In-Depth Bicycle Collision Reconstruction: From a Crash Helmet to Brain Injury Evaluation
Abstract
1. Introduction
2. Methods
2.1. Background of the Accident
2.2. Analysis of the Helmet Damage
2.3. Helmet Impact Testing
2.4. Finite Element Modelling
2.5. Brain Injury Metrics and Evaluation
3. Results
3.1. Helmet Damage Profile and Hypothesised Accident Scenario
3.1.1. The Severe Impact
3.1.2. The Moderate Impact
3.1.3. Mild Impacts
3.2. Helmet Impact Testing Results
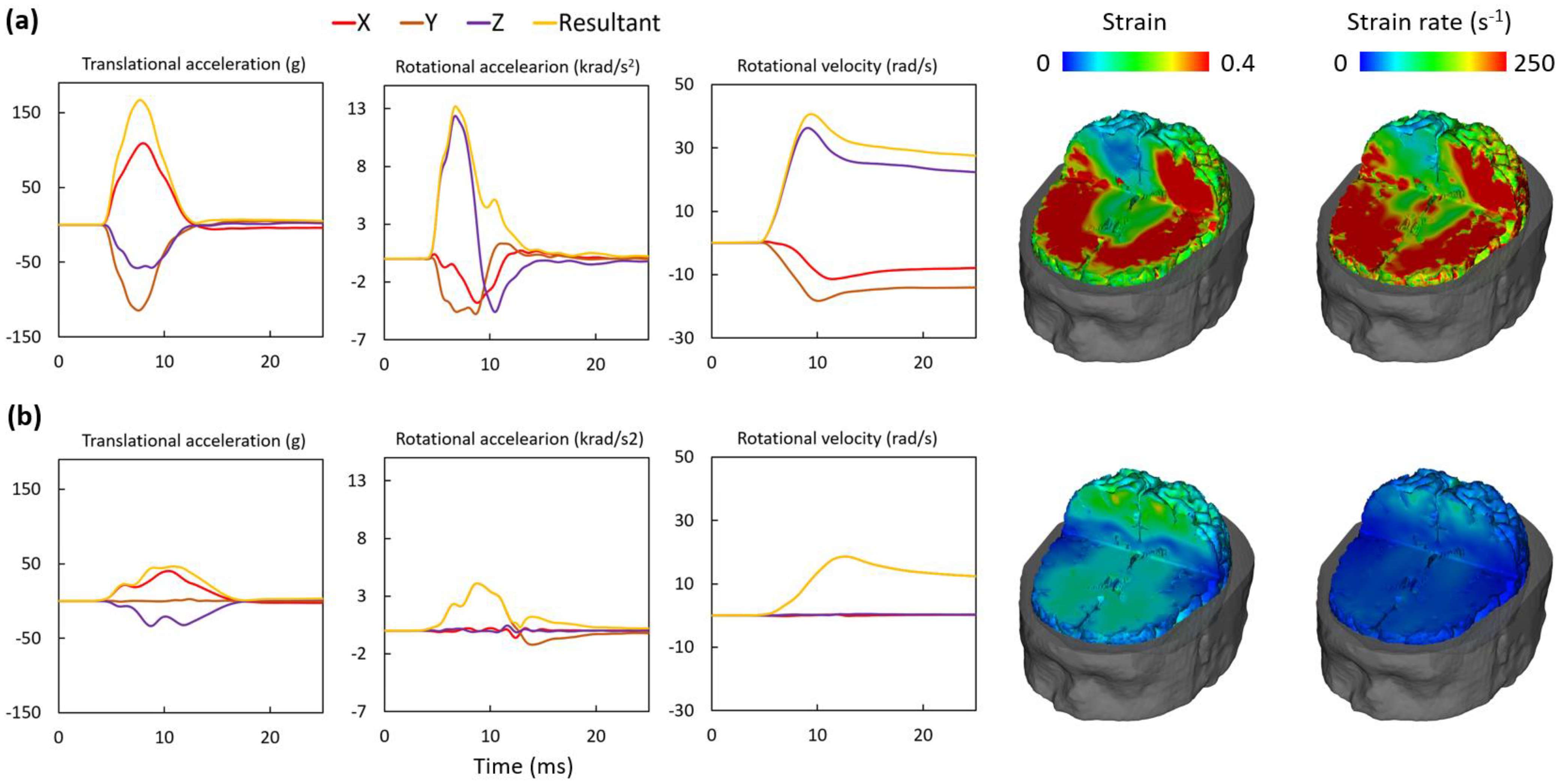
3.2.1. Reconstruction of the Severe Impact
3.2.2. Reconstruction of the Moderate Impact
3.3. Brain Injury Metrics and Injury Evaluation
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The Impact of the Coronavirus Pandemic on Walking and Cycling Statistics, England. 2020. Available online: https://www.gov.uk/government/statistics/walking-and-cycling-statistics-england-2020/the-impact-of-the-coronavirus-pandemic-on-walking-and-cycling-statistics-england-2020 (accessed on 1 January 2023).
- Murphy, A. Reported Road Casualties in Great Britain: 2020 Annual Report; Department of Transport: London, UK, 2021.
- Thompson, D.C.; Rivara, F.; Thompson, R. Helmets for Preventing Head and Facial Injuries in Bicyclists; Cochrane Database of Systematic Reviews; Cochrane: London, UK, 1999. [Google Scholar]
- Baker, C.E.; Martin, P.; Wilson, M.H.; Ghajari, M.; Sharp, D.J. The Relationship between Road Traffic Collision Dynamics and Traumatic Brain Injury Pathology; Brain Communications: Oxford, UK, 2022. [Google Scholar]
- Mesfin, F.B.; Gupta, N.; Taylor, R. Diffuse Axonal Injury; National Institutes of Health: Bethesda, MD, USA, 2017.
- Dodds, N.; Johnson, R.; Walton, B.; Bouamra, O.; Yates, D.; Lecky, F.E.; Thompson, J. Evaluating the impact of cycle helmet use on severe traumatic brain injury and death in a national cohort of over 11000 pedal cyclists: A retrospective study from the NHS England Trauma Audit and Research Network dataset. BMJ Open 2019, 9, e027845. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, R.W.; Daphalapurkar, N.P. The importance of structural anisotropy in computational models of traumatic brain injury. Front. Neurol. 2015, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, D.; Deck, C.; Willinger, R. Brain injury tolerance limit based on computation of axonal strain. Accid. Anal. Prev. 2016, 92, 53–70. [Google Scholar] [CrossRef] [PubMed]
- Tenovuo, O.; Diaz-Arrastia, R.; Goldstein, L.E.; Sharp, D.J.; van der Naalt, J.; Zasler, N.D. Assessing the severity of traumatic brain injury—Time for a change? J. Clin. Med. 2021, 10, 148. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee on the Review of the Department of Veterans Affairs Examinations for Traumatic Brain Injury. Evaluation of the Disability Determination Process for Traumatic Brain Injury in Veterans; National Institutes of Health: Bethesda, MD, USA, 2019.
- Freeman, L.C.; Ting, J.P.Y. The pathogenic role of the inflammasome in neurodegenerative diseases. J. Neurochem. 2016, 136, 29–38. [Google Scholar] [CrossRef]
- Gardner, R.C.; Burke, J.F.; Nettiksimmons, J.; Kaup, A.; Barnes, D.E.; Yaffe, K. Dementia risk after traumatic brain injury vs. nonbrain trauma: The role of age and severity. JAMA Neurol. 2014, 71, 1490–1497. [Google Scholar] [CrossRef]
- Smith, D.H.; Johnson, V.E.; Stewart, W. Chronic neuropathologies of single and repetitive TBI: Substrates of dementia? Nat. Rev. Neurol. 2013, 9, 211–221. [Google Scholar] [CrossRef]
- Vincent, A.S.; Roebuck-Spencer, T.M.; Cernich, A. Cognitive changes and dementia risk after traumatic brain injury: Implications for aging military personnel. Alzheimer’s Dement. 2014, 10, S174–S187. [Google Scholar] [CrossRef]
- Bourdet, N.; Deck, C.; Tinard, V.; Willinger, R.M. Behaviour of helmets during head impact in real accident cases of motorcyclists. Int. J. Crashworthiness 2012, 17, 51–61. [Google Scholar] [CrossRef]
- Carter, E.; Neal-Sturgess, C.E. Madymo reconstruction of a real-world collision between a vehicle and cyclist. Int. J. Crashworthiness 2009, 14, 379–390. [Google Scholar] [CrossRef]
- Bourdet, N.; Deck, C.; Serre, T.; Perrin, C.; Llari, M.; Willinger, R. In-depth real-world bicycle accident reconstructions. Int. J. Crashworthiness 2014, 19, 222–232. [Google Scholar] [CrossRef]
- Zimmerman, K.; Kim, J.; Karton, C.; Lochhead, L.; Sharp, D.; Hoshizaki, T.; Ghajari, M. Player position in American football influences the magnitude of mechanical strains produced in the location of chronic traumatic encephalopathy pathology: A computational modelling study. J. Biomech. 2021, 118, 110256. [Google Scholar] [CrossRef] [PubMed]
- Snedeker, J.G.; Walz, F.H.; Muser, M.H.; Lanz, C.; Schroeder, G. Assessing femur and pelvis injury risk in car-pedestrian collisions: Comparison of full body PMTO impacts, and a human body finite element model. In Proceedings of the 19th International Technical Conference on the Enhanced Safety of Vehicles (ESV), Washington, DC, USA, 6–9 June 2005. [Google Scholar]
- McIntosh, A.S.; A Patton, D. Impact reconstruction from damage to pedal and motorcycle helmets. Proc. Inst. Mech. Eng. Part P J. Sports Eng. Technol. 2012, 226, 274–281. [Google Scholar] [CrossRef]
- Bland, M.L.; McNally, C.; Cicchino, J.B.; Zuby, D.S.; Mueller, B.C.; McCarthy, M.L.; Newgard, C.D.; Kulie, P.E.; Arnold, B.N.; Rowson, S. Laboratory reconstructions of bicycle helmet damage: Investigation of cyclist head impacts using oblique impacts and computed tomography. Ann. Biomed. Eng. 2020, 48, 2783–2795. [Google Scholar] [CrossRef] [PubMed]
- Williams, M. The protective performance of bicyclists’ helmets in accidents. Accid. Anal. Prev. 1991, 23, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.A.; Tees, D.; Thom, D.R.; Hurt, H.H., Jr. Evaluation and replication of impact damage to bicycle helmets. Accid. Anal. Prev. 1994, 26, 795–802. [Google Scholar] [CrossRef]
- Yu, X.; Logan, I.; Sarasola, I.d.P.; Dasaratha, A.; Ghajari, M. The Protective Performance of Modern Motorcycle Helmets Under Oblique Impacts. Ann. Biomed. Eng. 2022, 50, 1674–1688. [Google Scholar] [CrossRef]
- Ghajari, M.; Hellyer, P.J.; Sharp, D.J. Computational modelling of traumatic brain injury predicts the location of chronic traumatic encephalopathy pathology. Brain 2017, 140, 333–343. [Google Scholar] [CrossRef]
- Yu, X.; Azor, A.; Sharp, D.J.; Mazdak, G. Mechanisms of tensile failure of cerebrospinal fluid in blast traumatic brain injury. Extrem. Mech. Lett. 2020, 38, 100739. [Google Scholar] [CrossRef]
- Yu, X.; Nguyen, T.-T.; Wu, T.; Ghajari, M. Non-Lethal Blasts Can Generate Cavitation in Cerebrospinal Fluid While Severe Helmeted Impacts Cannot: A Novel Mechanism for Blast Brain Injury; Frontiers in Bioengineering and Biotechnology: Lausanne, Switzerland, 2022; Volume 10. [Google Scholar]
- LS-DYNA Keyword User’s Manual, Version 971; Livermore Software Technology Corporation: Livermore, CA, USA, 2007; Volume 7374, p. 354.
- Yu, X.; Ghajari, M. An Assessment of Blast Modelling Techniques for Injury Biomechanics Research. Int. J. Numer. Methods Biomed. Eng. 2019, 35, e3258. [Google Scholar] [CrossRef]
- Allsop, D.L.; Warner, C.Y.; Wille, M.G.; Schneider, D.C.; Nahum, A.M. Facial Impact Response—A Comparison of the Hybrid III Dummy and Human Cadaver; SAE Transactions: New York, NY, USA, 1988; pp. 1224–1240. [Google Scholar]
- Gurdjian, E.S.; Roberts, V.; Thomas, L.M. Tolerance curves of acceleration and intracranial pressure and protective index in experimental head injury. J. Trauma Acute Care Surg. 1966, 6, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Depreitere, B.; Van Lierde, C.; Vander Sloten, J.; Van Audekercke, R.; Van der Perre, G.; Plets, C.; Goffin, J. Mechanics of Acute Subdural Hematomas Resulting From Bridging Vein Rupture. J. Neurosurg. 2006, 104, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Gennarelli, T.A.; Thibault, L.E. Biomechanics of acute subdural hematoma. J. Trauma 1982, 22, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Margulies, S.S.; Thibault, L.E. A proposed tolerance criterion for diffuse axonal injury in man. J. Biomech. 1992, 25, 917–923. [Google Scholar] [CrossRef]
- Bain, A.C.; Meaney, D.F. Tissue-level thresholds for axonal damage in an experimental model of central nervous system white matter injury. J. Biomech. Eng. 2000, 122, 615–622. [Google Scholar] [CrossRef]
- Yu, X.; Ghajari, M. Protective Performance of Helmets and Goggles in Mitigating Brain Biomechanical Response to Primary Blast Exposure. Ann. Biomed. Eng. 2022, 50, 1579–1595. [Google Scholar] [CrossRef]
- Yu, X.; Halldin, P.; Ghajari, M. Oblique Impact Responses of Hybrid III and a New Headform with More Biofidelic Coefficient of Friction and Moments of Inertia; Frontiers in Bioengineering and Biotechnology: Lausanne, Switzerland, 2022; p. 1620. [Google Scholar]
- Abayazid, F.; Ding, K.; Zimmerman, K.; Stigson, H.; Ghajari, M. A New Assessment of Bicycle Helmets: The Brain Injury Mitigation Effects of New Technologies in Oblique Impacts. Ann. Biomed. Eng. 2021, 49, 2716–2733. [Google Scholar] [CrossRef]
- Whyte, T.; Stuart, C.; Mallory, A.; Ghajari, M.; Plant, D.; Siegmund, G.P.; Cripton, P.A. A review of impact testing methods for headgear in sports: Considerations for improved prevention of head injury through research and standards. J. Biomech. Eng. 2019, 141. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, K.H.; King, A.I. A proposed injury threshold for mild traumatic brain injury. J. Biomech. Eng. 2004, 126, 226–236. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, K.H.; King, A.I.; Viano, D.C. A New Biomechanical Predictor for Mild Traumatic Brain Injury—A Preliminary Finding; Proceedings Summer Bioengineering Conference: Key Biscayne, FL, USA, 2003; pp. 137–138. [Google Scholar]
- Chinn, B.; Canaple, B.; Derler, S.; Doyle, D.; Otte, D.; Schuller, E.; Willinger, R. COST 327 Motorcycle Safety Helmets; European Commission, Directorate General for Energy and Transport: Brussels, Belgium, 2001. [Google Scholar]
- Kinnunen, K.M.; Greenwood, R.; Powell, J.H.; Leech, R.; Hawkins, P.C.; Bonnelle, V.; Patel, M.C.; Counsell, S.J.; Sharp, D.J. White matter damage and cognitive impairment after traumatic brain injury. Brain 2011, 134, 449–463. [Google Scholar] [CrossRef]
- Donat, C.K.; Yanez Lopez, M.; Sastre, M.; Baxan, N.; Goldfinger, M.; Seeamber, R.; Müller, F.; Davies, P.; Hellyer, P.; Siegkas, P. From biomechanics to pathology: Predicting axonal injury from patterns of strain after traumatic brain injury. Brain 2021, 144, 70–91. [Google Scholar] [CrossRef]
- McAllister, T.W.; Ford, J.C.; Ji, S.; Beckwith, J.G.; Flashman, L.A.; Paulsen, K.; Greenwald, R.M. Maximum principal strain and strain rate associated with concussion diagnosis correlates with changes in corpus callosum white matter indices. Ann. Biomed. Eng. 2012, 40, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Kleiven, S. Predictors for Traumatic Brain Injuries Evaluated through Accident Reconstructions; SAE Technical Paper: San Diego, CA, USA, 2007. [Google Scholar]
- Sharp, D.J.; Ham, T.E. Investigating white matter injury after mild traumatic brain injury. Curr. Opin. Neurol. 2011, 24, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, G.; Maas, A.; Lecky, F.; Manley, G.; Stocchetti, N.; Murray, G. The Glasgow Coma Scale at 40 years: Standing the test of time. Lancet Neurol. 2014, 13, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Juste-Lorente, Ó.; Maza, M.; Piccand, M.; López-Valdés, F.J. The Influence of Headform/Helmet Friction on Head Impact Biomechanics in Oblique Impacts at Different Tangential Velocities. Appl. Sci. 2021, 11, 11318. [Google Scholar] [CrossRef]
- Mills, N.; Gilchrist, A. Oblique impact testing of bicycle helmets. Int. J. Impact Eng. 2008, 35, 1075–1086. [Google Scholar] [CrossRef]
- Bourdet, N.; Deck, C.; Carreira, R.P.; Willinger, R. Head impact conditions in the case of cyclist falls. Proc. Inst. Mech. Eng. Part P J. Sport. Eng. Technol. 2012, 226, 282–289. [Google Scholar] [CrossRef]
- Harlos, A.R.; Rowson, S. Laboratory reconstructions of real-world bicycle helmet impacts. Ann. Biomed. Eng. 2021, 49, 2827–2835. [Google Scholar] [CrossRef]
- Bonin, S.J.; Luck, J.; Bass, C.; Gardiner, J.; Onar-Thomas, A.; Asfour, S.S.; Siegmund, G. Dynamic response and residual helmet liner crush using cadaver heads and standard headforms. Ann. Biomed. Eng. 2017, 45, 656–667. [Google Scholar] [CrossRef]
- FIM. FIM Racing Homologation Programme for Helmets; FIM: Mies, Switzerland, 2016. [Google Scholar]
- Uniform Provisions Concerning the Approval of Protective Helmets and of Their Visors for Drivers and Passengers of Motor Cycles and Mopeds.ECE/TRANS/WP.29/2020/60. 2020. Available online: https://unece.org/2020-16 (accessed on 1 January 2023).
- Aare, M.; Halldin, P. A new laboratory rig for evaluating helmets subject to oblique impacts. Traffic Inj. Prev. 2003, 4, 240–248. [Google Scholar] [CrossRef]
- Bliven, E.; Rouhier, A.; Tsai, S.; Willinger, R.; Bourdet, N.; Deck, C.; Madey, S.M.; Bottlang, M. Evaluation of a novel bicycle helmet concept in oblique impact testing. Accid. Anal. Prev. 2019, 124, 58–65. [Google Scholar] [CrossRef]
- Trotta, A.; Annaidh, A.N.; Burek, R.O.; Pelgrims, B.; Ivens, J. Evaluation of the head-helmet sliding properties in an impact test. J. Biomech. 2018, 75, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Trotta, A.; Zouzias, D.; De Bruyne, G.; Annaidh, A.N. The importance of the scalp in head impact kinematics. Ann. Biomed. Eng. 2018, 46, 831–840. [Google Scholar] [CrossRef]
- Connor, T.A.; Stewart, M.; Burek, R.; Gilchrist, M.D. Influence of headform mass and inertia on the response to oblique impacts. Int. J. Crashworthiness 2018, 24, 677–698. [Google Scholar] [CrossRef]
- Halldin, P.; Gilchrist, A.; Mills, N. A new oblique impact test for motorcycle helmets. Int. J. Crashworthiness 2001, 6, 53–64. [Google Scholar] [CrossRef]
- Pintar, F.A.; Sances, A.; Yoganandan, N.; Reinartz, J.; Maiman, D.J.; Suh, J.K.; Unger, G.; Cusick, J.F.; Larson, S.J. Biodynamics of the Total Human Cadaveric Cervical Spine; 0148-7191; SAE Technical Paper: Orlando, FL, USA, 1990. [Google Scholar]
- Ghajari, M.; Galvanetto, U.; Iannucci, L.; Willinger, R. The Influence of the Body on the Response of the Helmeted Head during Impact. Int. J. Crashworthiness 2011, 16, 285–295. [Google Scholar] [CrossRef]
- Ghajari, M.; Peldschus, S.; Galvanetto, U.; Iannucci, L. Effects of the presence of the body in helmet oblique impacts. Accid. Anal. Prev. 2013, 50, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, R.W.; McElhaney, J.H.; Richardson, W.J.; Myers, B.S. Dynamic responses of the head and cervical spine to axial impact loading. J. Biomech. 1996, 29, 307–318. [Google Scholar] [CrossRef]
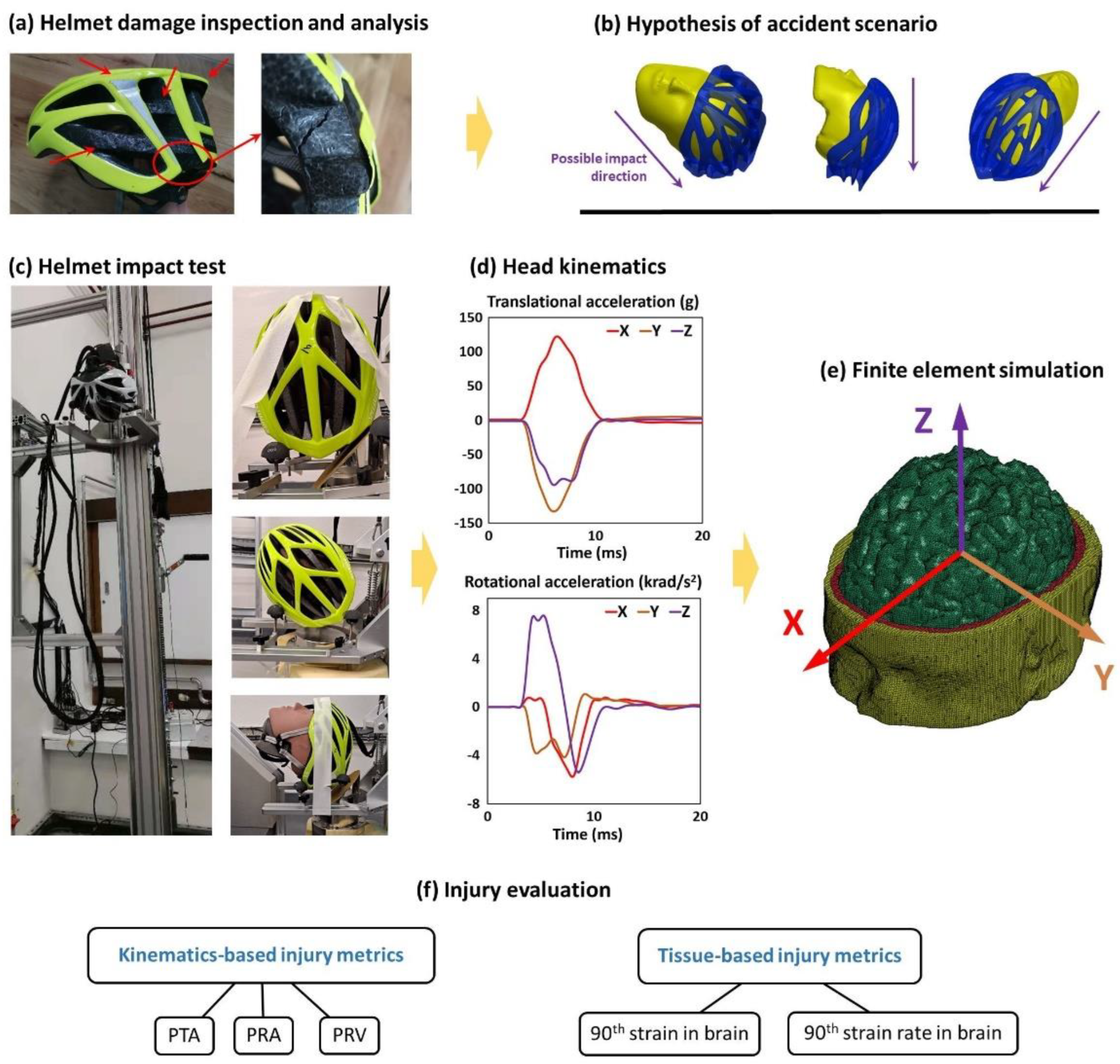




| Cause | Damages | Location | Details |
|---|---|---|---|
| Severe impact | Crushed surface | Rear left | 1. 75 mm in diameter with small pits, produced by road aggregates compression 2. Crush depth of the flattened surface is 4.9 mm 3. Delamination between shell and liner |
| Crack trajectory | Rear left | Five major cracks | |
| Localised shell buckling | Rear left-top | Localised shell buckling with scrapes | |
| Moderate impact | Crushed rear end | Rear top | 1. Crush depth is 6 mm 2. Delamination between shell and the liner |
| Mild impacts | Delamination | Rear right | 1. Delamination between shell and liner 2. No obvious crush of liner |
| Localised dents | Rear | Small pits on the EPS form and shell |
| Test No | Anvil | Velocity (m/s) | Crush Depth of Flatten Area (mm) | Recreated Cracks in Tested Helmet * | |||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | No. of Additional Cracks | ||||
| 1 | Flat | 5.8 | 6.2 | ● | × | ● | × | × | 2 |
| 2 | Flat | 6.3 | 6.3 | ● | × | ● | ● | × | 2 |
| 3 | Flat | 6.9 | 7 | ● | × | ● | ● | ● | 2 |
| 4 | Oblique | 6.4 | 2.7 | ● | × | ● | × | × | 0 |
| 5 | Oblique | 6.8 | 6.1 | ● | × | ● | ● | ● | 1 |
| 6 | Oblique | 8 | 3 | ● | × | × | × | × | 3 |
| 7 | Oblique | 8 | 3.5 | ● | ● | ● | ● | × | 2 |
| 8 | Oblique | 7.3 | 5.4 | ● | ● | ● | ● | ● | 2 |
| 9 | Oblique | 7.3 | 5.2 | ● | ● | ● | ● | ● | 0 |
| 10 | Oblique | 7.3 | 5.6 | ● | ● | ● | ● | ● | 2 |
| No | Anvil | Velocity (m/s) | Results |
|---|---|---|---|
| 11 | Oblique | 8 | Severe crushing at the rear upper point, which indicates much higher kinematic energy than in accident |
| 12 | Oblique | 3.2 | 5 mm crushing, which is close to the 6 mm crushing in the cyclist’s helmet |
| Test No. | PTA (g) | PRA (rad/s2) | PRV (rad/s) | 90th Percentile Brain Strain | 90th Percentile Brain Strain Rate (s−1) |
|---|---|---|---|---|---|
| 9 | 167 | 13,206 | 40.7 | 0.541 | 415 |
| 12 | 46 | 4102 | 18.6 | 0.045 | 178 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, X.; Baker, C.E.; Brown, M.; Ghajari, M. In-Depth Bicycle Collision Reconstruction: From a Crash Helmet to Brain Injury Evaluation. Bioengineering 2023, 10, 317. https://doi.org/10.3390/bioengineering10030317
Yu X, Baker CE, Brown M, Ghajari M. In-Depth Bicycle Collision Reconstruction: From a Crash Helmet to Brain Injury Evaluation. Bioengineering. 2023; 10(3):317. https://doi.org/10.3390/bioengineering10030317
Chicago/Turabian StyleYu, Xiancheng, Claire E. Baker, Mike Brown, and Mazdak Ghajari. 2023. "In-Depth Bicycle Collision Reconstruction: From a Crash Helmet to Brain Injury Evaluation" Bioengineering 10, no. 3: 317. https://doi.org/10.3390/bioengineering10030317
APA StyleYu, X., Baker, C. E., Brown, M., & Ghajari, M. (2023). In-Depth Bicycle Collision Reconstruction: From a Crash Helmet to Brain Injury Evaluation. Bioengineering, 10(3), 317. https://doi.org/10.3390/bioengineering10030317





