Solar Heat for Materials Processing: A Review on Recent Achievements and a Prospect on Future Trends
Abstract
1. Introduction
2. Other Recent Overviews
3. Examples of Innovative Topics of Research
3.1. Process Chambers for the Thermal Dissociation of ZnO
3.2. Solar Heat for Glass Production/Melting
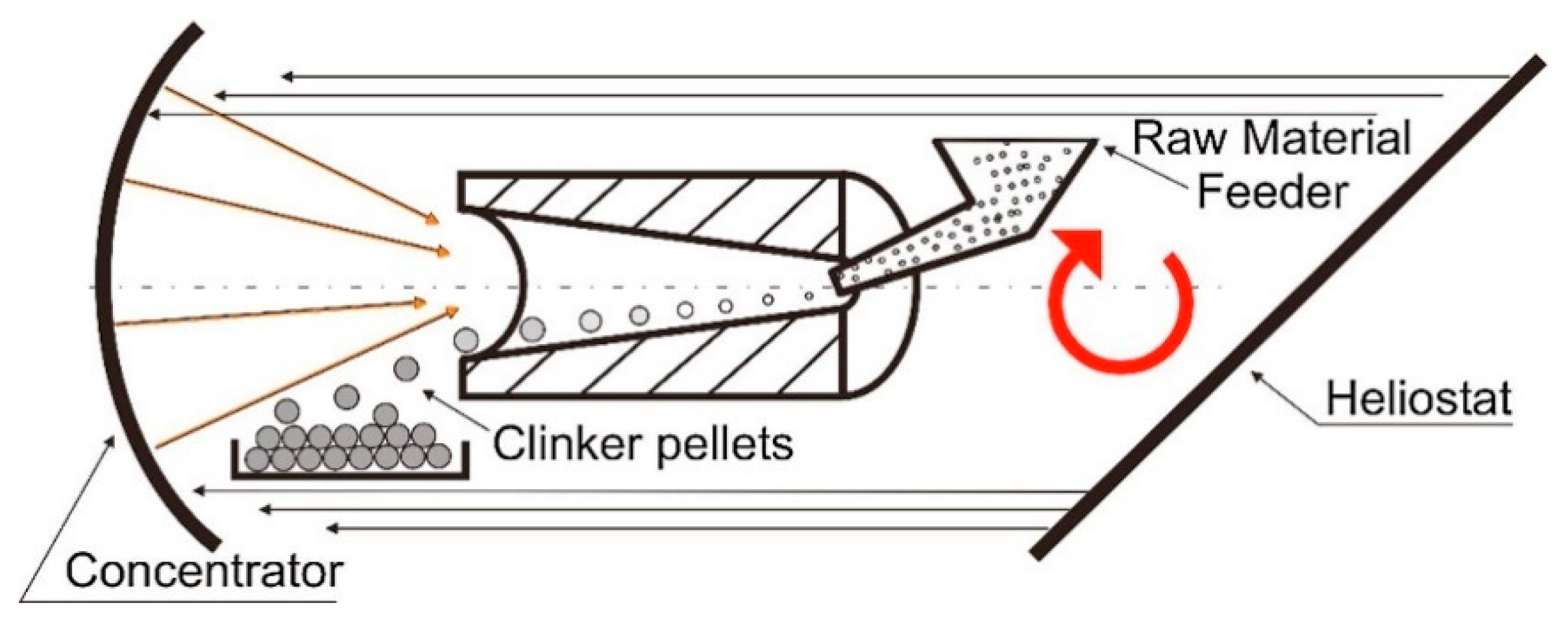
3.3. Solar Heat for Lime and Portland Cement Clinker Production
3.4. Solar Heat for Treatment of Waste Materials
3.5. Opportunities for Solar Heat in the Minerals Processing and Metallurgical Industries
- Successful results were obtained from solar thermal treatment of mercury mine wastes at temperatures higher than 400 °C in order to lower the Hg content [48];
- Thermal decomposition and preheating of manganese ores with solar thermal energy was conceptually proven at temperature measurements up to circa 1000 °C [49];
- The potential for alumina to be calcined with concentrated solar thermal heat has been assessed with a packed bed of boehmite (an aluminium oxyhydroxide) in a crucible positioned at the focal plane of a Fresnel concentrator by Padilla et al. [50]. The solar installation provided a power density of 260 W·cm−2 which allowed reaching temperatures higher than 1000 °C at few minutes of exposure. They reported 75% conversion after 10 min of exposure to solar radiation and the complete dehydration of boehmite, Al(OH)3, and its conversion to alumina, Al2O3, was attained after 90 min of solar radiation exposure under the static conditions described in the work [50]. Based on the fact that modern plants typically process alumina within flash calciners using particles of ~100 μm in diameter transported in a gas suspension through the reactor with residence times on the order of a few seconds, Davis et al. [51] have used a solar vortex transport reactor to processes powders of similar size to a flash calciner. They reported chemical conversion from aluminium hydroxide, Al(OH)3, to aluminium oxide or gibbsite, Al2O3, of up to 95.8% at nominal reactor temperatures over the range 890–1280 °C, and nominal residence times of approximately 3 s [51]. It is therefore proven that it is technically possible to calcine alumina without combustion and its concomitant CO2 emissions, at least during those periods when the solar resource is available. And, it is worth to remember that alumina is an intermediate product in the production of aluminium, but it is also a product in its own right. The industrial gibbsite calcination process shares similarities with the calcination of limestone, which takes place at a slightly lower range of temperature [40,41,42].
- Studies sponsored by CSIRO (Australia’s national science research agency) have identified the potential to use solar in high-temperature processing of ores such as bauxite, copper and iron ore [52]. Solar thermal energy works best at temperatures between 800 °C and 1600 °C [53] which can be achieved with existing technology. It should be noted that traditionally the conversion of heat to electricity generally operates below 600 °C.
- As an example of a high-temperature solar process allowing direct thermal route from the ore to metal, there is a recent publication [54] reporting the feasibility of using concentrated solar energy to the reduction of copper (II) oxide to metallic copper, in hydrogen atmosphere. Using a 1.5 kW thermal power vertical axis parabolic concentrator, the successful experiments were carried out using a stream of gaseous mixture 5/95 v/v H2/N2 for the reduction of CuO in H2.
- Carrying out the solar experiments also in a stream of gas, which in this case was uncracked ammonia NH3 gas (NH3 gas with suppressed extent of dissociation by flowing), other researches [55] have shown that higher nitrides of Mo (δ-MoN) and Fe (ε-Fe2N) can be successfully synthesized. For that they have developed an experimental setup consisting of a linear reaction tube made of silica glass and sample holder made of refractory steel in order to carry out nitriding experiments for powder specimens of Mo and Fe in uncracked NH3 gas at specified linear flow rate under irradiation of concentrated solar beam.
3.6. Integration of Solar Heat in the Regenerative Calcium Cycle
4. Some Important Characteristics of Solar Furnaces
4.1. Main Advantages of Direct Application of Concentrated Solar Radiation
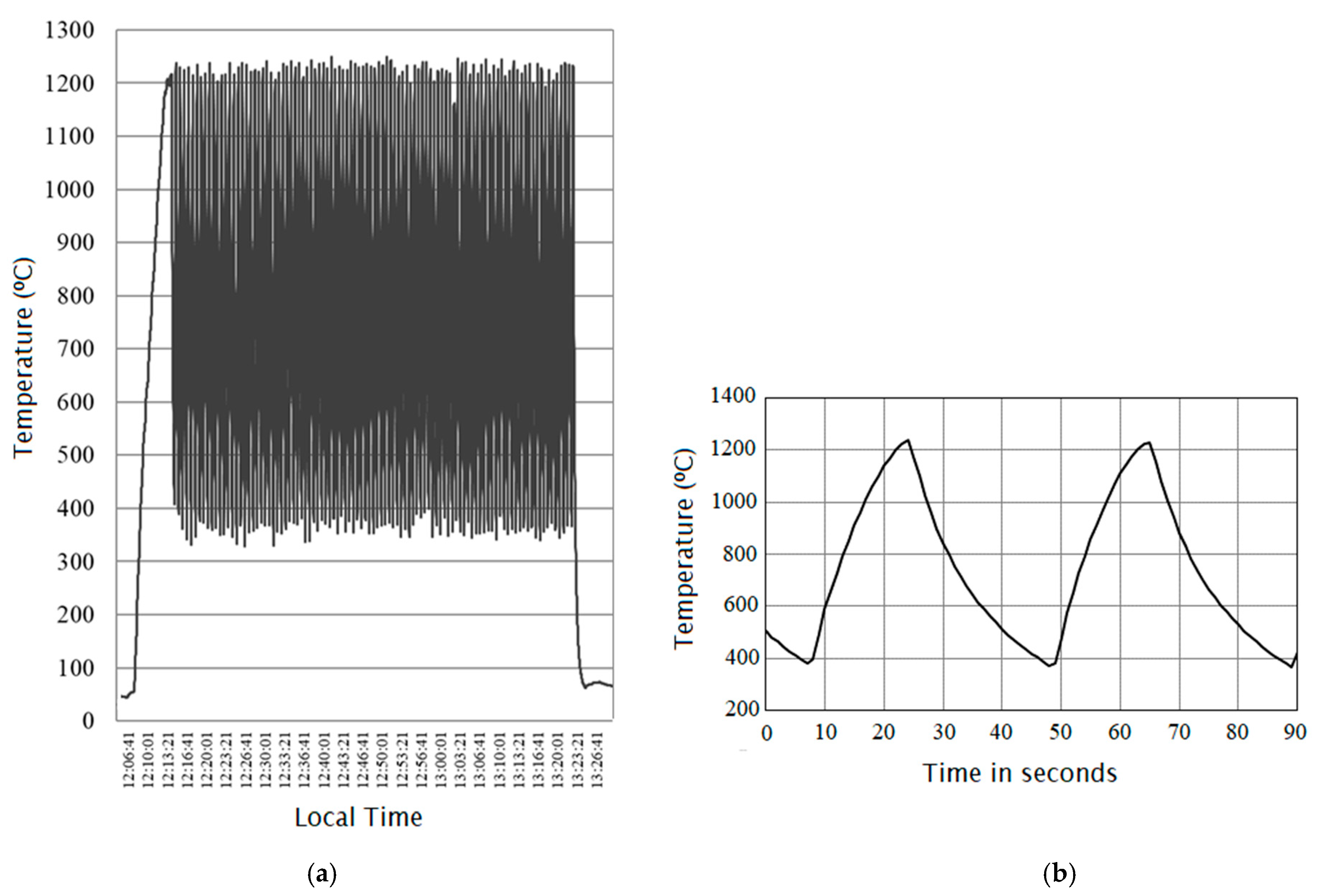
4.1.1. Rapid Heating and Rapid Thermal Cycling
4.1.2. A Natural Wide-Spectrum Radiation
4.2. Main Difficulties in the Use of Concentrated Solar Radiation
- The solar radiation depends on the atmospheric conditions (especially if clouds appear), on the solar time and on the latitude of the site.
- After concentration, the solar radiation is essentially unidirectional. Then, the targeted objects are usually irradiated/heated in a single direction, which is not the case for most of industrial furnaces, typically dealing with temperatures higher than 400 °C, like gas furnaces, electric furnaces, micro-wave furnaces, or even optical furnaces that use a radiant energy different from the solar radiation. Note that artificial radiation sources may consist of incandescent lamps, graphite heaters, arc lamps, super high-pressure xenon gas-discharge tubes, and plasma radiators.
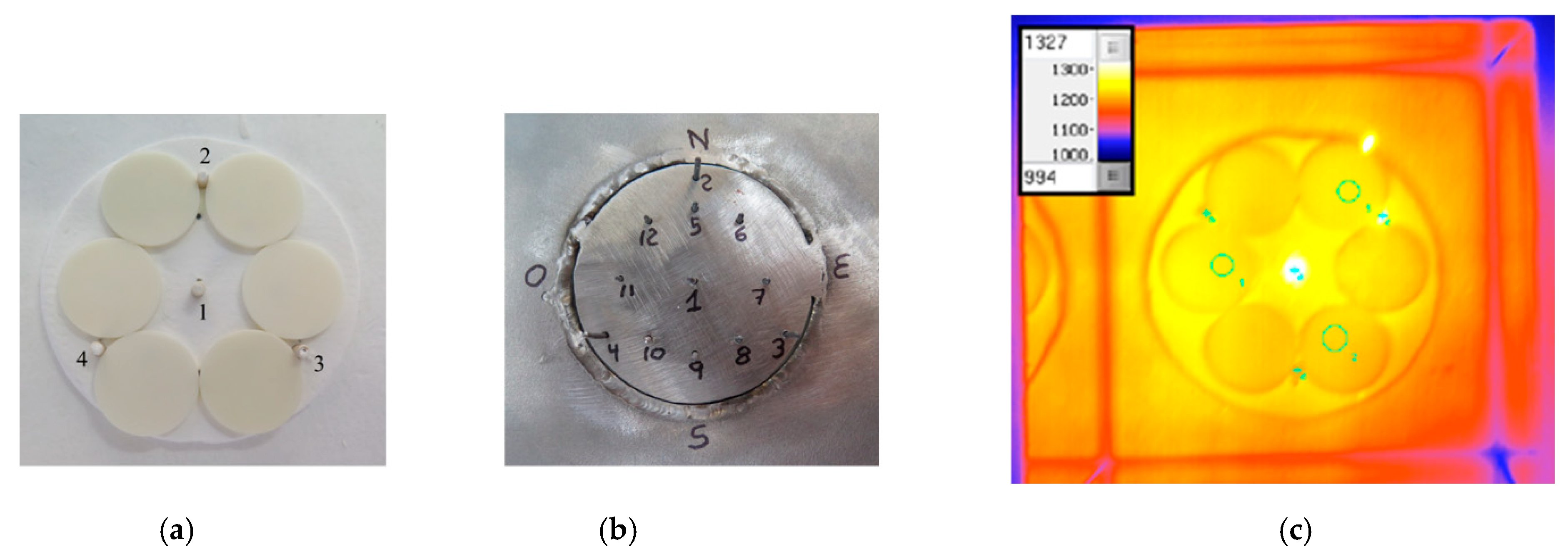
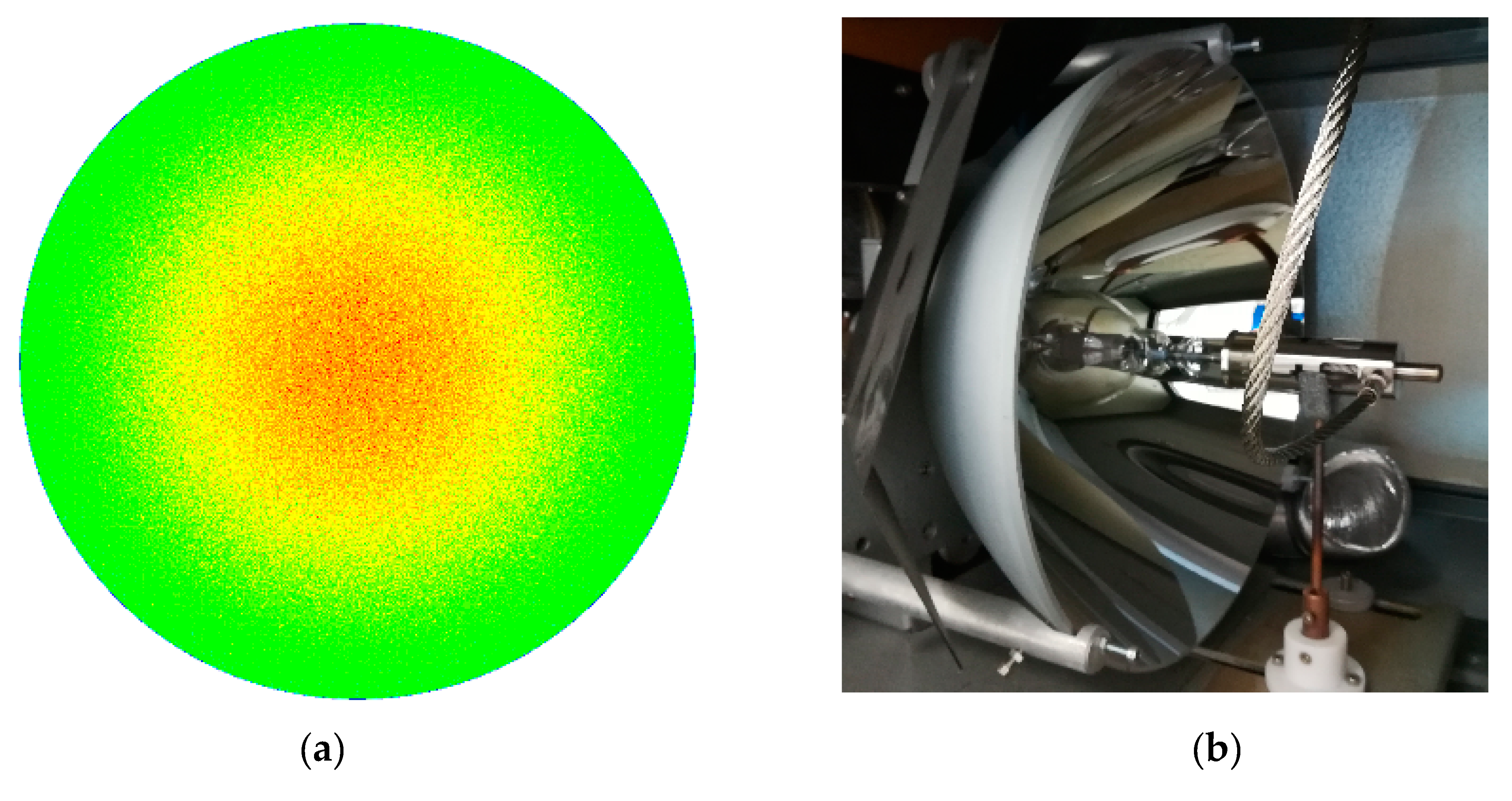
- Additionally, the flux of solar radiation that reaches the target is theoretically non-homogeneous. In fact, a circle illuminated with a higher concentration in the middle (see Figure 5) is theoretically obtained when the paraboloid reflection model [1] is applied to the traditional solar concentrators utilizing a point focusing solar concentrating panel assembly.
5. On-Going Activities and Prospects on Future Trends for Solar Processing of Materials
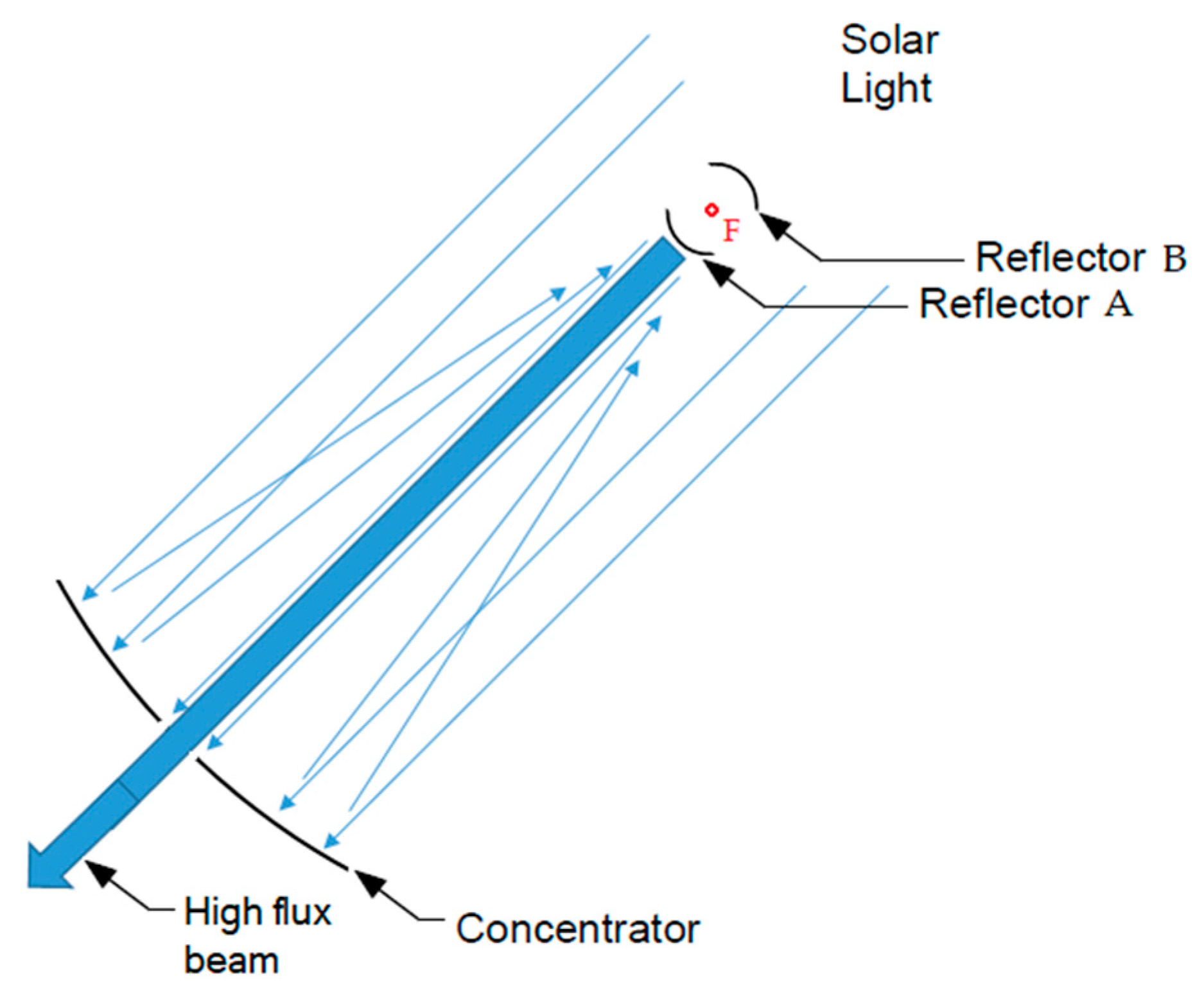
5.1. Novel Approach Based on Heating by Indirect Irradiation
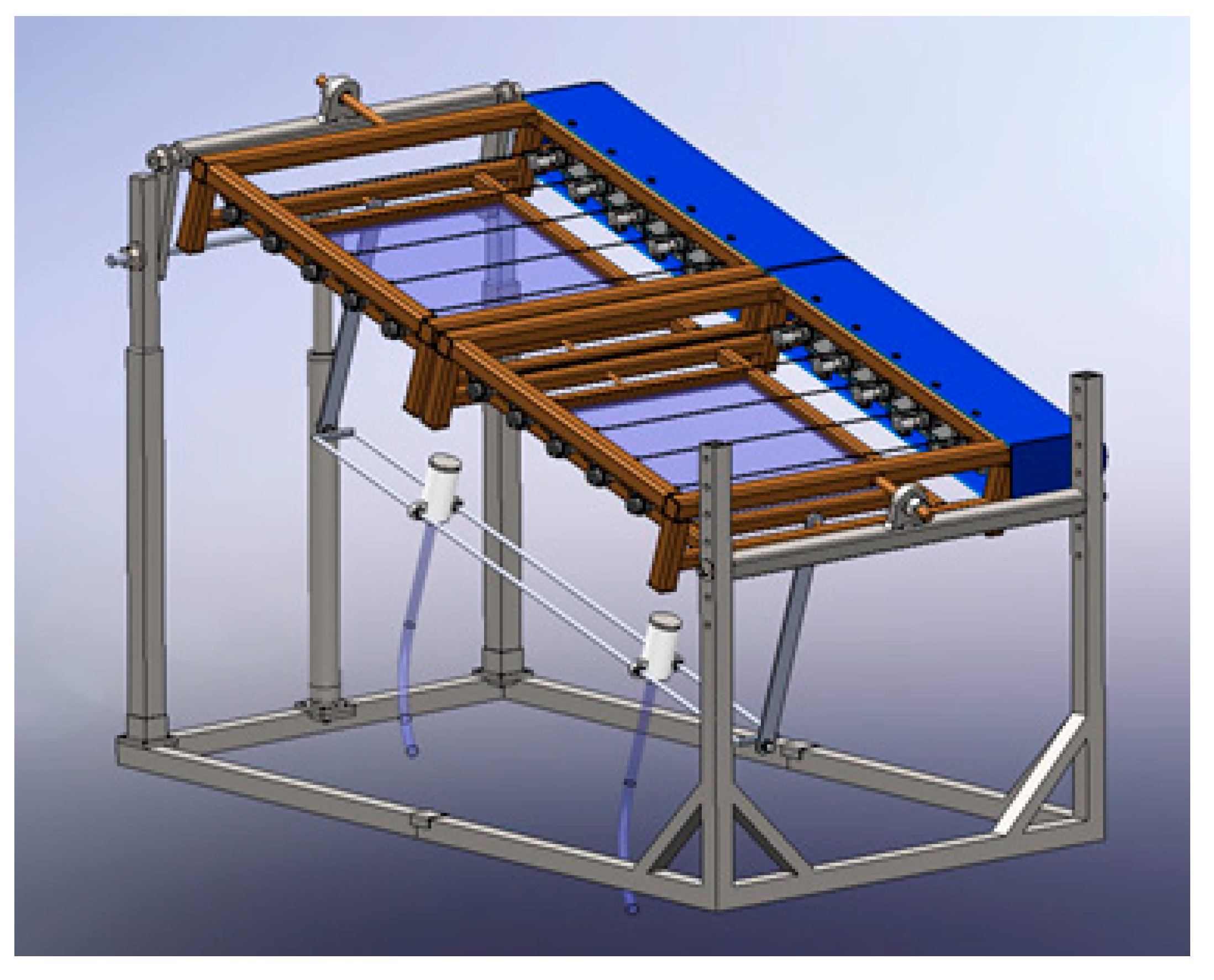
5.2. Modular Systems for Capture, Concentration, Control and Conduction of Solar Radiation
6. Conclusions
Funding
Conflicts of Interest
References
- Pereira, J.C.G.; Fernandes, J.C.; Rosa, L.G. Mathematical Models for Simulation and Optimization of High-Flux Solar Furnaces. Math. Comput. Appl. 2019, 24, 65. [Google Scholar] [CrossRef]
- Monreal, A.; Riveros-Rosas, D.; Sanchez, M. Analysis of the influence of the site in the final energy cost of solar furnaces for its use in industrial applications. Sol. Energy 2015, 118, 286–294. [Google Scholar] [CrossRef]
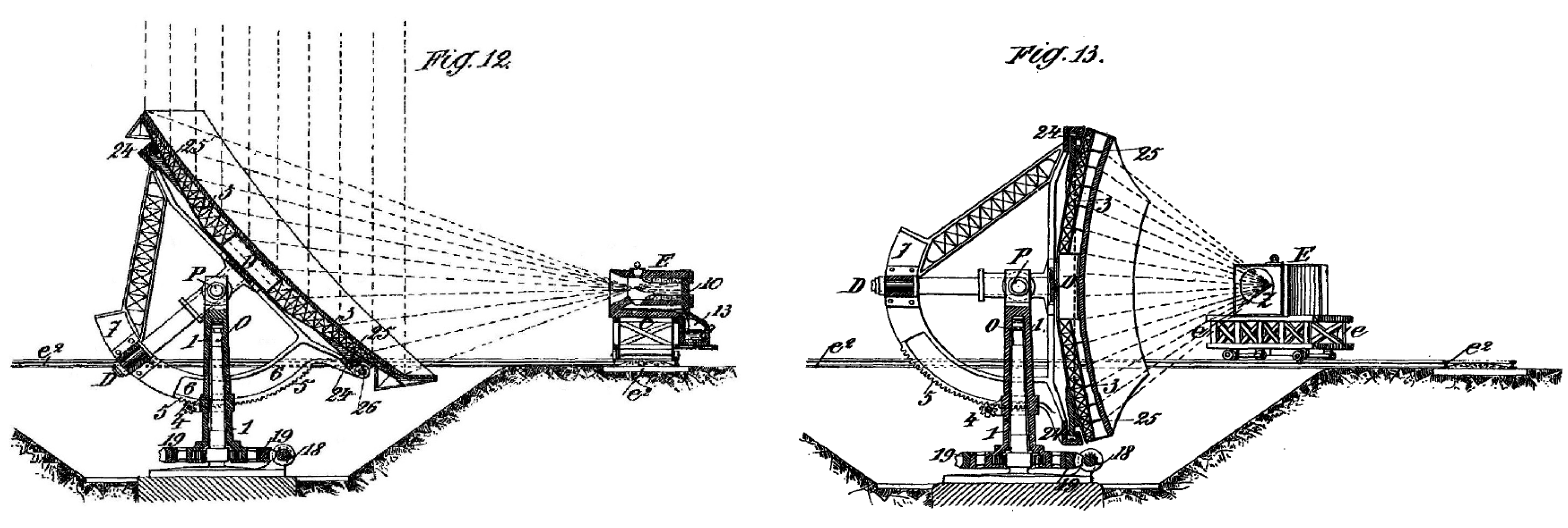
- Himalaya, M.A.G. Improved Apparatus for Making Industrial Use of the Heat of the Sun and Obtaining High Temperatures. Patent GB190116181, 26 October 1901. Available online: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=19011026&DB=EPODOC&locale=en_EP&CC=GB&NR=190116181A&KC=A&ND=2 (accessed on 7 July 2019).
- Himalaya, M.A.G. Solar Apparatus for Producing High Temperatures. U.S. Patent 797,891, 22 August 1905. Available online: https://patents.google.com/patent/US797891 (accessed on 7 July 2019).
- Rodrigues, J. A Conspiração Solar do Padre Himalaya – Esboço Biográfico Dum Português pioneiro da Ecologia (The Solar Conspiration of Father Himalaya—Biographic Outline of a Portuguese Pioneer in Ecology); Árvore—Cooperativa de Actividades Artísticas: Porto, Portugal, 1999; ISBN 972-9089-44-2. [Google Scholar]
- Tinoco, A. Portugal na Exposição Universal de 1904: O Padre Himalaia e o Pirelióforo. Cadernos Sociomuseologia 2012, 42, 113–127. [Google Scholar]
- Trombe, F.; Le Phat Vinh, A. Thousand kW solar furnace, built by the National Center of Scientific Research, in Odeillo (France). Sol. Energy 1973, 15, 57–61. [Google Scholar] [CrossRef]
- Răboacă, M.S.; Badea, G.; Enache, A.; Filote, C.; Răsoi, G.; Rata, M.; Lavric, A.; Felseghi, R. Concentrating Solar Power Technologies. Energies 2019, 12, 1048. [Google Scholar] [CrossRef]
- Yadav, D.; Banerjee, R. A review of solar thermochemical processes. Renew. Sustain. Energ. Rev. 2016, 54, 497–532. [Google Scholar] [CrossRef]
- Bushra, N.; Hartmann, T. A review of state-of-the-art reflective two-stage solar concentrators: Technology categorization and research trends. Renew. Sustain. Energ. Rev. 2019, 114, 109307. [Google Scholar] [CrossRef]
- Levêque, G.; Bader, R.; Lipinski, W.; Haussener, S. High-flux optical systems for solar thermochemistry. Sol. Energy 2017, 156, 133–148. [Google Scholar] [CrossRef]
- Petrasch, J.; Coray, P.; Meier, A.; Brack, M.; Haberling, P.; Wuillemin, D.; Steinfeld, A. A novel 50kW 11,000 suns high-flux solar simulator based on an array of Xenon arc lamps. J. Sol. Energy Eng. 2007, 129, 405–411. [Google Scholar] [CrossRef]
- Alxneit, I.; Schmit, H. Spectral characterization of PSI’s high-flux solar simulator. J. Sol. Energy Eng. 2012, 134. [Google Scholar] [CrossRef]
- Fernández-González, D.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Mochón-Csataños, J.; Sancho-Gorostiaga, J.; Verdeja, L.F. Concentrated solar energy applications in materials science and metallurgy. Sol. Energy 2018, 170, 520–540. [Google Scholar] [CrossRef]
- Alonso, E.; Gallo, A.; Roldan, M.I.; Perez-Rabago, C.A.; Fuentealba, E. Use of rotary kilns for solar thermal applications: Review of developed studies and analysis of their potential. Sol. Energy 2017, 144, 90–104. [Google Scholar] [CrossRef]
- Ho, C.K. A review of high-temperature particle receivers for concentrating solar power. Appl. Therm. Eng. 2016, 109, 958–969. [Google Scholar] [CrossRef]
- Rosa, L.G.; Rosa, N.F. Aplicação direta da radiação solar concentrada em fornos e câmaras de processo: Desenvolvimentos recentes e tendências futuras. In Proceedings of the XVI Congreso Ibérico y XII Congreso Iberoamericano de Energía Solar, Madrid, Spain, 20–24 June 2018; Available online: https://www.researchgate.net/publication/325881511_Aplicacao_direta_da_radiacao_solar_concentrada_em_fornos_e_camaras_de_processo_Desenvolvimentos_recentes_e_tendencias_futuras (accessed on 20 September 2019).
- Koepf, E.; Alxneit, I.; Wieckert, C.; Meier, A. A review of high temperature solar driven reactor technology: 25 years of experience in research and development at the Paul Scherrer Institute. Appl. Energy 2017, 188, 620–651. [Google Scholar] [CrossRef]
- Villasmil, W.; Cooper, T.; Koepf, E.; Meier, A.; Steinfeld, A. Coupled concentrating optics, heat transfer, and thermochemical modeling of a 100-kW(th) high-temperature solar reactor for the thermal dissociation of ZnO. J. Sol. Energy Eng. 2017, 139, 021015. [Google Scholar] [CrossRef]
- Palumbo, R.; Lede, J.; Boutin, O.; Ricart, E.E.; Steinfeld, A.; Möller, S.; Weidenkaff, A.; Fletcher, E.; Bielicki, J. The production of Zn from ZnO in a high temperature solar decomposition quench process ‒ I. The scientific framework for the process. Chem. Eng. Sci. 1998, 53, 2503–2517. [Google Scholar] [CrossRef]
- Haueter, P.; Möller, S.; Palumbo, R.; Steinfeld, A. The production of zinc by thermal dissociation of zinc oxide—Solar chemical reactor design. Sol. Energy 1999, 67, 161–167. [Google Scholar] [CrossRef]
- Ho, D.; Sobon, E.L. Extraterrestrial fiberglass production using solar energy. In Space Resources and Space Settlements (NASA SP-428); Billingham, J., Gilbreath, W., Eds.; National Aeronautics Space Administratio: Washington, DC, USA, 1979; pp. 225–232. Available online: https://space.nss.org/settlement/nasa/spaceres/V-2.html (accessed on 20 September 2019).
- Ahrnad, S.Q.S.; Hand, R.J.; Wieckert, C. Use of concentrated radiation for solar powered glass melting experiments. Sol. Energy 2014, 109, 174–182. [Google Scholar] [CrossRef]
- Romero, M.; Robla, J.I.; Padilla, I.; García-Hierro, J.; López-Delgado, A. Eco-efficient melting of glass frits by concentrated solar energy. Sol. Energy 2018, 174, 321–327. [Google Scholar] [CrossRef]
- Yaphary, Y.L.; Lam, R.H.W.; Lau, D. Chemical technologies for modern concrete production. Procedia Eng. 2017, 172, 1270–1277. [Google Scholar] [CrossRef]
- U.S. Geological Survey. Cement. In Mineral commodity summaries 2019; U.S. Geological Survey: Reston, VA, USA, 2019. [Google Scholar] [CrossRef]
- Rehan, R.; Nehdi, M. Carbon dioxide emissions and climate change: Policy implications for the cement industry. Environ. Sci. Policy 2005, 8, 105–114. [Google Scholar] [CrossRef]
- Worrell, E.; Price, L.; Martin, N.; Hendriks, C.; Meida, L.O. Carbon dioxide emissions from the global cement industry. Annu. Rev. Energy Environ. 2001, 26, 303–329. [Google Scholar] [CrossRef]
- Chen, C.; Habert, G.; Bouzidi, Y.; Jullien, A. Environmental impact of cement production: Detail of the different processes and cement plant variability evaluation. J. Clean. Prod. 2010, 18, 478–485. [Google Scholar] [CrossRef]
- Mikulčić, H.; Klimeš, J.J.; Vujanović, M.; Urbaniec, K.; Duić, N. Reducing greenhouse gasses emissions by fostering the deployment of alternative raw materials and energy sources in the cleaner cement manufacturing process. J. Clean. Prod. 2016, 136, 119–132. [Google Scholar] [CrossRef]
- Andrew, R.M. Global CO2 emissions from cement production. Earth Syst. Sci. Data 2018, 10, 195–217. [Google Scholar] [CrossRef]
- Meier, A.; Gremaud, N.; Steinfeld, A. Economic evaluation of the industrial solar production of lime. Energy Convers. Manag. 2005, 46, 905–926. [Google Scholar] [CrossRef]
- Hasanbeigi, A.; Price, L.; Lin, E. Emerging energy-efficiency and CO2 emission-reduction technologies for cement and concrete production: A technical review. Renew. Sustain. Energy Rev. 2012, 16, 6220–6238. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, S.; Shao, S.; Chen, Y.; Liu, S.; Zhang, S. Aspen Plus-based simulation of a cement calciner and optimization analysis of air pollutants emission. Clean. Technol. Environ. Policy 2011, 13, 459–468. [Google Scholar] [CrossRef]
- Rahman, A.; Rasul, M.G.; Khan, M.M.K.; Sharma, S. Recent development on the uses of alternative fuels in cement manufacturing process. Fuel 2015, 145, 84–99. [Google Scholar] [CrossRef]
- Telesca, A.; Marroccoli, M.; Ibris, N.; Lupiáñez, C.; Díez, L.I.; Romeo, L.M.; Montagnaro, F. Use of oxyfuel combustion ash for the production of blended cements: A synergetic solution toward reduction of CO2 emissions. Fuel Process. Technol. 2017, 156, 211–220. [Google Scholar] [CrossRef]
- Barcelo, L.; Kline, J.; Walenta, G.; Gartner, E. Cement and carbon emissions. Mater. Struct. 2014, 47, 1055–1065. [Google Scholar] [CrossRef]
- Koutník, P. Preparation of β-belite using liquid alkali silicates. Mater. Construcc. 2017, 67. [Google Scholar] [CrossRef]
- Miller, S.A.; Horvath, A.; Monteiro, P.J.M. Readily implementable techniques can cut annual CO2 emissions from the production of concrete by over 20%. Environ. Res. Lett. 2016, 11. [Google Scholar] [CrossRef]
- Meier, A.; Bonaldi, E.; Cella, G.M.; Lipinski, W.; Wuillemin, D. Solar chemical reactor technology for industrial production of lime. Sol. Energy 2006, 80, 1355–1362. [Google Scholar] [CrossRef]
- Abanades, S.; André, L. Design and demonstration of a high temperature solar-heated rotary tube reactor for continuous particles calcination. Appl. Energy 2018, 212, 1310–1320. [Google Scholar] [CrossRef]
- Tregambi, C.; Solimene, R.; Montagnaro, F.; Salatino, P.; Marroccoli, M.; Ibris, N.; Telesca, A. Solar-driven production of lime for ordinary Portland cement formulation. Sol. Energy 2018, 173, 759–768. [Google Scholar] [CrossRef]
- Oliveira, F.A.C.; Fernandes, J.C.; Galindo, J.; Rodríguez, J.; Cañadas, I.; Vermelhudo, V.; Nunes, A.; Rosa, L.G. Portland cement clinker production using concentrated solar energy—A proof-of-concept approach. Sol. Energy 2019, 183, 677–688. [Google Scholar] [CrossRef]
- Ugwuishiwu, B.O.; Owoh, I.P.; Udom, I.J. Solar energy application in waste treatment—A review. Nigerian J. Technol. 2016, 35, 432–440. [Google Scholar] [CrossRef]
- Mueller, F.; Patel, H.; Blumenthal, D.; Pozivil, P.; Das, P.; Wieckert, C.; Maiti, P.; Maiti, S.; Steinfeld, A. Co-production of syngas and potassium-based fertilizer by solar-driven thermochemical conversion of crop residues. Fuel Process. Technol. 2018, 171, 89–99. [Google Scholar] [CrossRef]
- Piatkowski, N.; Wieckert, C.; Weimer, A.W.; Steinfeld, A. Solar-driven gasification of carbonaceous feedstock—A review. Energy Environ. Sci. 2011, 4, 73–82. [Google Scholar] [CrossRef]
- Moustakas, K.; Loizidou, M. Solid waste management through the application of thermal methods. In Waste Management; Kumar, E.S., Ed.; IntechOpen: Rijeka, Croatia, 2010; ISBN 978-953-7619-84-8. Available online: http://www.intechopen.com/books/waste-management/solid-waste-management-through-the-application-of-thermal-methods (accessed on 20 September 2019). [CrossRef]
- Navarro, A.; Cañadas, I.; Rodríguez, J. Thermal treatment of mercury mine wastes using a rotary solar kiln. Minerals 2014, 4, 37–51. [Google Scholar] [CrossRef]
- Hockaday, S.A.C.; Reynolds, Q.G.; Dinter, F.; Harms, F. Solar thermal treatment of manganese ores. AIP Conf. Proc. 2018, 2033. [Google Scholar] [CrossRef]
- Padilla, I.; López-Delgado, A.; López-Andrés, S.; Álvarez, M.; Galindo, R.; Vázquez-Vaamonde, A.J. The application of thermal solar energy to high temperature processes: Case study of the synthesis of alumina from boehmite. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.; Müller, F.; Saw, W.L.; Steinfeld, A.; Nathan, G.J. Solar-driven alumina calcination for CO2 mitigation and improved product quality. Green Chem. 2017, 19, 2992–3005. [Google Scholar] [CrossRef]
- Eglinton, T.; Hinkley, J.; Beath, A.; Dell’Amico, M. Potential applications of concentrated solar thermal technologies in the australian minerals processing and extractive metallurgical industry. JOM 2013, 65, 1710–1720. [Google Scholar] [CrossRef]
- Ekman, B.M.; Brooks, G.; Rhamdhani, M.A. A review: Solar thermal reactors for materials production. In Energy Technology 2014: Carbon Dioxide Management and Other Technologies; Wang, C., de Bakker, J., Belt, C.A., Jha, A., Neelameggham, N.R., Pati, N.S., Prentice, L.H., Trnell, G., Brinkman, K.S., Eds.; Wiley: Hoboken, NJ, USA, 2014; pp. 1–14. [Google Scholar] [CrossRef]
- Arenas, M.A.; Padilla, I.; Robla, J.A.; Vázquez, A.J.; López-Delgado, A. The use of concentrated solar energy for the reduction of CuO in H2. Sol. Energy 2019, 180, 640–647. [Google Scholar] [CrossRef]
- Shohoji, N.; Oliveira, F.A.C.; Galindo, J.; Fernandes, J.C.; Rodríguez, J.; Cañadas, I.; Rosa, L.G. Influence of Linear Flow Velocity of Uncracked Ammonia (NH3) Gas on Formation of Higher Nitrides, 𝛅-MoN and 𝛆-Fe2N, under Concentrated Solar Irradiation in the SF40 Solar Furnace at PSA. Internat. J. Mater. Chem. 2019, 9, 1–12. [Google Scholar] [CrossRef][Green Version]
- Alovisio, A.; Chacartegui, R.; Ortiz, C.; Valverde, J.M.; Verda, V. Optimizing the CSP-calcium looping integration for thermochemical energy storage. Energy Convers. Manag. 2017, 136, 85–98. [Google Scholar] [CrossRef]
- Ortiz, C.; Romano, M.C.; Valverde, J.M.; Binotti, M.; Chacartegui, R. Process integration of Calcium-Looping thermochemical energy storage system in concentrating solar power plants. Energy 2018, 155, 535–551. [Google Scholar] [CrossRef]
- Tregambi, C.; Salatino, P.; Solimene, R.; Montagnaro, F. An experimental characterization of Calcium Looping integrated with concentrated solar power. Chem. Eng. J. 2018, 331, 794–802. [Google Scholar] [CrossRef]
- Zhai, R.; Li, C.; Qi, J.; Yang, Y. Thermodynamic analysis of CO2 capture by calcium looping process driven by coal and concentrated solar power. Energy Convers. Manag. 2016, 117, 251–263. [Google Scholar] [CrossRef]
- Douale, P.; Serror, S.; Duval, R.M.P.; Serra, J.J.; Felder, E. Thermal shocks on an electrolytic chromium coating in a solar furnace. J. Phys. IV Fr. 1999, 9, 429–434. [Google Scholar] [CrossRef]
- Kováčik, J.; Emmer, S.; Rodriguez, J.; Cañadas, I. Solar furnace: Thermal shock behaviour of TiB2 coating on steel. In Proceedings of the METAL 2014, Brno, Czech Republic, 21–23 May 2014; pp. 863–868. Available online: https://www.academia.edu/24388464/Solar_Furnace_Thermal_Shock_Behaviour_of_TIB2_Coating_on_Steel (accessed on 20 September 2019).
- Sallaberry, F.; García de Jalón, A.; Zaversky, F.; Vázquez, A.J.; López-Delgado, A.; Tamayo, A.; Mazo, M.A. Towards standard testing materials for high temperature solar receivers. Energy Procedia 2015, 69, 532–542. [Google Scholar] [CrossRef][Green Version]
- Rosa, L.G.; Rodríguez, J.; Pereira, J.; Fernandes, J.C. Thermal cycling behaviour of dense monolithic alumina. In Proceedings of the Second International Conference on Materials Chemistry and Environmental Protection—MEEP 2018, Sanya, China, 23–25 November 2018; pp. 173–179, ISBN 978-989-758-360-5. [Google Scholar] [CrossRef]
- Oliveira, F.A.C.; Fernandes, J.C.; Galindo, J.; Rodríguez, J.; Canãdas, I.; Rosa, L.G. Thermal resistance of solar volumetric absorbers made of mullite, brown alumina and ceria foams under concentrated solar radiation. Sol. Energy Mater. Sol. Cells 2019, 194, 121–129. [Google Scholar] [CrossRef]
- MacCallum, J.R. Photodegradation. In Comprehensive Polymer Science; Allen, G., Bevington, J.C., Eds.; Elsevier, B.V.: Amsterdam, The Netherlands, 1989; Volume 6, pp. 529–537. [Google Scholar] [CrossRef]
- McKeen, L.W. Introduction to the weathering of plastics. In The Effect of UV Light and Weather on Plastics and Elastomers, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 17–41. [Google Scholar] [CrossRef]
- Gomez-Garcia, F.; Santiago, S.; Luque, S.; Romero, M.; Gonzalez-Aguilar, J. A new laboratory-scale experimental facility for detailed aerothermal characterizations of volumetric absorbers. AIP Conf. Proc. 2016, 1734, 030018. [Google Scholar] [CrossRef]
- Luque, S.; Bai, F.; González-Aguilar, J.; Wang, Z.; Romero, M. A parametric experimental study of aerothermal performance and efficiency in monolithic volumetric absorbers. AIP Conf. Proc. 2017, 1850, 030034. [Google Scholar] [CrossRef]
- Li, B.; Oliveira, F.A.C.; Rodríguez, J.; Fernandes, J.C.; Rosa, L.G. Numerical and experimental study on improving temperature uniformity of solar furnaces for materials processing. Sol. Energy 2015, 115, 95–108. [Google Scholar] [CrossRef]
- Oliveira, F.A.C.; Fernandes, J.C.; Rodríguez, J.; Cañadas, I.; Galindo, J.; Rosa, L.G. Temperature uniformity improvement in a solar furnace by indirect heating. Sol. Energy 2016, 140, 141–150. [Google Scholar] [CrossRef]
- Martins, C.A.B. Aparelho Concentrador e Estabilizador de Raios Solares e Sistema de Transmissão de um Feixe de Raios Solares Concentrados e Estabilizados que o Contém. Portuguese Patent Pending Request No. PT20160109071, 4 January 2016. Available online: https://pt.espacenet.com/searchResults?CY=pt&DB=EPODOC&F=0&FIRST=1&LG=pt&PN=PT109071&Submit=PESQUISAR&bookmarkedResults=true&locale=pt_pt&sf=n (accessed on 22 July 2019).
- Costa, B.A.; Lemos, J.M.; Rosa, L.G. Temperature control of a solar furnace for material testing. Int. J. Syst. Sci. 2011, 42, 1253–1264. [Google Scholar] [CrossRef]
- Costa, B.A.; Lemos, J.M. Predictive adaptive temperature control in a solar furnace for material stress tests. In Proceedings of the 2012 IEEE Multi-conference on Systems and Control, Dubrovnik, Croatia, 3–5 October 2012; pp. 1340–1345. [Google Scholar] [CrossRef]
- Costa, B.A.; Lemos, J.M.; Guillot, E. Solar furnace temperature control with active cooling. Sol. Energy 2018, 159, 66–77. [Google Scholar] [CrossRef]
- Nakamura, T. Optical waveguide system for solar power applications in space. In Nonimaging Optics: Efficient Design for Illumination and Solar Concentration VI; International Society for Optics and Photonics: San Francisco, CA, USA, 2009; Volume 7423, p. 74230C. [Google Scholar] [CrossRef]
- Nakamura, T.; Smith, B.K. Solar thermal system for lunar ISRU applications: Development and field operation at Mauna Kea, HI. In Proceedings of the SPIE 8124, Nonimaging Optics: Efficient Design for Illumination and Solar Concentration VIII; 81240B (2011), San Diego, CA, USA, 21–25 August 2011. [Google Scholar] [CrossRef]
- Nakamura, T.; Smith, B.K.; Irvin, B.R. Optical waveguide solar power system for material processing in space. J. Aerosp. Eng. 2015, 28, 04014051. [Google Scholar] [CrossRef]
- Henshall, P.; Palmer, P. Concentrator pointing control concept for fiber optic augmented solar thermal propulsion systems. J. Spacecr. Rockets 2016, 53, 230–234. [Google Scholar] [CrossRef]
- Alwahabi, Z.T.; Kueh, K.C.; Nathan, G.J.; Cannon, S. Novel solid-state solar thermal simulator supplying 30,000 suns by a fibre optical probe. Opt. Express 2016, 24, A1444–A1453. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Fan, G.H.; Zhang, Y.Q.; Ji, X.F. A fresnel concentrator with fiber-optic bundle based space solar power satellite design. Acta Astronaut. 2018, 153, 122–129. [Google Scholar] [CrossRef]
- De Almeida, G. Proposta: Estrutura de Suporte do Concentrador e Dispositivos de Fixação e Regulação dos Seus Elementos Ópticos; Ed. do Autor: Lisboa, Portugal, 2019. [Google Scholar]



© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosa, L.G. Solar Heat for Materials Processing: A Review on Recent Achievements and a Prospect on Future Trends. ChemEngineering 2019, 3, 83. https://doi.org/10.3390/chemengineering3040083
Rosa LG. Solar Heat for Materials Processing: A Review on Recent Achievements and a Prospect on Future Trends. ChemEngineering. 2019; 3(4):83. https://doi.org/10.3390/chemengineering3040083
Chicago/Turabian StyleRosa, Luís Guerra. 2019. "Solar Heat for Materials Processing: A Review on Recent Achievements and a Prospect on Future Trends" ChemEngineering 3, no. 4: 83. https://doi.org/10.3390/chemengineering3040083
APA StyleRosa, L. G. (2019). Solar Heat for Materials Processing: A Review on Recent Achievements and a Prospect on Future Trends. ChemEngineering, 3(4), 83. https://doi.org/10.3390/chemengineering3040083




