3.2.1. Texture and Structure Properties
The positive effect of the addition cerium species to support on the texture parameters (the growth of surface area, total pore volume, and average pore size) was confirmed by the results of the adsorption and desorption of nitrogen (
Table 4). It can be explained by the better dissolution of cerium nitrate in the solution of methanol and Pluronic P-123, than zirconium oxonitrate during the preparation of oxides, and also the longer crystallization of ceria than zirconia. It can be a reason why the real molar ratios between ceria and zirconia on the external surface of catalysts, which were estimated using the XPS, were higher, than the nominal molar ratios (
Table 1). The modification of oxides by sulfur species led to the significant decrease of surface area (from around 30% in the case of pure zirconia to around 45% in pure ceria) and the increase of average pore diameter (excluding Ce
xZr
yO
z(1:4) and ZrO
2).
It suggests that hydrogen sulfide was adsorbed mainly in the internal pores of materials. After the modification of materials with gold, the surface area increased to the values similar before the modification with hydrogen sulfide. It can be explained by the partial removal of sulfur species during the thermal treatment of supports after gold loading, and the results of elemental analysis can confirm this phenomena (
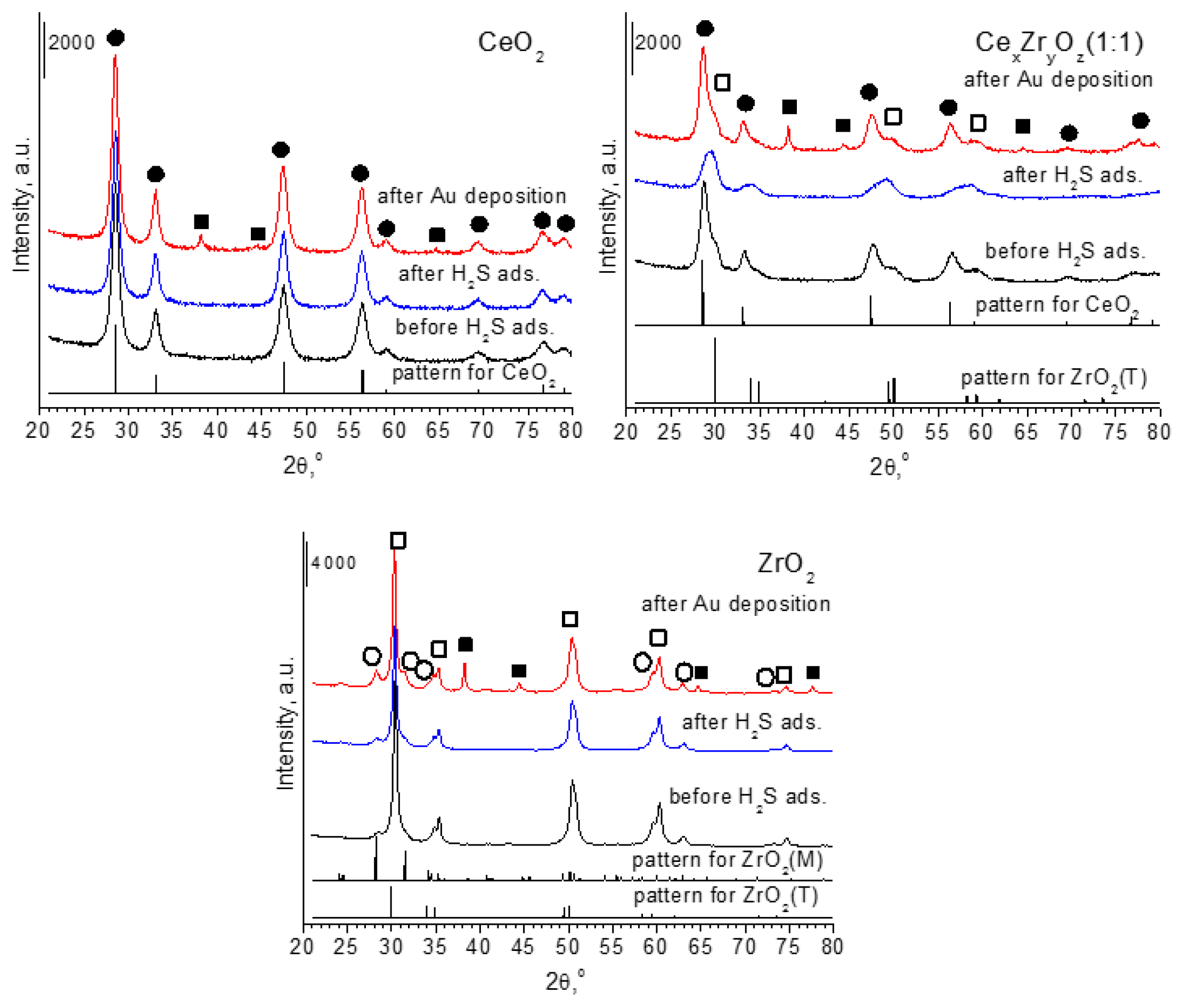
Table 3). The XRD diffraction analysis showed that ceria in all of the samples with cerium were in the form of cubic (JCPDS ICDD PDF Card–00-043-1002) and zirconia in the samples with zirconium species crystallized in the form of monoclinic (JCPDS ICDD PDF Card–00-007-0343) or tetragonal (JCPDS ICDD PDF Card–00-042-1164) (
Figure 1).
It is worth to note, that very small changes in the XRD patterns are observed after the modification of oxides with hydrogen sulfide, because the position and intensities of reflections are very similar before and after the treatment of hydrogen sulfide (excluding CexZryOz(1:1)).
It is worth to note, that the XRD method can be used to estimate the size of crystal phase of particles, which are larger than 5 nm. It means that particles, which are smaller than 5 nm, cannot give any reflections in the XRD patterns. The reflections due to metallic gold particles were observed in the XRD patterns recorded for all of the samples that are modified with this metal (
Figure 1). The average size of metallic gold particles was calculated using the Scherrer’s formula (
Table 2) [
47]. The results show that the increase of specific surface area (
Table 4) and cerium content (
Table 1) had a positive effect on the decrease of the average size of gold particles, but this correlation was not linear, because gold particles were much smaller in the case of Au/Ce
xZr
yO
zS
(2−z)(1:2), than Au/Ce
xZr
yO
zS
(2−z)(1:1) (in the case of gold particles estimated using the Scherrer’s formula [
47]). It can be explained by the changes in the average size of pore diameter (
Table 4). The decrease of pore diameters favored the agglomeration of gold on the external surface of catalysts. The comparison of gold content in the bulk and on the external surface of materials shows that gold was preferentially located on the external surface in all of the catalysts (
Table 2), especially in Au/ZrO
zS
(2−z).
The TEM images confirmed the presence of metallic gold particles in the materials that were modified with gold and the mesoporous structure of the obtained catalysts (
Table 2 and
Figure S1). The results that were obtained using TEM show that the increase of zirconia content in mixed cerium-zirconium oxides had positive effect on the decrease of the average gold particles, and a better dispersion of gold particles (from 5.3 nm in Au/Ce
xZr
yO
zS
(2−z)(1:1) to 4.7 nm in Au/Ce
xZr
yO
zS
(2−z)(1:4)). It can suggest that the direct contact gold, sulfur, and zirconium species can protect gold particles in their agglomeration. It means that the size of gold particles loading on ceria, zirconia, and mixed cerium-zirconium oxides can be controlled by the chemical composition of supports.
3.2.2. The Oxidation State of Metal Species
The main form of cerium species on the external surface of studied samples was Ce
4+ (
Table 5). The occurred differences between the catalysts can be explained by the partial reduction of cerium species (from Ce
4+ to Ce
3+) during the flow of inert gas (helium) before the modification of materials with hydrogen sulfide. The phenomenon of cerium species reduction during the activation of catalysts in the flow of inert gas was explained in earlier studies [
32]. The presence of zirconium species in the support led to the easier reduction of cerium species in the mixed oxides before their modification with sulfur and gold species. In the case of mixed cerium-zirconium oxides that were modified with sulfur species, the increase of zirconium content promoted also the reduction of cerium species.
The distribution of metallic gold (Au
0) species that were estimated using the XPS method was higher in the catalysts based on mixed cerium-zirconium oxides than pure ceria or zirconia (
Table 5). It can suggest the presence of synergetic effect supported on the electron interaction between cerium and zirconium atoms, which led to the reduction of gold species. In the case of pure ceria or zirconia modified with sulfur and gold, the distribution of anionic gold species on the external surface of metallic gold particles ((Au
0)
δ−) was higher than in the mixed cerium-zirconium oxides (
Table 5 and
Table 6). It is worth to note, that in earlier study [
32], gold loading on the external surface of ceria, zirconia, and mixed cerium-zirconium oxides was in the form of metallic (Au
0) and cationic species (Au
δ+). The anionic species ((Au
0)
δ−) were current only in the bimetallic copper-gold catalysts [
32]. It suggests that the sulfur species can play the same role as copper species in the bimetallic copper-gold catalysts. Sulfur species (e.g., as like thiol groups -SH) can reduce gold species, and they are oxidized to sulfate groups (e.g., as like sulfonic groups –SO
3).
The results of TG-DTA, XPS, and ATR-FTIR measurements confirmed the presence of both groups in the materials modified with hydrogen sulfide. It is worth to note that the mixed oxides modified with sulfur were characterised also by the lower values of stoichiometric coefficient of oxygen in chemical formulas estimated using XPS than pure ceria or zirconia (
Table 4).
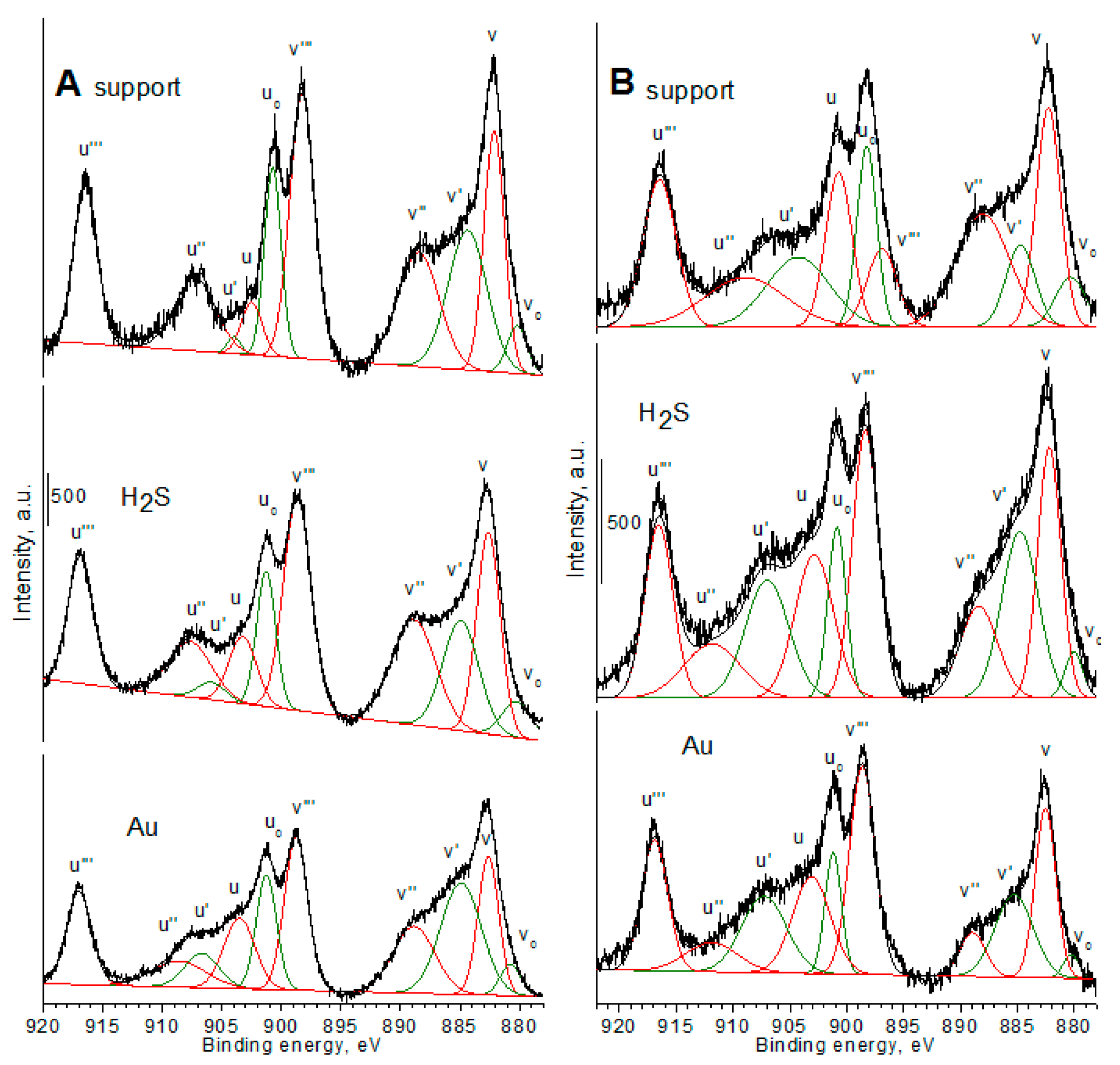
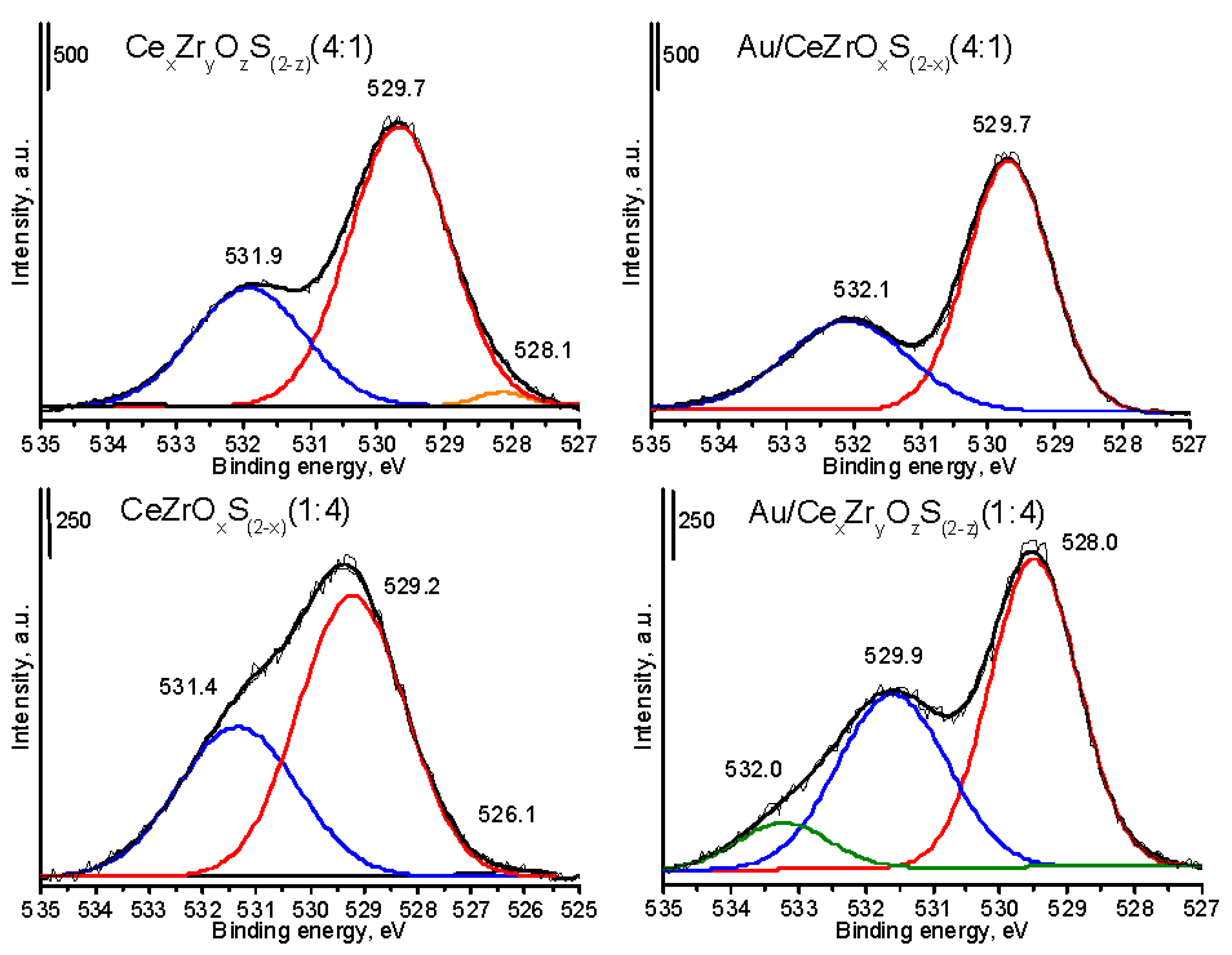
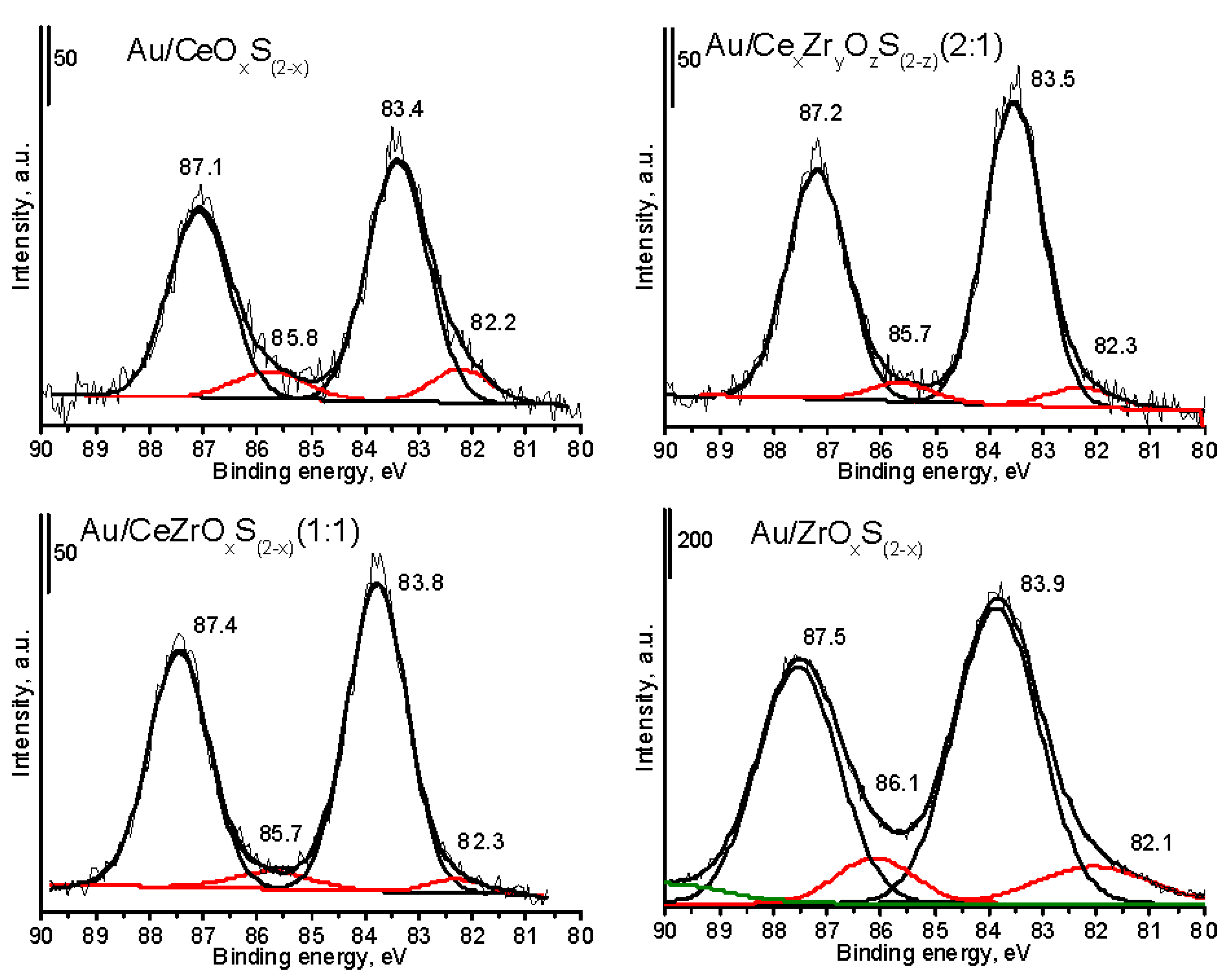
The XPS spectra (
Figure 2,
Figure 3 and
Figure 4 and
Figures S2–S5) were recorded for the assessment of oxidation state of chemical elements, and proved the changes in the contents of oxygen and sulfur. The increase of sulfur content (
Table 3) led to the decrease of gold content on an external surface (
Table 2). The comparison of zirconium and sulfur contents showed that the growth of zirconium concentration on external surface led to the decrease of sulfur on the surface of materials. The correlations between sulfur content on square meter of materials or cubic meter of pore volume of catalysts and the molar ratio of oxides for selected surface parameters were shown in
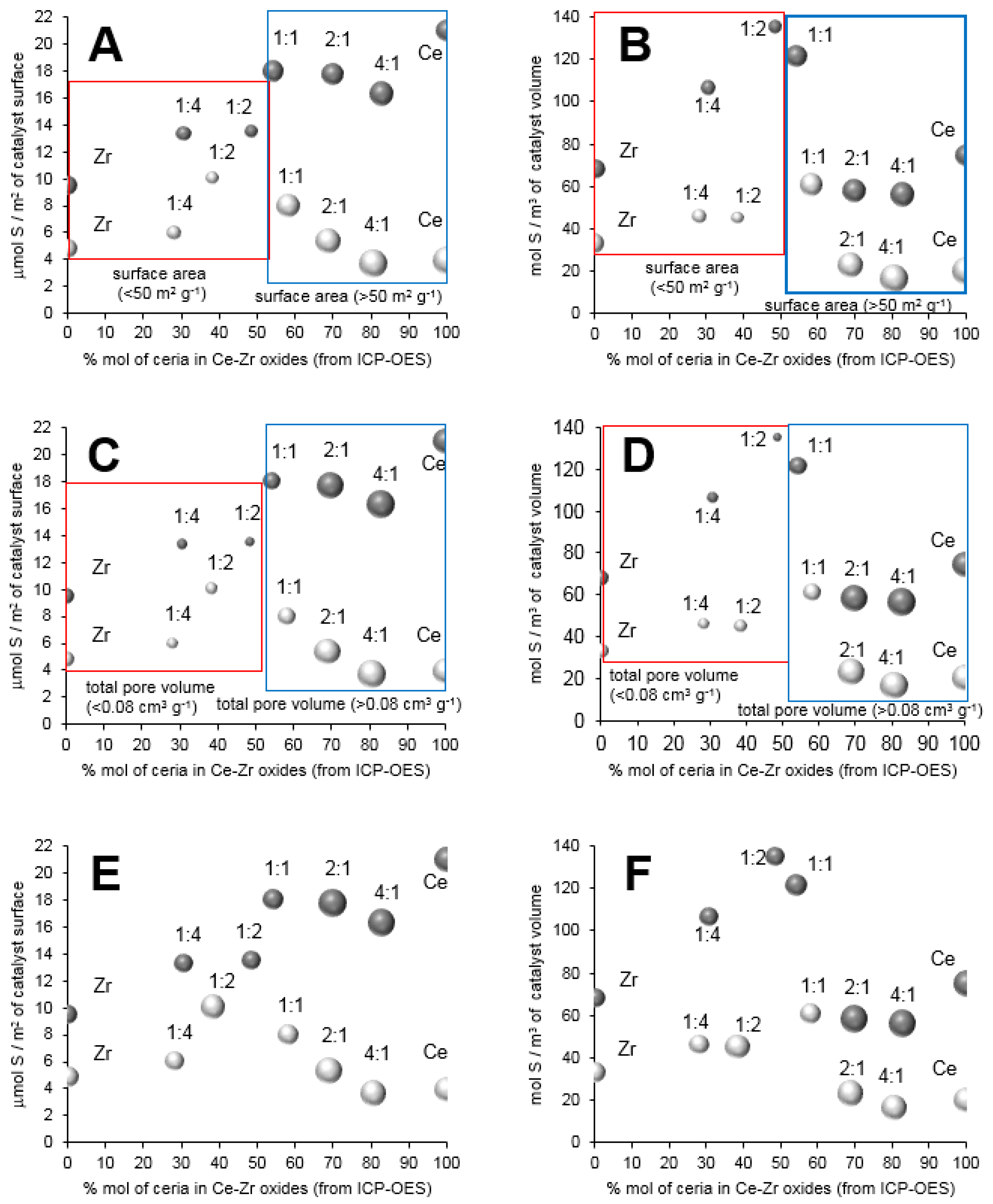
Figure 5.
The molar ratio between ceria and zirconia around 1 in mixed oxides led to the highest sulfur content (on the external surface) on square meter of materials that were modified with gold species. The lack of linear correlation between sulfur content and cerium or zirconium content proved that the chemical composition of supports influenced on the surface parameters. This parameter could play a crucial role in the possibility of the migration of gold species to the bulk of materials, and the agglomeration of sulfur species on the external surface during the thermal adsorption of hydrogen sulfide.
The largest surface specific area of materials were measured for the materials based on pure ceria CeO
2 and mixed oxide Ce
xZr
yO
z(4:1). The decrease of specific surface area below 30 m
2 g
−1 and the average pore size below 9.0 nm (in the case of Ce
xZr
yO
zS
(2−z)(1:2) and Ce
xZr
yO
zS
(2−z)(1:4)) led to the increase of gold content (above 3.0 at % and sulfur below 3.5 at %). If the specific surface area was around 40 m
2 g
−1 and the average pore size was around 6.0 nm (in ZrO
zS
(2−z)), then the gold and sulfur contents on the external surface were higher. It can suggest that the presence of zirconium species had positive effect on the migration of gold and sulfur species on the external surface. The XP spectra for O 2p region showed that the chemical composition of supports influenced on the distribution of the bands due to oxygen species (
Figure 3). The high cerium content in the mixed cerium-zirconium oxides led to the shift of a main band to the lower values of binding energy (BE) and the lower distribution of the area of band at 531–532 eV. It can suggest a stronger interaction between oxygen and zirconium species and easier removal from the crystal structure of oxygen atoms, which were in the neighborhood of cerium atoms. The XP spectra of Au 4f region recorded for selected gold catalysts (
Figure 4) showed that the presence of cerium can lead to the shift of the bands in the XP spectra to the lower values of binding energy (
Table 6).
It can be explained by the stronger interaction between cerium and gold species and the easier reducibility of cerium species, as confirmed by TPR by H2 study.
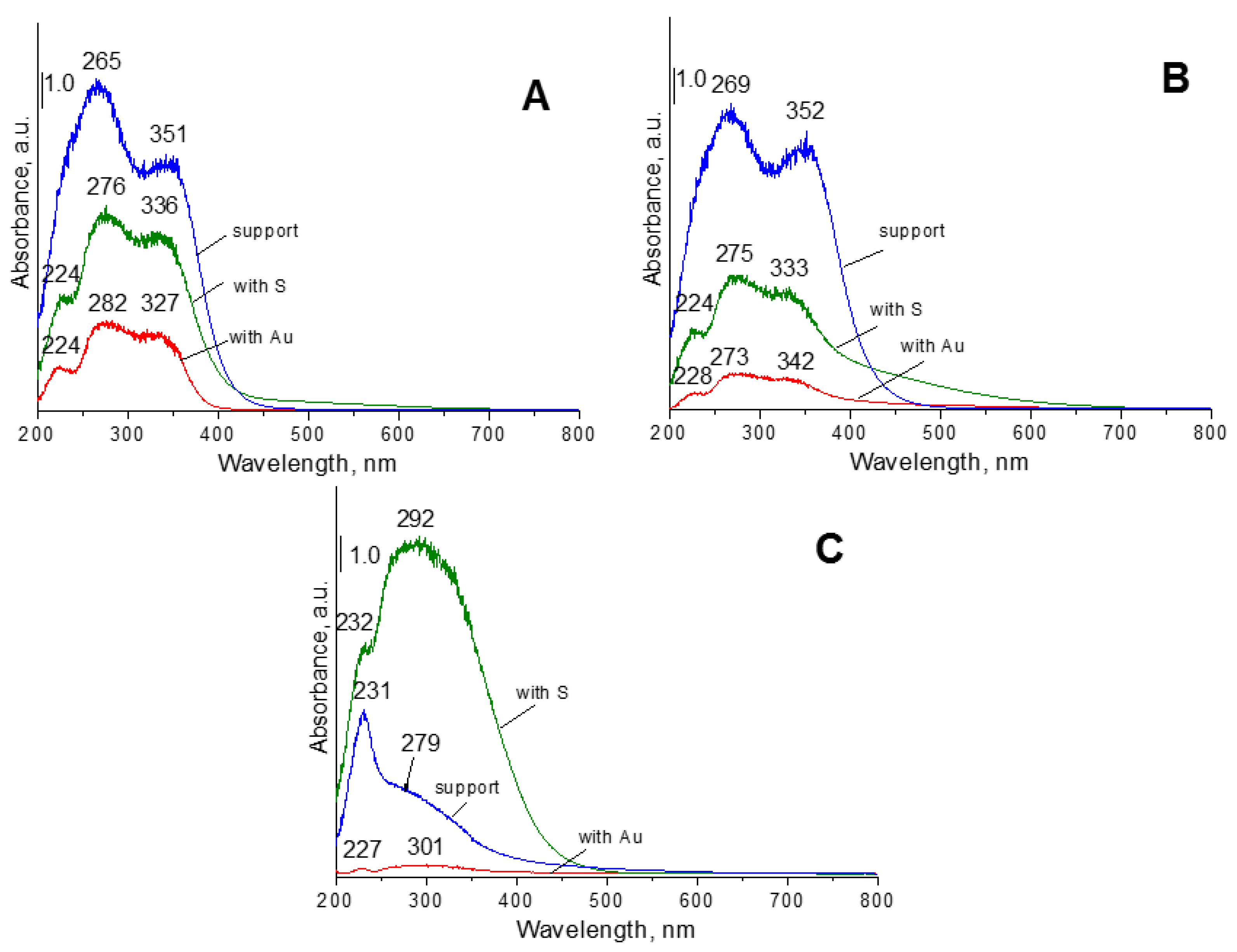
UV-vis spectroscopy is a method, which can be used to the estimation of oxidation state and the coordination of metals.
Figure 6 and
Figure S6 display the UV-vis spectra of selected calcined catalysts that are studied in this work. In the case of samples with cerium species, the bands in the range of 200–400 nm can be due to the ligand-metal charge transfer (LMCT) between oxygen ions (O
2−) to cerium cations (Ce
4+). According to the literature data [
21,
52], the position of a band coming from ligand to metal charge transfer depends on the ligand field symmetry surrounding the cerium center.
It can explain why the charge transfer from oxygen ions to cerium cations requires higher energy of tetra-coordinated cerium cations than that of e.g., hexa-coordinated or octa-coordinated species. In the UV-vis spectra recorded for the materials with cerium species are seen tree bands at around 225, 265, and 350 nm in the UV-vis spectra recorded for the catalysts with cerium (
Figure 6A,B) can be assigned to the different LMCT: in crystalline ceria (from O
2− to Ce
4+), from oxygen species O
2− to cerium cations Ce
4+ in tetrahedral coordination, and with a higher coordination number than four, respectively. The shape of the UV-vis spectra recorded for samples with cerium species after modification with hydrogen sulfide changed and the intensity of the bands decreased, but the band at around 225 nm is not covered by the band at around 265 nm.
The UV-vis spectra recorded for samples based on zirconia (
Figure 6C) show a band around 230 and 280 nm, which can be due to the LMCT from oxygen ions (O
2−) to zirconium ions (Zr
4+) with octahedral configuration in zirconia crystallites. In the case of catalysts based on pure zirconia, after its modification with sulfur species, the intensity of band at around 292 nm increased significantly. For support with sulfur, the band at ca. 220 nm is visible. It can suggest the presence of the new kind of coordination of zirconium species by sulfur and/or oxygen atoms.
The discussed UV-vis region (200–400 nm) is also typical of the LMCT of gold species (Au
3+ and Au
+ ions) with ligands [
52,
53,
54,
55,
56,
57,
58,
59], or for the absorption bands due to the transition of electrons between molecular orbitals of the few-atomic clusters Au
n (
n < 10). It means that this region cannot be used to the identification of the form of cationic gold species. The range of 500–580 nm can be applied to the estimation of metallic gold nanoparticles (
Figure S6). The bands in this region can be due to the optical absorption of the light excited oscillating conductivity electrons of metallic gold particles [
60,
61]. The positions of bands above 500 nm can give information about the kind of metallic gold species, e.g., a band at 567 nm (
Figure S6) can be due to the plasmon resonance in metallic gold particles [
62]. This band was found only in the catalyst that was modified with gold based on pure ceria. It can suggest that the composition of the support can determine the presence and the position of this band.
It is worth to note that the modification of the catalysts with gold led to the decrease of the intensity of these bands, which were assigned to cerium species. It can confirm that the decrease of cerium species contents on the external surface upon the modification of catalysts with gold, which was documented by XPS study.
3.2.3. The Form of Sulfuric Species
The XPS, TG-DTA, TPR by H2, and ATR-FTIR measurements were applied to the identification of the form of sulfuric species.
The comparison of selected XP spectra recorded for the region of sulfur species (S 2p) (
Figure S1) showed the shift of binding energy (BE) to higher values after the modification of samples with gold species (
Table 5). According to the literature data [
63], the bands of XPS spectra at around 168 eV could be correspond to sulfur species as oxidized sulfur groups. It was reported [
64] that the bands at 167.4 and 168.7 eV can be attributed to sulfur in sulfonyl (S–O) and/or sulfate (S=O) groups, respectively, and these groups are generated in the presence of oxygen. It was published in [
65,
66,
67] that the band at 168.1 eV can be assigned to the binding energy of sulfur in sulfonic groups (–SO
3H). The results of BE present in
Table 5 showed that after the modification of oxides with hydrogen sulfide, sulfonyl, and/or sulfonic groups could be located on the external surface of materials. The increase of zirconium content favored the presence of sulfonyl groups (S–O) and higher cerium content led to the growth of the distribution of sulfate groups (S=O). These differences can be explained by the changes in the coordination of metal (cerium and zirconium) species and the oxidation state of cerium species. The loading of gold on the surface of samples with high zirconium content changed the main form sulfonyl groups (S–O) to sulfate groups (S=O). In the mixed oxides [Ce
xZr
yO
zS
(−z)(2:1); Ce
xZr
yO
zS
(2−z)(1:1); Ce
xZr
yO
zS
(2−z)(1:2)] before and after their modification with gold, sulfonic groups were the main form of sulfur species on the external surface. These materials were characterised by the molar ratio between CeO
2 and ZrO
2 in the range 0.62 and 2.20 (according to the XPS study), and it suggests the positive effect of interaction between cerium and zirconium species on the creation of sulfonic groups.
The oxides before and after their modification with hydrogen sulfide and gold species were studied using TG-DTA in the flow of air in the order to know the form of sulfur species on the surface of catalysts and to estimate their thermal stability. The analysis of TGA and DTA profiles of samples that were modified with sulfur showed that this chemical element was in several forms (
Figure S7). In the supports modified with sulfur, in which the nominal (assumed) content of ceria was higher than zirconia [CeO
zS
(2−z), Ce
xZr
yO
zS
(2−z)(4:1) and Ce
xZr
yO
zS
(2−z)(2:1)] were observed two peaks. The first peak at lower temperature was more intense than the second at higher temperature. The first peak, at around 500 K, could be described to thiol groups (-SH), and the second peak at around 850 K could be due to sulfonic groups (–SO
3H). In the case of samples with higher amount of zirconium after the catalysts modification with sulfur species (Ce
xZr
yO
zS
(2−z)(2:1), (Ce
xZr
yO
zS
(2−z)(1:1), (Ce
xZr
yO
zS
(2−z)(1:2) and Ce
xZr
yO
zS
(2−z)(1:4)) were observed two peaks at around 500 and 600 K, which could be due to two forms of thiol groups being located on the external surface and in the bulk of samples, respectively. In the case of ZrO
zS
(2−z), only one intense peak in the DTA profile was seen, which could be due to sulfonic groups on the external surface. The results of DTA confirmed that the chemical composition of supports had influence on the chemical adsorption of hydrogen sulfide.
The samples were heated in the flow of inert gas (helium) before the adsorption of hydrogen sulfur, and then the modification of sulfur species was performed in the flow of the mixture of gases (helium and hydrogen sulfur). The lack of oxygen in the flow of gases before and during the modification of samples can suggest that the mechanism of modification thiol groups to sulfonic groups which was based on the exchanging between hydrogen atoms from hydrogen sulfide and oxygen atoms from the structure of oxides. This mechanism assumes the presence of oxygen vacancies in the structure of materials created during the activation of materials in the flow of inert gas and it is according to the literature data [
32]. The comparison of the stoichiometric formulas of oxides before and after their modification with sulfur species confirms lower oxygen distribution on the external surface after the adsorption of hydrogen sulfur (
Table 4). The increase of signals in the temperature range of 500–1000 K with the maximum at around 700 K was observed in the case of selected TGA profiles. These wide peaks were seen for Ce
xZr
yO
zS
(2−z)(2:1), Ce
xZr
yO
zS
(2−z)(1:1), Ce
xZr
yO
zS
(2−z)(1:2), and Ce
xZr
yO
zS
(2−z)(1:4). For the same samples, were observed peaks at around 600 K in the DTA profiles, which can be due to the thiol groups in the bulk materials (
Figure S7).
The catalysts were characterised using a temperature programmed reduction (TPR) by hydrogen in the order to know the redox properties of samples. The TPR profiles were shown in
Figure S8. The maximums of hydrogen consumption depend on the chemical composition of samples and the size of crystals. The TPR profile of pure ceria shows the consumption of hydrogen in the wide temperature range of 450–700 K, with the maximum at 522 K, which can be due to the reduction of cerium species (from Ce
4+ to Ce
3+). The consumption of hydrogen at this temperature can prove the presence of small cerium oxides particles, which are smaller than in the mixed cerium-zirconium oxides. The broad temperature range of hydrogen consumption suggests that the crystals of ceria are characteristic of the broad range of sizes. The modification of ceria with hydrogen sulfide led to the disappearance of hydrogen consumption at lower temperature. After medication of ceria with gold and sulfur species, a very weak peak is observed in the broad temperature range (500–900 K). It can suggest that sulfur and gold species covered the external surface of supports, and the oxidized cerium species (Ce
4+) were not able to reduce to the reduced form of cerium (Ce
3+). The addition of zirconium led to the increase of temperature, in which hydrogen is consumed or the disappearance of this process. It means that the presence of zirconium leads to the protection of cerium species against their reduction at lower temperature.
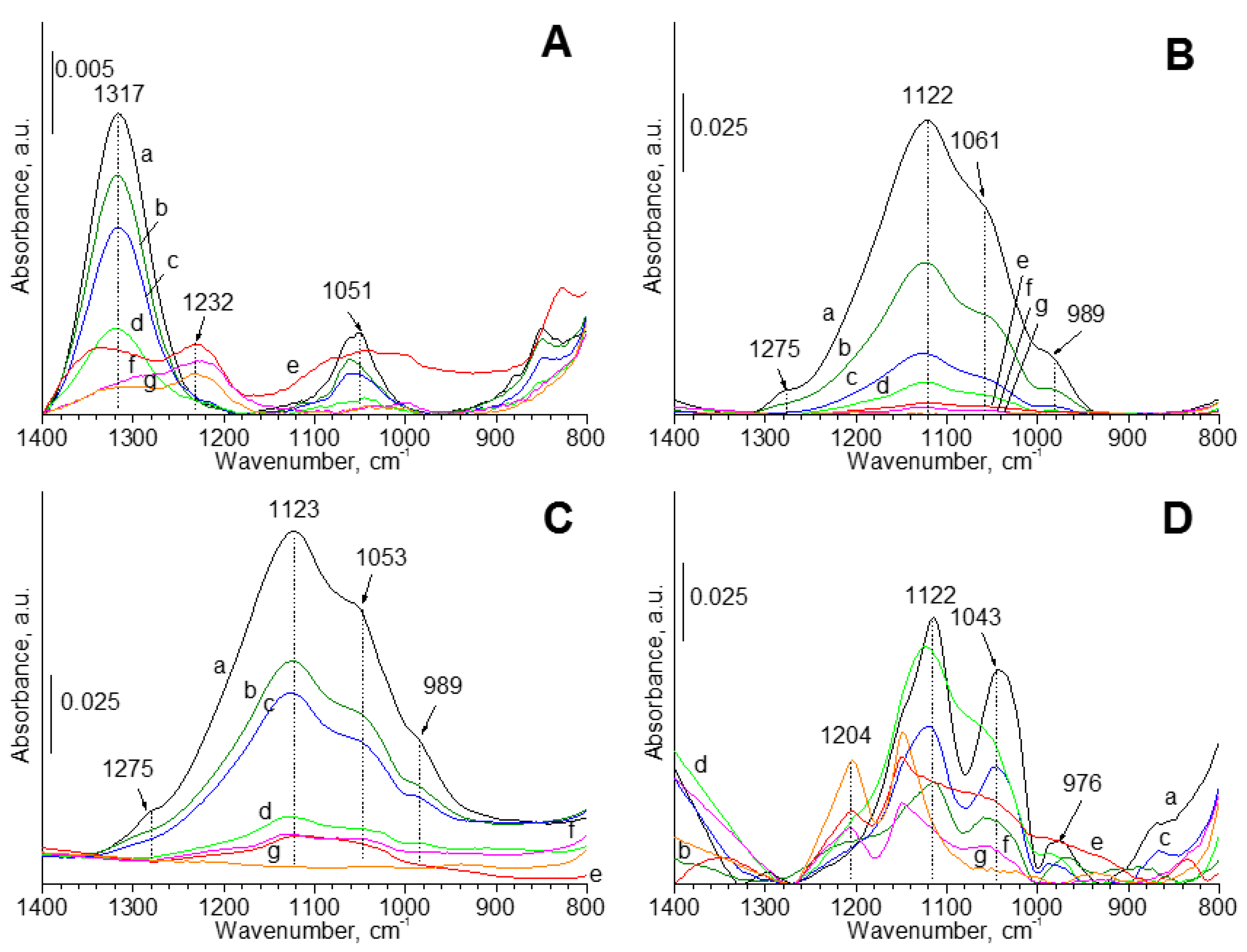
The ATR-FTIR spectra were recorded at room temperature in the atmosphere of air for all of the samples (
Figure 7). In the case of samples that are modified with hydrogen sulfide, four bands in the region of 1400–800 cm
−1 were observed. The most intense band at 1122 cm
−1 was not observed on the spectra of pure oxides. The intensity of this band was also smaller in the case of samples modified with sulfur and gold. The increase of cerium content had a positive effect on the increase of intensity of this band. The sulfur content increased with the growth of cerium content, so it can suggest that the band at 1122 cm
−1 can be assigned to the sulfur species. It has been reported [
68] that this band can be assigned to the symmetric stretching band of the sulphone groups, as like (–O–SO
2).