Premium Intraocular Lenses in Glaucoma—A Systematic Review
Abstract
1. Introduction
1.1. Overview of IOL Types
1.1.1. Monofocal IOLs
1.1.2. Multifocal IOLs (MFIOLs)
1.1.3. Extended Depth of Focus (EDOF)
1.1.4. Accommodative IOLs
1.1.5. Toric IOLs
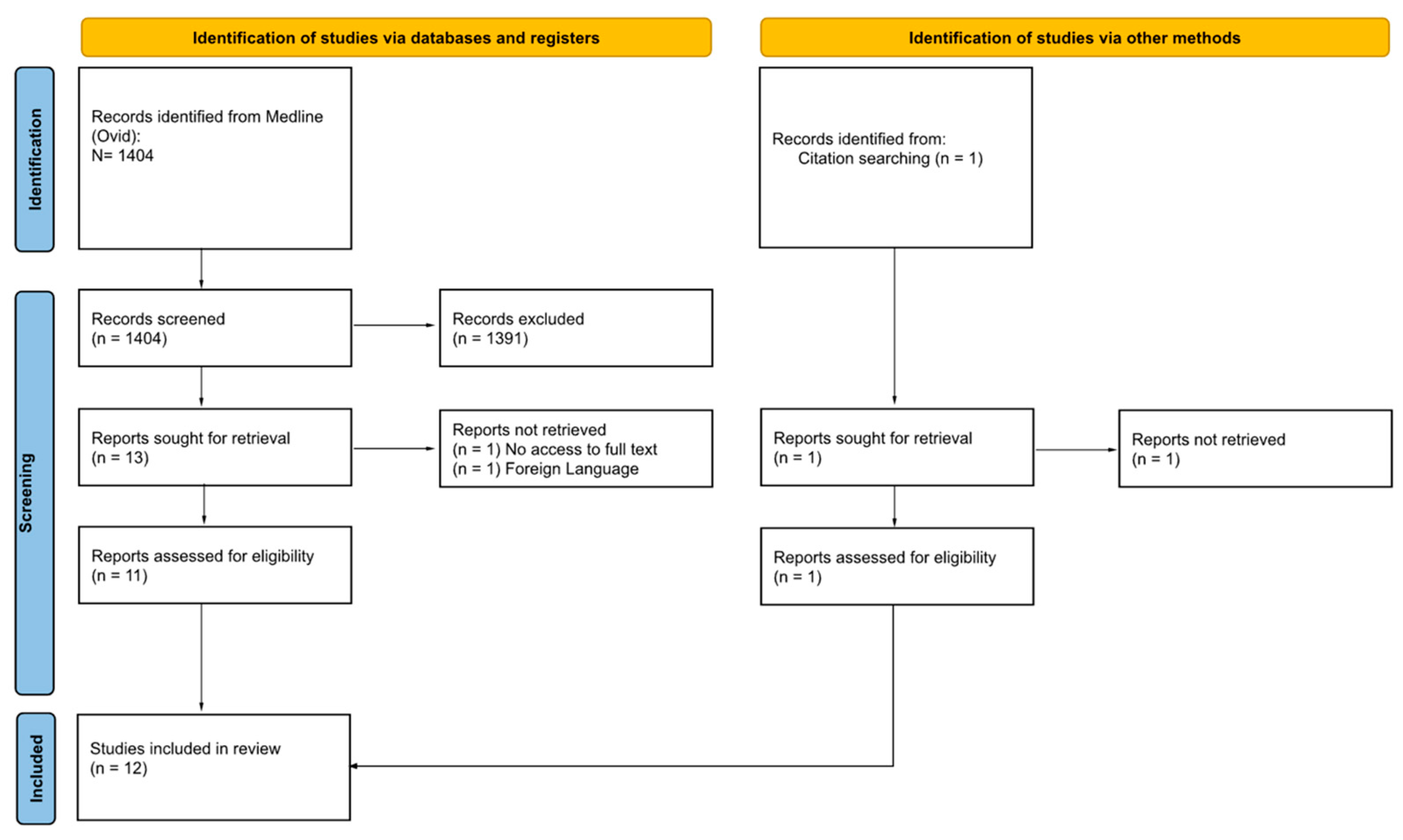
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Quality and Risk of Bias Assessment
3. Results
3.1. Methodology Quality and Risk of Bias
- Selection: (Question 1) Does/Do the patient(s) represent(s) the whole experience of the investigator (center), or is the selection method unclear to the extent that other patients with similar presentations may not have been reported?
- Ascertainment: (Question 2) Was the exposure adequately ascertained? (Question 3) Was the outcome adequately ascertained?
- Causality: (Question 4) Were other alternative causes that may explain the observation ruled out? (Question 5) Was there a challenge/rechallenge phenomenon? (Question 6) Was there a dose-response effect? (Question 7) Was follow-up long enough for outcomes to occur?
- Reporting: (Question 8) Is/Are the case(s) described in sufficient detail to allow other investigators to replicate the research or to allow practitioners to make inferences related to their own practice?
3.2. Patient Characteristics
3.3. Surgical Outcomes from Trials
3.3.1. Spectacle Independence
3.3.2. Contrast Sensitivity
3.3.3. Visual Acuity
3.3.4. Astigmatism
3.3.5. Patient Satisfaction
3.3.6. Glares and Haloes
4. Discussion: Considerations in Premium IOL Implantation in Glaucoma
4.1. Contrast Sensitivity
4.2. Glaucomatous Visual Field (VF) Defects
4.3. Glaucoma Subtype
4.4. Ocular Surface Disease (OSD)
4.5. Axial Length (AL) and Anterior Chamber Depth (ACD)
4.6. Type of Glaucoma Surgery
4.6.1. Combined Phacoemulsification and Trabeculectomy Surgery
4.6.2. MIGS (Minimally Invasive Glaucoma Surgery)
4.7. Functional and Structural Monitoring of Glaucoma
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Grzybowski, A. Recent developments in cataract surgery. Ann. Transl. Med. 2020, 8, 1540. [Google Scholar] [CrossRef]
- Zvorničanin, J.; Zvorničanin, E. Premium intraocular lenses: The past, present and future. J. Curr. Ophthalmol. 2018, 30, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Research, G.V. Global Intraocular Lens Market Outlook Report 2023–2028: Adoption & Penetration of Premium IOLs Gaining Momentum—ResearchAndMarkets.com. Available online: https://www.businesswire.com/news/home/20230421005192/en/Global-Intraocular-Lens-Market-Outlook-Report-2023-2028-Adoption-Penetration-of-Premium-IOLs-Gaining-Momentum—ResearchAndMarkets.com (accessed on 6 June 2023).
- Ling, J.D.; Bell, N.P. Role of Cataract Surgery in the Management of Glaucoma. Int. Ophthalmol. Clin. 2018, 58, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Leibowitz, H.M.; Krueger, D.E.; Maunder, L.R.; Milton, R.C.; Kini, M.M.; Kahn, H.A.; Nickerson, R.J.; Pool, J.; Colton, T.L.; Ganley, J.P.; et al. The Framingham Eye Study monograph: An ophthalmological and epidemiological study of cataract, glaucoma, diabetic retinopathy, macular degeneration, and visual acuity in a general population of 2631 adults, 1973–1975. Surv. Ophthalmol. 1980, 24, 335–610. [Google Scholar] [PubMed]
- Ichhpujani, P.; Bhartiya, S.; Sharma, A. Premium IOLs in Glaucoma. J. Curr. Glaucoma Pract. 2013, 7, 54–57. [Google Scholar] [CrossRef]
- Mencucci, R.; Cennamo, M.; Venturi, D.; Vignapiano, R.; Favuzza, E. Visual outcome, optical quality, and patient satisfaction with a new monofocal IOL, enhanced for intermediate vision: Preliminary results. J. Cataract Refract. Surg. 2020, 46, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Evans, B.J. Monovision: A review. Ophthalmic Physiol. Opt. 2007, 27, 417–439. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.V.; Phillips, R.P.; Prasad, S. Multifocal intraocular lenses in the setting of glaucoma. Curr. Opin. Ophthalmol. 2007, 18, 62–66. [Google Scholar] [CrossRef]
- Grzybowski, A.; Kanclerz, P.; Tuuminen, R. Multifocal intraocular lenses and retinal diseases. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 805–813. [Google Scholar] [CrossRef]
- Nuzzi, R.; Tripoli, F.; Ghilardi, A. Evaluation of the Effects of Multifocal Intraocular Lens Oculentis LENTIS Mplus LS-313 MF30 on Visual Performance in Patients Affected by Bilateral Cataract and Treated with Phacoemulsification. J. Ophthalmol. 2022, 2022, 1315480. [Google Scholar] [CrossRef] [PubMed]
- Artigas, J.M.; Menezo, J.L.; Peris, C.; Felipe, A.; Díaz-Llopis, M. Image quality with multifocal intraocular lenses and the effect of pupil size: Comparison of refractive and hybrid refractive-diffractive designs. J. Cataract Refract. Surg. 2007, 33, 2111–2117. [Google Scholar] [CrossRef]
- Palomino Bautista, C.; Carmona González, D.; Castillo Gómez, A.; Bescos, J.A. Evolution of visual performance in 250 eyes implanted with the Tecnis ZM900 multifocal IOL. Eur. J. Ophthalmol. 2009, 19, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Nuzzi, R.; Tridico, F. Comparison of visual outcomes, spectacles dependence and patient satisfaction of multifocal and accommodative intraocular lenses: Innovative perspectives for maximal refractive-oriented cataract surgery. BMC Ophthalmol. 2017, 17, 12. [Google Scholar] [CrossRef][Green Version]
- Javitt, J.C.; Steinert, R.F. Cataract extraction with multifocal intraocular lens implantation: A multinational clinical trial evaluating clinical, functional, and quality-of-life outcomes. Ophthalmology 2000, 107, 2040–2048. [Google Scholar] [CrossRef] [PubMed]
- Martínez Palmer, A.; Gómez Faiña, P.; España Albelda, A.; Comas Serrano, M.; Nahra Saad, D.; Castilla Céspedes, M. Visual function with bilateral implantation of monofocal and multifocal intraocular lenses: A prospective, randomized, controlled clinical trial. J. Refract. Surg. 2008, 24, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Pieh, S.; Lackner, B.; Hanselmayer, G.; Zöhrer, R.; Sticker, M.; Weghaupt, H.; Fercher, A.; Skorpik, C. Halo size under distance and near conditions in refractive multifocal intraocular lenses. Br. J. Ophthalmol. 2001, 85, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Balgos, M.; Vargas, V.; Alió, J.L. Correction of presbyopia: An integrated update for the practical surgeon. Taiwan J. Ophthalmol. 2018, 8, 121–140. [Google Scholar] [CrossRef] [PubMed]
- Farid, M.; Chak, G.; Garg, S.; Steinert, R.F. Reduction in mean deviation values in automated perimetry in eyes with multifocal compared to monofocal intraocular lens implants. Am. J. Ophthalmol. 2014, 158, 227–231.e221. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Friedman, D.S.; Jin, S.; Yusufu, M.; Zhang, J.; Wang, J.; Hou, S.; Zhu, G.; Wang, B.; Xiong, Y.; et al. Multifocal versus monofocal intraocular lenses for age-related cataract patients: A system review and meta-analysis based on randomized controlled trials. Surv. Ophthalmol. 2019, 64, 647–658. [Google Scholar] [CrossRef]
- Akella, S.S.; Juthani, V.V. Extended depth of focus intraocular lenses for presbyopia. Curr. Opin. Ophthalmol. 2018, 29, 318–322. [Google Scholar] [CrossRef]
- Ouchi, M.; Kinoshita, S. Implantation of refractive multifocal intraocular lens with a surface-embedded near section for cataract eyes complicated with a coexisting ocular pathology. Eye 2015, 29, 649–655. [Google Scholar] [CrossRef][Green Version]
- de Vries, N.E.; Webers, C.A.; Verbakel, F.; de Brabander, J.; Berendschot, T.T.; Cheng, Y.Y.; Doors, M.; Nuijts, R.M. Visual outcome and patient satisfaction after multifocal intraocular lens implantation: Aspheric versus spherical design. J. Cataract Refract. Surg. 2010, 36, 1897–1904. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, G.S.; Ramamurthy, S.; Sharma, U.; Dandapani, R. Visual outcomes of patients bilaterally implanted with the extended range of vision intraocular lens: A prospective study. Indian J. Ophthalmol. 2018, 66, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Auffarth, G.U.; Gerl, M.; Tsai, L.; Janakiraman, D.P.; Jackson, B.; Alarcon, A.; Dick, H.B. Clinical evaluation of a new monofocal IOL with enhanced intermediate function in patients with cataract. J. Cataract Refract. Surg. 2021, 47, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Unsal, U.; Sabur, H. Comparison of new monofocal innovative and standard monofocal intraocular lens after phacoemulsification. Int. Ophthalmol. 2021, 41, 273–282. [Google Scholar] [CrossRef]
- Liu, J.; Dong, Y.; Wang, Y. Efficacy and safety of extended depth of focus intraocular lenses in cataract surgery: A systematic review and meta-analysis. BMC Ophthalmol. 2019, 19, 198. [Google Scholar] [CrossRef]
- Gatinel, D.; Loicq, J. Clinically Relevant Optical Properties of Bifocal, Trifocal, and Extended Depth of Focus Intraocular Lenses. J. Refract. Surg. 2016, 32, 273–280. [Google Scholar] [CrossRef]
- Pedrotti, E.; Bruni, E.; Bonacci, E.; Badalamenti, R.; Mastropasqua, R.; Marchini, G. Comparative Analysis of the Clinical Outcomes with a Monofocal and an Extended Range of Vision Intraocular Lens. J. Refract. Surg. 2016, 32, 436–442. [Google Scholar] [CrossRef]
- Pedrotti, E.; Carones, F.; Aiello, F.; Mastropasqua, R.; Bruni, E.; Bonacci, E.; Talli, P.; Nucci, C.; Mariotti, C.; Marchini, G. Comparative analysis of visual outcomes with 4 intraocular lenses: Monofocal, multifocal, and extended range of vision. J. Cataract Refract. Surg. 2018, 44, 156–167. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, Y.; Hao, R.; Jiang, X.; Liu, Z.; Li, X. Comparison of Patient Outcomes following Implantation of Trifocal and Extended Depth of Focus Intraocular Lenses: A Systematic Review and Meta-Analysis. J. Ophthalmol. 2021, 2021, 1115076. [Google Scholar] [CrossRef] [PubMed]
- Mencucci, R.; Favuzza, E.; Caporossi, O.; Savastano, A.; Rizzo, S. Comparative analysis of visual outcomes, reading skills, contrast sensitivity, and patient satisfaction with two models of trifocal diffractive intraocular lenses and an extended range of vision intraocular lens. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 1913–1922. [Google Scholar] [CrossRef] [PubMed]
- Montés-Micó, R.; Ferrer-Blasco, T.; Charman, W.N.; Cerviño, A.; Alfonso, J.F.; Fernández-Vega, L. Optical quality of the eye after lens replacement with a pseudoaccommodating intraocular lens. J. Cataract Refract. Surg. 2008, 34, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Iancu, R.; Corbu, C. Premium intraocular lenses use in patients with cataract and concurrent glaucoma: A review. Maedica 2013, 8, 290–296. [Google Scholar] [PubMed]
- Ong, H.S.; Evans, J.R.; Allan, B.D. Accommodative intraocular lens versus standard monofocal intraocular lens implantation in cataract surgery. Cochrane Database Syst. Rev. 2014, Cd009667. [Google Scholar] [CrossRef] [PubMed]
- Petermeier, K.; Messias, A.; Gekeler, F.; Szurman, P. Effect of +3.00 diopter and +4.00 diopter additions in multifocal intraocular lenses on defocus profiles, patient satisfaction, and contrast sensitivity. J. Cataract Refract. Surg. 2011, 37, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Woodward, M.A.; Randleman, J.B.; Stulting, R.D. Dissatisfaction after multifocal intraocular lens implantation. J. Cataract Refract. Surg. 2009, 35, 992–997. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 5 June 2023).
- Murad, M.H.; Sultan, S.; Haffar, S.; Bazerbachi, F. Methodological quality and synthesis of case series and case reports. BMJ Evid. Based. Med. 2018, 23, 60–63. [Google Scholar] [CrossRef]
- Ferguson, T.J.; Wilson, C.W.; Shafer, B.M.; Berdahl, J.P.; Terveen, D.C. Clinical Outcomes of a Non-Diffractive Extended Depth-of-Focus IOL in Eyes with Mild Glaucoma. Clin. Ophthalmol. 2023, 17, 861–868. [Google Scholar] [CrossRef]
- Bissen-Miyajima, H.; Ota, Y.; Yuki, K.; Minami, K. Implantation of diffractive extended depth-of-focus intraocular lenses in normal tension glaucoma eyes: A case series. Am. J. Ophthalmol. Case Rep. 2023, 29, 101792. [Google Scholar] [CrossRef]
- Ichioka, S.; Ishida, A.; Takayanagi, Y.; Manabe, K.; Matsuo, M.; Tanito, M.; Tanito, M. Roles of Toric intraocular Lens implantation on visual acuity and astigmatism in glaucomatous eyes treated with iStent and cataract surgery. BMC Ophthalmol. 2022, 22, 487. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Sánchez, C.; Rementería-Capelo, L.A.; Puerto, B.; López-Caballero, C.; Morán, A.; Sánchez-Pina, J.M.; Contreras, I. Visual Function and Patient Satisfaction with Multifocal Intraocular Lenses in Patients with Glaucoma and Dry Age-Related Macular Degeneration. J. Ophthalmol. 2021, 2021, 9935983. [Google Scholar] [CrossRef]
- Takai, Y.; Sugihara, K.; Mochiji, M.; Manabe, K.; Tsutsui, A.; Tanito, M. Refractive Status in Eyes Implanted with Toric and Nontoric Intraocular Lenses during Combined Cataract Surgery and Microhook Ab Interno Trabeculotomy. J. Ophthalmol. 2021, 2021, 5545007. [Google Scholar] [CrossRef]
- López-Caballero, C.; Sánchez-Sánchez, C.; Puerto, B.; Blázquez, V.; Sánchez-Pina, J.M.; Contreras, I. Refractive outcomes of toric intraocular lens in combined trabecular micro bypass stent implantation and cataract surgery in glaucomatous eyes. Int. Ophthalmol. 2022, 42, 2711–2718. [Google Scholar] [CrossRef] [PubMed]
- Ichioka, S.; Manabe, K.; Tsutsui, A.; Takai, Y.; Tanito, M. Effect of Toric Intraocular Lens Implantation on Visual Acuity and Astigmatism Status in Eyes Treated With Microhook Ab Interno Trabeculotomy. J. Glaucoma 2021, 30, 94–100. [Google Scholar] [CrossRef]
- Brown, R.H.; Zhong, L.; Bozeman, C.W.; Lynch, M.G. Toric Intraocular Lens Outcomes in Patients With Glaucoma. J. Refract. Surg. 2015, 31, 366–372. [Google Scholar] [CrossRef]
- Kamath, G.G.; Prasad, S.; Danson, A.; Phillips, R.P. Visual outcome with the array multifocal intraocular lens in patients with concurrent eye disease. J. Cataract Refract. Surg. 2000, 26, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Rementería-Capelo, L.A.; Lorente, P.; Carrillo, V.; Sánchez-Pina, J.M.; Ruiz-Alcocer, J.; Contreras, I. Patient Satisfaction and Visual Performance in Patients with Ocular Pathology after Bilateral Implantation of a New Extended Depth of Focus Intraocular Lens. J. Ophthalmol. 2022, 2022, 4659309. [Google Scholar] [CrossRef] [PubMed]
- Kerr, N.M.; Moshegov, S.; Lim, S.; Simos, M. Visual Outcomes, Spectacle Independence, and Patient-Reported Satisfaction of the Vivity Extended Range of Vision Intraocular Lens in Patients with Early Glaucoma: An Observational Comparative Study. Clin. Ophthalmol. 2023, 17, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, A.S.; Szlyk, J.P.; Ardickas, Z.; Alexander, K.R.; Wilensky, J.T. Comparison of contrast sensitivity, visual acuity, and Humphrey visual field testing in patients with glaucoma. J. Glaucoma 2003, 12, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Fatehi, N.; Nowroozizadeh, S.; Henry, S.; Coleman, A.L.; Caprioli, J.; Nouri-Mahdavi, K. Association of Structural and Functional Measures With Contrast Sensitivity in Glaucoma. Am. J. Ophthalmol. 2017, 178, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Richman, J.; Lorenzana, L.L.; Lankaranian, D.; Dugar, J.; Mayer, J.; Wizov, S.S.; Spaeth, G.L. Importance of visual acuity and contrast sensitivity in patients with glaucoma. Arch. Ophthalmol. 2010, 128, 1576–1582. [Google Scholar] [CrossRef]
- Burton, R.; Crabb, D.P.; Smith, N.D.; Glen, F.C.; Garway-Heath, D.F. Glaucoma and reading: Exploring the effects of contrast lowering of text. Optom. Vis. Sci. 2012, 89, 1282–1287. [Google Scholar] [CrossRef] [PubMed]
- Korth, M.J.; Jünemann, A.M.; Horn, F.K.; Bergua, A.; Cursiefen, C.; Velten, I.; Budde, W.M.; Wisse, M.; Martus, P. Synopsis of various electrophysiological tests in early glaucoma diagnosis--temporal and spatiotemporal contrast sensitivity, light- and color-contrast pattern-reversal electroretinogram, blue-yellow VEP. Klin. Monbl. Augenheilkd. 2000, 216, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Yeu, E.; Cuozzo, S. Matching the Patient to the Intraocular Lens: Preoperative Considerations to Optimize Surgical Outcomes. Ophthalmology 2021, 128, e132–e141. [Google Scholar] [CrossRef] [PubMed]
- Trueb, P.R.; Albach, C.; Montés-Micó, R.; Ferrer-Blasco, T. Visual acuity and contrast sensitivity in eyes implanted with aspheric and spherical intraocular lenses. Ophthalmology 2009, 116, 890–895. [Google Scholar] [CrossRef]
- Deshpande, R.; Satijia, A.; Dole, K.; Mangiraj, V.; Deshpande, M. Effects on ocular aberration and contrast sensitivity after implantation of spherical and aspherical monofocal intraocular lens—A comparative study. Indian J. Ophthalmol. 2022, 70, 2862–2865. [Google Scholar] [CrossRef] [PubMed]
- Aychoua, N.; Junoy Montolio, F.G.; Jansonius, N.M. Influence of multifocal intraocular lenses on standard automated perimetry test results. JAMA Ophthalmol. 2013, 131, 481–485. [Google Scholar] [CrossRef]
- Wilensky, J.T.; Hawkins, A. Comparison of contrast sensitivity, visual acuity, and Humphrey visual field testing in patients with glaucoma. Trans. Am. Ophthalmol. Soc. 2001, 99, 213–217, discussion 217–218. [Google Scholar]
- Kang, S.G.; Lee, J.H. The change of visual acuity and visual field by diminished illumination in eyes with multifocal intraocular lens. Korean J. Ophthalmol. 1994, 8, 72–76. [Google Scholar] [CrossRef]
- Bi, H.; Cui, Y.; Ma, X.; Cai, W.; Wang, G.; Ji, P.; Xie, X. Early clinical evaluation of AcrySof ReSTOR multifocal intraocular lens for treatment of cataract. Ophthalmologica 2008, 222, 11–16. [Google Scholar] [CrossRef]
- Athanasiadis, Y.; de Wit, D.W.; Nithyanandrajah, G.A.; Patel, A.; Sharma, A. Neodymium:YAG laser peripheral iridotomy as a possible cause of zonular dehiscence during phacoemulsification cataract surgery. Eye 2010, 24, 1424–1425. [Google Scholar] [CrossRef]
- Mutoh, T.; Barrette, K.F.; Matsumoto, Y.; Chikuda, M. Lens dislocation has a possible relationship with laser iridotomy. Clin. Ophthalmol. 2012, 6, 2019–2022. [Google Scholar] [CrossRef]
- Hu, R.; Wang, X.; Wang, Y.; Sun, Y. Occult lens subluxation related to laser peripheral iridotomy: A case report and literature review. Medicine 2017, 96, e6255. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Varma, R. Natural history of glaucoma. Indian J. Ophthalmol. 2011, 59, S19–S23. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Dai, Y.; Chen, Y.; Yu, D.Y.; Cringle, S.J.; Chen, J.; Kong, X.; Wang, X.; Jiang, C. Primary angle closure glaucoma: What we know and what we don’t know. Prog. Retin. Eye Res. 2017, 57, 26–45. [Google Scholar] [CrossRef] [PubMed]
- Levkovitch-Verbin, H.; Habot-Wilner, Z.; Burla, N.; Melamed, S.; Goldenfeld, M.; Bar-Sela, S.M.; Sachs, D. Intraocular pressure elevation within the first 24 hours after cataract surgery in patients with glaucoma or exfoliation syndrome. Ophthalmology 2008, 115, 104–108. [Google Scholar] [CrossRef]
- Law, S.K.; Riddle, J. Management of cataracts in patients with glaucoma. Int. Ophthalmol. Clin. 2011, 51, 1–18. [Google Scholar] [CrossRef]
- Drolsum, L.; Haaskjold, E.; Davanger, M. Pseudoexfoliation syndrome and extracapsular cataract extraction. Acta Ophthalmol. 1993, 71, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Drolsum, L.; Ringvold, A.; Nicolaissen, B. Cataract and glaucoma surgery in pseudoexfoliation syndrome: A review. Acta Ophthalmol. Scand. 2007, 85, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Scorolli, L.; Scorolli, L.; Campos, E.C.; Bassein, L.; Meduri, R.A. Pseudoexfoliation syndrome: A cohort study on intraoperative complications in cataract surgery. Ophthalmologica 1998, 212, 278–280. [Google Scholar] [CrossRef]
- Ritch, R. Exfoliation syndrome. Curr. Opin. Ophthalmol. 2001, 12, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Riedl, J.C.; Rings, S.; Schuster, A.K.; Vossmerbaeumer, U. Intraocular lens dislocation: Manifestation, ocular and systemic risk factors. Int. Ophthalmol. 2023, 43, 1317–1324. [Google Scholar] [CrossRef] [PubMed]
- Shingleton, B.J.; Neo, Y.N.; Cvintal, V.; Shaikh, A.M.; Liberman, P.; O’Donoghue, M.W. Outcome of phacoemulsification and intraocular lens implantion in eyes with pseudoexfoliation and weak zonules. Acta Ophthalmol. 2017, 95, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Jehan, F.S.; Mamalis, N.; Crandall, A.S. Spontaneous late dislocation of intraocular lens within the capsular bag in pseudoexfoliation patients. Ophthalmology 2001, 108, 1727–1731. [Google Scholar] [CrossRef]
- Davis, D.; Brubaker, J.; Espandar, L.; Stringham, J.; Crandall, A.; Werner, L.; Mamalis, N. Late in-the-bag spontaneous intraocular lens dislocation: Evaluation of 86 consecutive cases. Ophthalmology 2009, 116, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Mönestam, E.I. Incidence of dislocation of intraocular lenses and pseudophakodonesis 10 years after cataract surgery. Ophthalmology 2009, 116, 2315–2320. [Google Scholar] [CrossRef]
- Shingleton, B.J.; Marvin, A.C.; Heier, J.S.; O’Donoghue, M.W.; Laul, A.; Wolff, B.; Rowland, A. Pseudoexfoliation: High risk factors for zonule weakness and concurrent vitrectomy during phacoemulsification. J. Cataract Refract. Surg. 2010, 36, 1261–1269. [Google Scholar] [CrossRef]
- Manoharan, N.; Patnaik, J.L.; Bonnell, L.N.; SooHoo, J.R.; Pantcheva, M.B.; Kahook, M.Y.; Wagner, B.D.; Lynch, A.M.; Seibold, L.K. Refractive outcomes of phacoemulsification cataract surgery in glaucoma patients. J. Cataract Refract. Surg. 2018, 44, 348–354. [Google Scholar] [CrossRef]
- Moon, Y.; Sung, K.R.; Kim, J.M.; Shim, S.H.; Yoo, C.; Park, J.H. Risk Factors Associated With Glaucomatous Progression in Pseudoexfoliation Patients. J. Glaucoma 2017, 26, 1107–1113. [Google Scholar] [CrossRef]
- Stewart, W.C.; Stewart, J.A.; Nelson, L.A. Ocular surface disease in patients with ocular hypertension and glaucoma. Curr. Eye Res. 2011, 36, 391–398. [Google Scholar] [CrossRef]
- Zhang, X.; Vadoothker, S.; Munir, W.M.; Saeedi, O. Ocular Surface Disease and Glaucoma Medications: A Clinical Approach. Eye Contact Lens. 2019, 45, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Kuppens, E.V.; van Best, J.A.; Sterk, C.C.; de Keizer, R.J. Decreased basal tear turnover in patients with untreated primary open-angle glaucoma. Am. J. Ophthalmol. 1995, 120, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Skalicky, S.E.; Goldberg, I.; McCluskey, P. Ocular surface disease and quality of life in patients with glaucoma. Am. J. Ophthalmol. 2012, 153, 1–9.e2. [Google Scholar] [CrossRef] [PubMed]
- Mocan, M.C.; Uzunosmanoglu, E.; Kocabeyoglu, S.; Karakaya, J.; Irkec, M. The Association of Chronic Topical Prostaglandin Analog Use With Meibomian Gland Dysfunction. J. Glaucoma 2016, 25, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, K.; Parkhurst, G.; Saenz, B.; Whitley, W.; Williamson, B.; Hovanesian, J. Call to action: Treating dry eye disease and setting the foundation for successful surgery. J. Cataract Refract. Surg. 2022, 48, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Starr, C.E.; Gupta, P.K.; Farid, M.; Beckman, K.A.; Chan, C.C.; Yeu, E.; Gomes, J.A.P.; Ayers, B.D.; Berdahl, J.P.; Holland, E.J.; et al. An algorithm for the preoperative diagnosis and treatment of ocular surface disorders. J. Cataract Refract. Surg. 2019, 45, 669–684. [Google Scholar] [CrossRef]
- Fu, L.; Chan, Y.K.; Li, J.; Nie, L.; Li, N.; Pan, W. Long term outcomes of cataract surgery in severe and end stage primary angle closure glaucoma with controlled IOP: A retrospective study. BMC Ophthalmol. 2020, 20, 160. [Google Scholar] [CrossRef]
- Mehta, R.; Tomatzu, S.; Cao, D.; Pleet, A.; Mokhur, A.; Aref, A.A.; Vajaranant, T.S. Refractive Outcomes for Combined Phacoemulsification and Glaucoma Drainage Procedure. Ophthalmol. Ther. 2022, 11, 311–320. [Google Scholar] [CrossRef]
- Chen, S.J.; Lu, P.; Zhang, W.F.; Lu, J.H. High myopia as a risk factor in primary open angle glaucoma. Int. J. Ophthalmol. 2012, 5, 750–753. [Google Scholar] [CrossRef]
- Kuzin, A.A.; Varma, R.; Reddy, H.S.; Torres, M.; Azen, S.P. Ocular biometry and open-angle glaucoma: The Los Angeles Latino Eye Study. Ophthalmology 2010, 117, 1713–1719. [Google Scholar] [CrossRef]
- Sherpa, D.; Badhu, B.P. Association between axial length of the eye and primary angle closure glaucoma. Kathmandu Univ. Med. J. KUMJ 2008, 6, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Jiang, W.; Lin, T.; Wu, X.; Lin, H.; Chen, W. Meta-analysis of accuracy of intraocular lens power calculation formulas in short eyes. Clin. Exp. Ophthalmol. 2018, 46, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Hou, Y.; Yang, H.; Sun, X. A systemic review and network meta-analysis of accuracy of intraocular lens power calculation formulas in primary angle-closure conditions. PLoS ONE 2022, 17, e0276286. [Google Scholar] [CrossRef] [PubMed]
- Ning, X.; Yang, Y.; Yan, H.; Zhang, J. Anterior chamber depth—A predictor of refractive outcomes after age-related cataract surgery. BMC Ophthalmol. 2019, 19, 134. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.S.; Sung, M.S.; Heo, H.; Ji, Y.S.; Park, S.W. Long-term outcomes of prediction error after combined phacoemulsification and trabeculectomy in glaucoma patients. BMC Ophthalmol. 2021, 21, 60. [Google Scholar] [CrossRef]
- Krupin, T.; Feitl, M.E.; Bishop, K.I. Postoperative intraocular pressure rise in open-angle glaucoma patients after cataract or combined cataract-filtration surgery. Ophthalmology 1989, 96, 579–584. [Google Scholar] [CrossRef]
- Murthy, S.K.; Damji, K.F.; Pan, Y.; Hodge, W.G. Trabeculectomy and phacotrabeculectomy, with mitomycin-C, show similar two-year target IOP outcomes. Can. J. Ophthalmol. 2006, 41, 51–59. [Google Scholar] [CrossRef]
- Tsai, H.Y.; Liu, C.J.; Cheng, C.Y. Combined trabeculectomy and cataract extraction versus trabeculectomy alone in primary angle-closure glaucoma. Br. J. Ophthalmol. 2009, 93, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Tzu, J.H.; Shah, C.T.; Galor, A.; Junk, A.K.; Sastry, A.; Wellik, S.R. Refractive outcomes of combined cataract and glaucoma surgery. J. Glaucoma 2015, 24, 161–164. [Google Scholar] [CrossRef]
- Vaideanu, D.; Mandal, K.; Hildreth, A.; Fraser, S.G.; Phelan, P.S. Visual and refractive outcome of one-site phacotrabeculectomy compared with temporal approach phacoemulsification. Clin. Ophthalmol. 2008, 2, 569–574. [Google Scholar] [CrossRef][Green Version]
- El Sayed, Y.M.; Elhusseiny, A.M.; Albalkini, A.S.; El Sheikh, R.H.; Osman, M.A. Mitomycin C-augmented Phacotrabeculectomy Versus Phacoemulsification in Primary Angle-closure Glaucoma: A Randomized Controlled Study. J. Glaucoma 2019, 28, 911–915. [Google Scholar] [CrossRef] [PubMed]
- Hugkulstone, C.E. Changes in keratometry following trabeculectomy. Br. J. Ophthalmol. 1991, 75, 217–218. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.; Nongpiur, M.; Peter, L.; Perera, S.A. Combined Approach to Phacoemulsification and Trabeculectomy Results in Less Ideal Refractive Outcomes Compared With the Sequential Approach. J. Glaucoma 2016, 25, e873–e878. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.W.; Lee, Y.H.; Kim, D.W.; Lee, T.; Hong, S.; Seong, G.J.; Kim, C.Y. Effect of trabeculectomy on the accuracy of intraocular lens calculations in patients with open-angle glaucoma. Clin. Exp. Ophthalmol. 2016, 44, 465–471. [Google Scholar] [CrossRef]
- Chan, J.C.; Lai, J.S.; Tham, C.C. Comparison of postoperative refractive outcome in phacotrabeculectomy and phacoemulsification with posterior chamber intraocular lens implantation. J. Glaucoma 2006, 15, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Muallem, M.S.; Nelson, G.A.; Osmanovic, S.; Quinones, R.; Viana, M.; Edward, D.P. Predicted refraction versus refraction outcome in cataract surgery after trabeculectomy. J. Glaucoma 2009, 18, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.Y.; Wu, S.C. Refractive error with optimum intraocular lens power calculation after glaucoma filtering surgery. J. Cataract Refract. Surg. 2004, 30, 2595–2597. [Google Scholar] [CrossRef]
- Zhang, N.; Tsai, P.L.; Catoira-Boyle, Y.P.; Morgan, L.S.; Hoop, J.S.; Cantor, L.B.; WuDunn, D. The effect of prior trabeculectomy on refractive outcomes of cataract surgery. Am. J. Ophthalmol. 2013, 155, 858–863. [Google Scholar] [CrossRef]
- Yeh, O.L.; Bojikian, K.D.; Slabaugh, M.A.; Chen, P.P. Refractive Outcome of Cataract Surgery in Eyes With Prior Trabeculectomy: Risk Factors for Postoperative Myopia. J. Glaucoma 2017, 26, 65–70. [Google Scholar] [CrossRef]
- Li, S.W.; Chen, Y.; Wu, Q.; Lu, B.; Wang, W.Q.; Fang, J. Angle parameter changes of phacoemulsification and combined phacotrabeculectomy for acute primary angle closure. Int. J. Ophthalmol. 2015, 8, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, K.; Takamura, Y.; Arimura, S.; Tsuji, T.; Matsumura, T.; Gozawa, M.; Inatani, M. Prospective Cohort Study on Refractive Changes after Trabeculectomy. J. Ophthalmol. 2019, 2019, 4731653. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Park, K.; Ha, S.J.; Yeom, H.Y.; Seong, G.J.; Hong, Y.J. Long-term intraocular pressure control of trabeculectomy and triple procedure in primary open angle glaucoma and chronic primary angle closure glaucoma. Ophthalmologica 2007, 221, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Z.; Koh, V.; Sng, C.; Aquino, M.C.; Chew, P. Complications and outcomes of primary phacotrabeculectomy with mitomycin C in a multi-ethnic asian population. PLoS ONE 2015, 10, e0118852. [Google Scholar] [CrossRef] [PubMed]
- Cashwell, L.F.; Martin, C.A. Axial length decrease accompanying successful glaucoma filtration surgery. Ophthalmology 1999, 106, 2307–2311. [Google Scholar] [CrossRef] [PubMed]
- Kook, M.S.; Kim, H.B.; Lee, S.U. Short-term effect of mitomycin-C augmented trabeculectomy on axial length and corneal astigmatism. J. Cataract Refract. Surg. 2001, 27, 518–523. [Google Scholar] [CrossRef]
- Pakravan, M.; Alvani, A.; Esfandiari, H.; Ghahari, E.; Yaseri, M. Post-trabeculectomy ocular biometric changes. Clin. Exp. Optom. 2017, 100, 128–132. [Google Scholar] [CrossRef]
- Olsen, T. Sources of error in intraocular lens power calculation. J. Cataract Refract Surg. 1992, 18, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Drexler, W.; Findl, O.; Menapace, R.; Rainer, G.; Vass, C.; Hitzenberger, C.K.; Fercher, A.F. Partial coherence interferometry: A novel approach to biometry in cataract surgery. Am. J. Ophthalmol. 1998, 126, 524–534. [Google Scholar] [CrossRef]
- Poon, L.Y.; Lai, I.C.; Lee, J.J.; Tsai, J.C.; Lin, P.W.; Teng, M.C. Comparison of surgical outcomes after phacotrabeculectomy in primary angle-closure glaucoma versus primary open-angle glaucoma. Taiwan J. Ophthalmol. 2015, 5, 28–32. [Google Scholar] [CrossRef]
- Law, S.K.; Mansury, A.M.; Vasudev, D.; Caprioli, J. Effects of combined cataract surgery and trabeculectomy with mitomycin C on ocular dimensions. Br. J. Ophthalmol. 2005, 89, 1021–1025. [Google Scholar] [CrossRef]
- Németh, J.; Horóczi, Z. Changes in the ocular dimensions after trabeculectomy. Int. Ophthalmol. 1992, 16, 355–357. [Google Scholar] [CrossRef] [PubMed]
- Volzhanin, A.V.; Petrov, S.Y.; Safonova, D.M.; Averich, V.V. On refraction shift after trabeculectomy. Vestn. Oftalmol. 2022, 138, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Husain, R.; Li, W.; Gazzard, G.; Foster, P.J.; Chew, P.T.; Oen, F.T.; Phillips, R.; Khaw, P.T.; Seah, S.K.; Aung, T. Longitudinal changes in anterior chamber depth and axial length in Asian subjects after trabeculectomy surgery. Br. J. Ophthalmol. 2013, 97, 852–856. [Google Scholar] [CrossRef]
- Dickens, M.A.; Cashwell, L.F. Long-term effect of cataract extraction on the function of an established filtering bleb. Ophthalmic Surg. Lasers 1996, 27, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Francis, B.A.; Wang, M.; Lei, H.; Du, L.T.; Minckler, D.S.; Green, R.L.; Roland, C. Changes in axial length following trabeculectomy and glaucoma drainage device surgery. Br. J. Ophthalmol. 2005, 89, 17–20. [Google Scholar] [CrossRef]
- Rosen, W.J.; Mannis, M.J.; Brandt, J.D. The effect of trabeculectomy on corneal topography. Ophthalmic Surg. 1992, 23, 395–398. [Google Scholar] [CrossRef]
- Claridge, K.G.; Galbraith, J.K.; Karmel, V.; Bates, A.K. The effect of trabeculectomy on refraction, keratometry and corneal topography. Eye 1995, 9 Pt 3, 292–298. [Google Scholar] [CrossRef]
- Investigators, A.A.G.I.S. The Advanced Glaucoma Intervention Study: 8. Risk of cataract formation after trabeculectomy. Arch. Ophthalmol. 2001, 119, 1771–1779. [Google Scholar] [CrossRef]
- Cunliffe, I.A.; Dapling, R.B.; West, J.; Longstaff, S. A prospective study examining the changes in factors that affect visual acuity following trabeculectomy. Eye 1992, 6 Pt 6, 618–622. [Google Scholar] [CrossRef]
- Dietze, P.J.; Oram, O.; Kohnen, T.; Feldman, R.M.; Koch, D.D.; Gross, R.L. Visual function following trabeculectomy: Effect on corneal topography and contrast sensitivity. J. Glaucoma 1997, 6, 99–103. [Google Scholar] [CrossRef]
- Vernon, S.A.; Zambarakji, H.J.; Potgieter, F.; Evans, J.; Chell, P.B. Topographic and keratometric astigmatism up to 1 year following small flap trabeculectomy (microtrabeculectomy). Br. J. Ophthalmol. 1999, 83, 779–782. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Egrilmez, S.; Ates, H.; Nalcaci, S.; Andac, K.; Yagci, A. Surgically induced corneal refractive change following glaucoma surgery: Nonpenetrating trabecular surgeries versus trabeculectomy. J. Cataract Refract. Surg. 2004, 30, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Willekens, K.; Pinto, L.A.; Delbeke, H.; Vandewalle, E.; Stalmans, I. Trabeculectomy With Moorfields Conjunctival Closure Technique Offers Safety Without Astigmatism Induction. J. Glaucoma 2016, 25, e531–e535. [Google Scholar] [CrossRef]
- Kumari, R.; Saha, B.C.; Puri, L.R. Keratometric astigmatism evaluation after trabeculectomy. Nepal. J. Ophthalmol. 2013, 5, 215–219. [Google Scholar] [CrossRef]
- Hong, Y.J.; Choe, C.M.; Lee, Y.G.; Chung, H.S.; Kim, H.K. The effect of mitomycin-C on postoperative corneal astigmatism in trabeculectomy and a triple procedure. Ophthalmic Surg. Lasers 1998, 29, 484–489. [Google Scholar] [CrossRef]
- Belov, D.F.; Nikolaenko, V.P. Calculation of intraocular lens power after trabeculectomy. Vestn. Oftalmol. 2021, 137, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Pakravan, M.; Alvani, A.; Yazdani, S.; Esfandiari, H.; Yaseri, M. Intraocular lens power changes after mitomycin trabeculectomy. Eur. J. Ophthalmol. 2015, 25, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Iijima, K.; Kamiya, K.; Iida, Y.; Kasahara, M.; Shoji, N. Predictability of combined cataract surgery and trabeculectomy using Barrett Universal Ⅱ formula. PLoS ONE 2022, 17, e0270363. [Google Scholar] [CrossRef]
- Saheb, H.; Ahmed, I.I. Micro-invasive glaucoma surgery: Current perspectives and future directions. Curr. Opin. Ophthalmol. 2012, 23, 96–104. [Google Scholar] [CrossRef]
- Pereira, I.C.F.; van de Wijdeven, R.; Wyss, H.M.; Beckers, H.J.M.; den Toonder, J.M.J. Conventional glaucoma implants and the new MIGS devices: A comprehensive review of current options and future directions. Eye 2021, 35, 3202–3221. [Google Scholar] [CrossRef]
- Shah, M. Micro-invasive glaucoma surgery—An interventional glaucoma revolution. Eye Vis. 2019, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, T.W.; Katz, L.J.; Wells, J.M.; Duh, Y.J.; Giamporcaro, J.E. Randomized evaluation of the trabecular micro-bypass stent with phacoemulsification in patients with glaucoma and cataract. Ophthalmology 2011, 118, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.A.; Ferguson, T.J.; Stephens, J.D.; Berdahl, J.P. Refractive outcomes after trabecular microbypass stent with cataract extraction in open-angle glaucoma. Clin. Ophthalmol. 2019, 13, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Ang, B.C.H.; Chiew, W.; Yip, V.C.H.; Chua, C.H.; Han, W.S.; Tecson, I.O.C.; Ogle, J.J.; Lim, B.A.; Hee, O.K.; Tay, E.L.Y.; et al. Prospective 12-month outcomes of combined iStent inject implantation and phacoemulsification in Asian eyes with normal tension glaucoma. Eye Vis. 2022, 9, 27. [Google Scholar] [CrossRef]
- Ioannidis, A.S.; Töteberg-Harms, M.; Hamann, T.; Hodge, C. Refractive Outcomes After Trabecular Micro-Bypass Stents (iStent Inject) with Cataract Extraction in Open-Angle Glaucoma. Clin. Ophthalmol. 2020, 14, 517–524. [Google Scholar] [CrossRef]
- Lim, R. The surgical management of glaucoma: A review. Clin. Exp. Ophthalmol. 2022, 50, 213–231. [Google Scholar] [CrossRef] [PubMed]
- Luebke, J.; Boehringer, D.; Neuburger, M.; Anton, A.; Wecker, T.; Cakir, B.; Reinhard, T.; Jordan, J.F. Refractive and visual outcomes after combined cataract and trabectome surgery: A report on the possible influences of combining cataract and trabectome surgery on refractive and visual outcomes. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 419–423. [Google Scholar] [CrossRef]
- Sieck, E.G.; Capitena Young, C.E.; Epstein, R.S.; SooHoo, J.R.; Pantcheva, M.B.; Patnaik, J.L.; Lynch, A.M.; Kahook, M.Y.; Seibold, L.K. Refractive outcomes among glaucoma patients undergoing phacoemulsification cataract extraction with and without Kahook Dual Blade goniotomy. Eye Vis. 2019, 6, 28. [Google Scholar] [CrossRef]
- Grover, D.S.; Flynn, W.J.; Bashford, K.P.; Lewis, R.A.; Duh, Y.J.; Nangia, R.S.; Niksch, B. Performance and Safety of a New Ab Interno Gelatin Stent in Refractory Glaucoma at 12 Months. Am. J. Ophthalmol. 2017, 183, 25–36. [Google Scholar] [CrossRef]
- Bormann, C.; Busch, C.; Rehak, M.; Schmidt, M.; Scharenberg, C.; Ziemssen, F.; Unterlauft, J.D. Refractive Changes after Glaucoma Surgery-A Comparison between Trabeculectomy and XEN Microstent Implantation. Life 2022, 12, 1889. [Google Scholar] [CrossRef] [PubMed]
- Hoeh, H.; Ahmed, I.I.; Grisanti, S.; Grisanti, S.; Grabner, G.; Nguyen, Q.H.; Rau, M.; Yoo, S.; Ianchulev, T. Early postoperative safety and surgical outcomes after implantation of a suprachoroidal micro-stent for the treatment of open-angle glaucoma concomitant with cataract surgery. J. Cataract Refract. Surg. 2013, 39, 431–437. [Google Scholar] [CrossRef]
- Lass, J.H.; Benetz, B.A.; He, J.; Hamilton, C.; Von Tress, M.; Dickerson, J.; Lane, S. Corneal Endothelial Cell Loss and Morphometric Changes 5 Years after Phacoemulsification with or without CyPass Micro-Stent. Am. J. Ophthalmol. 2019, 208, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Sarkisian, S.R., Jr.; Radcliffe, N.; Harasymowycz, P.; Vold, S.; Patrianakos, T.; Zhang, A.; Herndon, L.; Brubaker, J.; Moster, R.; Francis, B. Visual outcomes of combined cataract surgery and minimally invasive glaucoma surgery. J. Cataract Refract. Surg. 2020, 46, 1422–1432. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Bissen-Miyajima, H.; Yoshino, M.; Suzuki, T. Wavy horizontal artifacts on optical coherence tomography line-scanning images caused by diffractive multifocal intraocular lenses. J. Cataract Refract. Surg. 2009, 35, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- Braga-Mele, R.; Chang, D.; Dewey, S.; Foster, G.; Henderson, B.A.; Hill, W.; Hoffman, R.; Little, B.; Mamalis, N.; Oetting, T.; et al. Multifocal intraocular lenses: Relative indications and contraindications for implantation. J. Cataract Refract. Surg. 2014, 40, 313–322. [Google Scholar] [CrossRef]
- Visser, N.; Bauer, N.J.; Nuijts, R.M. Toric intraocular lenses: Historical overview, patient selection, IOL calculation, surgical techniques, clinical outcomes, and complications. J. Cataract Refract. Surg. 2013, 39, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Racette, L.; Chen, K.S.; Johnson, C.A. Functional assessment of glaucoma: Uncovering progression. Surv. Ophthalmol. 2020, 65, 639–661. [Google Scholar] [CrossRef]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
Population:
| Population
|
| Author (Year) | Modified Newcastle-Ottawa Scale | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Selection | Comparability | Outcome | Total Score | ||||||
| Representativeness of Exposed Cohort (Maximum:⋆) | Selection of Non-Exposed Cohort (Maximum:⋆) | Ascertainment of Exposure (Maximum:⋆) | Demonstration that the Current Outcome of Interest Was Not Present at Start of Study (Maximum:⋆) | Comparability of Cohorts on the Basis of the Design or Analysis (Maximum:⋆) | Assessment of Outcome (Maximum:⋆) | Was Follow Up Long Enough for Outcomes to Occur (Maximum:⋆) | Adequacy of Follow Up of Cohorts (Maximum:⋆) | ||
| Ichioka 2022 [4] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 7 | |
| Sanchez-Sanchez 2021 [7] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 8 |
| Takai 2021 [10] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 8 |
| Lopez Caballero 2022 [6] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 8 |
| Ichioka 2021 [3] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 7 | |
| Kamath 2000 [8] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 8 |
| Rementería-Capelo 2022 [5] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 8 |
| Kerr 2023 [9] | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 7 | |
| Author (Year) | * Selection | Ascertainment | Causality | Reporting | ||||
|---|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | |
| Bissen Miyajima 2023 [11] | No | Yes | Yes | No | No | No | Yes | Yes |
| Brown 2015 [12] | No | Yes | Yes | No | No | No | Yes | Yes |
| Ouchi 2015 [13] | No | Yes | Yes | No | No | No | Yes | Yes |
| Ferguson 2023 [14] | No | Yes | Yes | No | No | No | Yes | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, A.S.Y.; Ang, B.C.H.; Dorairaj, E.; Dorairaj, S. Premium Intraocular Lenses in Glaucoma—A Systematic Review. Bioengineering 2023, 10, 993. https://doi.org/10.3390/bioengineering10090993
Hong ASY, Ang BCH, Dorairaj E, Dorairaj S. Premium Intraocular Lenses in Glaucoma—A Systematic Review. Bioengineering. 2023; 10(9):993. https://doi.org/10.3390/bioengineering10090993
Chicago/Turabian StyleHong, Ashley Shuen Ying, Bryan Chin Hou Ang, Emily Dorairaj, and Syril Dorairaj. 2023. "Premium Intraocular Lenses in Glaucoma—A Systematic Review" Bioengineering 10, no. 9: 993. https://doi.org/10.3390/bioengineering10090993
APA StyleHong, A. S. Y., Ang, B. C. H., Dorairaj, E., & Dorairaj, S. (2023). Premium Intraocular Lenses in Glaucoma—A Systematic Review. Bioengineering, 10(9), 993. https://doi.org/10.3390/bioengineering10090993







