Utilization of Spent Sorbent in the Production of Ceramic Bricks
Abstract
1. Introduction
- -
- Sludge from domestic or industrial wastewater, which, as a rule, contains a lot of organic matter and significant amounts of heavy metals;
- -
- Ash from thermal power plants, boiler houses, waste incineration plants, etc. The composition of the ashes depends on the origin of the fuel, the technological parameters of its preparation and combustion, and the storage conditions in the dumps;
- -
- Inorganic waste, mainly waste from mining and smelting complexes. The largest number of studies is devoted to the use of dust and slag;
- -
- Organic waste: agricultural, pulp and paper, and woodworking industries, which are used mainly as blowing agents and energy savers during firing.
- -
- Possibility of obtaining ceramic bricks on the basis of these wastes;
- -
- Use of basalt fiber in ceramic bricks as thinners and sintering intensifiers;
- -
- Phase transformations occurring during the firing of ceramic bricks using bentonite clay and basalt fiber;
- -
- Influence of the used sorbent on the physical and mechanical characteristics of ceramic bricks.
2. Materials and Methods
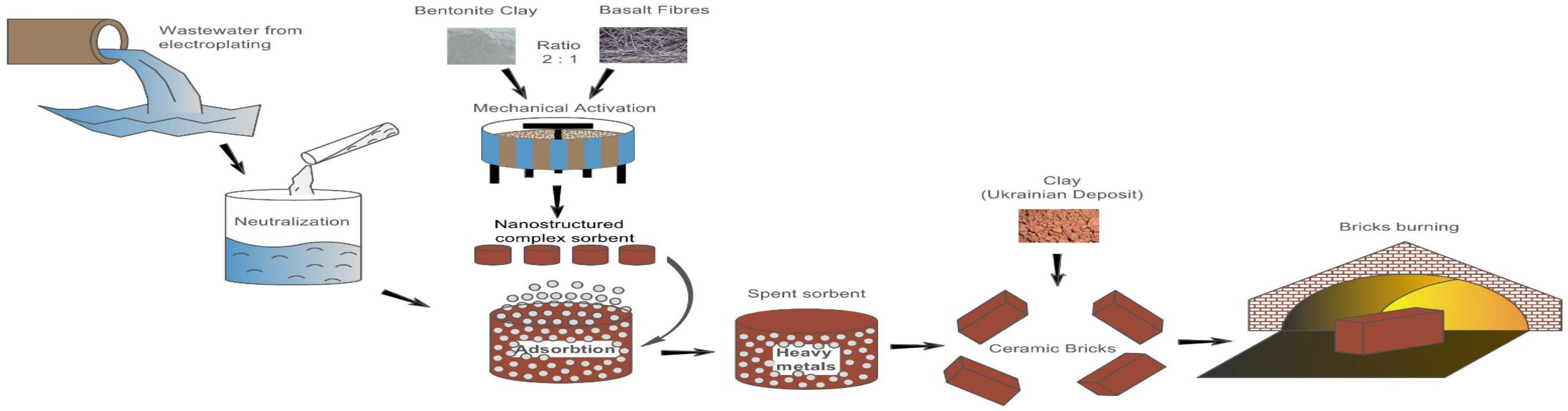
2.1. Preparation of Raw Material (Batch) for Bricks
2.2. Characteristics of the Raw Material
2.3. Research Methods
3. Results and Discussion
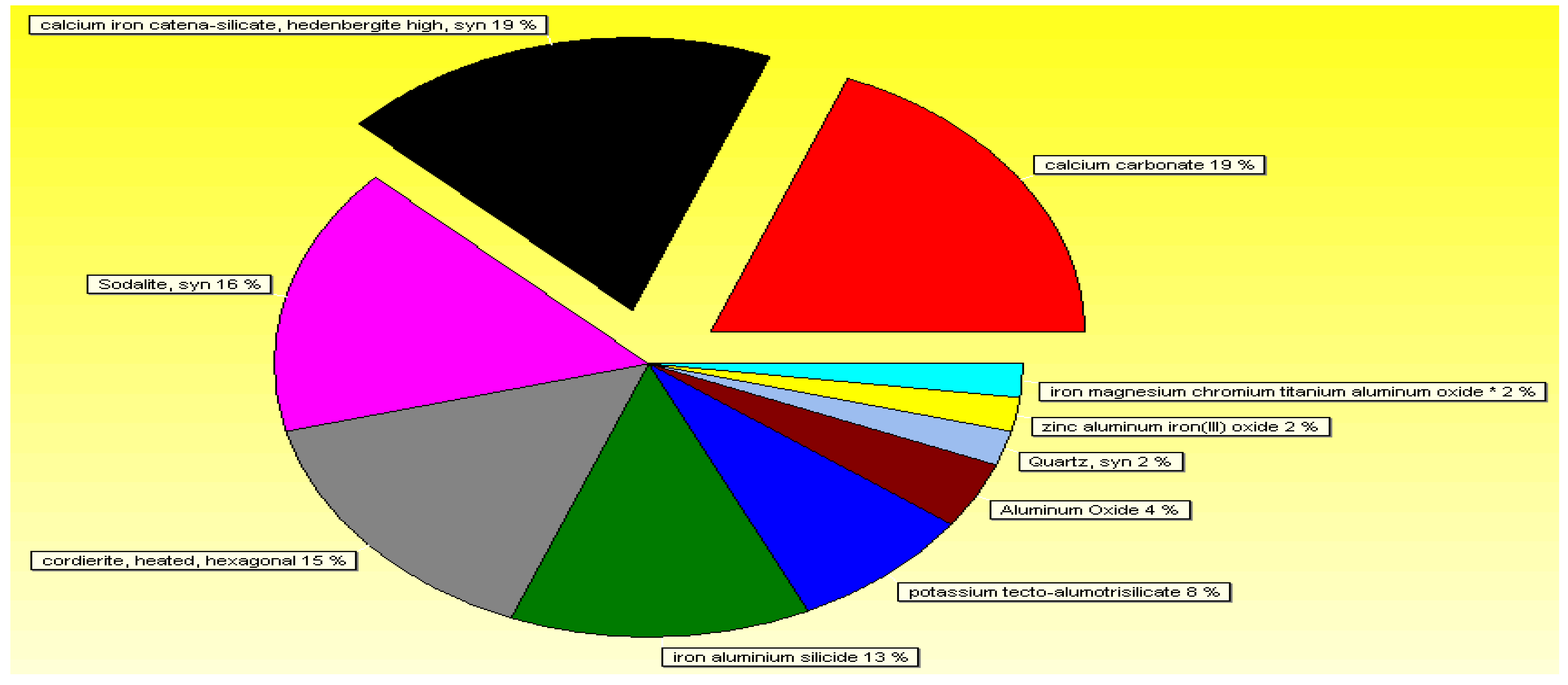
3.1. Characteristics and Chemical Composition of the Raw Material
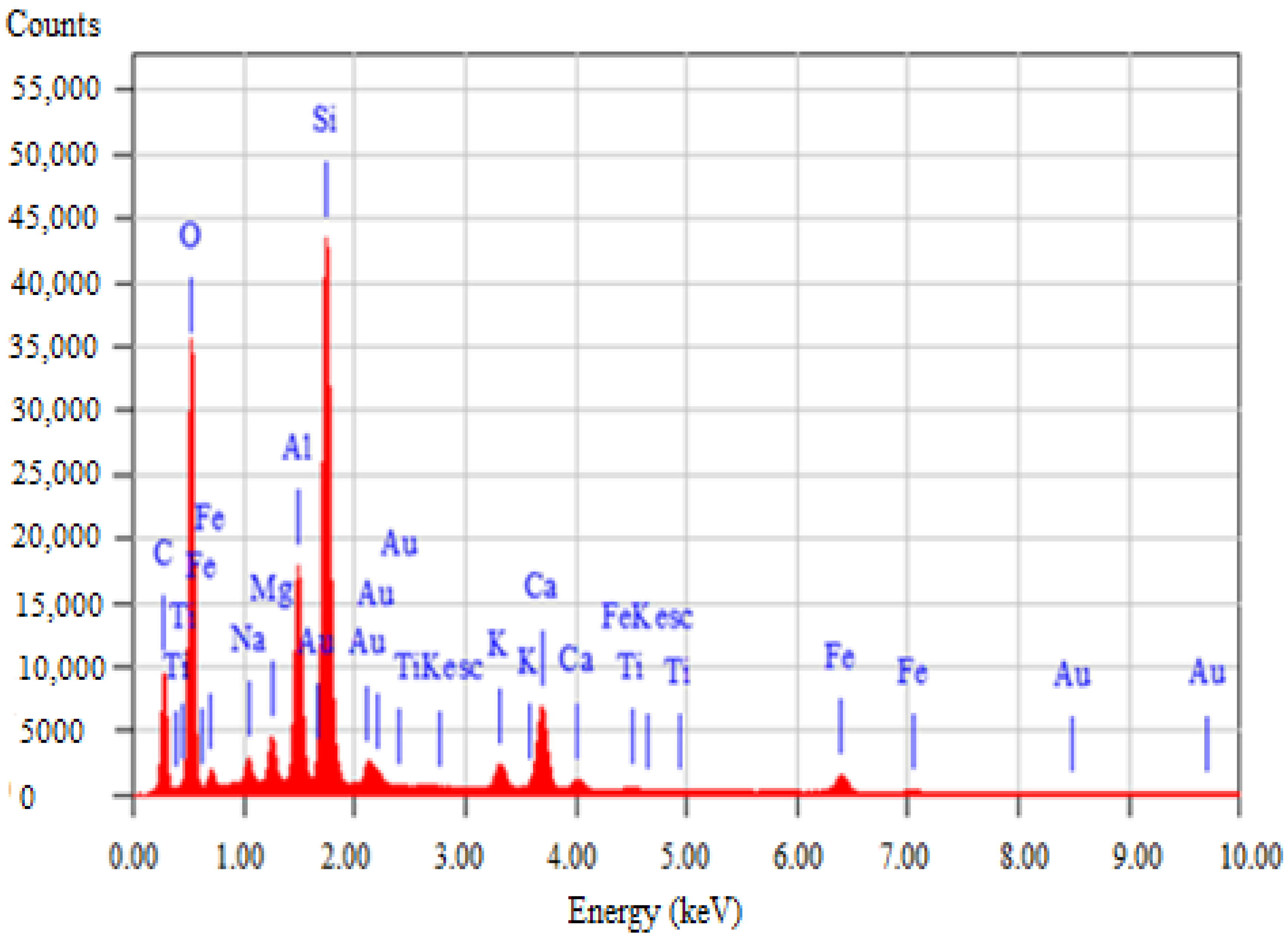
3.2. Characteristics and Chemical Composition of the Spent Sorbent
3.3. Test Results for Freshly Molded Samples
- -
- Mode 1—48 h at 20 °C and 24 h at 20–105 °C;
- -
- Mode 2—24 h at 20 °C and 24 h at 20–105 °C;
- -
- Mode 3—12 h at 20 to 60 °C and 12 h at 60–105 °C;
- -
- Mode 4—12 h at 20 to 105 °C.
3.4. Firing of Ceramic Materials
3.5. Physical and Mechanical Testing of Samples
3.6. Influence of the Spent Sorbent on the Technological Properties of Ceramic Bricks
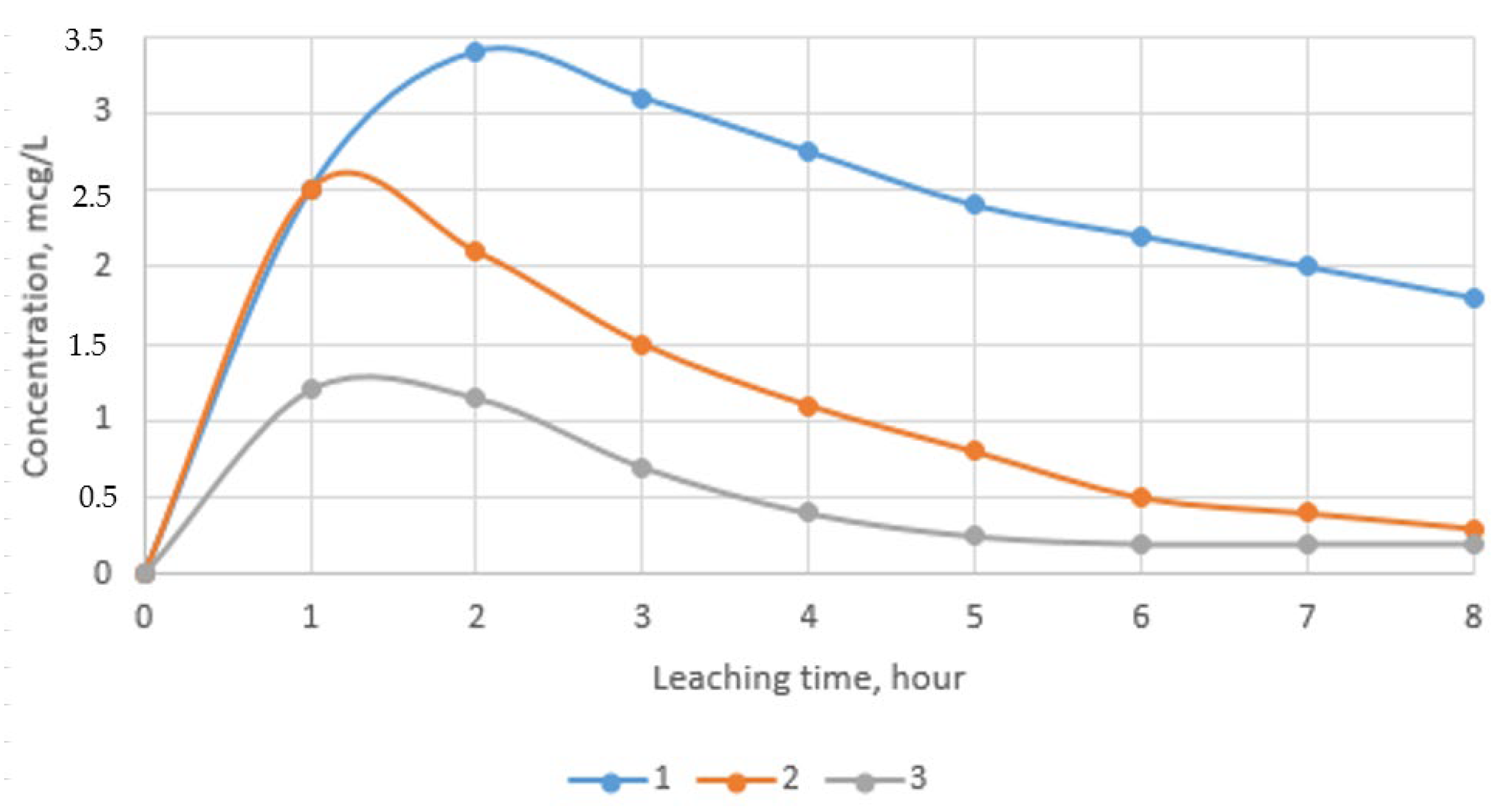
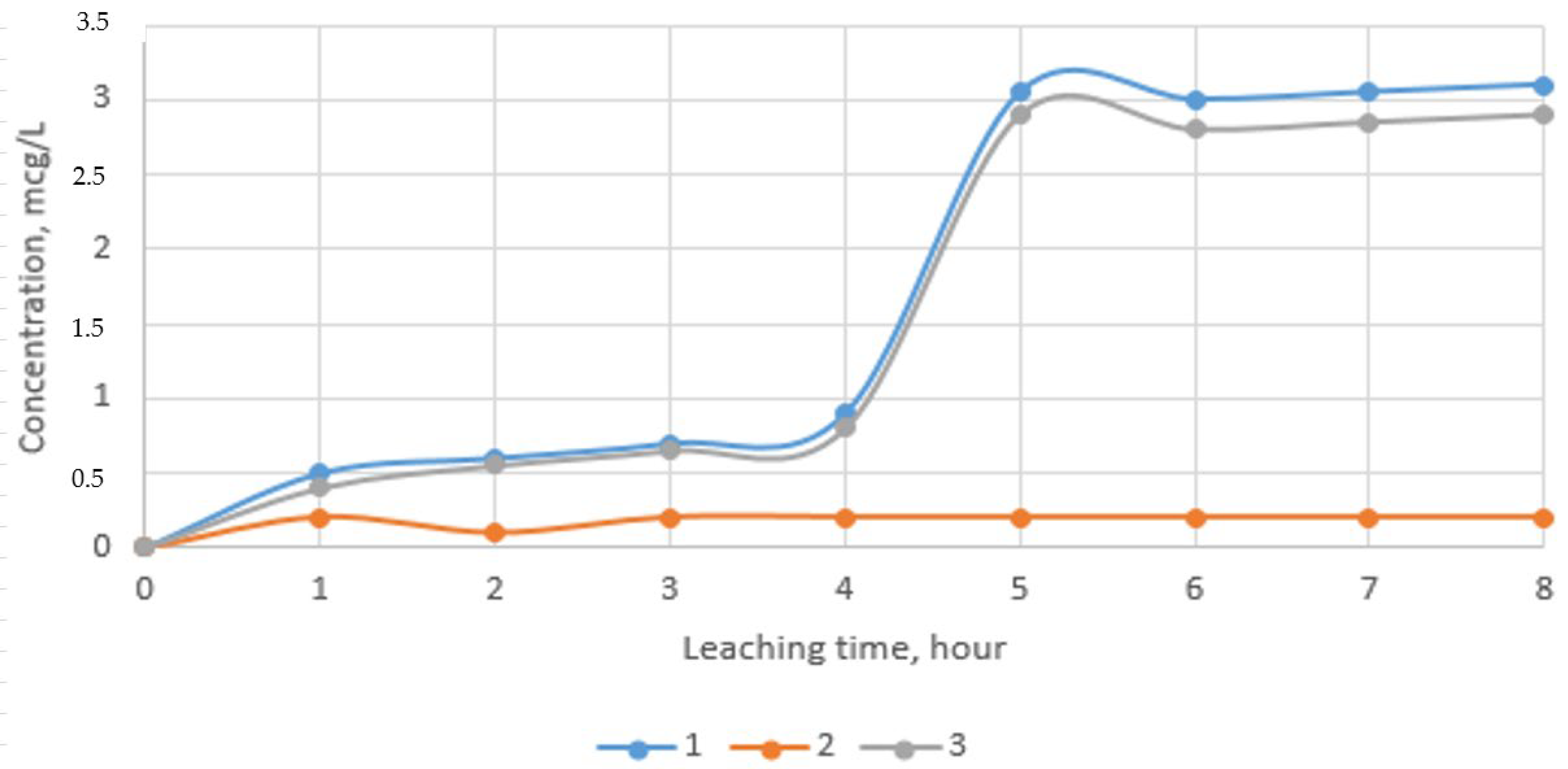
3.7. Study of Environmental Safety of Waste Sorbents Disposal
4. Conclusions
- -
- The resulting ceramic bricks have better compressive strength than reference bricks without the addition of spent sorbent. From the moisture values of the compared samples (8, 10, 12%), it was found that with an increase in the moisture content of the bricks, the compressive strength increases within the specified limits.
- -
- Among the studied firing temperatures (950, 1000, 1050, 1100 °C), the highest value of compressive strength was obtained for brick fired at a temperature of 950 °C.
- -
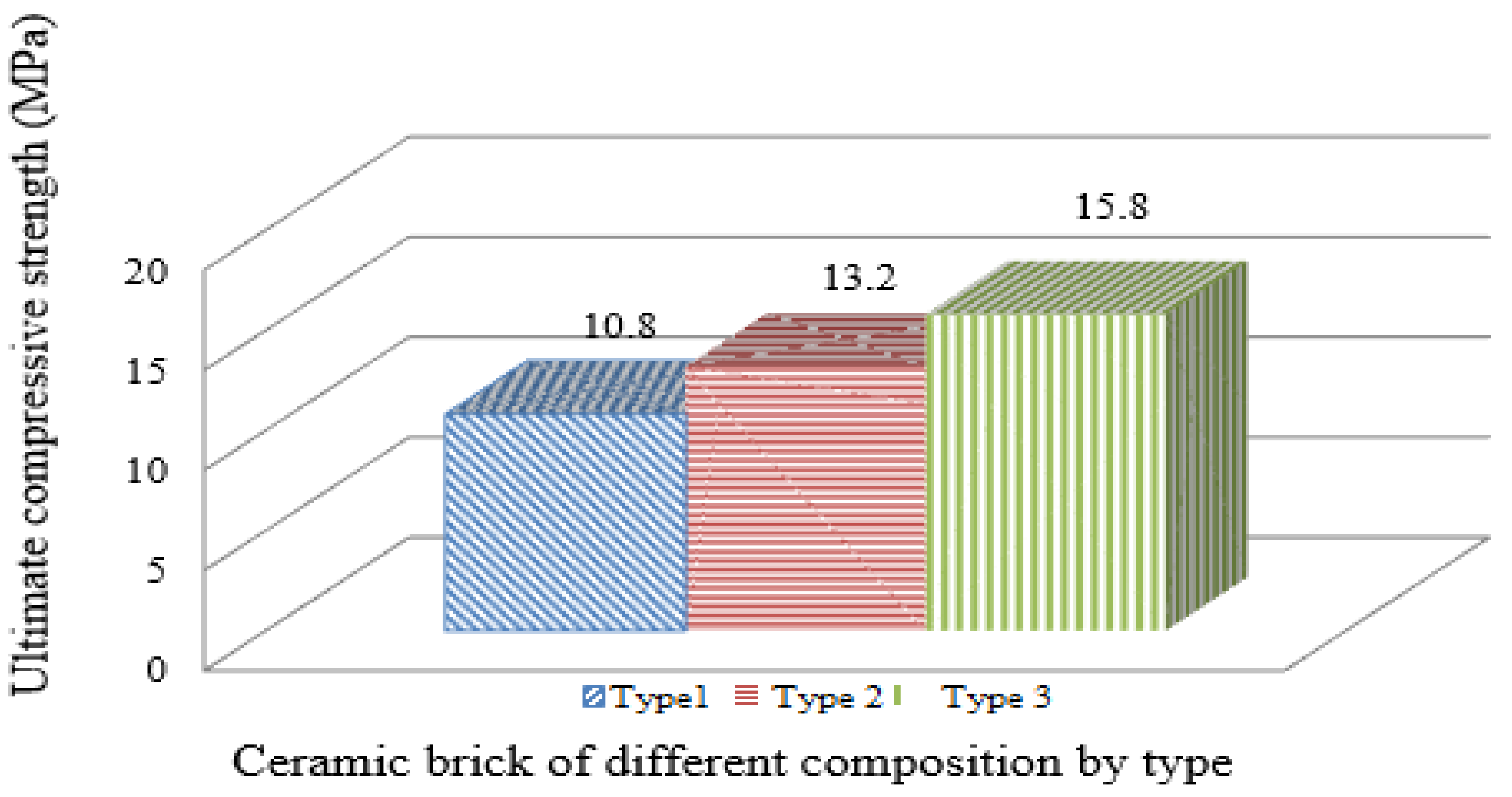
- When used as part of a ceramic mass with 10% used sorbent additive, the maximum compressive strength is 13.2 MPa. Moreover, for a brick containing 25% of the spent sorbent, the compressive strength increased to 15.8 MPa compared to the reference sample (10.8 MPa).
- -
- The frost resistance of the obtained bricks averaged 35 cycles.
- -
- Air shrinkage for the obtained samples has a zero value, which indicates the expediency of using the spent sorbent as part of the ceramic mass of the brick. With fire shrinkage, the average value is 2%. The presence of basalt fiber in the spent sorbent stabilizes shrinkage deformations.
- -
- The produced ceramic brick with an additive of 25% with improved physical and mechanical characteristics has the M125 grade, higher than the reference sample of the M100 grade.
- -
- When studying the microstructure, it was found that the reference sample has a microporous structure, in contrast to the brick sample with the addition of spent sorbent. The addition of 25% of the spent sorbent to the ceramic mass led to the ordering of the structure and an increase in homogeneity. The spent sorbent with the additive has secondary macropores of an elongated or irregular shape.
- -
- The environmental safety of waste sorbent disposal was established. According to the results of experimental studies, it was shown that the concentration of chromium and zinc ions in extracts in various media is not more than 3.5 μg/L, which is associated with the transition of heavy metals into strong and sparingly soluble compounds during high-temperature processing.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramos Huarachi, D.A.; Gonçalves, G.; de Francisco, A.C.; Canteri, M.H.G.; Piekarski, C.M. Life cycle assessment of traditional and alternative bricks: A review. Environ. Impact Assess. Rev. 2020, 80, 106335. [Google Scholar] [CrossRef]
- Boltakova, N.V.; Faseeva, G.R.; Kabirov, R.R.; Nafikov, R.M.; Zakharov, Y.A. Utilization of inorganic industrial wastes in producing construction ceramics. Review of Russian experience for the years 2000–2015. Waste Manag. 2017, 60, 230–246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wong, Y.C.; Arulrajah, A.; Horpibulsuk, S. A review of studies on bricks using alternative materials and approaches. Constr. Build. Mater. 2018, 188, 1101–1118. [Google Scholar] [CrossRef]
- Wolff, E.; Schwabe, W.K.; Conceição, S.V. Utilization of water treatment plant sludge in structural ceramics. J. Clean. Prod. 2015, 96, 282–289. [Google Scholar] [CrossRef]
- Atan, E.; Sutcu, M.; Cam, A.S. Combined effects of bayer process bauxite waste (red mud) and agricultural waste on technological properties of fired clay bricks. J. Build. Eng. 2021, 43, 103194. [Google Scholar] [CrossRef]
- Barnabas, A.A.; Balogun, O.A.; Akinwande, A.A.; Ogbodo, J.F.; Ademati, A.O.; Dongo, E.I.; Romanovski, V. Reuse of walnut shell waste in the development of fired ceramic bricks. Environ. Sci. Pollut. Res. 2022, 1–5. [Google Scholar] [CrossRef]
- Zhang, L. Production of bricks from waste materials—A review. Constr. Build. Mater. 2013, 47, 643–655. [Google Scholar] [CrossRef]
- Muñoz Velasco, P.; Morales Ortíz, M.P.; Mendívil Giry, M.A.; Muñoz Velasco, L. Fired clay bricks manufactured by adding wastes as sustainable construction material—A review. Constr. Build. Mater. 2014, 63, 97–107. [Google Scholar] [CrossRef]
- Yang, F.; Zhou, W.; Zhu, R.; Dai, G.; Wang, W.; Wang, W.; Wang, Z. Synergistic effects of amorphous porous materials and anhydrous Na2CO3 on the performance of bricks with high municipal sewage sludge content. J. Clean. Prod. 2021, 280, 124338. [Google Scholar] [CrossRef]
- Zat, T.; Bandieira, M.; Sattler, N.; Segadães, A.M.; Cruz, R.C.D.; Mohamad, G.; Rodríguez, E.D. Potential re-use of sewage sludge as a raw material in the production of eco-friendly bricks. J. Environ. Manag. 2021, 297, 113238. [Google Scholar] [CrossRef]
- Bandieira, M.; Zat, T.; Schuster, S.L.; Justen, L.H.; Weide, H.; Rodríguez, E.D. Water treatment sludge in the production of red-ceramic bricks: Effects on the physico-mechanical properties. Mater. Struct. 2021, 54, 168. [Google Scholar] [CrossRef]
- Cangussu, N.; Vasconcelos, L.; Maia, L. Environmental benefits of using sewage sludge in the production of ceramic bricks. Environ. Sci. Pollut. Res. 2022, 1–12. [Google Scholar] [CrossRef]
- Zhang, X.; Jiao, Y.; Yu, L.; Liu, L.; Wang, X.; Zhang, Y. Effect of Sewage Sludge Addition on Microstructure and Mechanical Properties of Kaolin-Sewage Sludge Ceramic Bricks. Coatings 2022, 12, 944. [Google Scholar] [CrossRef]
- Bubalo, A.; Vouk, D.; Stirmer, N.; Nad, K. Use of Sewage Sludge Ash in the Production of Innovative Bricks-An Example of a Circular Economy. Sustainability 2021, 13, 9330. [Google Scholar] [CrossRef]
- Amin, F.; Abbas, S.; Abbass, W.; Salmi, A.; Ahmed, A.; Saeed, D.; Sufian, M.; Sayed, M.M. Potential Use of Wastewater Treatment Plant Sludge in Fabrication of Burnt Clay Bricks. Sustainability 2022, 14, 6711. [Google Scholar] [CrossRef]
- Wu, K.; Hu, Y.; Zhang, L.; Xu, L.; Yang, Z. Promoting the sustainable fabrication of bricks from municipal sewage sludge through modifying calcination: Microstructure and performance characterization. Constr. Build. Mater. 2022, 324, 126401. [Google Scholar] [CrossRef]
- Chang, Z.; Long, G.; Zhou, J.L.; Ma, C. Valorization of sewage sludge in the fabrication of construction and building materials: A review. Resour. Conserv. Recycl. 2020, 154, 104606. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Jia, L.T.; Mei, H.; Cui, Q.; Zhang, P.G.; Sun, Z.M. Fabrication, microstructure and properties of bricks fired from lake sediment, cinder and sewage sludge. Constr. Build. Mater. 2016, 121, 154–160. [Google Scholar] [CrossRef]
- Benlalla, A.; Elmoussaouiti, M.; Dahhou, M.; Assafi, M. Utilization of water treatment plant sludge in structural ceramics bricks. Appl. Clay Sci. 2015, 118, 171–177. [Google Scholar] [CrossRef]
- Zasidko, I.; Polutrenko, M.; Mandryk, O.; Stakhmych, Y.; Petroshchuk, N. Complex Technology of Sewage Purification from Heavy-Metal Ions by Natural Adsorbents and Utilization of Sewage Sludge. J. Ecol. Eng. 2019, 20, 209–216. [Google Scholar] [CrossRef]
- Pérez-Villarejo, L.; Martínez-Martínez, S.; Carrasco-Hurtado, B.; Eliche-Quesada, D.; Ureña-Nieto, C.; Sánchez-Soto, P.J. Valorization and inertization of galvanic sludge waste in clay bricks. Appl. Clay Sci. 2015, 105–106, 89–99. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, C.; Mao, L. Use of electroplating sludge in production of fired clay bricks: Characterization and environmental risk evaluation. Constr. Build. Mater. 2018, 159, 27–36. [Google Scholar] [CrossRef]
- Mymrine, V.; Ponte, M.J.J.S.; Ponte, H.A.; Kaminari, N.M.S.; Pawlowsky, U.; Solyon, G.J.P. Oily diatomite and galvanic wastes as raw materials for red ceramics fabrication. Constr. Build. Mater. 2013, 41, 360–364. [Google Scholar] [CrossRef]
- Mymrin, V.A.; Alekseev, K.P.; Zelinskaya, E.V.; Tolmacheva, N.A.; Catai, R.E. Industrial sewage slurry utilization for red ceramics production. Constr. Build. Mater. 2014, 66, 368–374. [Google Scholar] [CrossRef]
- Li, M.; Su, P.; Guo, Y.; Zhang, W.; Mao, L. Effects of SiO2, Al2O3 and Fe2O3 on leachability of Zn, Cu and Cr in ceramics incorporated with electroplating sludge. J. Environ. Chem. Eng. 2017, 5, 3143–3150. [Google Scholar] [CrossRef]
- De Carvalho Gomes, S.; Zhou, J.L.; Li, W.; Long, G. Progress in manufacture and properties of construction materials incorporating water treatment sludge: A review. Resour. Conserv. Recycl. 2019, 145, 148–159. [Google Scholar] [CrossRef]
- Wei, N. Leachability of Heavy Metals from Lightweight Aggregates Made with Sewage Sludge and Municipal Solid Waste Incineration Fly Ash. Int. J. Environ. Res. Public Health 2015, 12, 4992–5005. [Google Scholar] [CrossRef] [PubMed]
- Dousova, B.; Kolousek, D.; Keppert, M.; Machovic, V.; Lhotka, M.; Urbanova, M.; Holcova, L. Use of waste ceramics in adsorption technologies. Appl. Clay Sci. 2016, 134, 145–152. [Google Scholar] [CrossRef]
- Svatovskaya, L.B.; Maslennikova, L.L.; Babak, N.A.; Kapustina, O.A. Ceramic Mass. RU Patent No. RU2416585, 20 April 2011. p. 3. [Google Scholar]
- Ibrahim, J.E.F.M.; Kotova, O.B.; Sun, S.; Kurovics, E.; Tihtih, M.; Gömze, L.A. Preparation of innovative eco-efficient composite bricks based on zeolite-poor rock and Hen′s eggshell. J. Build. Eng. 2022, 45, 103491. [Google Scholar] [CrossRef]
- Abdrakhimov, V.Z. The use of waste mineral wool in the production of ceramic wall materials. Constr. Geotech. 2019, 10, 53–60. [Google Scholar] [CrossRef]
- Daumova, G.K.; Abdulina, S.A.; Kokayeva, G.A.; Adilkanova, M.A. Experimental studies on wastewater sorption treatment with subsequent disposal of used sorbents. Chem. Eng. Trans. 2018, 70, 2125–2130. [Google Scholar] [CrossRef]
- GOST 21216-2014; Clay Raw Materials. Test Methods. Publisher Standardinform: Moscow, Russia, 2015. Available online: https://docs.cntd.ru/document/1200115068 (accessed on 1 July 2015).
- GOST 9169-75; Clayish Materials for Ceramic Industry. Classification. Publisher Standardinform: Moscow, Russia, 1976. Available online: https://rosgosty.com/down/r-82.html (accessed on 1 July 1976).
- GOST 12536-2014; Soils. Methods of Laboratory Granulometric (Grain Size) and Macroaggregate Distribution. Publisher Standardinform: Moscow, Russia, 2015. Available online: https://files.stroyinf.ru/Data2/1/4293766/4293766967.htm (accessed on 7 January 2015).
- GOST 8462-85; Wall materials. Methods for Determination of Ultimate Compressive and Bending Strength. Publisher Standardinform: Moscow, Russia, 1985. Available online: https://files.stroyinf.ru/Index2/1/4294853/4294853175.htm (accessed on 1 July 1985).
- Alemu, A.; Lemma, B.; Gabbiye, N.; Alula, M.T.; Desta, M.T. Removal of chromium (VI) from aqueous solution using vesicular basalt: A potential low cost wastewater treatment system. Heliyon 2018, 4, e00682. [Google Scholar] [CrossRef]
- Mamyachenkov, S.V.; Adryshev, A.K.; Seraya, N.V.; Khairullina, A.A.; Daumova, G.K. Nanostructured Complex Sorbent for Cleaning Heavy Metal Ions from Industrial Effluent. Metallurgist 2017, 61, 615–623. [Google Scholar] [CrossRef]
- GOST 530-2012; Ceramic Brick and Stone. General Specifications. Publisher Standardinform: Moscow, Russia, 2013. Available online: https://ru.scribd.com/document/507788314/GOST-530-2012-Ceramic-brick-and-stone-General-specifications-GOST-dated-December-27-2012-530 (accessed on 1 July 2013).
- Vakalova, T.V.; Revva, I.B. Use of zeolite rocks for ceramic bricks based on brick clays and clay loams with high drying sensitivity. Constr. Build. Mater. 2020, 255, 119324. [Google Scholar] [CrossRef]









| Oxide | SiO2 | Al2O3 + TiO2 | Fe2O3 | CaO | MgO | R2O | SO3 | LOI |
|---|---|---|---|---|---|---|---|---|
| % | 58.17 | 14.21 | 4.54 | 6.95 | 2.30 | 3.42 | 0.37 | 10.04 |
| Sampling Depth of Clay Raw Materials, m | Granulometric Composition According to the Rutkovsky Method | Plasticity Number | Drying Sensitivity Coefficient | ||
|---|---|---|---|---|---|
| Clay Fraction < 0.005 mm | Dust Fraction 0.005 ÷ 0.05 mm | Sand Fraction 0.05 ÷ 5 mm | |||
| 0,3–4 | 17.88 | 63.12 | 27.0 | 8.5 | 0.61 |
| 4–8 | 9.12 | 40.0 | 28.9 | 9.4 | 0.91 |
| 8–11,0 | 8.5 | 78.0 | 8.0 | 11.1 | 0.59 |
| 11–14 | 7.64 | 68.36 | 24.0 | 7.7 | 0.52 |
| 14–17 | 18.78 | 71.22 | 20.0 | 9.0 | 0.54 |
| Spectrum | O | Na | Mg | Al | Si | Ca | Fe | Sum |
|---|---|---|---|---|---|---|---|---|
| Spectrum 1 | 49.66 | 0.32 | 1.86 | 5.28 | 7.86 | 18.34 | 16.68 | 100.00 |
| Spectrum 2 | 59.49 | 0.51 | 1.46 | 4.17 | 12.52 | 19.16 | 2.70 | 100.00 |
| Spectrum 3 | 59.80 | 0.77 | 1.83 | 4.78 | 7.81 | 22.11 | 2.89 | 100.00 |
| Max. | 59.80 | 0.77 | 1.86 | 5.28 | 12.52 | 22.11 | 16.68 | - |
| Min. | 49.66 | 0.32 | 1.46 | 4.17 | 7.81 | 18.34 | 2.70 | - |
| Spectrum | O | Na | Mg | Al | Si | S | Ca | Cr | Fe | K | Sum |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Spectrum 1 | 46.48 | 0.54 | 1.40 | 2.00 | 6.20 | 4.55 | 16.87 | 0.19 | 2.43 | 19.34 | 100.00 |
| Spectrum 2 | 37.60 | 0.54 | 1.53 | 2.27 | 6.86 | 0.17 | 13.57 | 0.41 | 37.05 | - | 100.00 |
| Spectrum 3 | 54.16 | 0.44 | 1.45 | 2.21 | 7.91 | 0.22 | 29.91 | 0.28 | 3.43 | - | 100.00 |
| Spectrum 4 | 49.35 | 0.42 | 1.82 | 2.82 | 9.39 | 0.47 | 24.00 | 0.26 | 11.47 | - | 100.00 |
| Max. | 54.16 | 0.54 | 1.82 | 2.82 | 9.39 | 4.55 | 29.91 | 0.41 | 37.05 | 19.34 | - |
| Min. | 37.60 | 0.42 | 1.40 | 2.00 | 6.20 | 0.17 | 13.57 | 0.19 | 2.43 | 19.34 | - |
| Series No. | Number of Samples | Moisture Content of Powder, % | Sample Weight before Drying, g | Sample Weight after Drying, g | Raw Strength, MPa |
|---|---|---|---|---|---|
| 1 | 4 | 8 | 220 | 200.92 | 1.0 |
| 2 | 4 | 10 | 220 | 196.73 | 1.1 |
| 3 | 4 | 12 | 220 | 196.14 | 1.3 |
| Series № | Moisture Content of Powder, % | Firing Temperature, °C | Sample Weight after Firing, g | Shrinkage, in% | Ultimate Compressive Strength of Samples, MPa | Frost Resistance, Cycles | |
|---|---|---|---|---|---|---|---|
| Air | Fire | ||||||
| 1 | 8 | 950 | 176.22 | no | 2 | 12.2 | 20 |
| 2 | 10 | 175.05 | 12.7 | 20 | |||
| 3 | 12 | 177.03 | 12.8 | 20 | |||
| 4 | 8 | 1000 | 175.10 | 12.8 | 20 | ||
| 5 | 10 | 176.00 | 12.8 | 20 | |||
| 6 | 12 | 176.10 | 13.0 | 25 | |||
| 7 | 8 | 1050 | 177.05 | 13.0 | 25 | ||
| 8 | 10 | 176.96 | 12.9 | 20 | |||
| 9 | 12 | 178.00 | 12.8 | 20 | |||
| 10 | 8 | 1100 | 177.35 | 12.9 | 20 | ||
| 11 | 10 | 178.24 | 12.8 | 20 | |||
| 12 | 12 | 175.98 | 13.0 | 25 | |||
| Experiment Number | Amount of Used Sorbent, % | Humidity, % | Firing Temperature, °C | Sample Weight after Firing, g | Ultimate Compressive Strength of Samples, MPa | Frost Resistance, Cycle |
|---|---|---|---|---|---|---|
| 1 | 10 | 8 | 950 | 180.75 | 12.9 | 30 |
| 2 | 1000 | 180.62 | 12.7 | 30 | ||
| 3 | 1050 | 180.74 | 12.7 | 35 | ||
| 4 | 1100 | 180.93 | 12.4 | 35 | ||
| 5 | 10 | 950 | 180.87 | 13.1 | 35 | |
| 6 | 1000 | 180.89 | 12.9 | 35 | ||
| 7 | 1050 | 180.91 | 12.7 | 35 | ||
| 8 | 1100 | 180.98 | 12.7 | 35 | ||
| 9 | 12 | 950 | 181.01 | 13.2 | 35 | |
| 10 | 1000 | 181.25 | 13.0 | 35 | ||
| 11 | 1050 | 181.47 | 12.9 | 35 | ||
| 12 | 1100 | 181.98 | 12.9 | 35 | ||
| 13 | 25 | 8 | 950 | 181.82 | 15.1 | 35 |
| 14 | 1000 | 181.87 | 14.9 | 35 | ||
| 15 | 1050 | 181.91 | 14.8 | 35 | ||
| 16 | 1100 | 181.93 | 14.8 | 35 | ||
| 17 | 10 | 950 | 182.90 | 15.5 | 35 | |
| 18 | 1000 | 182.91 | 15.2 | 35 | ||
| 19 | 1050 | 183.41 | 15.3 | 35 | ||
| 20 | 1100 | 183.78 | 15.1 | 35 | ||
| 21 | 12 | 950 | 184.02 | 15.8 | 35 | |
| 22 | 1000 | 184.23 | 15.4 | 35 | ||
| 23 | 1050 | 184.34 | 15.3 | 35 | ||
| 24 | 1100 | 184.41 | 15.1 | 35 |
| Properties | Compositions | ||
|---|---|---|---|
| Ceramic Bricks with Loam (Reference Sample) | Ceramic Brick with the Addition of 10% Spent Sorbent | Ceramic Brick with the Addition of 25% Spent Sorbent | |
| Sample weight after firing at 950 °C, g | 177 | 181 | 184 |
| Average density, kg/m3 | 1619 | 1630 | 1640 |
| Air shrinkage, % | - | - | - |
| Fire shrinkage, % | 2 | - | - |
| Compressive strength, MPa | 10.8 | 13.2 | 15.8 |
| Brick brand according to samples | M100 | M125 | M125 |
| Sample | The Content of Oxides, wt.% | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | CaO | MgO | Fe2O3 | TiO2 | K2O | Na2O | Cr2O3 | |
| Fired sample with loam | 55.53 | 18.30 | 9.79 | 4.36 | 6.55 | 0.71 | 2.27 | 2.49 | - |
| Fired sample with addition of 25% spent sorbent | 46.07 | 16.90 | 14.54 | 3.73 | 13.03 | 1.01 | 1.83 | 2.63 | 0.26 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daumova, G.; Seraya, N.; Azbanbayev, E.; Assanov, D.; Aubakirova, R.; Reutova, G. Utilization of Spent Sorbent in the Production of Ceramic Bricks. ChemEngineering 2022, 6, 82. https://doi.org/10.3390/chemengineering6050082
Daumova G, Seraya N, Azbanbayev E, Assanov D, Aubakirova R, Reutova G. Utilization of Spent Sorbent in the Production of Ceramic Bricks. ChemEngineering. 2022; 6(5):82. https://doi.org/10.3390/chemengineering6050082
Chicago/Turabian StyleDaumova, Gulzhan, Natalya Seraya, Eldar Azbanbayev, Daulet Assanov, Roza Aubakirova, and Galina Reutova. 2022. "Utilization of Spent Sorbent in the Production of Ceramic Bricks" ChemEngineering 6, no. 5: 82. https://doi.org/10.3390/chemengineering6050082
APA StyleDaumova, G., Seraya, N., Azbanbayev, E., Assanov, D., Aubakirova, R., & Reutova, G. (2022). Utilization of Spent Sorbent in the Production of Ceramic Bricks. ChemEngineering, 6(5), 82. https://doi.org/10.3390/chemengineering6050082











