Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate
Abstract
1. Introduction
2. Materials and Methods
2.1. Adsorbent Materials
2.2. Methods
2.2.1. Lab-Scale Desulfurization
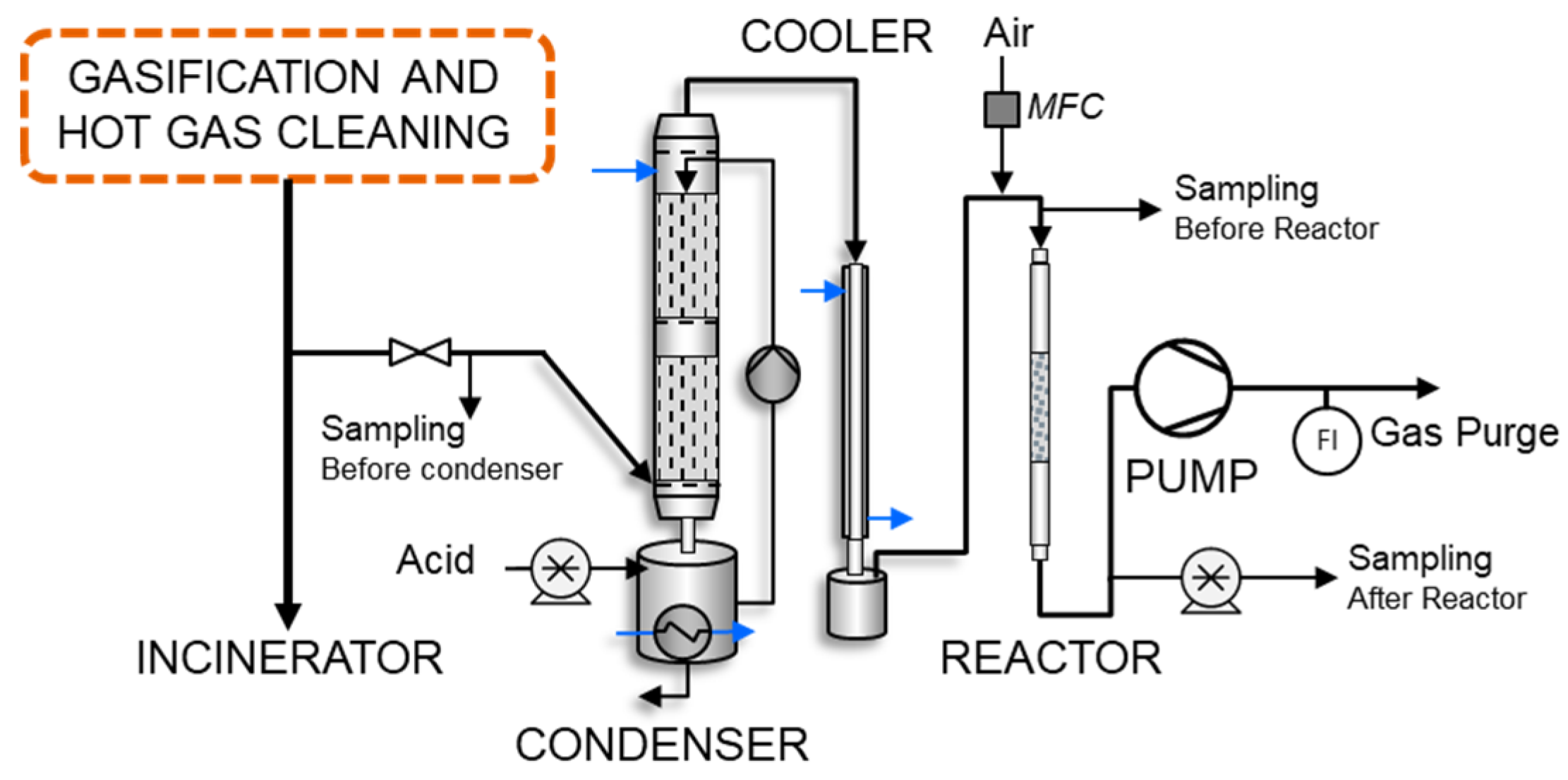
2.2.2. Bench-Scale Desulfurization
2.3. Analytics
2.3.1. Lab-Scale Gas Analytics
2.3.2. Bench-Scale Gas Analytics
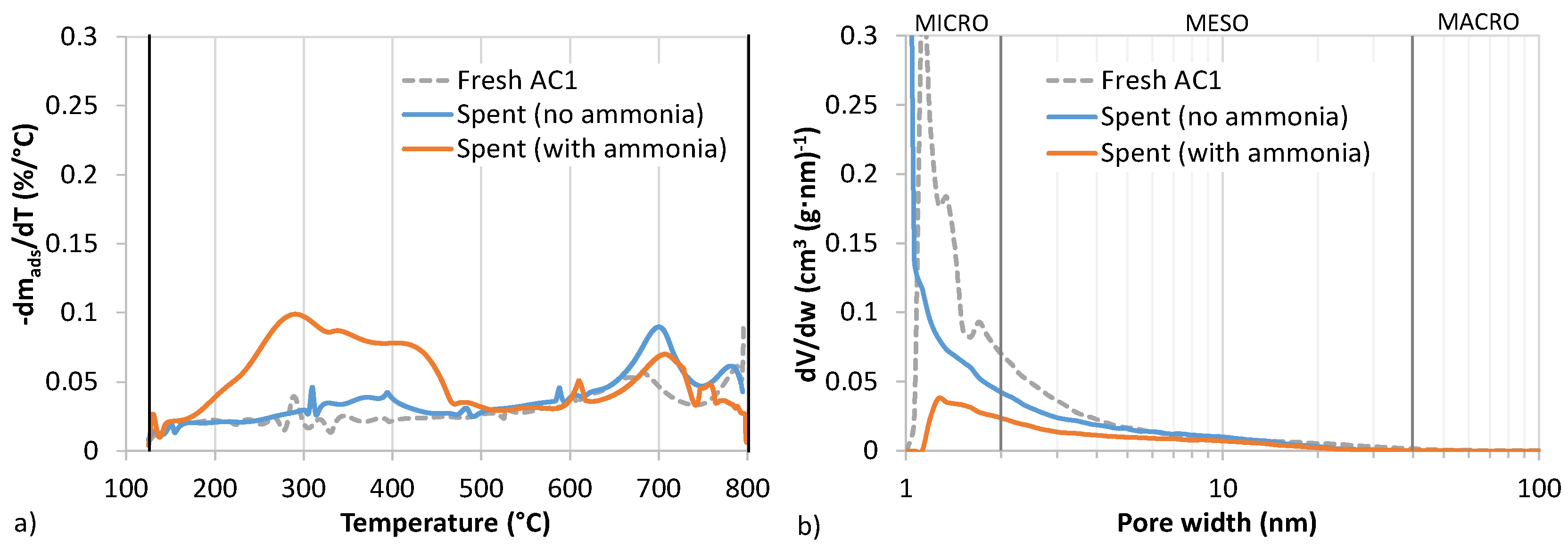
2.3.3. Adsorbent Characterization
Adsorbent Surface pH
N2 Adsorption/Desorption
Thermal Analysis
Elemental Analysis
3. Results
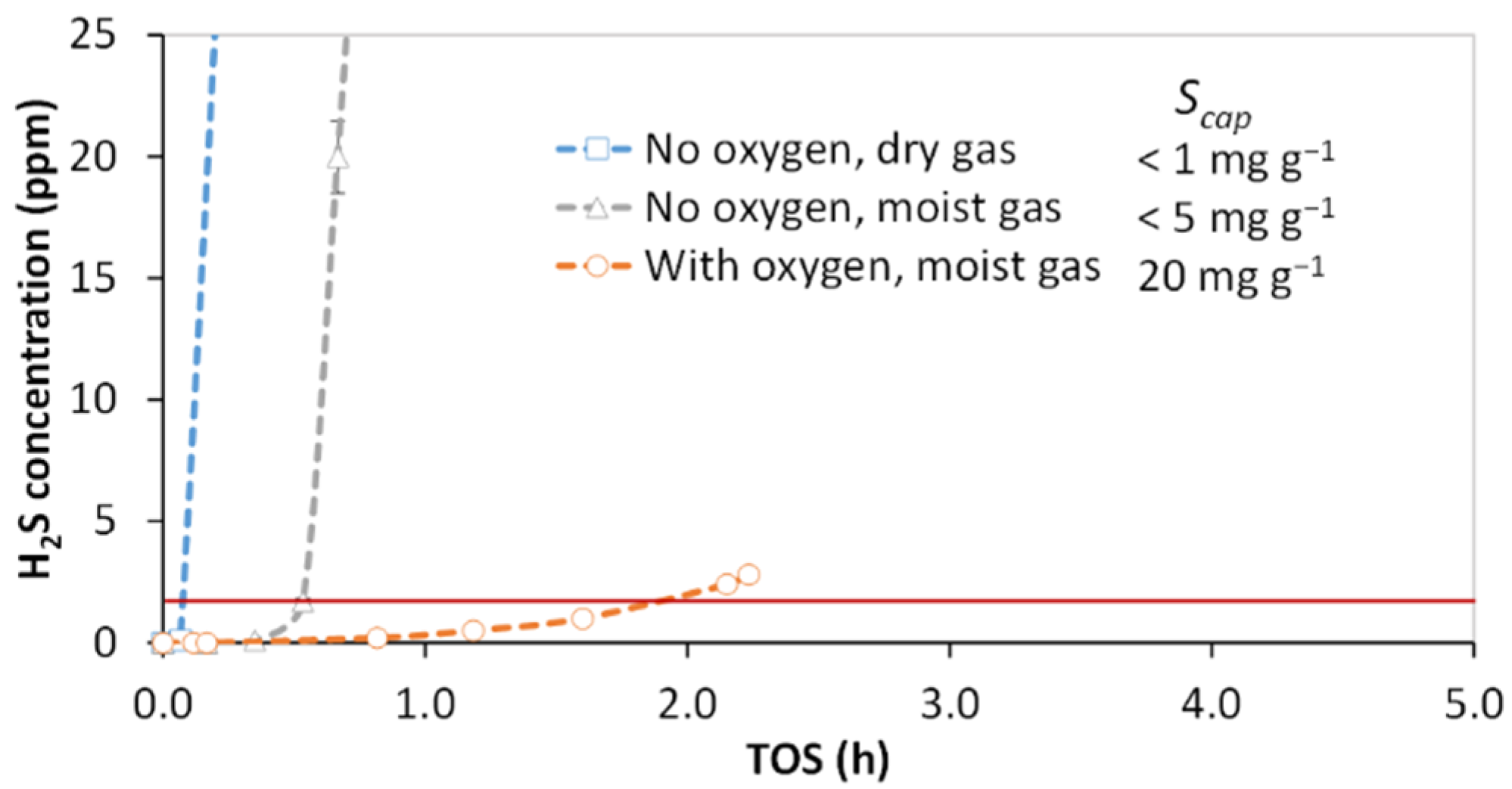
3.1. Effect of Oxygen and Moisture Content
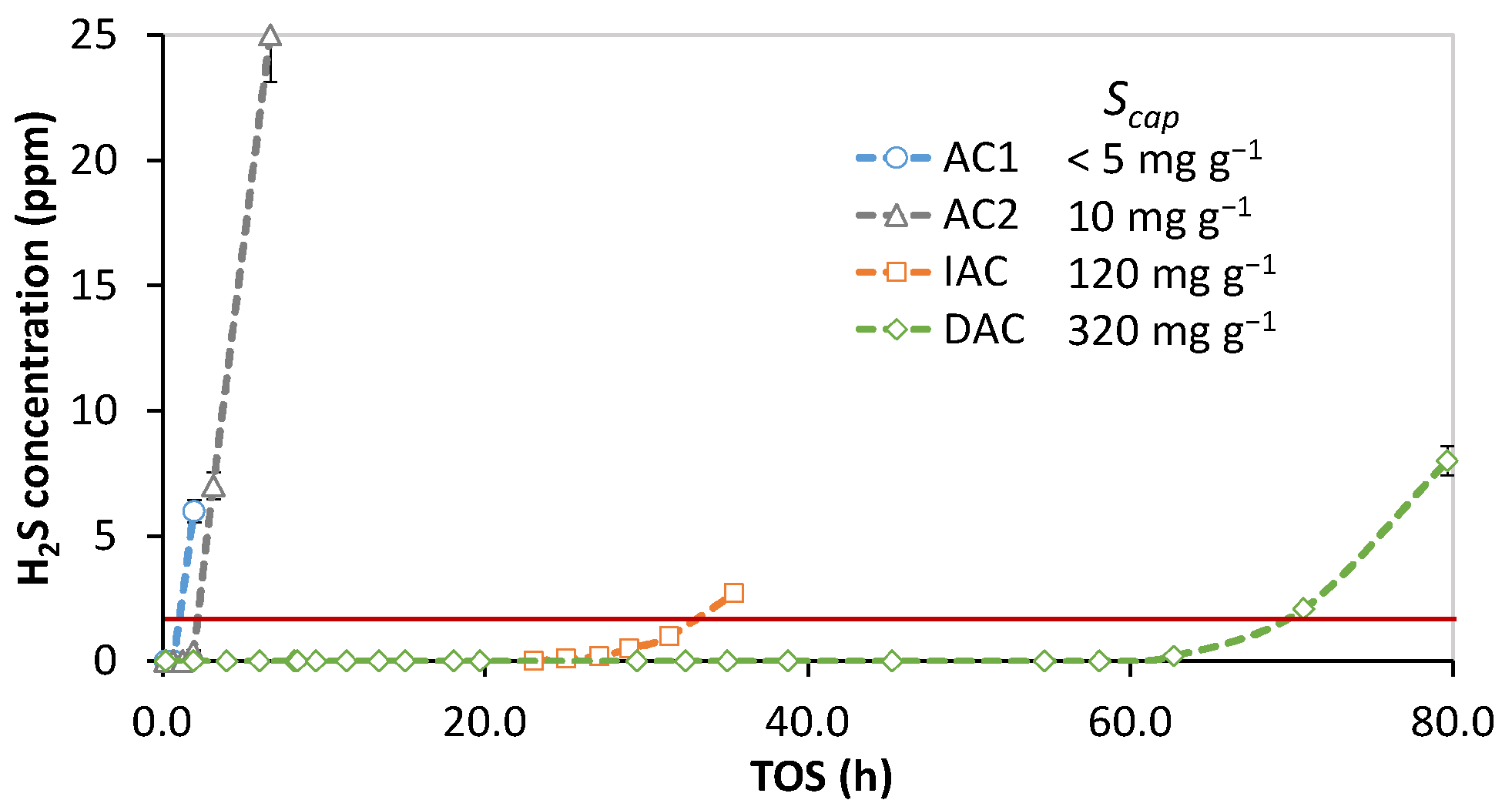
3.2. Adsorbent Relative Desulfurization Performance
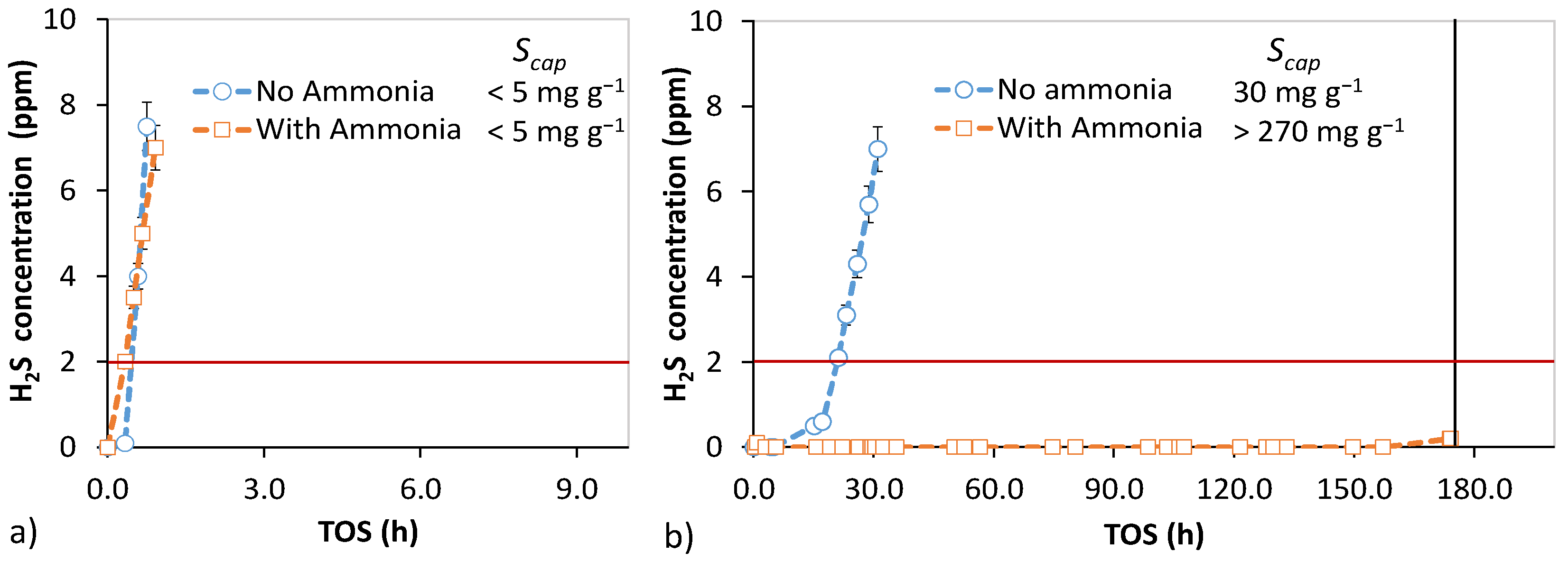
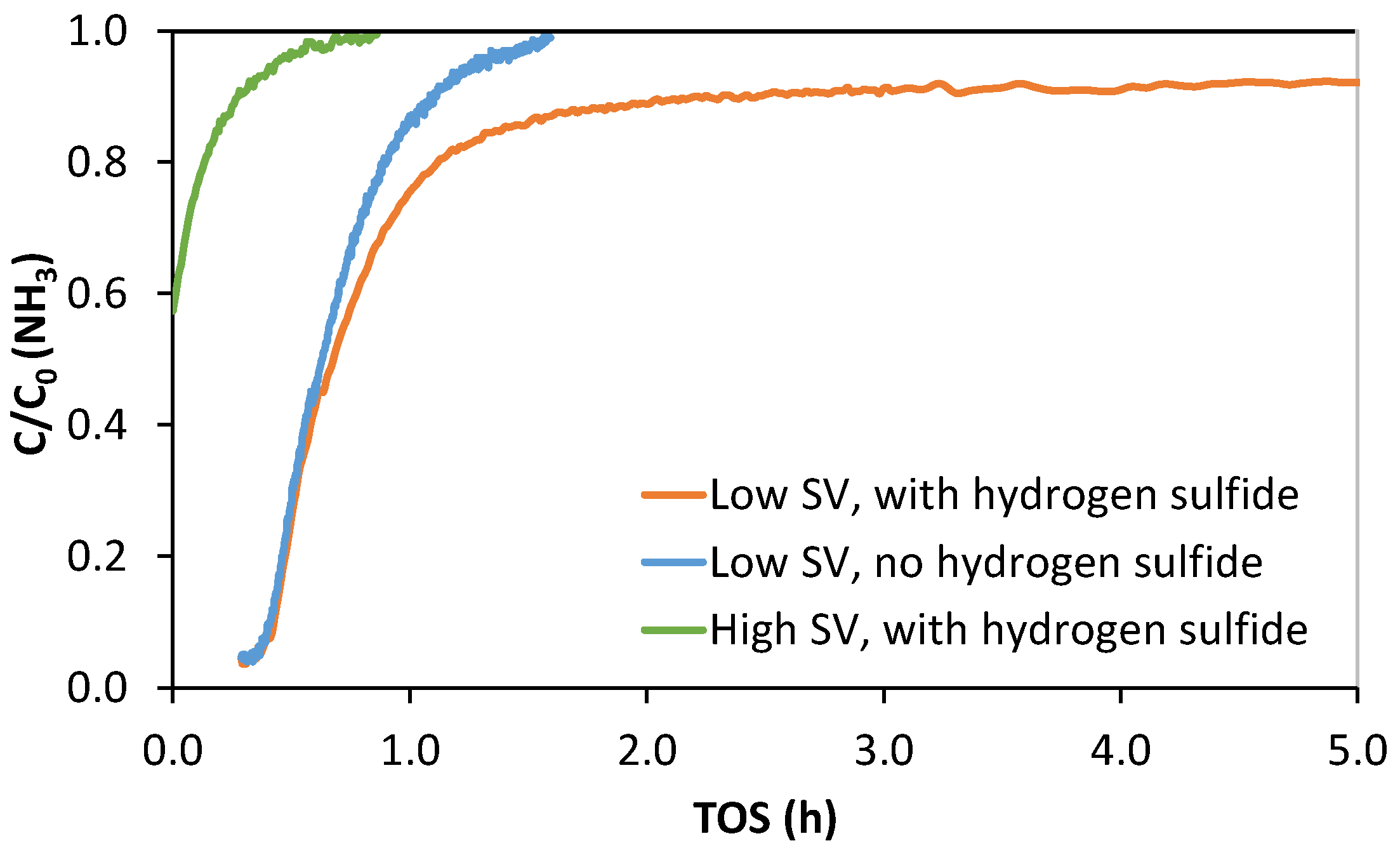
3.3. Ammonia-Enhanced H2S Oxidation
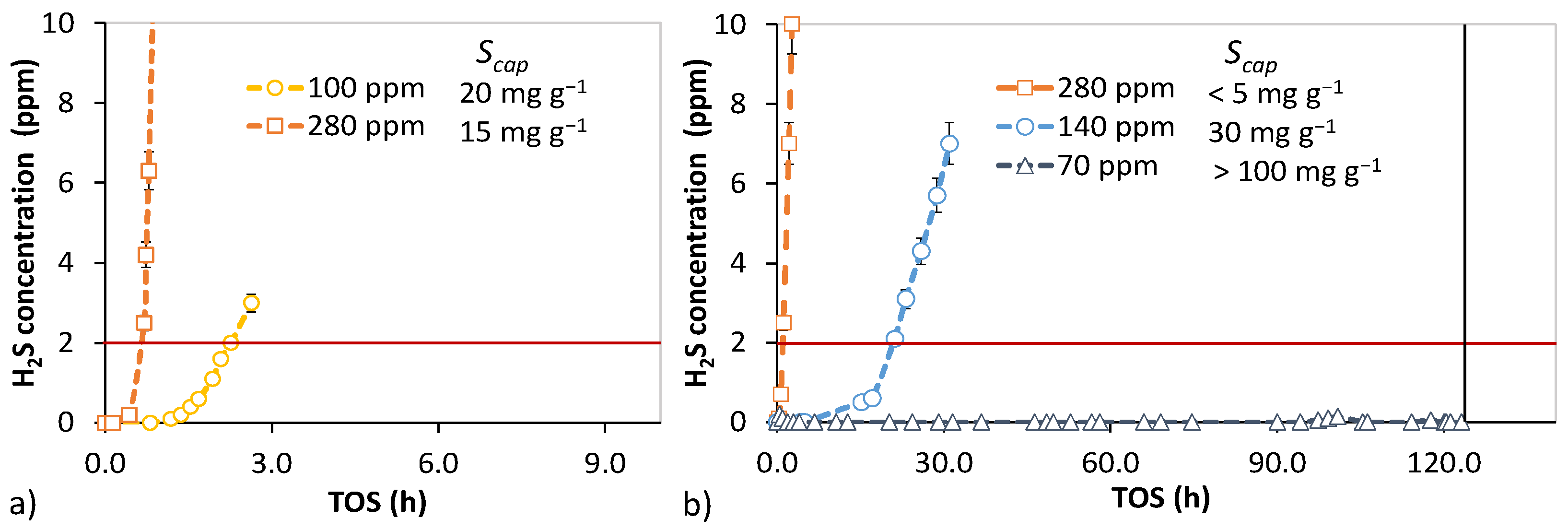
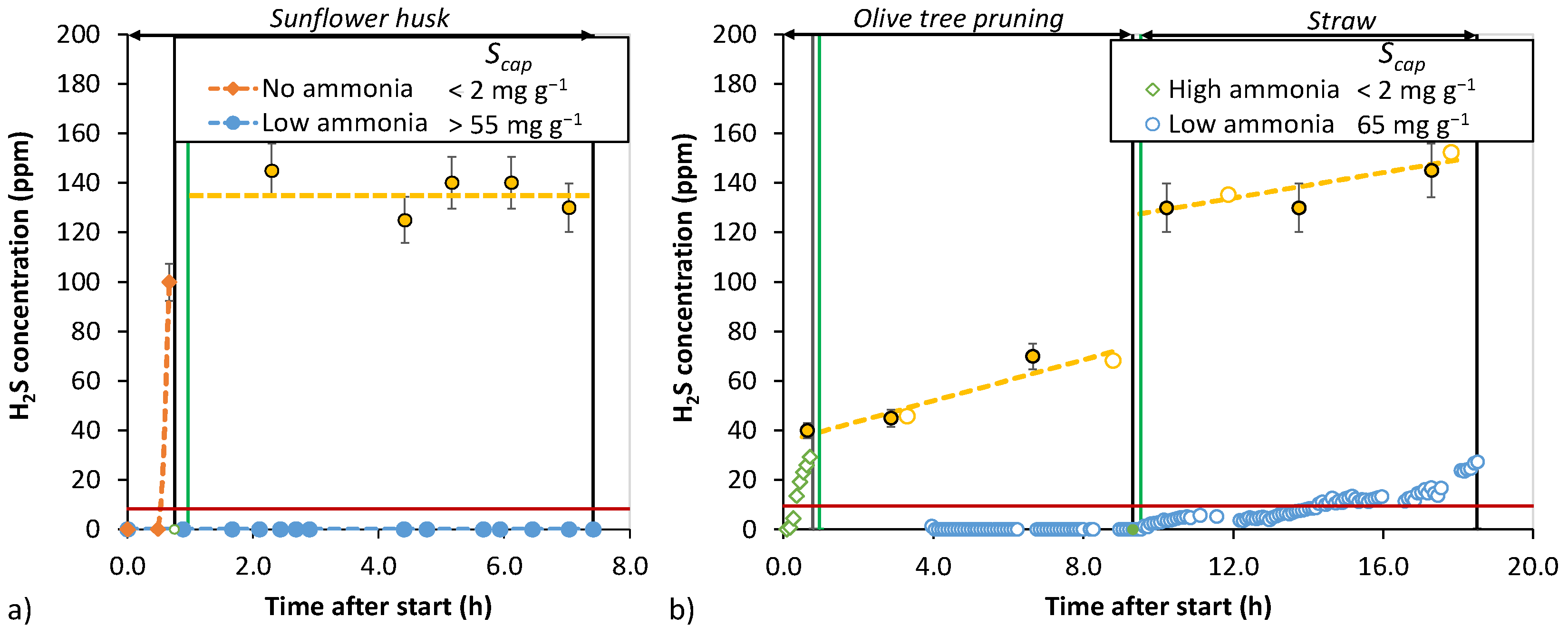
3.4. Bench-Scale Desulfurization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AC | Activated carbon |
| BJH | Barrett–Joyner–Halenda |
| DAC | Doped activated carbon |
| EDS | Energy dispersive X-ray spectroscopy |
| FPD | Flame photometric detector |
| FTIR | Fourier-transform infrared spectroscopy |
| IAC | Impregnated activated carbon |
| n.a. | Not analyzed |
| RH | Relative humidity |
| Rt | Residence time |
| SA | Surface area |
| Scap | Sulfur adsorption capacity |
| SEM | Scanning electron microscope |
| SV | Space velocity (volumetric) |
| TCD | Thermal conductivity detector |
| TOS | Time on stream |
| w | Pore width |
References
- Shah, M.S.; Tsapatsis, M.; Siepmann, J.I. Hydrogen Sulfide Capture: From Absorption in Polar Liquids to Oxide, Zeolite, and Metal-Organic Framework Adsorbents and Membranes. Chem. Rev. 2017, 117, 9755–9803. [Google Scholar] [CrossRef]
- Benedetti, V.; Patuzzi, F.; Baratieri, M. Gasification Char as a Potential Substitute of Activated Carbon in Adsorption Applications. Energy Procedia 2017, 105, 712–717. [Google Scholar] [CrossRef]
- Coppola, G.; Papurello, D. Biogas Cleaning: Activated Carbon Regeneration for H2S Removal. Clean Technol. 2018, 1, 40–57. [Google Scholar] [CrossRef]
- Nakamura, S.; Kitano, S.; Yoshikawa, K. Biomass gasification process with the tar removal technologies utilizing bio-oil scrubber and char bed. Appl. Energy 2016, 170, 186–192. [Google Scholar] [CrossRef]
- Sisani, E.; Cinti, G.; Discepoli, G.; Penchini, D.; Desideri, U.; Marmottini, F. Adsorptive removal of H2S in biogas conditions for high temperature fuel cell systems. Int. J. Hydrogen Energy 2014, 39, 21753–21766. [Google Scholar] [CrossRef]
- Wang, S.; Nam, H.; Nam, H. Preparation of activated carbon from peanut shell with KOH activation and its application for H2S adsorption in confined space. J. Environ. Chem. Eng. 2020, 8, 103683. [Google Scholar] [CrossRef]
- Bagreev, A.; Rahman, H.; Bandosz, T.J. Study of H2S adsorption and water regeneration of spent coconut-based activated carbon. Environ. Sci. Technol. 2000, 34, 4587–4592. [Google Scholar] [CrossRef]
- Le Leuch, L.M.; Subrenat, A.; Le Cloirec, P. Hydrogen sulfide adsorption and oxidation onto activated carbon cloths: Applications to odorous gaseous emission treatments. Langmuir 2003, 19, 10869–10877. [Google Scholar] [CrossRef]
- Habeeb, O.A.; Ramesh, K.; Gomaa, A.M.; Ali, G.A.M.; Yunus, R.M.; Thanusha, T.K.; Olalere, O.A. Modeling and Optimization for H2S Adsorption from Wastewater Using Coconut Shell Based Activated Carbon. Aust. J. Basic Appl. Sci. 2016, 10, 136–147. [Google Scholar]
- Li, Y.; Lin, Y.; Xu, Z.; Wang, B.; Zhu, T. Oxidation mechanisms of H 2 S by oxygen and oxygen-containing functional groups on activated carbon. Fuel Process. Technol. 2019, 189, 110–119. [Google Scholar] [CrossRef]
- Choi, D.-Y.; Lee, J.; Jang, S.; Ahn, B.; Choi, D. Adsorption dynamics of hydrogen sulfide in impregnated activated carbon bed. Adsorption 2008, 14, 533–538. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, S.; Wu, D.; Yuan, Q. Experimental and simulation study of hydrogen sulfide adsorption on impregnated activated carbon under anaerobic conditions. J. Hazard. Mater. 2008, 153, 1193–1200. [Google Scholar] [CrossRef]
- Isik-Gulsac, I. Investigation of impregnated activated carbon properties used in hydrogen sulfide fine removal. Brazilian J. Chem. Eng. 2016, 33, 1021–1030. [Google Scholar] [CrossRef]
- Guo, J.; Luo, Y.; Chong Lua, A.; Chi, R.A.; Chen, Y.; Bao, X.; Xiang, S. Adsorption of hydrogen sulphide (H2S) by activated carbons derived from oil-palm shell. Carbon N. Y. 2007, 45, 330–336. [Google Scholar] [CrossRef]
- Feng, W.; Kwon, S. Adsorption of Hydrogen Sulfide onto Activated Carbon Fibers: Effect of Pore Structure and Surface Chemistry. Environ. Sci. Technol. 2005, 39, 9744–9749. [Google Scholar] [CrossRef] [PubMed]
- Barelli, L.; Bidini, G.; de Arespacochaga, N.; Pérez, L.; Sisani, E. Biogas use in high temperature fuel cells: Enhancement of KOH-KI activated carbon performance toward H2S removal. Int. J. Hydrogen Energy 2017, 42, 10341–10353. [Google Scholar] [CrossRef]
- Georgiadis, A.G.; Charisiou, N.D.; Goula, M.A. Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials. Catalysts 2020, 10, 521. [Google Scholar] [CrossRef]
- Engelhardt, A. Sulphur recovery and gas purification. Conversion of hydrogen sulphide into sulphur by means of activated charcoal. Z. Angew. Chem. 1921, 34, 293–295. [Google Scholar] [CrossRef][Green Version]
- Turk, A.; Sakalis, E.; Lessuck, J.; Karamitsos, H.; Rago, O. Ammonia injection enhances capacity of activated carbon for hydrogen sulfide and methyl mercaptan. Environ. Sci. Technol. 1989, 23, 1242–1245. [Google Scholar] [CrossRef]
- Frilund, C.; Tuomi, S.; Kurkela, E.; Simell, P. Small- to medium-scale deep syngas purification: Biomass-to-liquids multi-contaminant removal demonstration. Biomass Bioenergy 2021, 148, 10. [Google Scholar] [CrossRef]
- Frilund, C.; Simell, P.; Kurkela, E.; Eskelinen, P. Experimental Bench-Scale Study of Residual Biomass Syngas Desulfurization Using ZnO-Based Adsorbents. Energy Fuels 2020, 34, 3326–3335. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Bandosz, T.J. On the adsorption/oxidation of hydrogen sulfide on activated carbons at ambient temperatures. J. Colloid Interface Sci. 2002, 246, 1–20. [Google Scholar] [CrossRef]
- Li, Q.; Lancaster, J.R. Chemical foundations of hydrogen sulfide biology. Nitric Oxide Biol. Chem. 2013, 35, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Tang, Y.; Qu, S.; Da, J.; Hao, Z. H2S-Selective Catalytic Oxidation: Catalysts and Processes. ACS Catal. 2015, 1067, 1053–1067. [Google Scholar] [CrossRef]
- Wu, X.; Schwartz, V.; Overbury, S.H.; Armstrong, T.R. Desulfurization of Gaseous Fuels Using Activated Carbons as Catalysts for the Selective Oxidation of Hydrogen Sulfide. Energy Fuels 2005, 19, 1774–1782. [Google Scholar] [CrossRef]
- Gardner, T.H.; Berry, D.A.; Lyons, K.D.; Beer, S.K.; Freed, A.D. Fuel processor integrated H2S catalytic partial oxidation technology for sulfur removal in fuel cell power plants. Fuel 2002, 81, 2157–2166. [Google Scholar] [CrossRef]
- Steijns, M.; Mars, P. The Role Partial of Sulfur Oxidation Trapped in Micropores of Hydrogen Sulfide in the Catalytic with Oxygen. J. Catal. 1974, 35, 11–17. [Google Scholar] [CrossRef]
- Steijns, M.; Derks, F.; Verloop, A.; Mars, P. The Mechanism of the Catalytic Oxidation of Hydrogen Sulfide II. J. Catal. 1976, 42, 87–95. [Google Scholar] [CrossRef]
- Dalai, A.K.; Tollefson, E.L. Kinetics and reaction mechanism of catalytic oxidation of low concentrations of hydrogen sulfide in natural gas over activated carbon. Can. J. Chem. Eng. 1998, 76, 902–914. [Google Scholar] [CrossRef]
- Bansal, R.C.; Goyal, M. Activated Carbon Adsorption; Taylor & Francis: Boca Raton, FL, USA, 2005; ISBN 9780824753443. [Google Scholar]
- Meeyoo, V.; Trimm, D.L.; Cant, N.W. Adsorption-reaction processes for the removal of hydrogen sulphide from gas streams. J. Chem. Technol. Biotechnol. 1997, 68, 411–416. [Google Scholar] [CrossRef]
- Yan, R.; Liang, D.T.; Tsen, L.; Tay, J.H. Kinetics and mechanisms of H2S adsorption by alkaline activated carbon. Environ. Sci. Technol. 2002, 36, 4460–4466. [Google Scholar] [CrossRef] [PubMed]
- Bashkova, S.; Armstrong, T.R.; Schwartz, V. Selective catalytic oxidation of hydrogen sulfide on activated carbons impregnated with sodium hydroxide. Energy Fuels 2009, 23, 1674–1682. [Google Scholar] [CrossRef]
- Bandosz, T.J.; Bagreev, A.; Adib, F.; Turk, A. Unmodified versus caustics-impregnated carbons for control of hydrogen sulfide emissions from sewage treatment plants. Environ. Sci. Technol. 2000, 34, 1069–1074. [Google Scholar] [CrossRef]
- Rossow, S.; Deerber, G.; Goetze, T.; Kanswohl, N.; Nelles, M. Biogas desulfurization with doped activated carbon. Landtechnik 2009, 3, 202–205. [Google Scholar]
- Sitthikhankaew, R.; Chadwick, D.; Assabumrungrat, S. Effect of KI and KOH Impregnations over Activated Carbon on H2S Adsorption Performance at Low and High Temperatures. Sep. Sci. Technol. 2014, 49, 354–366. [Google Scholar] [CrossRef]
- Sun, F.; Liu, J.; Chen, H.; Zhang, Z.; Qiao, W.; Long, D.; Ling, L. Nitrogen-rich mesoporous carbons: Highly efficient, regenerable metal-free catalysts for low-temperature oxidation of H2S. ACS Catal. 2013, 3, 862–870. [Google Scholar] [CrossRef]
- Nguyen-Thanh, D.; Bandosz, T.J. Activated carbons with metal containing bentonite binders as adsorbents of hydrogen sulfide. Carbon N. Y. 2005, 43, 359–367. [Google Scholar] [CrossRef]
- Bagreev, A.; Menendez, J.A.; Dukhno, I.; Tarasenko, Y.; Bandosz, T.J. Bituminous coal-based activated carbons modified with nitrogen as adsorbents of hydrogen sulfide. Carbon N. Y. 2004, 42, 469–476. [Google Scholar] [CrossRef]
- Adib, F.; Bagreev, A.; Bandosz, T.J. Effect of pH and surface chemistry on the mechanism of H2S removal by activated carbons. J. Colloid Interface Sci. 1999, 216, 360–369. [Google Scholar] [CrossRef]
- Yan, R.; Chin, T. Influence of Surface Properties on the Mechanism of H2S Removal by Alkaline Activated Carbons. Environ. Sci. Technol. 2004, 38, 316–323. [Google Scholar] [CrossRef]
- Adib, F.; Bagreev, A.; Bandosz, T.J. On the possibility of water regeneration of unimpregnated activated carbons used as hydrogen sulfide adsorbents. Ind. Eng. Chem. Res. 2000, 39, 2439–2446. [Google Scholar] [CrossRef]
- Bagreev, A.; Rahman, H.; Bandosz, T.J. Study of regeneration of activated carbons used as H2S adsorbents in water treatment plants. Adv. Environ. Res. 2002, 6, 303–311. [Google Scholar] [CrossRef]
- Bagreev, A.; Rahman, H.; Bandosz, T.J. Thermal regeneration of a spent activated carbon previously used as hydrogen sulfide adsorbent. Carbon N. Y. 2001, 39, 1319–1326. [Google Scholar] [CrossRef]
- Turk, A.; Mahmood, K.; Mozaffari, J. Activated Carbon For Air Purification In New York City’s Sewage Treatment Plants. Wat. Sci.Tech 1993, 27, 121–126. [Google Scholar] [CrossRef]
- Masuda, J.; Fukuyama, J.; Fujii, S. Influence of concurrent substances on removal of hydrogen sulfide by activated carbon. Chemosphere 1999, 39, 1611–1616. [Google Scholar] [CrossRef]
- Vaziri, R.S.; Babler, M.U. Removal of hydrogen sulfide with metal oxides in packed bed reactors-A review from a modeling perspective with practical implications. Appl. Sci. 2019, 9. [Google Scholar] [CrossRef]
- Frilund, C.; Simell, P.; Kaisalo, N.; Kurkela, E.; Koskinen-Soivi, M.-L. Desulfurization of Biomass Syngas Using ZnO-Based Adsorbents: Long-Term Hydrogen Sulfide Breakthrough Experiments. Energy Fuels 2020, 34, 3316–3325. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.C.; de Moraes, D.; da Nóbrega, S.W.; Barboza, M.G. Ammonia adsorption in a fixed bed of activated carbon. Bioresour. Technol. 2007, 98, 886–891. [Google Scholar] [CrossRef]
- Guo, J.; Xu, W.S.; Chen, Y.L.; Lua, A.C. Adsorption of NH3 onto activated carbon prepared from palm shells impregnated with H2SO4. J. Colloid Interface Sci. 2005, 281, 285–290. [Google Scholar] [CrossRef]
| Name | Material Type | Density 1 (m2 g−1) | BET-SA (m2 g−1) | Vmic (cm3 g−1) | Vmes (cm3 g−1) | pH |
|---|---|---|---|---|---|---|
| AC1 | Non-impregnated activated carbon | 0.43 | 520 | 0.13 | 0.28 | 10.8 |
| AC2 | Non-impregnated activated carbon | 0.39 | 1000 | 0.35 | 0.11 | 10.1 |
| IAC | KI impregnated activated carbon | 0.45 | 980 | 0.37 | 0.05 | n.a. |
| DAC | Doped activated carbon | 0.36 | 880 | 0.32 | 0.04 | 10.4 |
| Sunflower Husk | Olive Tree Pruning | Straw | |
|---|---|---|---|
| Feedstock S-content (%) 1 | 0.14 | 0.08 | 0.11 |
| CO (%) | 20.7 | 22.0 | 18.4 |
| H2 (%) | 40.0 | 40.6 | 38.1 |
| CO2 (%) | 24.7 | 18.8 | 21.8 |
| CH4 (%) | 1.7 | 2.1 | 2.4 |
| N2 (%) | 12.8 | 16.5 | 19.3 |
| Benzene and tars (mg m−3) 2 | 70 | 70 | 180 |
| NH3 (ppm) 2 | 260 | 770 | 460 |
| AC | Gas | H2S (ppm) | O2 (%) | RH (%) | T (°C) | SV | Scap–H2S (mg g−1) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Non-impr. | N2 | 200 | 0 | 50 | 30 | ~2100 h−1 | 4 (C/Co = 0.25%) | [12] |
| Na2CO3-impr. | 13 (C/Co = 0.25%) | |||||||
| KI-impr. | He | 3000 | 0 | 0 | 30 | 6000 cm3 (g·h)−1 | 24 (C/Co = 10%) | [37] |
| Non-impr. | 17 (C/Co = 10%) | |||||||
| KOH-KI-impr. | Biogas | 100 | 2 | 90 | 45 | 10,000 h−1 | 40 (C/Co = 50%) | [16] |
| KOH-KI-impr. | 0 | 0 | 24 (C/Co = 50%) | |||||
| KOH-impr. | Biogas | 400 | 2 | 90 | 25 | 5000 h−1 | 130 (C/Co = 0.25%) | [13] |
| KOH-impr. | 15,000 h−1 | 5 (C/Co = 0.25%) | ||||||
| Non-impr. | Biogas | - | 0.3 | 0 | 20 | ~2700 h−1 | 4 (0.5 ppm limit) | [3] |
| Doped | Biogas | 2000 | 0.4 | 50 | 50 | - Rt: 2 s | ~1000 (C/Co = 0.5%) | [36] |
| N-doped | Air | 1000 | 21 | 80 | 25 | 5.5 g (g·h)−1 | 2770 (C/Co = 0.5%) | [38] |
| Non-impr. | Air | 3000 | 21 | 80 | 20 | ~7900 h−1 | 17 (C/Co = 3%) | [39] |
| Cu-impr. | 490 (C/Co = 3%) (Partial conv. to SO2) | |||||||
| Melamine-impr. | Air | 3000 | 21 | 80 | 25 | ~1300 h−1 | 250 (C/Co = 17%) | [40] |
| Non-impr. | Air | 3000 | 21 | 80 | 25 | ~1300 h−1 | 290 (C/Co = 17%) | [41] |
| Surface pH | H2S Adsorption Mechanism | Reaction Mechanism and Final Product |
|---|---|---|
| >7 | H2S chemisorption on OH− sites | HS− on water film and high HS−/O* ratio leads to oxidation of solid S |
| 4.5–7 | Physical adsorption on wetted AC surfaces | Low HS−/O* ratio results in SOx species which acidify and block pores in the form of H2SO4 |
| <4.5 | Physical adsorption on wetted AC surfaces | Strong H2SO4 formation, partial H2S redox with H2SO4 to form solid S |
| Fresh | Spent | ||
|---|---|---|---|
| AC1 | No Ammonia | With Ammonia | |
| Bed Mass Change (%) | 4 | 31 | |
| pH | 10.8 | 9.4 | 8.0 |
| EDS Surface Analysis (%): Sulfur 1 | 1.3 ± 0.3 | 3.7 ± 2.0 | 18.8 ± 5.5 |
| Fresh | Spent | |||
|---|---|---|---|---|
| AC1 | Top | Middle | Bottom | |
| N2 adsorption/desorption analysis 1 | ||||
| BET-SA (m2 g−1) | 520 | 140 | 230 | 330 |
| Vmic (cm3 g−1) | 0.13 | 0 | 0.04 | 0.07 |
| Vmes (cm3 g−1) | 0.28 | 0.21 | 0.17 | 0.23 |
| Ultimate analysis (%) 2 | ||||
| C | 66.2 ±1.6 | 57.8 ± 1.2 | 55.4 ± 1.3 | 62.1 ± 2.3 |
| H | 0.5 ±0.0 | 0.6 ± 0.1 | 0.6 ± 0.0 | 0.6 ± 0.1 |
| N | 0.5 ±0.1 | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.4 ± 0.1 |
| S | 1.0 ±0.1 | 16.0 ± 0.2 | 11.1 ± 0.3 | 7.0 ± 0.5 |
| Adsorbent Properties | AC | AC (NH3 Enhanced) | Impregnated AC | Doped AC |
|---|---|---|---|---|
| Capture capacity | + | ++ | + | ++ |
| Capture rate | – | ++ | + | ++ |
| Removal level | ++ | ++ | ++ | ++ |
| Selectivity | – | – | – | – |
| Regenerability | + | – | – | – |
| Safety and environment | + | + | – | + |
| Cost | ++ | ++ | + | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frilund, C.; Hiltunen, I.; Simell, P. Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate. ChemEngineering 2021, 5, 23. https://doi.org/10.3390/chemengineering5020023
Frilund C, Hiltunen I, Simell P. Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate. ChemEngineering. 2021; 5(2):23. https://doi.org/10.3390/chemengineering5020023
Chicago/Turabian StyleFrilund, Christian, Ilkka Hiltunen, and Pekka Simell. 2021. "Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate" ChemEngineering 5, no. 2: 23. https://doi.org/10.3390/chemengineering5020023
APA StyleFrilund, C., Hiltunen, I., & Simell, P. (2021). Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate. ChemEngineering, 5(2), 23. https://doi.org/10.3390/chemengineering5020023







