Abstract
Assisted reproductive technology (ART) is used to enhance pregnancy in infertile women. In this technique, the eggs are removed from the ovary and fertilized and injected with sperm to make embryos. Unfortunately, embryo implantation failures still occur in many of these women. Platelet-rich plasma (PRP) therapies use a patient’s own platelets to promote tissue healing and growth, including endometrium. The growth factors provided by the platelets play a criterial role on the regenerative ability of PRP. In the last years, PRP treatments have been gaining a lot of popularity to treat women with repeated ART failures. In this study, we collected and summarized all information published in the scientific literature to assess the evidence of the PRP effect on pregnancy. We only considered randomized controlled trials (RCT), a type of study designed to be unbiased and considered at the highest level of evidence. Our analysis indicates that PRP therapies might be an effective treatment in cases of poor responsiveness to conventional ART. However, additional studies (well-designed) are necessary to confirm this beneficial effect of PRP.
1. Introduction
Around 10–15% of couples suffer from infertility or have difficulties conceiving [1,2]. The introduction of assisted reproductive technology (ART) has had a favorable impact on the pregnancy rate in infertile women. However, implantation failures still occur in a significant percentage of these patients. Recurrent implantation failure (RIF) can be defined as the absence of a positive pregnancy after multiple/repeated good-quality embryo transfers in women under the age of 40. Implantation failure is diagnosed by embryo failure to progress to a stage where intrauterine gestational sac is ultrasonography identified [3,4]. However, there is no consensus on the definition for such condition [5]. RIF is associated with a variety of factors, such as maternal age, lifestyle, obesity, genetic disorders or immunological conditions [6].
Endometrial receptivity is considered one of the most critical prognostic markers in the success of a pregnancy following embryo transfer [7]. Endometrial thickness of <7 mm has been linked to poor pregnancy outcomes, RIF or high rate of cycle cancelation rate [8,9,10,11,12]. Recent studies suggest that the embryonic implantation process is highly dependent on interleukins, growth factors or cytokines among others [13,14,15]. Recently, different therapies have been assessed to treat infertility such as estradiol hormonal supplementation [16], low-dose aspirin [17,18], vitamin E [19], vaginal sildenafil [20,21,22], pentoxifylline [19], tamoxifen [23] and stem cell therapy [24]. In spite of these therapeutic alternatives, many patients still are suffering from implantation failure, especially women with a thin endometrium (TE). Therefore, there is a need for new therapeutic approaches to the treatment of women with poor responsiveness to conventional ART [25,26].
Autologous platelet concentrates have emerged as an alternative to promoting endometrium receptivity and enhancing pregnancy outcomes. Of the available formulations, platelet-rich plasma (PRP) has been applied in multiple medical fields including oral and maxillofacial surgery [27], dermatology [28], traumatology [29], ophthalmology [30], and, most recently, reproductive medicine. PRP is prepared by gradient density centrifugation of blood to obtain a concentrated platelets in plasma. Following platelet activation, platelets granules release their contents.
In the last years, several controlled trials have been published, evaluating the efficacy of intrauterine PRP infusion over nonintervention therapy. However, whether PRP is an effective treatment in women with RIF or TE remains controversial. Some studies described effects of PRP that were generally positive in terms of endometrial thickness and pregnancy outcomes, but no meta-analysis that specifically evaluates randomized clinical trials (RCT) has been conducted so far [11,31,32]. According to in vitro data, the beneficial effects of PRP on endometrial cells have enhanced their response (proliferation, migration and differentiation) as well as an angiogenesis [33,34]. Interestingly, favorable outcomes have also been reported in patients with low ovarian reserve [35], premature ovarian insufficiency [36] and post-menopausal women [36] after intraovarian PRP administration. By contrast, some researchers observed no benefit for embryo implantation, pregnancy rate or live birth rate when PRP was administered in the uterus [37,38]. The aim of our study was to investigate whether intrauterine PRP infusion can improve embryo implantation and its potential for enhancing pregnancy outcomes in women with a history of ART failure, through an analysis of the available RCTs.
2. Materials and Methods
2.1. Protocol Registration and Reporting Format
This systematic review with meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [39]. The protocol was registered and assigned in the PROSPERO database (CRD42021287963).
2.2. Focus Question
This review aimed to answer the following question:
Does intrauterine infusion of PRP improve pregnancy outcomes in women with a history of embryo transfer failure?
2.3. PICO Strategy
The following strategy was constructed according to PICO study design:
- The participants (P) included were women undergoing assisted reproduction with a history of embryo transfer failure;
- The intervention (I) was intrauterine PRP infusion before embryo implantation;
- The comparison (C) was to no intervention or placebo;
- The outcomes (O) were implantation rate, biochemical pregnancy rate, clinical pregnancy rate, live birth rate and miscarriage rate.
2.4. Eligibility Criteria
Participants included in the study had to be healthy women with no underlying severe diseases. Subjects had a history of failed embryo transfer and there was not a minimum number of participants or a limit on the number of patients treated.
The inclusion criteria had been: (a) study on humans only, (b) intervention of PRP by intrauterine administration before embryo implantation, (c) comparator was no intervention or placebo, and (d) study was designed as RCT.
The following publications were excluded: (a) observational studies; (b) trials with inadequate information or insufficient information regarding selected topic.
2.5. Data Sources and Search Strategy
The following databases were used for the systematic search from inception to 19 August 2022: MEDLINE/PubMed, Cochrane Central Register of Controlled Trials and Ovid. The following keywords were used in the search strategy: (Platelet-rich plasma OR platelet-rich fibrin OR plasma rich in growth factors OR PRP OR PRF OR PRGF) AND (infertility OR pregnancy OR endometrium OR thin endometrium OR endometrial thickness OR embryo transfer OR ovary). The search did not apply any language restriction. A screening of other systematic reviews was also carried out for possible additional trials.
2.6. Study Selection
Two reviewers independently (M.A. and M.d.l.F.) screened the search hits (the title and abstract) to exclude studies that had not met the research question and selection criteria. In case of disagreement, discussion was implemented to reach consensus. Then, full texts of the eligible articles or those with insufficient information in the title and abstract were obtained. The reviewers independently selected the studies that met the selection criteria. A third reviewer (M.H.A.) solved disagreement between the reviewers. The excluded publications were listed with the reason for exclusion (Supplementary Table S2).
2.7. Data Extraction
Two authors (M.A. and M.d.l.F.) independently extracted relevant data on study, participants and PRP protocol and characteristics into a specific Excel (Microsoft) spreadsheet. Discrepancies were eliminated by referring back to the full-text of the article. The extracted data were: (a) study characteristics—primary author, year of publication, number of participants and country; (b) patient characteristics—age and cause of failed embryo transfer; (c) PRP characteristics—obtention protocol, anticoagulant, presence of leukocytes, platelet concentration, PRP activation, method of application, dose, time to embryo transfer; (d) outcome assessment—implantation rate, biochemical pregnancy rate, clinical pregnancy rate, live birth rate and miscarriage rate. One of the reviewers (M.A.) included the data in Review Manager 5.4. Data accuracy was double-checked.
2.8. Risk of Bias Assessment
The methodologic quality of each trial was investigated by using risk of bias assessment in accordance with the Cochrane Handbook for Systematic Reviews of Interventions [40]. Each trial was classified at high, low or unclear risk of bias. Two authors independently evaluated the selected studies.
Using the Cochrane Risk of Bias Tool, each study was classified at high, moderate or low risk of bias. If none of the six domains were found to be at high risk, and if three or fewer domains were found to be at unclear risk, an overall low risk rating was assigned. An overall rating of moderate risk was attributed when one of the domains was high risk; or when no domain was high risk, but four or more were unclear risk. In all other cases, the study was considered to have an overall high risk of bias.
2.9. Outcomes
The primary outcome was clinical pregnancy rate, whereas secondary outcomes were implantation rate, biochemical pregnancy rate, live birth rate and miscarriage rate. All outcomes were defined according to the study authors (Supplementary Table S1).
2.10. Quality of Evidence
Two authors (M.A. and M.H.A.) independently evaluated the overall certainty of evidence using the grades recommendation, assessment, development and evaluation (GRADE) approach [40]. GRADEpro Guideline Development Tool (McMaster University, Hamilton, Canada, 2020, developed by Evidence Prime, Inc., Krakow, Poland, available from https://www.gradepro.org/, accessed on 8 February 2023) was used to assess the quality of the body of retrieved evidence [41].
2.11. Statistical Analysis
The software Review Manager 5.4 (The Nordic Cochrane Centre, Copenhagen, Denmark) was used to conduct the meta-analysis. Outcomes were assessed as dichotomous variables using the Mantel–Haenszel method and recorded as risk ratio (RR) with 95% confidence interval (CI). A pooled RR was interpreted as follows: a value higher than 1 means that the event occurs more in the exposure group. However, a RR value inferior to 1 indicates that the event occurs less in the exposure group.
Heterogeneity among the selected studies was evaluated using the I2 statistic (substantial heterogeneity is indicated by a value > 50%). This allowed to select either a fixed effects model (heterogeneity was not significant) or a random effects model (heterogeneity was significant). When significant heterogeneity was identified, the random effects model was used. The results were shown in a forest plot of interventions. Sensitivity analyses of reporting bias by funnel plots were not performed (the small number of studies).
3. Trial Sequential Analysis
Trial Sequential Analysis (TSA) was undertaken for the primary and secondary outcomes, to estimate the power of the meta-analysis results and to consider types I and II errors. The software TSA 0.9.5.10 Beta (Copenhagen Trial Unit Centre for Clinical Intervention Research Department, Copenhagen, Denmark) was used. Fixed or random effects model was chosen for meta-analysis, as appropriate. The 95% confidence intervals for inconsistency (I2) were also estimated with TSA software. The type I and type II errors were set at 5% and 20% (80% power), respectively, to estimate the required information size (RIS) and alpha monitoring limits. For the calculation of the RIS, the incidence rates in the test (PRP) and control arms were estimated in accordance with the results of the meta-analysis. No correction for heterogeneity was applied. Graphical analysis showed whether the cumulative Z curve (blue) crossed the trial sequence monitoring threshold (horizontal red line) and RIS threshold (vertical red line).
4. Results
4.1. Summary of the Literature Search
The Flow chart of the selected studies is depicted in Figure 1. The literature search yielded 2053 publications. The titles and abstracts were then screened, resulting in 26 studies possibly eligible selection. After reading the full-text publications, 14 studies were excluded (Supplementary Table S2). Qualitative synthesis was performed with the remaining 12 studies. Two studies did not provide enough data and were excluded from the quantitative me-ta-analysis [42,43].
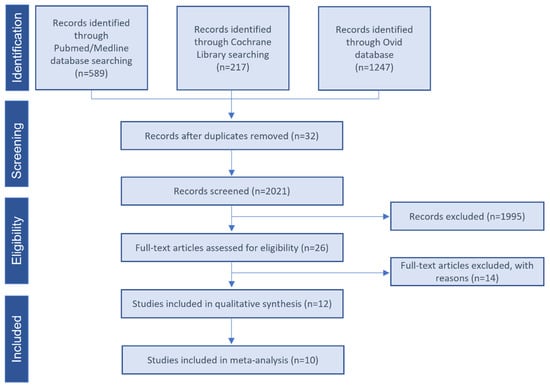
Figure 1.
Study selection flow diagram. PRISMA flow diagram of the screening and selection process.
4.2. Study Characteristics
In this systematic review, only RCTs that compared intrauterine PRP infusion before embryo transfer to no intervention or placebo group were included. Participants were women with a history of RIF [31,32,37,42,43,44,45,46,47] or TE [11,12]. Table 1 describes the principal characteristics of the selected studies. All trials were conducted between 2017 and 2022, of which eight studies were published after 2020. Eleven out of twelve studies were conducted in Iran and one in Russia. The number of patients in each study varied from 40 to 393. Hormone replacement therapy (HRT) was applied to all participants, regardless the experimental group. Nine studies infused ≤1 mL PRP [11,12,31,32,37,42,44,45,46], and two studies administered >1 mL [38,47], whereas one trial did not provide this information [43]. None of the studies reported on the activation of the PRP before application. The following outcomes were evaluated: implantation rate, biochemical pregnancy rate, clinical pregnancy rate, live birth rate and miscarriage rate (Table 2).

Table 1.
Main characteristics of the selected studies.
4.3. Risk of Bias of Included Trials
From a total of twelve studies, ten were selected for the quantitative analysis and subjected to a risk of bias assessment (Figure 2). The item of selection bias was judged low in eight studies that provided a random sequence generation description, while it was unclear in the remaining two studies. The item of selection bias in relation to allocation concealment was judged low risk of bias in seven studies and unclear risk of bias in three studies. The item performance bias was judged low risk of bias in three studies and unclear risk of bias in seven studies. The attrition bias was judged low risk of bias in nine studies and unclear risk of bias in one study (it was unclear in one study whether the follow-up reports were completed). The reporting bias was judged low risk of bias in three studies, unclear risk of bias in five studies and high risk of bias in two studies (did not report all planned outcomes).
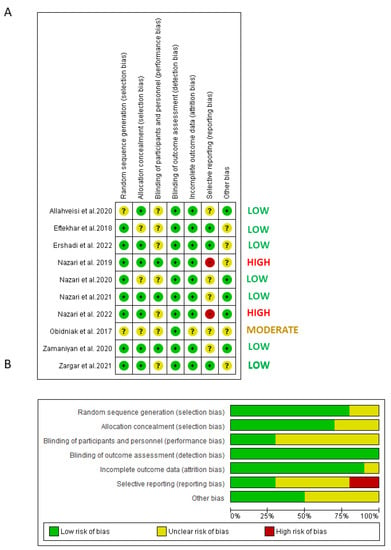
Figure 2.
Quality assessment of the included RCTs. (A) Risk of bias summary: review authors’ judgments about each risk of bias item for each included study: (+), low risk of bias; (−): high risk of bias; (?): unclear risk of bias. (B) Risk of bias graph: review authors’ judgments about each risk of bias item presented as percentages across all included studies.
After the assessment, the overall risk of bias was low in seven studies, moderate in one study and high in two studies.

Table 2.
Outcomes of the selected studies.
Table 2.
Outcomes of the selected studies.
| Study | Sample Size | Implantation Rate | Biochemical Pregnancy Rate | Clinical Pregnancy Rate | Live Birth Rate | Miscarriage Rate | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | PRP | Control | PRP | Control | PRP | Control | PRP | Control | PRP | Control | PRP | |
| Allahveisi et al., 2020 | 25 | 25 | 36% (n = 9) | 28% (n = 7) | NR | NR | 36% (n = 9) | 28% (n = 7) | 28% (n = 6) | 24% (n = 7) | NR | NR |
| Eftekhar et al., 2018 | 43 | 40 | 9.37% * | 21% * | 24.2% (n = 8) | 42.4% (n = 14) | 18.2% (n = 6) | 39.4% (n = 13) | 18.2% (n = 6) | 33.3% (n = 11) | 6% (n = 2) | 9% (n = 3) |
| Ershadi et al., 2022 | 45 | 40 | 0.38 ± 0.16% | 0.36 ± 0.24% | 27% (n = 12) | 40% (n = 16) | 24% (n = 11) | 33% (n = 13) | NR | NR | 31.25% (n = 5) | 8.33% (n = 1) |
| Nazari et al., 2020 | 48 | 49 | NR | NR | 27.08% (n = 13) | 53.06% (n = 26) | 16.66% (n = 8) | 44.89% (n = 22) | NR | NR | NR | NR |
| Nazari et al., 2019 | 30 | 30 | NR | NR | 6.7% (n = 2) | 40% (n = 12) | 3.3% (n = 1) | 33.3% (n = 10) | NR | NR | NR | NR |
| Nazari et al., 2021 | 197 | 196 | NR | NR | 24.87% (n = 49) | 51.53% (n = 101) | 19.28% (n = 38) | 48.97% (n = 96) | 5.58% (n = 11) | 39.28% (n = 77) | NR | NR |
| Nazari et al., 2022 | 20 | 20 | NR | NR | NR | NR | 20% (n = 4) | 35% (n = 7) | 0% (n = 0) | 15% (n = 3) | 20% (n = 4) | 20% (n = 4) |
| Obidniak et al., 2017 | 45 | 45 | 20.9% * | 40.5% * | NR | NR | 24.4% (n = 11) | 53.3% (n = 24) | NR | NR | NR | NR |
| Zamaniyan et al., 2020 | 43 | 55 | 34.9% (n = 15) | 63.6% (n = 35) | 23.3% (n = 10) | 36.2% (n = 20) | 23.3% (n = 10) | 52.7% (n = 29) | NR | NR | 4.6% (n = 2) | 1.8% (n = 1) |
| Zargar et al., 2021 | 40 | 40 | 5% (n = 2) | 15% (n = 6) | NR | NR | 2.5% (n = 1) | 12.5% (n = 5) | 2.5% (n = 1) | 12.5% (n = 5) | 2.5% (n = 1) | 2.5% (n = 1) |
* Data excluded from the meta-analysis; NR: not reported.
4.4. Quality of Evidence
GRADE was used to qualify the evidence of the study outcomes (Table 3). Regarding to the studies with RIF patients, we downgraded the certainty of the implantation rate by two levels to low due to very serious limitation in the imprecision (optimal information size is not met and the 95% CI of the relative risk (RR) included RR of 1.25). The same was for the certainty of live birth rate and miscarriage rate. Remaining outcomes (biochemical pregnancy rate and clinical pregnancy rate) were judged as high certainty of evidence. With regard to TE studies, the level of evidence of biochemical pregnancy rate was downgraded to low due to the unclear risk of bias in one study, the optimal information size was not met, and the RR included a value of 1.25 (Table 4). The same limitations applied to the clinical pregnancy outcome The certainty of the outcomes of the live birth rate and miscarriage rate was downgraded by three levels to very low due to the unclear risk of bias in one study, the optimal information size was not met, the 95% CI of the RR included a RR of 0.75 or 1.25 and a wide range of the 95% CI of the RR (miscarriage rate).

Table 3.
Summary of the quality assessment by GRADE approach of outcomes included in the meta-analysis of repeated implantation failure patients.

Table 4.
Summary of the quality assessment by GRADE approach of outcomes included in the meta-analysis of thin endometrium patients.
4.5. Effect of PRP on Clinical Pregnancy Rate
Ten studies evaluated clinical pregnancy in 933 women with RIF [31,32,37,38,44,45,46,47] and 143 women with TE [11,12]. Clinical pregnancy was interpreted as the presence of fetal heartbeat in transvaginal ultrasound 5–6 weeks after embryo transfer in six studies [11,12,31,32,45,46]. Other study measured clinical pregnancy rate by dividing the number of fetal poles with an observed heartbeat in the 6-week-old sonogram by the number of the transferred embryos [37]. Other study defined it as the presence of an embryonic sac at 5–6 weeks gestation [44]. Two publications did not define the term [38,47].
A total of 325 pregnancies were reported; the percentage of pregnancies was 41.85% (226 out of 540) and 18.47% (99 out of 536) in the PRP and control groups, respectively (Figures S1 and S2, Supplementary Table S3 and S4).
The meta-analysis including eight studies (excluding high-risk bias studies) showed higher pregnancy rate when intrauterine PRP was administered (RR: 2.19 (95% CI: 1.78 to 2.70), I2: 30%). In this line, statistical trend was maintained when RIF studies were analyzed separately (RR: 2.18 (95% CI: 1.76 to 2.70), I2: 40%). No meta-analysis could be performed for the TE sub-group (Figure 3A).

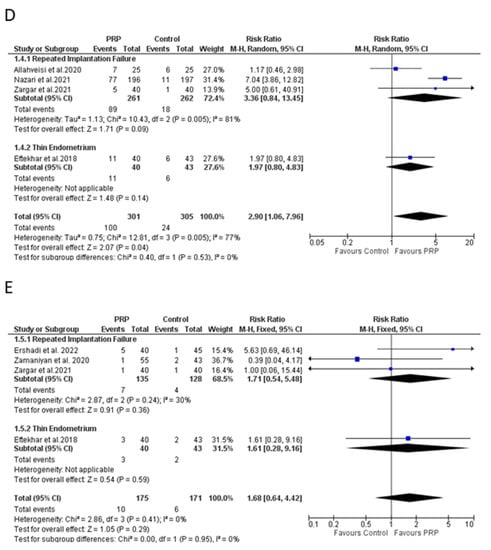
Figure 3.
Meta-analysis of the studies evaluating (A) clinical pregnancy rate, (B) implantation rate, (C) biochemical pregnancy rate, (D) live-birth rate, and (E) miscarriage rate. CI: confidence interval; PRP: platelet-rich plasma. Studies with high-risk bias were excluded.
4.6. Effect of PRP on Implantation Rate
Five studies analyzed implantation rate after PRP treatment [12,37,38,46,47]. These studies were composed of a total of 401 infertile women who had experienced RIF (four studies) or TE (one study). Implantation rate was defined as the ratio of gestational sacs to the number of embryos transferred in three trials [12,37,46]; authors did not provide a detailed description in two publications [38,47].
Out of four trials investigating women with RIF, two studies reported a beneficial effect for PRP [46,47], whereas no statistical differences were found in other two trials [37,38]. In this line, significative difference between groups was observed in the trial composed of a TE cohort [12]. Due to missing data, two studies were finally excluded from the meta-analysis [12,47]. The remaining studies corresponded to women with RIF and showed a better performance in terms of implantation rate when PRP was administered before embryo transfer RR: 1.57 (95% CI: 1.07 to 2.30), I2: 0% (Figure 3B). No meta-analysis could be performed for the TE sub-group.
4.7. Effect of PRP on Biochemical Pregnancy Rate
Six studies including 816 participants (410 cases and 406 controls) evaluated the effect of intrauterine PRP infusion before embryo transfer on biochemical pregnancy rate [11,12,31,32,45,46]. Four studies included infertile women who had experienced RIF [31,32,45,46], whereas TE pathology was studied in two trials [11,12]. All studies defined biochemical pregnancy as a positive serum β-human chorionic gonadotropin after 14 days from embryo transfer.
Women subjected to PRP treatment showed a biochemical pregnancy rate of 46.10% (189 out of 410); on the contrary, the control group displayed a 23.15% (94 out of 406) rate (Figures S1 and S2, Supplementary Tables S3 and S4). The meta-analysis, including five studies (excluding high-risk bias study), showed a significant positive effect for the PRP group in comparison to conventional treatment (RR: 1.91 (95% CI: 1.55 to 2.35), I2: 0%). When studies were evaluated separately by type of pathology, a beneficial effect was only observed for the PRP in women with RIF (RR: 1.91 (95% CI: 1.54 to 2.37), I2: 0%) (Figure 3C).
4.8. Effect of PRP on Live Birth Rate
Five RCTs composed of a total of 646 patients assessed the effect of PRP on live birth rate [12,32,37,38,44]. Four trials were conducted on women presenting a history of RIF [32,37,38] and one study was performed on women with TE [12]. Four studies considered birth after 20 to 24 weeks of gestation as a live birth [12,37,38,44]. Authors did not provide a clear definition in another trial [32].
Live birth rate reached to 32.09% (103 out of 321) when PRP treatment was applied, whereas the rate was reduced to 7.38% (24 out of 325) in its absence. Four out of five studies reported a nonsignificant difference [12,37,38,44] (Supplementary Materials). However, the overall meta-analysis (excluding one high-bias study) showed better outcomes following a PRP-based protocol (RR: 2.90 (95% CI: 1.06 to 7.96), I2: 77%). Analyzing RIF studies separately, the risk ratio increased to 3.36 (95% CI: 0.84 to 13.45), I2: 81% (Figure 3D).
4.9. Effect of PRP on Miscarriage Rate
Five studies evaluated the occurrence of miscarriages following intrauterine PRP infusion [12,38,44,45,46]. From a total of 386 participants, PRP was administered to 195 women (Supplementary Materials). Two studies [12,44] considered a miscarriage as pregnancy loss before 20 weeks of gestation. A clear definition was absent in the other three trials [38,45,46].
Our meta-analysis (excluding one high-risk bias study) did not reveal a significant effect of PRP on the miscarriage rate (RR: 1.68 (95% CI: 0.64 to 4.42), I2: 0%). Nonsignificant differences remained when a subgroup analysis was performed in RIF patients (RR: 1.71 (95% CI: 0.54 to 5.48), I2: 30%). Only one trial evaluated miscarriages on TE cohort and no meta-analysis could be performed (Figure 3E).
4.10. Trial Sequential Analysis
Trial sequential analysis was only undertaken for RIF, as many subgroups with TE had insufficient data to perform the analysis. TSA showed that the cumulative Z-curve was maintained well beyond the significance threshold in favor of the PRP group for all outcomes, except for miscarriage rate. In addition, the total sample size for RIF was above the required information size (RIS) for biochemical pregnancy rate (RIS = 199), clinical pregnancy rate (RIS = 234) and live birth rate (RIS = 331), but it was inferior for implantation rate (RIS = 806) and for miscarriage rate (RIS = 3756). This suggested that the meta-analysis had sufficient power to detect the beneficial effect of PRP for biochemical pregnancy rate, clinical pregnancy rate and live birth rate. Conversely, more studies are required to achieve significance regarding the advantageous effect of PRP for implantation rate and, especially, for miscarriage rate. From the available evidence regarding the last outcome, there is the possibility that additional studies might confirm the conclusion that PRP does not provide advantages for miscarriage rate.
5. Discussion
During the last decades, a variety of therapeutic approaches and interventions have been proposed to manage poor responsiveness to in vitro fertilization techniques, including uterine interventions [48,49,50,51,52], gynecological surgical procedures [53,54,55,56,57,58,59,60], immunomodulatory therapies [61,62,63,64,65,66,67,68] or treatments to enhance endometrial receptivity [69,70,71,72,73]. However, it was not until 2015 [8] that autologous platelet concentrates were used to promote endometrial growth and improve pregnancy outcome in infertile women with TE. Since then, the available clinical studies are increasing rapidly and PRPs are becoming the treatment of choice for many physicians. The present systematic review investigated the available literature to analyze the ability of PRP to manage infertility problems in women with a history of embryo transfer failure.
Ten RCTs were selected for our meta-analysis and a total of 540 women receiving intrauterine PRP infusion were included. According to TSA results, our meta-analysis fulfilled the information size requirement providing sufficient evidence for biochemical pregnancy rate, clinical pregnancy rate and live birth rate outcomes. To our knowledge, there are currently no published meta-analyses in the field of reproduction that specifically conduct a TSA to estimate the level of evidence. Conversely, information size requirement was found to be insufficient for implantation rate and miscarriage rate. This finding can be explained by the small number of patients recruited for this outcome and the high heterogeneity observed among studies. Future clinical trials and high-standardized protocols for PRP preparation and infusion are needed.
Since its discovery, PRP has been prescribed in the field of regenerative medicine for a wide variety of pathologies, including dermatology [74], maxillofacial surgery [75,76], traumatology [77,78] or in vitro reproduction [79,80,81] among others. Only two systematic reviews with meta-analysis have been published evaluating the beneficial effect of PRP in assisted reproduction. The first study was published by Maleki-Hajiagha et al. in 2020 and reported favorable outcomes for PRP group in women undergoing assisted reproduction when comparing to nonintervention control subjects [82]. More recently, Busnelli and colleagues performed a meta-analysis analyzing all available treatments and interventions in women with RIF [83]. In agreement with the first review, a beneficial effect was observed in terms of successful pregnancies when autologous PRP was infused before embryo transfer. The overall conclusion reached by these studies was similar to the present review, showing that PRP therapy confers additional benefits over conventional hormone replacement treatments. Nevertheless, the number of RCTs included in our systematic reviews is markedly higher compared to the previous ones. Moreover, no strength of evidence assessment was attempted, and no TSA analysis was presented to evaluate the power of the meta-analysis. In addition, nonrandomized controlled trials were also included in previously published systematic reviews. These types of studies might be an important source of bias.
Autologous PRP can be manufactured using different protocols that might lead to products with different composition and functionality [76]. It is likely that PRP composition plays a pivotal role in the results of the trial. In fact, the presence or absence of leukocyte is considered a key differentiator for many authors [76,84]. Unfortunately, none of the selected studies provided an accurate description of leukocyte concentration, and two trials did not even mention whether leukocytes were present or not. The different methodology used to prepare PRP (manufacturer, a comprehensive protocol description or leukocyte and platelet counts, among others) should be clearly provide by the authors, as these factors might have a clear impact on the measured clinical outcomes.
According to the published studies, the prevalence of TE ranged from 2.4% to 8.5% [85,86,87]. The management of women with insufficient endometrial thickness or a refractory endometrium is one of biggest challenges in assisted reproduction. In the last years, PRP therapies have emerged as a cutting-edge treatment to promote endometrial growth and enhance the success of embryo transfer. However, meta-analysis for TE studies could not be performed due to the exclusion of high-risk bias studies (leaving only one study). The current literature establishes 7 mm as the minimum endometrium thickness needed to accomplish the embryo transfer. Three of the selected studies administered a second intrauterine PRP infusion if the thickness did not reach 7 mm [11,12,38]. However, a meta-analysis suggests the cutoff of 7 mm has limited prognostic value for clinical pregnancy [88] and questions the requirement of this second infusion. Future studies should clarify the clinical usefulness of this thickness limit.
Another critical parameter for autologous PRP performance is the dose infused in the uterus. In the meta-analysis performed by Maleki-Hajiagha et al. [82], a dose of 0.5–1 mL showed a higher effect in comparison to doses of <0.5 mL in terms of clinical pregnancy. Our study did not consider this factor in the meta-analysis; however, a qualitative assessment of the data showed a high performance for both doses. The standardization of the PRP dose should be the focus of future clinical trials.
Although autologous PRP are being adopted by many fertility clinics, the data describing the safety profile are still limited. Only five out of the ten selected studies reported the incidence of miscarriage after PRP infusion. More importantly, none of the trials monitored PRP-related adverse events in the months following the childbirth or their effect on conceived children. Consequently, this aspect could be an interesting topic for upcoming clinical trials. PRP has been used in the context of in vitro fertilization to treat patients with repeated pregnancy loss. However, the level of the available evidence is not sufficient to propose the use of in vitro fertilization to treat patients with unexplained recurrent pregnancy loss [89].
The number of clinical trials using PRP in the field of reproduction is increasing rapidly. However, the underlying molecular mechanism of PRP to enhance endometrial receptivity has not been clearly described. In view of the experience derived from other medical fields, it is likely that PRP plays a pivotal role in endometrium tissue growth and regeneration by releasing growth factors and cytokines. Consequently, a higher migration and proliferation rate have been observed on different human endometrial cells following PRP application [7]. In this line, increased levels of the proliferative marker Ki67 have been measured in the endometrial stromal cell line ICE7 [90] and uterine horn [91]. More recently, the expression of Ki67 and homeobox A10 (Hoxa10) was found to be upregulated in the endometrium of mice subjected to a uterine damage model [92]. The potential role of PRP in inflammation has also been extensively studied. In fact, anti-inflammatory properties of PRP involve the downregulation of several key players in the inflammatory pathway [93], including the nuclear factor kappa-B (NF-κB) [94]. In addition, PRP is able to modulate a wide array of chemokines such as chemokine ligand 5 (CCL5) [7], lipoxin A4 [94], IL-1B, IL-8, TNF-A, COX-2 or iNOS [95]. Some authors have speculated these factors result in a reduced intrauterine fluid accumulation and, therefore, diminishing inflammation [94,96]. Other evidence suggests decreased fibrosis and a restoration of endometrial structure following PRP administration in a murine model of uterine injury [97]. A reduced expression in collagen type 1A, transforming growth factor β1 (TGF-β1) and tissue inhibitor of metalloproteinase 1 (TIMP1) were reported, supporting the notion that PRP might promote endometrial regeneration when the endometrium is damaged. Although there is a lack of studies associating directly immune response to PRP infusion in the endometrium, it is well known that platelets participate in innate and adaptive immunity [98,99], with the ability to interact with many immune cells. The interaction of platelets with neutrophils occurs via P-selectin/PSGL-1 and, subsequently, is able to induce leukocyte signaling and neutrophil integrins activation to promote degranulation [100,101]. The initial contact between platelets and monocyte also involves P-selectin and PSGL-1, and this interaction enhance expression of β1-integrin, β2-integrin and BF-κB pathways [102]. Overall, evidence of the underlying mechanism of PRP is still sparce, and it is likely that several factors and molecular pathways are involved (Figure 4).
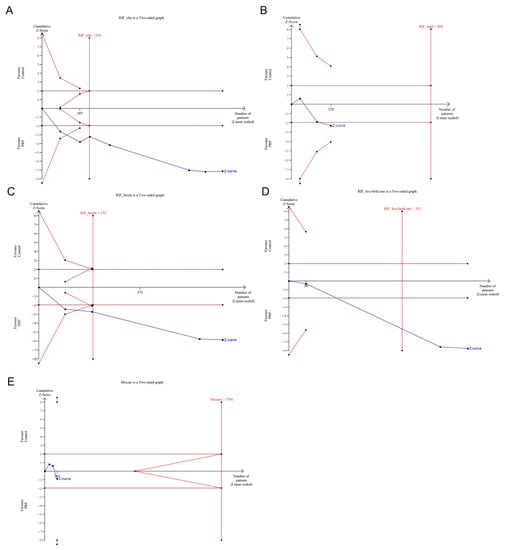
Figure 4.
Trial sequential analysis for (A) clinical pregnancy rate, (B) implantation pregnancy rate, (C) biochemical pregnancy rate, (D) live birth rate and (E) miscarriage rate in recurrent implantation failure (RIF) studies. Studies with high-risk bias were excluded.
The strengths of this systematic review included a restriction to RCTs to avoid biases from observational studies, the assessment of bias with the Cochrane Handbook for Systematic Reviews of Interventions [40], the evaluation of the strength of evidence with the grade approach [41] and the implementation of TSA to address information size requirement for each outcome [103]. In addition, most of the studies used similar outcomes definitions and the follow-up time was comparable among studies, avoiding biases caused by the different methodology or trial design.
However, several limitations need to be considered. First, the search strategy did not incorporate MeSH (Medical Subject Headings) terms. Second, there is no standardized protocol for PRP products, with some preparations including leukocytes among their composition and, therefore, conclusions should be interpreted with caution. Third, most of available trials were performed in the same country (Iran), the ethnicity being an important confounding factor that can lead to a data misinterpretation. Fourth, some publications reported an additional infusion of PRP when endometrium thickness was inadequate for embryo implantation. This factor was not considered in the meta-analysis due to the absence of information provided by the authors.
The present systematic review demonstrates that PRP might be an effective therapeutic approach in cases of poor responsiveness to conventional treatments in assisted reproductive techniques. Based on our meta-analysis and the subsequent TSA evaluation, autologous PRP could enhance biochemical pregnancy rate, clinical pregnancy rate and live birth rate. Additional well-designed trials are needed to confirm the beneficial effect of PRP over the implantation rate and miscarriage rate. Accordingly, there is lack of evidence to draw conclusions on whether PRP can bring benefit on women with TE due to the limited number and quality of the studies. Future studies should also elucidate the safety profile of intrauterine PRP administration with long-term follow-ups.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/bioengineering10030303/s1, Table S1: Outcome definitions and time of measurement; Table S2: Excluded studies (with reasons), Figure S1: Meta-analysis of all studies (not excluding high-risk bias studies) evaluating (A) clinical pregnancy rate, (B) implantation rate, (C) biochemical pregnancy rate, (D) live-birth rate, and (E) miscarriage rate. CI: confidence interval; PRP: platelet-rich plasma; Figure S2: Trial sequential analysis for (A) clinical pregnancy rate, (B) implantation pregnancy rate, (C) biochemical pregnancy rate, (D) live-birth rate and (E) miscarriage rate in Recurrent Implantation Failure (RIF) studies. Trial sequential analysis for (F) clinical pregnancy in Thin Endometrium (TE) studies; Table S3: Summary of the quality assessment by GRADE approach of outcomes included in the meta-analysis of Repeated Implantation Failure patients; Table S4: Summary of the quality assessment by GRADE approach of outcomes included in the meta-analysis of Thin Endometrium patients. References [104,105,106,107,108,109,110,111,112] are cited in the supplementary materials.
Author Contributions
E.A. and M.H.A. designed the study. M.A. and M.d.l.F. extracted data and performed statistical analysis. M.H.A., M.D.F. and M.A. interpreted the data. M.A. wrote the manuscript. E.A., M.H.A., M.d.l.F. and M.D.F. critically reviewed the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Data will be made available to the editors of the journal for review or query upon request.
Conflicts of Interest
E.A. is the head of Eduardo Anitua Foundation and the Scientific Director of BTI Biotechnology Institute. MA, M.d.l.F. and M.H.A. are researchers in BTI Biotechnology Institute, a biotechnological company that investigates in the field of regenerative medicine and PRGF-Endoret technology. M.D.F. has no conflict of interest.
References
- Mascarenhas, M.N.; Flaxman, S.R.; Boerma, T.; Vanderpoel, S.; Stevens, G.A. National, Regional, and Global Trends in Infertility Prevalence since 1990: A Systematic Analysis of 277 Health Surveys. PLoS Med. 2012, 9, e1001356. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.K.; Vandekerckhove, P.; Kennedy, R.; Keay, S.D. Investigation and Current Management of Recurrent IVF Treatment Failure in the UK. BJOG 2005, 112, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Garneau, A.S.; Young, S.L. Defining Recurrent Implantation Failure: A Profusion of Confusion or Simply an Illusion? Fertil. Steril. 2021, 116, 1432–1435. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Definitions of Infertility and Recurrent Pregnancy Loss: A Committee Opinion. Fertil. Steril. 2013, 99, 63. [Google Scholar] [CrossRef] [PubMed]
- Franasiak, J.M.; Alecsandru, D.; Forman, E.J.; Gemmell, L.C.; Goldberg, J.M.; Llarena, N.; Margolis, C.; Laven, J.; Schoenmakers, S.; Seli, E. A Review of the Pathophysiology of Recurrent Implantation Failure. Fertil. Steril. 2021, 116, 1436–1448. [Google Scholar] [CrossRef]
- Bashiri, A.; Halper, K.I.; Orvieto, R. Recurrent Implantation Failure-Update Overview on Etiology, Diagnosis, Treatment and Future Directions. Reprod. Biol. Endocrinol. 2018, 16, 121. [Google Scholar] [CrossRef]
- Aghajanova, L.; Houshdaran, S.; Balayan, S.; Manvelyan, E.; Irwin, J.C.; Huddleston, H.G.; Giudice, L.C. In Vitro Evidence That Platelet-Rich Plasma Stimulates Cellular Processes Involved in Endometrial Regeneration. J. Assist. Reprod. Genet. 2018, 35, 757–770. [Google Scholar] [CrossRef]
- Chang, Y.; Li, J.; Chen, Y.; Wei, L.; Yang, X.; Shi, Y.; Liang, X. Autologous Platelet-Rich Plasma Promotes Endometrial Growth and Improves Pregnancy Outcome during in Vitro Fertilization. Int. J. Clin. Exp. Med. 2015, 8, 1286–1290. [Google Scholar]
- Coksuer, H.; Akdemir, Y.; Ulas Barut, M. Improved in Vitro Fertilization Success and Pregnancy Outcome with Autologous Platelet-Rich Plasma Treatment in Unexplained Infertility Patients That Had Repeated Implantation Failure History. Gynecol. Endocrinol. 2019, 35, 815–818. [Google Scholar] [CrossRef]
- Farimani, M.; Poorolajal, J.; Rabiee, S.; Bahmanzadeh, M. Successful Pregnancy and Live Birth after Intrauterine Administration of Autologous Platelet-Rich Plasma in a Woman with Recurrent Implantation Failure: A Case Report. Int. J. Reprod. Biomed. 2017, 15, 803–806. [Google Scholar] [CrossRef]
- Nazari, L.; Salehpour, S.; Hoseini, S.; Zadehmodarres, S.; Azargashb, E. Effects of Autologous Platelet-Rich Plasma on Endometrial Expansion in Patients Undergoing Frozen-Thawed Embryo Transfer: A Double-Blind RCT. Int. J. Reprod. Biomed. 2019, 17, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Eftekhar, M.; Neghab, N.; Naghshineh, E.; Khani, P. Can Autologous Platelet Rich Plasma Expand Endometrial Thickness and Improve Pregnancy Rate during Frozen-Thawed Embryo Transfer Cycle? A Randomized Clinical Trial. Taiwan J. Obstet. Gynecol. 2018, 57, 810–813. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.B.; Jeelall, Y.; Pennell, C.E.; Hart, R.; McLean-Tooke, A.; Lucas, M. The Role of Immunological Testing and Intervention in Reproductive Medicine: A Fertile Collaboration? Am. J. Reprod. Immunol. 2018, 79, e12784. [Google Scholar] [CrossRef] [PubMed]
- Ghaebi, M.; Abdolmohammadi-Vahid, S.; Ahmadi, M.; Eghbal-Fard, S.; Dolati, S.; Nouri, M.; Talebi, M.; Hamdi, K.; Marofi, F.; Aghebati-Maleki, L.; et al. T Cell Subsets in Peripheral Blood of Women with Recurrent Implantation Failure. J. Reprod. Immunol. 2019, 131, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Lédée, N.; Petitbarat, M.; Chevrier, L.; Vitoux, D.; Vezmar, K.; Rahmati, M.; Dubanchet, S.; Gahéry, H.; Bensussan, A.; Chaouat, G. The Uterine Immune Profile May Help Women with Repeated Unexplained Embryo Implantation Failure after In Vitro Fertilization. Am. J. Reprod. Immunol. 2016, 75, 388–401. [Google Scholar] [CrossRef] [PubMed]
- Davar, R.; Janati, S.; Mohseni, F.; Khabazkhoob, M.; Asgari, S. A Comparison of the Effects of Transdermal Estradiol and Estradiol Valerate on Endometrial Receptivity in Frozen-Thawed Embryo Transfer Cycles: A Randomized Clinical Trial. J. Reprod. Infertil. 2016, 17, 97–103. [Google Scholar]
- Weckstein, L.N.; Jacobson, A.; Galen, D.; Hampton, K.; Hammel, J. Low-Dose Aspirin for Oocyte Donation Recipients with a Thin Endometrium: Prospective, Randomized Study. Fertil. Steril. 1997, 68, 927–930. [Google Scholar] [CrossRef]
- Wang, L.; Huang, X.; Li, X.; Lv, F.; He, X.; Pan, Y.; Wang, L.; Zhang, X. Efficacy Evaluation of Low-Dose Aspirin in IVF/ICSI Patients Evidence from 13 RCTs: A Systematic Review and Meta-Analysis. Medicine 2017, 96, e7720. [Google Scholar] [CrossRef]
- Acharya, S.; Yasmin, E.; Balen, A.H. The Use of a Combination of Pentoxifylline and Tocopherol in Women with a Thin Endometrium Undergoing Assisted Conception Therapies—A Report of 20 Cases. Hum. Fertil. 2009, 12, 198–203. [Google Scholar] [CrossRef]
- Zinger, M.; Liu, J.H.; Thomas, M.A. Successful Use of Vaginal Sildenafil Citrate in Two Infertility Patients with Asherman’s Syndrome. J. Women’s Health 2006, 15, 442–444. [Google Scholar] [CrossRef]
- Sher, G.; Fisch, J.D. Effect of Vaginal Sildenafil on the Outcome of in Vitro Fertilization (IVF) after Multiple IVF Failures Attributed to Poor Endometrial Development. Fertil. Steril. 2002, 78, 1073–1076. [Google Scholar] [CrossRef] [PubMed]
- Refai, H.; Hassan, D.; Abdelmonem, R. Development and Characterization of Polymer-Coated Liposomes for Vaginal Delivery of Sildenafil Citrate. Drug Deliv. 2017, 24, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Ke, H.; Jiang, J.; Xia, M.; Tang, R.; Qin, Y.; Chen, Z.-J. The Effect of Tamoxifen on Thin Endometrium in Patients Undergoing Frozen-Thawed Embryo Transfer. Reprod. Sci. 2018, 25, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Kunicki, M.; Łukaszuk, K.; Liss, J.; Skowrońska, P.; Szczyptańska, J. Granulocyte Colony Stimulating Factor Treatment of Resistant Thin Endometrium in Women with Frozen-Thawed Blastocyst Transfer. Syst. Biol. Reprod. Med. 2017, 63, 49–57. [Google Scholar] [CrossRef]
- Bos-Mikich, A.; De Oliveira, R.; Frantz, N. Platelet-Rich Plasma Therapy and Reproductive Medicine. J. Assist. Reprod. Genet. 2018, 35, 753–756. [Google Scholar] [CrossRef]
- Gat, I.; Levron, J.; Yerushalmi, G.; Dor, J.; Brengauz, M.; Orvieto, R. Should Zygote Intrafallopian Transfer Be Offered to All Patients with Unexplained Repeated In-Vitro Fertilization Cycle Failures? J. Ovarian Res. 2014, 7, 7. [Google Scholar] [CrossRef]
- Xu, J.; Gou, L.; Zhang, P.; Li, H.; Qiu, S. Platelet-Rich Plasma and Regenerative Dentistry. Aust. Dent. J. 2020, 65, 131–142. [Google Scholar] [CrossRef]
- Merchán, W.H.; Gómez, L.A.; Chasoy, M.E.; Alfonso-Rodríguez, C.A.; Muñoz, A.L. Platelet-Rich Plasma, a Powerful Tool in Dermatology. J. Tissue Eng. Regen. Med. 2019, 13, 892–901. [Google Scholar] [CrossRef]
- Urits, I.; Viswanath, O.; Galasso, A.C.; Sottosani, E.R.; Mahan, K.M.; Aiudi, C.M.; Kaye, A.D.; Orhurhu, V.J. Platelet-Rich Plasma for the Treatment of Low Back Pain: A Comprehensive Review. Curr. Pain Headache Rep. 2019, 23, 52. [Google Scholar] [CrossRef]
- Sanchez-Avila, R.M.; Merayo-Lloves, J.; Muruzabal, F.; Orive, G.; Anitua, E. Plasma Rich in Growth Factors for the Treatment of Dry Eye from Patients with Graft versus Host Diseases. Eur. J. Ophthalmol. 2020, 30, 94–103. [Google Scholar] [CrossRef]
- Nazari, L.; Salehpour, S.; Hosseini, M.S.; Hashemi Moghanjoughi, P. The Effects of Autologous Platelet-Rich Plasma in Repeated Implantation Failure: A Randomized Controlled Trial. Hum. Fertil. 2020, 23, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Nazari, L.; Salehpour, S.; Hosseini, S.; Sheibani, S.; Hosseinirad, H. The Effects of Autologous Platelet-Rich Plasma on Pregnancy Outcomes in Repeated Implantation Failure Patients Undergoing Frozen Embryo Transfer: A Randomized Controlled Trial. Reprod. Sci. 2022, 29, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; de la Fuente, M.; Ferrando, M.; Quintana, F.; Larreategui, Z.; Matorras, R.; Orive, G. Biological Effects of Plasma Rich in Growth Factors (PRGF) on Human Endometrial Fibroblasts. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 206, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, L.; Mou, S.; Zhao, H.; Fang, J.; Xiang, Y.; Zhao, T.; Sha, T.; Ding, J.; Hao, C. Investigation of Platelet-Rich Plasma in Increasing Proliferation and Migration of Endometrial Mesenchymal Stem Cells and Improving Pregnancy Outcome of Patients with Thin Endometrium. J. Cell. Biochem. 2018, 120, 7403–7411. [Google Scholar] [CrossRef] [PubMed]
- Melo, P.; Navarro, C.; Jones, C.; Coward, K.; Coleman, L. The Use of Autologous Platelet-Rich Plasma (PRP) versus No Intervention in Women with Low Ovarian Reserve Undergoing Fertility Treatment: A Non-Randomized Interventional Study. J. Assist. Reprod. Genet. 2020, 37, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Pantos, K.; Simopoulou, M.; Pantou, A.; Rapani, A.; Tsioulou, P.; Nitsos, N.; Syrkos, S.; Pappas, A.; Koutsilieris, M.; Sfakianoudis, K. A Case Series on Natural Conceptions Resulting in Ongoing Pregnancies in Menopausal and Prematurely Menopausal Women Following Platelet-Rich Plasma Treatment. Cell Transplant. 2019, 28, 1333–1340. [Google Scholar] [CrossRef]
- Allahveisi, A.; Seyedoshohadaei, F.; Rezaei, M.; Bazrafshan, N.; Rahimi, K. The Effect of Platelet-Rich Plasma on the Achievement of Pregnancy during Frozen Embryo Transfer in Women with a History of Failed Implantation. Heliyon 2020, 6, e03577. [Google Scholar] [CrossRef]
- Zargar, M.; Pazhouhanfar, R.; Najafian, M.; Choghakabodi, P.M. Effects of Intrauterine Autologous Platelet-Rich Plasma Infusions on Outcomes in Women with Repetitive in Vitro Fertilization Failures: A Prospective Randomized Study. Clin. Exp. Obstet. Gynecol. 2021, 48, 180–185. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. PLoS Med. 2021, 18, e1003583. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Schünemann, H.; Brożek, J.; Guyatt, G.; Oxman, A. GRADE Handbook for Grading Quality of Evidence and Strength of Recommendations; The GRADE Working Group, McMaster University: Hamilton, ON, Canada, 2013; Available online: https://www.guidelinedevelopment.org/handbook (accessed on 8 February 2023).
- Bakhsh, A.S.; Maleki, N.; Sadeghi, M.R.; SadeghiTabar, A.; Tavakoli, M.; Zafardoust, S.; Karimi, A.; Askari, S.; Jouhari, S.; Mohammadzadeh, A. Effects of Autologous Platelet-Rich Plasma in Women with Repeated Implantation Failure Undergoing Assisted Reproduction. JBRA Assist. Reprod. 2021, 26, 84–87. [Google Scholar] [CrossRef]
- Ghasemi, M. Evaluation of the Effect of Intrauterine Injection of Platelet-Rich Plasma on the Pregnancy Rate of Patients with a History of Implantation Failure in the in Vitro Fertilization Cycle. Int. J. Gynecol. Cancer 2020, 30, A1–A142. [Google Scholar] [CrossRef] [PubMed]
- Nazari, L.; Salehpour, S.; Hosseini, S.; Hashemi, T.; Borumandnia, N.; Azizi, E. Effect of Autologous Platelet-Rich Plasma for Treatment of Recurrent Pregnancy Loss: A Randomized Controlled Trial. Obstet. Gynecol. Sci. 2022, 65, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Ershadi, S.; Noori, N.; Dashipoor, A.; Ghasemi, M.; Shamsa, N. Evaluation of the Effect of Intrauterine Injection of Platelet-Rich Plasma on the Pregnancy Rate of Patients with a History of Implantation Failure in the in Vitro Fertilization Cycle. J. Fam. Med. Prim. Care 2022, 11, 2162–2166. [Google Scholar] [CrossRef]
- Zamaniyan, M.; Peyvandi, S.; Heidaryan Gorji, H.; Moradi, S.; Jamal, J.; Yahya Poor Aghmashhadi, F.; Hossein Mohammadi, M. Effect of Platelet-Rich Plasma on Pregnancy Outcomes in Infertile Women with Recurrent Implantation Failure: A Randomized Controlled Trial. Gynecol. Endocrinol. 2021, 37, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Obidniak, D.; Gzgzyan, A.; Feoktistov, A.; Niauri, D. Randomized Controlled Trial Evaluating Efficacy of Autologous Platelet-Rich Plasma Therapy for Patients with Recurrent Implantation Failure. Fertil. Steril. 2017, 108, e370. [Google Scholar] [CrossRef]
- Baum, M.; Yerushalmi, G.M.; Maman, E.; Kedem, A.; Machtinger, R.; Hourvitz, A.; Dor, J. Does Local Injury to the Endometrium before IVF Cycle Really Affect Treatment Outcome? Results of a Randomized Placebo Controlled Trial. Gynecol. Endocrinol. 2012, 28, 933–936. [Google Scholar] [CrossRef]
- Cicinelli, E.; Matteo, M.; Tinelli, R.; Lepera, A.; Alfonso, R.; Indraccolo, U.; Marrocchella, S.; Greco, P.; Resta, L. Prevalence of Chronic Endometritis in Repeated Unexplained Implantation Failure and the IVF Success Rate after Antibiotic Therapy. Hum. Reprod. 2015, 30, 323–330. [Google Scholar] [CrossRef]
- Gao, M.; Sun, Y.; Xie, H.; Fang, S.; Zhao, X. Hysteroscopy Prior to Repeat Embryo Transfer May Improve Pregnancy Outcomes for Asymptomatic Women with Repeated Implantation Failure. J. Obstet. Gynaecol. Res. 2015, 41, 1569–1576. [Google Scholar] [CrossRef]
- Mao, X.; Zhang, J.; Chen, Q.; Kuang, Y.; Zhang, S. Short-Term Copper Intrauterine Device Placement Improves the Implantation and Pregnancy Rates in Women with Repeated Implantation Failure. Fertil. Steril. 2017, 108, 55–61.e1. [Google Scholar] [CrossRef]
- He, Y.; Wu, H.; He, X.; Xing, Q.; Zhou, P.; Cao, Y.; Wei, Z. Application of Atosiban in Frozen-Thawed Cycle Patients with Different Times of Embryo Transfers. Gynecol. Endocrinol. 2016, 32, 811–815. [Google Scholar] [CrossRef]
- Tehraninejad, E.S.; Raisi, E.; Ghaleh, F.B.; Rashidi, B.H.; Aziminekoo, E.; Kalantari, V.; Haghollahi, F.; Shariat, M. The Sequential Embryo Transfer Compared to Blastocyst Embryo Transfer in in Vitro Fertilization (IVF) Cycle in Patients with the Three Repeated Consecutive IVF. A Randomized Controlled Trial. Gynecol. Endocrinol. 2019, 35, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Yu, M.; Zhang, X.-J. Effect of Hyaluronic Acid-Enriched Transfer Medium on Frozen-Thawed Embryo Transfer Outcomes. J. Obstet. Gynaecol. Res. 2018, 44, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Benkhalifa, M.; Demirol, A.; Sari, T.; Balashova, E.; Tsouroupaki, M.; Giakoumakis, Y.; Gurgan, T. Autologous Embryo-Cumulus Cells Co-Culture and Blastocyst Transfer in Repeated Implantation Failures: A Collaborative Prospective Randomized Study. Zygote 2012, 20, 173–180. [Google Scholar] [CrossRef]
- Delaroche, L.; Yazbeck, C.; Gout, C.; Kahn, V.; Oger, P.; Rougier, N. Intracytoplasmic Morphologically Selected Sperm Injection (IMSI) after Repeated IVF or ICSI Failures: A Prospective Comparative Study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 167, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Gao, Y.; Liu, W.; Liu, J.; Wu, L.; Xiong, S.; Zhu, J.; Han, W.; Wang, J.; Hao, X.; et al. Frozen Blastocyst Embryo Transfer vs. Frozen Cleavage-Stage Embryo Transfer in Couples with Recurrent Implantation Failure: A Cohort Study. Hum. Fertil. 2021, 24, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Levran, D.; Mashiach, S.; Dor, J.; Levron, J.; Farhi, J. Zygote Intrafallopian Transfer May Improve Pregnancy Rate in Patients with Repeated Failure of Implantation. Fertil. Steril. 1998, 69, 26–30. [Google Scholar] [CrossRef]
- Edirisinghe, W.R.; Ahnonkitpanit, V.; Promviengchai, S.; Suwajanakorn, S.; Pruksananonda, K.; Chinpilas, V.; Virutamasen, P. A Study Failing to Determine Significant Benefits from Assisted Hatching: Patients Selected for Advanced Age, Zonal Thickness of Embryos, and Previous Failed Attempts. J. Assist. Reprod. Genet. 1999, 16, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.; Bellver, J.; Rodrigo, L.; Bosch, E.; Mercader, A.; Vidal, C.; De los Santos, M.J.; Giles, J.; Labarta, E.; Domingo, J.; et al. Preimplantation Genetic Screening Using Fluorescence in Situ Hybridization in Patients with Repetitive Implantation Failure and Advanced Maternal Age: Two Randomized Trials. Fertil. Steril. 2013, 99, 1400–1407. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.-K.; Chen, H.-H.; Huang, C.-C.; Lee, C.-I.; Lin, P.-Y.; Lee, M.-S.; Lee, T.-H. Peripheral CD56(+)CD16(+) NK Cell Populations in the Early Follicular Phase Are Associated With Successful Clinical Outcomes of Intravenous Immunoglobulin Treatment in Women With Repeated Implantation Failure. Front. Endocrinol. 2019, 10, 937. [Google Scholar] [CrossRef]
- Li, J.; Chen, Y.; Liu, C.; Hu, Y.; Li, L. Intravenous Immunoglobulin Treatment for Repeated IVF/ICSI Failure and Unexplained Infertility: A Systematic Review and a Meta-Analysis. Am. J. Reprod. Immunol. 2013, 70, 434–447. [Google Scholar] [CrossRef]
- Li, J.; Mo, S.; Chen, Y. The Effect of G-CSF on Infertile Women Undergoing IVF Treatment: A Meta-Analysis. Syst. Biol. Reprod. Med. 2017, 63, 239–247. [Google Scholar] [CrossRef]
- Li, S.; Wang, J.; Cheng, Y.; Zhou, D.; Yin, T.; Xu, W.; Yu, N.; Yang, J. Intrauterine Administration of HCG-Activated Autologous Human Peripheral Blood Mononuclear Cells (PBMC) Promotes Live Birth Rates in Frozen/Thawed Embryo Transfer Cycles of Patients with Repeated Implantation Failure. J. Reprod. Immunol. 2017, 119, 15–22. [Google Scholar] [CrossRef]
- Nakagawa, K.; Kwak-Kim, J.; Ota, K.; Kuroda, K.; Hisano, M.; Sugiyama, R.; Yamaguchi, K. Immunosuppression with Tacrolimus Improved Reproductive Outcome of Women with Repeated Implantation Failure and Elevated Peripheral Blood TH1/TH2 Cell Ratios. Am. J. Reprod. Immunol. 2015, 73, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Aghajanzadeh, F.; Esmaeilzadeh, S.; Basirat, Z.; Mahouti, T.; Heidari, F.N.; Golsorkhtabaramiri, M. Using Autologous Intrauterine Platelet-Rich Plasma to Improve the Reproductive Outcomes of Women with Recurrent Implantation Failure. JBRA Assist. Reprod. 2020, 24, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Al-Zebeidi, J.; Agdi, M.; Lary, S.; Al-Obaid, S.; Salim, G.; Al-Jaroudi, D. Effect of Empiric Intravenous Intralipid Therapy on Pregnancy Outcome in Women with Unexplained Recurrent Implantation Failure Undergoing Intracytoplasmic Sperm Injection-Embryo Transfer Cycle: A Randomized Controlled Trial. Gynecol. Endocrinol. 2020, 36, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Wei, L.; Li, X.; Qin, A. Effects of Intrauterine Perfusion of Human Chorionic Gonadotropin in Women with Different Implantation Failure Numbers. Am. J. Reprod. Immunol. 2018, 79, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.A.; Eljabu, H.; Hopkisson, J.; Raine-Fenning, N.; Quenby, S.; Jayaprakasan, K. Aspirin and Heparin as Adjuvants during IVF Do Not Improve Live Birth Rates in Unexplained Implantation Failure. Reprod. Biomed. Online 2013, 26, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.A.; Eljabu, H.; Hopkisson, J.; Raine-Fenning, N.; Quenby, S.; Jayaprakasan, K.; Altmäe, S.; Mendoza-Tesarik, R.; Mendoza, C.; Mendoza, N.; et al. Effect of Growth Hormone on Uterine Receptivity in Women With Repeated Implantation Failure in an Oocyte Donation Program: A Randomized Controlled Trial. Fertil. Steril. 2018, 100, 818–824. [Google Scholar] [CrossRef]
- Berker, B.; Taşkin, S.; Kahraman, K.; Taşkin, E.A.; Atabekoğlu, C.; Sönmezer, M. The Role of Low-Molecular-Weight Heparin in Recurrent Implantation Failure: A Prospective, Quasi-Randomized, Controlled Study. Fertil. Steril. 2011, 95, 2499–2502. [Google Scholar] [CrossRef]
- Ruiz-Alonso, M.; Blesa, D.; Díaz-Gimeno, P.; Gómez, E.; Fernández-Sánchez, M.; Carranza, F.; Carrera, J.; Vilella, F.; Pellicer, A.; Simón, C. The Endometrial Receptivity Array for Diagnosis and Personalized Embryo Transfer as a Treatment for Patients with Repeated Implantation Failure. Fertil. Steril. 2013, 100, 818–824. [Google Scholar] [CrossRef]
- Siristatidis, C.; Dafopoulos, K.; El-Khayat, W.; Salamalekis, G.; Anifandis, G.; Vrantza, T.; Elsadek, M.; Papantoniou, N. Administration of Prednisolone and Low Molecular Weight Heparin in Patients with Repeated Implantation Failures: A Cohort Study. Gynecol. Endocrinol. 2018, 34, 136–139. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, L.; Martínez-Escribano, J.; Roo, E.; Pino, A.; Anitua, E. Plasma Rich in Growth Factor Gel as an Autologous Filler for Facial Volume Restoration. J. Cosmet. Dermatol. 2020, 19, 2552–2559. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Murias-Freijo, A.; Alkhraisat, M.H.; Orive, G. Clinical, Radiographical, and Histological Outcomes of Plasma Rich in Growth Factors in Extraction Socket: A Randomized Controlled Clinical Trial. Clin. Oral Investig. 2015, 19, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Fernández-de-Retana, S.; Alkhraisat, M.H. Platelet Rich Plasma in Oral and Maxillofacial Surgery from the Perspective of Composition. Platelets 2021, 32, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, M.; Azofra, J.; Anitua, E.; Andía, I.; Padilla, S.; Santisteban, J.; Mujika, I. Plasma Rich in Growth Factors to Treat an Articular Cartilage Avulsion: A Case Report. Med. Sci. Sports Exerc. 2003, 35, 1648–1652. [Google Scholar] [CrossRef]
- Padilla, S.; Sánchez, M.; Vaquerizo, V.; Malanga, G.A.; Fiz, N.; Azofra, J.; Rogers, C.J.; Samitier, G.; Sampson, S.; Seijas, R.; et al. Platelet-Rich Plasma Applications for Achilles Tendon Repair: A Bridge between Biology and Surgery. Int. J. Mol. Sci. 2021, 22, 824. [Google Scholar] [CrossRef]
- Sharara, F.I.; Lelea, L.-L.; Rahman, S.; Klebanoff, J.S.; Moawad, G.N. A Narrative Review of Platelet-Rich Plasma (PRP) in Reproductive Medicine. J. Assist. Reprod. Genet. 2021, 38, 1003–1012. [Google Scholar] [CrossRef]
- Cavalcante, M.B.; de Melo Bezerra Cavalcante, C.T.; Sarno, M.; Barini, R. Intrauterine Perfusion Immunotherapies in Recurrent Implantation Failures: Systematic Review. Am. J. Reprod. Immunol. 2020, 83, e13242. [Google Scholar] [CrossRef]
- Ferrari, A.R.; Cortrezzi, S.; Borges, E.J.; Braga, D.; do Carmo Borges de Souza, M.; de Azevedo Antunes, R. Evaluation of the Effects of Platelet-Rich Plasma on Follicular and Endometrial Growth: A Literature Review. JBRA Assist. Reprod. 2021, 25, 601–607. [Google Scholar] [CrossRef]
- Maleki-Hajiagha, A.; Razavi, M.; Rouholamin, S.; Rezaeinejad, M.; Maroufizadeh, S.; Sepidarkish, M. Intrauterine Infusion of Autologous Platelet-Rich Plasma in Women Undergoing Assisted Reproduction: A Systematic Review and Meta-Analysis. J. Reprod. Immunol. 2020, 137, 103078. [Google Scholar] [CrossRef]
- Busnelli, A.; Somigliana, E.; Cirillo, F.; Baggiani, A.; Levi-Setti, P.E. Efficacy of Therapies and Interventions for Repeated Embryo Implantation Failure: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 1747. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Park, Y.-B.; Ha, C.-W.; Roh, Y.J.; Park, J.-G. Adverse Reactions and Clinical Outcomes for Leukocyte-Poor Versus Leukocyte-Rich Platelet-Rich Plasma in Knee Osteoarthritis: A Systematic Review and Meta-Analysis. Orthop. J. Sports Med. 2021, 9, 23259671211011948. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Cui, X.; Li, S.; Ding, P.; Zhang, S.; Zhang, Y. Endometrial Thickness and IVF Cycle Outcomes: A Meta-Analysis. Reprod. Biomed. Online 2020, 40, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Kasius, A.; Smit, J.; Torrance, H.; Eijkemans, M.; Mol, B.; Opmeer, B.; Broekmans, F. Endometrial Thickness and Pregnancy Rates after IVF: A Systematic Review and Meta-Analysis. Hum. Reprod. Update 2014, 20, 530–541. [Google Scholar] [CrossRef]
- Ribeiro, V.C.; Santos-Ribeiro, S.; de Munck, N.; Drakopoulos, P.; Polyzos, N.P.; Schutyser, V.; Verheyen, G.; Tournaye, H.; Blockeel, C. Should We Continue to Measure Endometrial Thickness in Modern-Day Medicine? The Effect on Live Birth Rates and Birth Weight. Reprod. Biomed. Online 2018, 36, 416–426. [Google Scholar] [CrossRef]
- Yuan, X.; Saravelos, S.H.; Wang, Q.; Xu, Y.; Li, T.-C.; Zhou, C. Endometrial Thickness as a Predictor of Pregnancy Outcomes in 10787 Fresh IVF-ICSI Cycles. Reprod. Biomed. Online 2016, 33, 197–205. [Google Scholar] [CrossRef]
- Kirshenbaum, M.; Orvieto, R. Should We Offer In Vitro Fertilization to Couples with Unexplained Recurrent Pregnancy Loss? J. Clin. Med. 2019, 8, 2001. [Google Scholar] [CrossRef]
- Lin, Y.; Qi, J.; Sun, Y. Platelet-Rich Plasma as a Potential New Strategy in the Endometrium Treatment in Assisted Reproductive Technology. Front. Endocrinol. 2021, 12, 707584. [Google Scholar] [CrossRef]
- Lange-Consiglio, A.; Cazzaniga, N.; Garlappi, R.; Spelta, C.; Pollera, C.; Perrini, C.; Cremonesi, F. Platelet Concentrate in Bovine Reproduction: Effects on in Vitro Embryo Production and after Intrauterine Administration in Repeat Breeder Cows. Reprod. Biol. Endocrinol. 2015, 13, 65. [Google Scholar] [CrossRef]
- de Miguel-Gómez, L.; López-Martínez, S.; Campo, H.; Francés-Herrero, E.; Faus, A.; Díaz, A.; Pellicer, A.; Domínguez, F.; Cervelló, I. Comparison of Different Sources of Platelet-Rich Plasma as Treatment Option for Infertility-Causing Endometrial Pathologies. Fertil. Steril. 2021, 115, 490–500. [Google Scholar] [CrossRef]
- Marini, M.G.; Perrini, C.; Esposti, P.; Corradetti, B.; Bizzaro, D.; Riccaboni, P.; Fantinato, E.; Urbani, G.; Gelati, G.; Cremonesi, F.; et al. Effects of Platelet-Rich Plasma in a Model of Bovine Endometrial Inflammation in Vitro. Reprod. Biol. Endocrinol. 2016, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Segabinazzi, L.G.; Friso, A.M.; Correal, S.B.; Crespilho, A.M.; Dell’Aqua, J.A.J.; Miró, J.; Papa, F.O.; Alvarenga, M.A. Uterine Clinical Findings, Fertility Rate, Leucocyte Migration, and COX-2 Protein Levels in the Endometrial Tissue of Susceptible Mares Treated with Platelet-Rich Plasma before and after AI. Theriogenology 2017, 104, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.Y.; Myoung, S.M.; Choe, J.M.; Kim, T.; Cheon, Y.P.; Kim, Y.M.; Park, H. Effects of Autologous Platelet-Rich Plasma on Regeneration of Damaged Endometrium in Female Rats. Yonsei Med. J. 2017, 58, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Reghini, M.F.S.; Ramires Neto, C.; Segabinazzi, L.G.; Castro Chaves, M.M.B.; de Paula, F. Dell’Aqua, C.; Bussiere, M.C.C.; Dell’Aqua, J.A.J.; Papa, F.O.; Alvarenga, M.A. Inflammatory Response in Chronic Degenerative Endometritis Mares Treated with Platelet-Rich Plasma. Theriogenology 2016, 86, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Park, M.; Paek, J.Y.; Lee, W.S.; Song, H.; Lyu, S.W. Intrauterine Infusion of Human Platelet-Rich Plasma Improves Endometrial Regeneration and Pregnancy Outcomes in a Murine Model of Asherman’s Syndrome. Front. Physiol. 2020, 11, 105. [Google Scholar] [CrossRef]
- Thomas, M.R.; Storey, R.F. The Role of Platelets in Inflammation. Thromb. Haemost. 2015, 114, 449–458. [Google Scholar] [CrossRef]
- Herter, J.M.; Rossaint, J.; Zarbock, A. Platelets in Inflammation and Immunity. J. Thromb. Haemost. 2014, 12, 1764–1775. [Google Scholar] [CrossRef]
- Assinger, A.; Laky, M.; Schabbauer, G.; Hirschl, A.M.; Buchberger, E.; Binder, B.R.; Volf, I. Efficient Phagocytosis of Periodontopathogens by Neutrophils Requires Plasma Factors, Platelets and TLR2. J. Thromb. Haemost. 2011, 9, 799–809. [Google Scholar] [CrossRef]
- Page, C.; Pitchford, S. Neutrophil and Platelet Complexes and Their Relevance to Neutrophil Recruitment and Activation. Int. Immunopharmacol. 2013, 17, 1176–1184. [Google Scholar] [CrossRef]
- van Gils, J.M.; Zwaginga, J.J.; Hordijk, P.L. Molecular and Functional Interactions among Monocytes, Platelets, and Endothelial Cells and Their Relevance for Cardiovascular Diseases. J. Leukoc. Biol. 2009, 85, 195–204. [Google Scholar] [CrossRef]
- Wetterslev, J.; Jakobsen, J.C.; Gluud, C. Trial Sequential Analysis in Systematic Reviews with Meta-Analysis. BMC Med. Res. Methodol. 2017, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Tandulwadkar, S.R.; Naralkar, M.V.; Surana, A.D.; Selvakarthick, M.; Kharat, A.H. Autologous Intrauterine Platelet-Rich Plasma Instillation for Suboptimal Endometrium in Frozen Embryo Transfer Cycles: A Pilot Study. J. Hum. Reprod. Sci. 2017, 10, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Zadehmodarres, S.; Salehpour, S.; Saharkhiz, N.; Nazari, L. Treatment of thin endometrium with autologous platelet-rich plasma: A pilot study. JBRA Assist. Reprod. 2017, 21, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G.V.L.; Fanton, V.; Sosa, D.; Criado Scholz, E.; Lotti, J.; Aragona, S.E.; Lotti, T. Use of platelet rich plasma in human infertility. J. Biol. Regul. Homeost. Agents 2017, 31, 179–182. [Google Scholar]
- Molina, A.; Sánchez, J.; Sánchez, W.; Vielma, V. Platelet-rich plasma as an adjuvant in the endometrial preparation of patients with refractory endometrium. JBRA Assist. Reprod. 2018, 22, 42–48. [Google Scholar] [CrossRef]
- Kim, H.; Shin, J.E.; Koo, H.S.; Kwon, H.; Choi, D.H.; Kim, J.H. Effect of Autologous Platelet-Rich Plasma Treatment on Refractory Thin Endometrium During the Frozen Embryo Transfer Cycle: A Pilot Study. Front. Endocrinol. 2019, 10, 61. [Google Scholar] [CrossRef]
- Chang, Y.; Li, J.; Wei, L.N.; Pang, J.; Chen, J.; Liang, X. Autologous platelet-rich plasma infusion improves clinical pregnancy rate in frozen embryo transfer cycles for women with thin endometrium. Medicine 2019, 98, e14062. [Google Scholar] [CrossRef]
- Frantz, N.; Ferreira, M.; Kulmann, M.I.; Frantz, G.; Bos-Mikich, A.; Oliveira, R. Platelet-Rich plasma as an effective alternative approach for improving endometrial receptivity—A clinical retrospective study. J. Bras. Reprod. Assist 2020, 24, 442–446. [Google Scholar] [CrossRef]
- Tehraninejad, E.S.; Kashani, N.G.; Hosseini, A.; Tarafdari, A. Autologous platelet-rich plasma infusion does not improve pregnancy outcomes in frozen embryo transfer cycles in women with history of repeated implantation failure without thin endometrium. J. Obstet. Gynaecol. Res. 2021, 47, 147–151. [Google Scholar] [CrossRef]
- Godha, Z.; Nayar, K.D.; Gupta, S.; Singh, M.; Gupta, M.; Kant, G.; Nayar, K. Randomized controlled trial of intrauterine infusion of autologous platelet rich plasma (PRP) versus granulocyte colony stimulating factor (G-CSF) in thin endometrium in frozen embryo transfer. In Human Reproduction; Oxford University Press: Oxford, UK, 2019; Volume 34, p. 277. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).