Emerging Smart and Adaptive Hydrogels for Next-Generation Tissue Engineering
Abstract
1. Introduction
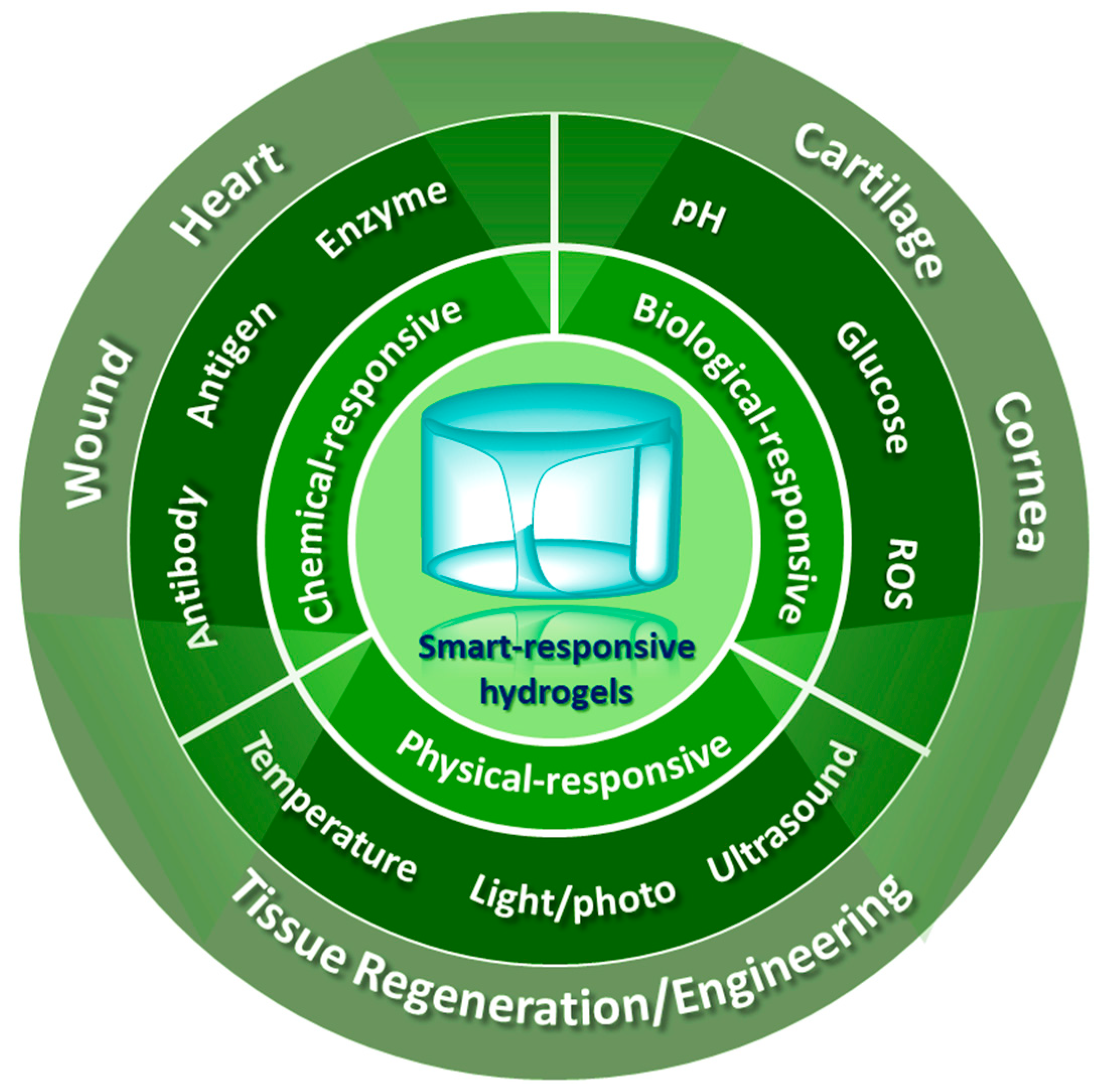
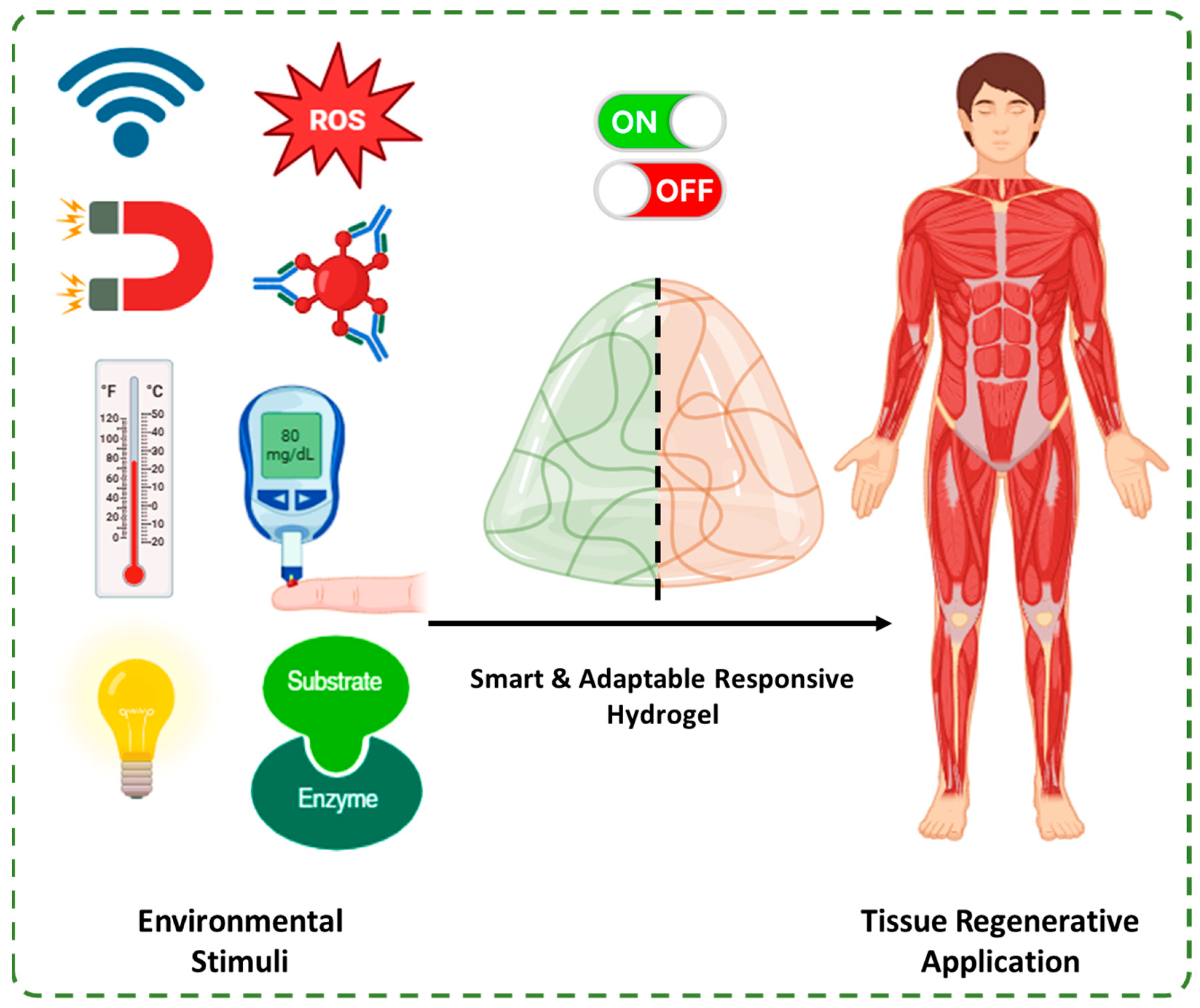
2. Physical, Chemical, and Biological Responsive Hydrogels
2.1. Physical Responsive Hydrogels
2.1.1. Ultrasound-Responsive Hydrogels
2.1.2. Magnetic-Responsive Hydrogels
2.1.3. Photo-Responsive Hydrogels
2.1.4. Thermo-Responsive Hydrogels
2.1.5. Pressure-Responsive Hydrogels
2.2. Chemical Responsive Hydrogels
2.2.1. ROS-Responsive Hydrogels
2.2.2. pH-Responsive Hydrogels
2.2.3. Glucose-Responsive Hydrogels
2.3. Biological Responsive Hydrogels
2.3.1. Enzyme-Responsive Hydrogels
2.3.2. Antigen/Antibody-Responsive Hydrogels
3. Mechanobiology-Informed Adaptive Scaffolds
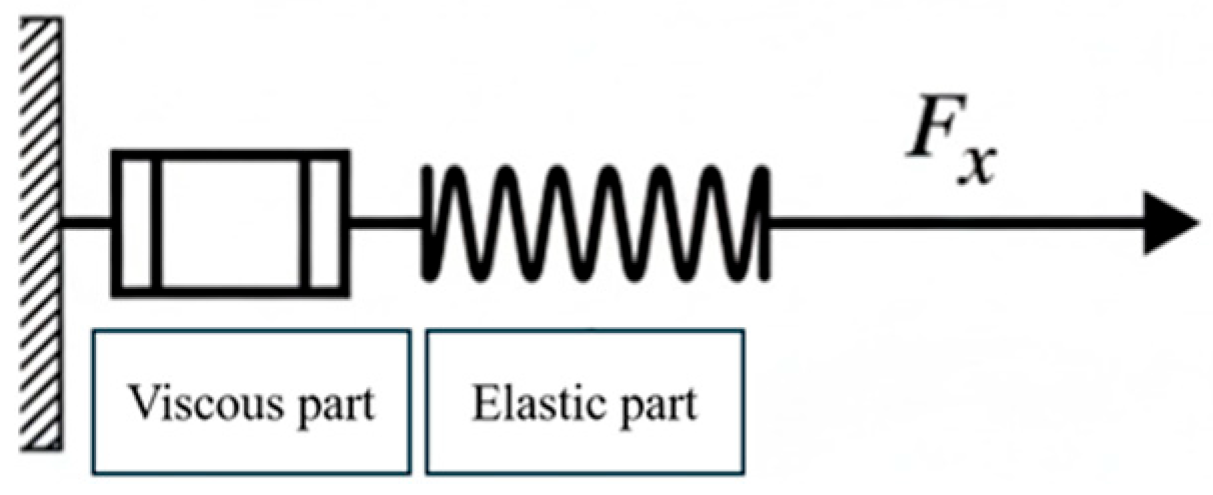
3.1. Mechanical Foundations of Mechanosensitive Materials
3.2. Mechanotransduction: Translating Force into Cellular Response
3.3. Mechanobiology-Guided Scaffold Design
4. Bioelectronic and Electroactive Hydrogels for Tissue Engineering
5. Advanced Biofabrication and Programmable Architectures
5.1. Design Constraints for 3D Printable Adaptive Biomaterials
5.2. 3D Printing Strategies for Mechanosensitive Constructs
5.3. 4D Biofabrication: Printing Time-Programmed Mechanics and Shape
5.4. In Situ Bioprinting and Minimally Invasive Deployment
5.5. Outlook: Integrating Fabrication, Mechanics, and Clinical Reality
6. Conclusions and Future Perceptions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wu, Z.; Shi, G.; Li, L.; Piao, Z.; Wang, J.; Chen, R.; Hao, Z.; Zhang, Z.; Li, Z.; Huang, Y. Recent advances in smart responsive hydrogel microspheres for tissue regeneration: Preparation, characteristics and applications. Mater. Horiz. 2025, 12, 8943–8988. [Google Scholar] [CrossRef]
- Asim, S.; Tuftee, C.; Qureshi, A.T.; Callaghan, R.; Geary, M.L.; Santra, M.; Pal, V.; Namli, I.; Yam, G.H.F.; Ozbolat, I.T. Multi-Functional Gelatin-Dithiolane Hydrogels for Tissue Engineering. Adv. Funct. Mater. 2025, 35, 2407522. [Google Scholar] [CrossRef]
- Sojdeh, S.; Panjipour, A.; Bejandi, Z.B.; Salehi, M.; Yaghmour, A.; Arabpour, Z.; Djalilian, A.R.; Chan, R.P. Hydrogel-Based Vitreous Substitutes. Int. J. Mol. Sci. 2025, 26, 8406. [Google Scholar] [CrossRef]
- Naderi, H.; Matin, M.M.; Bahrami, A.R. Critical issues in tissue engineering: Biomaterials, cell sources, angiogenesis, and drug delivery systems. J. Biomater. Appl. 2011, 26, 383–417. [Google Scholar] [CrossRef]
- Kumar, H.; Justa, P.; Jaswal, N.; Pani, B.; Kumar, P. Biomimetic and bioinspired composite processing for biomedical applications. Adv. Mater. Manuf. Tech. Biomed. Appl. 2023, 9, 211–239. [Google Scholar]
- Nazeri, N.; Tajerian, R.; Arabpour, Z.; Hadjighassem, M.R.; Gheibi, N.; Manouchehrabadi, M.; Ghanbari, H. Bioinspired immobilization of carbon nanotubes on scaffolds for nerve regeneration. Bioinspired Biomim. Nanobiomater. 2019, 8, 198–205. [Google Scholar] [CrossRef]
- Xue, H.; Zhu, C.; Wang, Y.; Gu, Q.; Shao, Y.; Jin, A.; Zhang, X.; Lei, L.; Li, Y. Stimulus-responsive cellulose hydrogels in biomedical applications and challenges. Mater. Today Bio 2025, 32, 101814. [Google Scholar] [CrossRef]
- Protsak, I.S.; Morozov, Y.M. Fundamentals and advances in stimuli-responsive hydrogels and their applications: A review. Gels 2025, 11, 30. [Google Scholar] [CrossRef] [PubMed]
- Sojdeh, S.; Bagherzadeh, M.; Daneshgar, H.; Edrisi, M.; Ahmadifar, M.; Rabiei, N. Dual-mode optical biocompatible-sensor enabled by enzyme-like activity of UiO-66 (Zr) for ultra-sensitive ROS detection in vitro. Talanta 2024, 280, 126742. [Google Scholar] [CrossRef]
- Rahman, M.M.; Giol, E.D.; Cama, G.; Van Vlierberghe, S.; Dubruel, P. Stimuli-Responsive Hydrogels for Tissue Engineering; Royal Society of Chemistry: London, UK, 2016. [Google Scholar]
- Garcia-Garcia, A.; Muñana-González, S.; Lanceros-Mendez, S.; Ruiz-Rubio, L.; Alvarez, L.P.; Vilas-Vilela, J.L. Biodegradable natural hydrogels for tissue engineering, controlled release, and soil remediation. Polymers 2024, 16, 2599. [Google Scholar] [CrossRef]
- Sojdeh, S.; Panjipour, A.; Yaghmour, A.; Arabpour, Z.; Djalilian, A.R. Click Chemistry-Based Hydrogels for Tissue Engineering. Gels 2025, 11, 724. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.U.A.; Stojanović, G.M.; Abdullah, M.F.B.; Dolatshahi-Pirouz, A.; Marei, H.E.; Ashammakhi, N.; Hasan, A. Fundamental properties of smart hydrogels for tissue engineering applications: A review. Int. J. Biol. Macromol. 2024, 254, 127882. [Google Scholar] [CrossRef]
- Solanki, R.; Bhatia, D. Stimulus-responsive hydrogels for targeted cancer therapy. Gels 2024, 10, 440. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Maruf, D.A.A.; Akin-Ige, F.; Amin, S. Stimuli-responsive hydrogels for skin wound healing and regeneration. Emergent Mater. 2025, 8, 1339–1356. [Google Scholar] [CrossRef]
- Sadraei, A.; Naghib, S.M. 4D printing of physical stimuli-responsive hydrogels for localized drug delivery and tissue engineering. Polym. Rev. 2025, 65, 104–168. [Google Scholar] [CrossRef]
- Yu, C.; Li, L.; Hu, P.; Yang, Y.; Wei, W.; Deng, X.; Wang, L.; Tay, F.R.; Ma, J. Recent advances in stimulus-responsive nanocarriers for gene therapy. Adv. Sci. 2021, 8, 2100540. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, G.; Guo, S. Advances in ultrasound-responsive hydrogels for biomedical applications. J. Mater. Chem. B 2022, 10, 3947–3958. [Google Scholar] [CrossRef]
- Cui, X.; Han, X.; Yu, L.; Zhang, B.; Chen, Y. Intrinsic chemistry and design principle of ultrasound-responsive nanomedicine. Nano Today 2019, 28, 100773. [Google Scholar] [CrossRef]
- Zhang, F.; Lv, M.; Wang, S.; Li, M.; Wang, Y.; Hu, C.; Hu, W.; Wang, X.; Wang, X.; Liu, Z. Ultrasound-triggered biomimetic ultrashort peptide nanofiber hydrogels promote bone regeneration by modulating macrophage and the osteogenic immune microenvironment. Bioact. Mater. 2024, 31, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Wang, Q.; Wan, X.; Yang, M.; Liu, C.; Xu, C.; Hu, B.; Feng, J.; Luo, Z. Wireless electrical stimulation of the vagus nerves by ultrasound-responsive programmable hydrogel nanogenerators for anti-inflammatory therapy in sepsis. Nano Energy 2021, 89, 106327. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, Y.; Meng, C.; Xie, X.; Cui, W.; Zuo, K. Tissue-Penetrating Ultrasound-Triggered Hydrogel for Promoting Microvascular Network Reconstruction. Adv. Sci. 2024, 11, 2401368. [Google Scholar] [CrossRef]
- Xiao, C.; Wang, R.; Fu, R.; Yu, P.; Guo, J.; Li, G.; Wang, Z.; Wang, H.; Nie, J.; Liu, W. Piezo-enhanced near infrared photocatalytic nanoheterojunction integrated injectable biopolymer hydrogel for anti-osteosarcoma and osteogenesis combination therapy. Bioact. Mater. 2024, 34, 381–400. [Google Scholar] [CrossRef]
- Zhou, S.; Xiao, C.; Fan, L.; Yang, J.; Ge, R.; Cai, M.; Yuan, K.; Li, C.; Crawford, R.W.; Xiao, Y. Injectable ultrasound-powered bone-adhesive nanocomposite hydrogel for electrically accelerated irregular bone defect healing. J. Nanobiotechnol. 2024, 22, 54. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, Y.; Chen, C.; Cheng, Y. Magnetic-responsive hydrogels: From strategic design to biomedical applications. J. Control. Release 2021, 335, 541–556. [Google Scholar] [CrossRef]
- Gonçalves, A.I.; Miranda, M.S.; Rodrigues, M.T.; Reis, R.L.; Gomes, M.E. Magnetic responsive cell-based strategies for diagnostics and therapeutics. Biomed. Mater. 2018, 13, 054001. [Google Scholar] [CrossRef]
- Zhu, K.; Ju, Y.; Xu, J.; Yang, Z.; Gao, S.; Hou, Y. Magnetic nanomaterials: Chemical design, synthesis, and potential applications. Acc. Chem. Res. 2018, 51, 404–413. [Google Scholar] [CrossRef]
- Ghazanfari, M.R.; Kashefi, M.; Shams, S.F.; Jaafari, M.R. Perspective of Fe3O4 nanoparticles role in biomedical applications. Biochem. Res. Int. 2016, 2016, 7840161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, Q.; Du, J. Recent advances in magnetic hydrogels. Polym. Int. 2016, 65, 1365–1372. [Google Scholar] [CrossRef]
- Ganguly, S.; Margel, S. Design of magnetic hydrogels for hyperthermia and drug delivery. Polymers 2021, 13, 4259. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Semeano, A.T.S.; Cornejo, D.R.; Ulrich, H.; Petri, D.F.S. Magnetic hydrogels for levodopa release and cell stimulation triggered by external magnetic field. Colloids Surf. B Biointerfaces 2018, 167, 415–424. [Google Scholar] [CrossRef]
- Iqbal, H.; Khan, B.A.; Khan, Z.U.; Razzaq, A.; Khan, N.U.; Menaa, B.; Menaa, F. Fabrication, physical characterizations and in vitro antibacterial activity of cefadroxil-loaded chitosan/poly (vinyl alcohol) nanofibers against Staphylococcus aureus clinical isolates. Int. J. Biol. Macromol. 2020, 144, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Lock, J.; Sallee, A.; Liu, H. Magnetic nanocomposite hydrogel for potential cartilage tissue engineering: Synthesis, characterization, and cytocompatibility with bone marrow derived mesenchymal stem cells. ACS Appl. Mater. Interfaces 2015, 7, 20987–20998. [Google Scholar] [CrossRef]
- Hu, Q.; Ding, B.; Yan, X.; Peng, L.; Duan, J.; Yang, S.; Cheng, L.; Chen, D. Polyethylene glycol modified PAMAM dendrimer delivery of kartogenin to induce chondrogenic differentiation of mesenchymal stem cells. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2189–2198. [Google Scholar] [CrossRef] [PubMed]
- Hazeri, Y.; Irani, S.; Zandi, M.; Pezeshki-Modaress, M. Polyvinyl alcohol/sulfated alginate nanofibers induced the neuronal differentiation of human bone marrow stem cells. Int. J. Biol. Macromol. 2020, 147, 946–953. [Google Scholar] [CrossRef]
- Omidinia-Anarkoli, A.; Boesveld, S.; Tuvshindorj, U.; Rose, J.C.; Haraszti, T.; De Laporte, L. An injectable hybrid hydrogel with oriented short fibers induces unidirectional growth of functional nerve cells. Small 2017, 13, 1702207. [Google Scholar] [CrossRef]
- Namdari, M.; Eatemadi, A. Cardioprotective effects of curcumin-loaded magnetic hydrogel nanocomposite (nanocurcumin) against doxorubicin-induced cardiac toxicity in rat cardiomyocyte cell lines. Artif. Cells Nanomed. Biotechnol. 2017, 45, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Scheiger, J.M.; Levkin, P.A. Design and applications of photoresponsive hydrogels. Adv. Mater. 2019, 31, 1807333. [Google Scholar] [CrossRef]
- Maleki, A.; He, J.; Bochani, S.; Nosrati, V.; Shahbazi, M.-A.; Guo, B. Multifunctional photoactive hydrogels for wound healing acceleration. ACS Nano 2021, 15, 18895–18930. [Google Scholar] [CrossRef]
- Lee, H.P.; Gaharwar, A.K. Light-responsive inorganic biomaterials for biomedical applications. Adv. Sci. 2020, 7, 2000863. [Google Scholar] [CrossRef]
- Tang, Y.; Wang, G. NIR light-responsive nanocarriers for controlled release. J. Photochem. Photobiol. C Photochem. Rev. 2021, 47, 100420. [Google Scholar] [CrossRef]
- Jochum, F.D.; Theato, P. Temperature-and light-responsive smart polymer materials. Chem. Soc. Rev. 2013, 42, 7468–7483. [Google Scholar] [CrossRef]
- Raza, A.; Hayat, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M. “Smart” materials-based near-infrared light-responsive drug delivery systems for cancer treatment: A review. J. Mater. Res. Technol. 2019, 8, 1497–1509. [Google Scholar] [CrossRef]
- Luo, R.; Xiang, X.; Jiao, Q.; Hua, H.; Chen, Y. Photoresponsive hydrogels for tissue engineering. ACS Biomater. Sci. Eng. 2024, 10, 3612–3630. [Google Scholar] [CrossRef]
- Tang, J.; Wu, C.; Qiao, Z.; Pi, J.; Zhang, Y.; Luo, F.; Sun, J.; Wei, D.; Fan, H. A photoelectric effect integrated scaffold for the wireless regulation of nerve cellular behavior. J. Mater. Chem. B 2022, 10, 1601–1611. [Google Scholar] [CrossRef]
- Jiang, Y.; Huang, Y.; Luo, X.; Wu, J.; Zong, H.; Shi, L.; Cheng, R.; Zhu, Y.; Jiang, S.; Lan, L. Neural stimulation in vitro and in vivo by photoacoustic nanotransducers. Matter 2021, 4, 654–674. [Google Scholar] [CrossRef]
- Zheng, N.; Fitzpatrick, V.; Cheng, R.; Shi, L.; Kaplan, D.L.; Yang, C. Photoacoustic carbon nanotubes embedded silk scaffolds for neural stimulation and regeneration. Acs Nano 2022, 16, 2292–2305. [Google Scholar] [CrossRef]
- Middha, E.; Liu, B. Nanoparticles of organic electronic materials for biomedical applications. ACS Nano 2020, 14, 9228–9242. [Google Scholar] [CrossRef]
- Zhou, K.; Chigan, D.; Xu, L.; Liu, C.; Ding, R.; Li, G.; Zhang, Z.; Pei, D.; Li, A.; Guo, B. Anti-sandwich structured photo-electronic wound dressing for highly efficient bacterial infection therapy. Small 2021, 17, 2101858. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Zhu, M.; Xu, G.; Liu, N.; Yu, C. A near-infrared light-responsive multifunctional nanocomposite hydrogel for efficient and synergistic antibacterial wound therapy and healing promotion. J. Mater. Chem. B 2020, 8, 3908–3917. [Google Scholar] [CrossRef]
- Kuang, L.; Huang, J.; Liu, Y.; Li, X.; Yuan, Y.; Liu, C. Injectable hydrogel with NIR light-responsive, dual-mode PTH release for osteoregeneration in osteoporosis. Adv. Funct. Mater. 2021, 31, 2105383. [Google Scholar] [CrossRef]
- Gao, D.; Li, G.; Qu, X.; Li, X.; Xu, X.; Li, Y.; Zhou, Z. Thermo-responsive hydrogel with tunable transition temperature for effective drug release. Mater. Lett. 2022, 321, 132367. [Google Scholar] [CrossRef]
- Bellotti, E.; Schilling, A.L.; Little, S.R.; Decuzzi, P. Injectable thermoresponsive hydrogels as drug delivery system for the treatment of central nervous system disorders: A review. J. Control. Release 2021, 329, 16–35. [Google Scholar] [CrossRef] [PubMed]
- Rafael, D.; Melendres, M.M.R.; Andrade, F.; Montero, S.; Martinez-Trucharte, F.; Vilar-Hernandez, M.; Durán-Lara, E.F.; Schwartz, S., Jr.; Abasolo, I. Thermo-responsive hydrogels for cancer local therapy: Challenges and state-of-art. Int. J. Pharm. 2021, 606, 120954. [Google Scholar] [CrossRef]
- Zhang, K.; Xue, K.; Loh, X.J. Thermo-responsive hydrogels: From recent progress to biomedical applications. Gels 2021, 7, 77. [Google Scholar] [CrossRef]
- Lee, A.-J.; Lee, Y.-J.; Jeon, H.-Y.; Kim, M.; Han, E.-T.; Park, W.S.; Hong, S.-H.; Kim, Y.-M.; Ha, K.-S. Application of elastin-like biopolymer-conjugated C-peptide hydrogel for systemic long-term delivery against diabetic aortic dysfunction. Acta Biomater. 2020, 118, 32–43. [Google Scholar] [CrossRef]
- Chou, P.-Y.; Chen, S.-H.; Chen, C.-H.; Chen, S.-H.; Fong, Y.T.; Chen, J.-P. Thermo-responsive in-situ forming hydrogels as barriers to prevent post-operative peritendinous adhesion. Acta Biomater. 2017, 63, 85–95. [Google Scholar] [CrossRef]
- Yu, R.; Li, M.; Li, Z.; Pan, G.; Liang, Y.; Guo, B. Supramolecular thermo-contracting adhesive hydrogel with self-removability simultaneously enhancing noninvasive wound closure and MRSA-infected wound healing. Adv. Healthc. Mater. 2022, 11, 2102749. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Gao, H.; Liu, J.; Liang, H. Edible thermosensitive chitosan/hydroxypropyl β-cyclodextrin hydrogel with natural licoricidin for enhancing oral health: Biofilm disruption and demineralization prevention. Int. J. Biol. Macromol. 2024, 282, 136647. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Ma, T.; Jin, Y.; Qiu, S. The effects of morin on bone regeneration to accelerate healing in bone defects in mice. Int. J. Immunopathol. Pharmacol. 2020, 34, 2058738420962909. [Google Scholar] [CrossRef]
- Sareethammanuwat, M.; Boonyuen, S.; Arpornmaeklong, P. Effects of beta-tricalcium phosphate nanoparticles on the properties of a thermosensitive chitosan/collagen hydrogel and controlled release of quercetin. J. Biomed. Mater. Res. Part A 2021, 109, 1147–1159. [Google Scholar] [CrossRef] [PubMed]
- Kocak, F.Z.; Talari, A.C.; Yar, M.; Rehman, I.U. In-situ forming pH and thermosensitive injectable hydrogels to stimulate angiogenesis: Potential candidates for fast bone regeneration applications. Int. J. Mol. Sci. 2020, 21, 1633. [Google Scholar] [CrossRef]
- Tian, M.-P.; Zhang, A.-D.; Yao, Y.-X.; Chen, X.-G.; Liu, Y. Mussel-inspired adhesive and polypeptide-based antibacterial thermo-sensitive hydroxybutyl chitosan hydrogel as BMSCs 3D culture matrix for wound healing. Carbohydr. Polym. 2021, 261, 117878. [Google Scholar] [CrossRef]
- Castillo-Henríquez, L.; Sanabria-Espinoza, P.; Murillo-Castillo, B.; Montes de Oca-Vásquez, G.; Batista-Menezes, D.; Calvo-Guzmán, B.; Ramírez-Arguedas, N.; Vega-Baudrit, J. Topical chitosan-based thermo-responsive scaffold provides dexketoprofen trometamol controlled release for 24 h use. Pharmaceutics 2021, 13, 2100. [Google Scholar] [CrossRef]
- Talaat, W.; Aryal AC, S.; Al Kawas, S.; Samsudin, A.R.; Kandile, N.G.; Harding, D.R.; Ghoneim, M.M.; Zeiada, W.; Jagal, J.; Aboelnaga, A. Nanoscale thermosensitive hydrogel scaffolds promote the chondrogenic differentiation of dental pulp stem and progenitor cells: A minimally invasive approach for cartilage regeneration. Int. J. Nanomed. 2020, 15, 7775–7789. [Google Scholar] [CrossRef]
- Eltahir, S.; Jagal, J.; Abdelkareem, M.A.; Ghoneim, M.M.; Rawas-Qalaji, M.M.; Greish, K.; Haider, M. Thermosensitive injectable graphene oxide/chitosan-based nanocomposite hydrogels for controlling the in vivo release of bupivacaine hydrochloride. Int. J. Pharm. 2022, 621, 121786. [Google Scholar]
- Chai, Y.; Long, Y.; Dong, X.; Liu, K.; Wei, W.; Chen, Y.; Qiu, T.; Dai, H. Improved functional recovery of rat transected spinal cord by peptide-grafted PNIPAM based hydrogel. Colloids Surf. B Biointerfaces 2022, 210, 112220. [Google Scholar] [CrossRef] [PubMed]
- Binaymotlagh, R.; Chronopoulou, L.; Palocci, C. Peptide-based hydrogels: Template materials for tissue engineering. J. Funct. Biomater. 2023, 14, 233. [Google Scholar] [CrossRef] [PubMed]
- Radojković, N.; Spasojević, J.; Vukoje, I.; Kačarević-Popović, Z.; Stamenović, U.; Vodnik, V.; Roglić, G.; Radosavljević, A. Gamma Irradiation-Induced Synthesis of Nano Au-PNiPAAm/PVA Bi-Layered Photo-Thermo-Responsive Hydrogel Actuators with a Switchable Bending Motion. Polymers 2025, 17, 1774. [Google Scholar] [CrossRef]
- El-Husseiny, H.M.; Mady, E.A.; Hamabe, L.; Abugomaa, A.; Shimada, K.; Yoshida, T.; Tanaka, T.; Yokoi, A.; Elbadawy, M.; Tanaka, R. Smart/stimuli-responsive hydrogels: Cutting-edge platforms for tissue engineering and other biomedical applications. Mater. Today Bio 2022, 13, 100186. [Google Scholar] [CrossRef]
- Yuan, B.; Aziz, M.R.F.; Li, S.; Wu, J.; Li, D.; Li, R.-K. An electro-spun tri-component polymer biomaterial with optoelectronic properties for neuronal differentiation. Acta Biomater. 2022, 139, 82–90. [Google Scholar] [CrossRef]
- Si, Y.; Wang, L.; Wang, X.; Tang, N.; Yu, J.; Ding, B. Ultrahigh-water-content, superelastic, and shape-memory nanofiber-assembled hydrogels exhibiting pressure-responsive conductivity. Adv. Mater. 2017, 29, 1700339. [Google Scholar] [CrossRef]
- Wang, X.; Ding, B.; Sun, G.; Wang, M.; Yu, J. Electro-spinning/netting: A strategy for the fabrication of three-dimensional polymer nano-fiber/nets. Prog. Mater. Sci. 2013, 58, 1173–1243. [Google Scholar] [CrossRef]
- Di, J.; Yao, S.; Ye, Y.; Cui, Z.; Yu, J.; Ghosh, T.K.; Zhu, Y.; Gu, Z. Stretch-triggered drug delivery from wearable elastomer films containing therapeutic depots. ACS Nano 2015, 9, 9407–9415. [Google Scholar] [CrossRef]
- Korin, N.; Kanapathipillai, M.; Ingber, D.E. Shear-responsive platelet mimetics for targeted drug delivery. Isr. J. Chem. 2013, 53, 610–615. [Google Scholar] [CrossRef]
- Zhu, T.; Cheng, Y.; Cao, C.; Mao, J.; Li, L.; Huang, J.; Gao, S.; Dong, X.; Chen, Z.; Lai, Y. A semi-interpenetrating network ionic hydrogel for strain sensing with high sensitivity, large strain range, and stable cycle performance. Chem. Eng. J. 2020, 385, 123912. [Google Scholar] [CrossRef]
- Ye, H.; Zhou, Y.; Liu, X.; Chen, Y.; Duan, S.; Zhu, R.; Liu, Y.; Yin, L. Recent advances on reactive oxygen species-responsive delivery and diagnosis system. Biomacromolecules 2019, 20, 2441–2463. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Liu, Z.; Yu, Q.; Ma, T. Preparation of reactive oxygen species-responsive antibacterial hydrogels for efficient anti-infection therapy. Mater. Lett. 2020, 263, 127254. [Google Scholar] [CrossRef]
- Fan, Z.; Xu, H. Recent progress in the biological applications of reactive oxygen species-responsive polymers. Polym. Rev. 2020, 60, 114–143. [Google Scholar] [CrossRef]
- Zhao, H.; Huang, J.; Li, Y.; Lv, X.; Zhou, H.; Wang, H.; Xu, Y.; Wang, C.; Wang, J.; Liu, Z. ROS-scavenging hydrogel to promote healing of bacteria infected diabetic wounds. Biomaterials 2020, 258, 120286. [Google Scholar] [CrossRef]
- Li, X.; Xu, K.; He, Y.; Tao, B.; Li, K.; Lin, C.; Hu, J.; Wu, J.; Wu, Y.; Liu, S. ROS-responsive hydrogel coating modified titanium promotes vascularization and osteointegration of bone defects by orchestrating immunomodulation. Biomaterials 2022, 287, 121683. [Google Scholar] [CrossRef]
- Zheng, Z.; Lei, C.; Liu, H.; Jiang, M.; Zhou, Z.; Zhao, Y.; Yu, C.Y.; Wei, H. A ROS-responsive liposomal composite hydrogel integrating improved mitochondrial function and pro-angiogenesis for efficient treatment of myocardial infarction. Adv. Healthc. Mater. 2022, 11, 2200990. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, T.; Ding, J.; Gu, H.; Wang, Q.; Wang, Y.; Zhang, D.; Gao, C. A reactive oxygen species-responsive hydrogel encapsulated with bone marrow derived stem cells promotes repair and regeneration of spinal cord injury. Bioact. Mater. 2023, 19, 550–568. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Zhang, J.; Yang, D.; Shao, J.; Wang, W.; Zhang, Q.; Dong, X. Recent advances in pH-responsive nanomaterials for anti-infective therapy. J. Mater. Chem. B 2020, 8, 10700–10711. [Google Scholar] [CrossRef] [PubMed]
- Ofridam, F.; Tarhini, M.; Lebaz, N.; Gagnière, É.; Mangin, D.; Elaissari, A. pH-sensitive polymers: Classification and some fine potential applications. Polym. Adv. Technol. 2021, 32, 1455–1484. [Google Scholar] [CrossRef]
- Raza, F.; Zhu, Y.; Chen, L.; You, X.; Zhang, J.; Khan, A.; Khan, M.W.; Hasnat, M.; Zafar, H.; Wu, J. Paclitaxel-loaded pH responsive hydrogel based on self-assembled peptides for tumor targeting. Biomater. Sci. 2019, 7, 2023–2036. [Google Scholar] [CrossRef]
- Hu, C.; Long, L.; Cao, J.; Zhang, S.; Wang, Y. Dual-crosslinked mussel-inspired smart hydrogels with enhanced antibacterial and angiogenic properties for chronic infected diabetic wound treatment via pH-responsive quick cargo release. Chem. Eng. J. 2021, 411, 128564. [Google Scholar] [CrossRef]
- Zhao, C.; Qazvini, N.T.; Sadati, M.; Zeng, Z.; Huang, S.; De La Lastra, A.L.; Zhang, L.; Feng, Y.; Liu, W.; Huang, B. A pH-triggered, self-assembled, and bioprintable hybrid hydrogel scaffold for mesenchymal stem cell based bone tissue engineering. ACS Appl. Mater. Interfaces 2019, 11, 8749–8762. [Google Scholar] [CrossRef]
- Lv, J.; Wu, G.; Liu, Y.; Li, C.; Huang, F.; Zhang, Y.; Liu, J.; An, Y.; Ma, R.; Shi, L. Injectable dual glucose-responsive hydrogel-micelle composite for mimicking physiological basal and prandial insulin delivery. Sci. China Chem. 2019, 62, 637–648. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Z.; Yu, J.; Kahkoska, A.R.; Buse, J.B.; Gu, Z. Glucose-responsive insulin and delivery systems: Innovation and translation. Adv. Mater. 2020, 32, 1902004. [Google Scholar] [CrossRef]
- Holz, E.; Rajagopal, K. In situ-forming glucose-responsive hydrogel from hyaluronic acid modified with a boronic acid derivative. Macromol. Chem. Phys. 2020, 221, 2000055. [Google Scholar] [CrossRef]
- Walter, S.V.; Ennen-Roth, F.; Büning, D.; Denizer, D.; Ulbricht, M. Glucose-responsive polymeric hydrogel materials: From a novel technique for the measurement of glucose binding toward swelling pressure sensor applications. ACS Appl. Bio Mater. 2019, 2, 2464–2480. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Mao, J.; Li, W.; Xu, C.; Lao, A.; Shin, A.; Wu, J.; Gu, A.; Zhang, Z.; Mao, L. Smart glucose-responsive hydrogel with ROS scavenging and homeostasis regulating properties for diabetic bone regeneration. Chem. Eng. J. 2024, 497, 154433. [Google Scholar] [CrossRef]
- Li, G.; Zhou, D.; Sheng, S.; Lin, Q.; Jing, Y.; Ren, X.; Su, J. Hydrogel for bone microenvironment: Strategy and application. Chem. Eng. J. 2024, 499, 156554. [Google Scholar] [CrossRef]
- Zhao, L.; Niu, L.; Liang, H.; Tan, H.; Liu, C.; Zhu, F. pH and glucose dual-responsive injectable hydrogels with insulin and fibroblasts as bioactive dressings for diabetic wound healing. ACS Appl. Mater. Interfaces 2017, 9, 37563–37574. [Google Scholar] [CrossRef]
- Chandrawati, R. Enzyme-responsive polymer hydrogels for therapeutic delivery. Exp. Biol. Med. 2016, 241, 972–979. [Google Scholar] [CrossRef]
- Shahriari, M.; Zahiri, M.; Abnous, K.; Taghdisi, S.M.; Ramezani, M.; Alibolandi, M. Enzyme responsive drug delivery systems in cancer treatment. J. Control. Release 2019, 308, 172–189. [Google Scholar] [CrossRef]
- Skaalure, S.C.; Chu, S.; Bryant, S.J. An enzyme-sensitive PEG hydrogel based on aggrecan catabolism for cartilage tissue engineering. Adv. Healthc. Mater. 2015, 4, 420–431. [Google Scholar] [CrossRef]
- Zhao, Z.; Shen, J.; Zhang, L.; Wang, L.; Xu, H.; Han, Y.; Jia, J.; Lu, Y.; Yu, R.; Liu, H. Injectable postoperative enzyme-responsive hydrogels for reversing temozolomide resistance and reducing local recurrence after glioma operation. Biomater. Sci. 2020, 8, 5306–5316. [Google Scholar] [CrossRef]
- Cai, C.; Wang, W.; Liang, J.; Li, Y.; Lu, M.; Cui, W.; Fan, C.; Deng, L.; Li, Y.; Wang, F. MMP-2 responsive unidirectional hydrogel-electrospun patch loading TGF-β1 siRNA polyplexes for peritendinous anti-adhesion. Adv. Funct. Mater. 2021, 31, 2008364. [Google Scholar] [CrossRef]
- Hendrickson, G.R.; Lyon, L.A. Bioresponsive hydrogels for sensing applications. Soft Matter 2009, 5, 29–35. [Google Scholar] [CrossRef]
- Ojha, S.; Singh, D.; Sett, A.; Chetia, H.; Kabiraj, D.; Bora, U. Nanotechnology in crop protection. In Nanomaterials in Plants, Algae, and Microorganisms; Elsevier: Amsterdam, The Netherlands, 2018; pp. 345–391. [Google Scholar]
- Choi, E.; Choi, Y.; Nejad, Y.H.P.; Shin, K.; Park, J. Label-free specific detection of immunoglobulin G antibody using nanoporous hydrogel photonic crystals. Sens. Actuators B Chem. 2013, 180, 107–113. [Google Scholar] [CrossRef]
- Liu, R.; Huang, Y.; Ma, Y.; Jia, S.; Gao, M.; Li, J.; Zhang, H.; Xu, D.; Wu, M.; Chen, Y. Design and synthesis of target-responsive aptamer-cross-linked hydrogel for visual quantitative detection of ochratoxin A. ACS Appl. Mater. Interfaces 2015, 7, 6982–6990. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Tao, Z.; Dai, Z.; Gao, Y.; Chu, C.; Fan, C.; Liu, S.; Ma, X.; Jin, F.; You, Z. Magnetically Guided Mechanoactive Mineralization Scaffolds for Enhanced Bone Regeneration. Adv. Funct. Mater. 2025, 35, 2503903. [Google Scholar] [CrossRef]
- Mierke, C.T. Viscoelasticity acts as a marker for tumor extracellular matrix characteristics. Front. Cell Dev. Biol. 2021, 9, 785138. [Google Scholar] [CrossRef]
- Song, J.; Deiss-Yehiely, E.; Yesilata, S.; McKinley, G.H. Strain-stiffening universality in composite hydrogels and soft tissues. Nat. Phys. 2025, 21, 1–9. [Google Scholar] [CrossRef]
- Broughton, K.M.; Sussman, M.A. Cardiac tissue engineering therapeutic products to enhance myocardial contractility. J. Muscle Res. Cell Motil. 2020, 41, 363–373. [Google Scholar] [CrossRef]
- Chachques, J.C.; Trainini, J.C.; Lago, N.; Cortes-Morichetti, M.; Schussler, O.; Carpentier, A. Myocardial assistance by grafting a new bioartificial upgraded myocardium (MAGNUM trial): Clinical feasibility study. Ann. Thorac. Surg. 2008, 85, 901–908. [Google Scholar] [CrossRef]
- Komeri, R.; Muthu, J. Injectable, cytocompatible, elastic, free radical scavenging and electroconductive hydrogel for cardiac cell encapsulation. Colloids Surf. B Biointerfaces 2017, 157, 381–390. [Google Scholar] [CrossRef]
- Walker, B.W.; Lara, R.P.; Mogadam, E.; Yu, C.H.; Kimball, W.; Annabi, N. Rational design of microfabricated electroconductive hydrogels for biomedical applications. Prog. Polym. Sci. 2019, 92, 135–157. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, N.; Ma, M. Electroconductive hydrogels for biomedical applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1568. [Google Scholar] [CrossRef]
- Ogle, B.M.; Bursac, N.; Domian, I.; Huang, N.F.; Menasché, P.; Murry, C.E.; Pruitt, B.; Radisic, M.; Wu, J.C.; Wu, S.M. Distilling complexity to advance cardiac tissue engineering. Sci. Transl. Med. 2016, 8, 342ps13. [Google Scholar] [CrossRef]
- Liang, S.; Zhang, Y.; Wang, H.; Xu, Z.; Chen, J.; Bao, R.; Tan, B.; Cui, Y.; Fan, G.; Wang, W. Paintable and rapidly bondable conductive hydrogels as therapeutic cardiac patches. Adv. Mater. 2018, 30, 1704235. [Google Scholar] [CrossRef]
- Dorsey, S.M.; Burdick, J.A. Hydrogels in cardiac tissue engineering. In GELS HANDBOOK: Fundamentals, Properties and Applications Volume 2: Applications of Hydrogels in Regenerative Medicine; World Scientific: Singapore, 2016; pp. 323–361. [Google Scholar]
- Florea, V.; Rieger, A.C.; DiFede, D.L.; El-Khorazaty, J.; Natsumeda, M.; Banerjee, M.N.; Tompkins, B.A.; Khan, A.; Schulman, I.H.; Landin, A.M. Dose comparison study of allogeneic mesenchymal stem cells in patients with ischemic cardiomyopathy (The TRIDENT Study). Circ. Res. 2017, 121, 1279–1290. [Google Scholar] [CrossRef]
- Menasché, P.; Vanneaux, V.; Hagège, A.; Bel, A.; Cholley, B.; Parouchev, A.; Cacciapuoti, I.; Al-Daccak, R.; Benhamouda, N.; Blons, H. Transplantation of human embryonic stem cell–derived cardiovascular progenitors for severe ischemic left ventricular dysfunction. J. Am. Coll. Cardiol. 2018, 71, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Broughton, K.M.; Sussman, M.A. Empowering adult stem cells for myocardial regeneration V2. 0: Success in small steps. Circ. Res. 2016, 118, 867–880. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Zhao, X.; Guo, B.; Ma, P.X. Self-healing conductive injectable hydrogels with antibacterial activity as cell delivery carrier for cardiac cell therapy. ACS Appl. Mater. Interfaces 2016, 8, 17138–17150. [Google Scholar] [CrossRef]
- Tomov, M.L.; Gil, C.J.; Cetnar, A.; Theus, A.S.; Lima, B.J.; Nish, J.E.; Bauser-Heaton, H.D.; Serpooshan, V. Engineering functional cardiac tissues for regenerative medicine applications. Curr. Cardiol. Rep. 2019, 21, 105. [Google Scholar] [CrossRef]
- Wang, W.; Tan, B.; Chen, J.; Bao, R.; Zhang, X.; Liang, S.; Shang, Y.; Liang, W.; Cui, Y.; Fan, G. An injectable conductive hydrogel encapsulating plasmid DNA-eNOs and ADSCs for treating myocardial infarction. Biomaterials 2018, 160, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Roshanbinfar, K.; Mohammadi, Z.; Mesgar, A.S.-M.; Dehghan, M.M.; Oommen, O.P.; Hilborn, J.; Engel, F.B. Carbon nanotube doped pericardial matrix derived electroconductive biohybrid hydrogel for cardiac tissue engineering. Biomater. Sci. 2019, 7, 3906–3917. [Google Scholar] [CrossRef]
- Karantalis, V.; DiFede, D.L.; Gerstenblith, G.; Pham, S.; Symes, J.; Zambrano, J.P.; Fishman, J.; Pattany, P.; McNiece, I.; Conte, J. Autologous mesenchymal stem cells produce concordant improvements in regional function, tissue perfusion, and fibrotic burden when administered to patients undergoing coronary artery bypass grafting: The Prospective Randomized Study of Mesenchymal Stem Cell Therapy in Patients Undergoing Cardiac Surgery (PROMETHEUS) trial. Circ. Res. 2014, 114, 1302–1310. [Google Scholar]
- Yang, B.; Yao, F.; Hao, T.; Fang, W.; Ye, L.; Zhang, Y.; Wang, Y.; Li, J.; Wang, C. Development of electrically conductive double-network hydrogels via one-step facile strategy for cardiac tissue engineering. Adv. Healthc. Mater. 2016, 5, 474–488. [Google Scholar] [CrossRef]
- Roshanbinfar, K.; Vogt, L.; Greber, B.; Diecke, S.; Boccaccini, A.R.; Scheibel, T.; Engel, F.B. Electroconductive biohybrid hydrogel for enhanced maturation and beating properties of engineered cardiac tissues. Adv. Funct. Mater. 2018, 28, 1803951. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, L.; Guo, B.; Ma, P.X. Interwoven aligned conductive nanofiber yarn/hydrogel composite scaffolds for engineered 3D cardiac anisotropy. Acs Nano 2017, 11, 5646–5659. [Google Scholar] [CrossRef]
- Navaei, A.; Eliato, K.R.; Ros, R.; Migrino, R.Q.; Willis, B.C.; Nikkhah, M. The influence of electrically conductive and non-conductive nanocomposite scaffolds on the maturation and excitability of engineered cardiac tissues. Biomater. Sci. 2019, 7, 585–595. [Google Scholar] [CrossRef]
- Hosoyama, K.; Ahumada, M.; McTiernan, C.D.; Davis, D.R.; Variola, F.; Ruel, M.; Liang, W.; Suuronen, E.J.; Alarcon, E.I. Nanoengineered electroconductive collagen-based cardiac patch for infarcted myocardium repair. ACS Appl. Mater. Interfaces 2018, 10, 44668–44677. [Google Scholar] [CrossRef]
- Jing, X.; Mi, H.-Y.; Napiwocki, B.N.; Peng, X.-F.; Turng, L.-S. Mussel-inspired electroactive chitosan/graphene oxide composite hydrogel with rapid self-healing and recovery behavior for tissue engineering. Carbon 2017, 125, 557–570. [Google Scholar] [CrossRef]
- Noshadi, I.; Walker, B.W.; Portillo-Lara, R.; Shirzaei Sani, E.; Gomes, N.; Aziziyan, M.R.; Annabi, N. Engineering biodegradable and biocompatible bio-ionic liquid conjugated hydrogels with tunable conductivity and mechanical properties. Sci. Rep. 2017, 7, 4345. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.R.; Zihlmann, C.; Akbari, M.; Assawes, P.; Cheung, L.; Zhang, K.; Manoharan, V.; Zhang, Y.S.; Yüksekkaya, M.; Wan, K.t. Reduced graphene oxide-gelMA hybrid hydrogels as scaffolds for cardiac tissue engineering. Small 2016, 12, 3677–3689. [Google Scholar] [CrossRef] [PubMed]
- Alegret, N.; Dominguez-Alfaro, A.; Mecerreyes, D. 3D scaffolds based on conductive polymers for biomedical applications. Biomacromolecules 2018, 20, 73–89. [Google Scholar] [CrossRef]
- Nezakati, T.; Seifalian, A.; Tan, A.; Seifalian, A.M. Conductive polymers: Opportunities and challenges in biomedical applications. Chem. Rev. 2018, 118, 6766–6843. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Patsis, P.A.; Hauser, S.; Voigt, D.; Rothe, R.; Günther, M.; Cui, M.; Yang, X.; Wieduwild, R.; Eckert, K. Cytocompatible, injectable, and electroconductive soft adhesives with hybrid covalent/noncovalent dynamic network. Adv. Sci. 2019, 6, 1802077. [Google Scholar] [CrossRef]
- Bertucci, C.; Koppes, R.; Dumont, C.; Koppes, A. Neural responses to electrical stimulation in 2D and 3D in vitro environments. Brain Res. Bull. 2019, 152, 265–284. [Google Scholar] [CrossRef]
- Xu, C.; Guan, S.; Wang, S.; Gong, W.; Liu, T.; Ma, X.; Sun, C. Biodegradable and electroconductive poly (3, 4-ethylenedioxythiophene)/carboxymethyl chitosan hydrogels for neural tissue engineering. Mater. Sci. Eng. C 2018, 84, 32–43. [Google Scholar] [CrossRef]
- Liu, X.; Miller, A.L.; Park, S.; Waletzki, B.E.; Zhou, Z.; Terzic, A.; Lu, L. Functionalized carbon nanotube and graphene oxide embedded electrically conductive hydrogel synergistically stimulates nerve cell differentiation. ACS Appl. Mater. Interfaces 2017, 9, 14677–14690. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, L.; Guo, B.; Shao, Y.; Ma, P.X. Electroactive biodegradable polyurethane significantly enhanced Schwann cells myelin gene expression and neurotrophin secretion for peripheral nerve tissue engineering. Biomaterials 2016, 87, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Manoharan, V.; Cheung, L.; Lee, S.; Cha, B.-H.; Newman, P.; Farzad, R.; Mehrotra, S.; Zhang, K.; Khan, F. Nanoparticle-based hybrid scaffolds for deciphering the role of multimodal cues in cardiac tissue engineering. ACS Nano 2019, 13, 12525–12539. [Google Scholar] [CrossRef]
- Baei, P.; Jalili-Firoozinezhad, S.; Rajabi-Zeleti, S.; Tafazzoli-Shadpour, M.; Baharvand, H.; Aghdami, N. Electrically conductive gold nanoparticle-chitosan thermosensitive hydrogels for cardiac tissue engineering. Mater. Sci. Eng. C 2016, 63, 131–141. [Google Scholar] [CrossRef]
- Zhou, L.; Fan, L.; Yi, X.; Zhou, Z.; Liu, C.; Fu, R.; Dai, C.; Wang, Z.; Chen, X.; Yu, P. Soft conducting polymer hydrogels cross-linked and doped by tannic acid for spinal cord injury repair. Acs Nano 2018, 12, 10957–10967. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Chang, Y.; Wu, P.; Liu, K.; Dong, X.; Nie, A.; Mu, C.; Liu, Z.; Dai, H.; Luo, Z. Two-dimensional-germanium phosphide-reinforced conductive and biodegradable hydrogel scaffolds enhance spinal cord injury repair. Adv. Funct. Mater. 2021, 31, 2104440. [Google Scholar] [CrossRef]
- Yan, L.; Zhou, T.; Han, L.; Zhu, M.; Cheng, Z.; Li, D.; Ren, F.; Wang, K.; Lu, X. Conductive cellulose bio-nanosheets assembled biostable hydrogel for reliable bioelectronics. Adv. Funct. Mater. 2021, 31, 2010465. [Google Scholar] [CrossRef]
- Zhang, B.; He, J.; Shi, M.; Liang, Y.; Guo, B. Injectable self-healing supramolecular hydrogels with conductivity and photo-thermal antibacterial activity to enhance complete skin regeneration. Chem. Eng. J. 2020, 400, 125994. [Google Scholar] [CrossRef]
- Yang, B.; Liang, C.; Chen, D.; Cheng, F.; Zhang, Y.; Wang, S.; Shu, J.; Huang, X.; Wang, J.; Xia, K. A conductive supramolecular hydrogel creates ideal endogenous niches to promote spinal cord injury repair. Bioact. Mater. 2022, 15, 103–119. [Google Scholar] [CrossRef]
- Chakraborty, P.; Oved, H.; Bychenko, D.; Yao, Y.; Tang, Y.; Zilberzwige-Tal, S.; Wei, G.; Dvir, T.; Gazit, E. Nanoengineered peptide-based antimicrobial conductive supramolecular biomaterial for cardiac tissue engineering. Adv. Mater. 2021, 33, 2008715. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Wei, J.; Xu, B.; Liu, X.; Wang, H.; Wang, W.; Wang, Q.; Liu, W. A robust, highly stretchable supramolecular polymer conductive hydrogel with self-healability and thermo-processability. Sci. Rep. 2017, 7, 41566. [Google Scholar] [CrossRef]
- Criado-Gonzalez, M.; Alegret, N.; Fracaroli, A.M.; Mantione, D.; Guzmán-González, G.; Del Olmo, R.; Tashiro, K.; Tomé, L.C.; Picchio, M.L.; Mecerreyes, D. Mixed conductive, injectable, and fluorescent supramolecular eutectogel composites. Angew. Chem. 2023, 135, e202301489. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Tsai, T.-Y.; Young, T.-H.; Yang, H.J.; Ji, Y.-R. An electroactive alginate hydrogel nanocomposite reinforced by functionalized graphite nanofilaments for neural tissue engineering. Carbohydr. Polym. 2019, 224, 115112. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Y.; Zhang, J.; Hu, X.; Yang, Z.; Guo, Y.; Wang, Y. In-situ doping of a conductive hydrogel with low protein absorption and bacterial adhesion for electrical stimulation of chronic wounds. Acta Biomater. 2019, 89, 217–226. [Google Scholar] [CrossRef]
- Jia, Z.; Gong, J.; Zeng, Y.; Ran, J.; Liu, J.; Wang, K.; Xie, C.; Lu, X.; Wang, J. Bioinspired conductive silk microfiber integrated bioelectronic for diagnosis and wound healing in diabetes. Adv. Funct. Mater. 2021, 31, 2010461. [Google Scholar] [CrossRef]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial anti-oxidant electroactive injectable hydrogel as self-healing wound dressing with hemostasis and adhesiveness for cutaneous wound healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef]
- Li, M.; Liang, Y.; He, J.; Zhang, H.; Guo, B. Two-pronged strategy of biomechanically active and biochemically multifunctional hydrogel wound dressing to accelerate wound closure and wound healing. Chem. Mater. 2020, 32, 9937–9953. [Google Scholar] [CrossRef]
- Jia, Z.; Lv, X.; Hou, Y.; Wang, K.; Ren, F.; Xu, D.; Wang, Q.; Fan, K.; Xie, C.; Lu, X. Mussel-inspired nanozyme catalyzed conductive and self-setting hydrogel for adhesive and antibacterial bioelectronics. Bioact. Mater. 2021, 6, 2676–2687. [Google Scholar] [CrossRef]
- Tamay, D.G.; Dursun Usal, T.; Alagoz, A.S.; Yucel, D.; Hasirci, N.; Hasirci, V. 3D and 4D printing of polymers for tissue engineering applications. Front. Bioeng. Biotechnol. 2019, 7, 164. [Google Scholar] [CrossRef]
- Mathur, V.; Agarwal, P.; Kasturi, M.; Varadharajan, S.; Devi, E.S.; Vasanthan, K.S. Transformative bioprinting: 4D printing and its role in the evolution of engineering and personalized medicine. Discov. Nano 2025, 20, 118. [Google Scholar] [CrossRef]
- Mohammadi, S.; Cidonio, G. Unravelling hierarchical patterning of biomaterial inks with 3D microfluidic-assisted spinning: A paradigm shift in bioprinting technologies. Front. Biomater. Sci. 2023, 2, 1279061. [Google Scholar] [CrossRef]
- Douglas, A.; Chen, Y.; Elloso, M.; Levschuk, A.; Jeschke, M.G. Bioprinting-By-Design of Hydrogel-Based Biomaterials for In Situ Skin Tissue Engineering. Gels 2025, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Blache, U.; Ford, E.M.; Ha, B.; Rijns, L.; Chaudhuri, O.; Dankers, P.Y.; Kloxin, A.M.; Snedeker, J.G.; Gentleman, E. Engineered hydrogels for mechanobiology. Nat. Rev. Methods Primers 2022, 2, 98. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, O.; Gu, L.; Klumpers, D.; Darnell, M.; Bencherif, S.A.; Weaver, J.C.; Huebsch, N.; Lee, H.-p.; Lippens, E.; Duda, G.N. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 2016, 15, 326–334. [Google Scholar] [CrossRef]
- Chaudhuri, O.; Gu, L.; Darnell, M.; Klumpers, D.; Bencherif, S.A.; Weaver, J.C.; Huebsch, N.; Mooney, D.J. Substrate stress relaxation regulates cell spreading. Nat. Commun. 2015, 6, 6365. [Google Scholar] [CrossRef]
- Gong, Z.; Szczesny, S.E.; Caliari, S.R.; Charrier, E.E.; Chaudhuri, O.; Cao, X.; Lin, Y.; Mauck, R.L.; Janmey, P.A.; Burdick, J.A. Matching material and cellular timescales maximizes cell spreading on viscoelastic substrates. Proc. Natl. Acad. Sci. USA 2018, 115, E2686–E2695. [Google Scholar] [CrossRef]
- Wei, Z.; Schnellmann, R.; Pruitt, H.C.; Gerecht, S. Hydrogel network dynamics regulate vascular morphogenesis. Cell Stem Cell 2020, 27, 798–812.e796. [Google Scholar] [CrossRef]
- Wang, W.; Xiang, L.; Diaz-Dussan, D.; Zhang, J.; Yang, W.; Gong, L.; Chen, J.; Narain, R.; Zeng, H. Dynamic flexible hydrogel network with biological tissue-like self-protective functions. Chem. Mater. 2020, 32, 10545–10555. [Google Scholar] [CrossRef]
- Fernandez-Castano Romera, M.; Lafleur, R.P.; Guibert, C.; Voets, I.K.; Storm, C.; Sijbesma, R.P. Strain stiffening hydrogels through self-assembly and covalent fixation of semi-flexible fibers. Angew. Chem. 2017, 129, 8897–8901. [Google Scholar] [CrossRef]
- Khaki, N.; Sharifi, E.; Solati-Hashjin, M.; Abolfathi, N. Influence of scaffold geometry on the degradation rate of 3D printed polylactic acid bone scaffold. J. Biomater. Appl. 2025, 39, 734–747. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Cai, L.; Li, X.; Ye, L.; Xie, J. Stiffness and topography of biomaterials dictate cell-matrix interaction in musculoskeletal cells at the bio-interface: A concise progress review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 2426–2440. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Han, Z.; Naito, M.; Maeyama, A.; Kim, S.H.; Kim, Y.H.; Matsuda, T. Articular cartilage tissue engineering based on a mechano-active scaffold made of poly (l-lactide-co-ε-caprolactone): In vivo performance in adult rabbits. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 94, 80–88. [Google Scholar] [CrossRef] [PubMed]



| Stimulus Type | Mechanism | Advantages | Key Applications | Ref. |
|---|---|---|---|---|
| Ultrasound | Acoustic waves trigger hydrogel deformation, payload release, or electrical output | Non-invasive, controllable, self-healing, deep tissue penetration | Bone repair, nerve stimulation, anticancer therapy, tissue regeneration | [21] |
| Magnetic Field | Magnetic nanoparticles respond via motion, heat, or deformation | Remote control, precise spatiotemporal regulation, deep tissue penetration | Bone, cartilage, nerve, cardiac, skin tissue engineering; targeted drug delivery | [28] |
| Light (UV/Vis/NIR) | Photon absorption induces swelling, phase transition, or chemical modification | High spatial/temporal resolution, non-invasive, tunable | Tissue engineering, neuronal differentiation, antimicrobial dressings, bone repair | [44] |
| Temperature | Sol-gel transitions via hydrophilic/hydrophobic balance | Injectable, in situ formation, biocompatible, minimally invasive | Wound healing, bone/dental regeneration, soft robotics, pain management | [53] |
| Pressure/Mechanical | Compression or shear stress induces network deformation or drug release | Mechanically sensitive, wearable/injectable, localized delivery | Drug delivery under strain, vascular-targeted therapy, strain sensors, tissue engineering | [71] |
| pH | Protonation/deprotonation of polymer chains | Site-specific, responsive to acidic microenvironments | Chronic wound healing, bone regeneration, injectable scaffolds | [84] |
| Glucose | Glucose-binding (GOx, Con A, PBA) triggers hydrogel response | Self-regulated, dynamic, multifunctional | Diabetic wound healing, bone/cartilage repair, biosensors, 3D-printed scaffolds | [90] |
| ROS | Oxidative stress induces degradation or drug release | Reduces oxidative damage, immunomodulatory | Wound healing, spinal cord repair, cardiovascular therapy, stem cell delivery | [77] |
| Enzyme | Hydrogel cleaved or restructured by specific enzymes (MMPs, aggrecanase) | Tissue-specific, controlled degradation, high precision | Cartilage engineering, glioma therapy, tendon anti-adhesion scaffolds | [96] |
| Antigen/Antibody | Specific binding alters hydrogel network | Highly selective, sensitive, label-free | Biosensing, immunodetection, controlled drug delivery | [101] |
| Hydrogel Component | Conductive Material | Application | Ref. |
|---|---|---|---|
| QCS-CD-AD/GO | GO | Wound healing | [144] |
| Aga/Gel/PPY (AGP3) | PPY | Spinal cord injury repair | [145] |
| Fmoc-K- (Fmoc)-RGDPANI | PANI | Cardiac tissue engineering | [146] |
| PNAGAPAMPS/PEDOT/PSS | PEDOT/PSS | Scaffold materials for soft tissue engineering | [147] |
| TA/PPY-Fe3+ | PPY | Spinal cord injury repair | [148] |
| Hyaluronic acid | Cardiac Tetraaniline | Cardiac tissue engineering | [121] |
| PGS | Aniline | Neural tissue engineering | [138] |
| Alginate | CAGNF | Neural tissue engineering | [149] |
| GelMA or PEGDA | Bio-IL | Cardiac tissue engineering | [130] |
| PDA@rGO/cellulose nanofibers | PDA@rGO | Skin tissue engineering | [143] |
| polyHEMA/PPy | PPY | Diabetic wound | [150] |
| PEDOT-PDA-mSF | PEDOT | Diabetic wound | [151] |
| QCS-g-PANi/PEGS-FA | PANI | Antibacterial-wound healing | [152] |
| QCS/PDA@rGO/PNIPAm | GO | Tissue adhesive | [153] |
| PAA/TA-Ag nanozyme | TA-Ag | Infected wound healing | [154] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sojdeh, S.; Panjipour, A.; Castillo, M.; Arabpour, Z.; Djalilian, A.R. Emerging Smart and Adaptive Hydrogels for Next-Generation Tissue Engineering. Bioengineering 2026, 13, 50. https://doi.org/10.3390/bioengineering13010050
Sojdeh S, Panjipour A, Castillo M, Arabpour Z, Djalilian AR. Emerging Smart and Adaptive Hydrogels for Next-Generation Tissue Engineering. Bioengineering. 2026; 13(1):50. https://doi.org/10.3390/bioengineering13010050
Chicago/Turabian StyleSojdeh, Soheil, Amirhosein Panjipour, Miranda Castillo, Zohreh Arabpour, and Ali R. Djalilian. 2026. "Emerging Smart and Adaptive Hydrogels for Next-Generation Tissue Engineering" Bioengineering 13, no. 1: 50. https://doi.org/10.3390/bioengineering13010050
APA StyleSojdeh, S., Panjipour, A., Castillo, M., Arabpour, Z., & Djalilian, A. R. (2026). Emerging Smart and Adaptive Hydrogels for Next-Generation Tissue Engineering. Bioengineering, 13(1), 50. https://doi.org/10.3390/bioengineering13010050








