Seasonal and Basinal Influences on the Formation and Transport of Dissolved Trace Metal Forms in a Mining-Impacted Riverine Environment
Abstract
1. Introduction
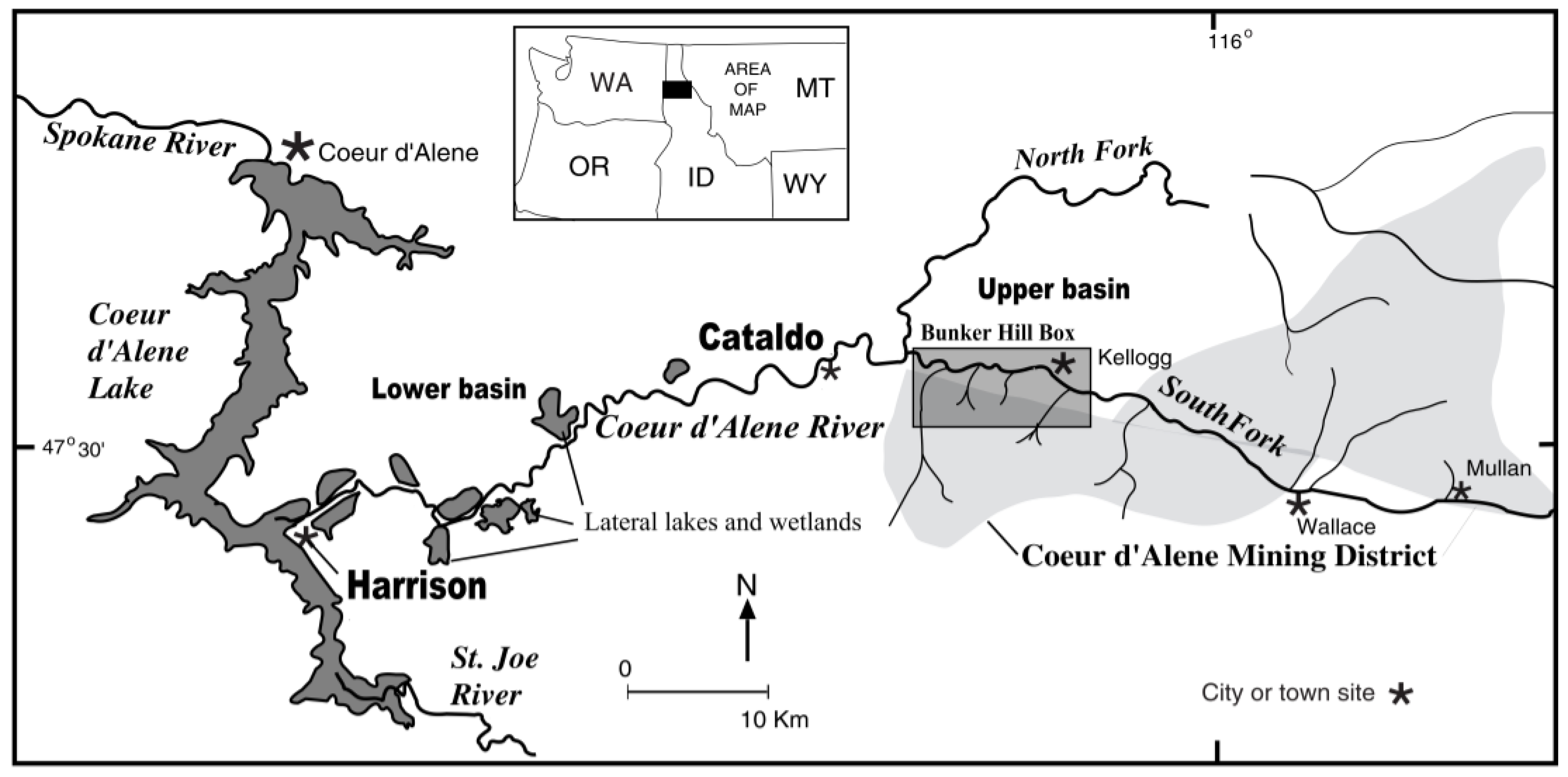
Study Area
2. Materials and Methods
3. Results
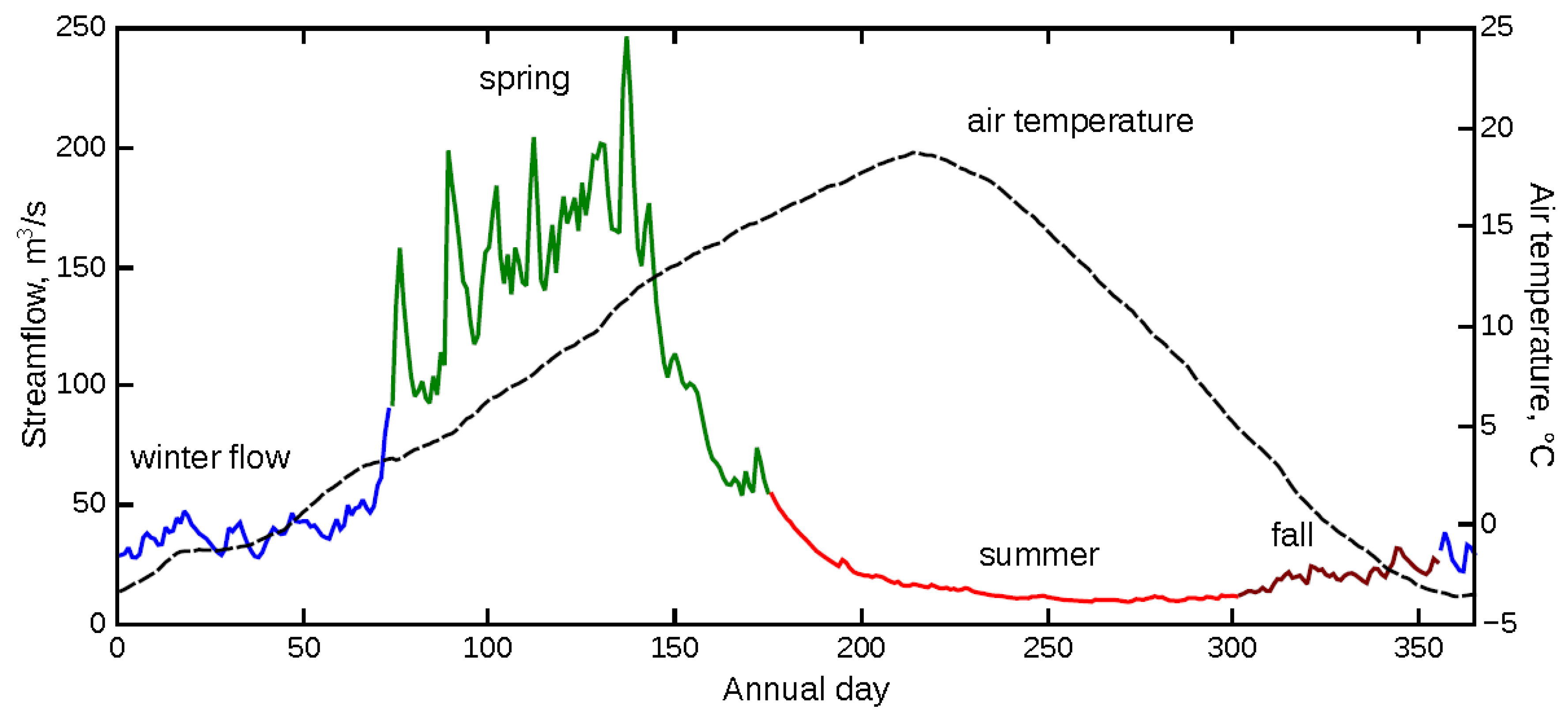
3.1. Physicochemical Parameters and Metal Concentrations
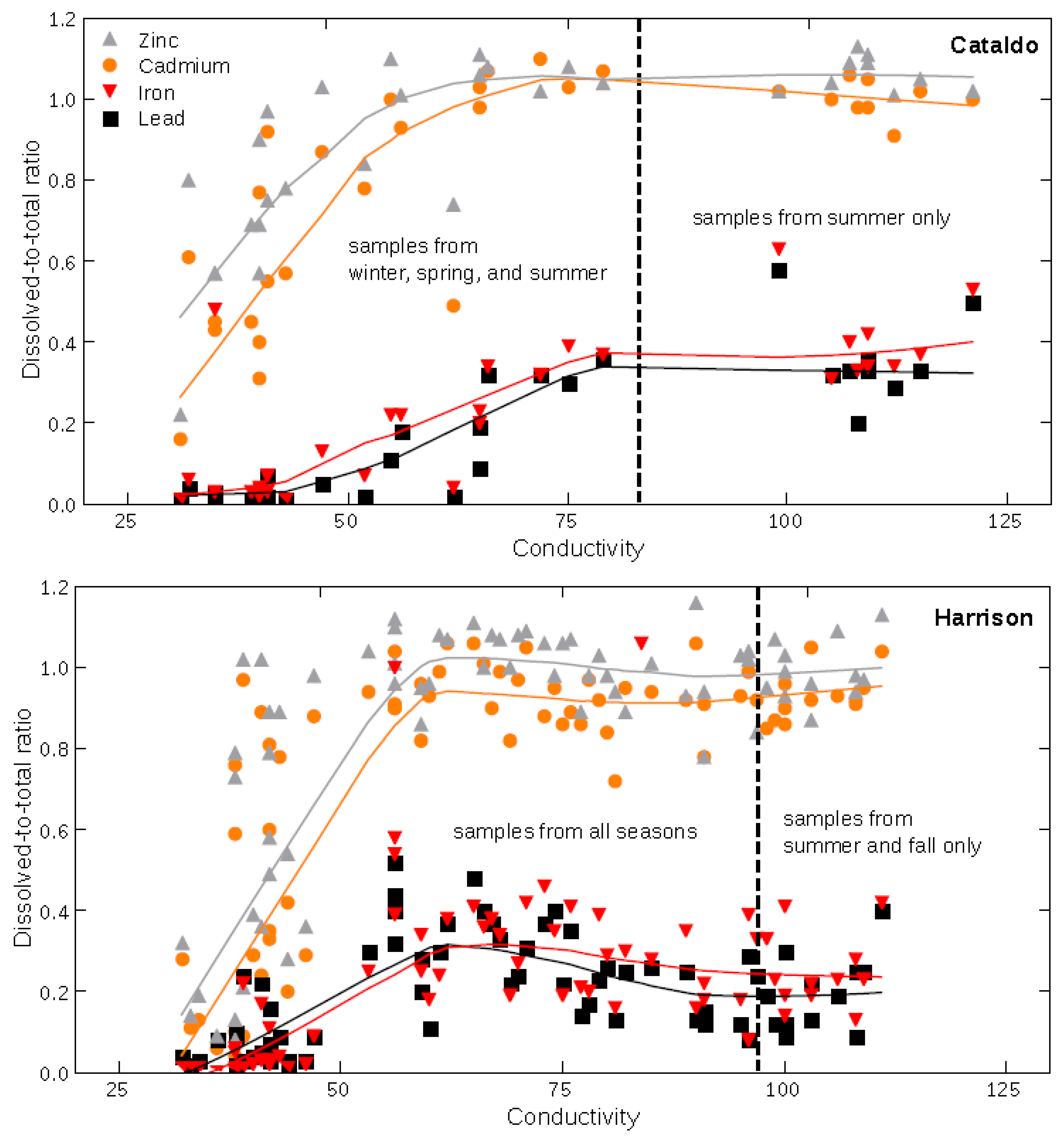
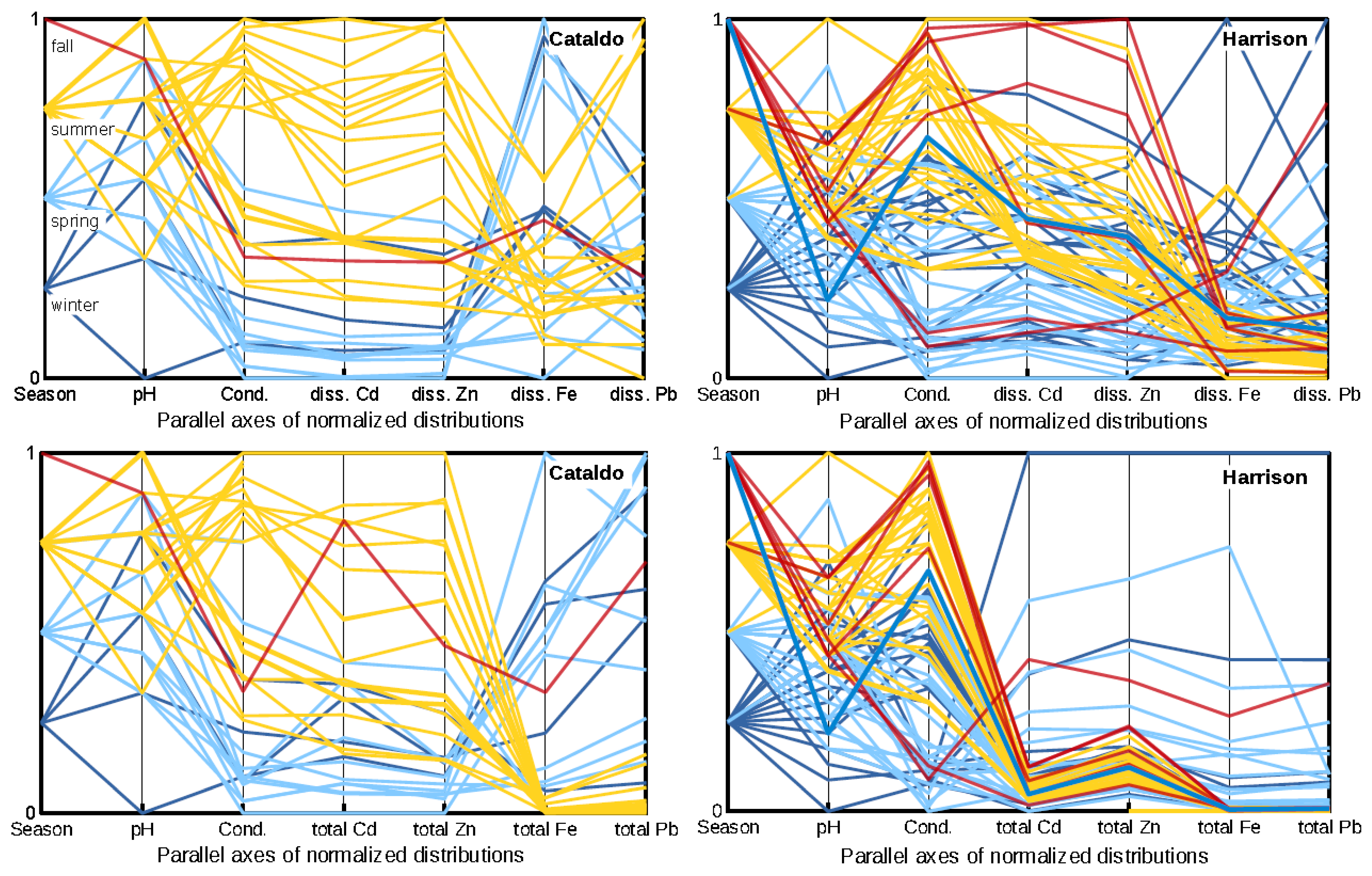
3.2. Available Ligands and Metal Transport Relations between Basins
3.3. Element Transport Differences in the Coeur d’Alene River
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moore, J.N.; Luoma, S.N. Hazardous wastes from large-scale metal extraction. A case study. Environ. Sci. Technol. 1990, 24, 1278–1285. [Google Scholar] [CrossRef]
- Bigham, J.M. Mineralogy of ochre deposits formed by sulfide oxidation. In Environmental Geochemistry of Sulfide Mine-Wastes; Jambor, J.L., Blowes, D.W., Eds.; Mineralogical Association of Canada: Québec, QC, Canada, 1994; Volume 22, pp. 103–132. ISBN 978-0-8412-2772-9. [Google Scholar]
- Dudka, S.; Adriano, D.C. Environmental impacts of metal ore mining and processing: A review. J. Environ. Qual. 1997, 26, 590–602. [Google Scholar] [CrossRef]
- Bigham, J.M.; Nordstrom, D.K. Iron and aluminum hydroxysulfates from acid sulfate water. In Sulfate Minerals: Crystallography, Geochemistry, and Environmental Significance; Alpers, C.N., Ed.; Reviews in Mineralogy; Mineralogical Society of America: Chantilly, VA, USA, 2000; Volume 40, pp. 351–403. [Google Scholar]
- Pyatt, F.B.; Grattan, J.P. Some consequences of ancient mining activities on the health of ancient and modern human populations. J. Public Health 2001, 23, 235–236. [Google Scholar] [CrossRef]
- Wigginton, N.S.; Haus, K.L.; Hochella, M.F., Jr. Aquatic environmental nanoparticles. J. Environ. Monit. 2007, 9, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Hochella, M.F., Jr.; Lower, S.K.; Maurice, P.A.; Penn, R.L.; Sahai, N.; Sparks, D.L.; Twining, B.S. Nanominerals, mineral nanoparticles, and Earth systems. Science 2008, 319, 1631–1635. [Google Scholar] [CrossRef] [PubMed]
- Ju-Nam, Y.; Lead, J.R. Manufactured nanoparticles: An overview of their chemistry, interactions and potential environmental implications. Sci. Total Environ. 2008, 400, 396–414. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; Wang, H.; Zhou, D.; Lenihan, H.S.; Cherr, G.; Cardinale, B.J.; Miller, R.; Ji, Z. Stability and aggregation of metal oxide nanoparticles in natural aqueous matrices. Environ. Sci. Technol. 2010, 44, 1962–1967. [Google Scholar] [CrossRef] [PubMed]
- Delay, M.; Frimmel, F.H. Nanoparticles in aquatic systems. Anal. Bioanal. Chem. 2011, 402, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Kretzschmar, R.; Schäfer, T. Metal retention and transport on colloidal particles in the environment. Elements 2005, 1, 205–210. [Google Scholar] [CrossRef]
- Hoffmann, S.R.; Shafer, M.M.; Armstrong, D.E. Strong colloidal and dissolved organic ligands binding copper and zinc in rivers. Environ. Sci. Technol. 2007, 41, 6996–7002. [Google Scholar] [CrossRef] [PubMed]
- Haus, K.L.; Hooper, R.L.; Strumness, L.A.; Mahoney, J.B. Analysis of arsenic speciation in mine contaminated lacustrine sediment using selective sequential extraction, HR-ICPMS and TEM. Appl. Geochem. 2008, 23, 692–704. [Google Scholar] [CrossRef]
- Aiken, G.R.; Hsu-Kim, H.; Ryan, J.N. Influence of dissolved organic matter on the environmental fate of metals, nanoparticles, and colloids. Environ. Sci. Technol. 2011, 45, 3196–3201. [Google Scholar] [CrossRef] [PubMed]
- Plathe, K.L.; von der Kammer, F.; Hassellöv, M.; Moore, J.N.; Murayama, M.; Hofmann, T.; Hochella, M.F., Jr. The role of nanominerals and mineral nanoparticles in the transport of toxic trace metals: Field-flow fractionation and analytical TEM analyses after nanoparticle isolation and density separation. Geochim. Cosmochim. Acta 2013, 102, 213–225. [Google Scholar] [CrossRef]
- Hochella, M.F., Jr.; Moore, J.N.; Putnis, C.V.; Putnis, A.; Kasama, T.; Eberl, D.D. Direct observation of heavy metal-mineral association from the Clark Fork River Superfund Complex: Implications for metal transport and bioavailability. Geochim. Cosmochim. Acta 2005, 69, 1651–1663. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Blank, R.G. Dissolved and labile concentrations of Cd, Cu, Pb, and Zn in the South Fork Coeur d’Alene River, Idaho: Comparisons among chemical equilibrium models and implications for biotic ligand models. Appl. Geochem. 2008, 23, 3355–3371. [Google Scholar] [CrossRef]
- Cornell, R.M. Simultaneous incorporation of Mn, Ni and Co in the goethite (alpha-FeOOH) structure. Clay Miner. 1991, 26, 427–430. [Google Scholar] [CrossRef]
- Galán, E.; Gómez-Ariza, J.L.; González, I.; Fernández-Caliani, J.C.; Morales, E.; Giráldez, I. Heavy metal partitioning in river sediments severely polluted by acid mine drainage in the Iberian Pyrite Belt. Appl. Geochem. 2003, 18, 409–421. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Box, S.E.; Tonkin, J.W. Modeling precipitation and sorption of elements during mixing of river water and porewater in the Coeur d’Alene River Basin. Environ. Sci. Technol. 2003, 37, 4694–4701. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Kan, A.T.; Tomson, M.B. Critical evaluation of desorption phenomena of heavy metals from natural sediments. Environ. Sci. Technol. 2003, 37, 5566–5573. [Google Scholar] [CrossRef] [PubMed]
- Boujelben, N.; Bouzid, J.; Elouear, Z. Adsorption of nickel and copper onto natural iron oxide-coated sand from aqueous solutions: Study in single and binary systems. J. Hazard. Mater. 2009, 163, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Stolpe, B.; Guo, L.; Shiller, A.M.; Aiken, G.R. Abundance, size distributions and trace-element binding of organic and iron-rich nanocolloids in Alaskan rivers, as revealed by field-flow fractionation and ICP-MS. Geochim. Cosmochim. Acta 2013, 105, 221–239. [Google Scholar] [CrossRef]
- Paulson, A.J. Biogeochemical removal of Zn and Cd in the Coeur d’Alene River (Idaho, USA), downstream of a mining district. Sci. Total Environ. 2001, 278, 31–44. [Google Scholar] [CrossRef]
- National Research Council. Superfund and Mining Megasites: Lessons from the Coeur d’Alene River Basin; U.S. Environmental Protection Agency: Washington, DC, USA, 2005; p. 484.
- Balistrieri, L.S.; Box, S.E.; Bookstrom, A.A.; Hooper, R.L.; Mahoney, J.B. Impacts of Historical Mining in the Coeur d‘Alene River Basin; Bulletin 2191-6; U.S. Geological Survey: Reston, VA, USA, 2010.
- Reece, D.E.; Felkey, J.R.; Wai, C.M. Heavy metal pollution in the sediments of the Coeur d’Alene River, Idaho. Environ. Geol. 1978, 2, 289–293. [Google Scholar] [CrossRef]
- Sprenke, K.F.; Rember, W.C.; Bender, S.F.; Hoffmann, M.L.; Rabbi, F.; Chamberlain, V.E. Toxic metal contamination in the lateral lakes of the Coeur d’Alene River valley, Idaho. Environ. Geol. 2000, 39, 575–586. [Google Scholar] [CrossRef]
- Barton, G.J. Dissolved Cadmium, Zinc, and Lead Loads from Ground-Water Seepage into the South Fork Coeur d’Alene River System, Northern Idaho, 1999; Water-Resources Investigations Report 2001-4274; U.S. Geological Survey: Reston, VA, USA, 2002; p. 134.
- Hickey, P.J.; McDaniel, P.A.; Strawn, D.G. Characterization of iron- and manganese-cemented redoximorphic aggregates in wetland soils contaminated with mine wastes. J. Environ. Qual. 2008, 37, 2375–2385. [Google Scholar] [CrossRef] [PubMed]
- Clark, G.M.; Mebane, C.A. Sources, Transport, and Trends for Selected Trace Metals and Nutrients in the Coeur d’Alene and Spokane River Basins, Northern Idaho, 1990–2013; Scientific Investigations Report 2014-5204; U.S. Geological Survey: Reston, VA, USA, 2014; p. 62.
- Egiebor, N.O.; Oni, B. Acid rock drainage formation and treatment: A review. Asia-Pac. J. Chem. Eng. 2007, 2, 47–62. [Google Scholar] [CrossRef]
- Nordstrom, D.K. Sulfide mineral oxidation. In Encyclopedia of Geobiology; Reitner, J., Thiel, V., Eds.; Encyclopedia of Earth Sciences Series; Springer: Dordrecht, The Netherlands, 2011; pp. 856–858. ISBN 978-1-4020-9211-4. [Google Scholar]
- Bookstrom, A.A.; Box, S.E.; Campbell, J.K.; Foster, K.I.; Jackson, B.L. Lead-Rich Sediments, Coeur d‘Alene River Valley, Idaho: Area, Volume, Tonnage, and Lead Content; Open File Report 2001-140; U.S. Geological Survey: Reston, VA, USA, 2001.
- Horowitz, A.J.; Elrick, K.A.; Cook, R.B. Effect of mining and related activities on the sediment trace element geochemistry of Lake Coeur d’Alene, Idaho, USA. Part I: Surface sediments. Hydrol. Process. 1993, 7, 403–423. [Google Scholar] [CrossRef]
- Harrington, J.M.; LaForce, M.J.; Rember, W.C.; Fendorf, S.E.; Rosenzweig, R.F. Phase associations and mobilization of iron and trace elements in Coeur d’Alene Lake, Idaho. Environ. Sci. Technol. 1998, 32, 650–656. [Google Scholar] [CrossRef]
- Bostick, B.C.; Hansel, C.M.; Fendorf, S. Seasonal Fluctuations in Zinc Speciation within a Contaminated Wetland. Environ. Sci. Technol. 2001, 35, 3823–3829. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, J.S.; Woods, P.F.; Berelson, W.M.; Balistrieri, L.S.; Carter, J.L.; Topping, B.R.; Fend, S.V. Importance of Sediment–Water Interactions in Coeur d’Alene Lake, Idaho, USA: Management Implications. Environ. Manag. 2003, 32, 348–359. [Google Scholar] [CrossRef]
- Moberly, J.G.; Borch, T.; Sani, R.K.; Spycher, N.F.; Şengör, S.S.; Ginn, T.R.; Peyton, B.M. Heavy metal–mineral associations in Coeur d’Alene River sediments: A synchrotron-based analysis. Water Air Soil Pollut. 2008, 201, 195–208. [Google Scholar] [CrossRef]
- Moberly, J.; D’Imperio, S.; Parker, A.; Peyton, B. Microbial community signature in Lake Coeur d’Alene: Association of environmental variables and toxic heavy metal phases. Appl. Geochem. 2016, 66, 174–183. [Google Scholar] [CrossRef]
- Garbarino, J.R.; Struzeski, T.M. Methods of analysis by the U.S. Geological Survey National Water Quality Laboratory—Determination of Elements in Whole-Water Digests Using Inductively Coupled Plasma-Optical Emission Spectrometry and Inductively Coupled Plasma-Mass Spectrometry; Open-File Report 98-165; U.S. Geological Survey: Reston, VA, USA, 1998.
- Garbarino, J.R. Methods of Analysis by the U.S. Geological Survey National Water Quality Laboratory—Determination of Dissolved Arsenic, Boron, Lithium, Selenium, Strontium, Thallium, and Vanadium Using Inductively Coupled Plasma-Mass Spectrometry; Open File Report 99-094; U.S. Geological Survey: Reston, VA, USA, 1999.
- Tuschall, J.R.; Brezonik, P.L. Characterization of organic nitrogen in natural waters: Its molecular size, protein content, and interactions with heavy metals. Limnol. Oceanogr. 1980, 25, 495–504. [Google Scholar] [CrossRef]
- Manceau, A.; Matynia, A. The nature of Cu bonding to natural organic matter. Geochim. Cosmochim. Acta 2010, 74, 2556–2580. [Google Scholar] [CrossRef]
- Hem, J.D. Chemistry and occurrence of cadmium and zinc in surface water and groundwater. Water Resour. Res. 1972, 8, 661–679. [Google Scholar] [CrossRef]
- Bruemmer, G.W.; Gerth, J.; Tiller, K.G. Reaction kinetics of the adsorption and desorption of nickel, zinc and cadmium by goethite. I. Adsorption and diffusion of metals. J. Soil Sci. 1988, 39, 37–52. [Google Scholar] [CrossRef]
- Krishnamurthy, S. Biomethylation and environmental transport of metals. J. Chem. Educ. 1992, 69, 347. [Google Scholar] [CrossRef]
- Benoit, G.; Rozan, T.F. The influence of size distribution on the particle concentration effect and trace metal partitioning in rivers. Geochim. Cosmochim. Acta 1999, 63, 113–127. [Google Scholar] [CrossRef]
- Adriano, D.C. Trace Elements in Terrestrial Environments; Springer: New York, NY, USA, 2001; ISBN 978-1-4684-9505-8. [Google Scholar]
- Zhang, X.; Sun, H.; Zhang, Z.; Niu, Q.; Chen, Y.; Crittenden, J.C. Enhanced bioaccumulation of cadmium in carp in the presence of titanium dioxide nanoparticles. Chemosphere 2007, 67, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Traina, S.J.; Laperche, V. Contaminant bioavailability in soils, sediments, and aquatic environments. Proc. Natl. Acad. Sci. USA 1999, 96, 3365–3371. [Google Scholar] [CrossRef] [PubMed]
- Kozyatnyk, I.; Bouchet, S.; Björn, E.; Haglund, P. Fractionation and size-distribution of metal and metalloid contaminants in a polluted groundwater rich in dissolved organic matter. J. Hazard. Mater. 2016, 318, 194–202. [Google Scholar] [CrossRef] [PubMed]




| Temp. (°C) | pH | Conductivity (µS/cm) | Sus. Sediment (mg/L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Season | Min | Med | Max | Min | Med | Max | Min | Med | Max | Min | Med | Max |
| Cataldo site (discharge from upper basin) | ||||||||||||
| Winter | 2.1 | 3.0 | 3.9 | 6.5 | 6.9 | 7.2 | 40 | 47 | 65 | 0.3 | 0.6 | 1 |
| Spring | 3.3 | 5.8 | 13.9 | 6.8 | 6.9 | 7.3 | 31 | 40 | 79 | 3 | 29 | 55 |
| Summer | 8.9 | 12.2 | 15.8 | 6.8 | 7.2 | 7.4 | 55 | 107 | 121 | 1 | 4 | 191 |
| Fall | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- |
| Harrison site (discharge from lower basin) | ||||||||||||
| Winter | 0.6 | 3.1 | 4.4 | 5.9 | 6.8 | 7.5 | 38 | 60 | 96 | 1 | 6 | 611 |
| Spring | 3.1 | 7.5 | 20.1 | 6.2 | 6.8 | 7.9 | 32 | 44 | 78 | 1 | 19 | 404 |
| Summer | 8.1 | 16.1 | 23.5 | 6.8 | 7.3 | 8.2 | 56 | 93 | 111 | 1 | 3 | 12 |
| Fall | 1.9 | 4.9 | 7.0 | 6.4 | 7.0 | 7.4 | 39 | 88 | 109 | 3 | 6 | 236 |
| Season | Diss. Cd | Total Cd | Diss. Fe | Total Fe | Diss. Pb | Total Pb | Diss. Zn | Total Zn |
|---|---|---|---|---|---|---|---|---|
| Cataldo site (discharge from the upper basin) | ||||||||
| Overall | 1.17 | 1.17 | 21.6 | 114 | 0.95 | 10.7 | 171* | 161* |
| Winter | 0.54 | 0.96 | 29.2 | 739 | 0.79 | 40.5 | 86.8 | 113 |
| Spring | 0.39 | 0.61 | 21.6 | 837 | 0.95 | 37.1 | 69.8 | 88.3 |
| Summer | 1.56 | 1.55 | 19.4 | 52.6 | 0.96 | 2.92 | 291* | 279* |
| Fall | --- | --- | --- | --- | --- | --- | --- | --- |
| Harrison site (discharge from the lower basin) | ||||||||
| Overall | 0.93 | 1.21 | 74.0 | 305 | 6.42 | 25.9 | 179 | 213 |
| Winter | 0.93 | 1.28 | 139 | 527 | 8.91 | 32.9 | 181 | 228 |
| Spring | 0.64 | 1.20 | 61.9 | 890 | 7.96 | 84.8 | 109 | 188 |
| Summer | 1.05 | 1.15 | 47.5 | 196 | 3.78 | 19.4 | 194 | 195 |
| Fall | 1.36 | 1.81 | 77.7 | 396 | 6.82 | 35.7 | 275 | 342 |
| Wilcoxon rank sum test: Ho: median difference = 0 (Ha ≠ 0), α= 0.05 | ||||||||
| Overall | 0.57 | 0.16 | 0.00 | 0.00 | 0.00 | 0.00 | 0.70 | 0.01 |
| Winter | ND | ND | D | D | D | D | D | D |
| Spring | 0.00 | 0.01 | 0.01 | 0.34 | 0.00 | 0.10 | 0.00 | 0.00 |
| Summer | 0.00 | 0.06 | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.22 |
| Fall | --- | --- | --- | --- | --- | --- | --- | --- |
| Cataldo | Harrison | |||||||
|---|---|---|---|---|---|---|---|---|
| Dissolved | Total | Dissolved | Total | |||||
| Constituent | Comp 1 | Comp 2 | Comp 1 | Comp 2 | Comp 1 | Comp 2 | Comp 1 | Comp 2 |
| % of variance | 51 | 18 | 53 | 20 | 46 | 22 | 59 | 19 |
| Streamflow | −0.88 | 0.09 | −0.91 | 0.26 | 0.48 | −0.32 | 0.37 | 0.58 |
| SS | −0.08 | 0.82 | −0.07 | −0.40 | 0.75 | −0.20 | 0.98 | −0.17 |
| Temperature | 0.80 | 0.33 | 0.78 | −0.02 | −0.57 | 0.46 | −0.43 | −0.71 |
| Conductivity | 0.95 | −0.17 | 0.93 | 0.26 | −0.85 | −0.35 | −0.53 | −0.49 |
| pH | 0.57 | 0.37 | 0.60 | −0.15 | −0.66 | 0.13 | −0.50 | −0.57 |
| Cd | 0.95 | −0.06 | 0.69 | 0.67 | −0.77 | −0.59 | 0.95 | −0.27 |
| Fe | −0.34 | −0.18 | −0.75 | 0.59 | 0.41 | −0.68 | 0.96 | −0.21 |
| Pb | −0.08 | 0.76 | −0.70 | 0.61 | 0.79 | −0.37 | 0.93 | −0.15 |
| Zn | 0.94 | −0.12 | 0.78 | 0.60 | −0.67 | −0.71 | 0.92 | −0.31 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langman, J.B.; Torso, K.; Moberly, J.G. Seasonal and Basinal Influences on the Formation and Transport of Dissolved Trace Metal Forms in a Mining-Impacted Riverine Environment. Hydrology 2018, 5, 35. https://doi.org/10.3390/hydrology5030035
Langman JB, Torso K, Moberly JG. Seasonal and Basinal Influences on the Formation and Transport of Dissolved Trace Metal Forms in a Mining-Impacted Riverine Environment. Hydrology. 2018; 5(3):35. https://doi.org/10.3390/hydrology5030035
Chicago/Turabian StyleLangman, Jeff B., Kathleen Torso, and James G. Moberly. 2018. "Seasonal and Basinal Influences on the Formation and Transport of Dissolved Trace Metal Forms in a Mining-Impacted Riverine Environment" Hydrology 5, no. 3: 35. https://doi.org/10.3390/hydrology5030035
APA StyleLangman, J. B., Torso, K., & Moberly, J. G. (2018). Seasonal and Basinal Influences on the Formation and Transport of Dissolved Trace Metal Forms in a Mining-Impacted Riverine Environment. Hydrology, 5(3), 35. https://doi.org/10.3390/hydrology5030035






