An Effective Microcurrent Stimulation Method for Inducing Non-Pharmacological Parasympathetic Nervous System Activity for Pain Relief
Abstract
1. Introduction
2. Method
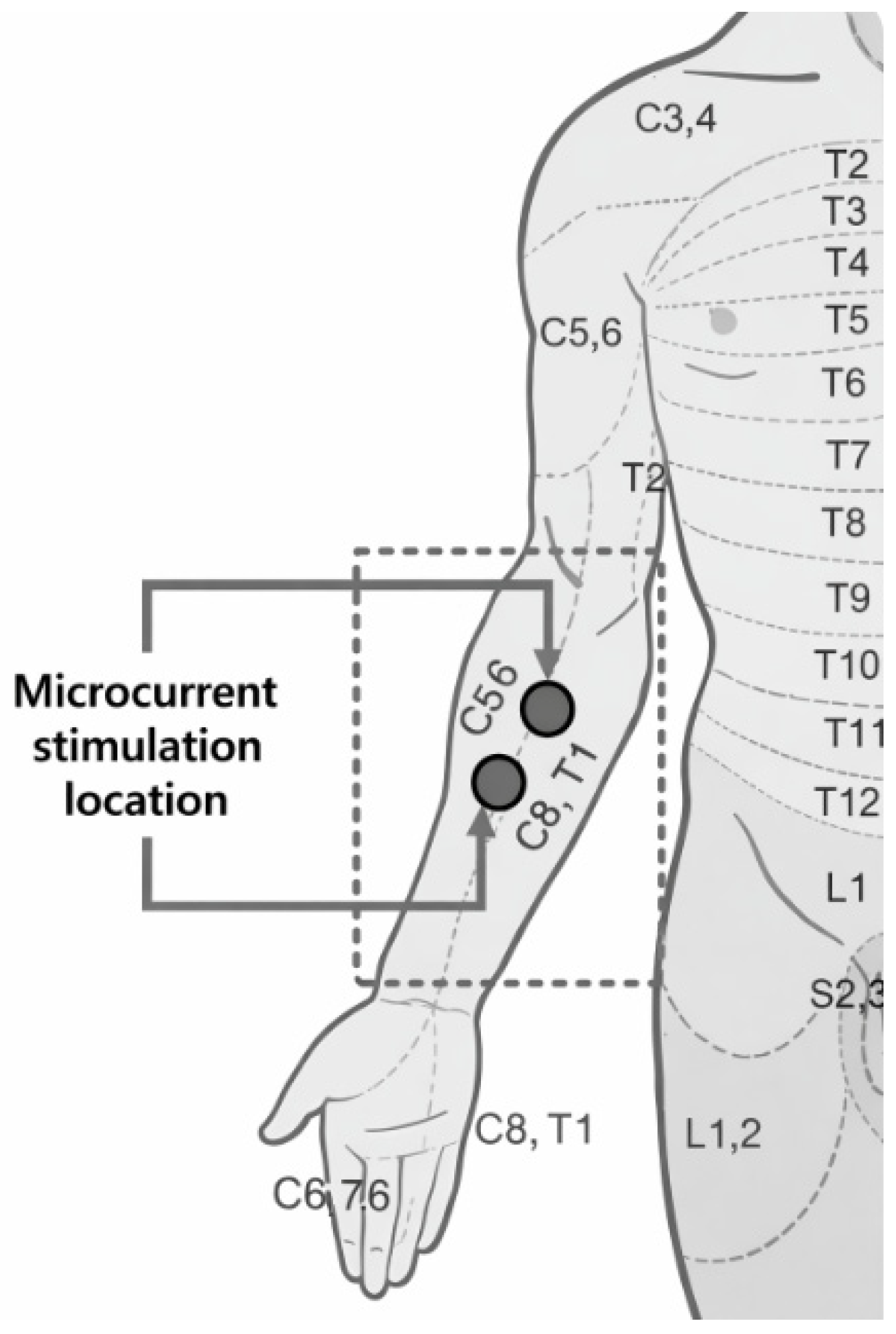
2.1. Selecting the Location of Stimulatio
2.2. Pulse Train Characteristics and Stimulation Procedure
2.3. Data Parameter Analysis Method
2.4. Data Statistical Analysis
3. Results
3.1. Nervous System Response to Microcurrent Stimulation
3.2. Stimulation Methods for Parasympathetic Nervous System Activity
3.3. Parameter Changes According to Frequency Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loeser, J.D.; Melzack, R. Pain: An overview. Lancet 1999, 353, 1607–1609. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. What is this thing called pain? J. Clin. Investig. 2010, 120, 3742–3744. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef] [PubMed]
- Saavedra-Hernández, M.M.; Castro-Sánchez, A.M.; Cuesta-Vargas, A.I.; Cleland, J.A.; Fernández-De-Las-Peñas, C.; Arroyo-Morales, M. The contribution of previous episodes of pain, pain intensity, physical impairment, and pain-related fear to disability in patients with chronic mechanical neck pain. Am. J. Phys. Med. Rehabil. 2012, 91, 1070–1076. [Google Scholar] [CrossRef]
- Fitzcharles, M.-A.; Cohen, S.P.; Clauw, D.J.; Littlejohn, G.; Usui, C.; Häuser, W. Nociplastic pain: Towards an understanding of prevalent pain conditions. Lancet 2021, 397, 2098–2110. [Google Scholar] [CrossRef]
- Fayaz, A.; Ayis, S.; Panesar, S.S.; Langford, R.M.; Donaldson, L.J. Assessing the relationship between chronic pain and cardiovasculardisease: A systematic review and meta-analysis. Scand. J. Pain 2016, 13, 76–90. [Google Scholar] [CrossRef]
- Schlereth, T.; Birklein, F. The sympathetic nervous system and pain. Neuromol. Med. 2008, 10, 141–147. [Google Scholar] [CrossRef]
- Terrier, L.M.; Hadjikhani, N.; Destrieux, C. The trigeminal pathways. J. Neurol. 2022, 269, 3443–3460. [Google Scholar] [CrossRef]
- Beckers, A.B.; van Oudenhove, L.; Weerts, Z.Z.R.; Jacobs, H.I.; Priovoulos, N.; Poser, B.A.; Ivanov, D.; Gholamrezaei, A.; Aziz, Q.; Elsenbruch, S.; et al. Evidence for engagement of the nucleus of the solitary tract in processing intestinal chemonociceptive input irrespective of conscious pain response in healthy humans. Pain 2022, 163, 1520–1529. [Google Scholar] [CrossRef]
- Benarroch, E.E. Pain-autonomic interactions: A selective review. Clin. Auton. Res. 2001, 11, 343–349. [Google Scholar] [CrossRef]
- McCarberg, B.; Peppin, J. Pain pathways and nervous system plasticity: Learning and memory in pain. Pain Med. 2019, 20, 2421–2437. [Google Scholar] [CrossRef]
- Yuan, H.; Silberstein, S.D. Vagus nerve and vagus nerve stimulation, a comprehensive review: Part I. Headache J. Head Face Pain 2016, 56, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Shore, S.E.; Vass, Z.; Wys, N.L.; Altschuler, R.A. Trigeminal ganglion innervates the auditory brainstem. J. Comp. Neurol. 2000, 419, 271–285. [Google Scholar] [CrossRef]
- Shore, S.E. Multisensory integration in the dorsal cochlear nucleus: Unit responses to acoustic and trigeminal ganglion stimulation. Eur. J. Neurosci. 2005, 21, 3334–3348. [Google Scholar] [CrossRef] [PubMed]
- Rönnegård, A.-S.; Nowak, C.; Äng, B.; Ärnlöv, J. The association between short-term, chronic localized and chronic wide-spread pain and risk for cardiovascular disease in the UK Biobank. Eur. J. Prev. Cardiol. 2022, 29, 1994–2002. [Google Scholar] [CrossRef]
- Tesarz, J.; Eich, W.; Baumeister, D.; Kohlmann, T.; D’AGostino, R.; Schuster, A.K. Widespread pain is a risk factor for cardiovascular mortality: Results from the Fram-ingham Heart Study. Eur. Heart J. 2019, 40, 1609–1617. [Google Scholar] [CrossRef]
- Macfarlane, G.J.; Barnish, M.S.; Jones, G.T. Persons with chronic widespread pain experience excess mortality: Longitudinal results from UK Biobank and meta-analysis. Ann. Rheum. Dis. 2017, 76, 1815–1822. [Google Scholar] [CrossRef]
- Nekovarova, T.; Yamamotova, A.; Vales, K.; Stuchlik, A.; Fricova, J.; Rokyta, R. Common mechanisms of pain and depression: Are antidepressants also an-algesics? Front. Behav. Neurosci. 2014, 8, 99. [Google Scholar] [CrossRef]
- Berrocoso, E.; Sanchez-Blazquez, P.; Garzon, J.; Mico, J.A. Opiates as antidepressants. Curr. Pharm. Des. 2009, 15, 1612–1622. [Google Scholar] [CrossRef]
- Watson, P.C.N. The treatment of neuropathic pain: Antidepressants and opioids. Clin. J. Pain 2000, 16, S49–S55. [Google Scholar] [CrossRef]
- Chau, D.L.; Walker, V.; Pai, L.; Cho, L.M. Opiates and elderly: Use and side effects. Clin. Interv. Aging 2008, 3, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Riediger, C.; Schuster, T.; Barlinn, K.; Maier, S.; Weitz, J.; Siepmann, T. Adverse effects of antidepressants for chronic pain: A systematic review and me-ta-analysis. Front. Neurol. 2017, 8, 307. [Google Scholar] [CrossRef] [PubMed]
- Alorfi, N.M. Pharmacological methods of pain management: Narrative review of medication used. Int. J. Gen. Med. 2023, 16, 3247–3256. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.M.; Ioannou, L.; Baker, K.S.; Gibson, S.J.; Georgiou-Karistianis, N.; Giummarra, M.J. Meta-analytic evidence for decreased heart rate variability in chronic pain impli-cating parasympathetic nervous system dysregulation. Pain 2016, 157, 7–29. [Google Scholar] [CrossRef]
- Ye, J.-J.; Lee, K.-T.; Lin, J.-S.; Chuang, C.-C. Observing continuous change in heart rate variability and photoplethysmogra-phy-derived parameters during the process of pain production/relief with thermal stimuli. J. Pain Res. 2017, 10, 527–533. [Google Scholar] [CrossRef]
- Yung, M.C.; Chang, Y.; Lai, S.T.; Tsou, M.Y.; Chan, K.H. Improved postoperative pain relief via preemptive analgesia in relation to heart rate variability for coronary artery bypass grafting: A preliminary report. Chin. Med. J. 1997, 60, 28–35. [Google Scholar]
- Yu, K.; Niu, X.; He, B. Neuromodulation management of chronic neuropathic pain in the central nervous system. Adv. Funct. Mater. 2020, 30, 1908999. [Google Scholar] [CrossRef]
- Knotkova, H.; Hamani, C.; Sivanesan, E.; Le Beuffe, M.F.E.; Moon, J.Y.; Cohen, S.P.; Huntoon, M.A. Neuromodulation for chronic pain. Lancet 2021, 397, 2111–2124. [Google Scholar] [CrossRef]
- Selva-Sarzo, F.; Romero, E.A.S.; Cuenca-Zaldívar, J.N.; García-Haba, B.; Akiyama, C.; Sillevis, R.; Fernández-Carnero, S. Effects on perceived pain and somatosensory function after transcutaneous neuromodulation in patients with chronic low back pain: A quasi-experimental study with a crossover intervention. Front. Pain Res. 2025, 6, 1525964. [Google Scholar] [CrossRef]
- Park, S.W.; Choi, J.W.; Jeong, M.G.; Lee, K.I.; Kim, H.S. Comparison and Evaluation of Autonomic Nervous System (ANS) Activity According to the Stimulus Position of Transcutaneous Electrical Nerve Stimulation (TENS). J. Biomed. Eng. Res. 2021, 42, 107–115. [Google Scholar]
- Sztajzel, J. Heart rate variability: A noninvasive electrocardiographic method to measure the autonomic nervous system. Swiss Med. Wkly. 2004, 134, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Abildskov, J.A.; Millar, K.; Burgess, M.J.; Vincent, W. The electrocardiogram and the central nervous system. Prog. Cardiovasc. Dis. 1970, 13, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Candemir, M.; Candemir, B.; Ertürk, A. Evaluation of cardiac autonomic nervous system in patients with ankylosing spondylitis using 12-lead electrocardiography and Holter monitoring. Clin. Rheumatol. 2020, 39, 2631–2639. [Google Scholar] [CrossRef] [PubMed]
- Belviso, I.; Palermi, S.; Sacco, A.M.; Romano, V.; Corrado, B.; Zappia, M.; Sirico, F. Brachial plexus injuries in sport medicine: Clinical evaluation, diagnostic ap-proaches, treatment options, and rehabilitative interventions. J. Funct. Morphol. Kinesiol. 2020, 5, 22. [Google Scholar] [CrossRef]
- Kawashima, T. The autonomic nervous system of the human heart with special reference to its origin, course, and peripheral distribution. Anat. Embryol. 2005, 209, 425–438. [Google Scholar] [CrossRef]
- Kawashima, T.; Sasaki, H. The morphological significance of the human sinuatrial nodal branch (artery). Heart Vessel. 2003, 18, 213–219. [Google Scholar] [CrossRef]
- Kandel, E.R.; Schwartz, J.H.; Jessell, T.M. (Eds.) Principles of Neural Science; McGraw-Hill: New York, NY, USA, 2000; Volume 4. [Google Scholar]
- Benarroch, E.E. The Central Autonomic Network: Functional Organization, Dysfunction, and Perspective; Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 1993; Volume 68. [Google Scholar]
- Ferrario, A.; Merrison-Hort, R.; Soffe, S.R.; Li, W.-C.; Borisyuk, R. Bifurcations of limit cycles in a reduced model of the Xenopus tadpole central pattern generator. J. Math. Neurosci. 2018, 8, 10. [Google Scholar] [CrossRef]
- Bucher, D.; Haspel, G.; Golowasch, J.; Nadim, F. Central pattern generators. eLS 2015, 11, 1–12. [Google Scholar]
- Stein, P.; Kleiger, R. Insights from the study of heart rate variability. Annu. Rev. Med. 1999, 50, 249–261. [Google Scholar] [CrossRef]
- Pham, T.; Lau, Z.J.; Chen, S.H.A.; Makowski, D. Heart rate variability in psychology: A review of HRV indices and an analysis tutorial. Sensors 2021, 21, 3998. [Google Scholar] [CrossRef]
- Khan, A.A.; Lip, G.Y.H.; Shantsila, A. Heart rate variability in atrial fibrillation: The balance between sympathetic and parasympathetic nervous system. Eur. J. Clin. Investig. 2019, 49, e13174. [Google Scholar] [CrossRef] [PubMed]
- Mirescu, S.-C.; Harden, S. Nonlinear dynamics methods for assessing heart rate varia-bility in patients with recent myocardial infarction. Rom. J. Biophys. 2012, 22, 117–124. [Google Scholar]
- Udhayakumar, R.K.; Karmakar, C.; Palaniswami, M. Understanding irreg-ularity characteristics of short-term HRV signals using sample entropy profile. IEEE Trans. Biomed. Eng. 2018, 65, 2569–2579. [Google Scholar] [CrossRef] [PubMed]
- Al-Angari, H.M.; Sahakian, A.V. Use of sample entropy approach to study heart rate variability in obstructive sleep apnea syndrome. IEEE Trans. Biomed. Eng. 2007, 54, 1900–1904. [Google Scholar] [CrossRef]
- Bakhchina, A.V.; Arutyunova, K.R.; Sozinov, A.A.; Demidovsky, A.V.; Alexandrov, Y.I. Sample entropy of the heart rate reflects properties of the system organi-zation of behaviour. Entropy 2018, 20, 449. [Google Scholar] [CrossRef]
- Shieh, J.-S.; Han, Y.-Y.; Wang, Y.-J.; Tseng, S.-C. Detrended fluctuation analyses of short-term heart rate variability in surgical in-tensive care units. Biomed. Eng. Appl. Basis Commun. 2006, 18, 67–72. [Google Scholar]
- Ashkenazy, Y.; Lewkowicz, M.; Levitan, J.; Havlin, S.; Saermark, K.; Moelgaard, H.; Thomsen, P.E.B. Discrimination between healthy and sick cardiac autonomic nervous system by detrended heart rate variability analysis. Fractals 1999, 7, 85–91. [Google Scholar] [CrossRef]
- Mizobuchi, A.; Osawa, K.; Tanaka, M.; Yumoto, A.; Saito, H.; Fuke, S. Detrended fluctuation analysis can detect the impairment of heart rate regulation in patients with heart failure with preserved ejection fraction. J. Cardiol. 2021, 77, 72–78. [Google Scholar] [CrossRef]
- Arsenault, M.; Ladouceur, A.; Lehmann, A.; Rainville, P.; Piché, M. Pain modulation induced by respiration: Phase and frequency effects. Neuroscience 2013, 252, 501–511. [Google Scholar] [CrossRef]
- Aslaksen, P.M.; Myrbakk, I.N.; Høifødt, R.S.; Flaten, M.A. The effect of experimenter gender on autonomic and subjective responses to pain stimuli. Pain 2007, 129, 260–268. [Google Scholar] [CrossRef]
- Forte, G.; Troisi, G.; Pazzaglia, M.; De Pascalis, V.; Casagrande, M. Heart rate variability and pain: A systematic review. Brain Sci. 2022, 12, 153. [Google Scholar] [CrossRef] [PubMed]
- Kolimechkov, S.; Seijo, M.; Swaine, I.; Thirkell, J.; Colado, J.C.; Naclerio, F. Physiological effects of microcurrent and its application for maximising acute responses and chronic adaptations to exercise. Eur. J. Appl. Physiol. 2023, 123, 451–465. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.V.; Heffner, K.; Gevirtz, R.; Zhang, Z.; Tadin, D.; Porsteinsson, A. Targeting autonomic flexibility to enhance cognitive training outcomes in older adults with mild cognitive impairment: Study protocol for a randomized controlled trial. Trials 2021, 22, 560. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-H. Frequency and intensity of electrical stimulation of human sympathetic ganglia affect heart rate variability and pain threshold. Appl. Sci. 2019, 9, 4490. [Google Scholar] [CrossRef]
- Messina, G.; Monda, A.; Messina, A.; Di Maio, G.; Monda, V.; Limone, P.; Dipace, A.; Monda, M.; Polito, R.; Moscatelli, F. Relationship between Non-Invasive Brain Stimulation and Autonomic Nervous System. Biomedicines 2024, 12, 972. [Google Scholar] [CrossRef]
- Korai, S.A.; Ranieri, F.; Di Lazzaro, V.; Papa, M.; Cirillo, G. Neurobiological after-effects of low intensity transcranial electric stimulation of the human nervous system: From basic mechanisms to metaplasticity. Front. Neurol. 2021, 12, 587771. [Google Scholar] [CrossRef]
- Yokota, H.; Edama, M.; Hirabayashi, R.; Sekine, C.; Otsuru, N.; Saito, K.; Kojima, S.; Miyaguchi, S.; Onishi, H. Effects of Stimulus Frequency, Intensity, and Sex on the Autonomic Response to Transcutaneous Vagus Nerve Stimulation. Brain Sci. 2022, 12, 1038. [Google Scholar] [CrossRef]
- Schmaußer, M.; Hoffmann, S.; Raab, M.; Laborde, S. The effects of noninvasive brain stimulation on heart rate and heart rate variability: A systematic review and meta-analysis. J. Neurosci. Res. 2022, 100, 1664–1694. [Google Scholar] [CrossRef]
- Beda, A.; Simpson, D.M.; Carvalho, N.C.; Carvalho, A.R.S. Low-frequency heart rate variability is related to the breath-to-breath variability in the respiratory pattern. Psychophysiology 2014, 51, 197–205. [Google Scholar] [CrossRef]
- Dayan, E.; Censor, N.; Buch, E.R.; Sandrini, M.; Cohen, L.G. Noninvasive brain stimulation: From physiology to network dynamics and back. Nat. Neurosci. 2013, 16, 838–844. [Google Scholar] [CrossRef]
- Cheing, G.; Chan, W. Influence of choice of electrical stimulation site on peripheral neurophysiological and hypoalgesic effects. J. Rehabil. Med. 2009, 41, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Nahman-Averbuch, H.; Sprecher, E.; Jacob, G.; Yarnitsky, D. The relationships between parasympathetic function and pain perception: The role of anxiety. Pain Pract. 2016, 16, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- De Couck, M.; Nijs, J.; Gidron, Y. You may need a nerve to treat pain: The neurobiological rationale for vagal nerve activation in pain management. Clin. J. Pain 2014, 30, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Hudlicka, O. Cardiovascular System: Changes with Exercise Training and Muscle Stimulation. In Application of Muscle/Nerve Stimulation in Health and Disease; Springer: Dordrecht, The Netherlands, 2008; pp. 23–53. [Google Scholar]
- Freyschuss, U.L.L.A. Elicitation of heart rate and blood pressure increase on muscle contraction. J. Appl. Physiol. 1970, 28, 758–761. [Google Scholar] [CrossRef]
- Al-Ani, M.; Munir, S.M.; White, M.; Townend, J.; Coote, J.H. Changes in RR variability before and after endurance training measured by power spectral analysis and by the effect of isometric muscle contraction. Eur. J. Appl. Physiol. Occup. Physiol. 1996, 74, 397–403. [Google Scholar]
- Katayama, K.; Saito, M. Muscle sympathetic nerve activity during exercise. J. Physiol. Sci. 2019, 69, 589–598. [Google Scholar] [CrossRef]
- Cui, J.; Blaha, C.; Moradkhan, R.; Gray, K.S.; Sinoway, L.I. Muscle sympathetic nerve activity responses to dynamic passive muscle stretch in humans. J. Physiol. 2006, 576, 625–634. [Google Scholar] [CrossRef]
- Saito, M.; Naito, M.; Mano, T. Different responses in skin and muscle sympathetic nerve activity to static muscle contraction. J. Appl. Physiol. 1990, 69, 2085–2090. [Google Scholar] [CrossRef]
- Schroeder, E.B.; Liao, D.; Chambless, L.E.; Prineas, R.J.; Evans, G.W.; Heiss, G. Hypertension, blood pressure, and heart rate variability: The Atherosclerosis Risk in Communities (ARIC) study. Hypertension 2003, 42, 1106–1111. [Google Scholar] [CrossRef]
- Xie, G.-L.; Wang, J.-H.; Zhou, Y.; Xu, H.; Sun, J.-H.; Yang, S.-R. Association of high blood pressure with heart rate variability in children. Iran. J. Pediatr. 2013, 23, 37. [Google Scholar]
- Nicoll, R.; Henein, M.Y. Caloric restriction and its effect on blood pressure, heart rate var-iability and arterial stiffness and dilatation: A review of the evidence. Int. J. Mol. Sci. 2018, 19, 751. [Google Scholar] [CrossRef] [PubMed]
- Bowsher, D. Trigeminal neuralgia: An anatomically oriented review. Clin. Anat. 1997, 10, 409–415. [Google Scholar] [CrossRef]
- Moeller, M.; Schroeder, C.F.; May, A. Vagus nerve stimulation modulates the cranial trigeminal autonomic reflex. Ann. Neurol. 2018, 84, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Agorastos, A.; Mansueto, A.C.; Hager, T.; Pappi, E.; Gardikioti, A.; Stiedl, O. Heart rate variability as a Translational dynamic biomarker of altered autonomic function in Health and Psychiatric disease. Biomedicines 2023, 11, 1591. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Mei, C.; Chen, P.; Chen, X. The contribution of neuro-immune crosstalk to pain in the peripheral nervous system and the spinal cord. Int. Immunopharmacol. 2022, 107, 108700. [Google Scholar] [CrossRef]
- Botha, C.; Farmer, A.D.; Nilsson, M.; Brock, C.; Gavrila, A.D.; Drewes, A.M.; Knowles, C.H.; Aziz, Q. Preliminary report: Modulation of parasympathetic nervous system tone influences oesophageal pain hypersensitivity. Gut 2015, 64, 611–617. [Google Scholar] [CrossRef]
- Hautala, A.J.; Karppinen, J.; Seppanen, T. Short-term assessment of autonomic nervous system as a potential tool to quantify pain experience. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; IEEE: New York, NY, USA, 2016. [Google Scholar]
- Luo, F.; Wang, Q.; Kashani, A.; Yan, J. Corticofugal modulation of initial sound processing in the brain. J. Neurosci. 2008, 28, 11615–11621. [Google Scholar] [CrossRef]
- Soeta, Y.; Nakagawa, S. Auditory evoked responses in human auditory cortex to the variation of sound intensity in an ongoing tone. Hear. Res. 2012, 287, 67–75. [Google Scholar] [CrossRef]
- Kim, D.; Kim, N.; Lee, Y.; Kim, S.; Kwon, J. Sound stimulation using the individual’s heart rate to improve the stability and ho-meostasis of the autonomic nervous system. Physiol. Rep. 2023, 11, e15816. [Google Scholar] [CrossRef]
- Kim, D.; Lee, Y.; Jeong, J.; Kim, S. Stimulation method and individual health index study for real-time cardiovascular and autonomic nervous system reactivity analysis using PPG signal. Biomed. Signal Process. Control. 2022, 76, 103714. [Google Scholar] [CrossRef]


| Participants (Count) | 24 | |||
|---|---|---|---|---|
| Male | 14 | Female | 10 | |
| Height (Cm mean ± std) | 176.08 ± 7.05 | 160.6 ± 5.88 | ||
| Weight (Kg mean ± std) | 77.07 ± 10.32 | 52.9 ± 9.03 | ||
| Age (Years mean ± std) | 26.35 ± 1.71 | 24 ± 1.61 | ||
| Parameter | Formula | Analyze |
|---|---|---|
| RR-interval | Heart rate variability | |
| The standard deviation of normal to normal R-R intervals (SDNN) | Sympathetic/parasympathetic nerves | |
| The square root of the mean squared differences of successive R-R intervals (RMSSD) | Parasympathetic nerves | |
| Instantaneous RR interval variability (SD1) | Short term volatility | |
| Continuous RR interval variability (SD2) | Long term volatility | |
| Entropy | Randomness | |
| Detrended fluctuation analysis (DFA) | Long-term correlation | |
| Low pass band power spectral density (LF norm) | Sympathetic/parasympathetic nerves | |
| High pass band power spectral density (HF norm) | Parasympathetic nerves | |
| LF/HF | Autonomic nervous system balance |
| Hz | Vpp | Before Stimulus | Stimulus 0–2 min Interval | Stimulus 4–6 min Interval | Stimulus 8–10 min Interval | End of Stimulus |
|---|---|---|---|---|---|---|
| 1 | 3 | 1.23 ± 0.24 | 1.12 ± 0.19 * | 1.11 ± 0.17 * | 1.12 ± 0.19 * | 1.23 ± 0.29 |
| 7 | 1.37 ± 0.23 | 1.14 ± 0.16 * | 1.14 ± 0.17 * | 1.16 ± 0.14 * | 1.31 ± 0.33 | |
| 11 | 1.32 ± 0.28 | 1.11 ± 0.17 * | 1.15 ± 0.16 * | 1.14 ± 0.19 * | 1.25 ± 0.20 | |
| 40 | 3 | 1.26 ± 0.19 | 1.07 ± 0.16 * | 1.09 ± 0.16 * | 1.09 ± 0.15 * | 1.20 ± 0.20 |
| 7 | 1.22 ± 0.20 | 1.09 ± 0.16 * | 1.10 ± 0.17 * | 1.09 ± 0.17 * | 1.24 ± 0.20 | |
| 11 | 1.28 ± 0.25 | 1.06 ± 0.16 * | 1.10 ± 0.14 * | 1.10 ± 0.16 * | 1.21 ± 0.22 | |
| 5–40 | 3 | 1.19 ± 0.20 | 1.06 ± 0.13 * | 1.06 ± 0.16 * | 1.07 ± 0.18 * | 1.16 ± 0.22 |
| 7 | 1.22 ± 0.27 | 1.04 ± 0.16 * | 1.03 ± 0.16 * | 1.09 ± 0.18 * | 1.15 ± 0.21 | |
| 11 | 1.25 ± 0.23 | 1.05 ± 0.15 * | 1.08 ± 0.16 * | 1.10 ± 0.18 * | 1.15 ± 0.19 |
| 3 Vpp | 7 Vpp | 11 Vpp | ||||
|---|---|---|---|---|---|---|
| RMSSD | ||||||
| Hz | Before Stimulus | End of Stimulus | Before Stimulus | End of Stimulus | Before Stimulus | End of Stimulus |
| 1 | 74.18 ± 63.85 | 75.36 ± 49.70 | 41.31 ± 34.13 | 65.09 ± 32.46 * | 47.04 ± 50.32 | 58.45 ± 23.78 |
| 40 | 47.00 ± 38.28 | 41.00 ± 33.41 | 38.25 ± 17.98 | 41.50 ± 41.05 | 37.33 ± 22.71 | 45.25 ± 41.32 |
| 5–40 | 47.79 ± 37.13 | 36.95 ± 24.77 | 44.45 ± 31.05 | 43.66 ± 23.50 | 47.91 ± 31.24 | 47.70 ± 31.06 |
| SD1 | ||||||
| 1 | 52.49 ± 45.08 | 53.28 ± 35.13 | 29.23 ± 24.14 | 44.56 ± 37.92 * | 33.15 ± 35.54 | 38.05 ± 27.86 |
| 40 | 33.22 ± 27.04 | 28.96 ± 23.56 | 27.06 ± 12.77 | 29.34 ± 28.98 | 26.41 ± 16.00 | 32.04 ± 29.27 |
| 5–40 | 33.83 ± 26.28 | 26.11 ± 17.45 | 31.40 ± 21.95 | 30.79 ± 16.58 | 33.85 ± 22.04 | 33.72 ± 21.95 |
| Type | Parameters | Before Stimulus | Stimulus 0–2 min Interval | Stimulus 4–6 min Interval | Stimulus 8–10 min Interval | End of Stimulus |
|---|---|---|---|---|---|---|
| 1 Hz | RR-intervals (s) | 0.75 ± 0.08 | 0.74 ± 0.07 | 0.74 ± 0.08 | 0.74 ± 0.08 | 0.75 ± 0.07 |
| SDNN (ms) | 54.68 ± 22.44 | 61.04 ± 19.16 * | 63.5 ± 28.35 | 60.40 ± 23.99 | 65.09 ± 32.46 | |
| RMSSD (ms) | 41.31 ± 34.13 | 47.68 ± 33.08 | 50.45 ± 43.38 | 60.40 ± 23.99 | 65.09 ± 32.46 * | |
| SD1 (ms) | 29.23 ± 24.14 | 33.62 ± 23.43 | 35.69 ± 30.62 | 34.02 ± 27.46 | 44.56 ± 37.92 * | |
| SD2 (ms) | 70.40 ± 24.06 | 77.77 ± 20.73 | 80.72 ± 31.00 * | 76.90 ± 24.76 | 78.07 ± 32.95 | |
| Entropy | 1.27 ± 0.34 | 1.21 ± 0.35 | 1.15 ± 0.31 * | 0.20 ± 0.31 | 1.26 ± 0.44 | |
| DFA | 1.37 ± 0.23 | 1.14 ± 0.16 * | 1.14 ± 0.17 * | 1.16 ± 0.14 * | 1.31 ± 0.33 | |
| LF norm | 0.38 ± 0.13 | 0.36 ± 0.15 | 0.36 ± 0.18 | 0.37 ± 0.13 | 0.36 ± 0.14 | |
| HF norm | 0.65 ± 0.56 | 0.73 ± 0.74 | 0.95 ± 1.15 | 0.63 ± 0.58 | 1.06 ± 1.02 | |
| LF/HF | 3.48 ± 3.77 | 5.50 ± 7.84 | 5.99 ± 8.11 | 4.36 ± 5.22 | 2.99 ± 3.30 | |
| 40 Hz | RR-intervals (s) | 0.76 ± 0.10 | 0.76 ± 0.10 | 0.76 ± 0.09 | 0.76 ± 0.10 | 0.75 ± 0.08 |
| SDNN (ms) | 49.41 ± 0.15 | 53.62 ± 14.31 | 53.54 ± 15.06 | 52.62 ± 16.60 | 57.87 ± 26.91 | |
| RMSSD (ms) | 38.25 ± 17.98 | 37.37 ± 13.88 | 37.93 ± 18.79 | 36.41 ± 17.68 | 41.50 ± 41.05 | |
| SD1 (ms) | 27.06 ± 12.77 | 26.41 ± 9.81 | 26.82 ± 13.28 | 25.81 ± 12.04 | 29.34 ± 28.98 | |
| SD2 (ms) | 64.10 ± 19.84 | 70.78 ± 18.98 | 70.04 ± 19.41 | 59.47 ± 21.19 | 75.01 ± 28.35 | |
| Entropy | 1.50 ± 0.43 | 1.36 ± 0.28 | 1.35 ± 0.28 | 1.39 ± 0.31 | 1.38 ± 0.33 | |
| DFA | 1.22 ± 0.20 | 1.09 ± 0.16 * | 1.10 ± 0.17 * | 1.09 ± 0.17 * | 1.24 ± 0.20 | |
| LF norm | 0.40 ± 0.14 | 0.36 ± 0.16 | 0.40 ± 0.17 | 0.39 ± 0.17 | 0.37 ± 0.17 | |
| HF norm | 0.69 ± 0.53 | 0.68 ± 0.52 | 0.70 ± 0.76 | 0.68 ± 0.69 | 0.62 ± 0.66 | |
| LF/HF | 2.84 ± 2.68 | 3.76 ± 4.54 | 4.58 ± 4.71 | 4.29 ± 4.53 | 4.24 ± 4.19 | |
| 5–40 Hz | RR-intervals (s) | 0.79 ± 0.11 | 0.80 ± 0.10 | 0.79 ± 0.10 | 0.79 ± 0.09 | 0.80 ± 0.10 |
| SDNN (ms) | 53.62 ± 19.02 | 73.12 ± 33.58 * | 61.50 ± 27.63 | 56.33 ± 19.46 | 58.50 ± 20.31 | |
| RMSSD (ms) | 44.45 ± 31.05 | 71.17 ± 55.93 * | 60.87 ± 48.27 | 46.87 ± 31.95 | 43.66 ± 23.50 | |
| SD1 (ms) | 31.40 ± 21.95 | 50.72 ± 39.58 * | 42.99 ± 34.03 | 33.15 ± 22.57 | 30.79 ± 16.58 | |
| SD2 (ms) | 67.26 ± 21.84 | 87.02 ± 34.68 * | 73.58 ± 25.99 | 70.89 ± 21.59 | 75.25 ± 28.04 | |
| Entropy | 1.37 ± 0.33 | 1.21 ± 0.34 * | 1.32 ± 0.36 | 1.36 ± 0.28 | 1.42 ± 0.29 | |
| DFA | 1.22 ± 0.27 | 1.04 ± 0.16 * | 1.03 ± 0.16 * | 1.09 ± 0.18 * | 1.15 ± 0.21 | |
| LF norm | 0.37 ± 0.17 | 0.22 ± 0.07 | 0.28 ± 0.09 | 0.28 ± 0.08 | 0.30 ± 0.12 | |
| HF norm | 1.13 ± 1.28 | 1.56 ± 1.39 | 1.22 ± 0.99 | 1.02 ± 0.91 | 0.99 ± 0.87 | |
| LF/HF | 2.85 ± 2.46 | 1.30 ± 1.19 * | 1.61 ± 1.31 * | 2.02 ± 1.59 | 2.39 ± 2.65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, D.; Ko, J.; Kim, S. An Effective Microcurrent Stimulation Method for Inducing Non-Pharmacological Parasympathetic Nervous System Activity for Pain Relief. Bioengineering 2026, 13, 52. https://doi.org/10.3390/bioengineering13010052
Kim D, Ko J, Kim S. An Effective Microcurrent Stimulation Method for Inducing Non-Pharmacological Parasympathetic Nervous System Activity for Pain Relief. Bioengineering. 2026; 13(1):52. https://doi.org/10.3390/bioengineering13010052
Chicago/Turabian StyleKim, Daechang, Jaeeun Ko, and Sungmin Kim. 2026. "An Effective Microcurrent Stimulation Method for Inducing Non-Pharmacological Parasympathetic Nervous System Activity for Pain Relief" Bioengineering 13, no. 1: 52. https://doi.org/10.3390/bioengineering13010052
APA StyleKim, D., Ko, J., & Kim, S. (2026). An Effective Microcurrent Stimulation Method for Inducing Non-Pharmacological Parasympathetic Nervous System Activity for Pain Relief. Bioengineering, 13(1), 52. https://doi.org/10.3390/bioengineering13010052






