Pilot Studies on Two Complementary Bath Products for Atopic Dermatitis Children: Pine-Tar and Tea
Abstract
1. Introduction
2. Materials and Methods
2.1. Subject Recruitment
2.2. Study Design
2.3. Studied Product
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Leung, A.K.; Hon, K.L.; Robson, W.L. Atopic dermatitis. Adv. Pediatr. 2007, 54, 241–273. [Google Scholar] [CrossRef] [PubMed]
- Leung, T.N.; Hon, K.L. Eczema therapeutics in children: What do the clinical trials say? Hong Kong Med. J. 2015, 21, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Van Zuuren, E.J.; Fedorowicz, Z.; Christensen, R.; Lavrijsen, A.; Arents, B.W.M. Emollients and moisturisers for eczema. Cochrane Database Syst. Rev. 2017, 2, CD012119. [Google Scholar] [CrossRef]
- Hon, K.L.; Kung, J.S.C.; Ng, W.G.G.; Leung, T.F. Emollient Treatment of Atopic Dermatitis: Latest Evidence and Clinical Considerations. Drugs Context 2018, 7, 212530. [Google Scholar] [CrossRef] [PubMed]
- D’Auria, E.; Banderali, G.; Barberi, S.; Gualandri, L.; Pietra, B.; Riva, E.; Cerri, A. Atopic dermatitis: Recent insight on pathogenesis and novel therapeutic target. Asian Pac. J. Allergy Immunol. 2016, 34, 98–108. [Google Scholar] [PubMed]
- Hon, K.L.; Wang, S.S.; Pong, N.H.; Leung, T.F. The ideal moisturizer: A survey of parental expectations and practice in childhood-onset eczema. J. Dermatol. Treat. 2013, 24, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.L.; Kung, J.S.C.; Tsang, K.Y.C.; Yu, J.W.S.; Lee, V.W.; Leung, T.F. Emollient acceptability in childhood atopic dermatitis: Not all emollients are equal? Curr. Pediatr. Rev. 2017, 14, 117–122. [Google Scholar] [CrossRef]
- Giam, Y.C.; Hebert, A.A.; Dizon, M.V.; Van Bever, H.; Tiongco-Recto, M.; Kim, K.H.; Soebono, H.; Munasir, Z.; Diana, I.A.; Luk, D.C. A review on the role of moisturizers for atopic dermatitis. Asia Pac. Allergy 2016, 6, 120–128. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, A.K.; Barankin, B. Barrier Repair Therapy in Atopic Dermatitis: An Overview. Am. J. Clin. Dermatol. 2013, 14, 389–399. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, A.K.C. Integrative, integrated medicine but no integration: Tarnishing steroid and Chinese medicine is vanity. Hong Kong J. Paediatr. 2018, 23, 192–194. [Google Scholar]
- Hon, K.L.; Leung, A.K.; Leung, T.N.; Lee, V.W. Complementary, Alternative and Integrative Medicine for Childhood Atopic Dermatitis. Recent Patents Inflamm. Allergy Drug Discov. 2017, 11, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Reuter, J.; Wölfle, U.; Korting, H.C.; Schempp, C. Which Plant for Which Skin Disease? Part 2: Dermatophytes, Chronic Venous Insufficiency, Photoprotection, Actinic Keratoses, Vitiligo, Hair Loss, Cosmetic Indications. J. Dtsch. Dermatol. Ges. 2010, 8, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Chang, H.K.; Baek, S.Y.; Chung, J.O.; Rha, C.S.; Kim, S.Y.; Kim, B.J.; Kim, M.N. Treatment of Atopic Dermatitis Associated with Malassezia sympodialis by Green Tea Extracts Bath Therapy: A Pilot Study. Mycobiology 2012, 40, 124–128. [Google Scholar] [CrossRef]
- Myers, R.A.; Fuller, E.; Yang, W. Identification of native catechin fatty acid esters in green tea (Camellia sinensis). J. Agric. Food Chem. 2013, 61, 11484–11493. [Google Scholar] [CrossRef] [PubMed]
- Unachukwu, U.J.; Ahmed, S.; Kavalier, A.; Lyles, J.T.; Kennelly, E.J. White and green teas (Camellia sinensis var. sinensis): Variation in phenolic, methylxanthine, and antioxidant profiles. J. Food Sci. 2010, 175, C541–C548. [Google Scholar] [CrossRef]
- Łuczaj, W.; Skrzydlewska, E. Antioxidative properties of black tea. Prev. Med. 2005, 40, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Henning, S.M.; Niu, Y.; Lee, R.; Scheuller, H.S.; Heber, D. Catechin and caffeine content of green tea dietary supplements and correlation with antioxidant capacity. J. Agric. Food Chem. 2006, 54, 1599–1603. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Daglia, M.; Ciampaglia, R.; Novellino, E. Exploring the nutraceutical potential of polyphenols from black, green and white tea infusions—An overview. Curr. Pharm. Biotechnol. 2015, 16, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Fechtner, S.; Singh, A.; Chourasia, M.; Ahmed, S. Molecular insights into the differences in anti-inflammatory activities of green tea catechins on IL-1β signaling in rheumatoid arthritis synovial fibroblasts. Toxicol. Appl. Pharmacol. 2017, 329, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.R.; Choi, H.J.; Kang, Y.G.; Kim, J.K.; Shin, J.-W. In vitro study on anti-inflammatory effects of epigallocatechin-3-gallate-loaded nano- and microscale particles. Int. J. Nanomed. 2017, 12, 7007–7013. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, Y.; Ito, M.; Kishi, M.; Mizutani, H.; Katada, T.; Konishi, H. Antiallergic constituents from oolong tea stem. Biol. Pharm. Bull. 1995, 18, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Shiozaki, T.; Sugiyama, K.; Nakazato, K.; Takeo, T. Effect of tea extracts, catechin and caffeine against type-I allergic reaction. Yakugaku Zasshi 1997, 117, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Reuter, J.; Wölfle, U.; Weckesser, S.; Schempp, C. Which plant for which skin disease? Part 1: Atopic dermatitis, psoriasis, acne, condyloma and herpes simplex. J. Dtsch. Dermatol. Ges. 2010, 8, 788–796. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.; Suzuki, M.; Miyase, T.; Yoshino, K.; Maeda-Yamamoto, M. Novel Antiallergic Catechin Derivatives Isolated from Oolong Tea. J. Agric. Food Chem. 1999, 47, 1906–1910. [Google Scholar] [CrossRef] [PubMed]
- Uehara, M.; Sugiura, H.; Sakurai, K. A trial of oolong tea in the management of recalcitrant atopic dermatitis. Arch. Dermatol. 2001, 137, 42–43. [Google Scholar] [CrossRef]
- Schmid, M.H.; Korting, H.C. Coal tar, pine tar and sulfonated shale oil preparations: Comparative activity, efficacy and safety. Dermatology 1996, 193, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Paghdal, K.V.; Schwartz, R.A. Topical tar: Back to the future. J. Am. Acad. Dermatol. 2009, 61, 294–302. [Google Scholar] [CrossRef]
- Hanifin, J.M.; Rajka, G. Diagnostic features of atopic dermatitis. Acta Derm. Venereol. 1980, 2, 44–47. [Google Scholar]
- Kunz, B.; Oranje, A.P.; Labrèze, L.; Stalder, J.F.; Ring, J.; Taïeb, A. Clinical validation and guidelines for the SCORAD index: Consensus report of the European Task Force on Atopic Dermatitis. Dermatology 1997, 195, 10–19. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, T.F.F.; Wong, K.Y.; Chow, C.M.; Chuh, A.; Ng, P.C. Does age or gender influence quality of life in children with atopic dermatitis? Clin. Exp. Dermatol. 2008, 33, 705–709. [Google Scholar] [CrossRef]
- Chuh, A.A. Validation of a Cantonese version of the Children’s Dermatology Life Quality Index. Pediatr. Dermatol. 2003, 20, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.L.; Kung, J.S.; Tsang, K.Y.; Yu, J.W.; Wong, N.; Leung, T.F. Do we need another symptom score for childhood eczema? J. Dermatol. Treat. 2018, 29. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.L.; Kung, J.; Ng, W.G.; Tsang, K.; Cheng, N.S.; Leung, T.F. Are skin equipment for assessing childhood eczema any good? J. Dermatol. Treat. 2018. [Google Scholar] [CrossRef] [PubMed]
- Roberts, G.; Hurley, C.; Lack, G. Development of a quality-of-life assessment for the allergic child or teenager with multisystem allergic disease. J. Allergy Clin. Immunol. 2003, 111, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.H.; To, C.Y.; Chan, E.Y.; Li, H.; Zhang, X.; Chow, P.Y.; Liu, P.L.; Leung, S.Y.; Chan, C.H.; Chan, K.Y.; et al. A randomized placebo-controlled trial of traditional Chinese medicine as an add-on therapy to oral montelukast in the treatment of mild persistent asthma in children. Complement Ther. Med. 2016, 29, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.R.; Simpson, E.; Apfelbacher, C.J.; Thomas, K.S.; von, K.L.; Schmitt, J.; Singh, J.A.; Svensson, Å.; Williams, H.C.; Abuabara, K.; et al. Report from the fourth international consensus meeting to harmonize core outcome measures for atopic eczema/dermatitis clinical trials (HOME initiative). Br. J. Dermatol. 2016, 175, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.L.E.; Wong, K.Y.; Leung, T.-F.F.; Chow, C.-M.M.; Ng, P.-C.C. Comparison of Skin Hydration Evaluation Sites and Correlations among Skin Hydration, Transepidermal Water Loss, SCORAD Index, Nottingham Eczema Severity Score, and Quality of Life in Patients with Atopic Dermatitis. Am. J. Clin. Dermatol. 2008, 9, 45–50. [Google Scholar] [CrossRef]
- Ahmad, M.; Baba, W.N.; Gani, A.; Wani, T.A.; Gani, A.; Masoodi, F.A. Effect of extraction time on antioxidants and bioactive volatile components of green tea (Camellia sinensis), using GC/MS. Cogent Food Agric. 2015, 1, 1106387. [Google Scholar] [CrossRef]
- Pandey, R.K.; Manimehalai, N. Production of Instant Tea Powder by Spray Drying. Int. J. Agric. Food Sci. Technol. 2014, 5, 197–202. [Google Scholar]
- Hon, K.L.; Tsang, Y.C.; Lee, V.W.; Pong, N.H.; Ha, G.; Lee, S.T.; Chow, C.M.; Leung, T.F. Efficacy of sodium hypochlorite (bleach) baths to reduce Staphylococcus aureus colonization in childhood onset moderate-to-severe eczema: A randomized, placebo-controlled cross-over trial. J. Dermatol. Treat. 2016, 27, 156–162. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, T.F.; Ng, P.C.; Lam, M.C.; Kam, W.Y.; Wong, K.Y.; Lee, K.C.; Sung, Y.T.; Cheng, K.F.; Fok, T.F.; et al. Efficacy and tolerability of a Chinese herbal medicine concoction for treatment of atopic dermatitis: A randomized, double-blind, placebo-controlled study. Br. J. Dermatol. 2007, 157, 357–363. [Google Scholar] [CrossRef]
- Barnes, T.M.; Greive, K.A. Topical pine tar: History, properties and use as a treatment for common skin conditions. Australas. J. Dermatol. 2017, 58, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Van den Bogaard, E.H.; Bergboer, J.G.; Vonk-Bergers, M.; van Vlijmen-Willems, I.M.; Hato, S.V.; van der Valk, P.G.; Schröder, J.M.; Joosten, I.; Zeeuwen, P.L.; Schalkwijk, J. Coal tar induces AHR-dependent skin barrier repair in atopic dermatitis. J. Clin. Investig. 2013, 123. [Google Scholar] [CrossRef] [PubMed]
- Takei, K.; Mitoma, C.; Hashimoto-Hachiya, A.; Uchi, H.; Takahara, M.; Tsuji, G.; Kido-Nakahara, M.; Nakahara, T.; Furue, M. Antioxidant soybean tar G lyteer rescues T-helper-mediated downregulation of filaggrin expression via aryl hydrocarbon receptor. J. Dermatol. 2015, 42, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Santer, M.; Ridd, M.J.; Francis, N.A.; Stuart, B.; Rumsby, K.; Chorozoglou, M.; Becque, T.; Roberts, A.; Liddiard, L.; Nollett, C.; et al. Emollient bath additives for the treatment of childhood eczema (BATHE): Multicentre pragmatic parallel group randomised controlled trial of clinical and cost effectiveness. BMJ 2018, 361, k1332. [Google Scholar]
- Warshaw, E.M.; Zug, K.A.; Belsito, D.V.; Fowler, J.F., Jr.; De Koven, J.G.; Sasseville, D.; Maibach, H.I.; Mathias, C.T.; DeLeo, V.A.; Taylor, J.S.; et al. Positive Patch-Test Reactions to Essential Oils in Consecutive Patients from North America and Central Europe. Dermatitis 2017, 28, 246–252. [Google Scholar] [CrossRef]
- Van Zuuren, E.J.; Fedorowicz, Z.; Arents, B.W.M. Performance and Tolerability of the Moisturizers Cetaphil((R)) and Excipial((R)) in Atopic Dermatitis: What is the Evidence Based on Randomized Trials? Dermatol. Ther. 2017, 7, 331–347. [Google Scholar] [CrossRef]
- Hon, K.L.; Pong, N.H.; Wang, S.S.; Lee, V.W.; Luk, N.M.; Leung, T.F. Acceptability and Efficacy of an Emollient Containing Ceramide-Precursor Lipids and Moisturizing Factors for Atopic Dermatitis in Pediatric Patients. Drugs R D 2013, 13, 37–42. [Google Scholar] [CrossRef]
- Kaplan, S.L.; Forbes, A.; Hammerman, W.A.; Lamberth, L.; Hulten, K.G.; Minard, C.G.; Mason, E.O. Randomized trial of “bleach baths” plus routine hygienic measures vs. routine hygienic measures alone for prevention of recurrent infections. Clin. Infect. Dis. 2014, 58, 679–682. [Google Scholar] [CrossRef]
| Patient Information | Median (IQR) for Tea Bath Study | Median (IQR) for Pine-Tar Bath Study |
|---|---|---|
| Sex (%) | 8 (40.0) Male 12 (60.0) Female | 5 (55.6) Male 4 (44.4) Female |
| Age (Years) | 10.8 (7.5–15.7) | 10.3 (7.1–13.9) |
| Objective SCORAD (SCORAD without the subjective components) | 33.3 (24.8–45.5) | 40.8 (22.2–56.6) |
| POEM | 18.5 (13.0–23.0) | 17.7 (12.0–23.0) |
| NESS | 15 Severe; 2 Moderate | 7 Severe; 2 Moderate |
| CDLQI | 9.0 (6.0–16.0) | 11.0 (10.0–16.5) |
| PADQLQ | - | 50.0 (24.5–54.0) |
| SH (a.u.) | 18.7 (14.7–31.5) | 10.2 (7.6–14.7) |
| TEWL (g/m2/h) | 7.1 (5.3–11.9) | 13.5 (7.4–18.0) |
| Erythema(a.u.) | - | 414.0 (393.0–418.5) |
| Eosinophil % | 7.0 (4.0–10.0) | 8.0 (6.3–9.8) |
| IgE (IU/mL) | 3581.5 (1650.5–5894.5) | 3169.0 (1041.5–8181.5) |
| Log [IgE] | 3.5 (3.2–3.7) | 3.5 (3.0–3.9) |
| S. aureus colonization (Number, %) | 12 (60.0) | 3 (42.9) |
| Days using topical steroid/week | 3.0 (1.0–7.0) | 2.0 (0.0–2.0) |
| Days using oral anti-histamines/week | 2.0 (0.0–6.5) | - |
| Tea Bath Study | Pine-Tar Bath Study | |||||
|---|---|---|---|---|---|---|
| Pre | Post | p-Value | Pre | Post | p-Value | |
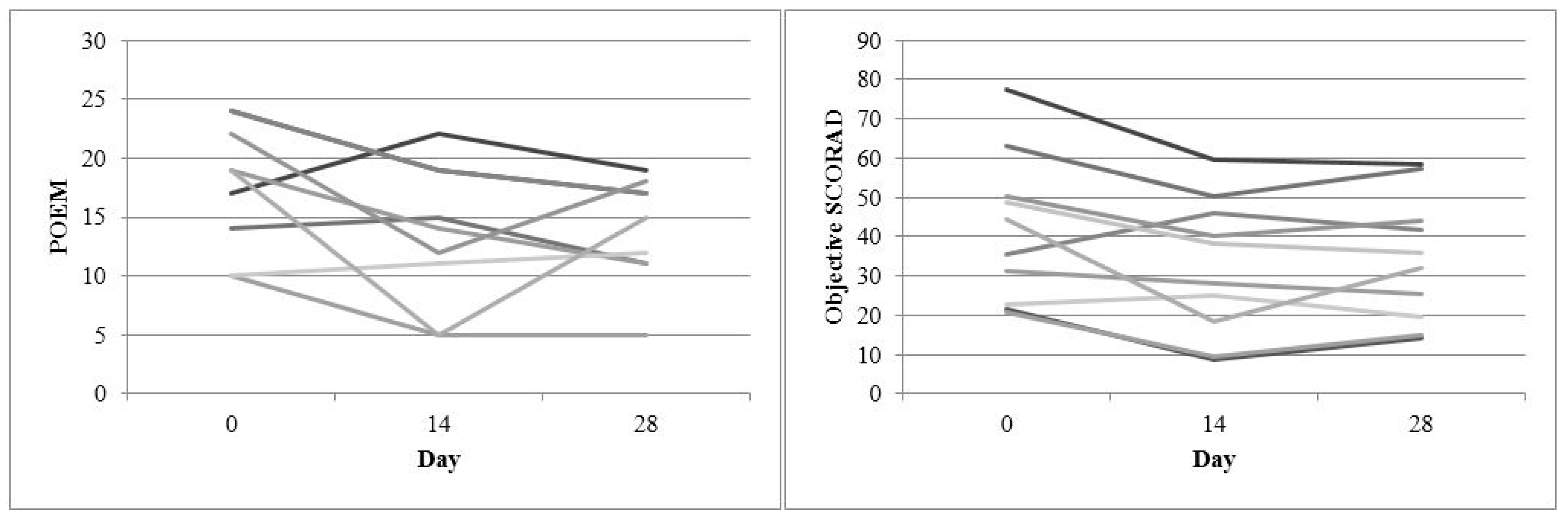
| Objective SCORAD | 33.3 (24.8–45.5) | 29.9 (16.4–37.4) | 0.324 | 40.8 (22.2–56.6) | 32.1 (17.4–50.7) | 0.050 |
| POEM | 18.5 (13.0–23.0) | 16.5 (12.0–23.0) | 0.393 | 17.7 (12.0–23.0) | 15.0 (11.0–17.5) | 0.021 |
| CDLQI | 9.0 (6.0–16.0) | 10.5 (8.0–19.0) | 0.305 | 11.0 (10.0–16.5) | 8.0 (4.0–11.5) | 0.011 |
| PADQLQ | - | - | - | 50.0 (24.5–54.0) | 29.0 (19.0–41.0) | 0.036 |
| SH (a.u.) | 18.7 (14.7–31.5) | 18.5 (14.1–34.6) | 0.575 | 10.2 (7.6–14.7) | 18.0 (11.4–20.5) | 0.192 |
| TEWL (g/m2/h) | 7.1 (5.3–11.9) | 8.8 (5.2–13.3) | 0.970 | 13.5 (7.4–18.0) | 10.7 (9.4–21.1) | 0.441 |
| Erythema (a.u.) | - | - | - | 414.0 (393.0–418.5) | 401.0 (396.5–415.5) | 0.722 |
| Eosinophil % | 7.0 (4.0–10.0) | 7.0 (4.0–9.0) | 0.858 | 8.0 (6.3–9.8) | 7.0 (7.0–10.5) | 0.172 |
| IgE (IU/mL) | 3581.5 (1650.5–5894.5) | 3277.0 (1611.0–6175.0) | 0.814 | 3169.0 (1041.5–8181.5) | 2355.0 (948.5–9032.8) | 0.141 |
| Log [IgE] | 3.5 (3.2–3.7) | 3.5 (3.2–3.7) | 3.5 (3.0–3.9) | 3.3 (3.0–4.0) | ||
| S. aureus status (no growth) | 8 (40.0) | 9 (45.0) | 0.257 | 3 (42.9) | 6 (66.7) | 0.386 |
| GAT | ||||||
| Very good or Good | 14 (70.0) | 7 (77.8) | ||||
| Fair or Poor | 6 (30.0) | 2 (22.2) | ||||
| One-Sided α= | 0.005 | 0.025 | 0.05 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Two-Sided α= | 0.01 | 0.05 | 0.10 | ||||||
| E/S* β= | 0.05 | 0.10 | 0.20 | 0.05 | 0.10 | 0.20 | 0.05 | 0.10 | 0.20 |
| 0.10 | 3565 | 2978 | 2338 | 2600 | 2103 | 1571 | 2166 | 1714 | 1238 |
| 0.15 | 1586 | 1325 | 1040 | 1157 | 935 | 699 | 963 | 762 | 551 |
| 0.20 | 893 | 746 | 586 | 651 | 527 | 394 | 542 | 429 | 310 |
| 0.25 | 572 | 478 | 376 | 417 | 338 | 253 | 347 | 275 | 199 |
| 0.30 | 398 | 333 | 262 | 290 | 235 | 176 | 242 | 191 | 139 |
| 0.40 | 225 | 188 | 148 | 164 | 133 | 100 | 136 | 108 | 78 |
| 0.50 | 145 | 121 | 96 | 105 | 86 | 64 | 88 | 70 | 51 |
| 0.60 | 101 | 85 | 67 | 74 | 60 | 45 | 61 | 49 | 36 |
| 0.70 | 75 | 63 | 50 | 55 | 44 | 34 | 45 | 36 | 26 |
| 0.80 | 58 | 49 | 39 | 42 | 34 | 26 | 35 | 28 | 21 |
| 0.90 | 46 | 39 | 32 | 34 | 27 | 21 | 28 | 22 | 16 |
| 1.00 | 38 | 32 | 26 | 27 | 23 | 17 | 23 | 18 | 14 |
| Steps | Calculation Example using Pine-Tar Study |
|---|---|
| Step 1—Determine effect size from pilot study: | Effect size = 40.8 − 32.1= 8.4 (obj. SCORAD score) |
| Step 2—Determine SD of outcome variable | SD = around 12 ** |
| Step 3—Determine Standardized effect size | Standardized effect size = effect size/SD = 8.4/12 = 0.725 |
| Step 4—Decide α (probability of Type I error) and β (probability of Type II error) | α (two sided) = 0.05; β = 1 − 0.80 = 0.20; |
| Step 5—Find out expected sample size per group from Table 3 | Using Table 3, around 34 participants per group would be required for a future randomized controlled trial. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hon, K.L.; Ng, W.G.G.; Kung, J.S.C.; Leung, P.C.; Leung, T.F. Pilot Studies on Two Complementary Bath Products for Atopic Dermatitis Children: Pine-Tar and Tea. Medicines 2019, 6, 8. https://doi.org/10.3390/medicines6010008
Hon KL, Ng WGG, Kung JSC, Leung PC, Leung TF. Pilot Studies on Two Complementary Bath Products for Atopic Dermatitis Children: Pine-Tar and Tea. Medicines. 2019; 6(1):8. https://doi.org/10.3390/medicines6010008
Chicago/Turabian StyleHon, Kam Lun, Wing Gi Gigi Ng, Jeng Sum C. Kung, Ping Chung Leung, and Ting Fan Leung. 2019. "Pilot Studies on Two Complementary Bath Products for Atopic Dermatitis Children: Pine-Tar and Tea" Medicines 6, no. 1: 8. https://doi.org/10.3390/medicines6010008
APA StyleHon, K. L., Ng, W. G. G., Kung, J. S. C., Leung, P. C., & Leung, T. F. (2019). Pilot Studies on Two Complementary Bath Products for Atopic Dermatitis Children: Pine-Tar and Tea. Medicines, 6(1), 8. https://doi.org/10.3390/medicines6010008







