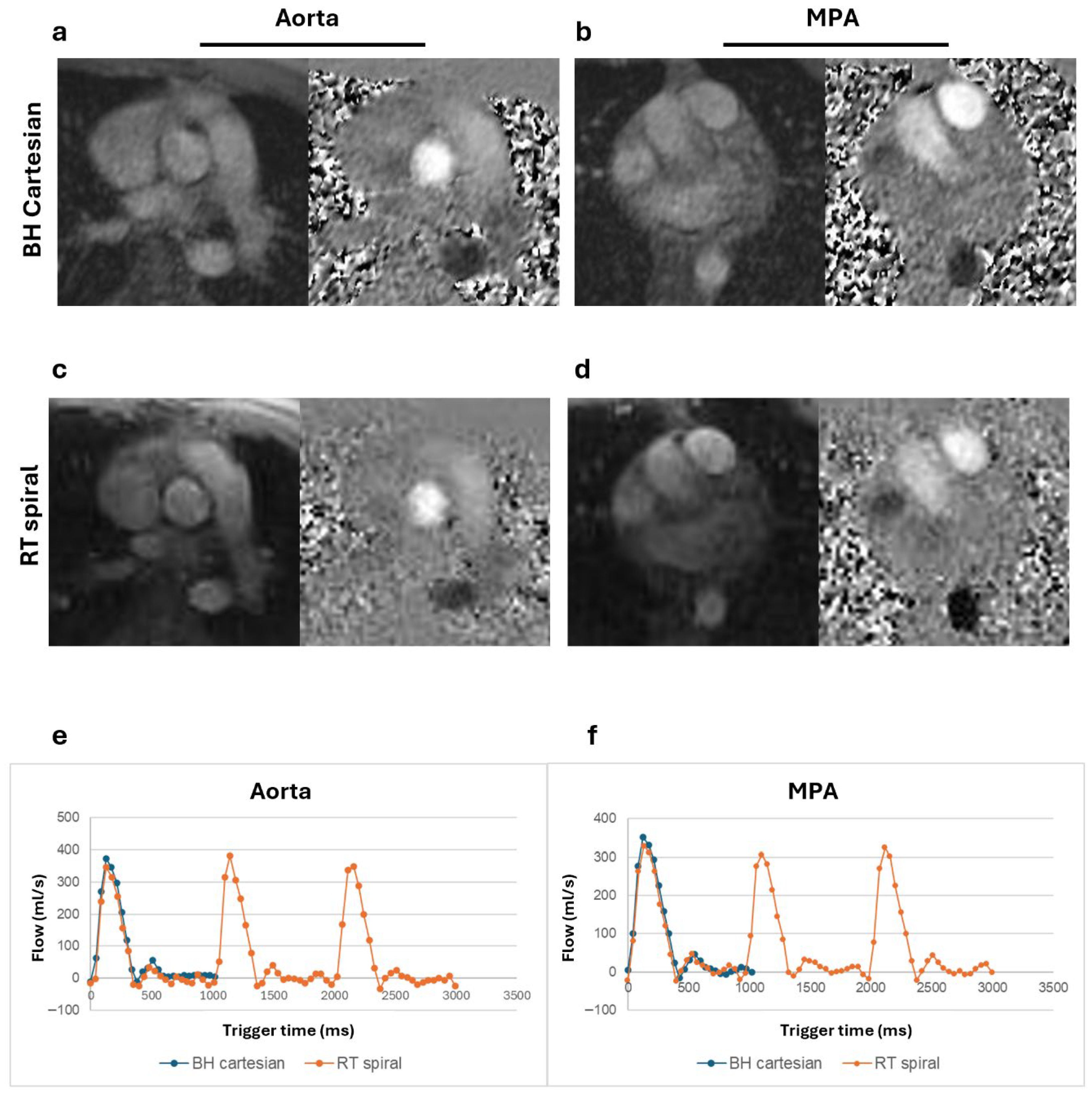
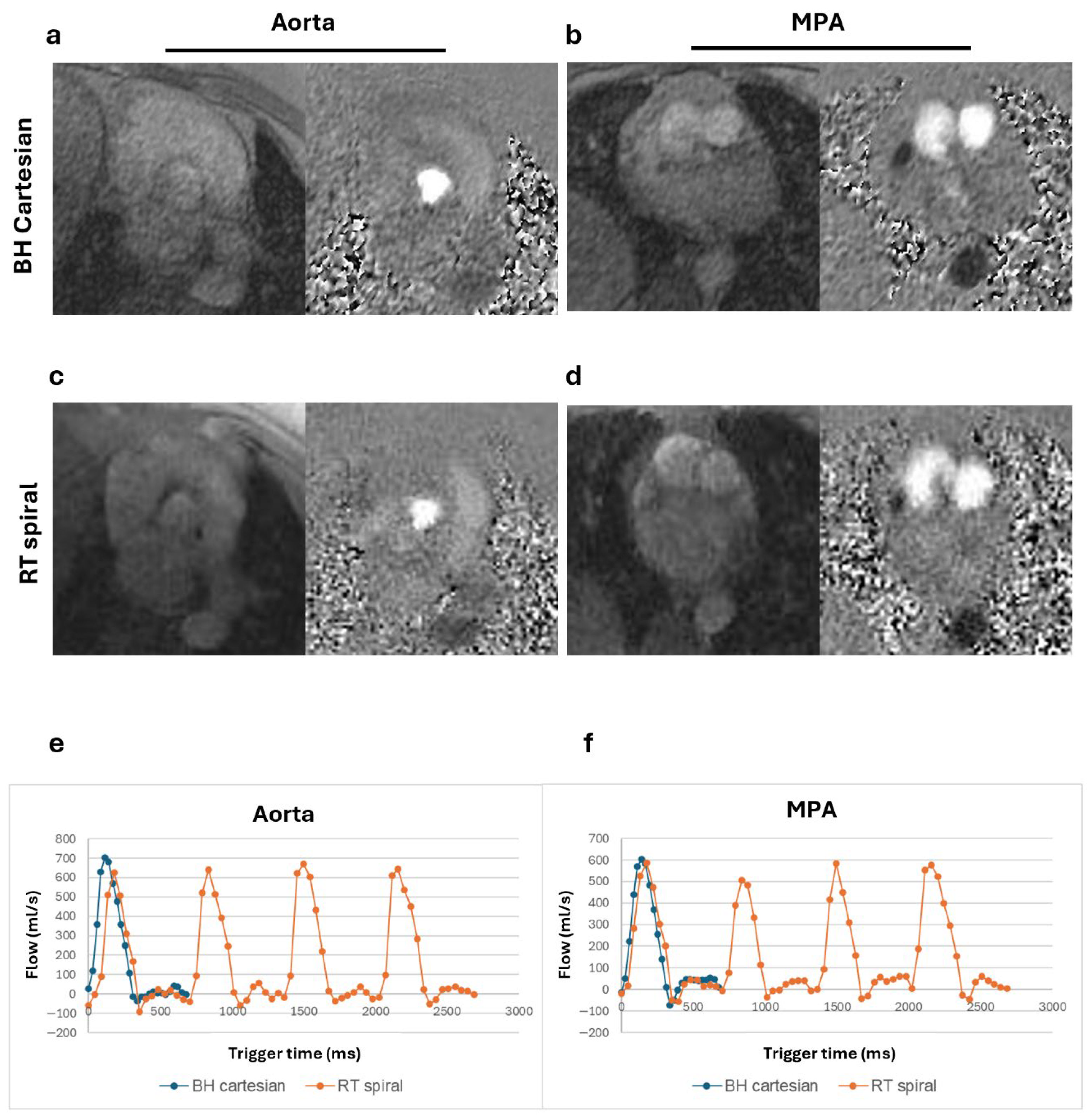
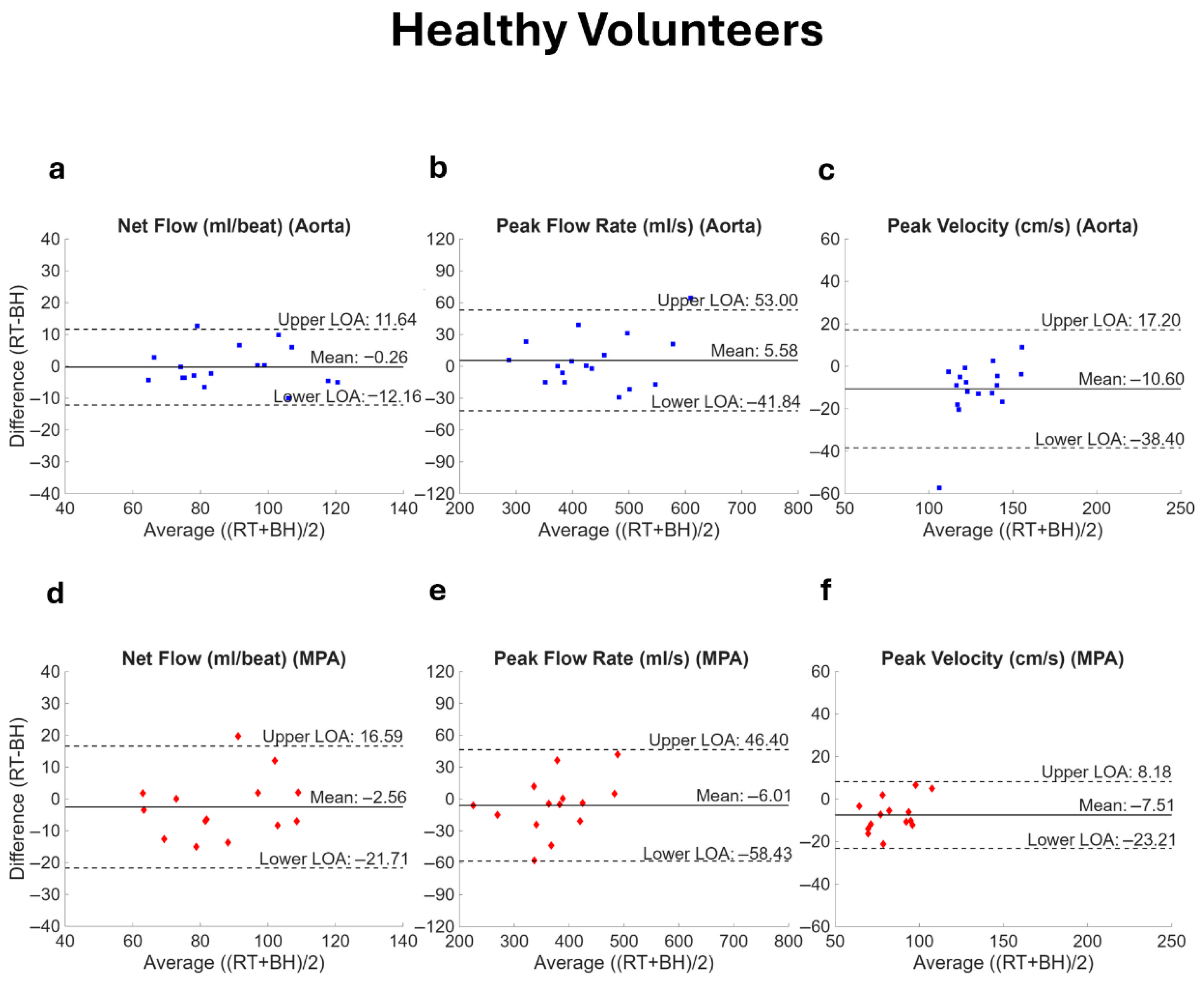
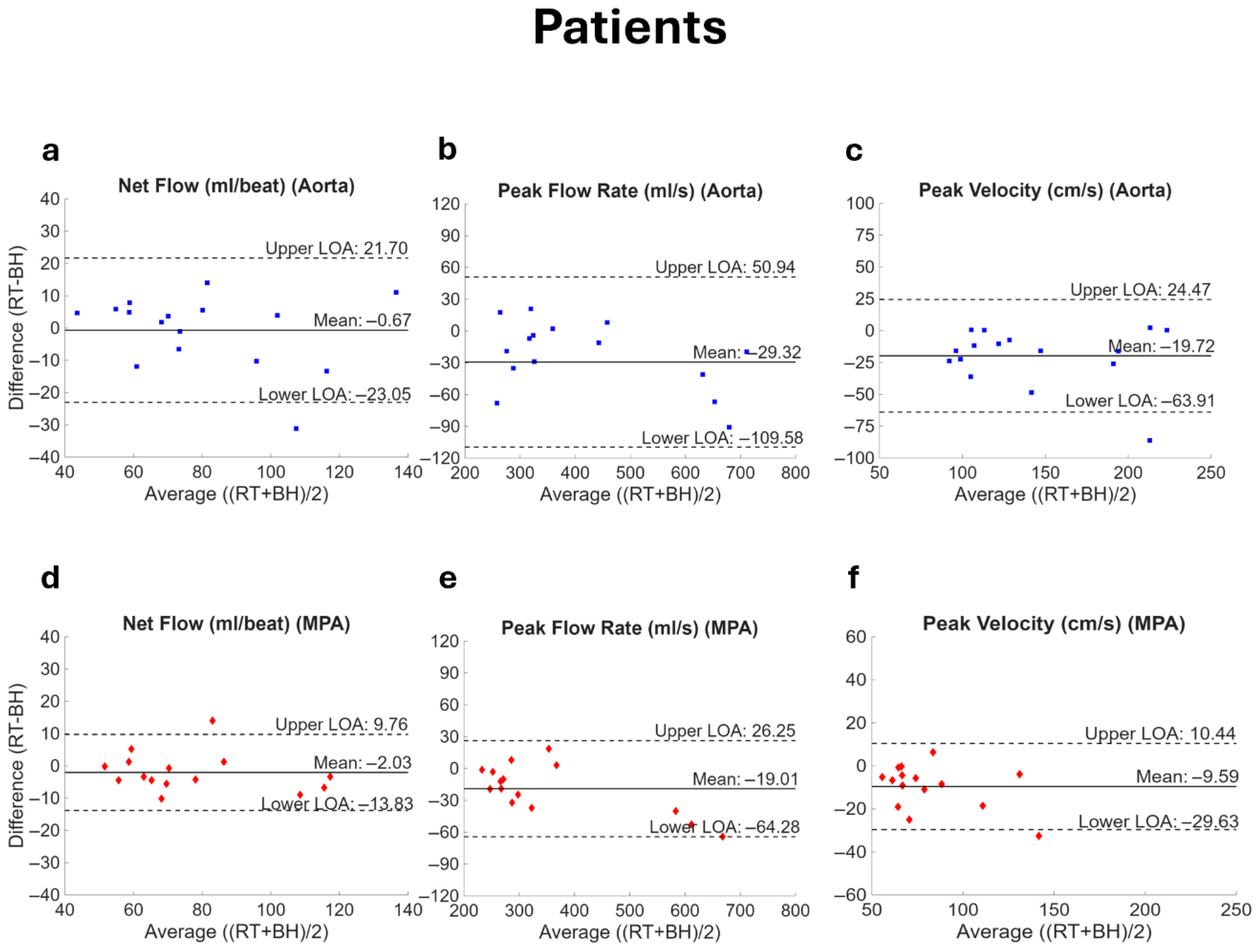
3. Results
4. Discussion
5. Study Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leiner, T.; Bogaert, J.; Friedrich, M.G.; Mohiaddin, R.; Muthurangu, V.; Myerson, S.; Powell, A.J.; Raman, S.V.; Pennell, D.J. SCMR Position Paper (2020) on clinical indications for cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2020, 22, 76. [Google Scholar] [CrossRef]
- Simonetti, O.P.; Ahmad, R. Low-Field Cardiac Magnetic Resonance Imaging: A Compelling Case for Cardiac Magnetic Resonance’s Future. Circ. Cardiovasc. Imaging 2017, 10, e005446. [Google Scholar] [CrossRef]
- Qin, C.; Murali, S.; Lee, E.; Supramaniam, V.; Hausenloy, D.J.; Obungoloch, J.; Brecher, J.; Lin, R.; Ding, H.; Akudjedu, T.N.; et al. Sustainable low-field cardiovascular magnetic resonance in changing healthcare systems. Eur. Heart J. Cardiovasc. Imaging 2022, 23, E246–E260. [Google Scholar] [CrossRef]
- Varghese, J.; Jin, N.; Giese, D.; Chen, C.; Liu, Y.; Pan, Y.; Nair, N.; Shalaan, M.T.; Khan, M.; Tong, M.S.; et al. Building a comprehensive cardiovascular magnetic resonance exam on a commercial 0.55 T system: A pictorial essay on potential applications. Front. Cardiovasc. Med. 2023, 10, 1120982. [Google Scholar] [CrossRef]
- Campbell-Washburn, A.E.; Ramasawmy, R.; Restivo, M.C.; Bhattacharya, I.; Basar, B.; Herzka, D.A.; Hansen, M.S.; Rogers, T.; Bandettini, W.P.; McGuirt, D.R.; et al. Opportunities in interventional and diagnostic imaging by using high-performance low-field-strength MRI. Radiology 2019, 293, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Bandettini, W.P.; Shanbhag, S.M.; Mancini, C.; McGuirt, D.R.; Kellman, P.; Xue, H.; Henry, J.L.; Lowery, M.; Thein, S.L.; Chen, M.Y.; et al. A comparison of cine CMR imaging at 0.55 T and 1.5 T. J. Cardiovasc. Magn. Reson. 2020, 22, 37. [Google Scholar] [CrossRef] [PubMed]
- McGrath, C.; Bieri, O.; Kozerke, S.; Bauman, G. Self-gated cine phase-contrast balanced SSFP flow quantification at 0.55 T. Magn. Reson. Med. 2024, 91, 174–189. [Google Scholar] [CrossRef] [PubMed]
- Nair, N.C.; Tong, M.S.; Gil, K.E.; Jin, N.; Han, Y.; Simonetti, O.P.; Varghese, J. Feasibility of Noncontrast 3D MR Angiography on a Commercial Wide-Bore 0.55-T System: Comparison with 1.5-T MR Angiography. Radiol. Cardiothorac. Imaging 2025, 7, e240252. [Google Scholar] [CrossRef]
- Bandettini, W.P.; Shanbhag, S.M.; Mancini, C.; Henry, J.L.; Lowery, M.; Chen, M.Y.; Xue, H.; Kellman, P.; Campbell-Washburn, A.E. Evaluation of Myocardial Infarction by Cardiovascular Magnetic Resonance at 0.55-T Compared to 1.5-T. JACC Cardiovasc. Imaging 2021, 14, 1866–1868. [Google Scholar] [CrossRef]
- Pelc, N.J.; Herfkens, R.J.; Shimakawa, A.; Enzmann, D.R. Phase Contrast Cine Magnetic Resonance Imaging. Magn. Reson. Q. 1991, 7, 229–254. [Google Scholar]
- Sun, A.; Zhao, B.; Li, Y.; He, Q.; Li, R.; Yuan, C. Real-time phase-contrast flow cardiovascular magnetic resonance with low-rank modeling and parallel imaging. J. Cardiovasc. Magn. Reson. 2017, 19, 19. [Google Scholar] [CrossRef] [PubMed]
- Guilfoyle, D.N.; Gibbs, P.; Ordidge, R.J.; Manwield, P. Real-time flow measurements using echo-planar imaging. Magn. Reson. Med. 1991, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gatehouse, P.D.; Firmin, D.N.; Collins, S.; Longmore, D.B. Real time blood flow imaging by spiral scan phase velocity mapping. Magn. Reson. Med. 1994, 31, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Nayak, K.S.; Pauly, J.M.; Kerr, A.B.; Hu, B.S.; Nishimura, D.G. Real-Time Color Flow MRI. Magn. Reson. Med. 2000, 43, 251–258. [Google Scholar] [CrossRef]
- Kowalik, G.T.; Knight, D.; Steeden, J.A.; Muthurangu, V. Perturbed spiral real-time phase-contrast MR with compressive sensing reconstruction for assessment of flow in children. Magn. Reson. Med. 2020, 83, 2077–2091. [Google Scholar] [CrossRef]
- Haji-Valizadeh, H.; Guo, R.; Kucukseymen, S.; Paskavitz, A.; Cai, X.; Rodriguez, J.; Pierce, P.; Goddu, B.; Kim, D.; Manning, W.; et al. Highly accelerated free-breathing real-time phase contrast cardiovascular MRI via complex-difference deep learning. Magn. Reson. Med. 2021, 86, 804–819. [Google Scholar] [CrossRef]
- Singh, V.; Pfeuffer, J.; Zhao, T.; Ress, D. Evaluation of spiral acquisition variants for functional imaging of human superior colliculus at 3T field strength. Magn. Reson. Med. 2018, 79, 1931–1940. [Google Scholar] [CrossRef]
- Pruessmann, K.P.; Weiger, M.; Scheidegger, M.B.; Boesiger, P. SENSE: Sensitivity encoding for fast MRI. Magn. Reson. Med. 1999, 42, 952–962. [Google Scholar] [CrossRef]
- Tian, Y.; Cui, S.X.; Lim, Y.; Lee, N.G.; Zhao, Z.; Nayak, K.S. Contrast-optimal simultaneous multi-slice bSSFP cine cardiac imaging at 0.55 T. Magn. Reson. Med. 2023, 89, 746–755. [Google Scholar] [CrossRef]
- Daudé, P.; Ramasawmy, R.; Javed, A.; Lederman, R.J.; Chow, K.; Campbell-Washburn, A.E. Inline automatic quality control of 2D phase-contrast flow MRI for subject-specific scan time adaptation. Magn. Reson. Med. 2024, 92, 751–760. [Google Scholar] [CrossRef]
- Varghese, J.; Goyal, A.; Han, Y.; Tong, M.; Jin, N.; Simonetti, O. Compressed Sensing Flow Quantification Validation on a Wide-bore 0.55T System in Volunteers and Patients. J. Cardiovasc. Magn. Reson. 2024, 26, 101018. [Google Scholar] [CrossRef]
- Lin, W.; Börnert, P.; Huang, F.; Duensing, G.R.; Reykowski, A. Generalized GRAPPA operators for wider spiral bands: Rapid self-calibrated parallel reconstruction for variable density spiral MRI. Magn. Reson. Med. 2011, 66, 1067–1078. [Google Scholar] [CrossRef] [PubMed]
- Winkelmann, S.; Schaeffter, T.; Koehler, T.; Eggers, H.; Doessel, O. An optimal radial profile order based on the golden ratio for time-resolved MRI. IEEE Trans. Med. Imaging 2007, 26, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Inati, S.J.; Naegele, J.D.; Zwart, N.R.; Roopchansingh, V.; Lizak, M.J.; Hansen, D.C.; Liu, C.; Atkinson, D.; Kellman, P.; Kozerke, S.; et al. ISMRM Raw data format: A proposed standard for MRI raw datasets. Magn. Reson. Med. 2017, 77, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.H.; Hu, B.S.; Nishimura, D.G.; Macovski, A. Fast Spiral Coronary Artery Imaging. Magn. Reson. Med. 1992, 28, 202–213. [Google Scholar] [CrossRef]
- Lim, Y.; Toutios, A.; Bliesener, Y.; Tian, Y.; Lingala, S.G.; Vaz, C.; Sorensen, T.; Oh, M.; Harper, S.; Chen, W. A multispeaker dataset of raw and reconstructed speech production real-time MRI video and 3D volumetric images. Sci. Data 2021, 8, 187. [Google Scholar] [CrossRef]
- Uecker, M.; Lai, P.; Murphy, M.J.; Virtue, P.; Elad, M.; Pauly, J.M.; Vasanawala, S.S.; Lustig, M. ESPIRiT—An eigenvalue approach to autocalibrating parallel MRI: Where SENSE meets GRAPPA. Magn. Reson. Med. 2014, 71, 990–1001. [Google Scholar] [CrossRef]
- Morales, M.A.; Ghanbari, F.; Demirel, Ö.B.; Street, J.A.; Wallace, T.E.; Davids, R.; Rodriguez, J.; Johnson, S.; Pierce, P.; Manning, W.J.; et al. Accelerated phase-contrast magnetic resonance imaging with use of resolution enhancement generative adversarial neural network. J. Cardiovasc. Magn. Reson. 2025, 27, 101128. [Google Scholar] [CrossRef]
- Schulz-Menger, J.; Bluemke, D.A.; Bremerich, J.; Flamm, S.D.; Fogel, M.A.; Friedrich, M.G.; Kim, R.J.; von Knobelsdorff-Brenkenhoff, F.; Kramer, C.M.; Pennell, D.J.; et al. Standardized image interpretation and post-processing in cardiovascular magnetic resonance—2020 update: Society for Cardiovascular Magnetic Resonance (SCMR): Board of Trustees Task Force on Standardized Post-Processing. J. Cardiovasc. Magn. Reson. 2020, 22, 19. [Google Scholar] [CrossRef]
- Traber, J.; Wurche, L.; Dieringer, M.A.; Utz, W.; von Knobelsdorff-Brenkenhoff, F.; Greiser, A.; Jin, N.; Schulz-Menger, J. Real-time phase contrast magnetic resonance imaging for assessment of haemodynamics: From phantom to patients. Eur. Radiol. 2016, 26, 986–996. [Google Scholar] [CrossRef]
- Macdonald, J.A.; François, C.J.; Forouzan, O.; Chesler, N.C.; Wieben, O. MRI assessment of aortic flow in patients with pulmonary arterial hypertension in response to exercise. BMC Med. Imaging 2018, 18, 55. [Google Scholar] [CrossRef] [PubMed]
- Kowallick, J.T.; A Joseph, A.; Unterberg-Buchwald, C.; Fasshauer, M.; van Wijk, K.; Merboldt, K.D.; Voit, D.; Frahm, J.; Lotz, J.; Sohns, J.M. Real-time phase-contrast flow MRI of the ascending aorta and superior vena cava as a function of intrathoracic pressure (Valsalva manoeuvre). Br. J. Radiol. 2014, 87, 20140401. [Google Scholar] [CrossRef] [PubMed]
- Xiong, F.; Emrich, T.; Schoepf, U.J.; Jin, N.; Hall, S.; Ruddy, J.M.; Giese, D.; Lautenschlager, C.; Emrich, A.L.; Varga-Szemes, A. Highly accelerated free-breathing real-time 2D flow imaging using compressed sensing and shared velocity encoding. Eur. Radiol. 2024, 34, 1692–1703. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Ramasawmy, R.; Seemann, F.; Peters, D.C.; Campbell-Washburn, A.E. Balanced steady-state free precession phase contrast at 0.55T applied to aortic flow. J. Cardiovasc. Magn. Reson. 2024, 26, 101098. [Google Scholar] [CrossRef]
- Santini, F.; Pansini, M.; Hrabak-Paar, M.; Yates, D.; Langenickel, T.H.; Bremerich, J.; Bieri, O.; Schubert, T. On the optimal temporal resolution for phase contrast cardiovascular magnetic resonance imaging: Establishment of baseline values. J. Cardiovasc. Magn. Reson. 2020, 22, 72. [Google Scholar] [CrossRef]
- Kramer, C.M.; Barkhausen, J.; Bucciarelli-Ducci, C.; Flamm, S.D.; Kim, R.J.; Nagel, E. Standardized cardiovascular magnetic resonance imaging (CMR) protocols: 2020 update. J. Cardiovasc. Magn. Reson. 2020, 22, 17. [Google Scholar] [CrossRef]
- Jaubert, O.; Steeden, J.; Montalt-Tordera, J.; Arridge, S.; Kowalik, G.T.; Muthurangu, V. Deep artifact suppression for spiral real-time phase contrast cardiac magnetic resonance imaging in congenital heart disease. Magn. Reson. Imaging 2021, 83, 125–132. [Google Scholar] [CrossRef]
- Thavendiranathan, P.; Verhaert, D.; Walls, M.C.; Bender, J.A.; Rajagopalan, S.; Chung, Y.-C.; Simonetti, O.P.; Raman, S.V. Simultaneous right and left heart real-time, free-breathing CMR flow quantification identifies constrictive physiology. JACC Cardiovasc. Imaging 2012, 5, 15–24. [Google Scholar] [CrossRef]
- DiCarlo, A.L.; Haji-Valizadeh, H.; Passman, R.; Greenland, P.; McCarthy, P.; Lee, D.C.; Kim, D.; Markl, M. Assessment of Beat-To-Beat Variability in Left Atrial Hemodynamics Using Real Time Phase Contrast MRI in Patients with Atrial Fibrillation. J. Magn. Reson. Imaging 2023, 58, 763–771. [Google Scholar] [CrossRef]
- Cherry, M.; Khatir, Z.; Khan, A.; Bissell, M. The impact of 4D-Flow MRI spatial resolution on patient-specific CFD simulations of the thoracic aorta. Sci. Rep. 2022, 12, 15128. [Google Scholar] [CrossRef]
- Joseph, A.A.; Merboldt, K.; Voit, D.; Zhang, S.; Uecker, M.; Lotz, J.; Frahm, J. Real-time phase-contrast MRI of cardiovascular blood flow using undersampled radial fast low-angle shot and nonlinear inverse reconstruction. NMR Biomed. 2012, 25, 917–924. [Google Scholar] [CrossRef]
- Kawel-Boehm, N.; Hetzel, S.J.; Ambale-Venkatesh, B.; Captur, G.; Chin, C.W.; François, C.J.; Jerosch-Herold, M.; Luu, J.M.; Raisi-Estabragh, Z.; Starekova, J.; et al. Society for Cardiovascular Magnetic Resonance reference values (“normal values”) in cardiovascular magnetic resonance: 2025 update. J. Cardiovasc. Magn. Reson. 2025, 27, 101853. [Google Scholar] [CrossRef]
- Kopanoglu, E.; Steckner, M.; Hoff, M.N.; Campbell-Washburn, A.E.; Webb, A.G.; Reeder, S.B.; Gulani, V. The ISMRM Safety Committee. MRI and Implant Safety at Low-Field and Ultralow-Field Strengths. J. Magn. Reson. Imaging 2025, 63, 364–377. [Google Scholar] [CrossRef]


| Parameters | Breath-Held Cartesian | Real-Time Spiral |
|---|---|---|
| TE/TR (ms) | 3.71/6.7 | 3/11 |
| FOV (Aorta) (mm × mm) | (240–420) × (225 × 436) | 300 × 300 |
| FOV (MPA) (mm × mm) | (262–436) × (225 × 440) | 300 × 300 |
| Spatial resolution (Aorta) (mm × mm) | (1.2–2.0) × (1.7–3.4) | 2.3 × 2.3 |
| Spatial resolution (MPA) (mm × mm) | (1.3–2.1) × (1.7–3.1) | 2.3 × 2.3 |
| Temporal resolution (ms) | 53 | 44 |
| Slice thickness (mm) | 8 | 8 |
| Acceleration factor (R) | 2 | 6 |
| Bandwidth (Hz/Px) | 401 | 1116 |
| Flip angle (degrees) | 12 | 20 |
| Acquisition time | 10 heart beats | 4 s |
| VENC Aorta (cm/s) | 200–250 | 150–250 |
| VENC MPA (cm/s) | 150–200 | 150–200 |
| Patient | Age (Years) | Sex | BMI (kg/m2) | Indications for CMR Evaluation |
|---|---|---|---|---|
| 1 | 65 | F | 26.3 | Chest pain and dyspnea on exertion. |
| 2 | 22 | M | 28.2 | Multiple arrythmias. History of supraventricular tachycardia, NSVT |
| 3 | 51 | F | 16.8 | Suspected cardiac dysfunction in the setting of PVCs, inconclusive echocardiogram. |
| 4 | 21 | F | 29.2 | Left ventricular dysfunction, abnormal echocardiogram. |
| 5 | 48 | F | 24.3 | Regional wall motion abnormalities. |
| 6 | 32 | F | 18.6 | NSVT. |
| * 7 | 67 | M | 32.3 | Renal amyloidosis. |
| 8 | 19 | F | 27 | Suspected bicuspid aortic valve. |
| 9 | 57 | F | 25.2 | Serial aortic surveillance, Takotsubo cardiomyopathy patient. |
| 10 | 47 | M | 52.8 | Hypertrophic cardiomyopathy, abnormal resting echocardiogram. |
| 11 | 34 | M | 48.8 | Suspected dilated aortic root and ascending aorta. |
| 12 | 55 | M | 29.5 | Cardiac viability in the setting of multiple comorbidities, claustrophobic. |
| 13 | 60 | F | 64 | Rule out infiltrative disease. |
| 14 | 67 | M | 48 | Suspected aortic dilation, high PVC burden present. |
| 15 | 63 | F | 30 | Evaluate aortic and mitral regurgitation, sub-aortic membrane. |
| 16 | 26 | F | 21.3 | Follow-up for hypertrophic cardiomyopathy. |
| 17 | 66 | F | 41.9 | Evaluation for left ventricular hypertrophy. |
| BH-PCMR | RT-PCMR | ICC (95%CI) | p-Value (ICC) | |
|---|---|---|---|---|
| Healthy Volunteers | ||||
| Net flow (ml/beat) | ||||
| Aorta (n = 17) | 89.4 ± 17.8 | 89.2 ± 17.5 | 0.97 (0.92–0.99) | <0.001 |
| MPA (n = 14) | 87.7 ± 15.6 | 85.1 ± 17.7 | 0.91 (0.71–0.97) | <0.001 |
| Peak flow rate (ml/s) | ||||
| Aorta (n = 17) | 434.5 ± 87.0 | 440.1 ± 93.2 | 0.98 (0.95–0.99) | <0.001 |
| MPA (n = 14) | 374.4 ± 67.8 | 368.4 ± 77.8 | 0.96 (0.90–0.99) | <0.001 |
| Peak positive velocity (cm/s) | ||||
| Aorta (n = 17) | 134.5 ± 12.7 | 125.2 ± 16.6 | 0.79 (0.12–0.93) | <0.001 |
| MPA (n = 14) | 87.5 ± 11.9 | 80.0 ± 15.3 | 0.84 (0.21–0.96) | <0.001 |
| Qp/Qs (n = 14) | ||||
| 1.00 ± 0.10 | 0.95 ± 0.08 | 0.56 (−0.18–0.85) | 0.056 | |
| Patients | ||||
| Net flow (ml/beat) | ||||
| Aorta (n = 16) | 80.4 ± 27.3 | 79.8 ± 24.4 | 0.95 (0.86–0.98) | <0.001 |
| MPA (n = 15) | 77.7 ± 22.4 | 75.7 ± 21.1 | 0.98 (0.94–0.99) | <0.001 |
| Peak flow rate (ml/s) | ||||
| Aorta (n = 16) | 433.3 ± 168.3 | 403.9 ± 154.6 | 0.98 (0.89–0.99) | <0.001 |
| MPA (n = 15) | 363.7 ± 152.1 | 344.7 ± 136.0 | 0.99 (0.94–0.99) | <0.001 |
| Peak positive velocity (cm/s) | ||||
| Aorta (n = 16) | 153.1 ± 50.6 | 133.3 ± 46.7 | 0.91 (0.50–0.97) | <0.001 |
| MPA (n = 15) | 86.4 ± 28.6 | 76.8 ± 24.3 | 0.93 (0.55–0.98) | <0.001 |
| Qp/Qs (n = 15) | ||||
| 1.03 ± 0.14 | 1.01 ± 0.13 | 0.71 (0.13–0.90) | 0.015 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Pervaiz, S.; Chen, C.; Liu, Y.; Binzel, K.; Chow, K.; Ahmad, R.; Han, Y.; Simonetti, O.P.; Jin, N.; Varghese, J. Feasibility of Golden Angle Spiral Real-Time Phase Contrast MRI at 0.55T: A Single-Center Prospective Study. Bioengineering 2026, 13, 166. https://doi.org/10.3390/bioengineering13020166
Pervaiz S, Chen C, Liu Y, Binzel K, Chow K, Ahmad R, Han Y, Simonetti OP, Jin N, Varghese J. Feasibility of Golden Angle Spiral Real-Time Phase Contrast MRI at 0.55T: A Single-Center Prospective Study. Bioengineering. 2026; 13(2):166. https://doi.org/10.3390/bioengineering13020166
Chicago/Turabian StylePervaiz, Salman, Chong Chen, Yingmin Liu, Katherine Binzel, Kelvin Chow, Rizwan Ahmad, Yuchi Han, Orlando P. Simonetti, Ning Jin, and Juliet Varghese. 2026. "Feasibility of Golden Angle Spiral Real-Time Phase Contrast MRI at 0.55T: A Single-Center Prospective Study" Bioengineering 13, no. 2: 166. https://doi.org/10.3390/bioengineering13020166
APA StylePervaiz, S., Chen, C., Liu, Y., Binzel, K., Chow, K., Ahmad, R., Han, Y., Simonetti, O. P., Jin, N., & Varghese, J. (2026). Feasibility of Golden Angle Spiral Real-Time Phase Contrast MRI at 0.55T: A Single-Center Prospective Study. Bioengineering, 13(2), 166. https://doi.org/10.3390/bioengineering13020166







