Progress in Modeling of Silica-Based Membranes and Membrane Reactors for Hydrogen Production and Purification
Abstract
:1. Introduction
- Cost-effective hydrogen generation in a carbon constrained global energy system: the challenges in this area involve hydrogen production from fossil fuels combined to carbon sequestration, in parallel with an increase in renewable sources exploitation.
- Hydrogen purification and storage technologies able to separate and purify hydrogen streams to the requirements of the end-users: efficient hydrogen separation and storage devices will have to match the United States—Department of Energy (US DOE) targets, such as hydrogen permeability and permselectivity (2015, last up-date), recognized worldwide as reference values for any hydrogen permselective membrane worthy of interest for its potential utilization in industrial applications.
- An efficient, widely available, and well-managed hydrogen delivery and distribution infrastructure.
- Efficient fuel cells and other energy conversion technologies fueled by hydrogen.
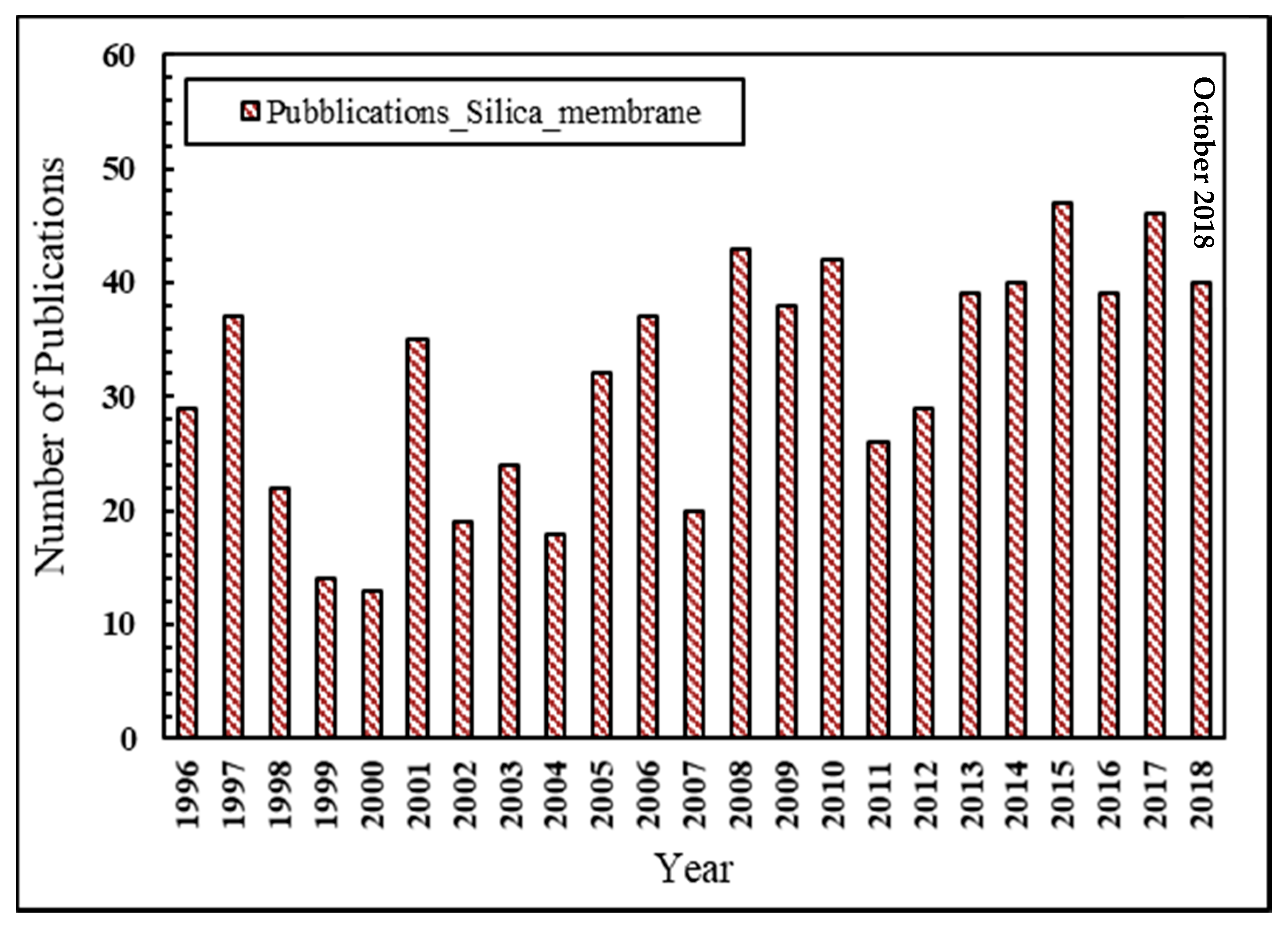
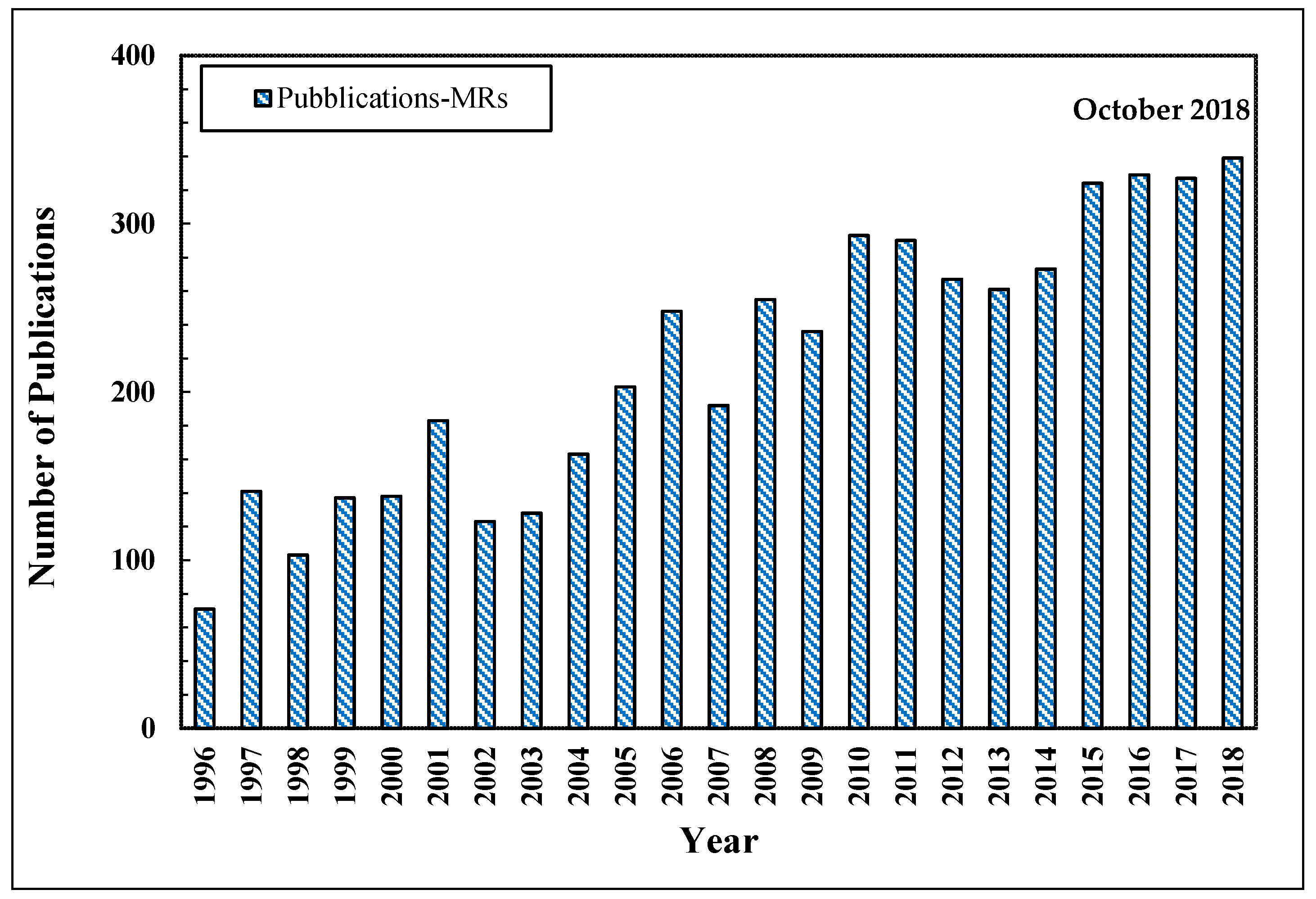
2. Silica Membranes
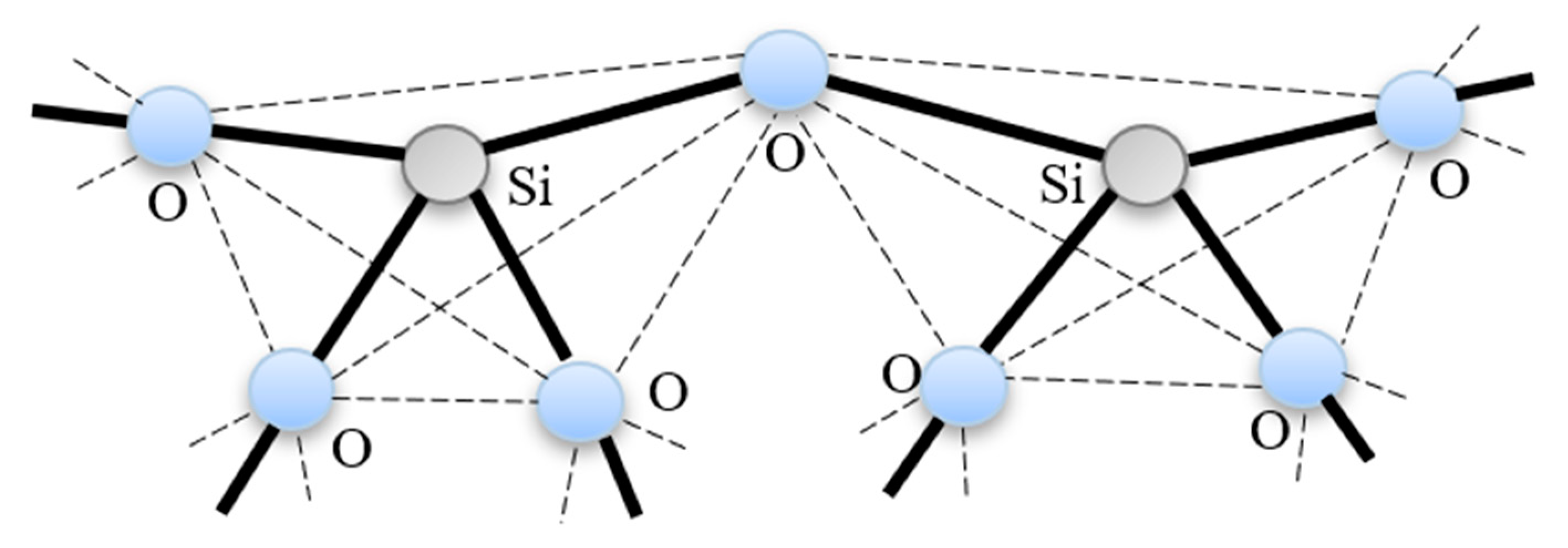
2.1. Silica Structure
2.2. Transport Mechanism in Microporous Silica Membrane
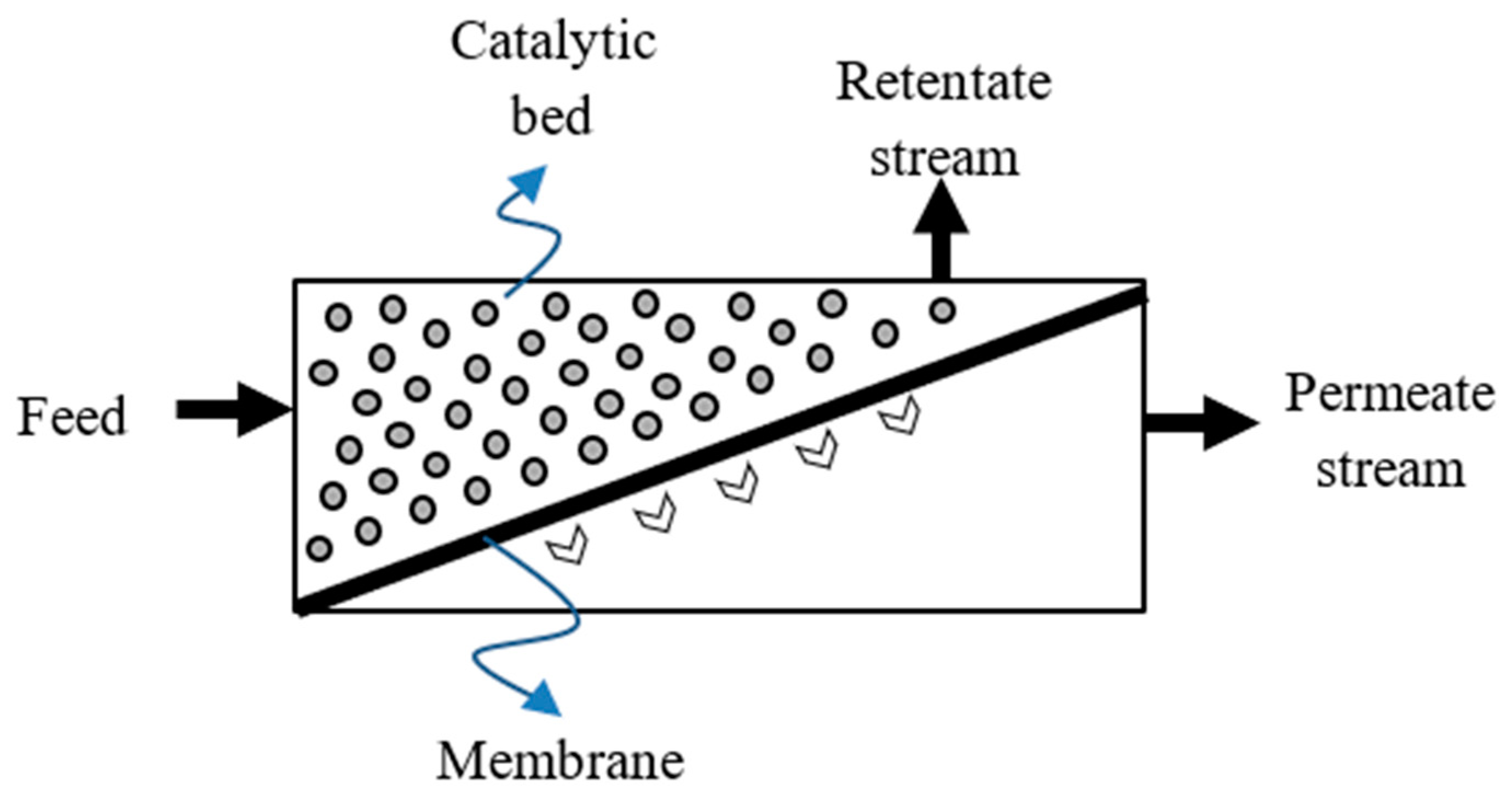
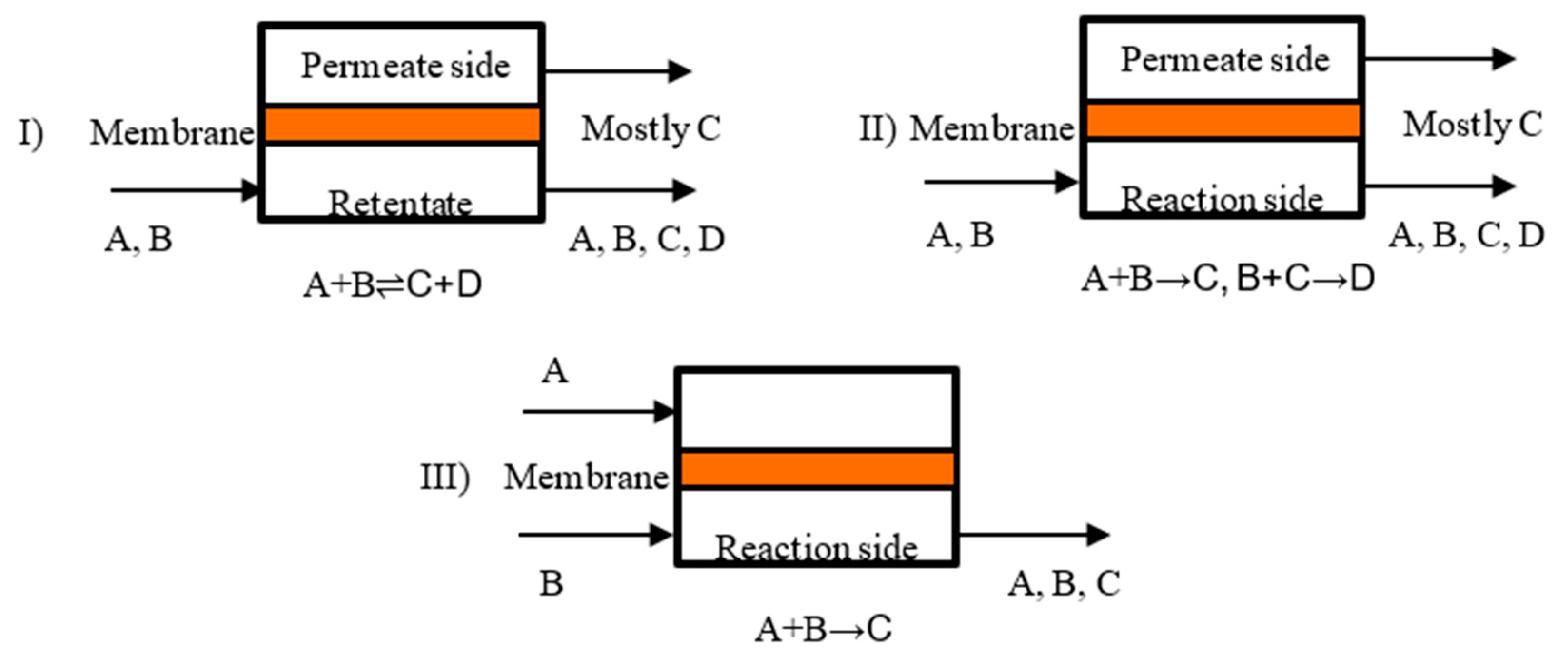
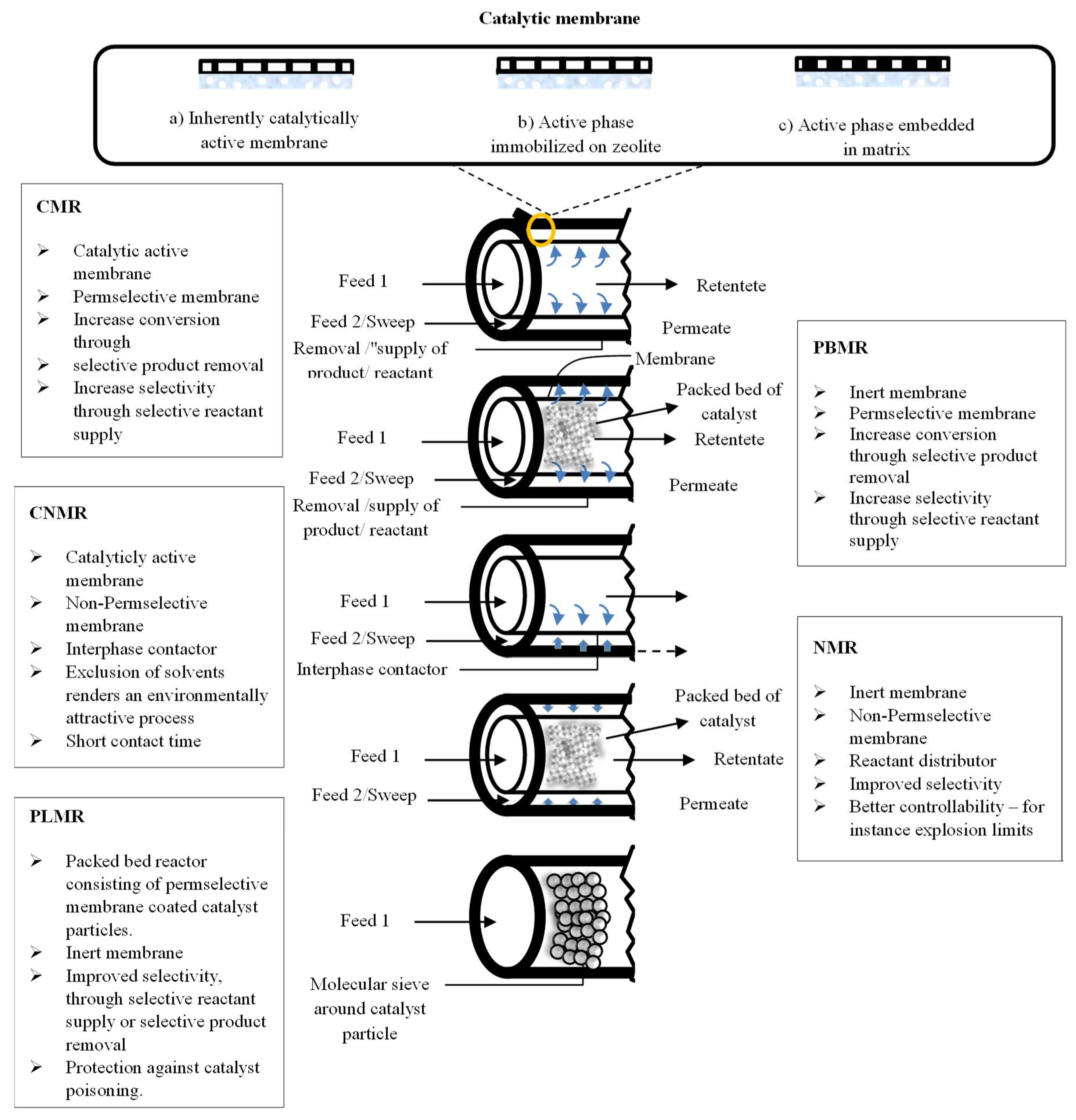
3. Membrane Reactor Technology
- catalytic membrane reactors (CMR);
- packed bed membrane reactors (PBMR);
- catalytic non-permselective membrane reactors (CNMR),
- non-permselective membrane reactors (NMR);
- reactant-selective packed bed reactors (RSPBR).
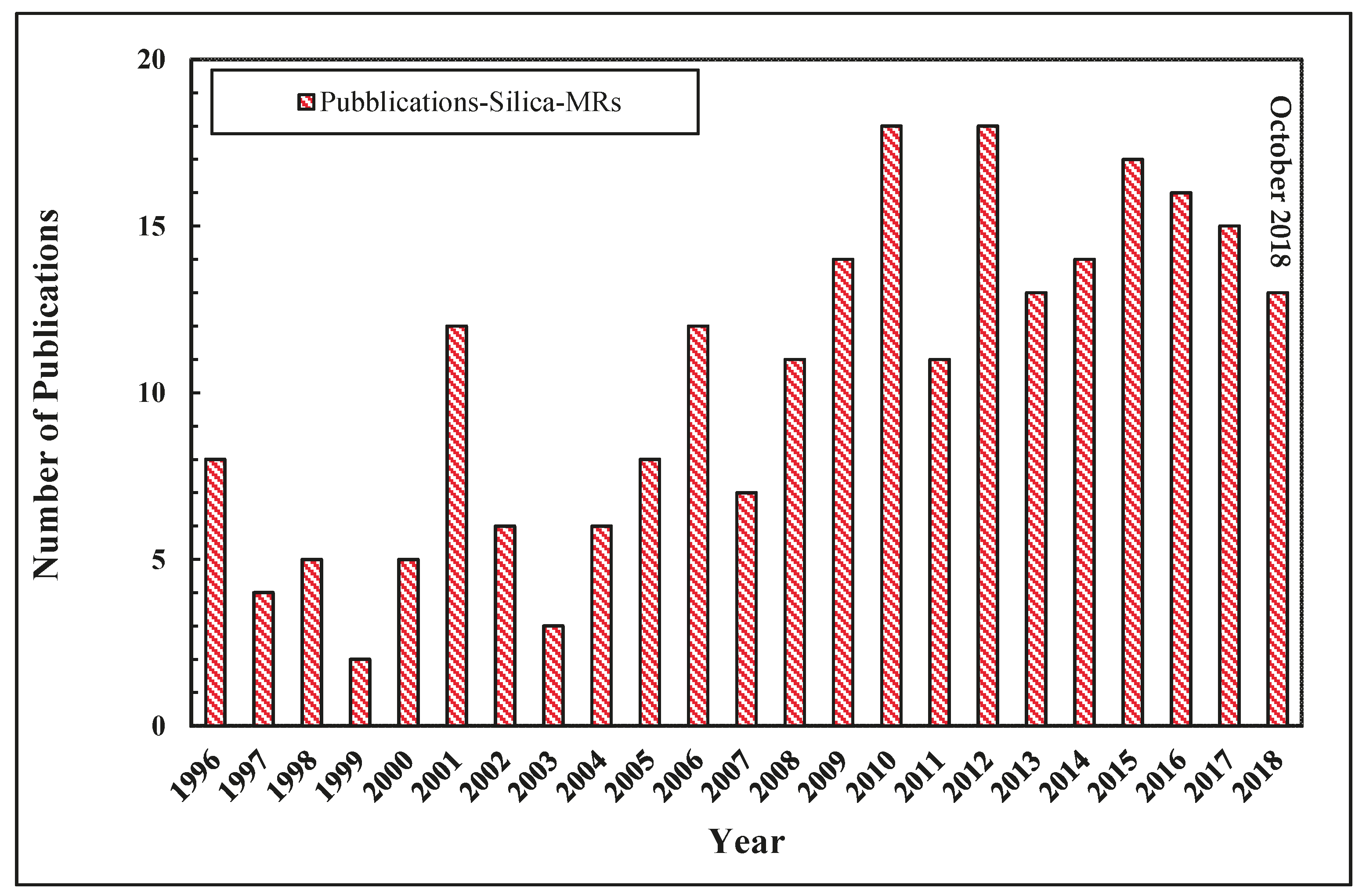
4. Application of Silica Membranes in MR Systems
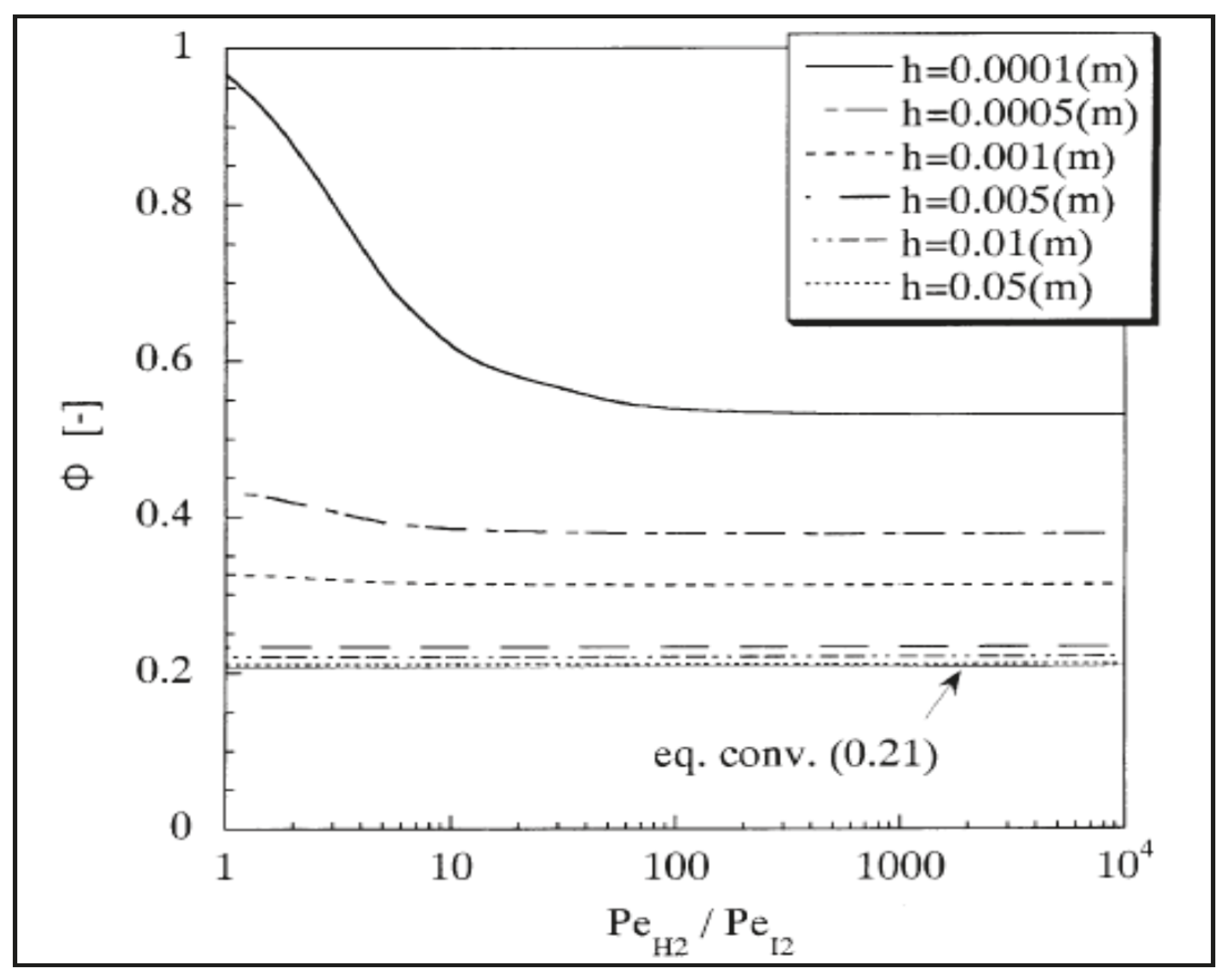
Modeling of Silica MRs
- the simple model represents a useful tool for a preliminary evaluation of the WGS-MR performance.
- The CFD-based model can be adopted for designing the WGS-MR and for optimising the silica MR performance.
- The desired MR performance can be provided with a high perm-selective silica membrane.
5. Conclusions and Future Trends
Funding
Conflicts of Interest
List of Acronyms
| ANN | Artificial Neural Network |
| CCS | Carbon capture and storage |
| CFD | Computational fluid dynamics |
| CMR | Catalytic membrane reactor |
| CNMR | Catalytic non perm-selective membrane reactors |
| FBR | Fixed bed reactor |
| MD | Molecular dynamics |
| MR | Membrane reactor |
| NMR | Non perm-selective membrane reactors |
| PBMR | Packed bed membrane reactors |
| PEMFC | Proton exchange membrane fuel cell |
| RSPBR | Reactant-selective packed bed reactors |
| TR | Traditional reactor |
| WGS | Water gas shift |
List of Symbols
| D0 | mean intrinsic diffusion coefficient for micropore diffusion (m2·s−1) |
| K0 | intrinsic Henry constant (-) |
| ε | membrane porosity (-) |
| L | membrane thickness (m) |
| bulk density (Kg/m3) | |
| qst | isosteric heat adsorption (J/mol) |
| Ei | activation energy for gas species (KJ/mol) |
| R | universal gas constant (J/mol·K) |
| T | temperature (K) |
References
- Bujnicki, J.; Dykstra, P.; Fortunato, E.; Heuer, R.-D.; Keskitalo, C.; Nurse, P. Novel carbon capture and utilisation technologies, Scientific Opinion 4/2018 (Supported by SAPEA Evidence Review Report No 2), European Commission—Directorate-General for Research and Innovation Unit RTD.DDG1.02—Scientific Advice Mechanism, Brussel. 23 May 2018. Available online: https://ec.europa.eu/research/sam/pdf/sam_ccu_report.pdf (accessed on 18 October 2018).
- Ji, G.; Zhao, M. Membrane separation technology in carbon capture. In Recent Advances in Carbon Capture and Storage; Yun, Y., Ed.; Intech Open Science: Rijeka, Croatia, 2017; pp. 59–90. [Google Scholar]
- Qiu, H.H.; Liu, L.G. A Study on the evolution of carbon capture and storage technology based on knowledge mapping. Energy 2018, 11, 1103. [Google Scholar] [CrossRef]
- AIulianelli, A.; Basile, A. Sulfonated PEEK-based polymers in PEMFC and DMFC applications: A review. Int. J. Hydrogen Eng. 2012, 37, 15241–15255. [Google Scholar] [CrossRef]
- Vivek, R.; Muthukumar, M.D. Performance improvement of Proton Exchange Membrane Fuel Cell. Innov. Energy Res. 2018, 7, 1–5. [Google Scholar]
- Basu, S. Proton Exchange Membrane Fuel Cell Technology: India’s Perspective. Proc. Indian Natl. Sci. Acad. 2015, 81, 865–890. [Google Scholar] [CrossRef]
- Basile, A.; Iulianelli, A. Advances in Hydrogen Production, Storage and Distribution, 1st ed.; Woodhead Publishing: Sawston, UK, 7 July 2014; pp. 1–546. ISBN 9780857097682. [Google Scholar]
- Voitic, G.; Pichler, B.; Basile, A.; Iulianelli, A.; Malli, K.; Bock, S.; Hacker, V. Chapter 10—Hydrogen Production. In Fuel Cells and Hydrogen; Hacker, V., Mitsushima, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 215–241. ISBN 9780128114599. [Google Scholar]
- Lu, N.; Xie, D. Novel membrane reactor concepts for hydrogen production from hydrocarbons: A review. Int. J. Chem. React. Eng. 2015, 14, 1–31. [Google Scholar] [CrossRef]
- Gallucci, F.; Fernandez, E.; Corengia, P.; van Sint, M. Annaland, Recent advances on membranes and membrane reactors for hydrogen production. Chem. Eng. Sci. 2013, 92, 40–66. [Google Scholar] [CrossRef]
- Lu, G.Q.; da Costa, J.C.D.; Duke, M.; Giessler, S.; Socolow, R.; Williams, R.H.; Kreutz, T. Inorganic membranes for hydrogen production and purification: A critical review and perspective. J. Coll. Interface Sci. 2007, 314, 589–603. [Google Scholar] [CrossRef]
- Pacheco, D.T.; van Sint Annaland, M.; Gallucci, F. Recent advances in Pd-based membranes for membrane reactors. Molecules 2017, 22, 51. [Google Scholar]
- Abdallah, H. A review on catalytic membranes production and applications. Bull. Chem. React. Eng. Catal. 2017, 12, 136–156. [Google Scholar] [CrossRef]
- Ayral, A.; Julbe, A.; Roualdes, S.; Rouessac, V.; Durand, J.; Sala, B. Silica membranes-basic principles. Chem. Eng. 2006, 50, 67–79. [Google Scholar]
- Sugawara, T.; Takagaki, A.; Kikuchi, R. Review on mechanisms of gas permeation through inorganic membranes. J. Jpn. Petrol. Inst. 2011, 54, 298–309. [Google Scholar]
- Lee, P.S.; Lee, K.H. Inorganic membranes for gas separation. In Membrane Engineering for the Treatment of Gases: Volume 2: Gas-Separation Issues Combined with Membrane Reactors, 2nd ed.; Drioli, E., Barbieri, G., Brunetti, A., Eds.; Royal Society of Chemistry: London, UK, 2017; Chapter 10; pp. 303–357. ISBN 978-1-78262-875-0. [Google Scholar]
- Li, C.; Meckler, S.M.; Smith, Z.P.; Bachman, J.E.; Maserati, L.; Long, J.R.; Helms, B.A. Engineered transport in microporous materials and membranes for clean energy technologies. Adv. Mater. 2018, 30, 1704953–1704986. [Google Scholar] [CrossRef] [PubMed]
- Fotou, G.P.; Lin, Y.S.; Pratsinis, S.E. Hydrothermal stability of pure and modified microporous silica membranes. J. Mater. Sci. 1995, 30, 2803–2808. [Google Scholar] [CrossRef]
- Rowe, B.W.; Robeson, L.M.; Freeman, B.D.; Paul, D.R. Influence of temperature on the upper bound: Theoretical considerations and comparison with experimental results. J. Membr. Sci. 2010, 360, 58–69. [Google Scholar] [CrossRef]
- De Lange, R.; Keizer, K.; Burggraf, A.J. Analysis and theory of gas transport in microporous sol–gel derived ceramic membranes. J. Membr. Sci. 1995, 104, 81–100. [Google Scholar] [CrossRef]
- Dixon, A.G. Recent research in catalytic inorganic membrane reactors. Int. J. Chem. React. Eng. 2003, 1, 1–35. [Google Scholar] [CrossRef]
- Alique, D.; Martinez-Diaz, D.; Sanz, R.; Calles, J. Review of supported Pd-based membranes preparation by electroless plating for ultra-pure hydrogen production. Membranes 2018, 8, 5–44. [Google Scholar] [CrossRef]
- Cardoso, S.P.; Azenha, I.S.; Lin, Z.; Portugal, I.; Rodrigues, A.E.; Silva, C.M. Inorganic membranes for hydrogen separation. Sep. Purif. Rev. 2018, 47, 229–266. [Google Scholar] [CrossRef]
- Rahimpour, M.R.; Samimi, F.; Babapoor, A.; Tohidian, T.; Mohebi, S. Palladium membranes applications in reaction systems for hydrogen separation and purification: A review. Chem. Eng. Proc.: Process Intens. 2017, 121, 24–49. [Google Scholar] [CrossRef]
- Dolan, M.D. Non-Pd BCC alloy membranes for industrial hydrogen separation. J. Membr. Sci. 2010, 362, 12–28. [Google Scholar] [CrossRef]
- Murmura, M.A.; Cerbelli, S.; Annesini, M.C. Modeling fixed bed membrane reactors for hydrogen production through steam reforming reactions: A critical analysis. Membranes 2018, 19, 8. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, M.; Aghili, F. Recent advances in membrane reactors for hydrogen production by steam reforming of ethanol as a renewable resource. Rev. Chem. Eng. 2018, in press. [Google Scholar] [CrossRef]
- Iulianelli, A.; Longo, T.; Liguori, S.; Basile, A. Production of hydrogen via glycerol steam reforming in a Pd-Ag membrane reactor over Co-Al2O3 catalyst. Asia-Pacific J. Chem. Eng. 2010, 5, 138–145. [Google Scholar] [CrossRef]
- Gallucci, F.; Medrano, J.; Fernandez, E.; Melendez, J.; Annaland, M.V.; Pacheco, A. Advances on high temperature Pd-based membranes and membrane reactors for hydrogen purification and production. J. Membr. Sci. Res. 2017, 3, 142–156. [Google Scholar]
- Ghasemzadeh, K.; Aghaeinejad-Meybodi, A.; Iulianelli, A.; Basile, A. Theoretical performance evaluation of not Pd-based membranes for hydrogen separation. J. Membr. Sci. Res. 2018, 4, 198–203. [Google Scholar]
- Yin, H.; Yip, A.C.K. A review on the production and purification of biomass-derived hydrogen using emerging membrane technologies. Catalysts 2018, 7, 297–327. [Google Scholar] [CrossRef]
- Ghasemzadeh, K.; Tilebon, S.M.S.; Basile, A. Modeling of silica membranes. In Current Trends and Future Developments on (Bio-) Membranes; Basile, A., Ghasemzadeh, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Chapter 6; pp. 135–153. ISBN 9780444638663. [Google Scholar]
- Ghasemzadeh, K.; Aghaeinejad-Meybodi, A.; Basile, A. Hydrogen production as a green fuel in silica membrane reactor: Experimental analysis and artificial neural network modeling. Fuel 2018, 222, 114–124. [Google Scholar] [CrossRef]
- Ghasemzadeh, K.; Zeynali, R.; Basile, A.; Iulianelli, A. CFD analysis of a hybrid sorption-enhanced membrane reactor for hydrogen production during WGS reaction. Int. J. Hydrog. Eng. 2017, 42, 26914–26923. [Google Scholar] [CrossRef]
- Meng, L.; Tsuru, T. Hydrogen production from energy carriers by silica-based catalytic membrane reactors, Catal. Today 2016, 268, 3–11. [Google Scholar] [CrossRef]
- Koukou, M.K.; Papayannakos, N.; Markatos, N.C.; Bracht, M.; Alderliesten, P.T. Simulation tools for the design of industrial-scale membrane reactors. Chem. Eng. Res. Des. 1998, 76, 911–920. [Google Scholar] [CrossRef]
- Prabhu, A.K.; Liu, A.; Lovell, L.G.; Ted Oyama, S. Modeling of the methane reforming reaction in hydrogen selective membrane reactors. J. Membr. Sci. 2000, 177, 83–95. [Google Scholar] [CrossRef]
- Hwang, G.J.; Onuki, K. Simulation study on the catalytic decomposition of hydrogen iodide in a membrane reactor with a silica membrane for the thermochemical water-splitting IS process. J. Membr. Sci. 2001, 194, 207–215. [Google Scholar] [CrossRef]
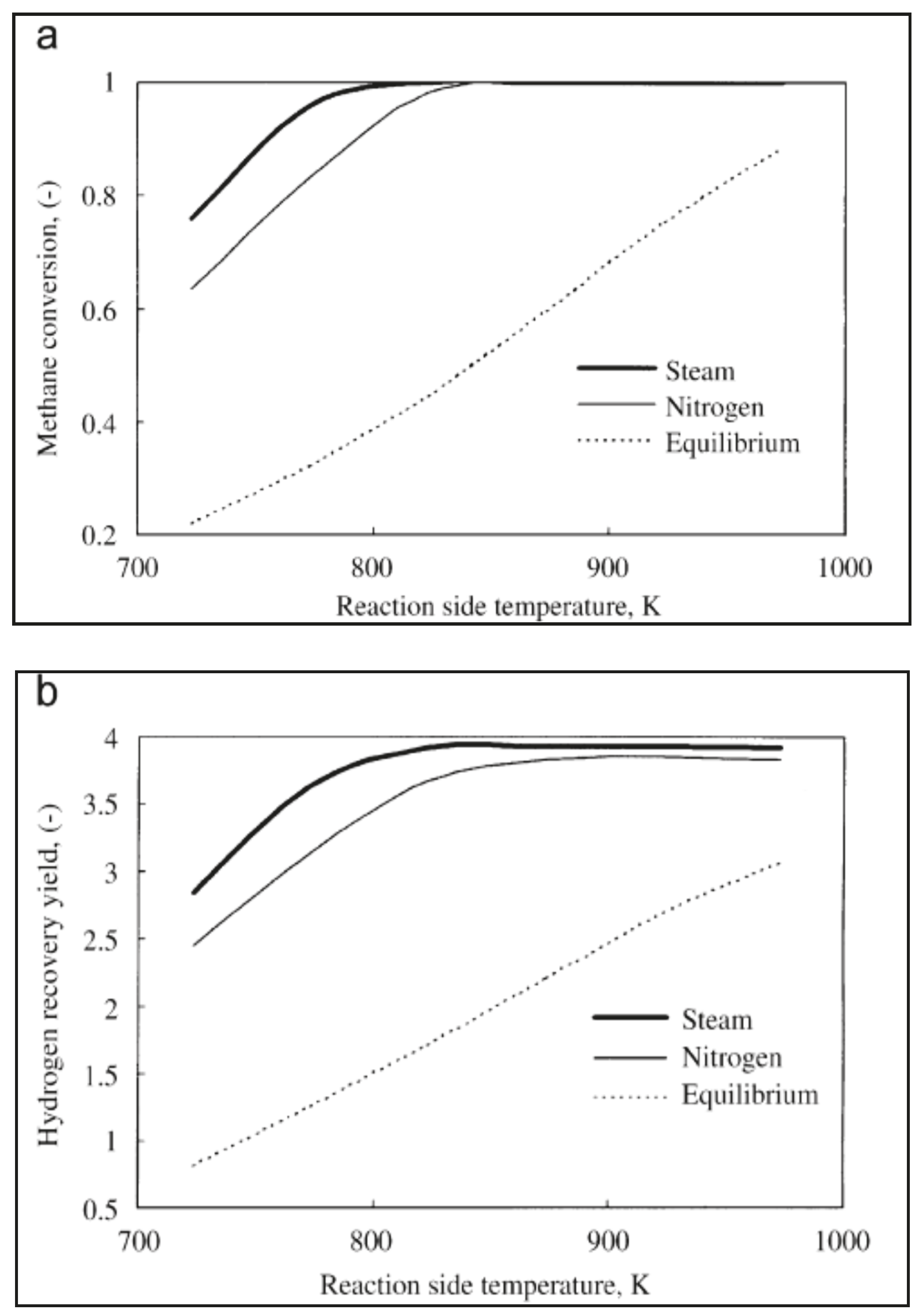
- Yu, W.; Ohmori, T.; Kataoka, S.; Yamamoto, T. A comparative simulation study of methane steam reforming in a porous ceramic membrane reactor using nitrogen and steam as sweep gases. Int. J. Hydrog. Eng. 2008, 33, 685–692. [Google Scholar] [CrossRef]
- Oyama, S.T.; Hacarlioglu, P. The boundary between simple and complex descriptions of membrane reactors: The transition between 1-D and 2-D analysis. J. Membr. Sci. 2009, 337, 188–199. [Google Scholar] [CrossRef]
- Tsuru, T.; Morita, T.; Shintani, H.; Yoshioka, T. Membrane reactor performance of steam reforming of methane using hydrogen-permselective catalytic SiO2 membranes. J. Membr. Sci. 2008, 316, 53–62. [Google Scholar] [CrossRef]
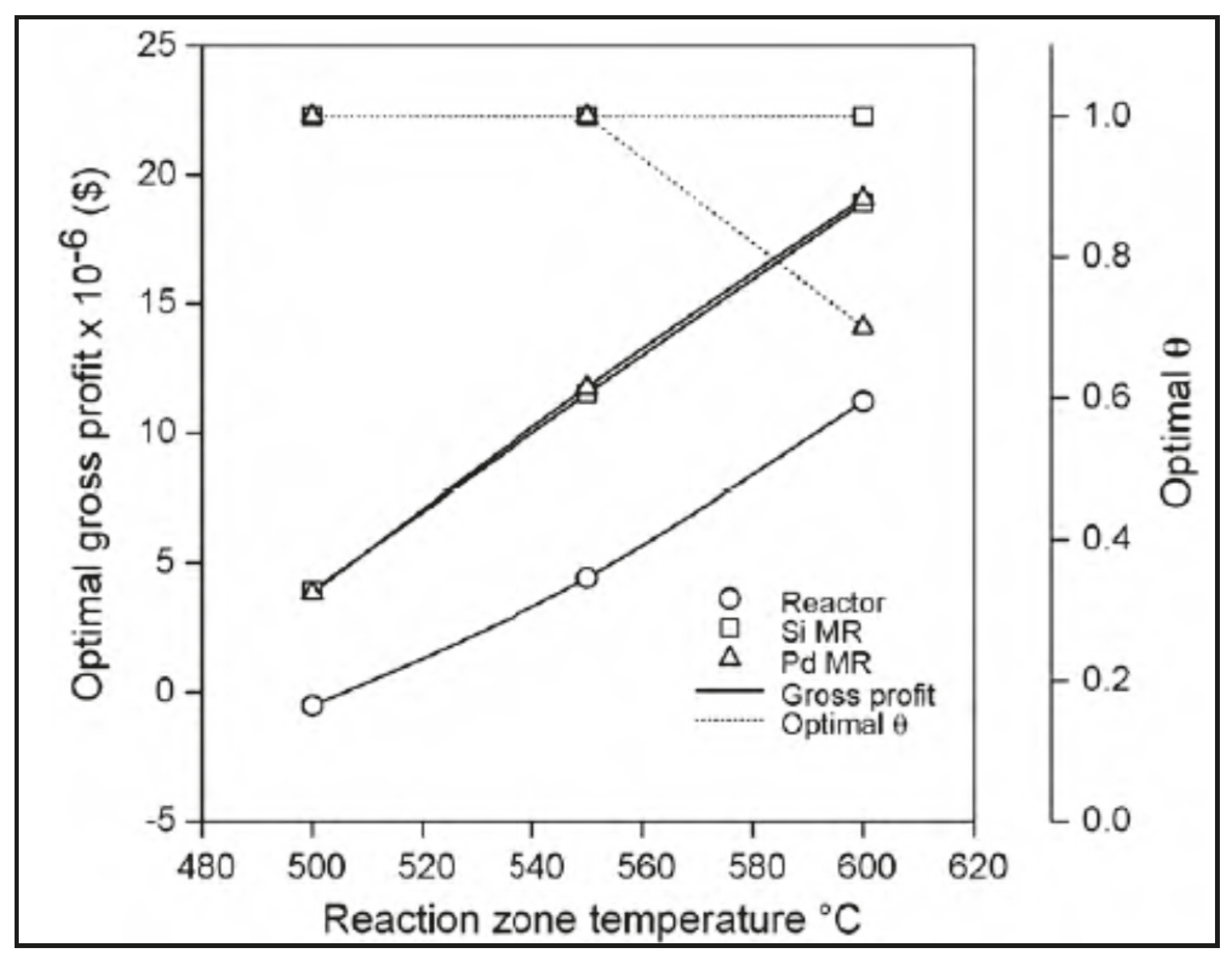
- Moparthi, A.; Uppaluri, R.; Gill, B.S. Economic feasibility of silica and palladium composite membranes for industrial dehydrogenation reactions. Chem. Eng. Res. Des. 2010, 88, 1088–1101. [Google Scholar] [CrossRef]
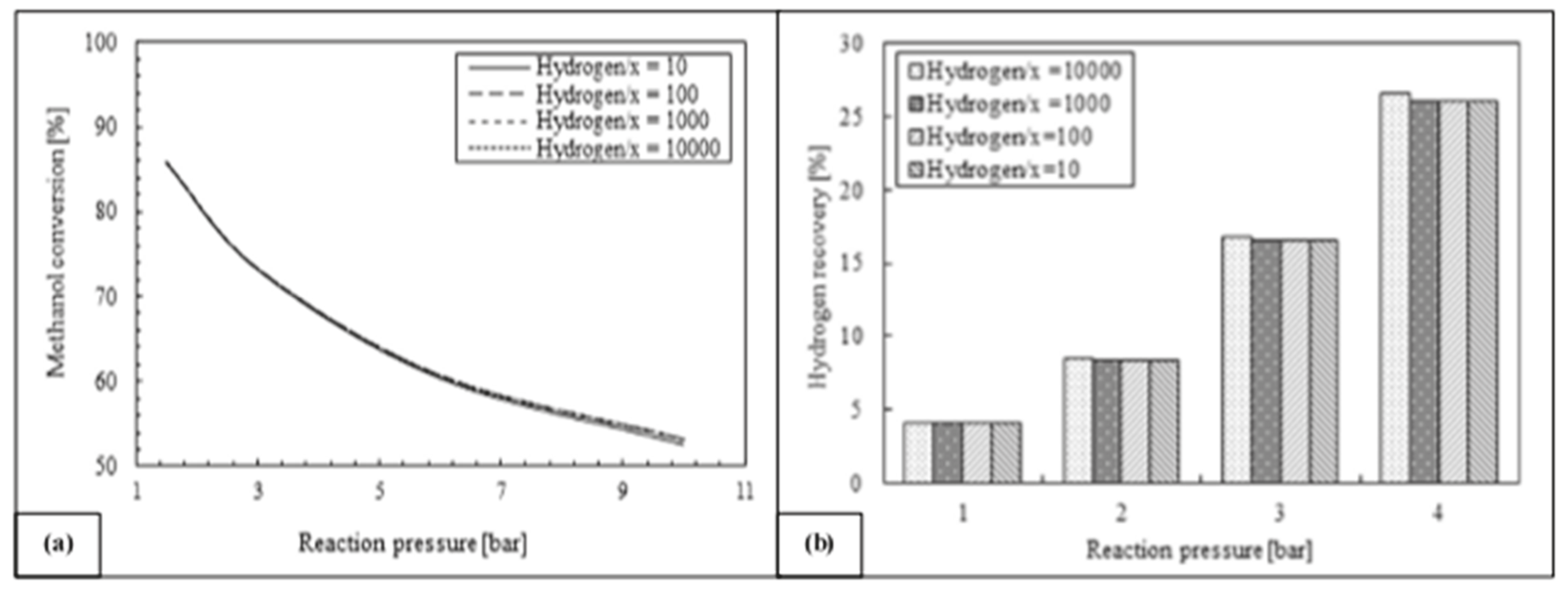
- Ghasemzadeh, K.; Morrone, P.; Liguori, S.; Babaluo, A.A.; Basile, A. Evaluation of silica membrane reactor performance for hydrogen production via methanol steam reforming: Modeling study. Int. J. Hydrog. Eng. 2013, 38, 16698–16709. [Google Scholar] [CrossRef]
- Ghasemzadeh, K.; Morrone, P.; Iulianelli, A.; Liguori, S.; Babaluo, A.A.; Basile, A. H2 production in silica membrane reactor via methanol steam reforming: Modeling and HAZOP analysis. Int. J. Hydrog. Eng. 2013, 38, 10315–10326. [Google Scholar] [CrossRef]
- Ghasemzadeh, K.; Morrone, P.; Babaluo, A.A.; Basile, A. A simulation study on methanol steam reforming in the silica membrane reactor for hydrogen production. Int. J. Hydrog. Eng. 2015, 39, 2–11. [Google Scholar] [CrossRef]
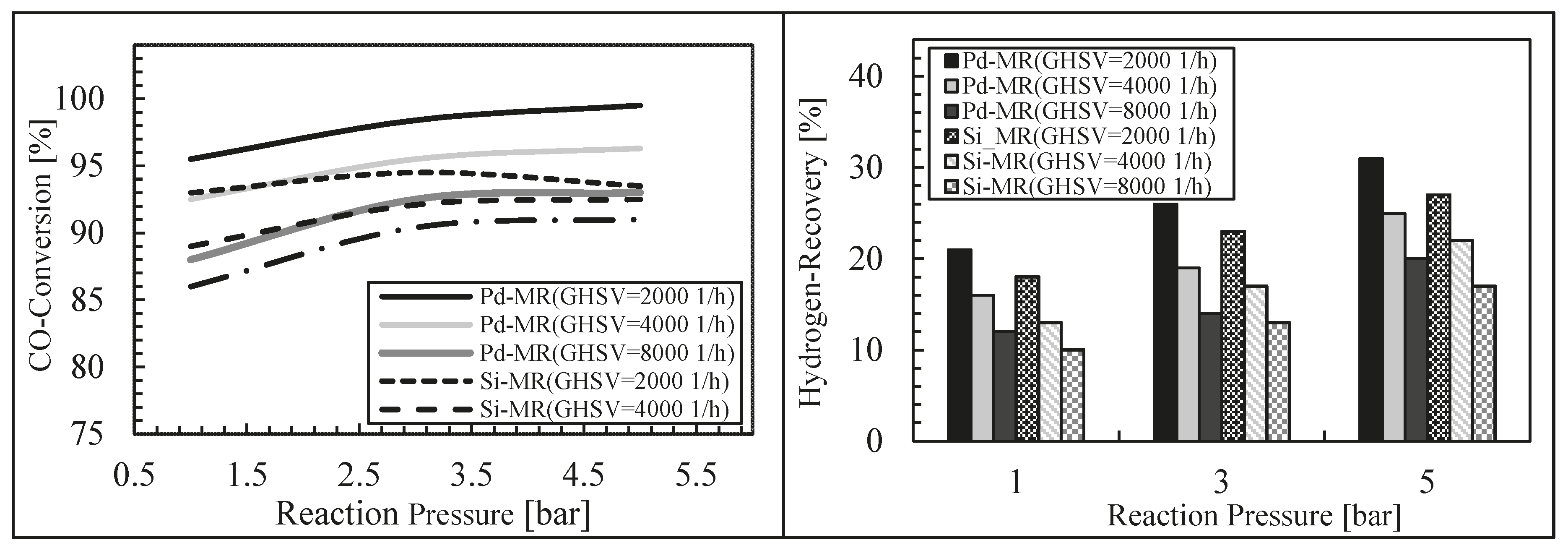
- Ghasemzadeh, K.; Zeynali, R.; Basile, A. Theoretical study of hydrogen production using inorganic membrane reactors during WGS reaction. Int. J. Hydrog. Eng. 2016, 41, 8696–8705. [Google Scholar] [CrossRef]
| Microporous (<2 nm) | Mesopores | Macropores | |
|---|---|---|---|
| Ultramicropores | Supermicropores | ||
| <0.7 nm | >0.7 nm | 2–50 nm | >50 nm |
| Type of Membrane | Transport Mechanism | Perm-Selectivity |
|---|---|---|
| Macroporous > 50 nm | Viscous flow Molecular diffusion  | 1 |
| 1 | ||
| Mesoporous, 2–50 nm | Knudsen diffusion Surface diffusion  Capillary Condensation, pore filling  | |
| More than Knudsen value | ||
| Much more than Knudsen value | ||
| Microporous, <2 nm | Micropore diffusion | Much more than Knudsen value |
| Silica MRs Advantages | Silica MRs Disadvantages |
|---|---|
|
|
|
|
|
|
| Model Type | Theoretical Assumptions | Reaction Process | Primary Target | References |
|---|---|---|---|---|
| Mass balance and CFD | 2-D Non-Isothermal (CFD model) Isothermal (simple model) | WGS reaction | Simulation of silica MR performance by two types of mathematical models | Kouku et al. [36] |
| Mass balance | 1-D Isothermal | Methane dry reforming | Comparison between silica MR and other kinds of reactors | Prabhu et al. [37] |
| Mass balance | One-dimensional Isothermal | HI decomposition reaction | Evaluation of silica MR performance | Hwang and Onuki [38] |
| Mass balance | 1-D Isothermal and pseudo-homogeneous | Methane steam reforming | Evaluation of silica MR performance | Yu et al. [39] |
| Mass balance | 1-D 2-D | Methane steam reforming | silica MR performance comparison between 1-D and 2-D models | Oyama and Hacarlioglu [40] |
| Mass balance | 1-D Isothermal | Methane steam reforming | Permselectivity variation effects on silica MR performance | Tsuru et al. [41] |
| Mass balance | 1-D Isothermal | WGS reaction | Evaluation of silica MR performance | Tsuru et al. [41] |
| Mass balance and economic analysis | 1-D | Propane and ethylbenzene dehydrogenations | Economic evaluation between silica and Pd-based MRs | Moparthi et al. [42] |
| Mass balance | 1-D 2-D | Methanol steam reforming and WGS | Evaluation of silica MR performance | Ghasemzadeh et al. [43,44,45,46] |
| K2 | Mole Fraction | |||||
|---|---|---|---|---|---|---|
| Experimental | Theoretical | H2O | H2 | CO | CO2 | CH4 |
| Fixed-bed Reactor | ||||||
| 0.37 | 0.37 | 0.03 | 0.2 | 0.26 | 0.11 | 0.14 |
| Vycor MR | ||||||
| 0.45 | 0.37 | 0.03 | 0.18 | 0.24 | 0.09 | 0.12 |
| Nanosil MR | ||||||
| 0.38 | 0.37 | 0.03 | 0.19 | 0.27 | 0.1 | 0.13 |
| T (K) | P (bar) | H2 yield × 10−6 (Experimental) | CO yield × 10−6 (Experimental) | CO2 yield × 10−6 (Experimental) | H2 yield × 10−6 (Theoretical) | CO yield × 10−6 (Theoretical) | CO2 yield × 10−6 (Theoretical) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PBR | MR | PBR | MR | PBR | MR | PBR | MR | PBR | MR | PBR | MR | ||
| 873 | 1 | 7.4 | 10.4 | 0.9 | 0.9 | 1.6 | 1.8 | 9 | 11.5 | 0.9 | 0.9 | 1.6 | 1.9 |
| 5 | 24.4 | 34.1 | 1.6 | 1.9 | 5.3 | 7.0 | 27.2 | 38.0 | 1.8 | 2.0 | 5.8 | 7.6 | |
| 10 | 38.6 | 53.1 | 1.7 | 2.3 | 7.9 | 11.5 | 41.2 | 57.9 | 1.9 | 2.5 | 8.7 | 12.9 | |
| 15 | 48.1 | 67.1 | 1.8 | 2.5 | 9.6 | 14.8 | 52.2 | 73.1 | 2 | 2.8 | 11.8 | 16.9 | |
| 20 | 55.6 | 77.7 | 1.8 | 2.7 | 10.9 | 17.5 | 60.4 | 85.7 | 2.2 | 3.1 | 13.2 | 20.4 | |
| 923 | 1 | 7.9 | 11.5 | 1.5 | 1.4 | 1.5 | 1.8 | 9.8 | 12.3 | 1.5 | 1.3 | 1.5 | 1.8 |
| 5 | 31.2 | 42.8 | 3.3 | 3.8 | 6.1 | 7.8 | 34.4 | 46.1 | 2.6 | 3.8 | 6.4 | 8.2 | |
| 10 | 50.1 | 68.9 | 3.7 | 4.9 | 9.7 | 13.5 | 53.9 | 76.1 | 3.1 | 5.1 | 10.2 | 14.5 | |
| 15 | 62.4 | 88.7 | 3.9 | 5.4 | 12.0 | 18.0 | 66.7 | 95.3 | 4.2 | 5.8 | 14.2 | 20.0 | |
| 20 | 73.1 | 104.7 | 4.0 | 5.7 | 13.8 | 21.8 | 79.2 | 112.7 | 4.6 | 6.0 | 16.7 | 25.0 | |
| Silica MR | Dense Pd-Ag MR | ||||
|---|---|---|---|---|---|
| H2/N2 = 14 | H2/N2 = 60 | H2/N2 = 600 | H2/N2 = 6000 | H2/N2 = ∞ | |
| Methanol conversion [%] | 59.33 | 70.21 | 75.41 | 76.11 | 80.95 |
| Hydrogen Recovery [%] | 30.97 | 58.29 | 67.03 | 67.29 | 52.07 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghasemzadeh, K.; Basile, A.; Iulianelli, A. Progress in Modeling of Silica-Based Membranes and Membrane Reactors for Hydrogen Production and Purification. ChemEngineering 2019, 3, 2. https://doi.org/10.3390/chemengineering3010002
Ghasemzadeh K, Basile A, Iulianelli A. Progress in Modeling of Silica-Based Membranes and Membrane Reactors for Hydrogen Production and Purification. ChemEngineering. 2019; 3(1):2. https://doi.org/10.3390/chemengineering3010002
Chicago/Turabian StyleGhasemzadeh, Kamran, Angelo Basile, and Adolfo Iulianelli. 2019. "Progress in Modeling of Silica-Based Membranes and Membrane Reactors for Hydrogen Production and Purification" ChemEngineering 3, no. 1: 2. https://doi.org/10.3390/chemengineering3010002
APA StyleGhasemzadeh, K., Basile, A., & Iulianelli, A. (2019). Progress in Modeling of Silica-Based Membranes and Membrane Reactors for Hydrogen Production and Purification. ChemEngineering, 3(1), 2. https://doi.org/10.3390/chemengineering3010002







