The Effects of Tai Chi and Qigong on Immune Responses: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
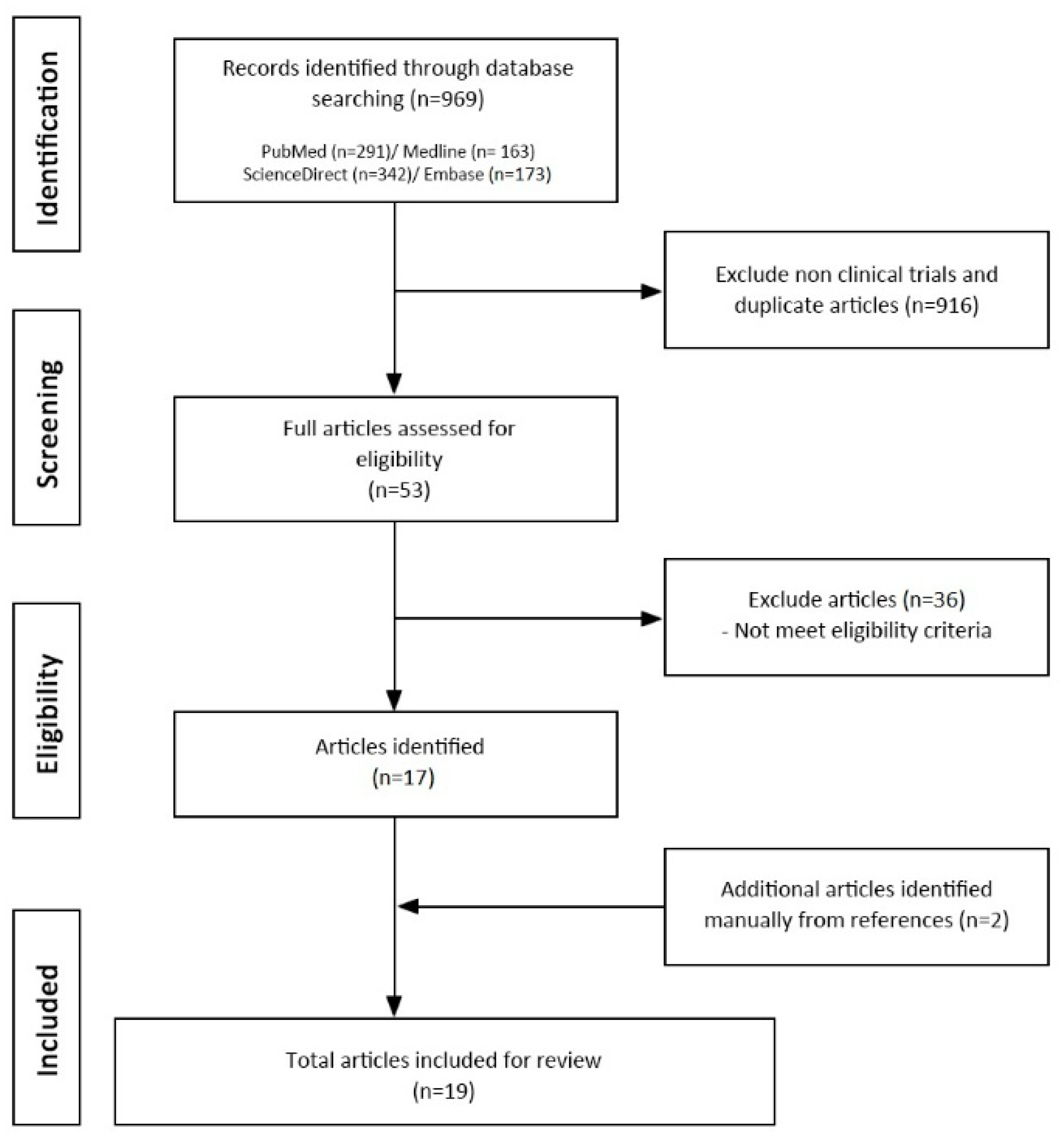
2. Methods
2.1. Data Analysis
2.2. Quality Assessment of Original Papers
3. Results
3.1. Characteristics of Clinical Studies and Quality of Evidence
3.2. Outcomes on the Immune System and Inflammation Associated Biomarkers
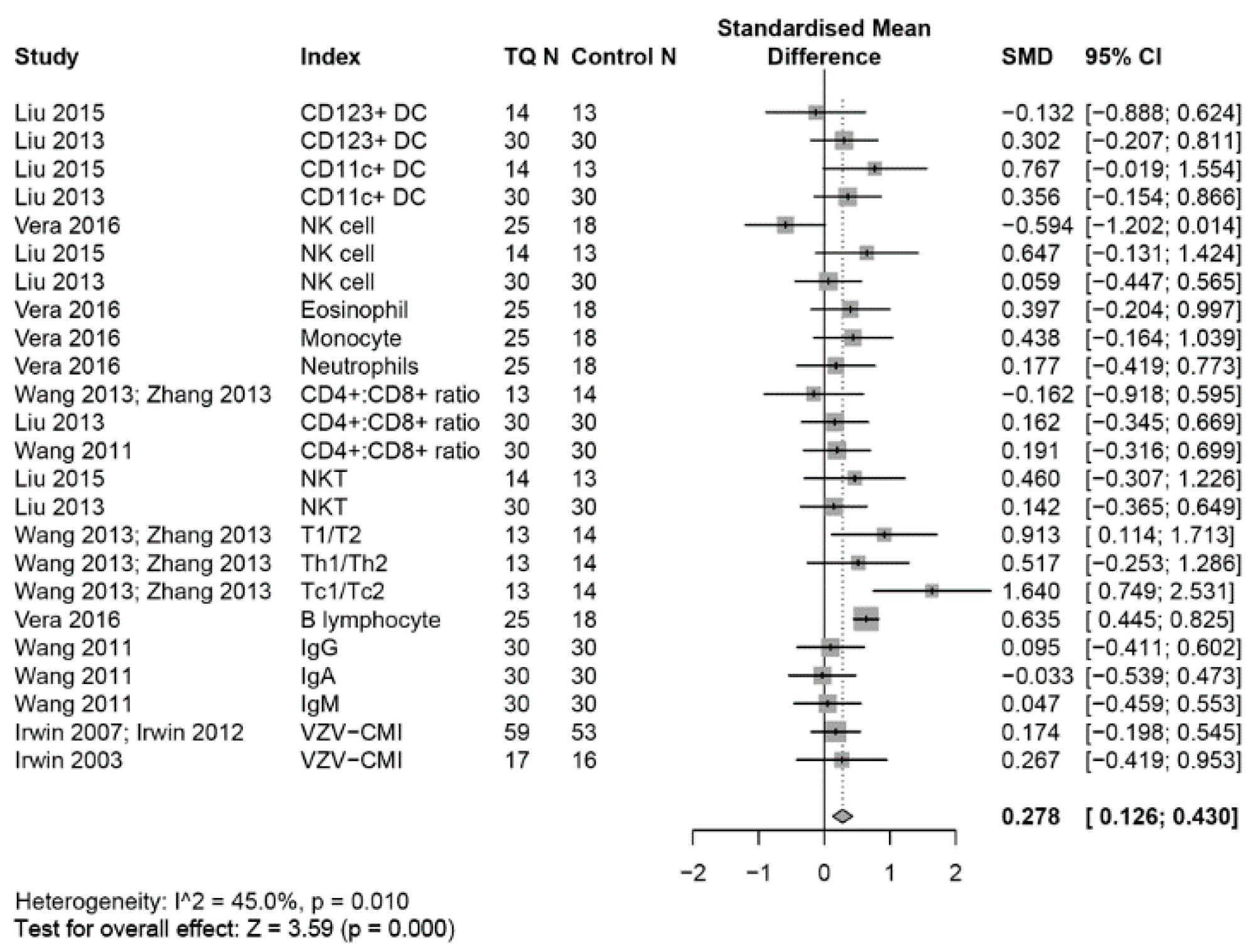
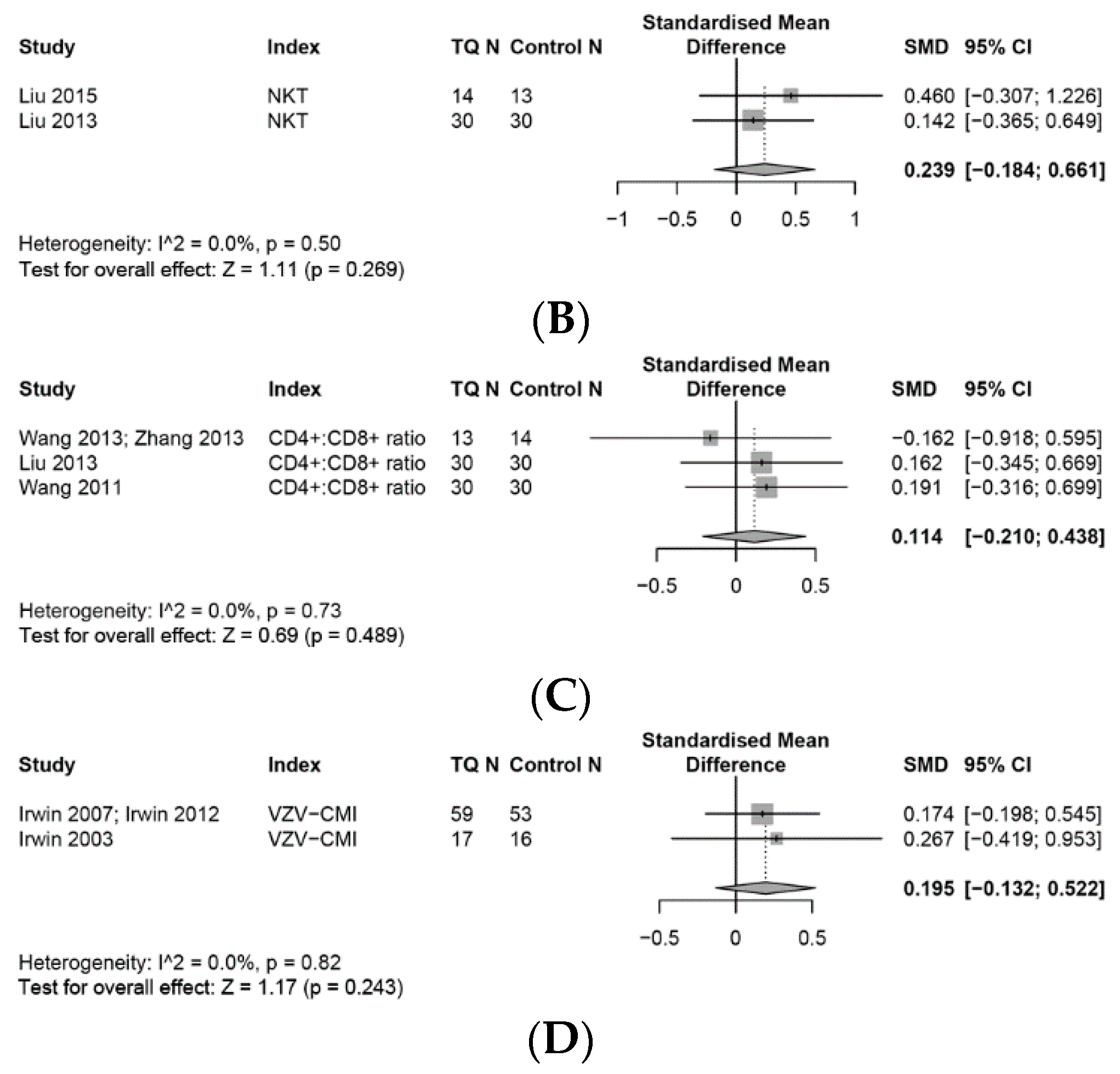
3.3. Outcomes on the Immune System
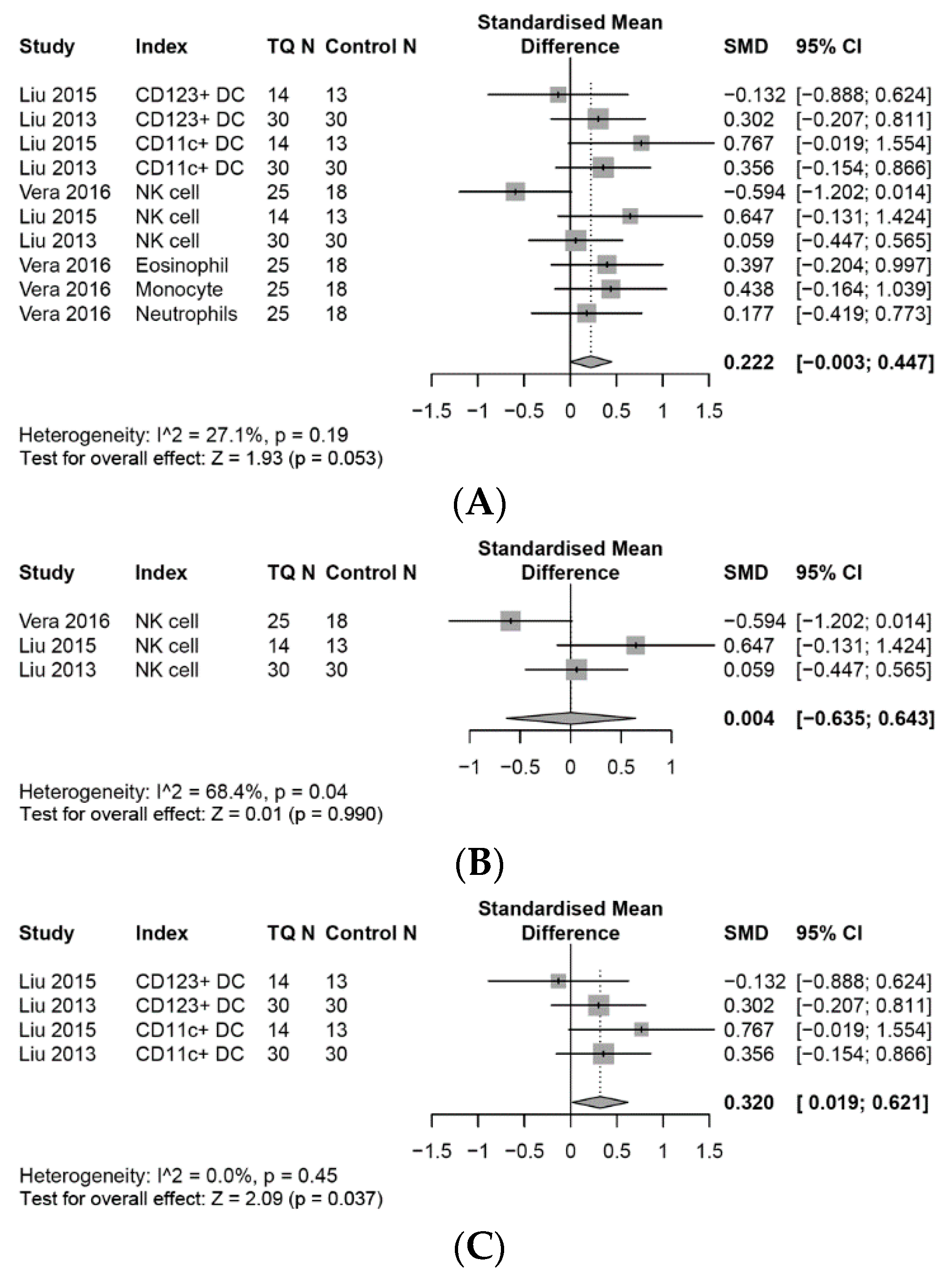
3.4. Effects on the Innate Immune System
3.4.1. NK Cells
3.4.2. Dendritic Cells (DCs)
3.5. Other Innate Immune Cells
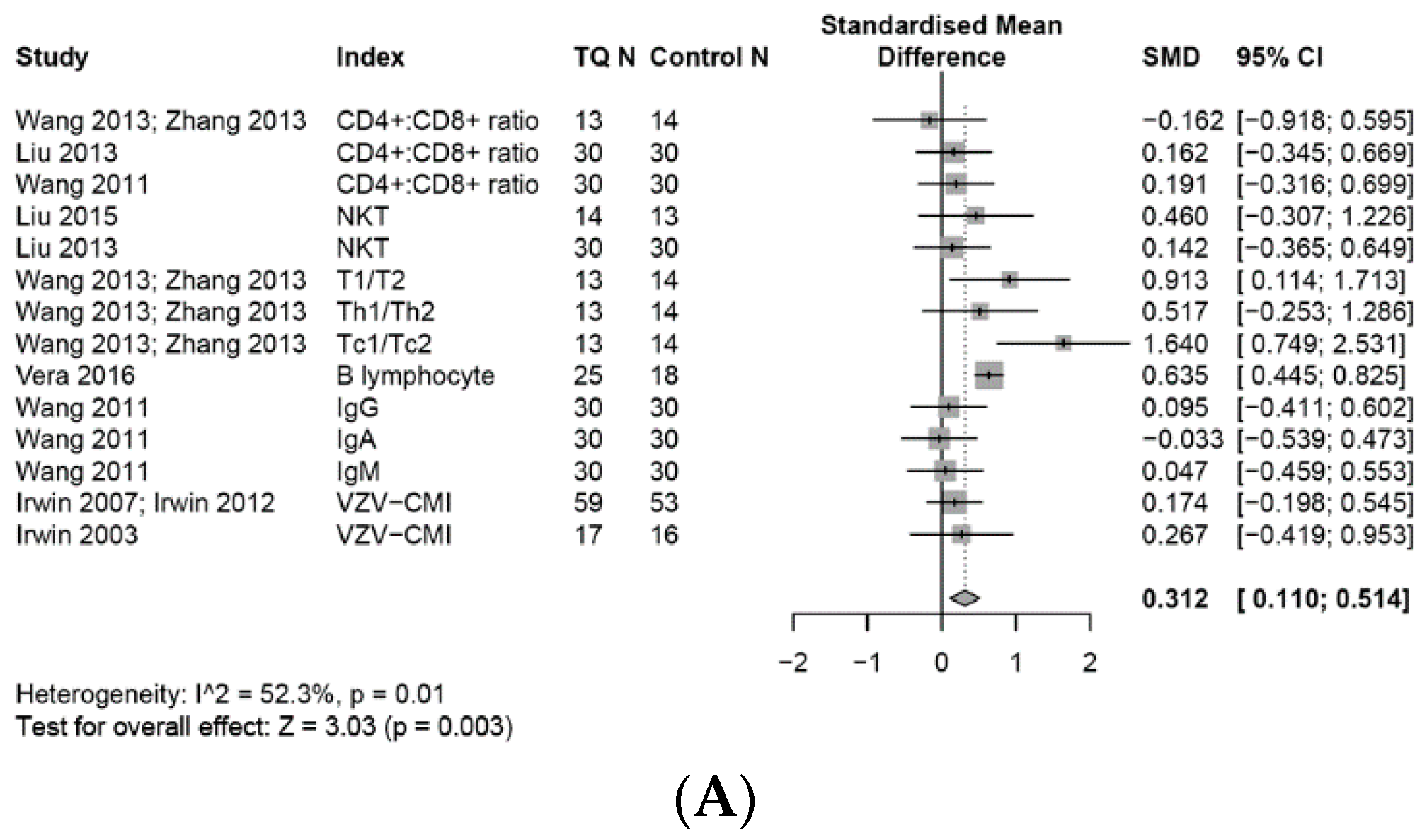
3.6. Effects on the Adaptive Immune System
3.6.1. Adaptive Immune Cells
3.6.2. T Cell Associated Adaptive Immune Cells
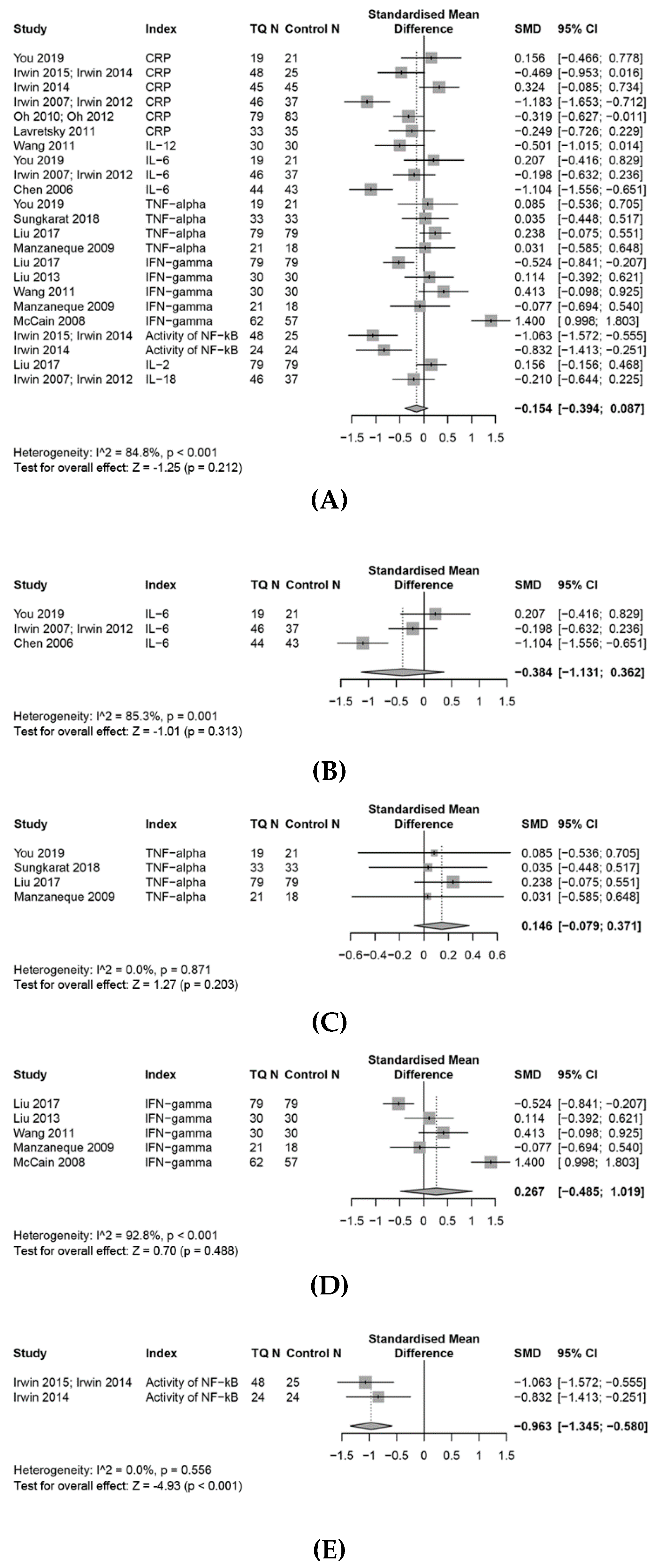
3.7. Effects on the Inflammation Response
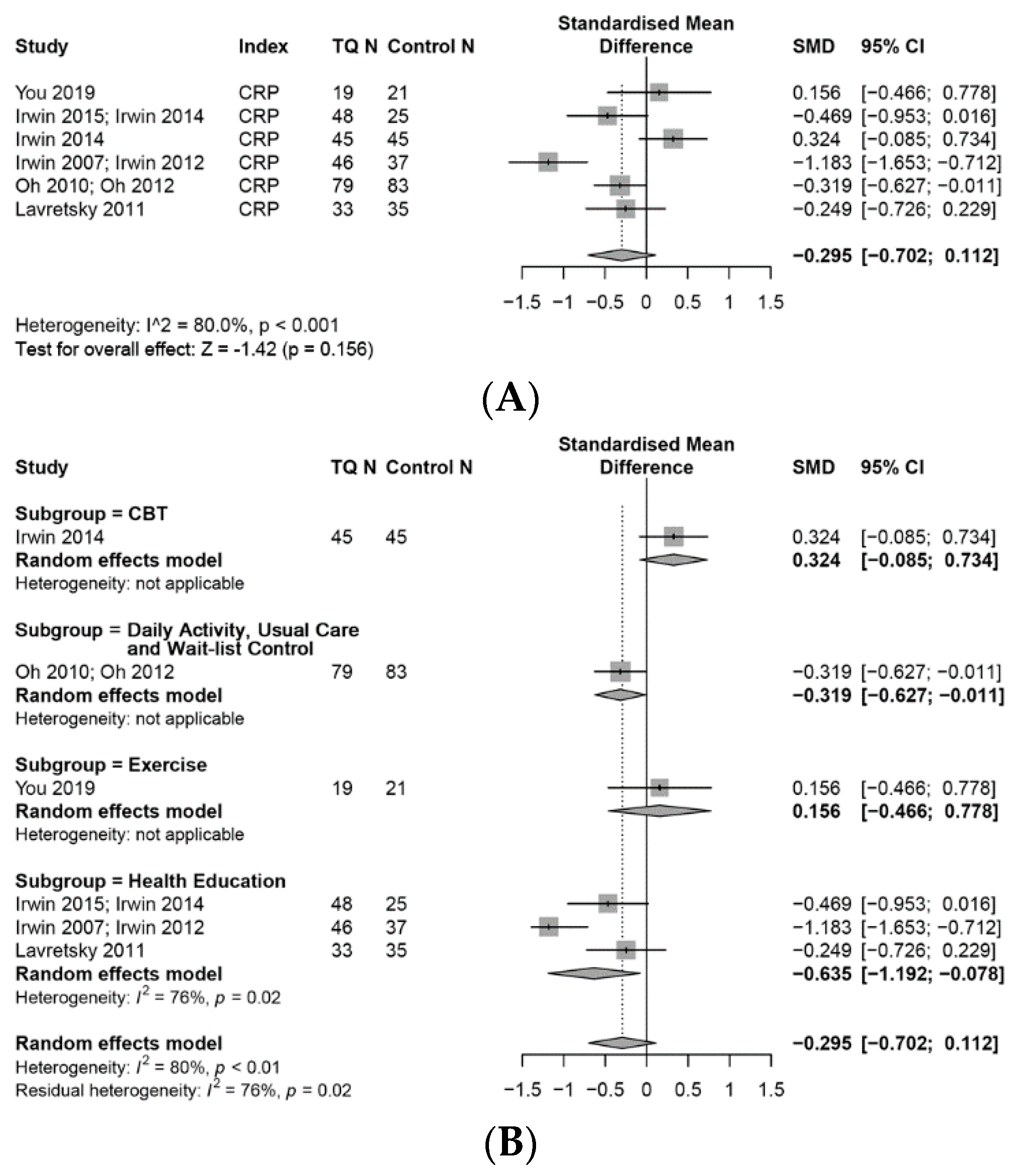
3.7.1. CRP
3.7.2. IL-6
3.7.3. TNF-α
3.7.4. INF-γ
3.7.5. Effects on Other Pro-Inflammatory Biomarkers
3.8. Effects on Anti-Inflammatory Biomarkers
IL-4
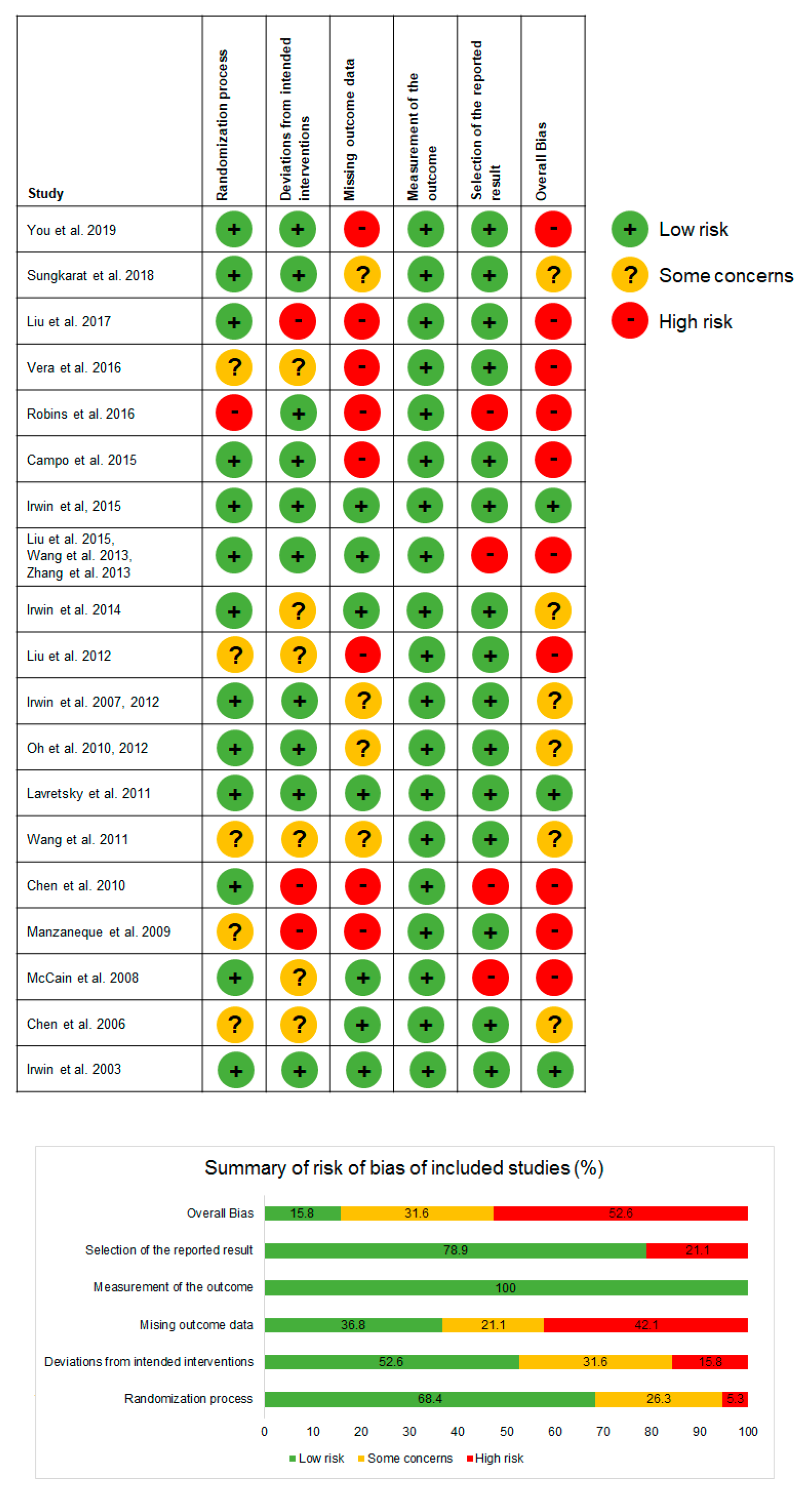
3.9. Assessment of Risk of Bias
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Bennett, J.M.; Reeves, G.; Billman, G.E.; Sturmberg, J.P. Inflammation-Nature’s way to efficiently respond to all types of challenges: Implications for understanding and managing “the epidemic” of chronic Diseases. Front. Med. 2018, 5, 316. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed]
- Kritas, S.K.; Ronconi, G.; Caraffa, A.L.; Gallenga, C.E.; Ross, R.; Conti, P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost Agents 2020, 34, 10–23812. [Google Scholar]
- Zhang, W.; Zhao, Y.; Zhang, F.; Wang, Q.; Li, T.; Liu, Z.; Wang, J.; Qin, Y.; Zhang, X.; Yan, X.; et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin. Immunol. 2020, 214, 108393. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef]
- Jakobsson, J.; Malm, C.; Furberg, M.; Ekelund, U.; Svensson, M. Physical Activity During the Coronavirus (COVID-19) Pandemic: Prevention of a decline in metabolic and immunological functions. Front. Sports Act. Living 2020, 2, 57. [Google Scholar] [CrossRef]
- Hong, K.S.; Lee, K.H.; Chung, J.H.; Shin, K.-C.; Choi, E.Y.; Jin, H.J.; Jang, J.G.; Lee, W.; Ahn, J.H. Clinical features and outcomes of 98 patients hospitalized with SARS-CoV-2 infection in Daegu, South Korea: A brief descriptive study. Yonsei Med. J. 2020, 61, 431–437. [Google Scholar] [CrossRef]
- Liu, K.; Chen, Y.; Lin, R.; Han, K. Clinical features of COVID-19 in elderly patients: A comparison with young and middle-aged patients. J. Infect. 2020, 80, e14–e18. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; The Northwell COVID-19 Research Consortium. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Keilich, S.R.; Bartley, J.M.; Haynes, L. Diminished immune responses with aging predispose older adults to common and uncommon influenza complications. Cell. Immunol. 2019, 345, 103992. [Google Scholar] [CrossRef]
- Thevarajan, I.; Nguyen, T.H.O.; Koutsakos, M.; Druce, J.; Caly, L.; Sandt, C.E.v.d.; Jia, X.; Nicholson, S.; Catton, M.; Cowie, B.; et al. Breadth of concomitant immune responses prior to patient recovery: A case report of non-severe COVID-19. Nat. Med. 2020, 26, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Nikolich-Zugich, J.; Knox, K.S.; Rios, C.T.; Natt, B.; Bhattacharya, D.; Fain, M.J. SARS-CoV-2 and COVID-19 in older adults: What we may expect regarding pathogenesis, immune responses, and outcomes. GeroScience 2020, 42, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Gao, Y.; Wang, G.; Song, G.; Liu, S.; Sun, D.; Xu, Y.; Tian, Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 2020, 17, 533–535. [Google Scholar] [CrossRef]
- Wang, X.; Xu, W.; Hu, G.; Xia, S.; Sun, Z.; Liu, Z.; Xie, Y.; Zhang, R.; Jiang, S.; Lu, L. SARS-CoV-2 infects T lymphocytes through its spike protein-mediated membrane fusion. Cell. Mol. Immunol. 2020, 7, 1–3. [Google Scholar] [CrossRef]
- di Mauro, G.; Cristina, S.; Concetta, R.; Francesco, R.; Annalisa, C. SARS-Cov-2 infection: Response of human immune system and possible implications for the rapid test and treatment. Int. Immunopharmacol. 2020, 84, 106519. [Google Scholar] [CrossRef]
- Febbraio, M.A. Health benefits of exercise—More than meets the eye! Nat. Rev. Endocrinol. 2017, 13, 72–74. [Google Scholar] [CrossRef] [PubMed]
- Black, D.S.; Slavich, G.M. Mindfulness meditation and the immune system: A systematic review of randomized controlled trials. Ann. N. Y. Acad. Sci. 2016, 1373, 13–24. [Google Scholar] [CrossRef]
- Nieman, D.C.; Wentz, L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2019, 8, 201–217. [Google Scholar] [CrossRef]
- Sellami, M.; Gasmi, M.; Denham, J.; Hayes, L.D.; Stratton, D.; Padulo, J.; Bragazzi, N. Effects of acute and chronic exercise on immunological parameters in the elderly aged: Can physical activity counteract the effects of aging? Front. Immunol. 2018, 9, 2187. [Google Scholar] [CrossRef]
- Birdee, G.S.; Cai, H.; Xiang, Y.-B.; Yang, G.; Li, H.; Gao, Y.; Zheng, W.; Shu, X.O. T’ai chi as exercise among middle-aged and elderly Chinese in urban China. J. Altern. Complement. Med. (N. Y.) 2013, 19, 550–557. [Google Scholar] [CrossRef]
- Lauche, R.; Wayne, P.M.; Dobos, G.; Cramer, H. Prevalence, patterns, and predictors of T’ai Chi and Qigong use in the United States: Results of a nationally representative survey. J. Altern. Complement Med. 2016, 22, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Qiu, P.; Liu, T. Tai Ji Quan: An overview of its history, health benefits, and cultural value. J. Sport Health Sci. 2014, 3, 3–8. [Google Scholar] [CrossRef]
- Lan, C.; Chen, S.-Y.; Lai, J.-S.; Wong, A.M.-K. Tai Chi Chuan in medicine and health promotion. Evid. Based Complementary Altern. Med. 2013, 2013, 502131. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Schmid, C.H.; Iversen, M.D.; Harvey, W.F.; Fielding, R.A.; Driban, J.B.; Price, L.L.; Wong, J.B.; Reid, K.F.; Rones, R.; et al. Comparative Effectiveness of Tai Chi Versus Physical Therapy for Knee Osteoarthritis: A Randomized Trial. Ann. Intern. Med. 2016, 165, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.-C.; Liu, J.; Fu, J.; Yin, H.Y.; Shen, L.; Liu, M.-L.; Lan, L.; Ying, J.; Qiao, X.-L.; Tang, C.-Z.; et al. Effect of Tai Chi Chuan in breast cancer patients: A systematic review and meta-analysis. Front. Oncol. 2020, 10, 607. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhou, R.; Li, K.; Zhu, Y.; Zhang, Z.; Luo, Y.; Luan, R. Effects of tai chi on physiology, balance and quality of life in patients with type 2 diabetes: A systematic review and meta-analysis. J. Rehabil. Med. 2019, 51, 405–417. [Google Scholar] [CrossRef]
- Liu, H.-H.; Yeh, N.-C.; Wu, Y.-F.; Yang, Y.-R.; Wang, R.-Y.; Cheng, F.-Y. Effects of Tai Chi exercise on reducing falls and improving balance performance in parkinson’s disease: A meta-analysis. Parkinson’s Dis. 2019, 2019, 9626934. [Google Scholar] [CrossRef]
- Cheng, C.-A.; Chiu, Y.-W.; Wu, D.; Kuan, Y.-C.; Chen, S.-N.; Tam, K.-W. Effectiveness of Tai Chi on fibromyalgia patients: A meta-analysis of randomized controlled trials. Complement. Ther. Med. 2019, 46, 1–8. [Google Scholar] [CrossRef]
- Kolasinski, S.L.; Neogi, T.; Hochberg, M.C.; Oatis, C.; Guyatt, G.; Block, J.; Callahan, L.; Copenhaver, C.; Dodge, C.; Felson, D.; et al. 2019 American College of Rheumatology/Arthritis Foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 2020, 72, 220–233. [Google Scholar] [CrossRef]
- Kong, L.J.; Lauche, R.; Klose, P.; Bu, J.H.; Yang, X.C.; Guo, C.Q.; Dobos, G.; Cheng, Y.W. Tai Chi for Chronic pain conditions: A systematic review and meta-analysis of randomized controlled trials. Sci. Rep. 2016, 6, 25325. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, L.; Tang, Q.; Wang, F.; Li, Y.; Peng, H.; Wang, S. Tai Chi is effective in delaying cognitive decline in older adults with mild cognitive impairment: Evidence from a systematic review and meta-analysis. Evid. Based Complementary Altern. Med. 2020, 2020, 3620534. [Google Scholar] [CrossRef]
- So, W.W.Y.; Cai, S.; Yau, S.Y.; Tsang, H.W.H. The neurophysiological and psychological mechanisms of Qigong as a treatment for depression: A systematic review and meta-analysis. Front. Psychiatry 2019, 10, 820. [Google Scholar] [CrossRef] [PubMed]
- Bower, J.E.; Irwin, M.R. Mind-body therapies and control of inflammatory biology: A descriptive review. Brain Behav. Immun. 2016, 51, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bottaccioli, A.G.; Bottaccioli, F.; Carosella, A.; Cofini, V.; Muzi, P.; Bologna, M. Psychoneuroendocrinoimmunology-based meditation (PNEIMED) training reduces salivary cortisol under basal and stressful conditions in healthy university students: Results of a randomized controlled study. EXPLORE 2020, 16, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Półrola, P.; Wilk-Franczuk, M.; Wilczyński, J.; Nowak-Starz, G.; Góral-Półrola, J.; Chruściński, G.; Bonisławska, I.; Pedrycz, A.; Żychowska, M. Anti-inflammatory effect on genes expression after four days of Qigong training in peripheral mononuclear blood cells in healthy women. Ann. Agric. Environ. Med. 2018, 25, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Klein, P.J.; Baumgarden, J.; Schneider, R. Qigong and Tai Chi as Therapeutic Exercise: Survey of Systematic Reviews and Meta-Analyses Addressing Physical Health Conditions. Altern. Ther. Health Med. 2019, 25, 48–53. [Google Scholar]
- Lauche, R.; Peng, W.; Ferguson, C.; Cramer, H.; Frawley, J.; Adams, J.; Sibbritt, D. Efficacy of Tai Chi and qigong for the prevention of stroke and stroke risk factors: A systematic review with meta-analysis. Medicine 2017, 96, e8517. [Google Scholar] [CrossRef]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; The PRISMA-DTA Group. Preferred reporting items for a systematic review and meta-analysis of diagnostic test accuracy studies: The PRISMA-DTA statement. Jama 2018, 319, 388–396. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef]
- Higgins, J.P. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (Updated July 2019). Cochrane. 2019. Available online: www.training.cochrane.org/handbook (accessed on 18 May 2020).
- You, T.; Ogawa, E.F.; Thapa, S.; Cai, Y.; Yeh, G.Y.; Wayne, P.M.; Shi, L.; Leveille, S.G. Effects of Tai Chi on beta endorphin and inflammatory markers in older adults with chronic pain: An exploratory study. Aging Clin. Exp. Res. 2019, 20, 20. [Google Scholar] [CrossRef]
- Sungkarat, S.; Boripuntakul, S.; Kumfu, S.; Lord, S.R.; Chattipakorn, N. Tai Chi improves cognition and plasma BDNF in Older adults with mild cognitive impairment: A randomized controlled trial. Neurorehabilit. Neural Repair 2018, 32, 142–149. [Google Scholar] [CrossRef]
- Liu, P.; You, J.; Loo, W.T.Y.; Sun, Y.; He, Y.; Sit, H.; Jia, L.; Wong, M.; Xia, Z.; Zheng, X.; et al. The efficacy of Guolin-Qigong on the body-mind health of Chinese women with breast cancer: A randomized controlled trial. Qual. Life Res. 2017, 26, 2321–2331. [Google Scholar] [CrossRef] [PubMed]
- Vera, F.M.; Manzaneque, J.M.; Rodríguez, F.M.; Bendayan, R.; Fernandez, N.; Alonso, A. Acute effects on the counts of innate and adaptive immune response cells after 1 month of Taoist Qigong Practice. Int. J. Behav. Med. 2016, 23, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Robins, J.L.; Elswick, R.K.; Sturgill, J., Jr.; McCain, N.L. The Effects of Tai Chi on Cardiovascular Risk in Women. Am. J. Health Promot. 2016, 30, 613–622. [Google Scholar] [CrossRef]
- Campo, R.A.; Light, K.C.; O’Connor, K.; Nakamura, Y.; Lipschitz, D.; LaStayo, P.C.; Pappas, L.M.; Boucher, K.M.; Irwin, M.R.; Hill, H.R.; et al. Blood pressure, salivary cortisol, and inflammatory cytokine outcomes in senior female cancer survivors enrolled in a tai chi chih randomized controlled trial. J. Cancer Surviv. Res. Pract. 2015, 9, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.R.; Olmstead, R.; Breen, E.C.; Witarama, T.; Carrillo, C.; Sadeghi, N.; Arevalo, J.M.G.; Ma, J.; Nicassio, P.; Bootzin, R.; et al. Cognitive behavioral therapy and Tai Chi reverse cellular and genomic markers of inflammation in late-life Insomnia: A randomized controlled trial. Biol. Psychiatry 2015, 78, 721–729. [Google Scholar] [CrossRef]
- Irwin, M.R.; Olmstead, R.; Carrillo, C.; Sadeghi, N.; Breen, E.C.; Witarama, T.; Yokomizo, M.; Lavretsky, H.; Carroll, J.E.; Motivala, S.J.; et al. Cognitive behavioral therapy vs. Tai Chi for late life insomnia and inflammatory risk: A randomized controlled comparative efficacy trial. Sleep 2014, 37, 1543–1552. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, P.; Wang, R.; Yuan, Y.; Wang, X.; Li, C. Effect of Tai Chi on mononuclear cell functions in patients with non-small cell lung cancer. BMC Complementary Altern. Med. 2015, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, J.; Chen, P.; Yu, D. Regular Tai Chi exercise decreases the percentage of type 2 cytokine–producing cells in postsurgical non–small cell lung cancer survivors. Cancer Nurs. 2013, 36, E27–E34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Wang, R.; Chen, P.-J.; Yu, D.-H. Effects of Tai Chi Chuan training on cellular immunity in post-surgical non-small cell lung cancer survivors: A randomized pilot trial. J. Sport Health Sci. 2013, 2, 104–108. [Google Scholar] [CrossRef]
- Irwin, M.R.; Olmstead, R.; Breen, E.C.; Witarama, T.; Carrillo, C.; Sadeghi, N.; Arevalo, J.M.G.; Ma, J.; Nicassio, P.; Ganz, P.A.; et al. Tai Chi, cellular inflammation, and transcriptome dynamics in breast cancer survivors with insomnia: A randomized controlled trial. JNCI Monogr. 2014, 2014, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, P.; Wang, R.; Yuan, Y.; Li, C. Effect of Tai Chi exercise on immune function in middle-aged and elderly women. J. Sports Med. Doping Stud. 2012, 2, 1–7. [Google Scholar] [CrossRef]
- Irwin, M.R.; Olmstead, R.; Oxman, M.N. Augmenting immune responses to varicella zoster virus in older adults: A randomized, controlled trial of Tai Chi. J. Am. Geriatr. Soc. 2007, 55, 511–517. [Google Scholar] [CrossRef]
- Irwin, M.R.; Olmstead, R. Mitigating cellular inflammation in older adults: A randomized controlled trial of Tai Chi Chih. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry 2012, 20, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.; Butow, P.; Mullan, B.; Clarke, S.; Beale, P.; Pavlakis, N.; Kothe, E.; Lam, L.; Rosenthal, D. Impact of medical Qigong on quality of life, fatigue, mood and inflammation in cancer patients: A randomized controlled trial. Ann. Oncol. 2010, 21, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.; Butow, P.N.; Mullan, B.A.; Clarke, S.J.; Beale, P.J.; Pavlakis, N.; Lee, M.S.; Rosenthal, D.S.; Larkey, L.; Vardy, J. Effect of medical Qigong on cognitive function, quality of life, and a biomarker of inflammation in cancer patients: A randomized controlled trial. Supportive Care Cancer 2012, 20, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Lavretsky, H.; Alstein, L.L.; Olmstead, R.E.; Ercoli, L.M.; Riparetti-Brown, M.; Cyr, N.S.; Irwin, M.R. Complementary use of tai chi chih augments escitalopram treatment of geriatric depression: A randomized controlled trial. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry 2011, 19, 839–850. [Google Scholar] [CrossRef]
- Wang, M.Y.; An, L.G. Effects of 12-Week’s Tai Chi Chuan practice on the immune function of female college students who lack physical exercise. Biol. Sport 2011, 28, 45–49. [Google Scholar] [CrossRef][Green Version]
- Chen, S.-C.; Ueng, K.-C.; Lee, S.-H.; Sun, K.-T.; Lee, M.-C. Effect of T’ai Chi Exercise on biochemical profiles and oxidative stress indicators in obese patients with type 2 diabetes. J. Altern. Complement. Med. 2010, 16, 1153–1159. [Google Scholar] [CrossRef]
- Manzaneque, J.M.; Vera, F.M.; Rodriguez, F.M.; Garcia, G.J.; Leyva, L.; Blanca, M.J. Serum Cytokines, mood and sleep after a Qigong program: Is Qigong an effective psychobiological tool? J. Health Psychol. 2009, 14, 60–67. [Google Scholar] [CrossRef]
- McCain, N.L.; Gray, D.P.; Elswick, R.K.; Robins, J.W.; Tuck, I.; Walter, J.M.; Rausch, S.M.; Ketchun, J.M. A randomized clinical trial of alternative stress management interventions in persons with HIV infection. J. Consult. Clin. Psychol. 2008, 76, 431–441. [Google Scholar] [CrossRef]
- Chen, H.-H.; Yeh, M.-L.; Lee, F.-Y. The Effects of baduanjin Qigong in the prevention of bone loss for middle-aged women. Am. J. Chin. Med. 2006, 34, 741–747. [Google Scholar] [CrossRef]
- Irwin, M.R.; Pike, J.L.; Cole, J.C.; Oxman, M.N. Effects of a behavioral intervention, Tai Chi Chih, on Varicella-Zoster virus specific immunity and health functioning in older adults. Psychosom. Med. 2003, 65, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Morgan, N.; Irwin, M.R.; Chung, M.; Wang, C. The effects of mind-body therapies on the immune system: Meta-Analysis. PLoS ONE 2014, 9, e100903. [Google Scholar] [CrossRef]
- You, T.; Ogawa, E.F. Effects of T’ai Chi on chronic systemic inflammation. J. Altern. Complement. Med. 2019, 25, 656–658. [Google Scholar] [CrossRef]
- Wang, C.-W.; Ng, S.-M.; Ho, R.-T.H.; Ziea, E.T.C.; Wong, V.C.W.; Chan, C.L.W. The effect of qigong exercise on immunity and infections: A systematic review of controlled trials. Am. J. Chin. Med. 2012, 40, 1143–1156. [Google Scholar] [CrossRef] [PubMed]
- Ho, R.T.H.; Wang, C.-W.; Ng, S.-M.; Ho, A.H.Y.; Ziea, E.T.C.; Wong, V.T.; Chan, C.L.W. The effect of T’ai Chi exercise on immunity and infections: A systematic review of controlled trials. J. Altern. Complement. Med. 2013, 19, 389–396. [Google Scholar] [CrossRef]
- Khosravi, N.; Stoner, L.; Farajivafa, V.; Hanson, E.D. Exercise training, circulating cytokine levels and immune function in cancer survivors: A meta-analysis. Brain. Behav. Immun. 2019, 81, 92–104. [Google Scholar] [CrossRef]
- Ben-Sasson, S.Z.; Hu-Li, J.; Quiel, J.; Cauchetaux, S.; Ratner, M.; Shapira, I.; Dinarello, C.A.; Paul, W.E. IL-1 acts directly on CD4 T cells to enhance their antigen-driven expansion and differentiation. Proc. Natl. Acad. Sci. USA 2009, 106, 7119. [Google Scholar] [CrossRef]
- Musolino, C.; Allegra, A.; Innao, V.; Allegra, A.G.; Pioggia, G.; Gangemi, S. Inflammatory and anti-inflammatory equilibrium, proliferative and antiproliferative balance: The role of cytokines in multiple myeloma. Mediat. Inflamm. 2017, 2017, 1852517. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zhang, Y.; Kong, Z.; Loprinzi, P.D.; Hu, Y.; Ye, J.; Liu, S.; Yu, J.J.; Zou, L. The effects of Tai Chi on markers of atherosclerosis, lower-limb physical function, and cognitive ability in adults aged over 60: A randomized controlled trial. Int. J. Environ. Res. Public Health 2019, 16, 753. [Google Scholar] [CrossRef] [PubMed]
| Author/Years/Reference | Population | Sample Size (% Female) | Mean Age (Range) | Intervention Duration, Type | Intervention Frequency, Style | Control Condition | Results ↑ ↓ |
|---|---|---|---|---|---|---|---|
| You et al. [41] 2019 China | Older adults with chronic pain | N = 40 (43%) TC (n = 19) CG (n = 21) | 74 (65–87) | 12 weeks; TC | 60 min, 2 times/week, 8-form Yang style | Light physical exercise | NS CRP NS IL-6 NS TNF-α |
| Sungkarat et al. [42] 2018 Thailand | Older adults with mild cognitive impairment | N = 56 (86%) TC (n = 29) CG (n = 27) | 68 | 6 months; TC | 50 min, 3 times/week, TC in-class for 9 sessions (3 times per week for 3 weeks) plus practice at home 3 times a week with video, 10 forms TC | Health Education | NS TNF-α NS IL-10 |
| Liu et al. [43] 2017 Hong Kong | Women with breast cancer | N = 158 (100%) QG (n = 50) CG (n = 51) | 51 (21–80) | 24 weeks; QG | 60 min session, 2 times/week for 24 weeks, Guolin-Qigong | Stretching exercise | NS IL-2 NS IFN-γ ↑ TNF-α: (95% CI, 0.48–3.41, p < 0.03) |
| Vera et al. [44] 2016 Spain | College students | N = 43 (79%) QQ (n = 23) CG (n = 18) | (18–21) | 4 weeks; QG | 25–30 min, 3 times/week In total 20 qigong sessions, Taoist QG | Daily activities | ↑ Number of B lymphocytes (p = 0.006) ↑ % of B lymphocytes (p = 0.006) ↓ % NK (p = 0.05) NS Number of NK NS Neutrophils, NS Monocytes, NS Eosinophils, |
| Robins et al. [45] 2016 USA | Women with high risk of cardiovascular disease | N = 96 (100%) TC (n = 47) CG (n = 49) | 43 (35–50) | 8 weeks;TC | 60 min, 1/week, TC (short form) | Wait-list control | ↓ TNF-α (p < 0.002) ↓ IFN-γ (p < 0.002) ↓ IL 8 (p < 0.026) ↓ IL-4 (p < 0.001) Result not reported (IL-1β, IL-2, IL-10, IL-12, IL-6, GCSF, CRP) |
| Campo et al. [46] 2015 USA | Older female cancer survivors | N = 63 (100%) TC (n = 29) CG (n = 25) | 67 | 12 weeks; TC | 60 min, 3 times/week, TC (19 movements) | Health education | NS IL-6 NS IL-12 NS TNF-α NS IL-4 NS IL-10 |
| Irwin et al. [47,48] 2015 and 2014 USA | Older adults with insomnia | N = 123 (73%) TC (n = 48) CBT (n = 50) CG (n = 25) | 65 (55–85) | 16 weeks; TC | 120 min, 1/week, TC | Health education, CBT | ↓ CRP (n = 0.06) ↓ % monocytes producing ↓ IL-6 (p < 0.01) ↓ % monocytes producing TNF-α (p < 0.01) ↓ % monocytes co-producing TNF-α and IL-6 (p < 0.01) at different time points ↓ Pro-inflammatory gene expression (NF-kB, IRF+, AP-1) ((month 4, p < 0.001). |
| Liu et al. [49] 2015 Wang et al. [50] 2013 Zhang et al. [51] 2013 China | Lung cancer survivors | N = 32 (50%) TC (n = 16) CG (n = 16) | 61 | 16-weeks; TC | 60 min, 3 times/week, Yang style 24-form | Daily activities | ↑ NKT cells (p < 0.05) ↑ % NKT cells (p < 0.05) NS NK cells NS % NK cells NS DC 123c cells NS % of DC 123c cells ↑ DC11c cells (p < 0.01) NS % DC11c cells T lymphocyte cells (T1) and (T2) ↑ T1/T2 ratio (p < 0.01) ↑ Tc1/Tc2 ratios (p < 0.01) NS TH1/TH2 ratio ↓ T2 (p < 0.001) ↓ Tc2 (p < 0.003) ↑ Th2 (p < 0.025) ↓ CD55 (p < 0.05) NS T-helper/T-suppressor (CD4+:CD8+ ratio) NS CD59 expression |
| Irwin et al. [52] 2014 USA | Breast cancer survivors with insomnia | N = 90 (100%) TC (n = 45) CG (n = 45) | 59 (42–83) | 12 weeks; TC | 120 min, 1/week, TC | CBT | NS CRP ↓ % monocytes producing IL-6 (p = 0.07) ↓ % monocytes producing TNF-α (p < 0.05) ↓ % monocytes coproducing TNF-α and IL-6 (p < 0.02) ↓ Proinflammatory mediators (NF-kB) (p = 0.001) |
| Liu et al. [53] 2012 China | Healthy middle-aged and older women | N = 60 TC (n = 30) CG (n = 30) | 54 (50–65) | 6 months; TC (24 forms and 42 Sword forms) | 60 min, 4 times/week TC (24 forms and 42 Sword forms) | Daily activities | ↑ % of CD4+ T lymphocytes (p < 0.05) NS CD3 NS CD8+ ↑ CD4+:CD8+ ratio (p < 0.05) ↑ % of NK cell (p < 0.05) ↑ % of NKT cells (p < 0.05) ↑ TNF- γ (p < 0.05) NS IL-4 ↑ % of CD123+ DCs (p < 0.01) ↑ % of CD11c+ DCs (p < 0.01) |
| Irwin et al. [54,55] 2007 and 2012 USA | Healthy older adults with history of varicella | N = 112 (61%) TC (n = 59) CG (n = 53) | 70 (59–86) | 16 weeks; TC | 40 min, 3 times/week for a total of 120 min, TC with 20 movements | Health education | ↑ Levels of VZV-CMI (p < 0.05) NS IL-6 (p = 0.06) NS IL-18 NS CRP NS sIL-1RA NS sIL-6R NS sICAM, |
| Oh et al. [56,57] 2012 and 2012 Australia | Cancer survivors | N = 162 (57%) TC (n = 79) CG (n = 83) | 60 (31–86) | 10 weeks; QG | 90 min, 2 times/week + individual practice recommended for 30 min per day, Medical QG | Usual Care | ↓ CRP (p < 0.044) |
| Lavretsky et al. [58] 2011 USA | Older adults with major depression | N = 73 (62%) TC (n = 36) CG (n = 37) | 71 | 10 weeks; TC | 120 min, 1 times/week TC Chih | Health education | ↓ CRP (p < 0.05) |
| Wang et al. [59] 2011, China | Healthy sedentary female college students | N = 60 (100%) TC (n = 30) CG (n = 30) | 19 | 12 weeks; TC | 45 min, 5 times/week; TC 24 standardized movements | Daily activities | NS IgG NS IgA NS IgM NS (CD3, CD4+, CD8+) NS IFN-γ NS IL-4 NS IL-12 |
| Chen et al. [60] 2010 Taiwan | Adults with diagnosis of Type II diabetes and BMI 30–35 | N = 104 (43%) TC (n = 56) CG (n = 48) | 58 | 12 weeks; TC | 60 min, 3 times/week; Chen-style 99-form | Conventional aerobic exercise | ↓CRP (p < 0.014) |
| Manzaneque et al. [61] 2009 Spain | Healthy college students | N = 39 (87%) QG (n = 21) CG (n = 18) | 18–21 | 4 weeks; QG | 30 min, 3 times/week; Ba Duan Jin Qg | Daily activities | NS TNF-α NS IFN-γ |
| McCain et al. [62] 2008 USA | Adults with diagnosis of HIV | N = 252 (40%) TC (n = 62) RLXN (n = 65) SPRT (n = 68) CG (n = 57) | 42 | 10 weeks; TC | 90 min, 1 time/week; Focused short form TC training with 8 movements | Wait-list control | NS CD4+, CD8+, and CD57+ T lymphocytes: NS NKC cytotoxicity NS Lymphocyte proliferation NS TNF-α NS IFN-γ NS IL-2 NS IL-4 NS IL-6 NS IL-10 |
| Chen et al. [63] 2006 Taiwan | Healthy middle-aged women with BMD T scores ≥ −2.5 | N = 87 (100%) QG (n = 44) CG (n = 43) | 45 (35–60) | 12 weeks; QG | 3 times/week; Ba Duan Jin Qg 8 sections | Daily activities | ↓ IL-6 (p < 0.001) |
| Irwin et al. [64] 2003 USA | Healthy older adults with history of varicella | N = 36 (72%) TC (n = 18) CG (n = 18) | 70 | 15 weeks; TC | 45 min, 3 times/week for a total of 45 sessions; TC with 20 standardized movements | Wait-list control (maintenance of routine activities | ↑ Levels of VZV-CMI (p < 0.05) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, B.; Bae, K.; Lamoury, G.; Eade, T.; Boyle, F.; Corless, B.; Clarke, S.; Yeung, A.; Rosenthal, D.; Schapira, L.; et al. The Effects of Tai Chi and Qigong on Immune Responses: A Systematic Review and Meta-Analysis. Medicines 2020, 7, 39. https://doi.org/10.3390/medicines7070039
Oh B, Bae K, Lamoury G, Eade T, Boyle F, Corless B, Clarke S, Yeung A, Rosenthal D, Schapira L, et al. The Effects of Tai Chi and Qigong on Immune Responses: A Systematic Review and Meta-Analysis. Medicines. 2020; 7(7):39. https://doi.org/10.3390/medicines7070039
Chicago/Turabian StyleOh, Byeongsang, Kyeore Bae, Gillian Lamoury, Thomas Eade, Frances Boyle, Brian Corless, Stephen Clarke, Albert Yeung, David Rosenthal, Lidia Schapira, and et al. 2020. "The Effects of Tai Chi and Qigong on Immune Responses: A Systematic Review and Meta-Analysis" Medicines 7, no. 7: 39. https://doi.org/10.3390/medicines7070039
APA StyleOh, B., Bae, K., Lamoury, G., Eade, T., Boyle, F., Corless, B., Clarke, S., Yeung, A., Rosenthal, D., Schapira, L., & Back, M. (2020). The Effects of Tai Chi and Qigong on Immune Responses: A Systematic Review and Meta-Analysis. Medicines, 7(7), 39. https://doi.org/10.3390/medicines7070039







