Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural
Abstract
1. Introduction
2. Definition of “Natural Polymers”—Clarifying Existing Ambiguities
- -
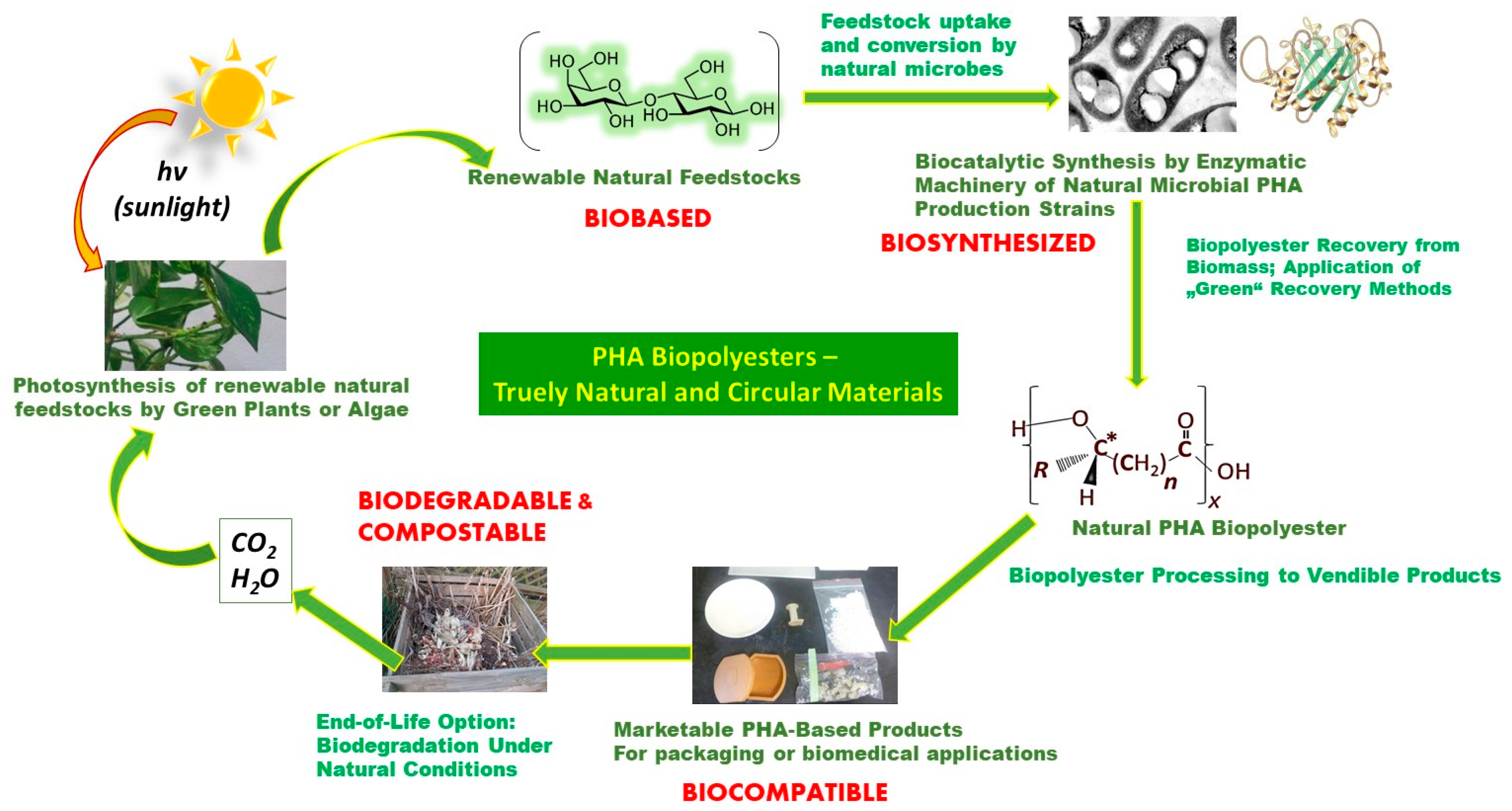
- Bio-based—Denoting a chemical compound or material produced from renewable carbon sources. “Bio-based” does not specify whether the product or material is synthesized chemically or biologically in plants, organisms, and animals.
- -
- Biosynthesis—The synthesis of a chemical or material through the actions in living organisms (plants, microorganisms, or animals) or parts thereof (enzymes). A biosynthesized polymer implies having been produced in plants, microorganisms, or animals through the action of enzymes in vivo. The word “polymerization” implies a synthetic or an anthropogenic process for generation of polymers from building blocks that are either bio-based, synthesized, or chemically synthesized.
- -
- Biodegradation—This implies the breakdown of a chemical or material through the actions of naturally occurring enzymes in vivo or by extracellular enzymes that are secreted by plants, microorganisms, or animals. Generally, breaking down chemicals or materials involves the eventual transformation to carbon dioxide (CO2) and water and complex biomass called humus, also known as organic fertilizer. Hence, “biodegradation” can be understood as nature’s way of material recycling.
- -
- Biocompatibility—Implies chemicals or materials that do not exert harmful effects on the environment or on living systems, including humans.
- Derived from renewable carbon such as sugar from sugar cane, fatty acids like canola oil or carbon dioxide, or methane from biogas or the atmosphere.
- Biosynthesized in plants, microorganisms, and animals.
- Biodegradable or turn into CO2, water, and humus.
- Are biocompatible.
3. PolyHydroxyAlkanoates Are Natural Polymers
Evidence: PHAs Are a Natural Polymer
4. PHAs Are Produced by Natural Microorganisms
5. Biosynthesis of PHA in Natural Strains Occurs via Natural Biocatalysts
6. PHA Biosynthesis Is Based on Natural Feedstocks
7. PHAs Are Biodegraded in Nature—Marine, Fresh Water, and Soil in Every Environment around the World
8. PHAs Do Not Create Recalcitrant Microplastics
9. Industrial PHA Production Is Analogous to PHAs Produced in Nature
10. Products Made of PHAs Are Natural and Biocompatible
11. Definition of Plastics and How PHAs Are Plastics
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leslie, H.A.; Van Velzen, M.J.; Brandsma, S.H.; Vethaak, A.D.; Garcia-Vallejo, J.J.; Lamoree, M.H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Svelato, A.; Santacroce, C.; Catalano, P.; Notarstefano, V.; Carnevali, O.; Papa, F.; Rongioletti, M.C.A.; Baiocco, F.; Draghi, S.; et al. Plasticenta: First evidence of microplastics in human placenta. Environ. Int. 2021, 146, 106274. [Google Scholar] [CrossRef] [PubMed]
- Schwabl, P.; Köppel, S.; Königshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of various microplastics in human stool: A prospective case series. Ann. Intern. Med. 2019, 171, 453–457. [Google Scholar] [CrossRef]
- Mohamed Nor, N.H.; Kooi, M.; Diepens, N.J.; Koelmans, A.A. Lifetime accumulation of microplastic in children and adults. Environ. Sci. Technol. 2021, 55, 5084–5096. [Google Scholar] [CrossRef] [PubMed]
- Gruber, E.S.; Stadlbauer, V.; Pichler, V.; Resch-Fauster, K.; Todorovic, A.; Meisel, T.C.; Trawoeger, S.; Hollóczki, O.; Turner, S.D.; Wadsak, W.; et al. To Waste or Not to Waste: Questioning Potential Health Risks of Micro-and Nanoplastics with a Focus on Their Ingestion and Potential Carcinogenicity. Expo. Health 2023, 15, 33–51. [Google Scholar] [CrossRef]
- Ramsperger, A.F.R.M.; Narayana, V.K.B.; Gross, W.; Mohanraj, J.; Thelakkat, M.; Greiner, A.; Schmalz, H.; Kress, H.; Laforsch, C. Environmental exposure enhances the internalization of microplastic particles into cells. Sci. Adv. 2020, 6, eabd1211. [Google Scholar] [CrossRef]
- Available online: https://openstates.org/ca/bills/20212022/AB1201/ (accessed on 5 June 2023).
- Available online: https://leginfo.legislature.ca.gov/faces/billTextClient.xhtml?bill_id=202120220SB54 (accessed on 5 June 2023).
- Available online: https://eur-lex.europa.eu/eli/dir/2019/904/oj (accessed on 5 June 2023).
- Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=LEGISSUM:l21207 (accessed on 5 June 2023).
- Available online: https://single-market-economy.ec.europa.eu/news/reach-committee-votes-restrict-intentional-microplastics-2023-04-27_en#:~:text=The%20Commission%20welcomes%20the%20positive,are%20intentionally%20added%20to%20products (accessed on 5 June 2023).
- Pathak, P.; Sharma, S.; Ramakrishna, S. Circular transformation in plastic management lessens the carbon footprint of the plastic industry. Mater. Today Sustain. 2023, 22, 100365. [Google Scholar] [CrossRef]
- Available online: https://openstates.org/ca/bills/20192020/AB1080/ (accessed on 5 June 2023).
- Available online: https://leginfo.legislature.ca.gov/faces/billTextClient.xhtml?bill_id=201320140SB270 (accessed on 5 June 2023).
- Available online: https://www.canada.ca/en/environment-climate-change/services/managing-reducing-waste/reduce-plastic-waste/single-use-plastic-overview.html#toc0 (accessed on 11 April 2023).
- Available online: https://www.loc.gov/item/global-legal-monitor/2021-03-23/china-single-use-plastic-straw-and-bag-ban-takes-effect/ (accessed on 11 April 2023).
- Available online: Circulareconomy.earth/publications/how-japan-is-using-the-circular-economy-to-recycle-plastics (accessed on 11 April 2023).
- Available online: https://www.weforum.org/agenda/2022/07/india-ban-policy-single-use-plastic-pollution (accessed on 11 April 2023).
- Aubin, S.; Beaugrand, J.; Berteloot, M.; Boutrou, R.; Buche, P.; Gontard, N.; Guillard, V. Plastics in a circular economy: Mitigating the ambiguity of widely-used terms from stakeholders consultation. Environ. Sci. Pol. 2022, 134, 119–126. [Google Scholar] [CrossRef]
- Koller, M.; Maršálek, L.; Miranda de Sousa Dias, M.; Braunegg, G. Producing microbial polyhydroxyalkanoate (PHA) biopolyesters in a sustainable manner. New Biotechnol. 2017, 37, 24–38. [Google Scholar] [CrossRef]
- Rodriguez-Contreras, A. Recent advances in the use of polyhydroyalkanoates in biomedicine. Bioengineering 2019, 6, 82. [Google Scholar] [CrossRef]
- Lavoisier, A. Traité Elémentaire de Chimie, Présenté Dans un Ordre Nouveau et d’Apres les Découvertes Modernes, 2 Vols; Cuchet Libraire: Paris, France, 1789. (In French) [Google Scholar]
- Anastas, P.T.; Warner, J.C. Green chemistry. Frontiers 1998, 640, 1998. [Google Scholar]
- Ivanković, A.; Dronjić, A.; Bevanda, A.M.; Talić, S. Review of 12 principles of green chemistry in practice. Int. J. Sustain. Green Energy 2017, 6, 39–48. [Google Scholar] [CrossRef]
- Sohn, Y.J.; Son, J.; Jo, S.Y.; Park, S.Y.; Yoo, J.I.; Baritugo, K.-A.; Na, J.G.; Choi, J.-I.; Kim, H.-T.; Joo, J.C.; et al. Chemoautotroph Cupriavidus necator as a potential game-changer for global warming and plastic waste problem: A review. Bioresour. Technol. 2021, 340, 125693. [Google Scholar] [CrossRef] [PubMed]
- Lemoigne, M. The origin of b-hydroxybutyric acid obtained by bacterial process. CR Acad. Sci. 1925, 180, 1539–1541. [Google Scholar]
- Tan, D.; Xue, Y.S.; Aibaidula, G.; Chen, G.-Q. Unsterile and continuous production of polyhydroxybutyrate by Halomonas TD01. Bioresour. Technol. 2011, 102, 8130–8136. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Zhang, Y.; Xi, L.; Huo, F.; Zhao, J.Y.; Li, J. Thermophilic production of polyhydroxyalkanoates by a novel Aneurinibacillus strain isolated from Gudao oilfield, China. J. Basic Microb. 2015, 55, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Rathour, R.; Singh, R.; Sun, Y.; Pandey, A.; Gnansounou, E.; Lin, K.-Y.A.; Tsang, D.C.W.; Thakur, I.S. Bacterial polyhydroxyalkanoates: Opportunities, challenges, and prospects. J. Clean Prod. 2020, 263, 121500. [Google Scholar] [CrossRef]
- Udayakumar, G.P.; Muthusamy, S.; Selvaganesh, B.; Sivarajasekar, N.; Rambabu, K.; Banat, F.; Sivamani, S.; Sivakumar, N.; Hosseini-Bandegharaei, A.; Show, P.L. Biopolymers and composites: Properties, characterization and their applications in food, medical and pharmaceutical industries. J. Environ. Chem. Eng. 2021, 9, 105322. [Google Scholar] [CrossRef]
- Koller, M.; Mukherjee, A.; Obruca, S.; Zinn, M. Polyhydroxyalkanoates (PHA): Microbial synthesis of natural polyesters. In Microbial Production of High-Value Products; Rehm, B.H.A., Wibowo, D., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 185–236. [Google Scholar]
- Avérous, L. Polylactic acid: Synthesis, properties and applications. In Monomers, Polymers and Composites from Renewable Resources; Naceur Belgacem, M., Gandini, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 433–450. [Google Scholar]
- Sadeghi, B.; Marfavi, Y.; AliAkbari, R.; Kowsari, E.; Borbor Ajdari, F.; Ramakrishna, S. Recent studies on recycled PET fibers: Production and applications: A review. Mater. Circ. Econ. 2021, 3, 4. [Google Scholar] [CrossRef]
- Huang, T.Y.; Duan, K.J.; Huang, S.Y.; Chen, C.W. Production of polyhydroxyalkanoates from inexpensive extruded rice bran and starch by Haloferax mediterranei. J. Ind. Microb. Biotechnol. 2006, 33, 701–706. [Google Scholar] [CrossRef]
- Oliveira-Filho, E.R.; Gomez, J.G.C.; Taciro, M.K.; Silva, L.F. Burkholderia sacchari (synonym Paraburkholderia sacchari): An industrial and versatile bacterial chassis for sustainable biosynthesis of polyhydroxyalkanoates and other bioproducts. Bioresour. Technol. 2021, 337, 125472. [Google Scholar] [CrossRef] [PubMed]
- Povolo, S.; Romanelli, M.G.; Basaglia, M.; Ilieva, V.I.; Corti, A.; Morelli, A.; Chiellini, E.; Casella, S. Polyhydroxyalkanoate biosynthesis by Hydrogenophaga pseudoflava DSM1034 from structurally unrelated carbon sources. New Biotechnol. 2013, 30, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.U.N.; Ramanaiah, S.V.; Reddy, M.V.; Chang, Y.C. Review of the developments of bacterial medium-chain-length polyhydroxyalkanoates (mcl-PHAs). Bioengineering 2022, 9, 225. [Google Scholar] [CrossRef]
- Lambauer, V.; Kratzer, R. Lab-scale cultivation of Cupriavidus necator on explosive gas mixtures: Carbon dioxide fixation into polyhydroxybutyrate. Bioengineering 2022, 9, 204. [Google Scholar] [CrossRef]
- Afreen, R.; Tyagi, S.; Singh, G.P.; Singh, M. Challenges and perspectives of polyhydroxyalkanoate production from microalgae/cyanobacteria and bacteria as microbial factories: An assessment of hybrid biological system. Front. Bioeng. Biotechnol. 2021, 9, 624885. [Google Scholar] [CrossRef]
- Rodríguez, Y.; Pérez, V.; López, J.C.; Bordel, S.; Firmino, P.I.; Lebrero, R.; Muñoz, R. Coupling biogas with PHA biosynthesis. In The Handbook of Polyhydroxyalkanoates, 1st ed.; Koller, M., Ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2020; Volume 1, Enzymology/Metabolism/Genome Aspects for Microbial PHA Biosynthesis; pp. 357–376. [Google Scholar]
- Do, Y.S.; Smeenk, J.; Broer, K.M.; Kisting, C.J.; Brown, R.; Heindel, T.J.; Bobik, T.A.; DiSpirito, A.A. Growth of Rhodospirillum rubrum on synthesis gas: Conversion of CO to H2 and poly-β-hydroxyalkanoate. Biotechnol. Bioeng. 2007, 97, 279–286. [Google Scholar] [CrossRef]
- Jendrossek, D.; Schirmer, A.; Schlegel, H.G. Biodegradation of polyhydroxyalkanoic acids. Appl. Microbiol. Biotechnol. 1996, 46, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.N.; Lee, A.R.; Yoon, J.S.; Chin, I.J. Biodegradation of poly(3-hydroxybutyrate), Sky-Green® and Mater-Bi® by fungi isolated from soils. Europ. Polym. J. 2008, 36, 1677–1685. [Google Scholar] [CrossRef]
- Vodicka, J.; Wikarska, M.; Trudicova, M.; Juglova, Z.; Pospisilova, A.; Kalina, M.; Slaninova, E.; Obruca, S.; Sedlacek, P. Degradation of P(3HB-co-4HB) films in simulated body fluids. Polymers 2022, 14, 1990. [Google Scholar] [CrossRef]
- Luzier, W.D. Materials derived from biomass/biodegradable materials. Proc. Natl. Acad. Sci. USA 1992, 89, 839–842. [Google Scholar] [CrossRef]
- Rosa, D.S.; Lotto, N.T.; Lopes, D.R.; Guedes, C.G.F. The use of roughness for evaluating the biodegradation of poly-β-(hydroxybutyrate) and poly-β-(hydroxybutyrate-co-β-valerate). Polym. Test 2004, 23, 3–8. [Google Scholar] [CrossRef]
- Available online: http://www.biomer.de/IndexE.html (accessed on 11 April 2023).
- Doi, Y.; Kawaguchi, Y.; Koyama, N.; Nakamura, S.; Hiramitsu, M.; Yoshida, Y.; Kimura, H. Synthesis and degradation of polyhydroxyalkanoates in Alcaligenes eutrophus. FEMS Microbiol. Rev. 1992, 9, 103–108. [Google Scholar] [CrossRef]
- Cinelli, P.; Seggiani, M.; Mallegni, N.; Gigante, V.; Lazzeri, A. Processability and degradability of PHA-based composites in terrestrial environments. Int. J. Mol. Sci. 2019, 20, 284. [Google Scholar] [CrossRef]
- Gómez-Gast, N.; López Cuellar, M.D.R.; Vergara-Porras, B.; Vieyra, H. Biopackaging potential alternatives: Bioplastic composites of polyhydroxyalkanoates and vegetal fibers. Polymers 2022, 14, 1114. [Google Scholar] [CrossRef]
- Meereboer, K.W.; Misra, M.; Mohanty, A.K. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. [Google Scholar] [CrossRef]
- Zhang, K.; Hamidian, A.H.; Tubić, A.; Zhang, Y.; Fang, J.K.; Wu, C.; Lam, P.K. Understanding plastic degradation and microplastic formation in the environment: A review. Environ. Pollut. 2021, 274, 116554. [Google Scholar] [CrossRef]
- Sorensen, R.M.; Jovanović, B. From nanoplastic to microplastic: A bibliometric analysis on the presence of plastic particles in the environment. Mar. Pollut. Bull. 2021, 163, 111926. [Google Scholar] [CrossRef] [PubMed]
- McIlwraith, H.K.; Kim, J.; Helm, P.; Bhavsar, S.P.; Metzger, J.S.; Rochman, C.M. Evidence of microplastic translocation in wild-caught fish and implications for microplastic accumulation dynamics in food webs. Environ. Sci. Technol. 2021, 55, 12372–12382. [Google Scholar] [CrossRef]
- Suzuki, G.; Uchida, N.; Tanaka, K.; Matsukami, H.; Kunisue, T.; Takahashi, S.; Viet, P.H.; Kuramochi, H.; Osako, M. Mechanical recycling of plastic waste as a point source of microplastic pollution. Environ. Pollut. 2022, 303, 119114. [Google Scholar] [CrossRef]
- Barrett, A. Frost & Sullivan Awards Bio-On for Best Cosmetic Innovation. 2018. Available online: https://bioplasticsnews.com/2018/10/08/frost-sullivan-awards-bio-best-cosmetic-innovation/ (accessed on 6 March 2023).
- TianAn Biopolymer: Nature’s Eco-Friendly Solution. Available online: Tianan-enmat.com (accessed on 12 December 2022).
- Corradini, F.; Meza, P.; Eguiluz, R.; Casado, F.; Huerta-Lwanga, E.; Geissen, V. Evidence of microplastic accumulation in agricultural soils from sewage sludge disposal. Sci. Total Environ. 2019, 671, 411–420. [Google Scholar] [CrossRef]
- Available online: https://www.nafigate.com/portfolio/naturetics/ (accessed on 7 April 2023).
- Braunegg, G.; Lefebvre, G.; Genser, K.F. Polyhydroxyalkanoates, biopolyesters from renewable resources: Physiological and engineering aspects. J. Biotechnol. 1998, 65, 127–161. [Google Scholar] [CrossRef]
- Koller, M. A review on established and emerging fermentation schemes for microbial production of polyhydroxyalkanoate (PHA) biopolyesters. Fermentation 2018, 4, 30. [Google Scholar] [CrossRef]
- Reed, G.; Nagodawithana, T.W. Baker’s yeast production. In Yeast Technology; Springer: Dordrecht, The Netherlands, 1990; pp. 261–314. [Google Scholar] [CrossRef]
- Hailu, S.; Admassu, S.; Jha, K. Vinegar production technology—An overview. Beverage Food World 2012, 2, 29–32. [Google Scholar]
- Bajpai, R.K.; Reuss, M. A mechanistic model for penicillin production. J. Chem. Technol. Biotechnol. 1980, 30, 332–344. [Google Scholar] [CrossRef]
- Angumeenal, A.R.; Venkappayya, D. An overview of citric acid production. LWT-Food Sci. Technol. 2013, 50, 367–370. [Google Scholar] [CrossRef]
- Koller, M. Established and advanced approaches for recovery of microbial polyhydroxyalkanoate (PHA) biopolyesters from surrounding microbial biomass. EuroBiotech J. 2020, 4, 113–126. [Google Scholar] [CrossRef]
- Puppi, D.; Chiellini, F. Additive manufacturing of PHA. In The Handbook of Polyhydroxyalkanoates, 1st ed.; Koller, M., Ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2020; Volume 3: Postsynthetic Treatment, Processing and Application, pp. 119–136. [Google Scholar]
- Gigante, V.; Cinelli, P.; Seggiani, M.; Alavarez, V.A.; Lazzeri, A. Processing and thermomechanical properties of PHA. In The Handbook of Polyhydroxyalkanoates; Taylor & Francis: Boca Raton, FL, USA, 2020; pp. 91–118. [Google Scholar]
- Peng, X.; Dong, K.; Wu, Z.; Wang, J.; Wang, Z.L. A review on emerging biodegradable polymers for environmentally benign transient electronic skins. J. Mater. Sci. 2021, 56, 16765–16789. [Google Scholar] [CrossRef]
- Mukherjee, A.; Koller, M. Polyhydroxyalkanoate (PHA) bio-polyesters—Circular materials for sustainable development and growth. Chem. Biochem. Eng. Q. 2022, 36, 273–293. [Google Scholar]
- Brigham, C.J.; Sinskey, A.J. Applications of polyhydroxyalkanoates in the medical industry. Int. J. Biotechnol. Wellness Ind. 2012, 1, 52. [Google Scholar] [CrossRef]
- Hänggi, U. Virgin PHB has Thermoplastic Properties, But Is not a Thermoplast. Presentation at the PHA Congress 2018 in Cologne. 2018. Available online: http://www.biomer.de/K%C3%96LN2018.pdf (accessed on 15 September 2021).
- Koller, M.; Mukherjee, A. A new wave of industrialization of PHA biopolyesters. Bioengineering 2022, 9, 74. [Google Scholar] [CrossRef]
| Material: | PHA | Carbohydrates (Starch, Cellulose, Alginates, Chitin, etc.) | Proteins | PLA | PCL | Bio-PE | Fossil Plastics (PE, PP, PVC, PET, etc.) |
|---|---|---|---|---|---|---|---|
| Criterion: | |||||||
| Production depletes fossil resources | NO | NO | NO | NO | YES | NO | YES |
| Biobased (produced from renewable natural resources or via photosynthesis) | YES | YES | YES | YES | NO | YES | NO |
| Biosynthesized (polymerization taking place in nature by nature´s enzymatic toolbox) | YES | YES | YES | NO | NO | NO | NO |
| Biodegradable (degradation in nature by enzymes present in living organisms) | YES | YES | YES | YES | YES | NO | NO |
| Biocompatible (not harmful to the biosphere) | YES | YES | YES | NO | YES | NO | NO |
| (Natural) chemical structure after (bio)synthesis can be maintained during processing | YES | YES | YES | YES | YES | YES | YES |
| Formation of persistent microparticles | NO | NO | NO | NO | NO | YES | YES |
| Incineration generates surplus atmospheric CO2, thus fueling global warming | NO | NO | NO | NO | YES | NO | YES |
| Summary: Natural polymer or not?? | YES | YES | YES | NO | NO | NO | NO |
| Principle | Description | Relevant to PHAs? | Explanation of the Relevance |
|---|---|---|---|
| Principle 1: Prevention | “It is better to prevent waste than to treat or clean up waste after it has been created.” | YES | Neither PHA biosynthesis nor PHA biodegradation generates any waste which needs to be treated, in contrast to production and disintegration of fossil plastics. This is especially true for the formation of microplastic particles, which, in the case of fossil plastics, are recalcitrant and endanger the eco- and biosphere, while micro-sized PHA particles undergo biodegradation. |
| Principle 2: Atom Economy | “Synthetic methods should be designed to maximize incorporation of all materials used in the process into the final product.” | - | Not applicable in the context of PHA. As natural, aerobic process, PHA biosynthesis creates CO2 as side product; however, generated CO2 is embedded into the natural carbon cycle, in contrast to CO2 stemming from petrochemical products. |
| Principle 3: Less Hazardous Chemical Syntheses | “Wherever practicable, synthetic methods should be designed to use and generate substances that possess little or no toxicity to human health and the environment.” | YES | PHA biopolyesters are highly biocompatible; they do not exert any risk to the environment or human health. |
| Principle 4: Designing Safer Chemicals | “Chemical products should be designed to preserve efficacy of function while reducing toxicity.” | YES | PHA biopolyesters can function as a replacement for plastics, but, in contrast to fossil plastics, do not generate any toxic compounds when disposed or recycled. |
| Principle 5: Safer Solvents and Auxiliaries | “The use of auxiliary substances (e.g., solvents, separation agents, etc.) should be made unnecessary wherever possible and, innocuous when used.” | YES | PHA biosynthesis occurs in the aqueous phase; hence, water, being the most sustainable and innocuous solvent, is used. For PHA recovery from biomass, ecologically benign “green solvents” of natural origin can be used. |
| Principle 6: Design for Energy Efficiency | “Energy requirements should be recognized for their environmental and economic impacts and should be minimized. Synthetic methods should be conducted at ambient temperature and pressure.” | YES | PHA biosynthesis typically occurs at room temperature and under ambient pressure conditions, which makes it a process of low energy requirements. Required energy supply is mainly due to upstream processing (e.g., sterilization of the bioreactor and media compounds), aerating, and stirring of the bioreactor, and downstream processing. In the case of using extremophilic organisms, energy for sterilization can even be avoided. |
| Principle 7: Use of Renewable Feedstocks | “A raw material or feedstock should be renewable rather than depleting whenever technically and economically practicable.” | YES | PHAs originate from renewable feedstocks. |
| Principle 8: Reduce Derivatives | “Unnecessary derivatization (use of blocking groups, protection/deprotection, temporary modification of physical/chemical processes) should be minimized or avoided if possible, because such steps require additional reagents and can generate waste.” | YES | Derivatization is not needed during PHA biosynthesis due to the efficiently coordinated enzymatic sequence in the intracellular biocatalytic cascade. |
| Principle 9: Catalysis | “Catalytic reagents (as selective as possible) are superior to stoichiometric reagents.” | YES | A highly efficient enzymatic cascade, starting from the biocatalysts responsible for substrate catabolism towards acetyl-CoA, propionyl-CoA, etc. (glycolysis, KDPG pathway, β-oxidation, oxidative pyruvate decarboxylation, etc.), via enzymes generating the PHA building blocks (3-ketothiolase and reductase) until PHA synthases catalyze PHA biosynthesis. Activities of enzymes involved in the entire pathway from substrate to bioproduct (PHA) are highly specific, and efficiently coordinated. |
| Principle 10: Design for Degradation | “Chemical products should be designed so that at the end of their function they break down into innocuous degradation products and do not persist in the environment.” | YES | Products made of PHAs degrade naturally after use, without harming the environment, into exactly those innocuous compounds they derive from: water and CO2 under aerobic conditions (composting), plus methane under anaerobic conditions (biogas plants). |
| Principle 11: Real-time analysis for Pollution Prevention | “Analytical methodologies need to be further developed to allow for real-time, in-process monitoring and control prior to the formation of hazardous substances.” | YES | Modern bioreactor equipment used for PHA production encompasses online analytical tools to monitor temperature, pH-value, dissolved oxygen concentration, redox potential, foam formation (via conductivity sensors), substrate consumption (e.g., glucose sensors), biomass formation (via turbidity sensors), and the CO2 level and remaining oxygen in exhaust gas. These parameters are processed via high-level digitalization, which control the process in real time by adjusting substrate supply, oxygen input, temperature, pH-value, etc. |
| Principle 12: Inherently Safer Chemistry for Accident Prevention | “Substances and the form of a substance used in a chemical process should be chosen to minimize the potential for chemical accidents, including releases, explosions, and fires.” | YES | PHA production is based on safe, renewable feedstocks. No risk of fire or explosion exists during PHA production, and no toxins are released during the process. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mukherjee, A.; Koller, M. Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural. Bioengineering 2023, 10, 855. https://doi.org/10.3390/bioengineering10070855
Mukherjee A, Koller M. Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural. Bioengineering. 2023; 10(7):855. https://doi.org/10.3390/bioengineering10070855
Chicago/Turabian StyleMukherjee, Anindya, and Martin Koller. 2023. "Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural" Bioengineering 10, no. 7: 855. https://doi.org/10.3390/bioengineering10070855
APA StyleMukherjee, A., & Koller, M. (2023). Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural. Bioengineering, 10(7), 855. https://doi.org/10.3390/bioengineering10070855








