1. Introduction
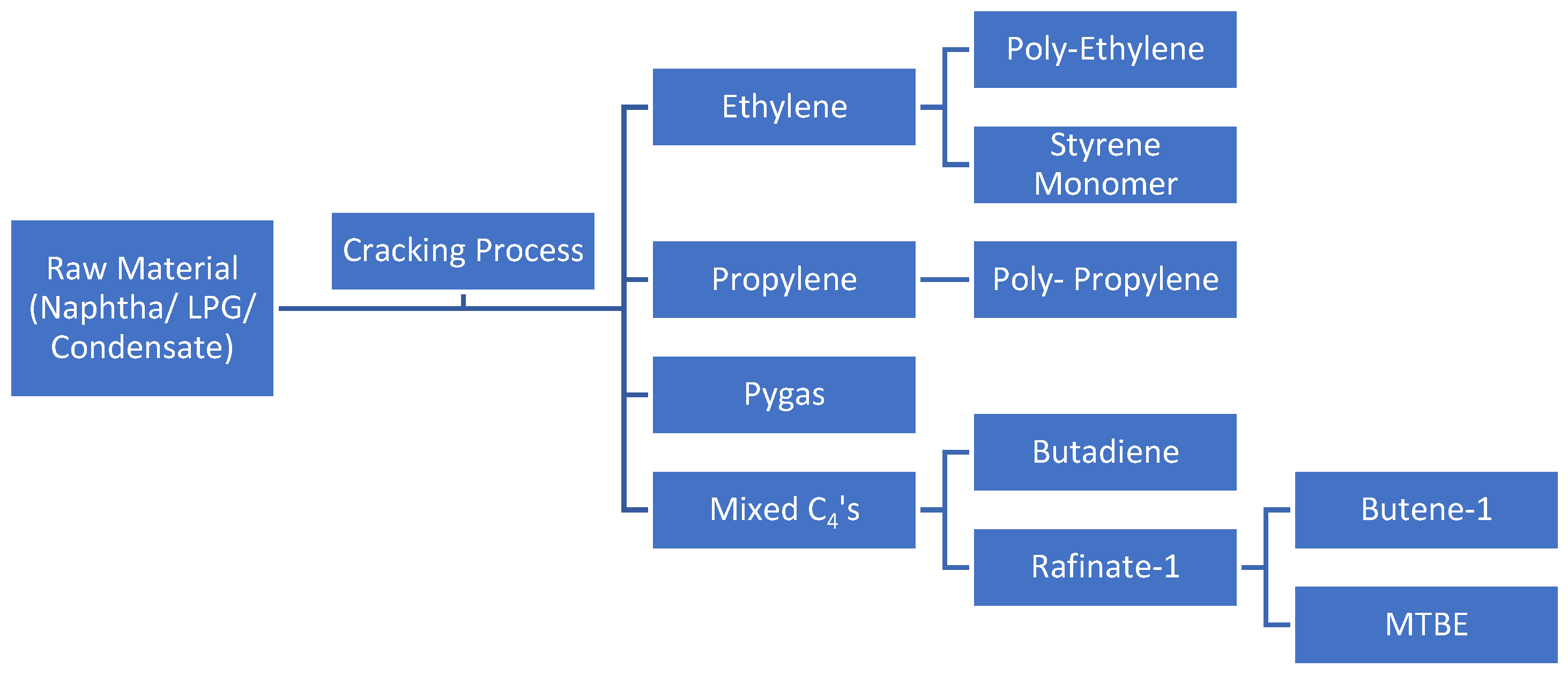
Ethylene, propylene, crude C4 (1,3-butadiene), and pygas (benzene, toluene, xylene) are the fundamental building blocks of the petrochemical industry. Naphtha cracking is the process that produces olefins most frequently (Kirk, 1988) [
1]. Up to 80% of the operational capacity of olefin plants is contributed by naphtha price. Olefins plants around the world offer the potential to replace naphtha feed into LPG due to ample LPG thanks to the shale oil production trend, LPG price decline, and LPG plenty. (Argus, 2018) [
2,
3].
However, switching from naphtha to LPG feed carries certain risks. The cracking furnace, where the feed undergoes steam cracking reactions, is a critical unit in olefin plants (Schaschke, 2014) [
4]. The cracking gas is then separated into ethylene, propylene, mixed C4 (including butadiene and other C4 compounds), and pygas (a mixture of benzene, toluene, and xylene streams), which are all pure olefins, in the distillation column. The process flow diagram is shown in
Figure 1.
Olefins plants utilizing LPG feed are expected to experience an increase in propylene and propane production (Argus, 2018) [
2]. As naphtha is commonly assumed to be the primary feed for most olefin plants, including the plant discussed in this article, it is crucial to consider the effects of LPG cracking on column hydraulics (Fakhroleslam, 2020) [
6].
According to Kister (1992) [
7], there are six key hydraulic parameters that significantly impact the operability of distillation columns:
Jet flooding: This occurs when a distillation column is operated with a high vapor load, causing liquid to be transported over the trays.
Down comer back flood: when a distillation column is run with a substantial liquid and vapor load, liquid accumulates in the top half of the column.
Down comer choke flood: excessive vapor flow results in liquid buildup in the trays’ down comers.
Weeping: low vapor load results in liquid draining from the tray channel.
Excessive pressure drops: the pressure drop across each tray should not exceed the design limit.
Turndown ratio: it is essential to feed the distillation column within its operational capacity to maintain acceptable efficiency.
The olefins plant under investigation consists of nine distillation columns, with the following configurations (Lummus, 2012) [
8]:
Demethanizer: This nine-bed distillation column operates at a pressure of 5.8 kg/cm2 and extracts heavier C2 compounds from methane. The top product is methane, with gauge and bottom side temperatures of −53 °C and −131 °C, respectively.
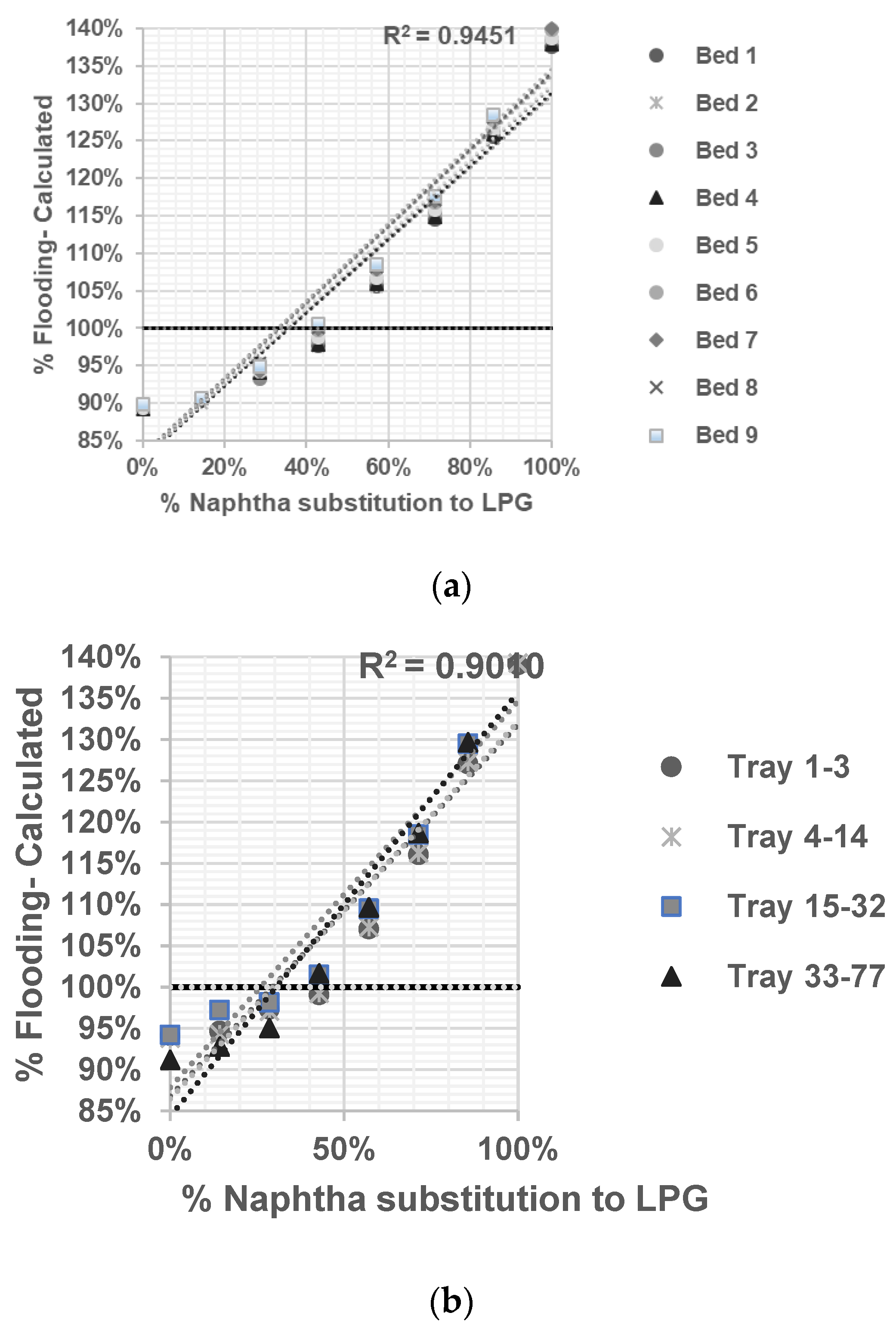
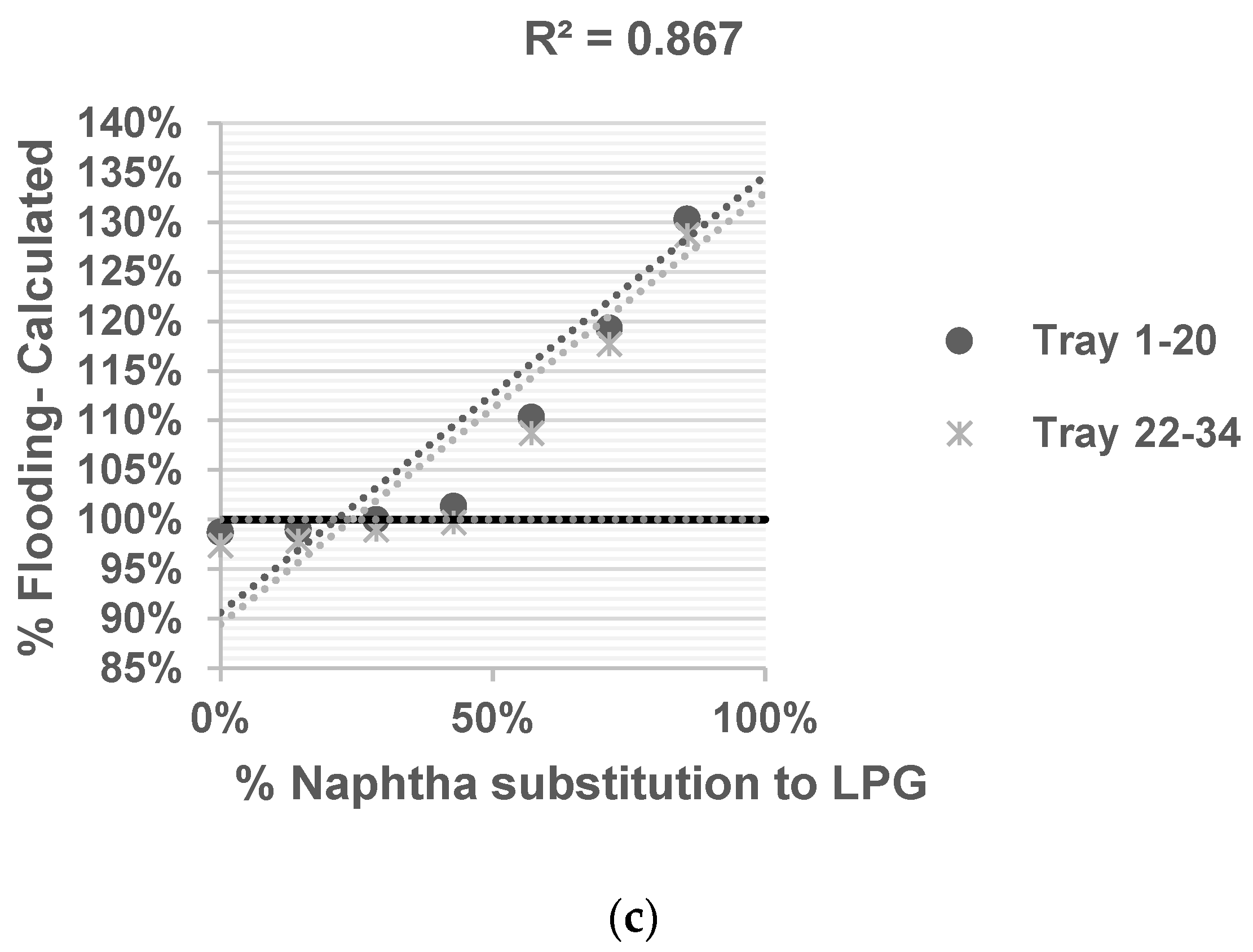
Deethanizer: A distillation column with 177 sieve trays is used to separate C2 compounds (the top product) from C3 compounds and heavier substances (the bottom product). It operates at a pressure of 21.3 kg/cm2, with top side and bottom side temperatures of −23 °C and 66 °C, respectively.
Ethylene Fractionator: With 137 sieve trays, this distillation column separates ethylene and ethane. It operates at pressures of 16.48 kg/cm2, with top side and bottom side temperatures of −35 °C and −12 °C, respectively.
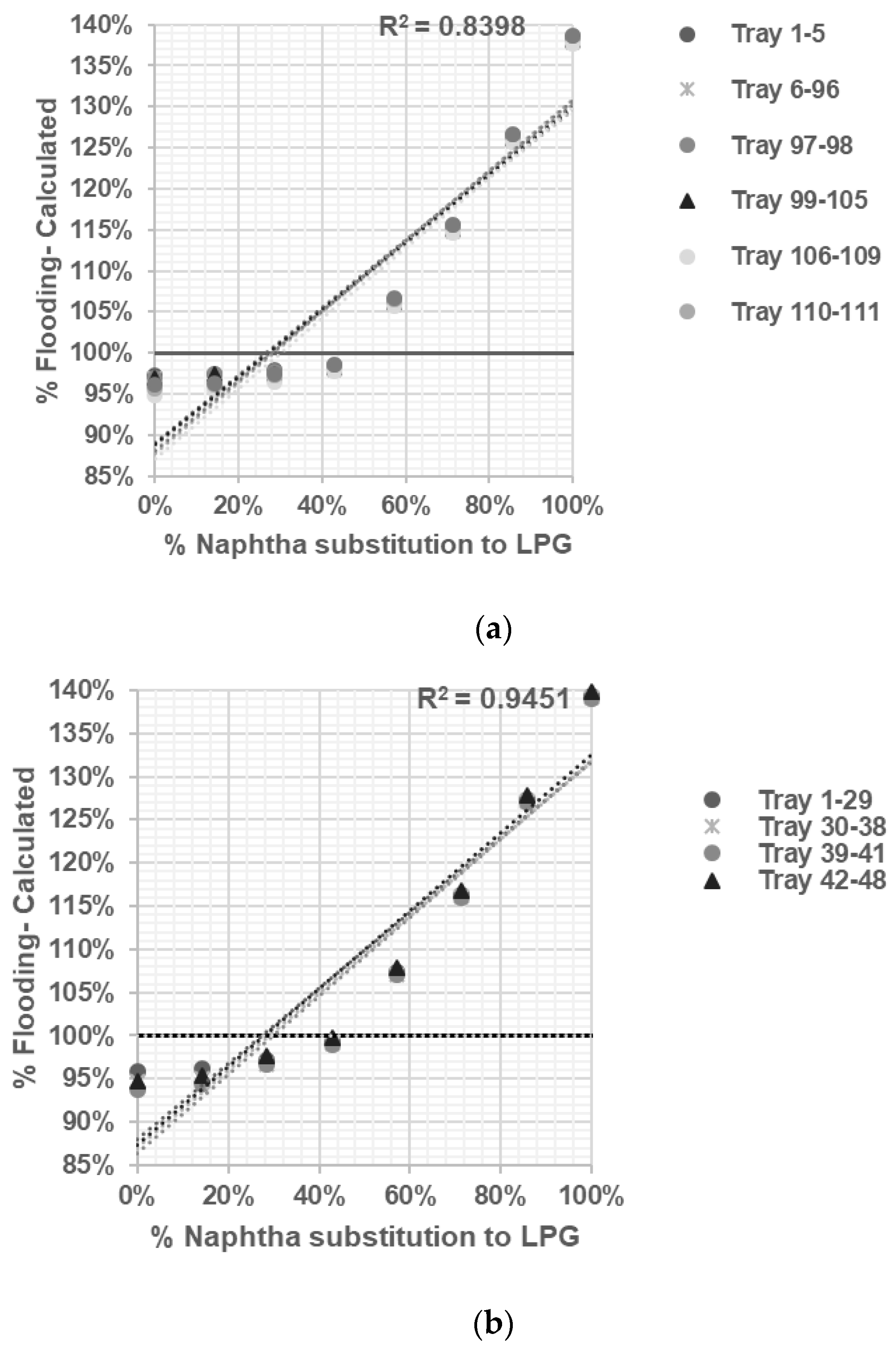
Depropanizer No. 1: This distillation column, equipped with 48 sieve trays, separates C3 compounds (the top product) from heavier C4 compounds (the bottom product). It operates at a pressure of 16.7 kg/cm2, with a top side temperature of 44 °C and a bottom side temperature of 82 °C.
Depropanizer No. 2: Using 30 sieve trays, this distillation column separates C3 compounds (the top product) from heavier C4 compounds (the bottom product). It operates at a pressure of 6.1 kg/cm2, with top side and bottom side temperatures of 38.2 °C and 82 °C, respectively.
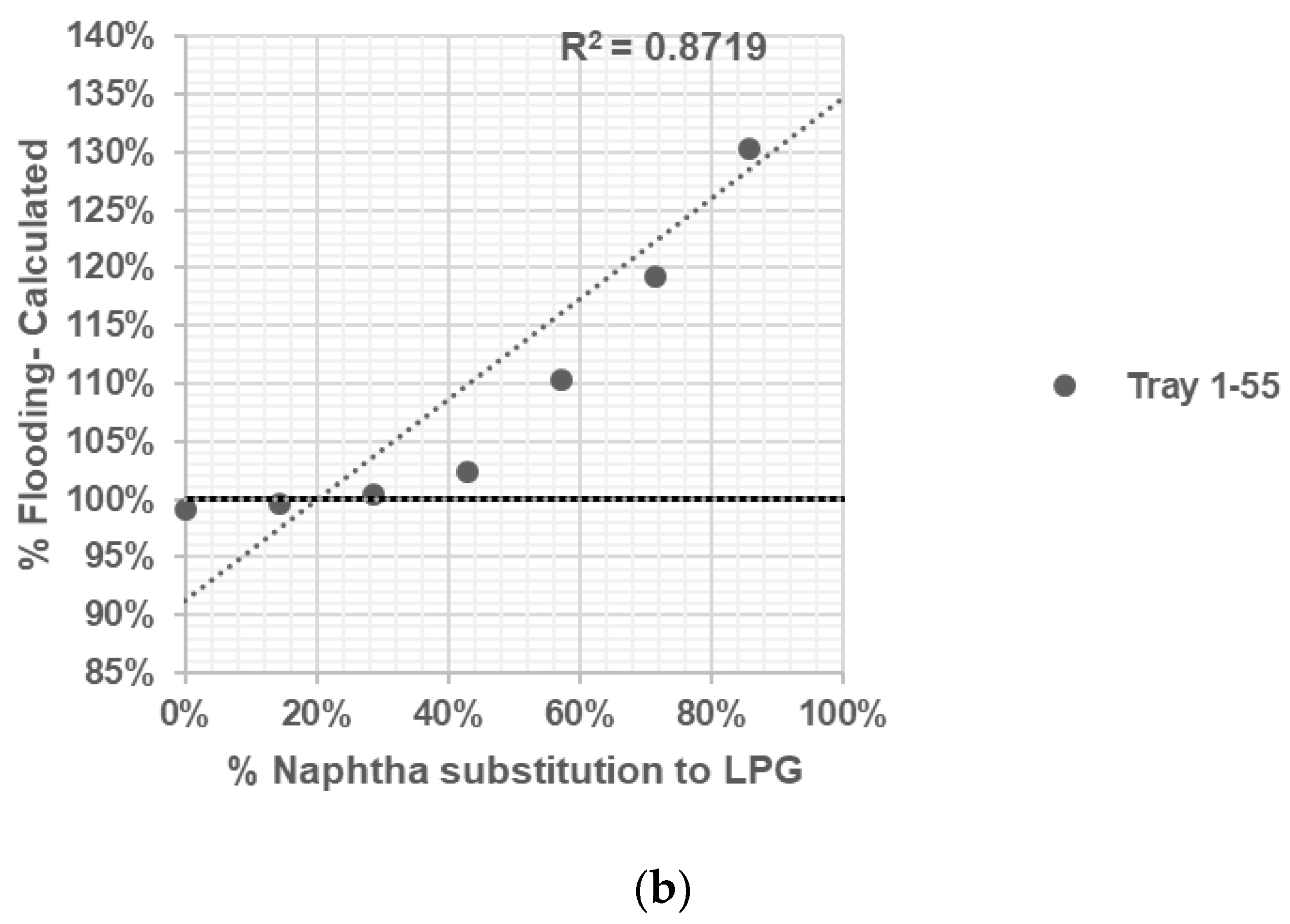
Propylene Fractionator No. 1: With 55 valve trays, this distillation column separates C3 compounds from propane. It operates at a pressure of 19.7 kg/cm2, with top side and bottom side temperatures of 50 °C and 58 °C, respectively.
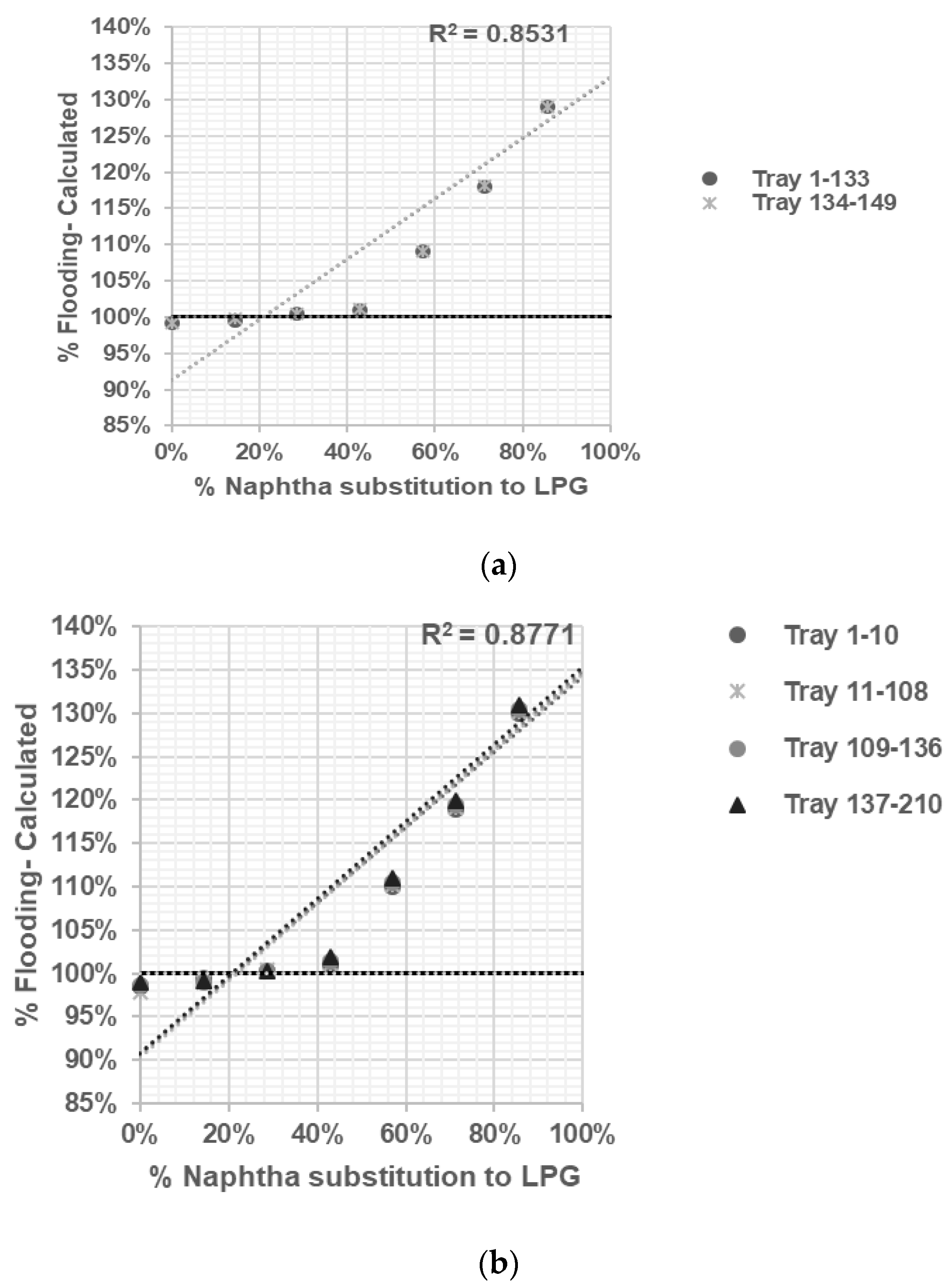
Propylene Fractionator No. 2: Propylene and propane are separated using 149 sieve trays in this distillation column, which operates at pressures of 19.2 kg/cm2 and temperatures of 46 °C (top side) and 50 °C (bottom side).
Propylene Fractionator No. 3: Propylene and propane are separated using 210 sieve trays in this distillation column, which operates at pressures of 18.3 kg/cm2 and temperatures of 46 °C (top side) and 58 °C (bottom side). This column can be interchanged with Propylene Fractionator No. 1 and No. 2.
Debutanizer: This distillation column, equipped with 34 valve trays, separates C4 compounds (the top product) from C5 compounds and heavier substances (the bottom product). It operates at a pressure of 4.34 kg/cm2, with top side and bottom side temperatures of 47 °C and 116 °C, respectively.
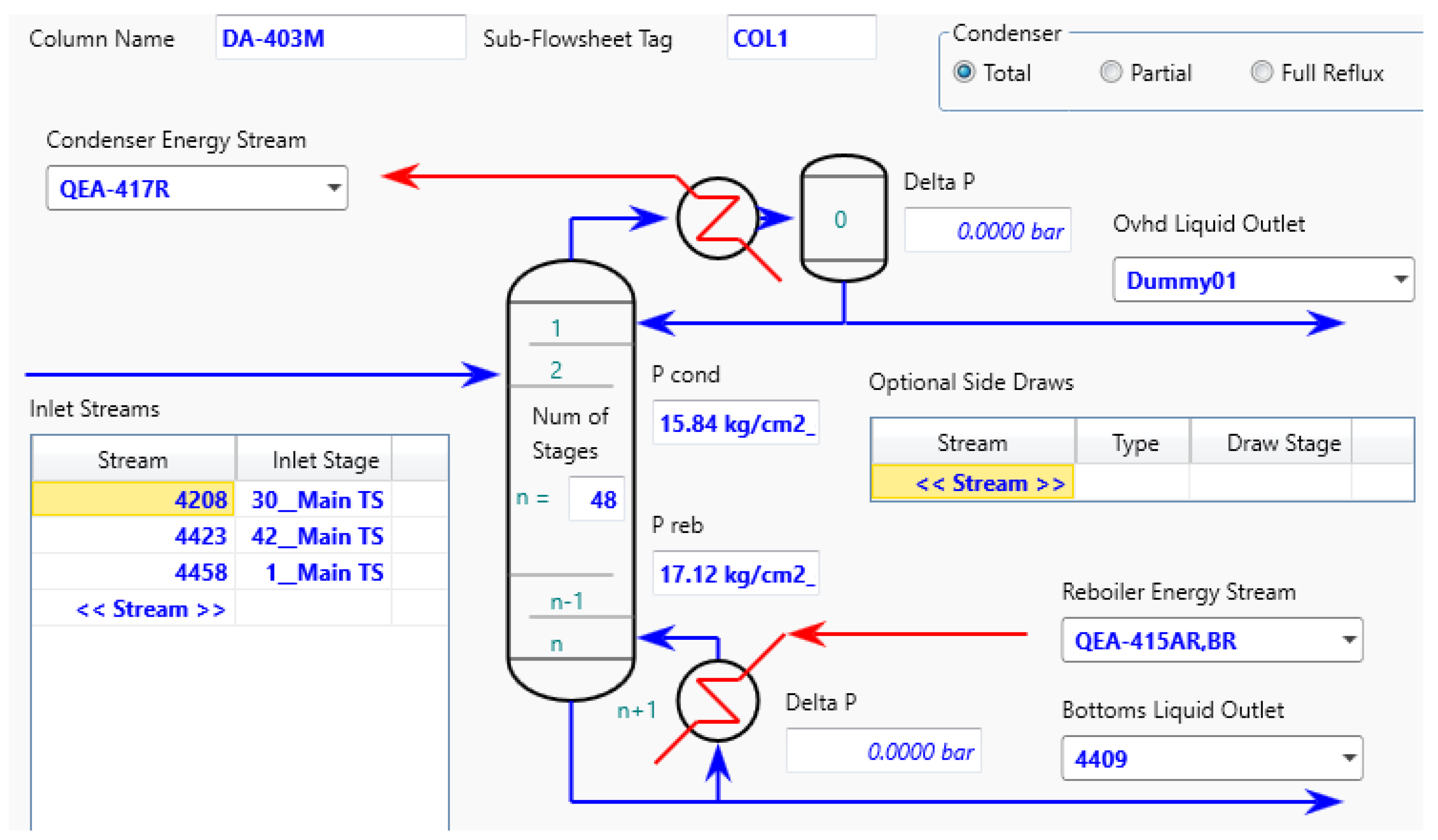
A model will be constructed for each distillation column based on actual data and simulated effects on column hydraulics. The objective is to determine the optimal substitution of naphtha with LPG without causing any adverse effects on the distillation column.
When evaluating the performance of distillation columns, the American Institute of Chemical Engineers (AIChE) Equipment Testing Procedures Committee considers the following parameters: ensuring that column performance meets vendor guarantees, identifying capacity bottlenecks, troubleshooting performance issues, determining the operating range of the column, defining optimum operating conditions, developing basic data and correlations for new designs, and calibrating computer simulations for use in optimization, bottleneck analysis, and design studies (CEP AIChE, 2013) [
9].
Kister (1992) [
3,
6,
10] explained that flooding can occur due to various mechanisms, including spray entrainment flooding, froth entrainment flooding, downcomer malfunctions, and defects in large diameter columns. Further details on these mechanisms can be found in the cited literature. For sieve trays, the entrainment flooding point can be predicted using the method developed by Kister and Haas [
3,
6,
10], shown in Equation (1). This method has been shown to accurately reproduce a large database of measured flood points within a 15 percent margin.
where, d
h = hole diameter, mm;
= surface tension, mN/m (dyn/cm);
,
= vapor and liquid densities, kg/m
3; TS = tray spacing, mm; h
ct = clear liquid height at froth to spray transition, mm; h
ct is obtained from the Equation (2):
, H2O derived from Equation (3)
In Equations (3) and (4),
= m
3 liquid down flow/(h,m weir length) and
= fractional hole based on active bubbling area; for example, derived from Equation (5).
The simulation tool used in this study is ASPEN HYSYS V.12, which is capable of simulating the thermodynamic characteristics and equilibrium of distillation column separations (Hanley, 2016) [
11]. The selection of properties for ASPEN HYSYS simulation is crucial to obtain reliable results (Luyben, 2014) [
12]. In this investigation, a fluid program was utilized to replicate the thermodynamic model, with guidance from (Yadav, 2020) [
13]. Here, the user must select the method based on the component type or process type. If one does not have an idea of selecting the appropriate method, there is an option called method assistant on the page. Method assistant gives the suitable method based on the component properties or specific area of application.
When developing a robust model, it is important to carefully choose the dependent and independent variables (Shinskey, 1991) [
14]. In this study, the independent variable is the LPG feed flow, while the dependent variables are the naphtha composition and LPG composition. The simulation model should be evaluated by adjusting tray efficiency to minimize the percentage error, and plant data should be used to validate the simulation against actual conditions (Loshchev, 2010) [
15].
4. Summary and Conclusions
In the present study, ASPEN HYSYS was utilized to conduct sensitivity analysis and assess the ability of the validated model in predicting the flooding phenomenon in distillation columns when substituting naphtha feed with LPG. ASPEN HYSYS is a simulation-based tool that incorporates thermodynamic properties’ packages, allowing for the selection of appropriate packages based on the system’s physical conditions.
By employing modeling, optimization, and prediction techniques within ASPEN HYSYS, the operating conditions and limitations of distillation columns—particularly regarding jet flooding—could be predicted without risking the actual plant conditions. This approach provides a safe and efficient way to explore different scenarios and evaluate the impact of modifications on column internals.
To further enhance the accuracy of the model and ensure its alignment with actual plant conditions, future efforts will be directed towards improving the model’s performance, and conducting thorough assessments of the modification effects on column internals. These ongoing developments will contribute to advancing the understanding and optimization of distillation column operations.