The Effect of Thermocycling on the Microhardness of Contemporary Glass Ionomer-Based Restorative Materials: An In Vitro Study
Abstract
1. Introduction
2. Material and Method
2.1. Study Design and Ethical Considerations
2.2. Materials
2.3. Specimen Preparation
- •
- Group 1: Beautifil Bulk Restorative;
- ○
- 1a: before thermocycling.
- ○
- 1b: after thermocycling.
- •
- Group 2: EQUIA Forte HT;
- ○
- 2a: before thermocycling.
- ○
- 2b: after thermocycling.
- •
- Group 3: Fuji II LC;
- ○
- 3a: before thermocycling.
- ○
- 3b: after thermocycling.
- •
- Teflon molds (diameter: 10 mm, height: 2 mm) were used to prepare 30 specimens per group. To minimize potential batch effects, all specimens were fabricated by the same operator, using identical molds, materials from the same batch numbers, and the same light-curing unit, within a single preparation period.
- •
- The setting and curing times for each restorative material were selected in strict accordance with the manufacturers’ instructions for a 2-mm material thickness to ensure complete setting and optimal polymerization. Since surface microhardness is highly dependent on the degree of polymerization, curing time and light intensity were carefully standardized for all specimens to allow reliable inter-material comparison.
- •
- Beautifil Bulk Restorative: The material was applied using a mouth spatula and light-cured with an LED unit (Bluephase G4, Ivoclar Vivadent, Liechtenstein) at 1500 mW/cm2 for 10 s per surface.
- •
- EQUIA Forte HT and Fuji II LC: They were mixed in an amalgamator (Rock-mix, Dentmark, China) for 10 s, placed in molds, and light-cured for 20 s per surface using LED.
- •
- The output irradiance of the LED curing unit was verified using a calibrated radiometer prior to specimen preparation to ensure consistent and accurate light intensity throughout the study.
- •
- A transparent strip (Universal strips, Extra Dental, Istanbul, Türkiye) and a glass slide were placed over the molds during setting to obtain smooth and standardized surfaces. After setting, all specimens were polished using rubber polishing discs (Astropol, Ivoclar Vivadent, Schaan, Liechtenstein) in a sequential order (gray, green, and pink) for 30 s per surface under water cooling at approximately 10,000 rpm. Following polishing, specimens were stored in distilled water at 37 °C for 24 h prior to microhardness testing (Figure 1).
2.4. Thermocycling Procedure
2.5. Microhardness Evaluation with Vickers Method
2.6. Statistical Analysis
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Colak, H.; Dulgergil, C.T.; Dalli, M.; Hamidi, M.M. Early childhood caries update: A review of causes, diagnoses, and treatments. J. Nat. Sc. Biol. Med. 2013, 4, 29. [Google Scholar]
- Sengul, F.; Urvasizoglu, G.; Derelioglu, S.; Seddik, T.; Celikel, P.; Bas, A. Early childhood caries in 4-to 5-year-old children in Erzurum, Turkey. Front. Public Health 2021, 9, 725501. [Google Scholar] [CrossRef]
- Sidhu, S.K.; Nicholson, J.W. A review of glass-ionomer cements for clinical dentistry. J. Funct. Biomater. 2016, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.W.; Sidhu, S.K.; Czarnecka, B. Fluoride exchange by glass-ionomer dental cements and its clinical effects: A review. Biomater. Investig. Dent. 2023, 10, 2244982. [Google Scholar] [CrossRef]
- Bas, A.; Sheikholaemeh, N.O.; Sengul, F.; Sarac, F.; Derelioglu, S.S.; Celikel, P. Effect of thickness on irradiance loss and temperature rise in indirect restorative materials: An in vitro study. BMC Oral Health 2025, 25, 716. [Google Scholar] [CrossRef]
- Ahmad, Z.H.; Anil, S.; Chalisserry, E.P.; Almuhaiza, M.; AlZailai, A.M. Study of the mechanical properties of the novel zirconia-reinforced glass ionomer cement. J. Contemp. Dent. Pract. 2016, 17, 394–398. [Google Scholar] [CrossRef]
- Obeid, A.; Lopez, A.; Candido, A.; Forcin, L.; Cristina Margarido Brondino, N.; Mondelli, R.F.L.; Alhotan, A.; Silikas, N.; Velo, M. Evaluating the physical-mechanical properties of flowable fiber-reinforced and bulk-fill Giomer composites: A comparative study of advanced technologies. Front. Dent. Med. 2025, 6, 1634533. [Google Scholar]
- Kimyai, S.; Mashayekhi, Z.; Mohammadi, N.; Bahari, M.; Kahnamouei, M.A.; Chaharom, M.E.E. Comparison of the effect of preheating on the flexural strength of giomer and nanohybrid composite resin. J. Dent. Res. Dent. Clin. Dent. Prospect. 2022, 16, 159. [Google Scholar] [CrossRef] [PubMed]
- Celikel, P.; Sengul, F. Investigating the impact of post-curing cycles on surface hardness and color stability in 3D printed resin crowns. Odontology 2025, 113, 156–162. [Google Scholar] [CrossRef]
- Kaya, N.; Sasany, R.; Yanıkoglu, N.; Tosun, B. Investigation of color and physicomechanical properties of peek and pekk after storage in a different medium. Sci. Rep. 2024, 14, 5357. [Google Scholar] [CrossRef]
- Collado-González, M.; Pecci-Lloret, M.R.; Tomás-Catalá, C.J.; García-Bernal, D.; Oñate-Sánchez, R.E.; Llena, C.; Forner, L.; Rosa, V.; Rodríguez-Lozano, F.J. Thermo-setting glass ionomer cements promote variable biological responses of human dental pulp stem cells. Dent. Mater. 2018, 34, 932–943. [Google Scholar] [CrossRef]
- Ravi, B.; Paulraj, J.; Maiti, S.; Shanmugam, R. Assessing the influence of thermocycling on compressive strength, flexural strength, and microhardness in green-mediated nanocomposite-enhanced glass ionomer cement compared to traditional glass ionomer cement. Cureus 2024, 16, e56078. [Google Scholar] [CrossRef]
- Buldur, M.; Karaarslan, E.S. Microhardness of glass carbomer and high-viscous glass Ionomer cement in different thickness and thermo-light curing durations after thermocycling aging. BMC Oral Health 2019, 19, 273. [Google Scholar] [CrossRef]
- Fidan, M.; Dereli, Z. Evaluation of the effect of two polishing techniques and thermocycling process on surface roughness, hardness, and color stability of composites. J. Dent. Indones. 2022, 29, 8–16. [Google Scholar] [CrossRef]
- Kara, O.; Sarı, T.; Ozyılmaz, O.Y.; Kurugol, P.İ.; Kara, H.B. Effect of thermal and mechanical load cycling on dentin bond strength of a self-etch resin luting cement. Selcuk. Dent. J. 2017, 4, 36–43. [Google Scholar]
- Chesterman, J.; Jowett, A.; Gallacher, A.; Nixon, P. Bulk-fill resin-based composite restorative materials: A review. Brit. Dent. J. 2017, 222, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.S. Effects of surface pre-reacted glass particles on fluoride release of dental restorative materials. World Appl. Sci. J. 2013, 28, 457–462. [Google Scholar]
- Desai, S.; Chandler, N. Calcium Hydroxide-Based Root Canal Sealers: A Review. J. Endodont. 2009, 35, 475–480. [Google Scholar] [CrossRef]
- Khoroushi, M.; Keshani, F. A review of glass-ionomers: From conventional glass-ionomer to bioactive glass-ionomer. Dent. Res. J. 2013, 10, 411–420. [Google Scholar]
- Abuzinadah, A.J.; Merdad, Y.M.; Aldharrab, R.S.; Almutairi, W.A.; Yeslam, H.E.; Hasanain, F.A. Microhardness and compressive strength of bulk fill glass hybrid material and other direct restorative materials. J. Compos. Sci. 2024, 8, 508. [Google Scholar] [CrossRef]
- Yap, A.U.J.; Tan, S.H.L.; Wee, S.S.C.; Lee, C.T.; Lim, E.L.C.; Zeng, K.Y. Chemical degradation of composite restoratives. J. Oral. Rehabil. 2001, 28, 1015–1021. [Google Scholar] [CrossRef]
- Bernardo, M.; Luis, H.; Martin, M.D.; Leroux, B.G.; Rue, T.; Leitao, J.; DeRouen, T.A. Survival and reasons for failure of amalgam versus composite posterior restorations placed in a randomized clinical trial. J. Am. Dent. Assoc. 2007, 138, 775–783. [Google Scholar] [CrossRef]
- Gale, M.S.; Darvell, B.W. Thermal cycling procedures for laboratory testing of dental restorations. J. Dent. 1999, 27, 89–99. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, S.; Fan, J.; Wang, K.; Huang, J.; Wang, J.; Zhang, S.; Tao, G.; Zhang, Z.-J. Surface Coatings of Glass Fiber: Exploring Emerging Functionalities and Beyond. Adv. Fiber Mater. 2025, 7, 1731–1765. [Google Scholar] [CrossRef]
- Alaida, W.S.; Gadi, S.A.; Al-Ghannam, R.E.; Alamri, M.F.; Mirdad, F.I.; Argaibeh, R.M.; Alqahtani, B.A.; Alqahtani, A.M.; Al Jaban, A.A.; Alkuraydimi, T.M. Biomechanical and Occlusal Factors Influencing the Longevity of Single-Unit Restorations: A Comprehensive Review. Cureus 2025, 17, e85998. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, W.; Wang, L.; Wang, X.; Wei, H.; Xu, Q.; Guan, R. A Comparative Study on the Contact Fatigue Failure Mechanisms of Mn-Cr Series and Cr-Mo Series Gear Steels Based on Surface Integrity and Damage Morphology. Coatings 2025, 15, 1452. [Google Scholar] [CrossRef]
- Yanikoglu, N.D.; Sakarya, R.E. Test methods used in the evaluation of the structure features of the restorative materials: A literature review. J. Mater. Res. Technol. 2020, 9, 9720–9734. [Google Scholar] [CrossRef]
- Carneiro, E.R.; Coelho, A.S.; Amaro, I.; Paula, A.B.; Marto, C.M.; Saraiva, J.; Ferreira, M.M.; Vilhena, L.; Ramalho, A.; Carrilho, E. Mechanical and tribological characterization of a bioactive composite resin. Appl. Sci. 2021, 11, 8256. [Google Scholar] [CrossRef]
- Xu, S.; Chen, J.W.; Roggenkamp, C.L.; Zhang, W. Evaluating Bonding Agent’s Effect in Microleakage of a Bioactive Restorative Material with Thermocycling and PH Challenge Test. Taiwan J. Pediatr. Dent. 2019, 19, 133–142. [Google Scholar]
- Sauro, S.; Makeeva, I.; Faus-Matoses, V.; Foschi, F.; Giovarruscio, M.; Maciel Pires, P.; Martins Moura, M.E.; Almeida Neves, A.; Faus-Llácer, V. Effects of ions-releasing restorative materials on the dentine bonding longevity of modern universal adhesives after load-cycle and prolonged artificial saliva aging. Materials 2019, 12, 722. [Google Scholar] [CrossRef] [PubMed]
- Taha, N.; Ghanim, A.; Tavangar, M. Comparison of mechanical properties of resin composites with resin modified glass ionomers. J. Dent. Biomater. 2015, 2, 47–53. [Google Scholar]
- Bilge, K.; Aşar, E.M.; İpek, İ. Evaluation of Flexural Strength and Microhardness of Different Type Glass Ionomer Cements. Eur. Ann. Dent. Sci. 2024, 51, 10–14. [Google Scholar] [CrossRef]

| Material | Composition | Manufacturer |
|---|---|---|
| Beautifil Bulk Restorative | Giomer-based, containing Bis-GMA, UDMA, Bis-MPEPP, TEGDMA, and surface pre-reacted glass (S PRG) fillers; includes polymerization initiators, pigments, and additives | SHOFU Inc., Kyoto, Japan |
| EQUIA Forte HT | High-viscosity glass ionomer reinforced with fluoroaluminosilicate glass, polyacrylic acid, iron oxide, polybasic carboxylic acid, and water | GC Co.,Tokyo, Japan |
| Fuji II LC | Resin-modified GIC containing HEMA, TEGDMA, polyacrylic acid, and fluoroaluminosilicate glass | GC Co., Tokyo, Japan |
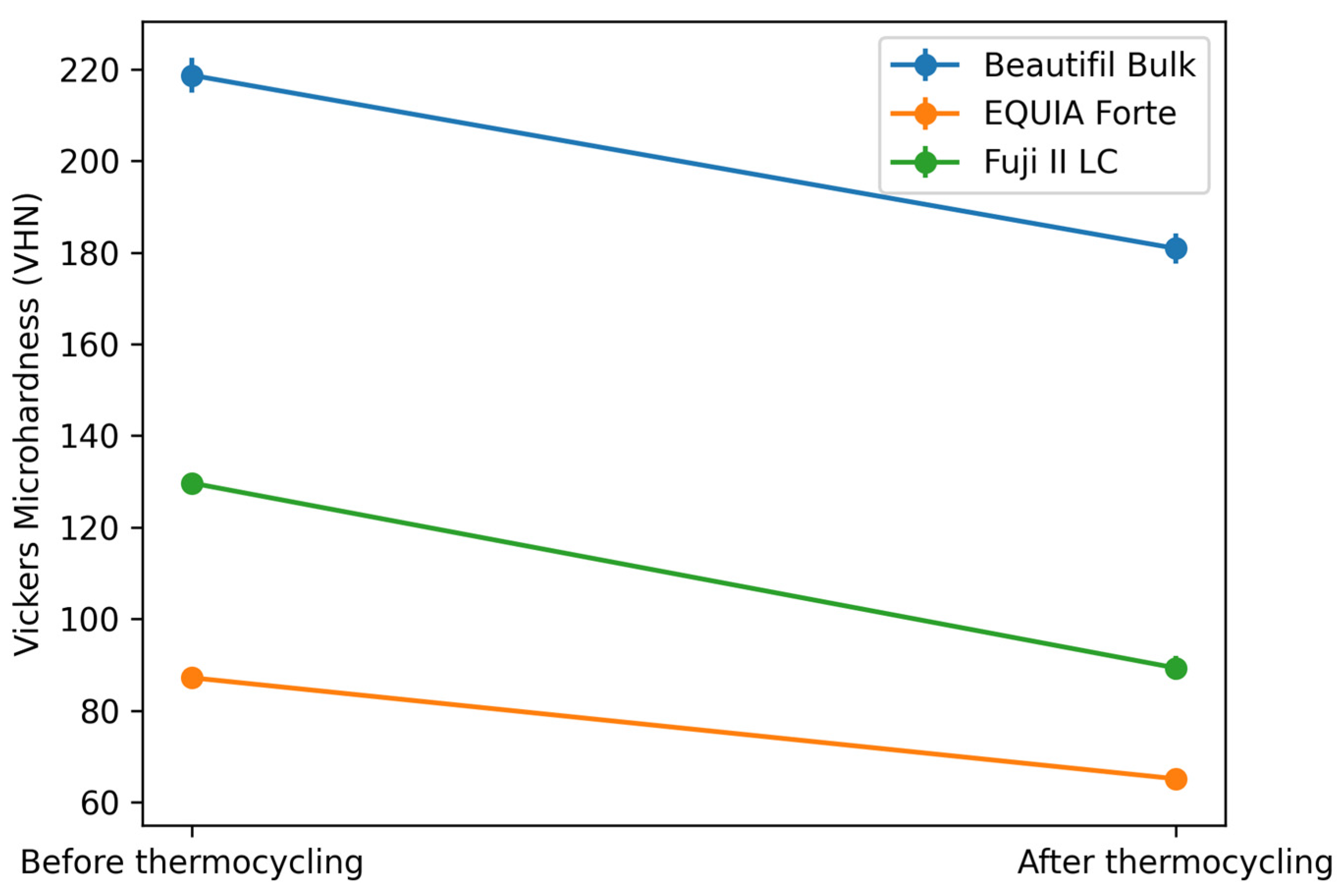
| Material | Condition | Mean ± SD | p Value |
|---|---|---|---|
| Beautifil Bulk Restorative Group 1 | Before thermal cycling Group 1a | 218.69 ± 3.77 | p< 0.001 |
| After thermal cycling Group 1b | 180.86 ± 3.37 | ||
| EQUIA Forte HT Group 2 | Before thermal cycling Group 2a | 87.14 ± 2.22 | p < 0.001 |
| After thermal cycling Group 2b | 65.08 ± 2.15 | ||
| Fuji II LC Group 3 | Before thermal cycling Group 3a | 129.66 ± 1.68 | p< 0.001 |
| After thermal cycling Group 3b | 89.27 ± 2.65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Bardakci, E.; Ozdemir Ozenen, D.; Yavuz, I. The Effect of Thermocycling on the Microhardness of Contemporary Glass Ionomer-Based Restorative Materials: An In Vitro Study. Bioengineering 2026, 13, 161. https://doi.org/10.3390/bioengineering13020161
Bardakci E, Ozdemir Ozenen D, Yavuz I. The Effect of Thermocycling on the Microhardness of Contemporary Glass Ionomer-Based Restorative Materials: An In Vitro Study. Bioengineering. 2026; 13(2):161. https://doi.org/10.3390/bioengineering13020161
Chicago/Turabian StyleBardakci, Enes, Didem Ozdemir Ozenen, and Izzet Yavuz. 2026. "The Effect of Thermocycling on the Microhardness of Contemporary Glass Ionomer-Based Restorative Materials: An In Vitro Study" Bioengineering 13, no. 2: 161. https://doi.org/10.3390/bioengineering13020161
APA StyleBardakci, E., Ozdemir Ozenen, D., & Yavuz, I. (2026). The Effect of Thermocycling on the Microhardness of Contemporary Glass Ionomer-Based Restorative Materials: An In Vitro Study. Bioengineering, 13(2), 161. https://doi.org/10.3390/bioengineering13020161







