Remote Symptom Alerts and Patient-Reported Outcomes (PROS) in Real-World Breast Cancer Practice: Innovative Data to Derive Symptom Burden and Quality of Life
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population and PROmpt®
2.2. Symptom Prevalence and Alerts
2.3. Trajectory of Neuropathy
2.4. Statistical Analysis
3. Results
3.1. Demographic and Baseline Characteristics
3.2. Symptoms, QoL, and Alerts
3.3. Clinical Actions Documented in the Alerts
3.4. Trajectory of Neuropathy
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hamer, J.; McDonald, R.; Zhang, L.; Verma, S.; Leahey, A.; Ecclestone, C.; Bedard, G.; Pulenzas, N.; Bhatia, A.; Chow, R.; et al. Quality of life (QOL) and symptom burden (SB) in patients with breast cancer. Support. Care Cancer 2017, 25, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Deal, A.M.; Kris, M.G.; Scher, H.I.; Hudis, C.A.; Sabbatini, P.; Rogak, L.; Bennett, A.V.; Dueck, A.C.; Atkinson, T.M.; et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: A randomized controlled trial. J. Clin. Oncol. 2016, 34, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Deal, A.M.; Dueck, A.C.; Scher, H.I.; Kris, M.G.; Hudis, C.; Schrag, D. Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA 2017, 318, 197–198. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Stover, A.M.; Schrag, D.; Chung, A.; Jansen, J.; Henson, S.; Carr, P.; Ginos, B.; Deal, A.; Spears, P.A.; et al. Clinical utility and user perceptions of a digital system for electronic patient-reported symptom monitoring during routine cancer care: Findings from the PRO-TECT trial. JCO Clin. Cancer Inform. 2020, 4, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Jauhari, Y.; Gannon, M.R.; Dodwell, D.; Horgan, K.; Tsang, C.; Clements, K.; Medina, J.; Tang, S.; Pettengell, R.; Cromwell, D.A. Addressing frailty in patients with breast cancer: A review of the literature. Eur. J. Surg. Oncol. 2020, 46, 24–32. [Google Scholar] [CrossRef]
- Warrington, L.; Absolom, K.; Conner, M.; Kellar, I.; Clayton, B.; Ayres, M.; Velikova, G. Electronic systems for patients to report and manage side effects of cancer treatment: Systematic review. J. Med. Internet Res. 2019, 21, e10875. [Google Scholar] [CrossRef]
- Kamgar, M.; Greenwald, R.K.; Asad, H.; Hastert, T.A.; McLaughlin, E.M.; Reding, K.W.; Paskett, E.D.; Bea, J.W.; Shadyab, A.H.; Neuhouser, M.L.; et al. Prevalence and predictors of peripheral neuropathy after breast cancer treatment. Cancer Med. 2021, 10, 6666–6676. [Google Scholar] [CrossRef]
- Bandos, H.; Melnikow, J.; Rivera, D.R.; Swain, S.M.; Sturtz, K.; Fehrenbacher, L.; Wade, J.L.; Brufsky, A.M.; Julian, T.B.; Margolese, R.G.; et al. Long-term peripheral neuropathy in breast cancer patients treated with adjuvant chemotherapy: NRG Oncology/NSABP B-30. J. Natl. Cancer Inst. 2017, 110, djx162. [Google Scholar] [CrossRef]
- Zhi, W.I.; Baser, R.E.; Kwon, A.; Chen, C.; Li, S.Q.; Piulson, L.; Seluzicki, C.; Panageas, K.S.; Harte, S.E.; Mao, J.J.; et al. Characterization of chemotherapy-induced peripheral neuropathy using patient reported outcomes and quantitative sensory testing. Breast Cancer Res. Treat. 2021, 186, 761–768. [Google Scholar] [CrossRef]
- National Cancer Institute. Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE®). 2023. Available online: https://healthcaredelivery.cancer.gov/pro-ctcae/ (accessed on 27 June 2024).
- Fayers, P.; Bottomley, A. EORTC Quality of Life Group; Quality of Life Unit; European Organisation for Research and Treatment of Cancer. Quality of life research within the EORTC—The EORTC QLQ-C30. Eur. J. Cancer 2002, 38 (Suppl. S4), S125–S133. [Google Scholar] [CrossRef]
- Jensen, R.E.; Potosky, A.L.; Reeve, B.B.; Hahn, E.; Cella, D.; Fries, J.; Smith, A.W.; Keegan, T.H.; Wu, X.C.; Paddock, L.; et al. Validation of the PROMIS physical function measures in a diverse US population-based cohort of cancer patients. Qual. Life Res. 2015, 24, 2333–2344. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pearman, T.P.; Beaumont, J.L.; Mroczek, D.; O’Connor, M.; Cella, D. Validity and usefulness of a single-item measure of patient-reported bother from side effects of cancer therapy. Cancer 2018, 124, 991–997. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Basch, E.; Becker, C.; Rogak, L.J.; Schrag, D.; Reeve, B.B.; Spears, P.; Smith, M.L.; Gounder, M.M.; Mahoney, M.R.; Schwartz, G.K.; et al. Composite grading algorithm for the National Cancer Institute’s Patient Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Clin. Trials 2021, 18, 104–114. [Google Scholar] [CrossRef]
- Palumbo, A.; Bringhen, S.; Mateos, M.V.; Larocca, A.; Facon, T.; Kumar, S.K.; Offidani, M.; McCarthy, P.; Evangelista, A.; Lonial, S.; et al. Geriatric assessment predicts survival and toxicities in elderly myeloma patients: An International Myeloma Working Group report. Blood 2015, 125, 2068–2074. [Google Scholar] [CrossRef]
- Katz, J.N.; Chang, L.C.; Sangha, O.; Fossel, A.H.; Bates, D.W. Can comorbidity be measured by questionnaire rather than medical record review? Med. Care 1996, 34, 73–84. [Google Scholar] [CrossRef]
- Hurria, A.; Gupta, S.; Zauderer, M.; Zuckerman, E.L.; Cohen, H.J.; Muss, H.; Rodin, M.; Panageas, K.S.; Holland, J.C.; Saltz, L.; et al. Developing a cancer-specific geriatric assessment. Cancer 2005, 104, 1998–2005. [Google Scholar] [CrossRef]
- Rockwood, K.; Mitnitski, A. Frailty in relation to the accumulation of deficits. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Searle, S.D.; Mitnitski, A.; Gahbauer, E.A.; Gill, T.M.; Rockwood, K. A standard procedure for creating a frailty index. BMC Geriatr. 2008, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the eastern cooperative oncology group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.; Watanabe, S.; Fainsinger, R.; Lau, F.; Ghosh, S.; Quan, H.; Atkins, M.; Fassbender, K.; Downing, G.M.; Baracos, V. Prognostic factors in patients with advanced cancer: Use of the patient-generated subjective global assessment in survival prediction. J. Clin. Oncol. 2010, 28, 4376–4383. [Google Scholar] [CrossRef]
- Basch, E.; Hudson, K.; Rocque, G. Implementation of electronic patient-reported outcomes for symptom monitoring during cancer treatment: The importance of getting it right. J. Comp. Eff. Res. 2023, 12, e230157. [Google Scholar] [CrossRef]
- Alhazmi, L.S.S.; Bawadood, M.A.A.; Aljohani, A.M.S.; Alzahrani, A.A.R.; Moshref, L.; Trabulsi, N.; Moshref, R. Pain management in breast cancer patients: A multidisciplinary approach. Cureus 2021, 13, e15994. [Google Scholar] [CrossRef] [PubMed]
- So, W.K.W.; Law, B.M.H.; Ng, M.S.N.; He, X.; Chan, D.N.S.; Chan, C.W.H.; McCarthy, A.L. Symptom clusters experienced by breast cancer patients at various treatment stages: A systematic review. Cancer Med. 2021, 10, 2531–2565. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Fontes, F.; Sonin, T.; Dias, T.; Fragoso, M.; Castro-Lopes, J.; Lunet, N. Neuropathic pain after breast cancer treatment: Characterization and risk factors. J. Pain Sym. Manag. 2017, 54, 877–888. [Google Scholar] [CrossRef] [PubMed]
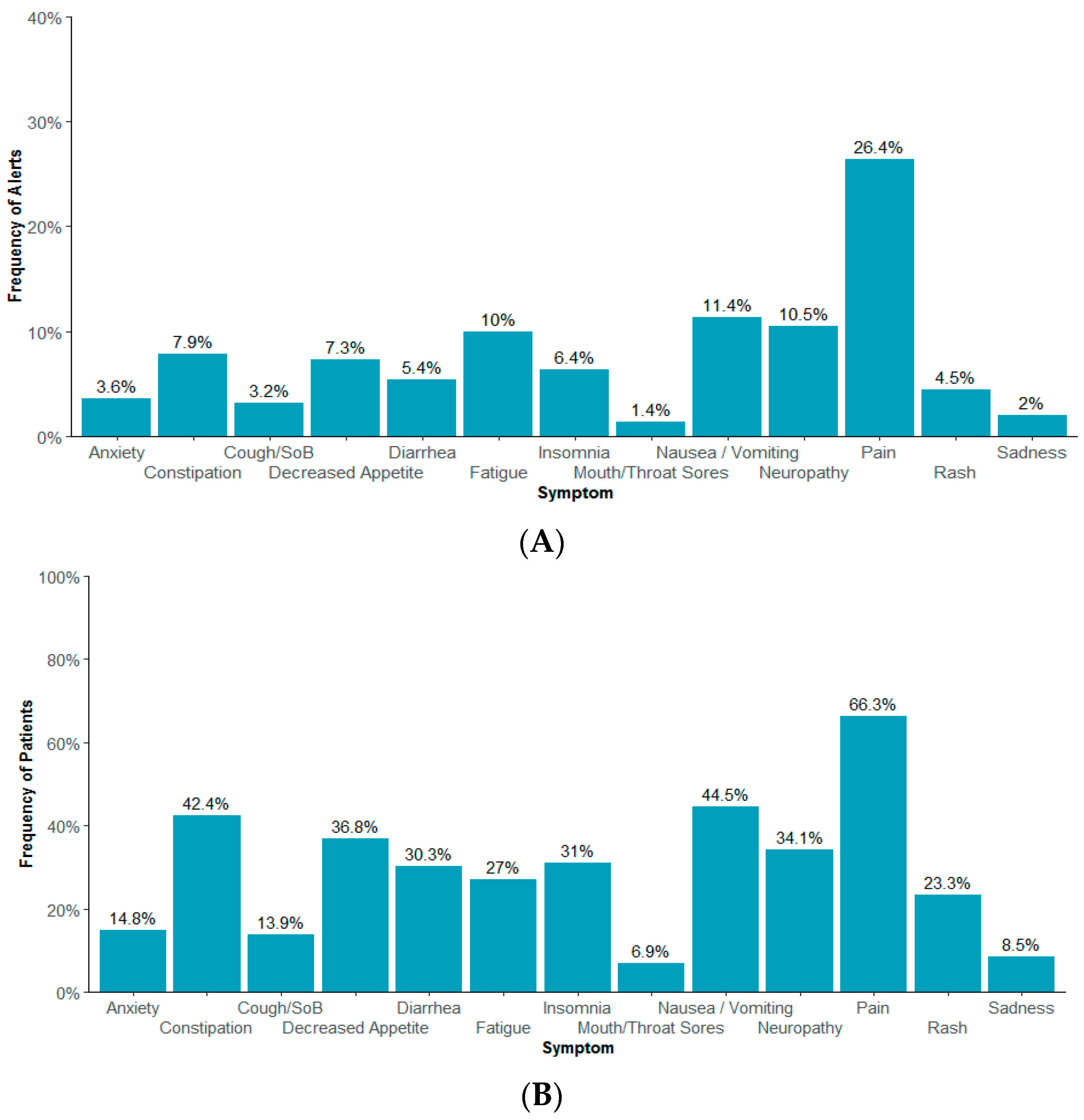



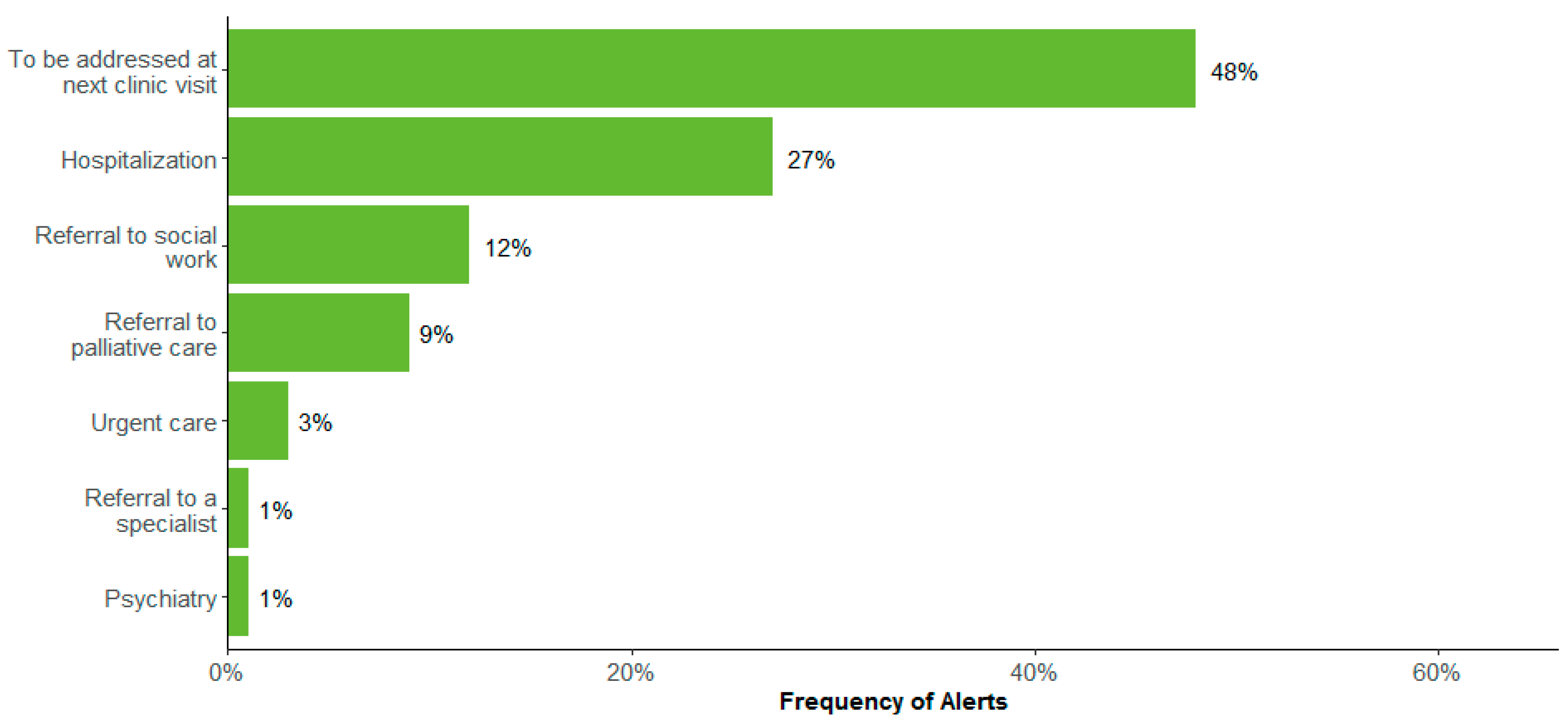
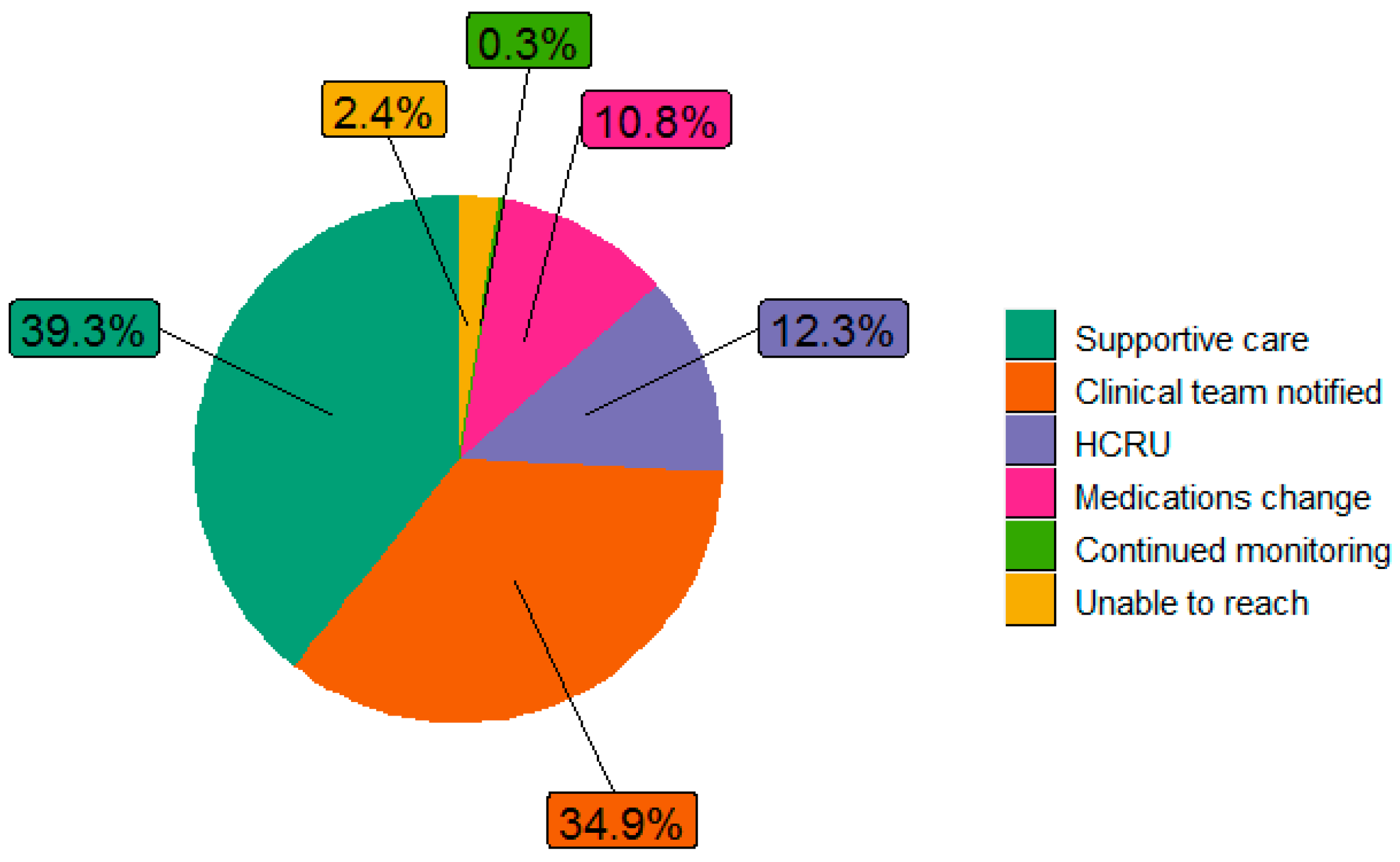


| All Patients (n = 646) | Reported at Least 1 Moderate or Severe Symptom (n = 519) | |
|---|---|---|
| No. of patients with at least one symptom alert, n (%) | 519 (80.3) | 519 (100) |
| No. of symptoms reported during study period | 19,425 | 18,506 |
| Total alerts generated during study period | 7641 | 7641 |
| PROs follow-up time (weeks) | ||
| Mean (SD) | 19.6 (21.7) | 22.3 (22.8) |
| Median (Range) | 12.3 (0–152) | 16.1 (0–152) |
| Age at enrollment (years) | ||
| Mean (SD) | 55.6 (12.6) | 54.8 (12.7) |
| Median (Range) | 56 (26–84) | 55 (26–81) |
| Age at enrollment (years), n (%) | ||
| <50 years old | 200 (31.0) | 174 (33.5) |
| 50–64 years old | 270 (41.8) | 211 (40.7) |
| 65–75 years old | 154 (23.8) | 118 (22.7) |
| >75 years old | 22 (3.4) | 16 (3.1) |
| Female, n (%) | 646 (100) | 519 (100) |
| Race, n (%) | ||
| American Indian or Alaskan Native | 7 (1.1) | 5 (1.0) |
| Asian | 11 (1.7) | 10 (1.9) |
| Black or African American | 132 (20.4) | 107 (20.6) |
| Native Hawaiian or Other Pacific Islander | 1 (0.2) | 1 (0.2) |
| White | 466 (72.1) | 370 (71.3) |
| Other | 9 (1.4) | 7 (1.3) |
| Unspecified | 20 (3.1) | 19 (3.7) |
| Biomarker status, n (%) | ||
| HR+/HER2− | 305 (47.2) | 247 (47.6) |
| HR+/HER2+ | 100 (15.5) | 85 (16.4) |
| Triple negative | 119 (18.4) | 90 (17.3) |
| Unspecified | 122 (18.9) | 97 (18.7) |
| Stage, n (%) | ||
| Early stage (0–IIIA) | 399 (61.8) | 325 (62.6) |
| Late stage (IIIB–IV) | 147 (22.8) | 114 (22.0) |
| Unspecified | 100 (15.4) | 80 (15.4) |
| Baseline frailty status, n (%) | ||
| Fit | 503 (77.9) | 393 (75.7) |
| Intermediate | 77 (11.9) | 69 (13.3) |
| Frail | 44 (6.8) | 41 (7.9) |
| Unspecified | 22 (3.4) | 16 (3.1) |
| ECOG status, n (%) | ||
| 0 | 144 (22.2) | 120 (23.1) |
| 1 | 134 (20.7) | 108 (20.8) |
| 2+ | 61 (9.4) | 58 (11.2) |
| Unspecified | 307 (47.7) | 233 (44.9) |
| Treatment closest to first symptom alert, n (%) | ||
| Chemotherapy | 164 (25.4) | 164 (31.6) |
| Anti-HER2 therapy | 101 (15.6) | 101 (19.5) |
| Mono Endocrine therapy (ET) | 91 (14.1) | 91 (17.5) |
| PD-1/L1 inhibitors | 45 (7.0) | 45 (8.7) |
| CDK 4/6 inhibitors | 36 (5.6) | 36 (6.9) |
| Other | 33 (5.1) | 33 (6.4) |
| Did not generate alerts | 127 (19.7) | 0 (0.0) |
| Unspecified | 49 (7.6) | 49 (9.4) |
| (a) | ||||||||||||||||
| All | Early Stage (0–IIIA) | Late Stage (IIIB–IV) | ||||||||||||||
| No. of patients with at least one symptom alert | 519 | 325 | 114 | |||||||||||||
| Total alerts generated during study period | 7641 | 4439 | 2088 | |||||||||||||
| No. of alerts per patient per week | ||||||||||||||||
| Mean (SD) | 2.0 (1.5) | 2.1 (1.5) | 2.1 (1.6) | |||||||||||||
| Median (Range) | 1.0 (1–16) | 1.0 (1–13) | 1.0 (1–16) | |||||||||||||
| (b) | ||||||||||||||||
| All | HER2−/HR+ | HER2+/HR+ | TNBC | |||||||||||||
| No. of patients with at least one symptom alert | 519 | 247 | 85 | 90 | ||||||||||||
| Total alerts generated during study period | 7641 | 3671 | 1245 | 1403 | ||||||||||||
| No. of alerts per patient per week | ||||||||||||||||
| Mean (SD) | 2.0 (1.5) | 2.0 (1.6) | 2.0 (1.5) | 2.1 (1.7) | ||||||||||||
| Median (Range) | 1.0 (1–16) | 1.0 (1–13) | 1.0 (1–12) | 2.0 (1–16) | ||||||||||||
| (c) | ||||||||||||||||
| All | <50 | 50–64 | 65–75 | >75 | ||||||||||||
| No. of patients with at least one symptom alert | 519 | 174 | 211 | 118 | 16 | |||||||||||
| Total alerts generated during study period | 7641 | 2547 | 3398 | 1520 | 176 | |||||||||||
| No. of alerts per patient per week | ||||||||||||||||
| Mean (SD) | 2.0 (1.5) | 2.0 (1.6) | 2.1 (1.5) | 1.9 (1.4) | 1.7 (1.0) | |||||||||||
| Median (Range) | 1.0 (1–16) | 1.0 (1–13) | 2.0 (1–16) | 1.0 (1–11) | 1.0 (1–5) | |||||||||||
| (d) | ||||||||||||||||
| All | Fit | Intermediate | Frail | ECOG 0 | ECOG 1 | ECOG 2+ | ||||||||||
| No. of patients with at least one symptom alert | 519 | 393 | 69 | 41 | 120 | 108 | 58 | |||||||||
| Total alerts generated during study period | 7641 | 5319 | 1400 | 621 | 1235 | 1579 | 1145 | |||||||||
| No. of alerts per patient per week | ||||||||||||||||
| Mean (SD) | 2.0 (1.5) | 2.0 (1.5) | 2.1 (1.4) | 2.5 (1.5) | 1.5 (0.9) | 1.9 (1.1) | 2.1 (1.3) | |||||||||
| Median (Range) | 1.0 (1–16) | 1.0 (1–16) | 2.0 (1–11) | 2.0 (1–10) | 1.0 (1–7) | 2.0 (1–7) | 2.0 (1–9) | |||||||||
| All Patients (n = 177) | No PX PN (n = 103) | Had PX PN (n = 74) | |
|---|---|---|---|
| Total neuropathy alerts generated, n (%) | 801 (100) | 453 (56.6) | 348 (43.4) |
| Age at first neuropathy (years) | |||
| Mean (SD) | 55.8 (12.8) | 55.1 (13.3) | 56.7 (12.1) |
| Median (Range) | 57 (26–79) | 56 (28–78) | 58.5 (26–79) |
| Female, n (%) | 177 (100) | 103 (100) | 74 (100) |
| Race, n (%) | |||
| American Indian or Alaskan Native | 2 (1.1) | 0 (0.0) | 2 (2.7) |
| Asian | 4 (2.3) | 2 (1.9) | 2 (2.7) |
| Black or African American | 55 (31.1) | 31 (30.1) | 24 (32.4) |
| Native Hawaiian or Other Pacific Islander | 1 (0.6) | 1 (1.0) | 0 (0.0) |
| White | 103 (58.2) | 60 (58.3) | 43 (58.1) |
| Other | 5 (2.8) | 4 (3.9) | 1 (1.4) |
| Unspecified | 7 (4.0) | 5 (4.9) | 2 (2.7) |
| Biomarker status, n (%) | |||
| HR+/HER2− | 76 (42.9) | 46 (44.7) | 30 (40.5) |
| HR+/HER2+ | 31 (17.5) | 15 (14.6) | 16 (21.6) |
| Triple negative | 35 (19.8) | 21 (20.4) | 14 (18.9) |
| Unspecified | 35 (19.8) | 21 (20.4) | 14 (18.9) |
| Stage, n (%) | |||
| Early stage (0–IIIA) | 104 (58.8) | 65 (63.1) | 39 (52.7) |
| Late stage (IIIB–IV) | 45 (25.4) | 22 (21.4) | 23 (31.1) |
| Unspecified | 28 (15.8) | 16 (15.5) | 12 (16.2) |
| Prior use of CIPN drugs, n (%) | 94 (53.1) | 49 (47.6) | 45 (60.8) |
| 0–28 Days (n = 28) | >28 Days (n = 27) | |
|---|---|---|
| Age at first neuropathy, years | ||
| Mean (SD) | 52.3 (12.5) | 52.4 (14.4) |
| Median (Range) | 51 (31–78) | 49 (28–77) |
| Late stage (IIIB–IV), n (%) | 5 (17.9) | 4 (14.8) |
| Race, n (%) | ||
| American Indian or Alaskan Native | 0 (0.0) | 0 (0.0) |
| Asian | 0 (0.0) | 1 (3.7) |
| Black or African American | 9 (32.1) | 6 (22.2) |
| Native Hawaiian or Other Pacific Islander | 0 (0.0) | 0 (0.0) |
| White | 15 (53.6) | 18 (66.7) |
| Other | 3 (10.7) | 0 (0.0) |
| Unspecified | 1 (3.6) | 2 (7.4) |
| Time since diagnosis (months) | ||
| Mean (SD) | 11.6 (24.9) | 18.4 (29.2) |
| Median (Range) | 5.7 (1, 134) | 5.7 (2.6, 120) |
| Median duration of treatment (months) | ||
| Mean (SD) | 7.0 (18.6) | 18.9 (34.8) |
| Median (Range) | 2.4 (0–98.9) | 4.4 (1.3–128) |
| CIPN drugs use at alert, n (%) | 14 (50.0) | 16 (59.3) |
| No. of neuropathy alerts per patient/week | ||
| Mean (SD) | 1.0 (0.1) | 1.0 (0.2) |
| Median (Range) | 1.0 (1–2) | 1.0 (1–2) |
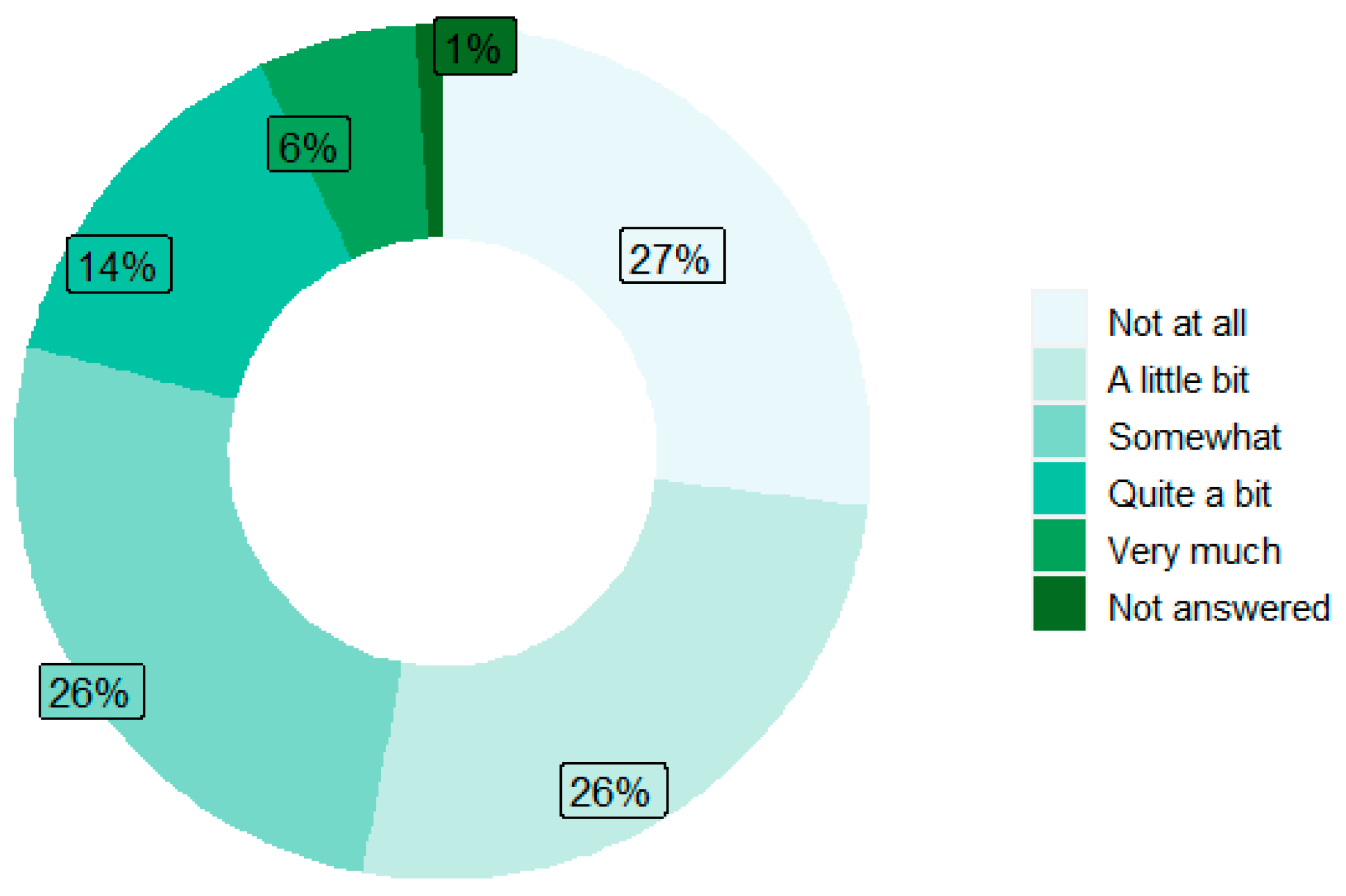
| Level of neuropathy interference at first alert, n (%) | ||
| Not at all | 1 (3.6) | 0 (0.0) |
| A little bit | 2 (7.1) | 2 (7.4) |
| Somewhat | 17 (60.7) | 14 (51.9) |
| Quite a bit | 7 (25.0) | 4 (14.8) |
| Very much | 0 (0.0) | 4 (14.8) |
| Not answered | 1 (3.6) | 3 (11.1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusli, E.; Wujcik, D.; Galaznik, A. Remote Symptom Alerts and Patient-Reported Outcomes (PROS) in Real-World Breast Cancer Practice: Innovative Data to Derive Symptom Burden and Quality of Life. Bioengineering 2024, 11, 846. https://doi.org/10.3390/bioengineering11080846
Rusli E, Wujcik D, Galaznik A. Remote Symptom Alerts and Patient-Reported Outcomes (PROS) in Real-World Breast Cancer Practice: Innovative Data to Derive Symptom Burden and Quality of Life. Bioengineering. 2024; 11(8):846. https://doi.org/10.3390/bioengineering11080846
Chicago/Turabian StyleRusli, Emelly, Debra Wujcik, and Aaron Galaznik. 2024. "Remote Symptom Alerts and Patient-Reported Outcomes (PROS) in Real-World Breast Cancer Practice: Innovative Data to Derive Symptom Burden and Quality of Life" Bioengineering 11, no. 8: 846. https://doi.org/10.3390/bioengineering11080846
APA StyleRusli, E., Wujcik, D., & Galaznik, A. (2024). Remote Symptom Alerts and Patient-Reported Outcomes (PROS) in Real-World Breast Cancer Practice: Innovative Data to Derive Symptom Burden and Quality of Life. Bioengineering, 11(8), 846. https://doi.org/10.3390/bioengineering11080846





