Addressing Glaucoma in Myopic Eyes: Diagnostic and Surgical Challenges
Abstract
:1. Introduction
2. Pathophysiology of Glaucoma in Myopic Eyes
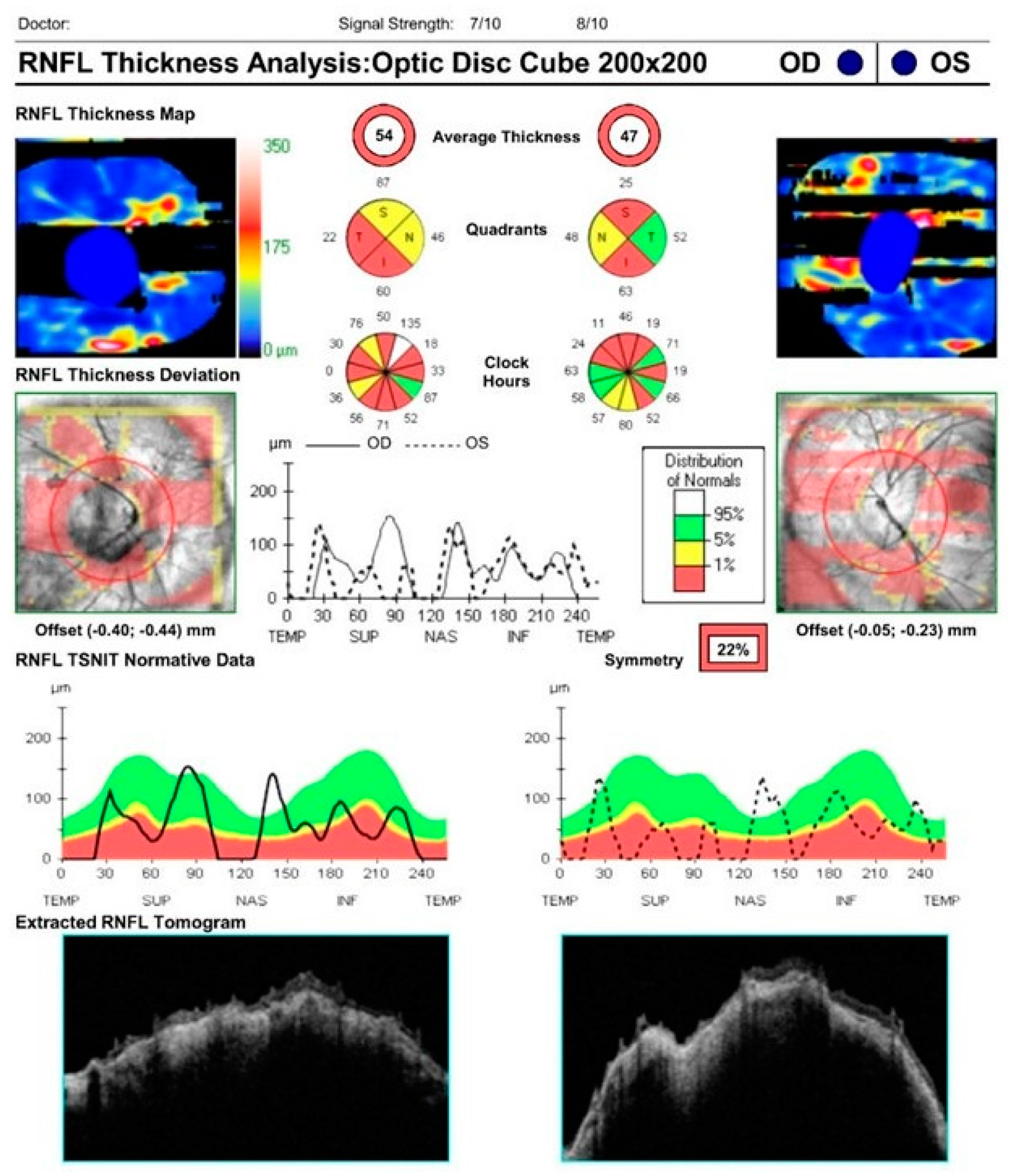
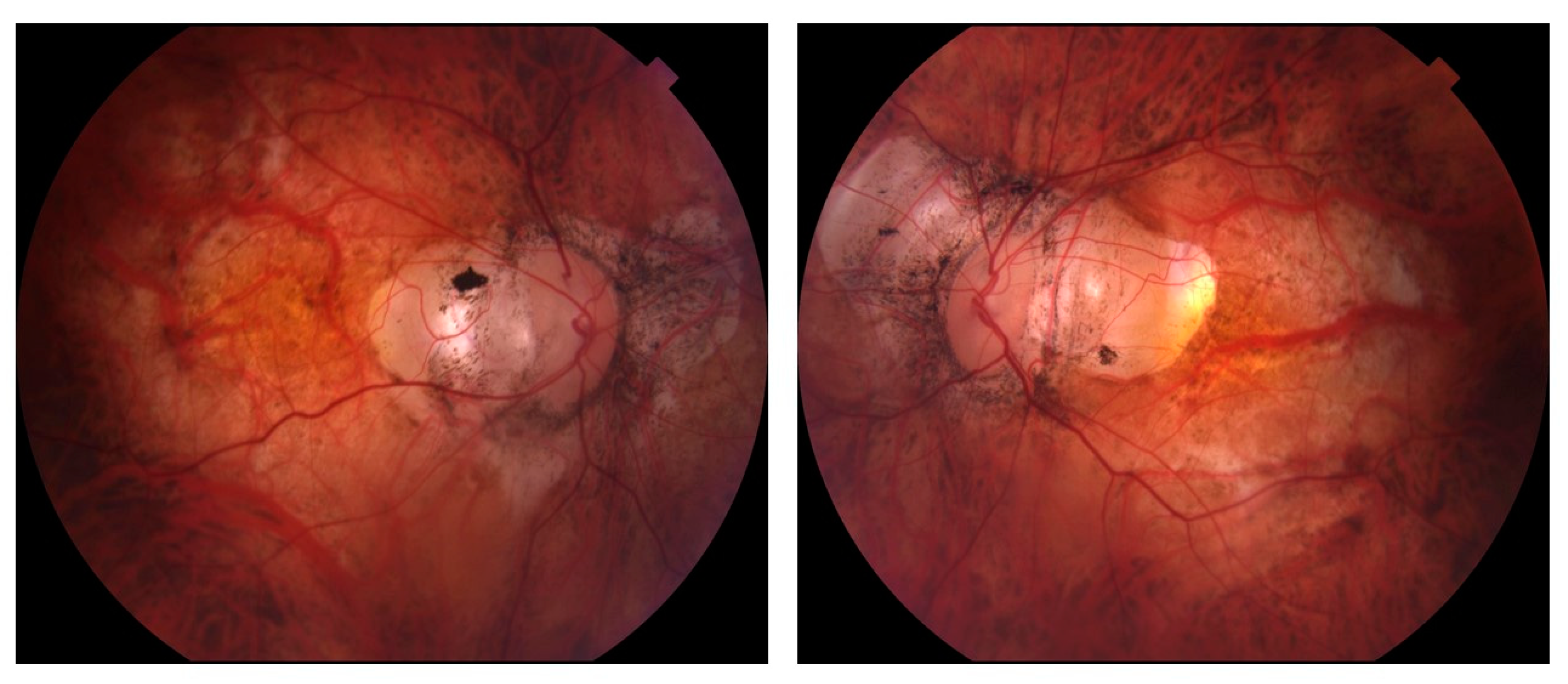
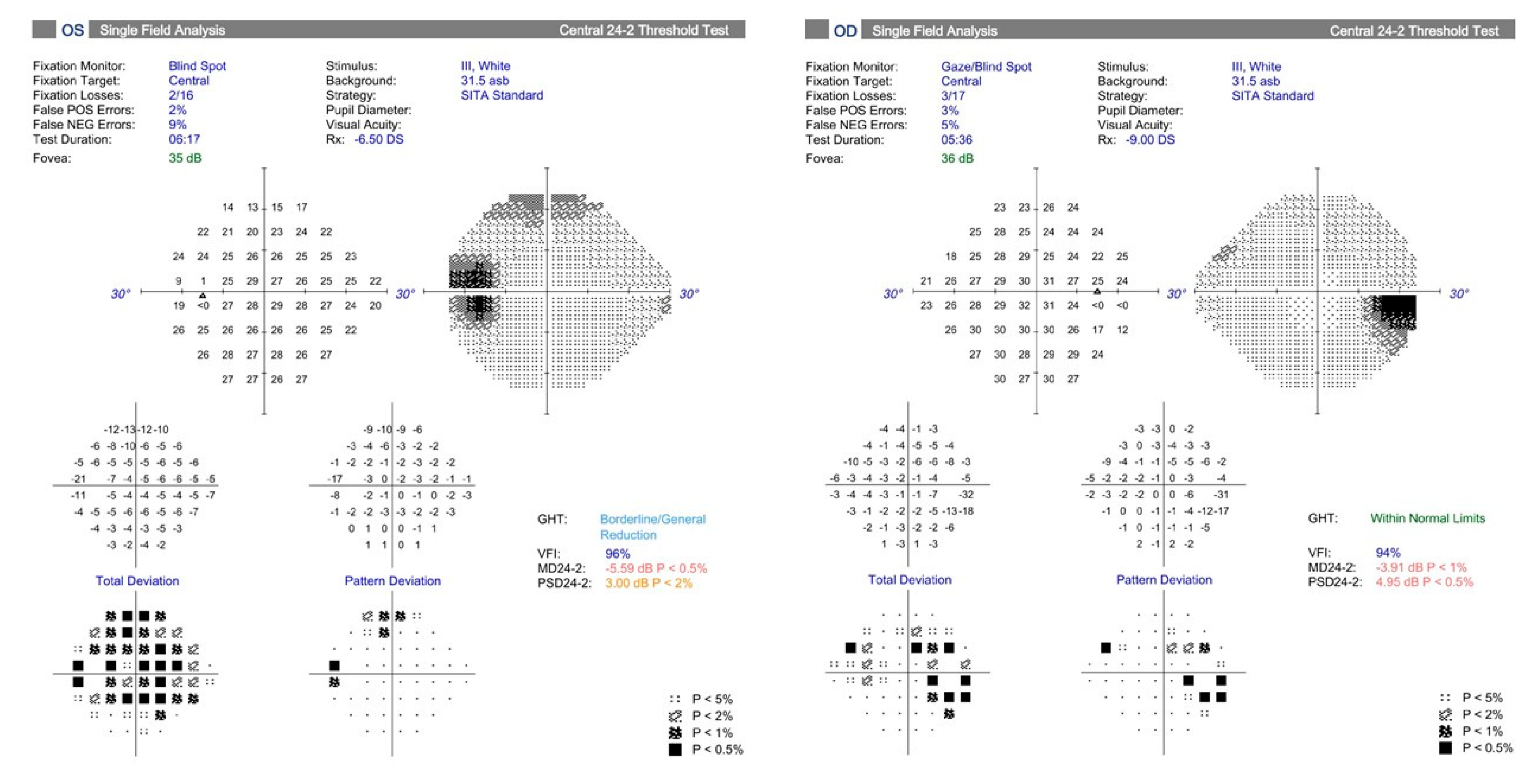
3. Diagnostic Structural and Functional Testing of Glaucoma in Myopic Eyes
4. Surgical Considerations in Myopic Eyes
5. Conclusions
6. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Morgan, I.G.; Ohno-Matsui, K.; Saw, S.M. Myopia. Lancet 2012, 379, 1739–1748. [Google Scholar] [CrossRef] [PubMed]
- Holden, B.A.; Fricke, T.R.; Wilson, D.A.; Jong, M.; Naidoo, K.S.; Sankaidurg, P.; Wong, T.Y.; Naduvilath, T.J.; Resnikoff, S. Global Prevalence of Myopia and High Myopia and Temporal Trends from 2000 through 2050. Ophthalmology 2016, 123, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Ha, A.; Kim, C.Y.; Shim, S.R.; Chang, I.B.; Kim, Y.K. Degree of Myopia and Glaucoma Risk: A Dose-Response Meta-analysis. Am. J. Ophthalmol. 2022, 236, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.; Hourihan, F.; Sandbach, J.; Wang, J.J. The relationship between glaucoma and myopia: The Blue Mountains Eye Study. Ophthalmology 1999, 106, 2010–2015. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, Y.; Wang, S.; Wang, Y.; Jonas, J.B. High myopia and glaucoma susceptibility the Beijing Eye Study. Ophthalmology 2007, 114, 216–220. [Google Scholar] [CrossRef]
- Wang, Y.X.; Yang, H.; Wei, C.C.; Xu, L.; Wei, W.B.; Jonas, J.B. High myopia as risk factor for the 10-year incidence of open-angle glaucoma in the Beijing Eye Study. Br. J. Ophthalmol. 2022, 107, 935–940. [Google Scholar] [CrossRef]
- Cahane, M.; Bartov, E. Axial length and scleral thickness effect on susceptibility to glaucomatous damage: A theoretical model implementing Laplace’s law. Ophthalmic Res. 1992, 24, 280–284. [Google Scholar] [CrossRef]
- Choquet, H.; Khawaja, A.P.; Jiang, C.; Yin, J.; Melles, R.B.; Glymour, M.M.; Hysi, P.G.; Jorgenson, E. Association Between Myopic Refractive Error and Primary Open-Angle Glaucoma: A 2-Sample Mendelian Randomization Study. JAMA Ophthalmol. 2022, 140, 864–871. [Google Scholar] [CrossRef]
- Chong, R.S.; Li, H.; Cheong, A.J.Y.; Fan, Q.; Koh, V.; Raghavan, L.; Nongpiur, M.E.; Cheng, C.Y. Mendelian Randomization Implicates Bidirectional Association between Myopia and Primary Open-Angle Glaucoma or Intraocular Pressure. Ophthalmology 2023, 130, 394–403. [Google Scholar] [CrossRef]
- Tham, Y.C.; Aung, T.; Fan, Q.; Saw, S.M.; Siantar, R.G.; Wong, T.Y.; Cheng, C.Y. Joint Effects of Intraocular Pressure and Myopia on Risk of Primary Open-Angle Glaucoma: The Singapore Epidemiology of Eye Diseases Study. Sci. Rep. 2016, 6, 19320. [Google Scholar] [CrossRef]
- Iglesias, A.I.; Ong, J.S.; Khawaja, A.P.; Gharahkhani, P.; Tedja, M.S.; Verhoeven, V.J.M.; Bonnemaijer, P.W.M.; Wolfs, R.C.W.; Young, T.L.; Jansonius, N.M.; et al. International Glaucoma Genetics Consortium (IGGC) and Consortium for Refractive Error and Myopia (CREAM). Determining Possible Shared Genetic Architecture between Myopia and Primary Open-Angle Glaucoma. Investig. Ophthalmol. Vis. Sci. 2019, 60, 3142–3149. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Lee, J.W.; Kim, E.A.; Caprioli, J. The effect of corneal biomechanical properties on rebound tonometer in patients with normal-tension glaucoma. Am. J. Ophthalmol. 2015, 159, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Dou, R.; Wang, Y. Comparison of Corneal Biomechanics between Low and High Myopic Eyes-A Meta-analysis. Am. J. Ophthalmol. 2019, 207, 419–425. [Google Scholar] [CrossRef]
- Leung, C.K.; Mohamed, S.; Leung, K.S.; Cheung, C.Y.L.; Chan, S.L.W.; Cheng, D.K.Y.; Lee, A.K.C.; Leung, G.Y.O.; Rao, S.K.; Lam, D.S.C. Retinal nerve fiber layer measurements in myopia: An optical coherence tomography study. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5171–5176. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Hong, S.W.; Im, S.K.; Lee, S.H.; Ahn, M.D. Effect of myopia on the thickness of the retinal nerve fiber layer measured by Cirrus HD optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4075–4083. [Google Scholar] [CrossRef]
- Leung, C.K.; Yu, M.; Weinreb, R.N.; Mak, H.K.; Lai, G.; Ye, C.; Lam, D.S.C. Retinal nerve fiber layer imaging with spectral-domain optical coherence tomography: Interpreting the RNFL maps in healthy myopic eyes. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7194–7200. [Google Scholar] [CrossRef]
- Chong, G.T.; Lee, R.K. Glaucoma versus red disease: Imaging and glaucoma diagnosis. Curr. Opin. Ophthalmol. 2012, 23, 79–88. [Google Scholar] [CrossRef]
- Zemborain, Z.Z.; Jarukasetphon, R.; Tsamis, E.; De Moraes, C.G.; Ritch, R.; Hood, D.C. Optical Coherence Tomography Can Be Used to Assess Glaucomatous Optic Nerve Damage in Most Eyes With High Myopia. J. Glaucoma 2020, 29, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wen, W.; Sun, X. Comparison of Several Parameters in Two Optical Coherence Tomography Systems for Detecting Glaucomatous Defects in High Myopia. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4910–4915. [Google Scholar] [CrossRef]
- Seol, B.R.; Jeoung, J.W.; Park, K.H. Glaucoma Detection Ability of Macular Ganglion Cell-Inner Plexiform Layer Thickness in Myopic Preperimetric Glaucoma. Investig. Ophthalmol. Vis. Sci. 2015, 56, 8306–8313. [Google Scholar] [CrossRef]
- Kim, N.R.; Lee, E.S.; Seong, G.J.; Kang, S.Y.; Kim, J.H.; Hong, S.; Kim, C.Y. Comparing the ganglion cell complex and retinal nerve fibre layer measurements by Fourier domain OCT to detect glaucoma in high myopia. Br. J. Ophthalmol. 2011, 95, 1115–1121. [Google Scholar] [CrossRef]
- Akashi, A.; Kanamori, A.; Nakamura, M.; Fujihara, M.; Yamada, Y.; Negi, A. The ability of macular parameters and circumpapillary retinal nerve fiber layer by three SD-OCT instruments to diagnose highly myopic glaucoma. Investig. Ophthalmol. Vis. Sci. 2013, 54, 6025–6032. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.H.; Kim, M.K.; Kim, D.W. Segmentation Errors in Macular Ganglion Cell Analysis as Determined by Optical Coherence Tomography. Ophthalmology 2016, 123, 950–958. [Google Scholar] [CrossRef]
- Biswas, S.; Lin, C.; Leung, C.K. Evaluation of a Myopic Normative Database for Analysis of Retinal Nerve Fiber Layer Thickness. JAMA Ophthalmol. 2016, 134, 1032–1039, Erratum in JAMA Ophthalmol. 2016, 134, 1336. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.U.; Kim, K.E.; Kim, Y.K.; Park, K.H.; Jeoung, J.W. Development of Topographic Scoring System for Identifying Glaucoma in Myopic Eyes: A Spectral-Domain OCT Study. Ophthalmology 2018, 125, 1710–1719. [Google Scholar] [CrossRef]
- Kim, Y.W.; Lee, J.; Kim, J.S.; Park, K.H. Diagnostic Accuracy of Wide-Field Map from Swept-Source Optical Coherence Tomography for Primary Open-Angle Glaucoma in Myopic Eyes. Am. J. Ophthalmol. 2020, 218, 182–191. [Google Scholar] [CrossRef]
- Vuori, M.L.; Mäntyjärvi, M. Tilted disc syndrome may mimic false visual field deterioration. Acta Ophthalmol. 2008, 86, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.S.; Baskaran, M.; Singh, K.; Aung, T. Clinical characterization of young chinese myopes with optic nerve and visual field changes resembling glaucoma. J. Glaucoma 2012, 21, 281–286. [Google Scholar] [CrossRef]
- Doshi, A.; Kreidl, K.O.; Lombardi, L.; Sakamoto, D.K.; Singh, K. Nonprogressive glaucomatous cupping and visual field abnormalities in young Chinese males. Ophthalmology 2007, 114, 472–479. [Google Scholar] [CrossRef]
- Han, J.C.; Lee, E.J.; Kim, S.H.; Kee, C. Visual Field Progression Pattern Associated with Optic Disc Tilt Morphology in Myopic Open-Angle Glaucoma. Am. J. Ophthalmol. 2016, 169, 33–45. [Google Scholar] [CrossRef]
- Lin, F.; Chen, S.; Song, Y.; Li, F.; Wang, W.; Zhao, Z.; Gao, X.; Wang, P.; Jin, L.; Liu, Y.; et al. Glaucoma Suspects with High Myopia Study Group. Classification of Visual Field Abnormalities in Highly Myopic Eyes without Pathologic Change. Ophthalmology 2022, 129, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Fannin, L.A.; Schiffman, J.C.; Budenz, D.L. Risk factors for hypotony maculopathy. Ophthalmology 2003, 110, 1185–1191. [Google Scholar] [CrossRef] [PubMed]
- Stamper, R.L.; McMenemy, M.G.; Lieberman, M.F. Hypotonous maculopathy after trabeculectomy with subconjunctival 5-fluorouracil. Am. J. Ophthalmol. 1992, 114, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Costa, V.P.; Arcieri, E.S. Hypotony maculopathy. Acta Ophthalmol. Scand. 2007, 85, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Kao, S.T.; Lee, S.H.; Chen, Y.C. Late-onset Hypotony Maculopathy after Trabeculectomy in a Highly Myopic Patient with Juvenile Open-angle Glaucoma. J. Glaucoma 2017, 26, e137–e141. [Google Scholar] [CrossRef]
- Saheb, H.; Ahmed, I.I. Micro-invasive glaucoma surgery: Current perspectives and future directions. Curr. Opin. Ophthalmol. 2012, 23, 96–104. [Google Scholar] [CrossRef]
- Huth, A.; Viestenz, A. High myopia in vitrectomized eyes: Contraindication for minimally invasive glaucoma surgery implant? Ophthalmologe 2020, 117, 461–466. [Google Scholar] [CrossRef]
- Fea, A.; Sacchi, M.; Franco, F.; Laffi, G.L.; Oddone, F.; Costa, G.; Serino, F.; Giansanti, F. Effectiveness and Safety of XEN45 in Eyes with High Myopia and Open Angle Glaucoma. J. Glaucoma 2023, 32, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.; Martin, K.R. Ciliary body position variability in glaucoma patients assessed by scleral transillumination. Eye 2008, 22, 1499–1503. [Google Scholar] [CrossRef]
- Gavrilova, B.; Roters, S.; Engels, B.F.; Konen, W.; Krieglstein, G.K. Late hypotony as a complication of viscocanalostomy: A case report. J. Glaucoma 2004, 13, 263–267. [Google Scholar] [CrossRef]
- Kora, Y.; Koike, M.; Suzuki, Y.; Inatomi, M.; Fukado, Y.; Ozawa, T. Errors in IOL power calculations for axial high myopia. Ophthalmic Surg. 1991, 22, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Tehrani, M.; Krummenauer, F.; Blom, E.; Dick, H.B. Evaluation of the practicality of optical biometry and applanation ultrasound in 253 eyes. J. Cataract. Refract. Surg. 2003, 29, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Elhusseiny, A.M.; Salim, S. Cataract surgery in myopic eyes. Curr. Opin. Ophthalmol. 2023, 34, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Omoto, M.; Sugawara, K.; Torii, H.; Yotsukura, E.; Masui, S.; Shigeno, Y.; Nishi, Y.; Negishi, K. Investigating the Prediction Accuracy of Recently Updated Intraocular Lens Power Formulas with Artificial Intelligence for High Myopia. J. Clin. Med. 2022, 11, 4848. [Google Scholar] [CrossRef] [PubMed]
- Ripandelli, G.; Scassa, C.; Parisi, V.; Gazzaniga, D.; D’Amico, D.J.; Stirpe, M. Cataract surgery as a risk factor for retinal detachment in very highly myopic eyes. Ophthalmology 2003, 110, 2355–2361. [Google Scholar] [CrossRef]
- Cionni, R.J.; Barros, M.G.; Osher, R.H. Management of lens-iris diaphragm retropulsion syndrome during phacoemulsification. J. Cataract. Refract. Surg. 2004, 30, 953–956. [Google Scholar] [CrossRef]
- Wang, D.; Yu, X.; Li, Z.; Ding, X.; Lian, H.; Mao, J.; Zhao, Y.; Zhao, Y.E. The Effect of Anterior Capsule Polishing on Capsular Contraction and Lens Stability in Cataract Patients with High Myopia. J. Ophthalmol. 2018, 2018, 8676451. [Google Scholar] [CrossRef]
- Wang, W.; Xu, D.; Liu, X.; Xu, W. Case series: “Double arch” changes caused by capsule contraction syndrome after cataract surgery in highly myopic eyes. BMC Ophthalmol. 2021, 21, 367. [Google Scholar] [CrossRef]
- Vetter, J.M.; Holzer, M.P.; Teping, C.; Weingartner, W.E.; Gericke, A.; Stoffelns, B.; Pfeiffer, N.; Sekundo, W. Intraocular pressure during corneal flap preparation: Comparison among four femtosecond lasers in porcine eyes. J. Refract. Surg. 2011, 27, 427–433. [Google Scholar] [CrossRef]
- Bamashmus, M.A.; Saleh, M.F. Post-LASIK interface fluid syndrome caused by steroid drops. Saudi J. Ophthalmol. 2013, 27, 125–128. [Google Scholar] [CrossRef]
- Shoji, N.; Ishida, A.; Haruki, T.; Matsumura, K.; Kasahara, M.; Shimizu, K. Interface Fluid Syndrome Induced by Uncontrolled Intraocular Pressure without Triggering Factors after LASIK in a Glaucoma Patient: A Case Report. Medicine 2015, 94, e1609. [Google Scholar] [CrossRef]
- Kim, C.Y.; Jung, Y.H.; Lee, E.J.; Hyon, J.Y.; Park, K.H.; Kim, T.W. Delayed-onset interface fluid syndrome after LASIK following phacotrabeculectomy. BMC Ophthalmol. 2019, 19, 74. [Google Scholar] [CrossRef]
- Grzybowski, A.; Kanclerz, P.; Tsubota, K.; Lanca, C.; Saw, S.M. A review on the epidemiology of myopia in school children worldwide. BMC Ophthalmol. 2020, 20, 27. [Google Scholar] [CrossRef]
- Rudnicka, A.R.; Kapetanakis, V.V.; Wathern, A.K.; Logan, N.S.; Gilmartin, B.; Whincup, P.H.; Cook, D.G.; Owen, C.G. Global variations and time trends in the prevalence of childhood myopia, a systematic review and quantitative meta-analysis: Implications for aetiology and early prevention. Br. J. Ophthalmol. 2016, 100, 882–890. [Google Scholar] [CrossRef]
- Modjtahedi, B.S.; Abbott, R.L.; Fong, D.S.; Lum, F.; Tan, D. Task Force on Myopia. Reducing the Global Burden of Myopia by Delaying the Onset of Myopia and Reducing Myopic Progression in Children: The Academy’s Task Force on Myopia. Ophthalmology 2021, 128, 816–826. [Google Scholar] [CrossRef] [PubMed]
- VanderVeen, D.K.; Kraker, R.T.; Pineles, S.L.; Hutchinson, A.K.; Wilson, L.B.; Galvin, J.A.; Lambert, S.R. Use of Orthokeratology for the Prevention of Myopic Progression in Children: A Report by the American Academy of Ophthalmology. Ophthalmology 2019, 126, 623–636. [Google Scholar] [CrossRef]
- Pineles, S.L.; Kraker, R.T.; VanderVeen, D.K.; Hutchinson, A.K.; Galvin, J.A.; Wilson, L.B.; Lambert, S.R. Atropine for the Prevention of Myopia Progression in Children: A Report by the American Academy of Ophthalmology. Ophthalmology 2017, 124, 1857–1866. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.S.; Ho, H.Y.; Ho, H.C.; Chen, Y.W.; Lee, C.K.; Chen, P.J.; Lai, F.; Roger Jang, J.S.; Ko, M.L. Use of multimodal dataset in AI for detecting glaucoma based on fundus photographs assessed with OCT: Focus group study on high prevalence of myopia. BMC Med. Imaging 2022, 22, 206. [Google Scholar] [CrossRef] [PubMed]
| Glaucoma Surgical Procedure | Advantages | Disadvantages | Special Considerations in Myopic Eyes |
|---|---|---|---|
| Microinvasive glaucoma surgery (MIGS) | Improved safety profile | Angle-based MIGS offer lower efficacy and may not be appropriate for patients with advanced glaucoma Hypotony may occur with bleb-based MIGS | Perform preoperative gonioscopy to assess angle anatomy in candidates for angle-based MIGS Use antimetabolites judiciously if performing bleb-based MIGS to avoid hypotony |
| Tube shunt surgery | Improved efficacy | Risk of hypotony and hypotony-related complications | Exercise caution while securing endplate to thin sclera Consider using valved or smaller surface area nonvalved implant Consider performing planned laser tube ligature release if using nonvalved implant |
| Trabeculectomy | Improved efficacy | Risk of hypotony and hypotony-related complications | Exercise caution during scleral flap dissection as sclera may be thin Use an increased number of, and tighter, scleral flap sutures to avoid hypotony Use antimetabolites judiciously to avoid hypotony |
| Trans-scleral cyclophotocoagulation | Nonincisional | Risk of hypotony and hypotony-related complications | Consider alternative to retrobulbar or peribulbar block to avoid risk of scleral perforation Consider scleral transillumination to confirm ciliary body location |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinod, K.; Salim, S. Addressing Glaucoma in Myopic Eyes: Diagnostic and Surgical Challenges. Bioengineering 2023, 10, 1260. https://doi.org/10.3390/bioengineering10111260
Vinod K, Salim S. Addressing Glaucoma in Myopic Eyes: Diagnostic and Surgical Challenges. Bioengineering. 2023; 10(11):1260. https://doi.org/10.3390/bioengineering10111260
Chicago/Turabian StyleVinod, Kateki, and Sarwat Salim. 2023. "Addressing Glaucoma in Myopic Eyes: Diagnostic and Surgical Challenges" Bioengineering 10, no. 11: 1260. https://doi.org/10.3390/bioengineering10111260
APA StyleVinod, K., & Salim, S. (2023). Addressing Glaucoma in Myopic Eyes: Diagnostic and Surgical Challenges. Bioengineering, 10(11), 1260. https://doi.org/10.3390/bioengineering10111260






