Jawbone Cavitations: Current Understanding and Conceptual Introduction of Covered Socket Residuum (CSR)
Abstract
1. Introduction
2. Methods
3. Results
3.1. Etiology of Jawbone Cavitations: Current Concepts and Theories
| Author (Year) | Study Type | Terminology Used | Primary Method | Etiological Concept |
|---|---|---|---|---|
| Roberts & Person (1979) [2] | Retrospective case series | Bone cavities at previous extraction sites | Clinical observation | Facial pain phenomena associated with cavitations at former extraction sites |
| Ratner et al. (1979) [14] | Retrospective case series | Bone cavities | Histopathological and microbiological analysis | Chronic lymphocytic inflammation with polymicrobial flora |
| Bouquot (1992) [23] | Retrospective observational study | Neuralgia-inducing cavitational osteonecrosis (NICO) | Histopathological examination | Chronic intraosseous inflammation, marrow fibrosis, necrotic bone fragments |
| Gruppo et al. (1996) [20] | Case–control study | Neuralgia-inducing cavitational osteonecrosis (NICO) | Blood coagulation and fibrinolysis analysis | Defects in thrombotic and fibrinolytic systems |
| Lechner & Mayer (2010) [11] | Retrospective case series | Neuralgia-inducing cavitational osteonecrosis (NICO) | Multiplex cytokine analysis | Overexpression of RANTES and IL-1 |
| Lechner (2014) [24] | Case–control study | Fatty degenerative osteolysis of the jawbone (FDOJ) | Correlation of radiography with inflammatory markers | Marked RANTES elevation; 2D radiography insufficient |
| Ghanaati et al. (2025) [10] | Retrospective case–control study | Jawbone cavitation | Three-dimensional radiological assessment (CBCT) | Cavitations as result of socket collapse after tooth loss |
| Ghanaati et al. (2025) [22] | Prospective randomized controlled trial | Covered Socket Residuum (CSR) | Three-dimensional radiological assessment (CBCT) | Covered non-mineralized intraosseous regions after extraction |
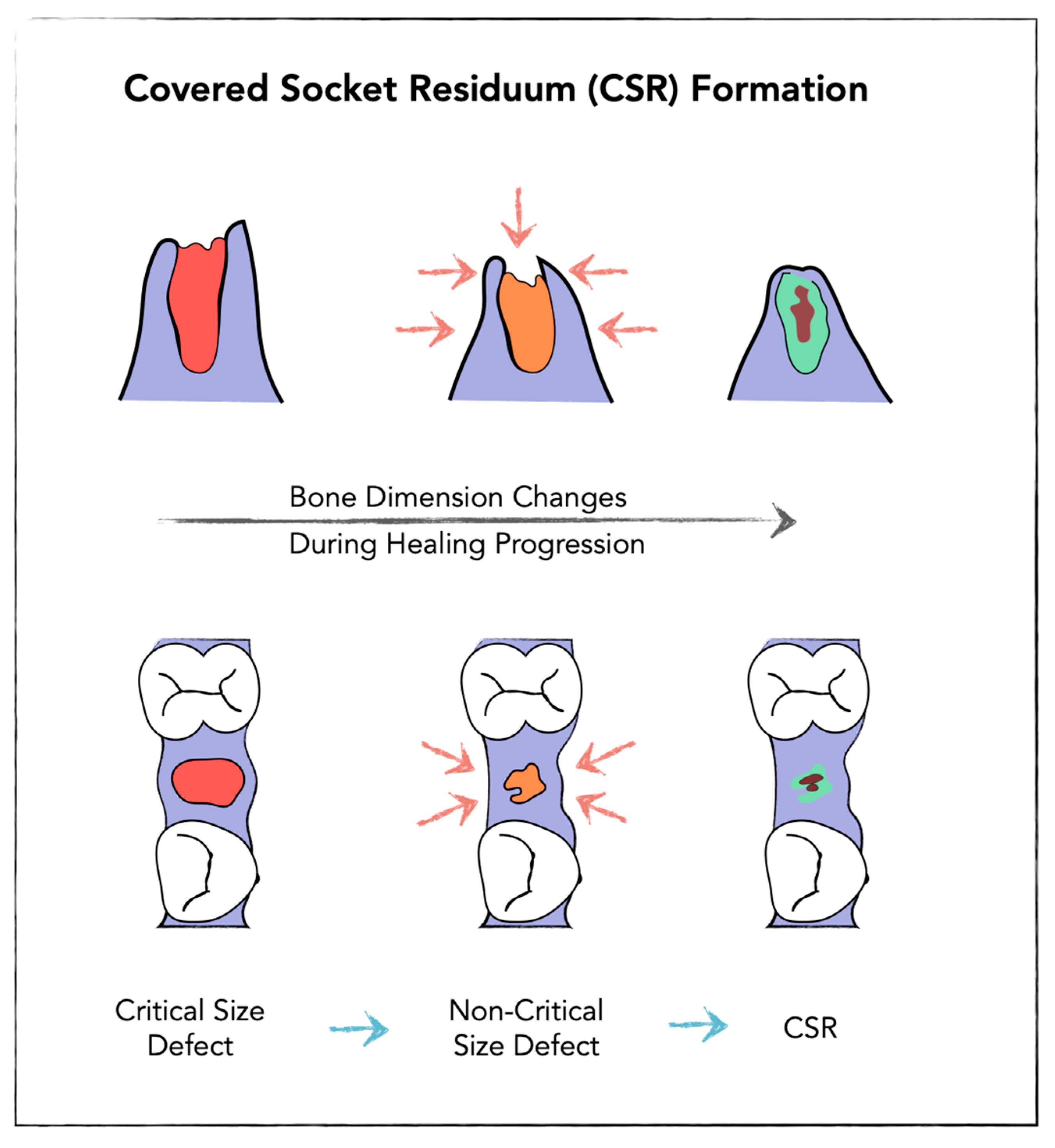
3.2. Conceptual Introduction of Covered Socket Residuum (CSR)
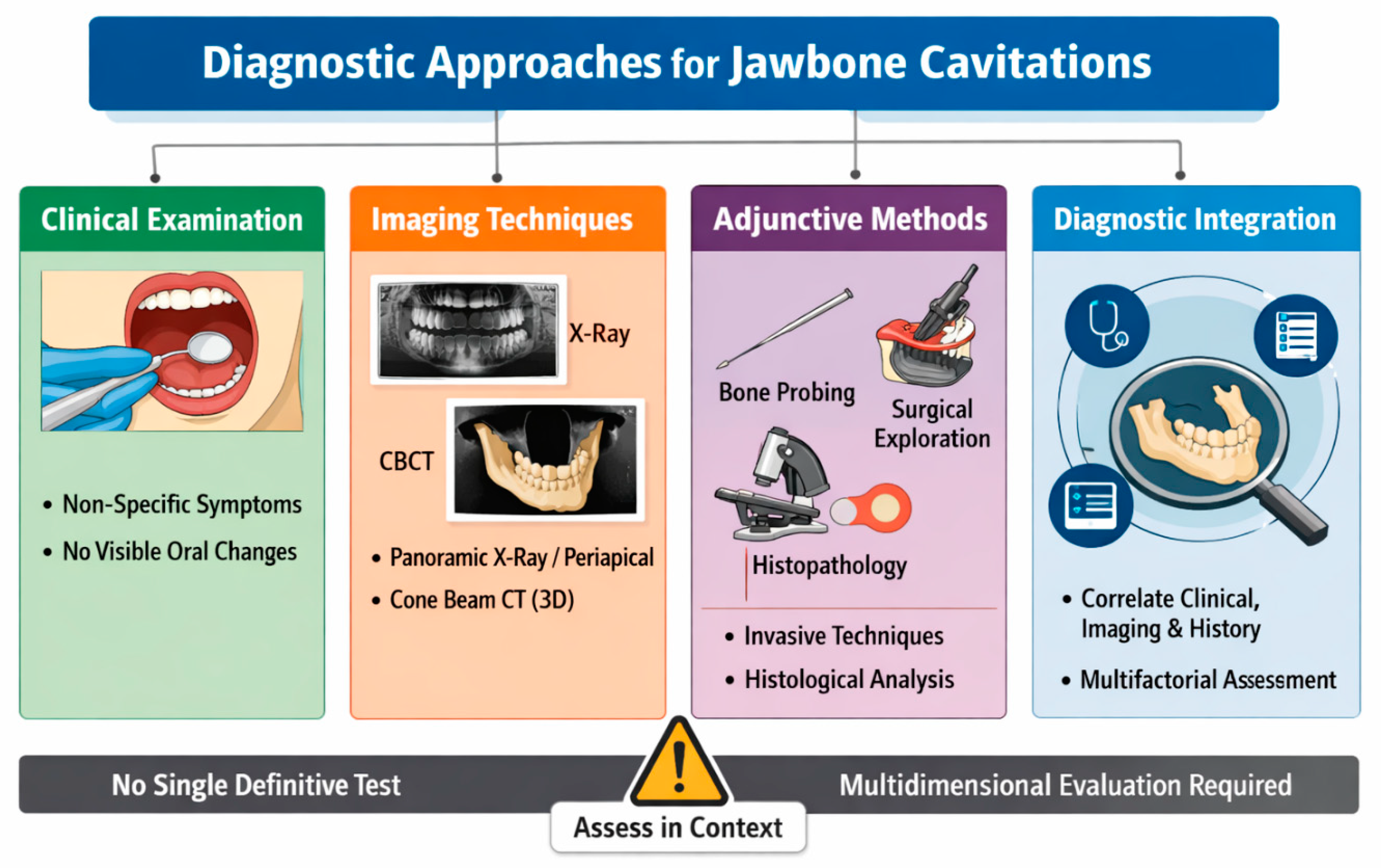
3.3. Diagnostic Approaches for CSR
3.3.1. Clinical Assessment
3.3.2. Radiological Evaluation
- Presence of non-mineralized or low-density areas within the former tooth socket area
- Non-mineralized area surrounded by mineralized bony layer in the crestal part of the jaw (figure)
- Absence of radiological criteria for other established diseases (e.g., jaw cyst and odontogeneic tumors)
- Absence of malignant criteria
3.3.3. Histopathological Analysis
3.4. Therapy
4. Discussion
4.1. Learning from Socket Healing
4.2. Clinical Implications
4.3. Prevention and Potential Therapeutic Concepts
5. Conclusions
6. Outlook
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bouquot, J.E.; Roberts, A.M.; Person, P.; Christian, J. Neuralgia-inducing cavitational osteonecrosis (NICO): Osteomyelitis in 224 jawbone samples from patients with facial neuralgia. Oral Surg. Oral Med. Oral Pathol. 1992, 73, 307–319. [Google Scholar] [CrossRef]
- Roberts, A.M.; Person, P. Etiology and treatment of idiopathic trigeminal and atypical facial neuralgias. Oral Surg. Oral Med. Oral Pathol. 1979, 48, 298–308. [Google Scholar] [CrossRef]
- Roberts, A.M.; Person, P.; Chandran, N.B.; Hori, J.M. Further observations on dental parameters of trigeminal and atypical facial neuralgias. Oral Surg. Oral Med. Oral Pathol. 1984, 58, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, Y.; Pal, U.; Singh, N. Neuralgia-inducing cavitational osteonecrosis in a patient seeking dental implants. Natl. J. Maxillofac. Surg. 2012, 3, 84. [Google Scholar] [CrossRef]
- Gandhi, Y. Neuralgia-inducing cavitational osteonecrosis—Fact or myth, the debate persists. Natl. J. Maxillofac. Surg. 2019, 10, 228–231. [Google Scholar] [CrossRef]
- Hassoun, M.R.; Cherayil, N.R. A 72-Year-Old Man with a History of Rheumatoid Arthritis Presenting with Unilateral Eye and Jaw Pain. Ann. Clin. Transl. Neurol. 2025, 12, 1942–1943. [Google Scholar] [CrossRef]
- Al-Quliti, K.W. Update on neuropathic pain treatment for trigeminal neuralgia. The pharmacological and surgical options. Neurosciences 2015, 20, 107–114. [Google Scholar] [CrossRef]
- Lechner, J.; von Baehr, V. Chemokine RANTES/CCL5 as an unknown link between wound healing in the jawbone and systemic disease: Is prediction and tailored treatments in the horizon? EPMA J. 2015, 6, 10. [Google Scholar] [CrossRef]
- Feinberg, L.S.; Stephan, R.B.; Fogarty, K.P.; Voortman, L.; Tiller, W.A.; Cassiani-Ingoni, R. Resolution of cavitational osteonecrosis through NeuroModulation Technique, a novel form of intention-based therapy: A clinical case study. J. Altern. Complement. Med. 2009, 15, 25–33. [Google Scholar] [CrossRef]
- Ghanaati, S.; Śmieszek-Wilczewska, J.; Al-Maawi, S.; Heselich, A.; Sader, R.; Extraction, A. Upper Premolars Undergo Programmed Socket Collapse with Development of Cavitations Rather than Complete Socket Healing: A Radiological Study. Bioengineering 2025, 12, 128. [Google Scholar] [CrossRef] [PubMed]
- Lechner, J.; Mayer, W. Immune messengers in Neuralgia Inducing Cavitational Osteonecrosis (NICO) in jaw bone and systemic interference. Eur. J. Integr. Med. 2010, 2, 71–77. [Google Scholar] [CrossRef]
- Freedman, P.D.; Reich, R.F.; Steinlauf, A.F. Neuralgia-inducing cavitational osteonecrosis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1998, 86, 3–5. [Google Scholar] [CrossRef]
- Zuniga, J.R. Challenging the neuralgia-inducing cavitational osteonecrosis concept. J. Oral Maxillofac. Surg. 2000, 58, 1021–1028. [Google Scholar] [CrossRef]
- Ratner, E.J.; Person, P.; Kleinman, D.J.; Shklar, G.; Socransky, S.S. Jawbone cavities and trigeminal and atypical facial neuralgias. Oral Surg. Oral Med. Oral Pathol. 1979, 48, 3–20. [Google Scholar] [CrossRef]
- Sekundo, C.; Wiltfang, J.; Schliephake, H.; Al-Nawas, B.; Rückschloß, T.; Moratin, J.; Hoffmann, J.; Ristow, O. Neuralgia-inducing cavitational osteonecrosis—A systematic review. Oral Dis. 2022, 28, 1448–1467. [Google Scholar] [CrossRef] [PubMed]
- Cruz, J.V.E.; Notter, F.; Schick, F.; Lechner, J. Comparison of Cytokine RANTES/CCL5 Inflammation in Apical Periodontitis and in Jawbone Cavitations—Retrospective Clinical Study. J. Inflamm. Res. 2024, 17, 67–80. [Google Scholar] [CrossRef]
- Klasser, G.D.; Epstein, J.B. Neuralgia-inducing cavitational osteonecrosis: A possible diagnosis for an orofacial pain complaint? J. Am. Dent. Assoc. 2011, 142, 651–653. [Google Scholar] [CrossRef]
- Lechner, J.; Rudi, T.; von Baehr, V. Osteoimmunology of tumor necrosis factor-alpha, IL-6, and RANTES/CCL5: A review of known and poorly understood inflammatory patterns in osteonecrosis. Clin. Cosmet. Investig. Dent. 2018, 10, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Ghanaati, S.; Dohle, E.; Schick, F.; Lechner, J. Quantitative Real-Time RT-PCR Verifying Gene Expression Profile of Cavitations Within Human Jaw Bone. Biomedicines 2025, 13, 1144. [Google Scholar] [CrossRef]
- Gruppo, R.; Glueck, C.J.; Mcmahon, R.E.; Bouquot, J.; Rabinovich, B.A.; Becker, A.; Tracy, T.; Wang, P. The pathophysiology of alveolar osteonecrosis of the jaw: Anticardiolipin antibodies, thrombophilia, and hypofibrinolysis. J. Lab. Clin. Med. 1996, 127, 481–488. [Google Scholar] [CrossRef]
- Bouquot, J.E.; LaMarche, M.G. Ischemic osteonecrosis under fixed partial denture pontics: Radiographicand microscopic features in 38 patients with chronic pain. J. Prosthet. Dent. 1999, 81, 148–158. [Google Scholar] [CrossRef]
- Ghanaati, S.; Kamalov, A.; Bouquot, J.; Sader, R.; Heselich, A.; Al-Maawi, S. Covered Socket Residuum (CSR) in Former Third Molar Sockets Despite Platelet-Rich Fibrin: A Prospective Randomized Controlled Split-Mouth Clinical Study. Bioengineering 2025, 12, 1242. [Google Scholar] [CrossRef] [PubMed]
- Bouquot, J.E. More about neuralgia-inducing cavitational osteonecrosis (NICO). Oral Surg. Oral Med. Oral Pathol. 1992, 74, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Lechner, J. Validation of dental X-ray by cytokine RANTES—Comparison of X-ray findings with cytokine overexpression in jawbone. Clin. Cosmet. Investig. Dent. 2014, 6, 71–79. [Google Scholar] [CrossRef]
- Araújo, M.G.; Silva, C.O.; Misawa, M.; Sukekava, F. Alveolar socket healing: What can we learn? Periodontology 2000 2015, 68, 122–134. [Google Scholar] [CrossRef]
- Juodzbalys, G.; Stumbras, A.; Goyushov, S.; Duruel, O.; Tözüm, T.F. Morphological Classification of Extraction Sockets and Clinical Decision Tree for Socket Preservation/Augmentation after Tooth Extraction: A Systematic Review. J. Oral Maxillofac. Res. 2019, 10, e3. [Google Scholar] [CrossRef]
- Pietrokovski, J.; Massler, M. Ridge Remodeling after Tooth Extraction in Rats. J. Dent. Res. 1967, 46, 222–231. [Google Scholar] [CrossRef]
- Kim, J.H.; Koo, K.T.; Capetillo, J.; Kim, J.J.; Yoo, J.M.; Amara, H.B.; Park, J.C.; Schwarz, F.; Wikesjö, U.M.E. Periodontal and endodontic pathology delays extraction socket healing in a canine model. J. Periodontal Implant. Sci. 2017, 47, 143–153. [Google Scholar] [CrossRef]
- Srinivas, B.; Das, P.; Rana, M.M.; Qureshi, A.Q.; Vaidya, K.C.; Raziuddin, S.J.A. Wound healing and bone regeneration in postextraction sockets with and without platelet-rich fibrin. Ann. Maxillofac. Surg. 2018, 8, 28–34. [Google Scholar] [CrossRef]
- MacBeth, N.; Trullenque-Eriksson, A.; Donos, N.; Mardas, N. Hard and soft tissue changes following alveolar ridge preservation: A systematic review. Clin. Oral Implant. Res. 2017, 28, 982–1004. [Google Scholar] [CrossRef]
- Peck, M.T.; Marnewick, J.; Stephen, L. Alveolar ridge preservation using leukocyte and platelet-rich fibrin: A report of a case. Case Rep. Dent. 2011, 2011, 345048. [Google Scholar] [CrossRef]
- Rothamel, D.; Schwarz, F.; Herten, M.; Engelhardt, E.; Donath, K.; Kuehn, P.; Becker, J. Dimensional ridge alterations following socket preservation using a nanocrystalline hydroxyapatite paste. A histomorpho-metrical study in dogs. Int. J. Oral Maxillofac. Surg. 2008, 37, 741–747. [Google Scholar] [CrossRef]
- Blanco, J.; Mareque, S.; Liñares, A.; Muñoz, F. Vertical and horizontal ridge alterations after tooth extraction in the dog: Flap vs. flapless surgery. Clin. Oral. Implant. Res. 2011, 22, 1255–1258. [Google Scholar] [CrossRef]
- Araujo, M.G.; Lindhe, J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J. Clin. Periodontol. 2005, 32, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Ono, T.; Takayanagi, H. Osteoimmunology in Bone Fracture Healing. Curr. Osteoporos. Rep. 2017, 15, 367–375. [Google Scholar] [CrossRef]
- Dahlin, C.; Linde, A.; Gottlow, J.; Nyman, S. Healing of bone defects by guided tissue regeneration. Plast. Reconstr. Surg. 1988, 81, 672–676. [Google Scholar] [CrossRef]
- Tan, W.L.; Wong, T.L.T.; Wong, M.C.M.; Lang, N.P. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin. Oral Implant. Res. 2012, 23, 1–21. [Google Scholar] [CrossRef]
- Fickl, S.; Zuhr, O.; Wachtel, H.; Bolz, W.; Huerzeler, M. Tissue alterations after tooth extraction with and without surgical trauma: A volumetric study in the beagle dog. J. Clin. Periodontol. 2008, 35, 356–363. [Google Scholar] [CrossRef]
- Tan, Z.; Kang, J.; Liu, W.; Wang, H. The effect of the heights and thicknesses of the remaining root segments on buccal bone resorption in the socket-shield tech-nique: An experimental study in dogs. Clin. Implant. Dent. Relat. Res. 2018, 20, 352–359. [Google Scholar] [CrossRef]
- Rossetti, P.H.O.; Bonachela, W.C.; Rossetti, L.M.N. Relevant anatomic and biomechanical studies for implant possibilities on the atrophic maxilla: Critical appraisal and literature review. J. Prosthodont. 2010, 19, 449–457. [Google Scholar] [CrossRef]
- Kalsi, A.S.; Kalsi, J.S.; Bassi, S. Alveolar ridge preservation: Why, when and how. Br. Dent. J. 2019, 227, 264–274. [Google Scholar] [CrossRef]
- Kondo, T.; Kanayama, K.; Egusa, H.; Nishimura, I. Current perspectives of residual ridge resorption: Pathological activation of oral barrier osteoclasts. J. Prosthodont. Res. 2023, 67, 12–22. [Google Scholar] [CrossRef]
- Anwandter, A.; Bohmann, S.; Nally, M.; Castro, A.B.; Quirynen, M.; Pinto, N. Dimensional changes of the post extraction alveolar ridge, preserved with Leukocyte- and Platelet Rich Fibrin: A clinical pilot study. J. Dent. 2016, 52, 23–29. [Google Scholar] [CrossRef]
- Shelley, A.M.; Ferrero, A.; Brunton, P.; Goodwin, M.; Horner, K. The impact of CBCT imaging when placing dental implants in the anterior edentulous mandible: A before-after study. Dentomaxillofac. Radiol. 2015, 44, 20140316. [Google Scholar] [CrossRef]
- Ibrahim, R.K.; Louis, B.B.; Ritter, M.; Varsani, R.; Nammour, S.; Arany, P.R. Feasibility of atraumatic extractions with surgical lasers: A pilot study in porcine jaws. J. Dent. 2025, 163, 106107. [Google Scholar] [CrossRef] [PubMed]
- Ghanaati, S.; Śmieszek-Wilczewska, J.; Al-Maawi, S.; Neff, P.; Zadeh, H.H.; Sader, R.; Heselich, A.; Rutkowski, J.L. Solid PRF Serves as Basis for Guided Open Wound Healing of the Ridge After Tooth Extraction by Accelerating the Wound Healing Time Course-A Prospective Parallel Arm Randomized Controlled Single Blind Trial. Bioengineering 2022, 9, 661. [Google Scholar] [CrossRef]
- Ghanaati, S.; Al-Maawi, S.; Conrad, T.; Lorenz, J.; Rössler, R.; Sader, R. Biomaterial-based bone regeneration and soft tissue management of the individualized 3D-titanium mesh: An alternative concept to autologous transplantation and flap mobilization. J. Cranio-Maxillofac. Surg. 2019, 47, 1633–1644. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ghanaati, S.; Heselich, A.; Lechner, J.; Sader, R.; Bouquot, J.E.; Al-Maawi, S. Jawbone Cavitations: Current Understanding and Conceptual Introduction of Covered Socket Residuum (CSR). Bioengineering 2026, 13, 106. https://doi.org/10.3390/bioengineering13010106
Ghanaati S, Heselich A, Lechner J, Sader R, Bouquot JE, Al-Maawi S. Jawbone Cavitations: Current Understanding and Conceptual Introduction of Covered Socket Residuum (CSR). Bioengineering. 2026; 13(1):106. https://doi.org/10.3390/bioengineering13010106
Chicago/Turabian StyleGhanaati, Shahram, Anja Heselich, Johann Lechner, Robert Sader, Jerry E. Bouquot, and Sarah Al-Maawi. 2026. "Jawbone Cavitations: Current Understanding and Conceptual Introduction of Covered Socket Residuum (CSR)" Bioengineering 13, no. 1: 106. https://doi.org/10.3390/bioengineering13010106
APA StyleGhanaati, S., Heselich, A., Lechner, J., Sader, R., Bouquot, J. E., & Al-Maawi, S. (2026). Jawbone Cavitations: Current Understanding and Conceptual Introduction of Covered Socket Residuum (CSR). Bioengineering, 13(1), 106. https://doi.org/10.3390/bioengineering13010106







