Changes in Metabolic Profiles of Human Oral Cells by Benzylidene Ascorbates and Eugenol
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
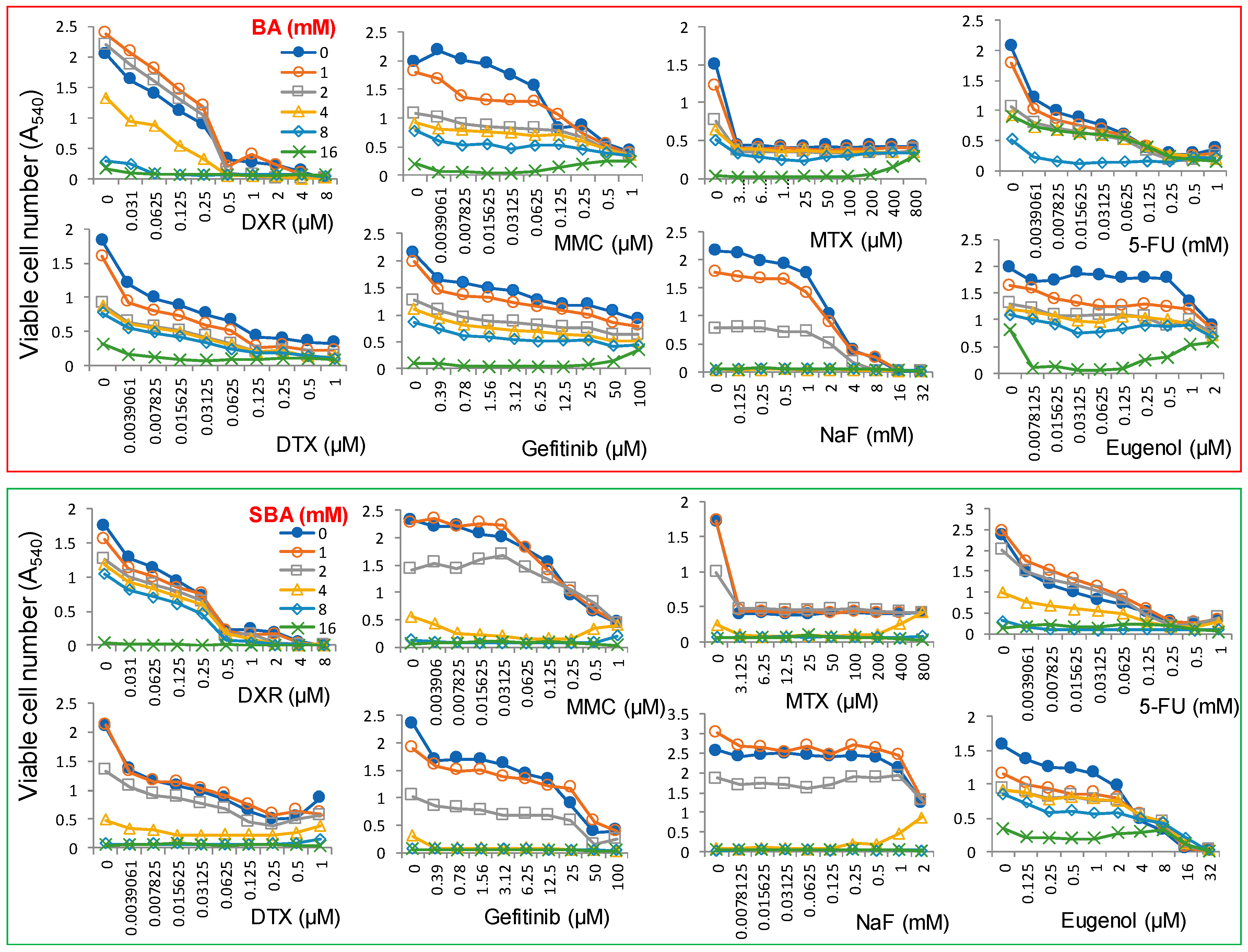
2.2. Cytotoxic Assay
2.3. Fine Cell Structure
2.4. Processing for Metabolomic Analysis
2.5. CE-MS Analysis
2.6. Statistical Analysis
3. Results
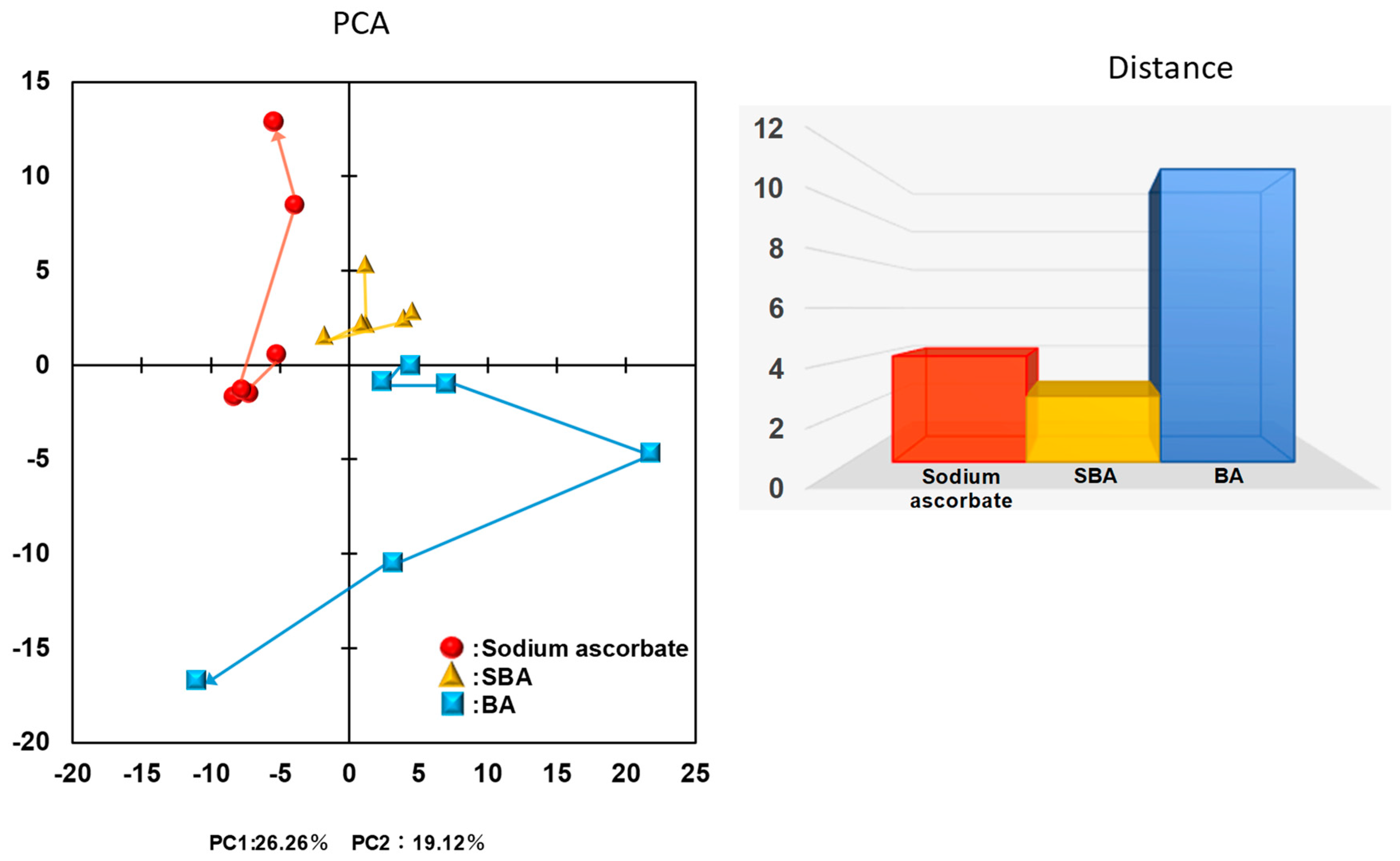
3.1. Distinct Biological Properties of BA from SBA and SA
3.1.1. Catalase Sensitivity
3.1.2. Minimum Exposure Time Required for Irreversible Cell Death Induction
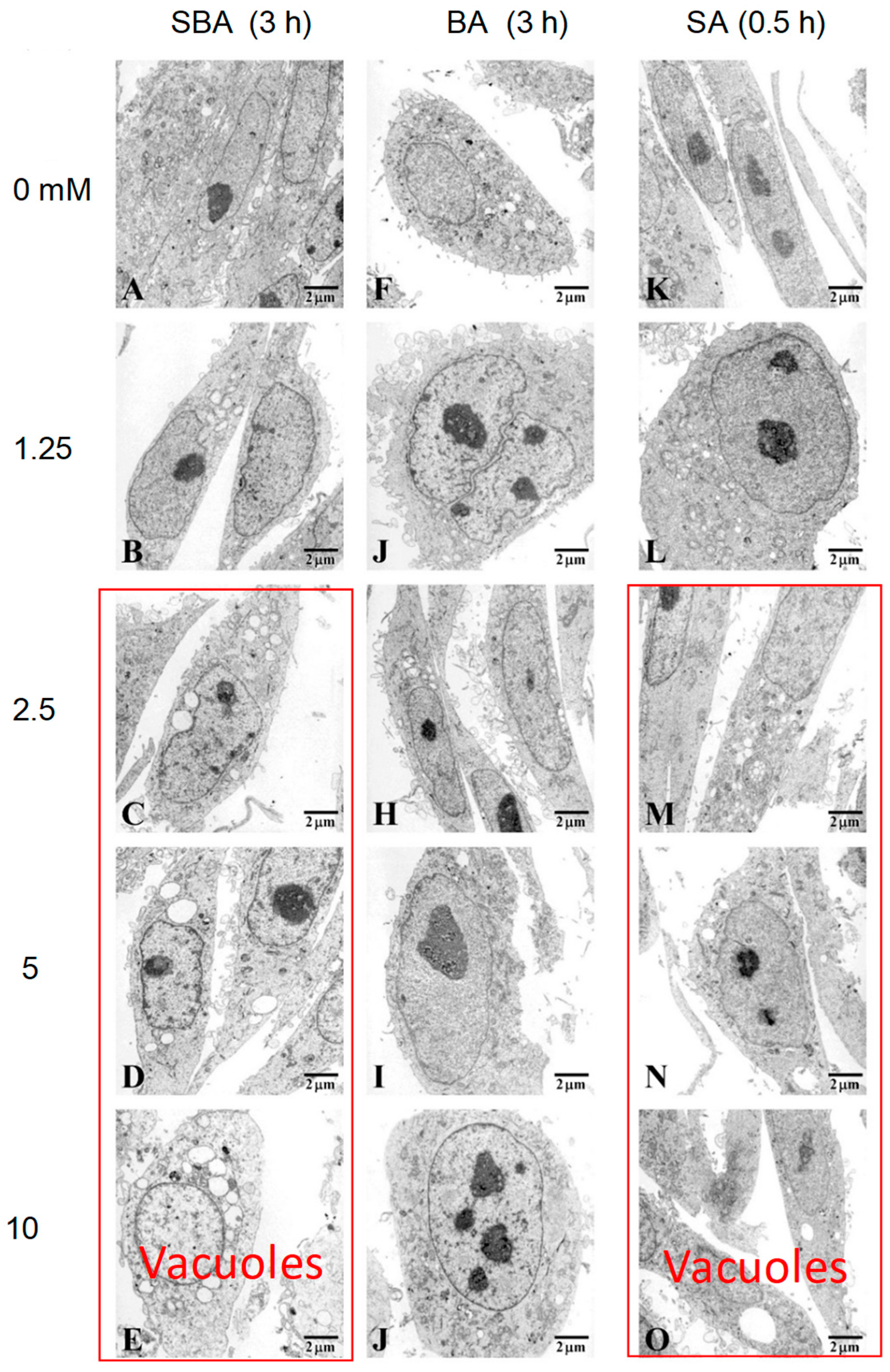
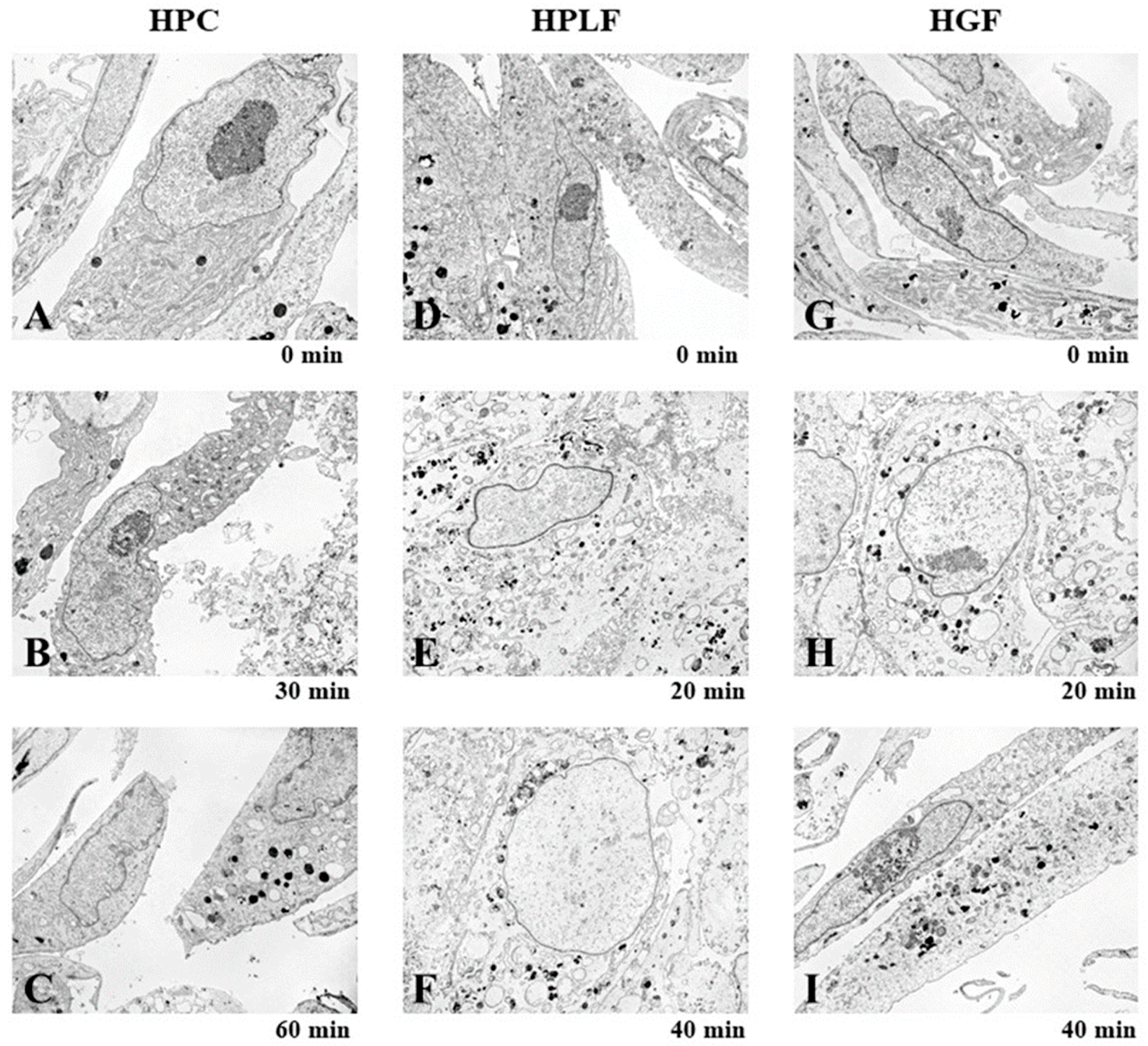
3.1.3. Induction of Mitochondrial Vacuolization
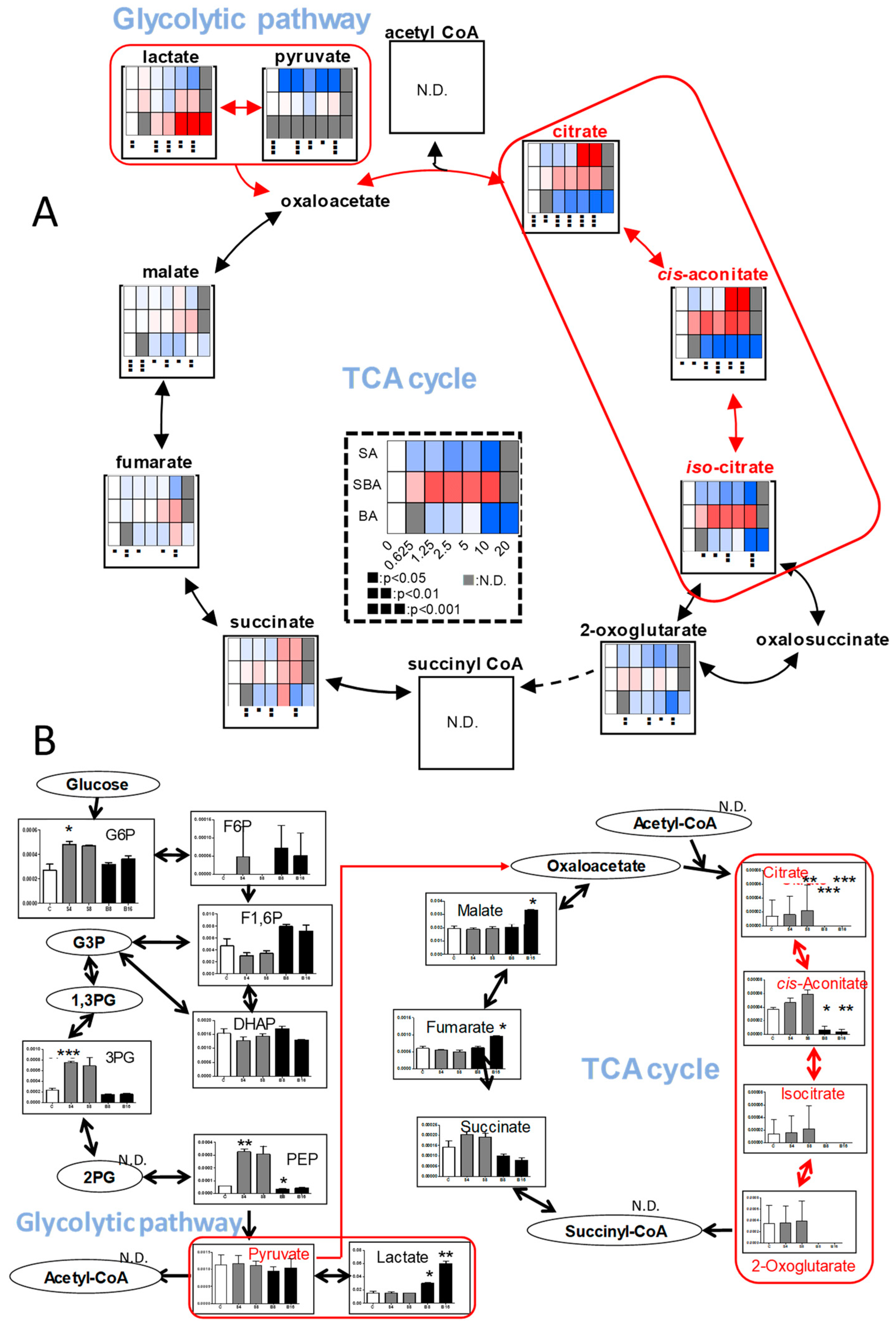
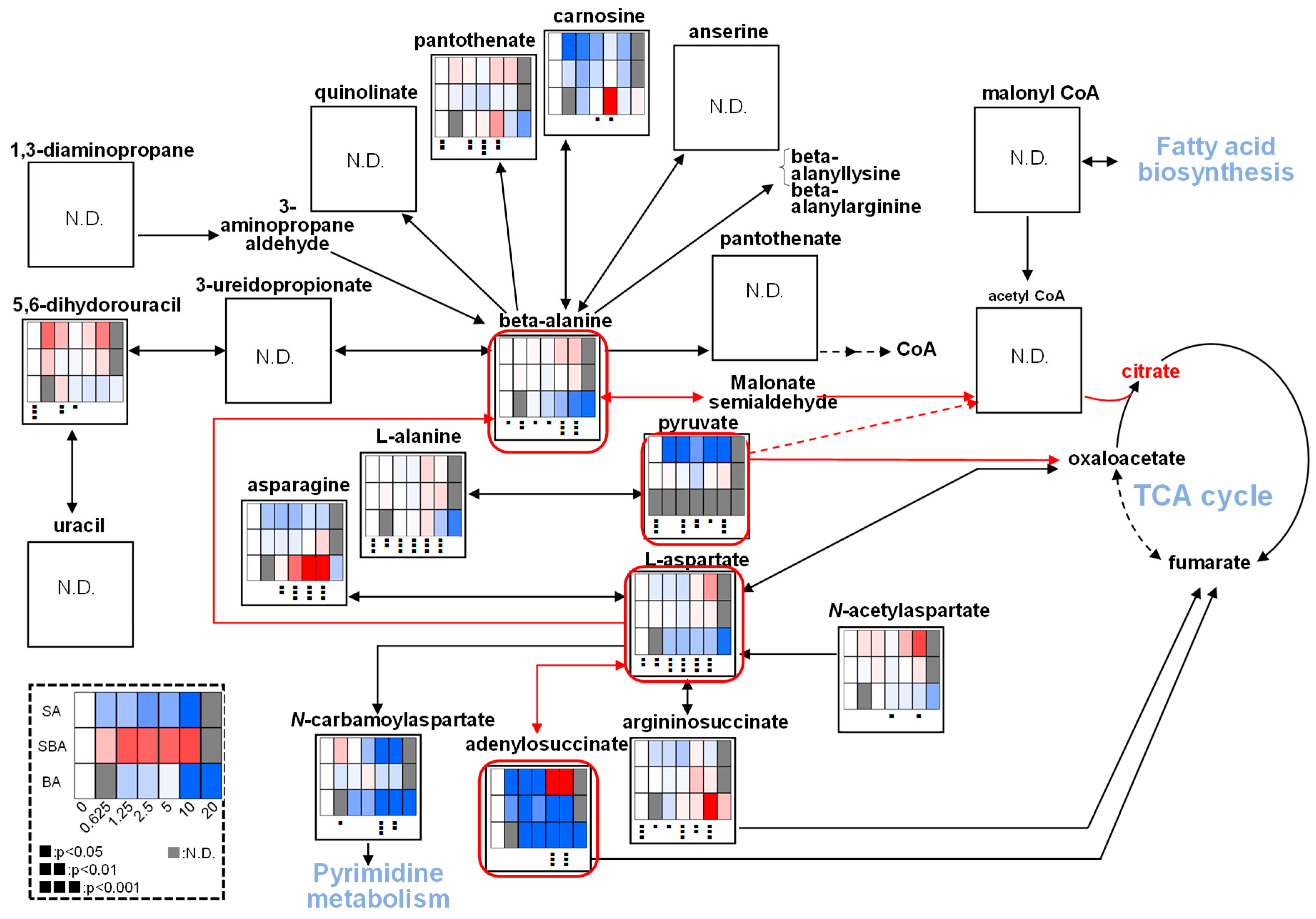
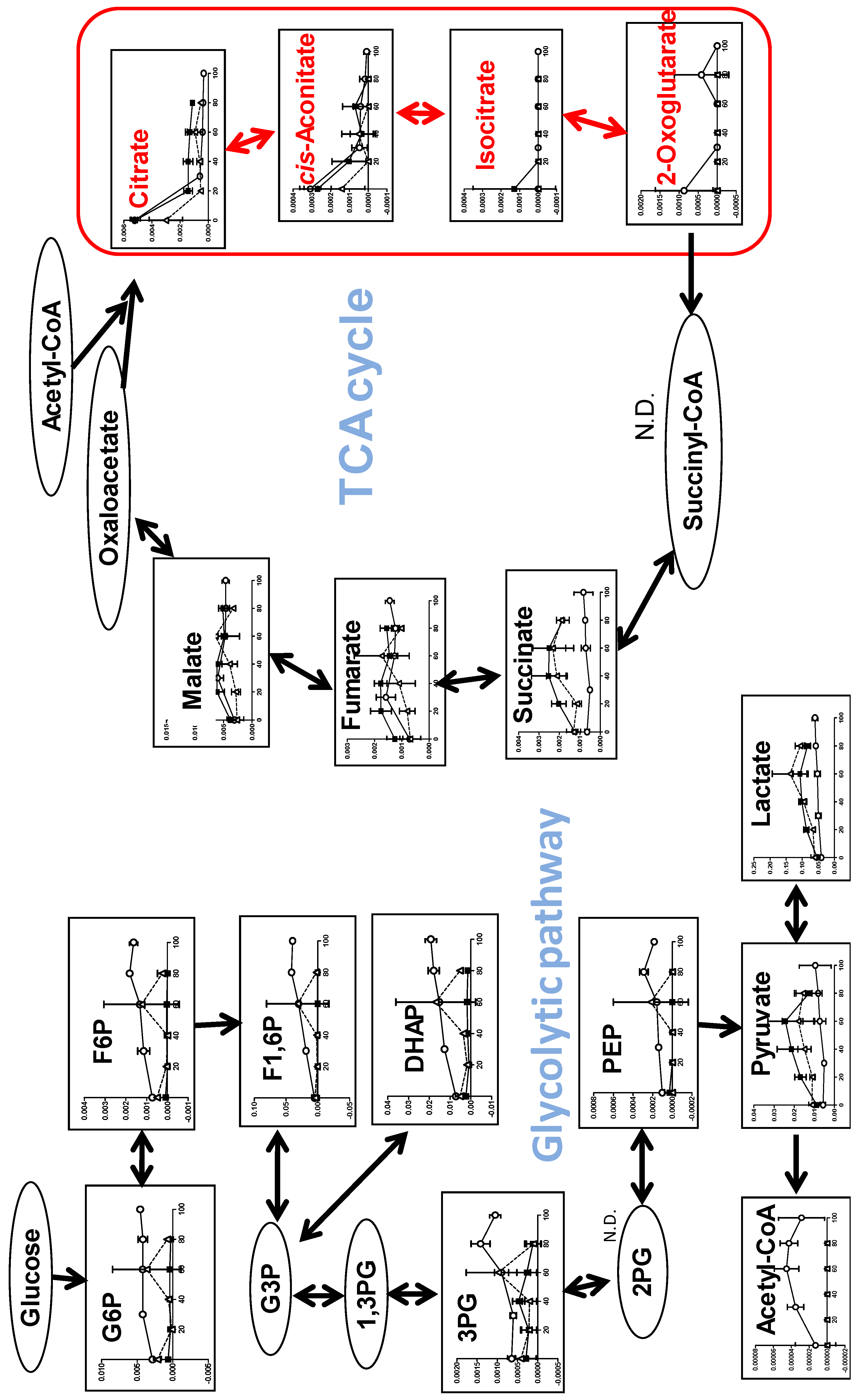
3.1.4. Effect on TCA Cycle Metabolites
3.2. Changes of Metabolic Profiles in Normal Oral Cells Induced by Eugenol
3.2.1. Eugenol Induced Rapid Collapse in Mitochondria
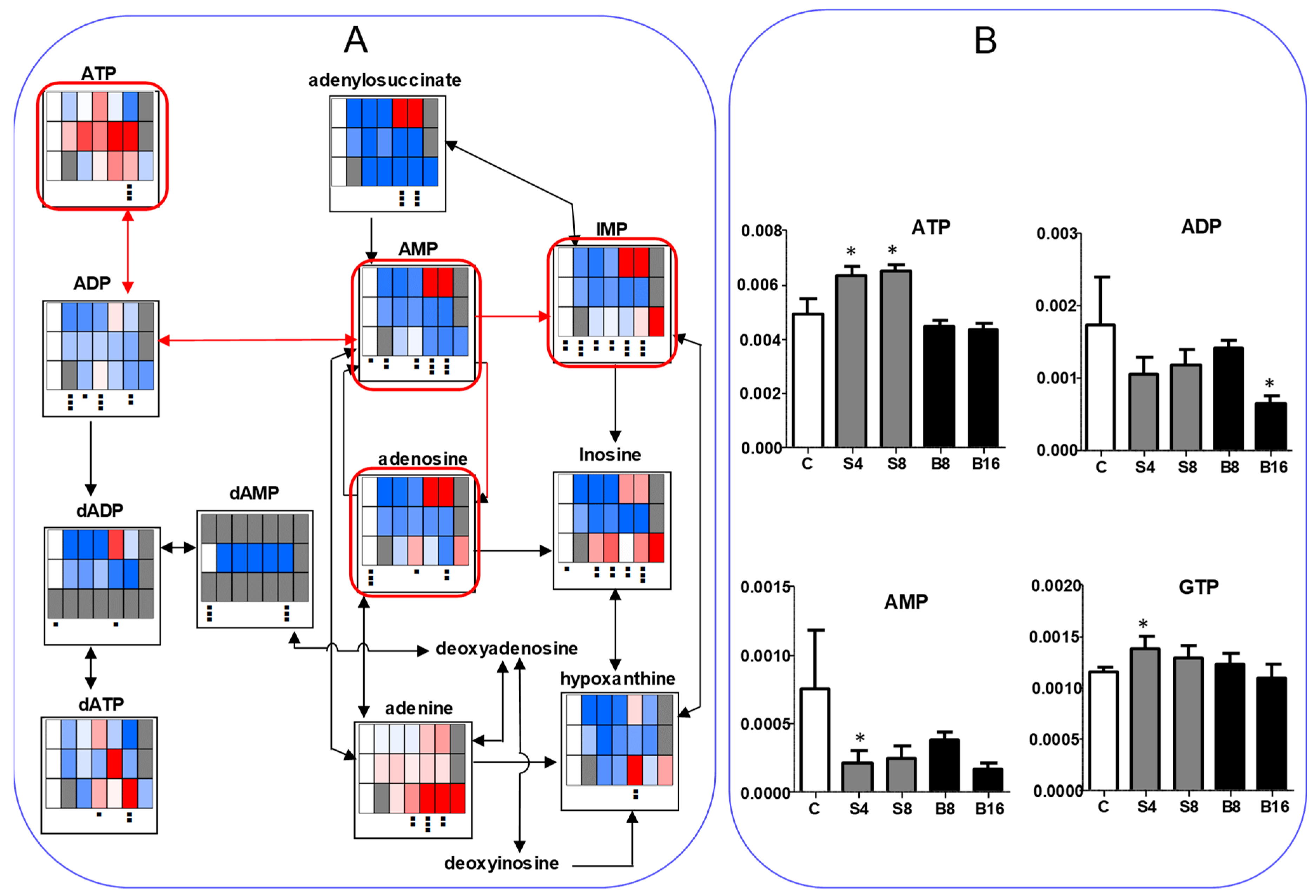
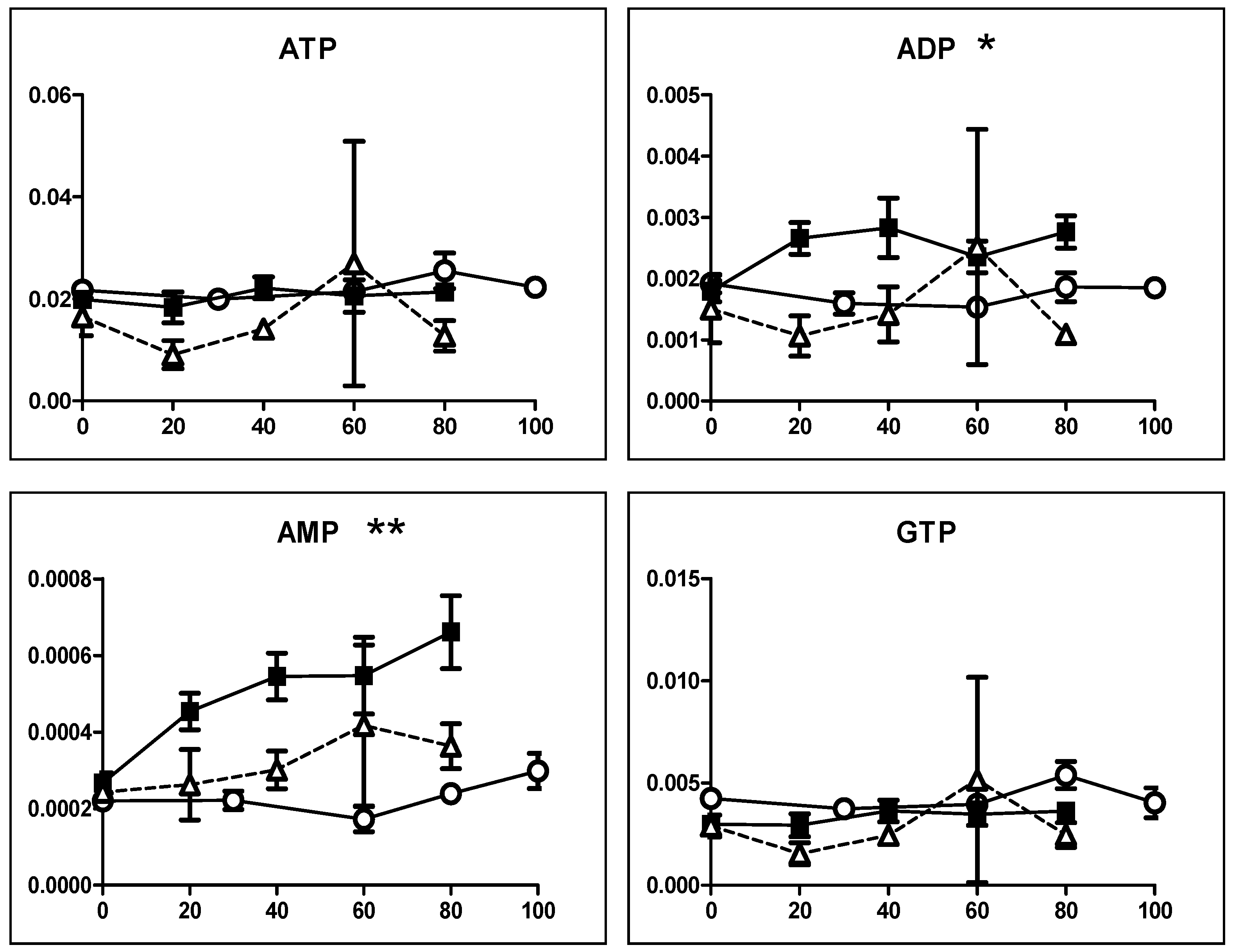
3.2.2. Eugenol Rapidly Suppressed TCA Cycle
4. Discussion
4.1. Inhibition of TCA Cycle by Benzaldehyde (BA) in Malignant Cells
4.2. Inhibition of TCA Cycle by Eugenol in Non-Malignant Cells
4.3. Combination Experiments with Anticancer Drugs
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Takeuchi, S.; Kochi, M.; Sakaguchi, K.; Nakagawa, K.; Mizutani, T. Benzaldehyde as a carcinostatic principle in figs. Agric. Biol. Chem. 1978, 42, 1449–1451. [Google Scholar] [CrossRef]
- Kochi, M.; Takeuchi, S.; Mizutani, T.; Mochizuki, K.; Matsumoto, Y.; Saito, Y. Antitumor activity of benzaldehyde. Cancer Treat. Rep. 1980, 64, 21–23. [Google Scholar]
- Kochi, M.; Isono, N.; Niwayama, M.; Shirakabe, K. Antitumor activity of a benzaldehyde derivative. Cancer Treat. Rep. 1985, 69, 533–537. [Google Scholar] [PubMed]
- Kochi, M.; Ueda, S.; Hagiwara, T. Antitumor activity of sodium benzylidenascorbate. In Progress in Cancer Research and Therapy 35: Hormones and Cancer 3; Bresciani, F., King, R.J.B., Eds.; Raven Press: New York, NY, USA, 1988; pp. 338–343. [Google Scholar]
- Sakagami, H.; Asano, K.; Fukuchi, K.; Gomi, K.; Ota, H.; Kazama, K.; Tanuma, S.; Kochi, M. Induction of tumor degeneration by sodium benzylideneascorbate. Anticancer Res. 1991, 11, 1533–1538. [Google Scholar] [PubMed]
- Sakagami, H.; Sakagami, T.; Takeda, M.; Iwaki, K.; Takeda, K. Determination of sodium 5,6-benzylidene-l-ascorbate and related compounds by high-performance liquid chromatography. J. Chromatogr. Coruña 1993, 653, 37–43. [Google Scholar] [CrossRef]
- Sakagami, H.; Sakagami, T.; Yamamura, M.; Takahashi, H.; Shibuya, I.; Takeda, M. Stability of sodium 5,6-benzylidene-l-ascorbate. Anticancer Res. 1995, 15, 1269–1274. [Google Scholar] [PubMed]
- Kishino, K.; Hashimoto, K.; Amano, O.; Kochi, M.; Sakagami, H. Tumor-specific cytotoxicity and type of cell death induced by sodium 5,6-benzylidene-l-ascorbate. Anticancer Res. 2008, 28, 2577–2584. [Google Scholar] [PubMed]
- Ariyoshi-Kishino, K.; Hashimoto, K.; Amano, O.; Kochi, M.; Sakagami, H. Tumor-specific cytotoxicity and type of cell death induced by benzaldehyde. Anticancer Res. 2010, 30, 5069–5076. [Google Scholar] [PubMed]
- Kuribayashi, N.; Sakagami, H.; Sakagami, T.; Niimi, E.; Shiokawa, D.; Ikekita, M.; Takeda, M.; Tanuma, S. Induction of DNA fragmentation in human myelogenous leukemic cell lines by sodium 5,6-benzylidene-l-ascorbate and its related compounds. Anticancer Res. 1994, 14, 969–976. [Google Scholar] [PubMed]
- Sakagami, H.; Satoh, K.; Kochi, M. Comparative study of the antitumor action between sodium 5,6-bnzylidene-l-ascorbate and sodium ascorbate (Minireview). Anticancer Res. 1997, 17, 4451–4452. [Google Scholar] [PubMed]
- Sakagami, H.; Satoh, K.; Hakeda, Y.; Kumegawa, M. Apoptosis-inducing activity of vitamin C and vitamin K. Cell Mol. Biol. 2000, 46, 129–143. [Google Scholar] [PubMed]
- Sakagami, H.; Kuribayashi, N.; Iida, M.; Hagiwara, T.; Takahashi, H.; Yoshida, H.; Shiota, F.; Ohata, H.; Momose, K.; Takeda, M. The requirement for and mobilization of calcium during induction by sodium ascorbate and by hydrogen peroxide of cell death. Life Sci. 1996, 58, 1131–1138. [Google Scholar] [CrossRef]
- Sakagami, H.; Satoh, K.; Kadofuku, T.; Takeda, M. Methionine oxidation and apoptosis induction by ascorbate, gallate and hydrogen peroxide. Anticancer Res. 1997, 17, 2565–2570. [Google Scholar] [PubMed]
- Sakagami, H.; Hosaka, M.; Arakawa, H.; Maeda, M.; Satoh, K.; Ida, Y.; Asano, K.; Hisamitsu, T.; Takimoto, M.; Ota, H.; et al. Role of hydrogen peroxide in antitumor activity induction by sodium 5,6-benzylidene-L-ascorbate. Anticancer Res. 1998, 18, 2519–2524. [Google Scholar] [PubMed]
- Sweet, C. Procedure for treatment of exposed and pulpless deciduous teeth. J. Am. Dent. Assoc. 1930, 17, 1150–1153. [Google Scholar]
- Primosch, R.E. Primary tooth pulp therapy as taught in predoctoral pediatric dental programs in the United States. Pediatr. Dent. 1997, 19, 118–122. [Google Scholar] [PubMed]
- Hume, W. An analysis of the release and the diffusion through dentin of eugenol from zinc oxide-eugenol mixtures. J. Dent. Res. 1984, 63, 881–884. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.; Zander, H. Pulp healing. J. Dent. Res. 1949, 28, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Barja-Fidalgo, F.; Moutinho-Ribeiro, M.; Oliveira, M.A.A.; de Oliveira, B.H. A systematic review of root canal filling materials for deciduous teeth: Is there an alternative for zinc oxide-eugenol? ISRN Dent. 2011, 2011, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hilton, T.J. Keys to clinical success with pulp capping: A review of the literature. Oper. Dent. 2009, 34, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Hume, W.R. Effect of eugenol on respiration and division in human pulp, mouse fibroblasts, and liver cells in vitro. J. Dent. Res. 1984, 63, 1262–1265. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.; Machino, M.; Murakami, Y.; Umemura, N.; Sakagami, H. Cytotoxicity of dental compounds against human oral squamous cell carcinoma and normal oral cells. In Vivo 2013, 27, 85–96. [Google Scholar] [PubMed]
- Atsumi, T.; Fujisawa, S.; Satoh, K.; Sakagami, H.; Iwakura, I.; Ueha, T.; Sugita, Y.; Yokoe, I. Cytotoxicity and radical intensity of eugenol, isoeugenol or related dimers. Anticancer Res. 2000, 20, 2519–2524. [Google Scholar] [PubMed]
- Jaqanathan, S.K.; Mazumdar, A.; Mondhe, D.; Mandal, M. Apoptotic effect of eugenol in human colon cancer cell lines. Cell Biol. Int. 2011, 35, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Vidhya, N.; Devaraj, S.N. Induction of apoptosis by eugenol in human breast cancer cells. Indian J. Exp. Biol. 2011, 49, 871–878. [Google Scholar] [PubMed]
- Majno, G.; Joris, I. Apoptosis, oncosis, and necrosis. An overview of cell death. Am. J. Pathol. 1995, 146, 3–15. [Google Scholar] [PubMed]
- Halestrap, A.P.; Clarke, S.J.; Javadov, S.A. Mitochondrial permeability transition pore opening during myocardial reperfusion—A target for cardioprotection. Cardiovasc. Res. 2004, 61, 372–385. [Google Scholar] [CrossRef]
- Halestrap, A. Biochemistry: A pore way to die. Nature 2005, 434, 578–579. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, S.; Morshed, S.R.M.; Chowdhury, S.A.; Takayama, F.; Satoh, T.; Hashimoto, K.; Sugiyama, K.; Amano, O.; Yasui, T.; Yokote, Y.; et al. Possible link between glycolysis and apoptosis induced by sodium fluoride. J. Dent. Res. 2005, 84, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, S.; Sugiyama, K.; Amano, O.; Yasui, T.; Sakagami, H. Negative regulation of NaF-induced apoptosis by Bad-CAII complex. Toxicology 2011, 287, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Sugimoto, M.; Tanaka, S.; Onuma, H.; Ota, S.; Kaneko, M.; Soga, T.; Tomita, M. Metabolomic profiling of sodium fluoride-induced cytotoxicity in an oral squamous cell carcinoma cell line. Metabolomics 2014, 10, 270–279. [Google Scholar] [CrossRef]
- Koh, T.; Murakami, Y.; Tanaka, S.; Machino, M.; Onuma, H.; Kaneko, M.; Sugimoto, M.; Soga, T.; Tomita, M.; Sakagami, H. Changes of metabolic profiles in an oral squamous cell carcinoma cell line induced by eugenol. In Vivo 2013, 27, 233–244. [Google Scholar] [PubMed]
- Kantoh, K.; Ono, M.; Nakamura, Y.; Nakamura, Y.; Hashimoto, K.; Sakagami, H.; Wakabayashi, H. Hormetic and anti-radiation effects of tropolone-related compounds. In Vivo 2010, 24, 843–852. [Google Scholar] [PubMed]
- Sakagami, H.; Shimada, C.; Kanda, Y.; Amano, O.; Sugimoto, M.; Ota, S.; Soga, T.; Tomita, M.; Sato, A.; Tanuma, S.; et al. Effects of 3-styrylchromones on metabolic profiles and cell death in oral squamous cell carcinoma cells. Toxocol. Rep. 2015, 2, 1281–1290. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Contreras1, R.; Sugimoto, M.; Umemura, N.; Kaneko, M.; Hatakeyama, Y.; Soga, T.; Tomita, M.; Scougall-Vilchis, R.J.; Contreras-Bulnes, R.; Nakajima, H.; et al. Alteration of metabolomic profiles by titanium dioxide nanoparticles in human gingivitis model. Biomaterials 2015, 57, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Soga, T.; Baran, R.; Suematsu, M.; Ueno, Y.; Ikeda, S.; Sakurakawa, T.; Kakazu, Y.; Ishikawa, T.; Robert, T.; Nishioka, T.; et al. Differential metabolomics reveals ophthalmic acid as an oxidative stress biomarker indicating hepatic glutathione consumption. J. Biol. Chem. 2006, 281, 16768–16776. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Sakagami, H.; Yokote, Y.; Onuma, H.; Kaneko, M.; Mori, M.; Sakaguchi, Y.; Soga, T.; Tomita, M. Non-targeted metabolite profiling in activated macrophage secretion. Metabolomics 2012, 8, 624–633. [Google Scholar] [CrossRef]
- Sugimoto, M.; Wong, D.T.; Hirayama, A.; Soga, T.; Tomita, M. Capillary electrophoresis mass spectrometry-based saliva metabolomics identified oral, breast and pancreatic cancer-specific profiles. Metabolomics 2010, 6, 78–95. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Kawakami, M.; Robert, M.; Soga, T.; Tomita, M. Bioinformatics tools for mass spectroscopy-based metabolomic data processing and analysis. Curr. Bioinform. 2012, 7, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Sugimori, N.; Espinoza, J.L.; Trung, L.Q.; Takami, A.; Kondo, Y.; An, D.T.; Sasaki, M.; Wakayama, T.; Nakao, S. Paraptosis cell death induction by the thiamine analog benfotiamine in leukemia cells. PLoS ONE 2015, 10, e0120709. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.; Murakami, Y.; Tanaka, S.; Machino, M.; Sakagami, H. Re-evaluation of anti-inflammatory potential of eugenol in IL-1β-stimulated gigngival fibroblast and pulp cells. In Vivo 2013, 27, 269–274. [Google Scholar] [PubMed]
- Ma, Y.; Jiang, J.; Gao, Y.; Shi, T.; Zhu, X.; Zhang, K.; Lu, K.; Xue, B. Research progress of the relationship between pyroptosis and disease. Am. J. Transl. Res. 2018, 10, 2213–2219. [Google Scholar] [PubMed]
- Guamán-Ortiz, L.M.; Orellana, M.I.; Ratovitski, E.A. Natural compounds as modulators of non-apoptotic cell death in cancer cells. Curr. Genom. 2017, 18, 132–155. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Okudaira, N.; Masuda, Y.; Amano, O.; Yokose, S.; Kanda, Y.; Suguro, M.; Natori, T.; Oizumi, H.; Oizumi, T. Induction of apoptosis in human oral keratinocyte by doxorubicin. Anticancer Res. 2017, 37, 1023–1029. [Google Scholar] [PubMed]



| Biological Activities | SBA | SA | BA | Ref. |
|---|---|---|---|---|
| Antitumor activity (in vivo) | Yes | No | Yes | [5] |
| Tumor-specificity (TS = CC50 (normal)/CC50 (tumor)) | 2 | 2.5 | 8.8 | [8,9] |
| Apoptosis-induction in HSC-2 cells | No | N.D. | No | [8,9] |
| Apoptosis-induction in HL-60 cells | Yes | Yes | No | [10] |
| Cytotoxicity by addition of copper | Increase | Increase | N.D. | [11,12] |
| Cytotoxicity by addition of iron, cysteine analog, catalase | Not clear | Decrease | N.D. | [11,12,13] |
| Radical generation | Yes | Yes | N.D. | [11,12] |
| Oxidation potential, H2O2 production, methionine oxidation | Yes | Yes | N.D. | [11,12,13,14,15] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakagami, H.; Sugimoto, M.; Kanda, Y.; Murakami, Y.; Amano, O.; Saitoh, J.; Kochi, A. Changes in Metabolic Profiles of Human Oral Cells by Benzylidene Ascorbates and Eugenol. Medicines 2018, 5, 116. https://doi.org/10.3390/medicines5040116
Sakagami H, Sugimoto M, Kanda Y, Murakami Y, Amano O, Saitoh J, Kochi A. Changes in Metabolic Profiles of Human Oral Cells by Benzylidene Ascorbates and Eugenol. Medicines. 2018; 5(4):116. https://doi.org/10.3390/medicines5040116
Chicago/Turabian StyleSakagami, Hiroshi, Masahiro Sugimoto, Yumiko Kanda, Yukio Murakami, Osamu Amano, Junko Saitoh, and Atsuko Kochi. 2018. "Changes in Metabolic Profiles of Human Oral Cells by Benzylidene Ascorbates and Eugenol" Medicines 5, no. 4: 116. https://doi.org/10.3390/medicines5040116
APA StyleSakagami, H., Sugimoto, M., Kanda, Y., Murakami, Y., Amano, O., Saitoh, J., & Kochi, A. (2018). Changes in Metabolic Profiles of Human Oral Cells by Benzylidene Ascorbates and Eugenol. Medicines, 5(4), 116. https://doi.org/10.3390/medicines5040116








