Anti-Apoptosis Therapy for Meniscal Avascular Zone Repair: A Proof-of-Concept Study in a Lapine Model
Abstract
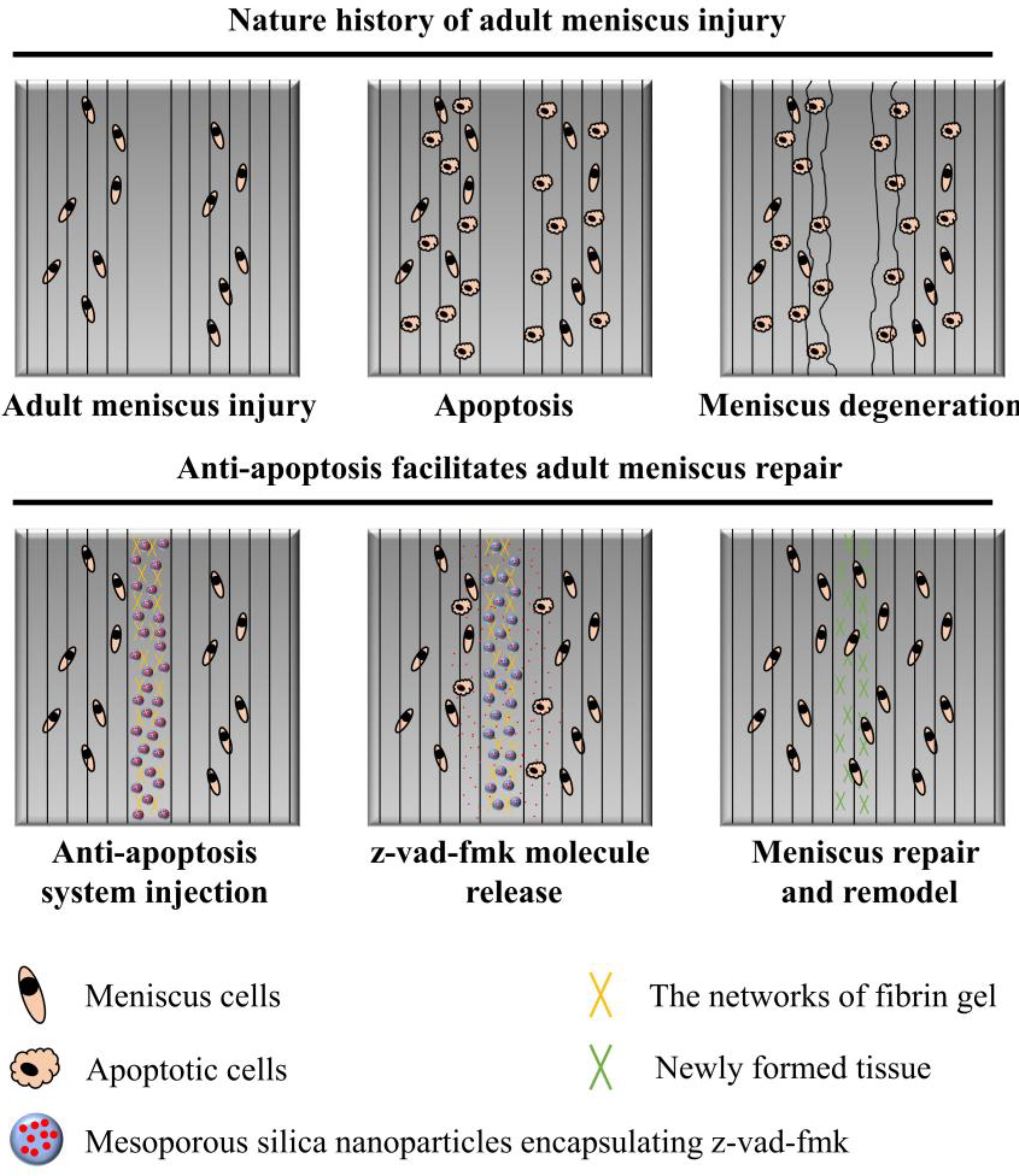
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Surgical Procedure
2.3. Fabrication of z-Vad-Fmk Encapsulated Aminated MSNs
2.4. Sample Collection and Processing
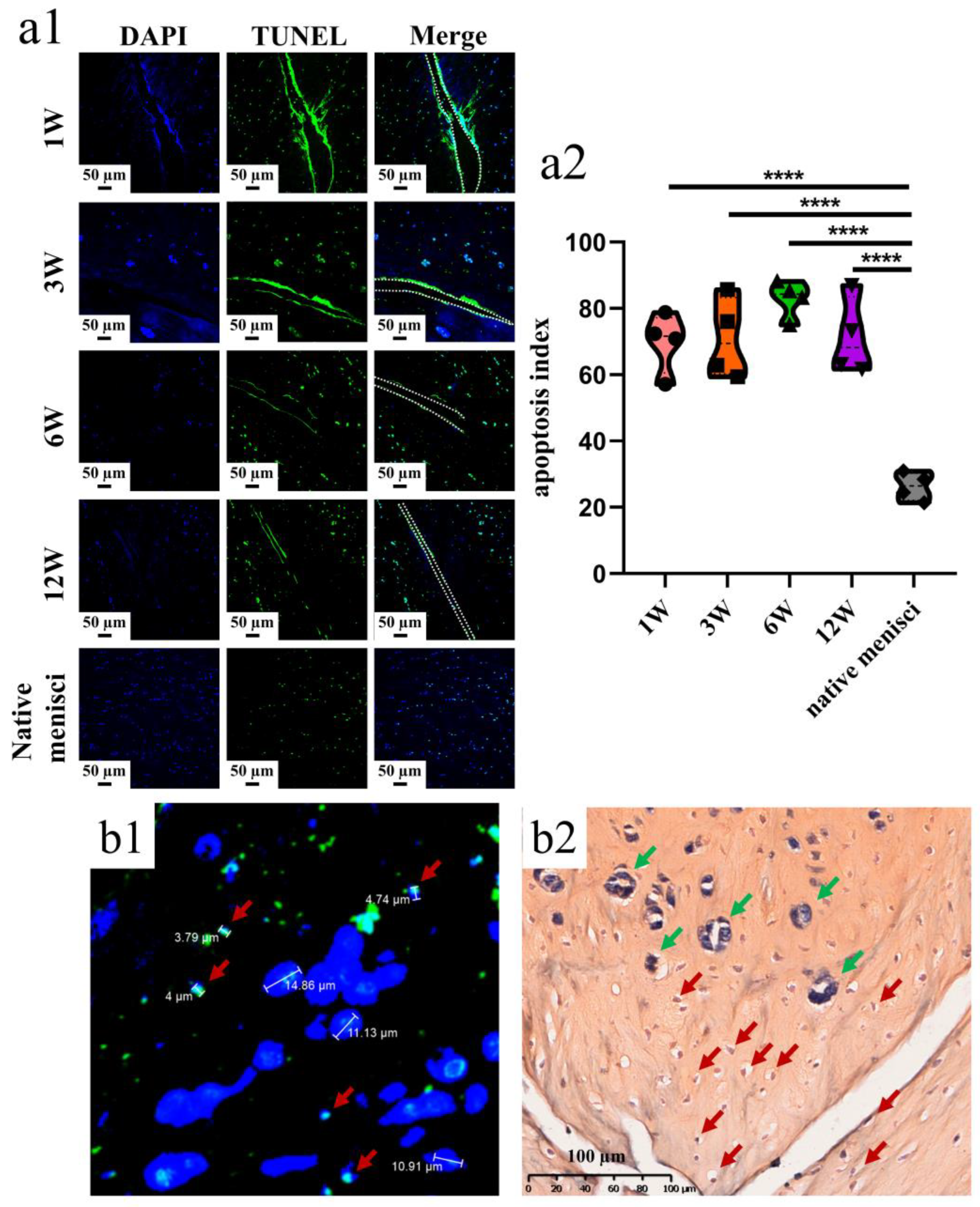
2.5. Apoptosis Tests
2.6. Tissue Immunofluorescence and Immunohistochemical Staining
2.7. Statistical Analysis
3. Results
3.1. Apoptosis Examination after Untreated Adult Meniscal Tears
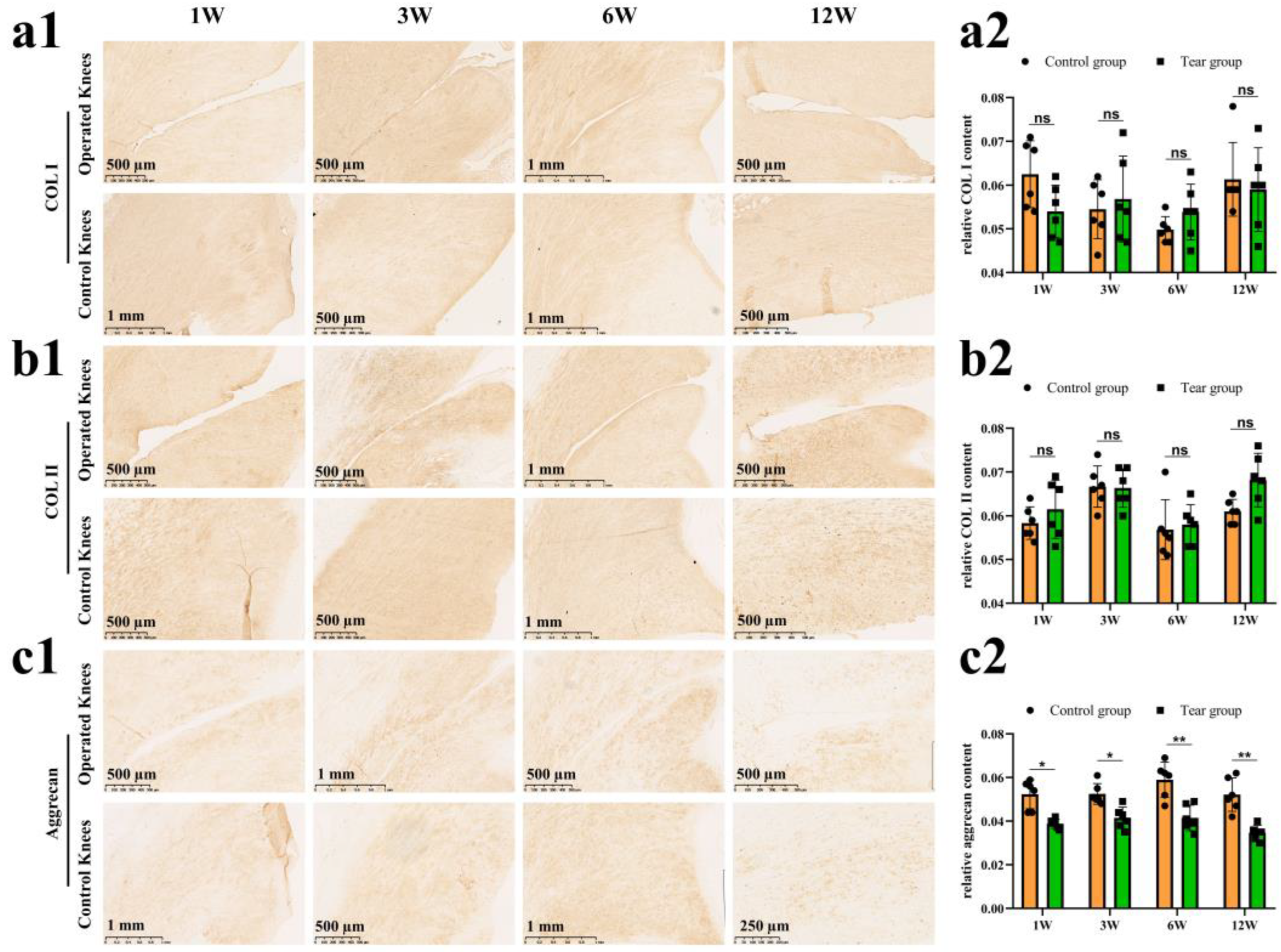
3.2. Meniscal Matrix Degradation after Untreated Adult Meniscal Tears
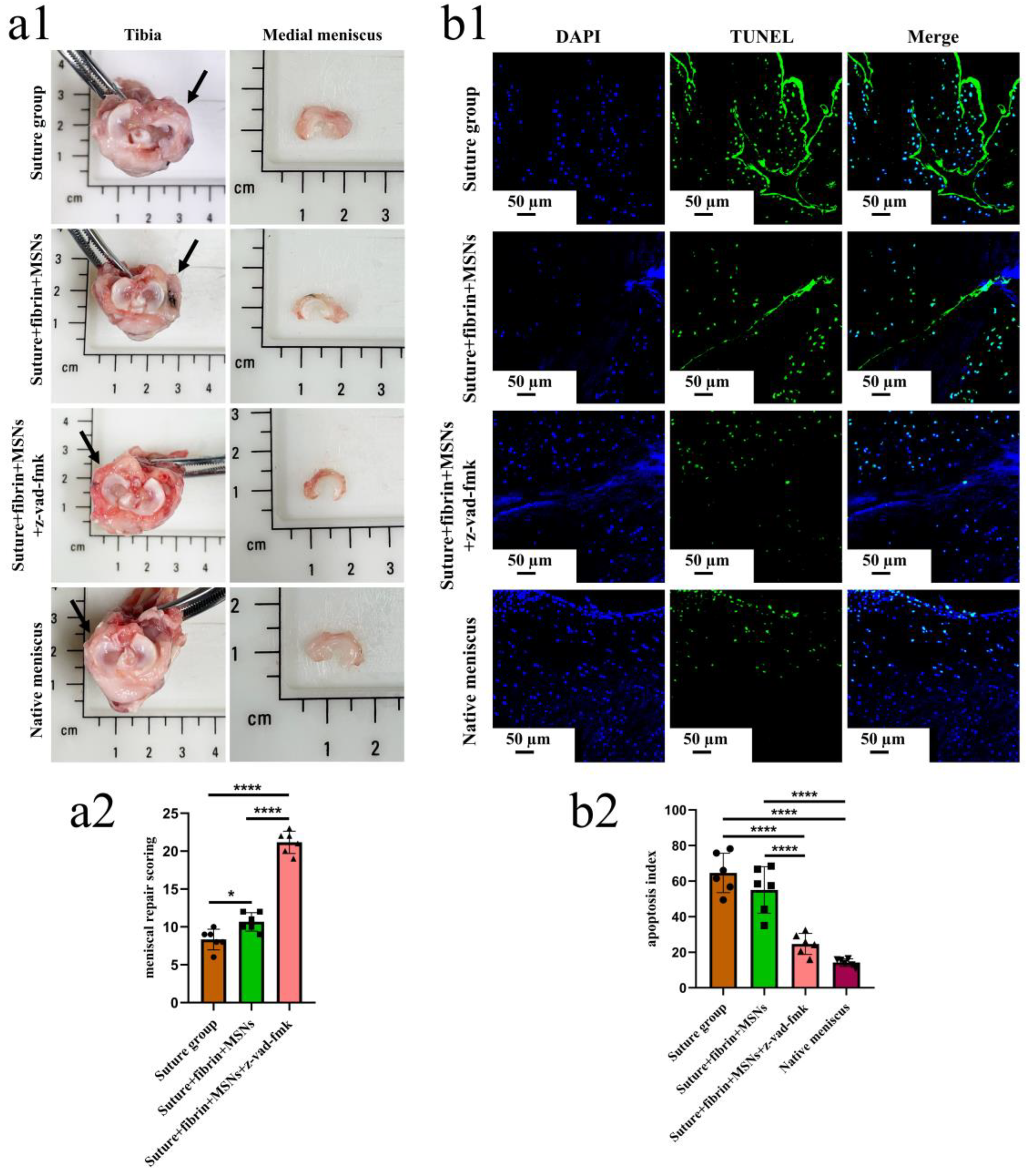
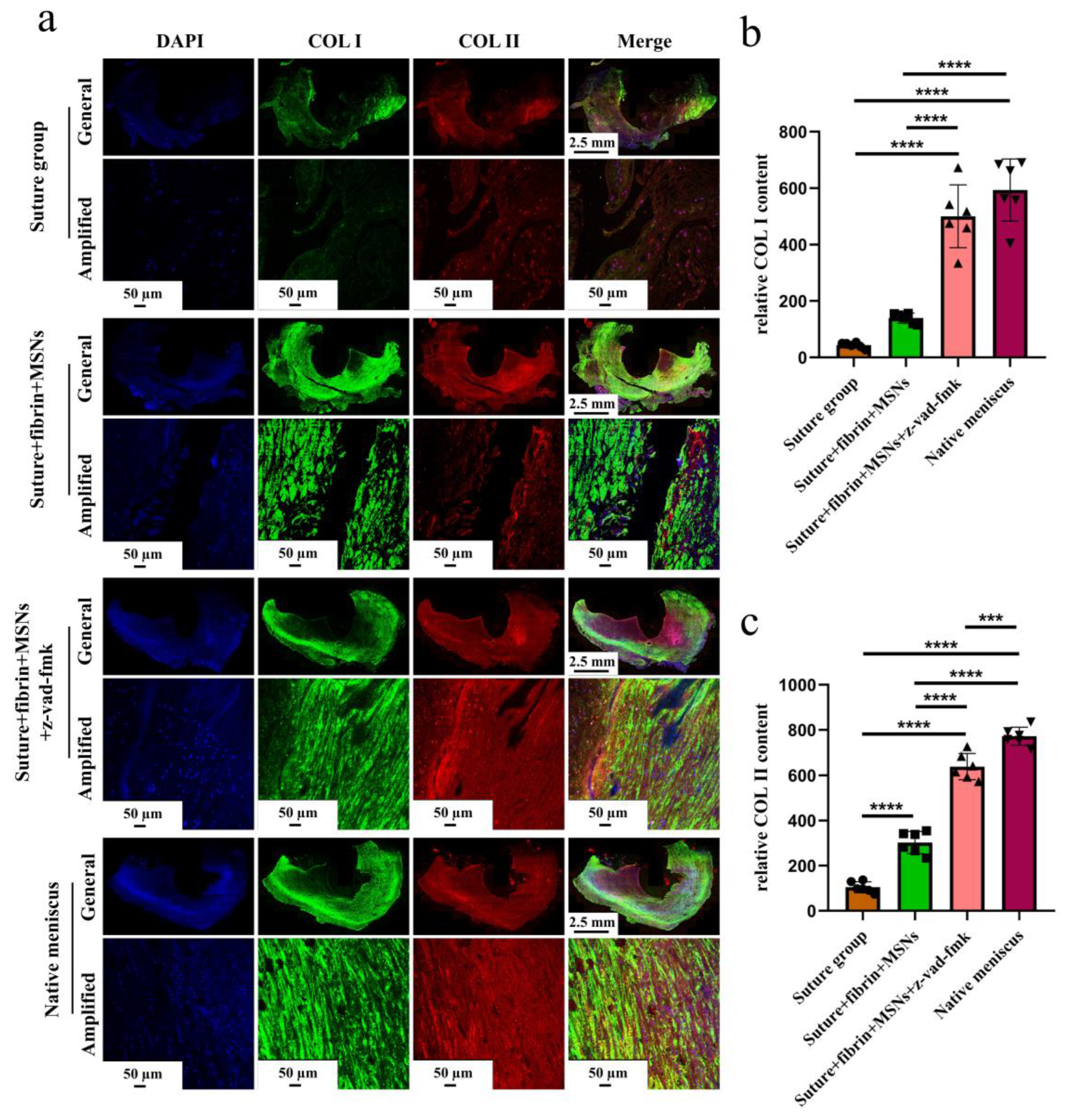
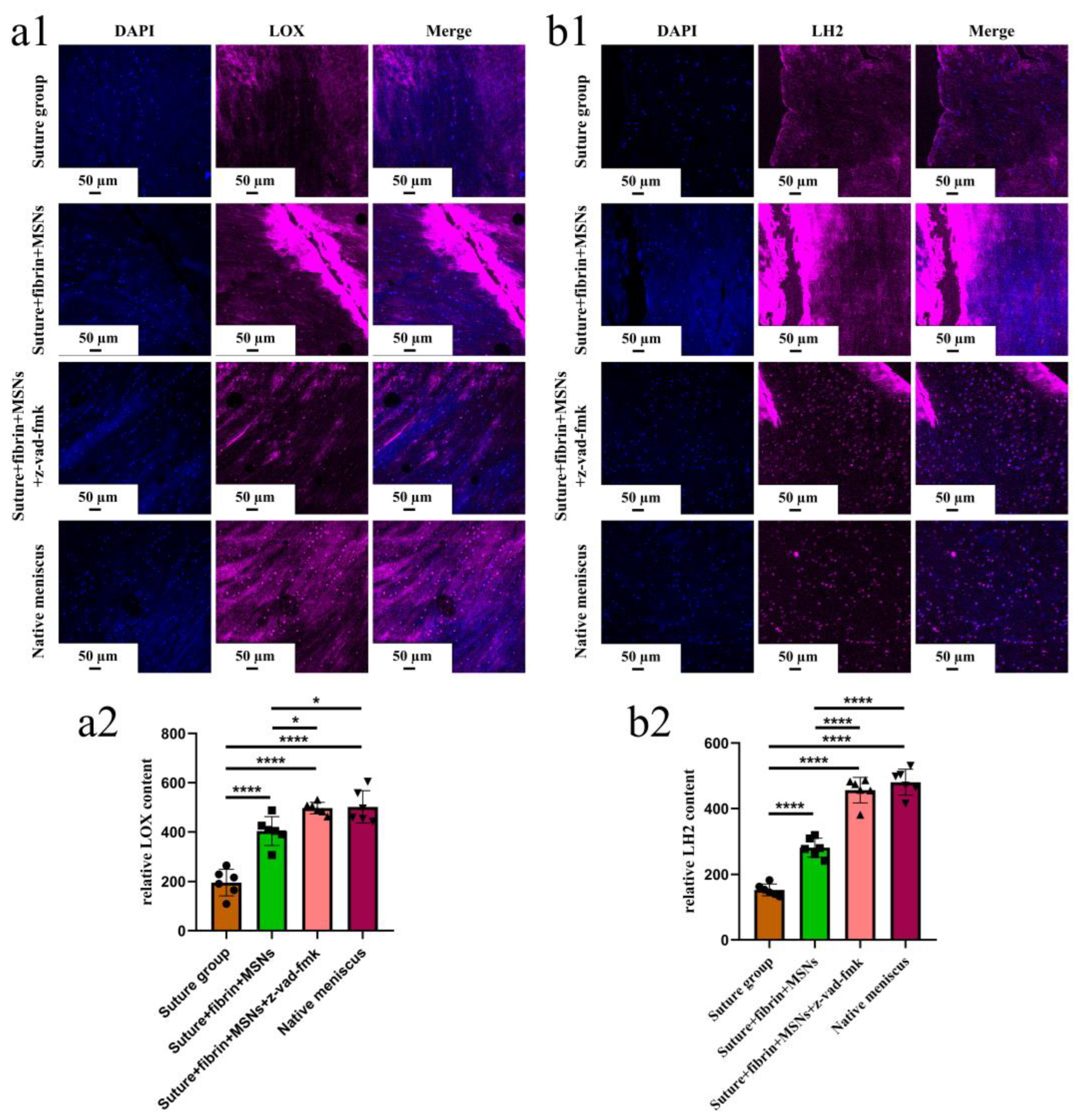
3.3. Sustained Release of z-Vad-Fmk Alleviated Apoptosis and Enhanced Adult Meniscal Repair
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Makris, E.A.; Hadidi, P.; Athanasiou, K.A. The knee meniscus: Structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials 2011, 32, 7411–7431. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Bosque, J.; Meehan, J.P.; Jamali, A.; Marder, R. Increase in outpatient knee arthroscopy in the United States: A comparison of National Surveys of Ambulatory Surgery, 1996 and 2006. J. Bone Jt. Surg. Am. 2011, 93, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Logerstedt, D.S.; Scalzitti, D.A.; Bennell, K.L.; Hinman, R.S.; Silvers-Granelli, H.; Ebert, J.; Hambly, K.; Carey, J.L.; Snyder-Mackler, L.; Axe, M.J.; et al. Knee Pain and Mobility Impairments: Meniscal and Articular Cartilage Lesions Revision 2018. J. Orthop. Sports Phys. Ther. 2018, 48, A1–A50. [Google Scholar] [CrossRef] [PubMed]
- Gee, S.M.; Tennent, D.J.; Cameron, K.L.; Posner, M.A. The Burden of Meniscus Injury in Young and Physically Active Populations. Clin. Sports Med. 2020, 39, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.; Graham, W.; Best, T.M.; Collins, C.; Currie, D.W.; Comstock, R.D.; Flanigan, D.C. Epidemiology of meniscal injuries in US high school athletes between 2007 and 2013. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Arandjelovic, S.; Ravichandran, K.S. Phagocytosis of apoptotic cells in homeostasis. Nat. Immunol. 2015, 16, 907–917. [Google Scholar] [CrossRef]
- Majno, G.; Joris, I. Apoptosis, oncosis, and necrosis. An overview of cell death. Am. J. Pathol. 1995, 146, 3–15. [Google Scholar] [PubMed]
- Rudin, C.M.; Thompson, C.B. Apoptosis and disease: Regulation and clinical relevance of programmed cell death. Annu. Rev. Med. 1997, 48, 267–281. [Google Scholar] [CrossRef]
- Hashimoto, S.; Takahashi, K.; Ochs, R.L.; Coutts, R.D.; Amiel, D.; Lotz, M. Nitric oxide production and apoptosis in cells of the meniscus during experimental osteoarthritis. Arthritis Rheum. 1999, 42, 2123–2131. [Google Scholar] [CrossRef]
- Hashimoto, S.; Ochs, R.L.; Rosen, F.; Quach, J.; McCabe, G.; Solan, J.; Seegmiller, J.E.; Terkeltaub, R.; Lotz, M. Chondrocyte-derived apoptotic bodies and calcification of articular cartilage. Proc. Natl. Acad. Sci. USA 1998, 95, 3094–3099. [Google Scholar] [CrossRef]
- Uysal, M.; Akpinar, S.; Bolat, F.; Cekin, N.; Cinar, M.; Cesur, N. Apoptosis in the traumatic and degenerative tears of human meniscus. Knee Surg. Sports Traumatol. Arthrosc. 2008, 16, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Alenzi, F.Q. Apoptosis and diseases: Regulation and clinical relevance. Saudi Med. J. 2005, 26, 1679–1690. [Google Scholar]
- Kim, H.A.; Lee, Y.J.; Seong, S.C.; Choe, K.W.; Song, Y.W. Apoptotic chondrocyte death in human osteoarthritis. J. Rheumatol. 2000, 27, 455–462. [Google Scholar] [PubMed]
- Rai, M.F.; Patra, D.; Sandell, L.J.; Brophy, R.H. Transcriptome analysis of injured human meniscus reveals a distinct phenotype of meniscus degeneration with aging. Arthritis Rheum. 2013, 65, 2090–2101. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Mishima, H.; Hashimoto, S.; Goomer, R.S.; Harwood, F.L.; Lotz, M.; Moriya, H.; Amiel, D. Chondrocyte apoptosis and regional differential expression of nitric oxide in the medial meniscus following partial meniscectomy. J. Orthop. Res. 2001, 19, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Thornberry, N.A.; Lazebnik, Y. Caspases: Enemies within. Science 1998, 281, 1312–1316. [Google Scholar] [CrossRef] [PubMed]
- Fischer, U.; Jänicke, R.U.; Schulze-Osthoff, K. Many cuts to ruin: A comprehensive update of caspase substrates. Cell Death Differ. 2003, 10, 76–100. [Google Scholar] [CrossRef]
- Costouros, J.G.; Dang, A.C.; Kim, H.T. Inhibition of chondrocyte apoptosis in vivo following acute osteochondral injury. Osteoarthr. Cartil. 2003, 11, 756–759. [Google Scholar] [CrossRef] [PubMed]
- Lo, M.Y.; Kim, H.T. Chondrocyte apoptosis induced by collagen degradation: Inhibition by caspase inhibitors and IGF-1. J. Orthop. Res. 2004, 22, 140–144. [Google Scholar] [CrossRef]
- Slee, E.A.; Zhu, H.; Chow, S.C.; MacFarlane, M.; Nicholson, D.W.; Cohen, G.M. Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone (Z-VAD.FMK) inhibits apoptosis by blocking the processing of CPP32. Biochem. J. 1996, 315 Pt 1, 21–24. [Google Scholar] [CrossRef]
- Yan, W.; Maimaitimin, M.; Zhao, F.; Fan, Y.; Yang, S.; Li, Y.; Cao, C.; Shao, Z.; Liu, Z.; Hu, X.; et al. The transplantation of particulated juvenile allograft cartilage and synovium for the repair of meniscal defect in a lapine model. J. Orthop. Translat. 2022, 33, 72–89. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Dai, W.; Cheng, J.; Fan, Y.; Zhao, F.; Li, Y.; Maimaitimin, M.; Cao, C.; Shao, Z.; Li, Q.; et al. Histologically Confirmed Recellularization is a Key Factor that Affects Meniscal Healing in Immature and Mature Meniscal Tears. Front. Cell Dev. Biol. 2021, 9, 793820. [Google Scholar] [CrossRef]
- Kim, H.A.; Suh, D.I.; Song, Y.W. Relationship between chondrocyte apoptosis and matrix depletion in human articular cartilage. J. Rheumatol. 2001, 28, 2038–2045. [Google Scholar] [PubMed]
- Zhang, X.B.; Hu, Y.C.; Cheng, P.; Zhou, H.Y.; Chen, X.Y.; Wu, D.; Zhang, R.H.; Yu, D.C.; Gao, X.D.; Shi, J.T.; et al. Targeted therapy for intervertebral disc degeneration: Inhibiting apoptosis is a promising treatment strategy. Int. J. Med. Sci. 2021, 18, 2799–2813. [Google Scholar] [CrossRef] [PubMed]
- Boraldi, F.; Lofaro, F.D.; Quaglino, D. Apoptosis in the Extraosseous Calcification Process. Cells 2021, 10, 131. [Google Scholar] [CrossRef]
- Zamli, Z.; Sharif, M. Chondrocyte apoptosis: A cause or consequence of osteoarthritis? Int. J. Rheum. Dis. 2011, 14, 159–166. [Google Scholar] [CrossRef]
- Gonzalez-Leon, E.A.; Bielajew, B.J.; Hu, J.C.; Athanasiou, K.A. Engineering self-assembled neomenisci through combination of matrix augmentation and directional remodeling. Acta Biomater. 2020, 109, 73–81. [Google Scholar] [CrossRef]
- Remst, D.F.; Blaney Davidson, E.N.; Vitters, E.L.; Blom, A.B.; Stoop, R.; Snabel, J.M.; Bank, R.A.; van den Berg, W.B.; van der Kraan, P.M. Osteoarthritis-related fibrosis is associated with both elevated pyridinoline cross-link formation and lysyl hydroxylase 2b expression. Osteoarthr. Cartil. 2013, 21, 157–164. [Google Scholar] [CrossRef]
- Dang, A.C.; Warren, A.P.; Kim, H.T. Beneficial effects of intra-articular caspase inhibition therapy following osteochondral injury. Osteoarthr. Cartil. 2006, 14, 526–532. [Google Scholar] [CrossRef]






| Abbreviations | Definition |
|---|---|
| z-vad-fmk | Benzyloxycarbonyl-Val-Ala-Asp-fluoromethyl ketone |
| GAG | Glycosaminoglycans |
| COL I | Type I collagen |
| COL II | Type II collagen |
| SOX9 | Sry-type HMG-box 9 |
| ECM | Extracellular matrix |
| caspase | Cysteinyl aspartate-specific proteases |
| MSNs | Mesoporous silica nanoparticles |
| PBS | Phosphate-buffered solution |
| HE | Hematoxylin-eosin |
| DMSO | Dimethylsulfoxide |
| IHC | Immunohistochemical |
| MFC | Medial femoral condyle |
| MTP | Medial tibial plateau |
| OARSI | Osteoarthritis Research Society International |
| LOX | Lysyl oxidase |
| LH | Lysyl hydroxylase |
| DAPI | 4′,6-diamidino2-phenylindole |
| ROI | Region of interest |
| HRP | Horseradish peroxidase |
| DAB | Diaminobenzidine |
| SD | Standard deviation |
| ANOVA | Analysis of variance |
| ns | No significant difference |
| IDD | Intervertebral disc degeneration |
| Reagent or Resource | Source | Identifier |
|---|---|---|
| Primary antibodies | ||
| Mouse anti collagen II monoclonal antibody | Invitrogen, Rockford, IL, USA | MA5-13026 |
| Rabbit anti SOX9 antibody | Sigma, Darmstadt, Germany | HPA001758 |
| Goat anti collagen I antibody | Arigo, China | ARG21965 |
| Mouse anti aggrecan antibody | MilliporeSigma, Darmstadt, Germany | C8035 |
| Rabbit anti LOX polyclonal antibody | Proteintech, Wuhan, China | 17958-1-AP |
| Rabbit anti PLODA2 (LH2) antibody | Proteintech, Wuhan, China | 21214-1-AP |
| Secondary antibodies | ||
| Donkey anti-rabbit IgG H & L (Alexa Fluor® 647) | Abcam, Cambridge, UK | ab150075 |
| Donkey anti-mouse IgG H & L (Alexa Fluor® 594) | Abcam, Cambridge, UK | ab150108 |
| Donkey anti-goat IgG H & L (Alexa Fluor® 488) | Abcam, Cambridge, UK | ab150129 |
| Goat anti-mouse IgG H & L (HRP) | Abcam, Cambridge, UK | ab6789 |
| Donkey anti-Goat IgG H & L (HRP) | Abcam, Cambridge, UK | ab6885 |
| Chemicals and reagent | ||
| z-vad-fmk | MCE, Monmouth Junction, NJ, USA | HY-16658B |
| TUNEL assay | Promega, Madison, MI, USA | G3250 |
| Software | ||
| GraphPad Prism software | GraphPad | version 8.0.1 |
| Image J software | NIH | version 1.8.0 |
| G*Power software | G*Power | version 3.1.9.2 |
| 0 | 1 | 2 | 3 | |
|---|---|---|---|---|
| Defect filling | No fill | <25% | 25–75% | >75% |
| Surface | No surface | Ruptured | Fissured/fibrillated | Meniscus-like |
| Integration | No integration | Partial, unilateral integration | Bilateral partial or unilateral complete integration | Bilateral complete integration |
| Cellularity | No cells | >10 cell cluster/slide | No cell cluster/slide, Cell–ECM ratio > 0.5 | Meniscus-like cell–ECM ratio |
| Cell morphology | No cells | <25% Meniscus-like cells | 25–75% Meniscus-like cells | >75% Meniscus-like cells |
| Content of proteoglycan | No staining for proteoglycan | <25% | 25–75% | >75% |
| Content of type 2 collagen | No staining for type 2 collagen | <25% | 25–75% | >75% |
| Stability | No stability | Weak | Stable in shape | Stable to pressure and pulling stress |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, W.; Wu, Y.; Zhao, F.; Dai, R.; Zhou, Y.; Liu, D.; Cheng, J.; Hu, X.; Ao, Y. Anti-Apoptosis Therapy for Meniscal Avascular Zone Repair: A Proof-of-Concept Study in a Lapine Model. Bioengineering 2023, 10, 1422. https://doi.org/10.3390/bioengineering10121422
Yan W, Wu Y, Zhao F, Dai R, Zhou Y, Liu D, Cheng J, Hu X, Ao Y. Anti-Apoptosis Therapy for Meniscal Avascular Zone Repair: A Proof-of-Concept Study in a Lapine Model. Bioengineering. 2023; 10(12):1422. https://doi.org/10.3390/bioengineering10121422
Chicago/Turabian StyleYan, Wenqiang, Yue Wu, Fengyuan Zhao, Ruilan Dai, Yunan Zhou, Dingge Liu, Jin Cheng, Xiaoqing Hu, and Yingfang Ao. 2023. "Anti-Apoptosis Therapy for Meniscal Avascular Zone Repair: A Proof-of-Concept Study in a Lapine Model" Bioengineering 10, no. 12: 1422. https://doi.org/10.3390/bioengineering10121422
APA StyleYan, W., Wu, Y., Zhao, F., Dai, R., Zhou, Y., Liu, D., Cheng, J., Hu, X., & Ao, Y. (2023). Anti-Apoptosis Therapy for Meniscal Avascular Zone Repair: A Proof-of-Concept Study in a Lapine Model. Bioengineering, 10(12), 1422. https://doi.org/10.3390/bioengineering10121422








