Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils
Abstract
1. Introduction
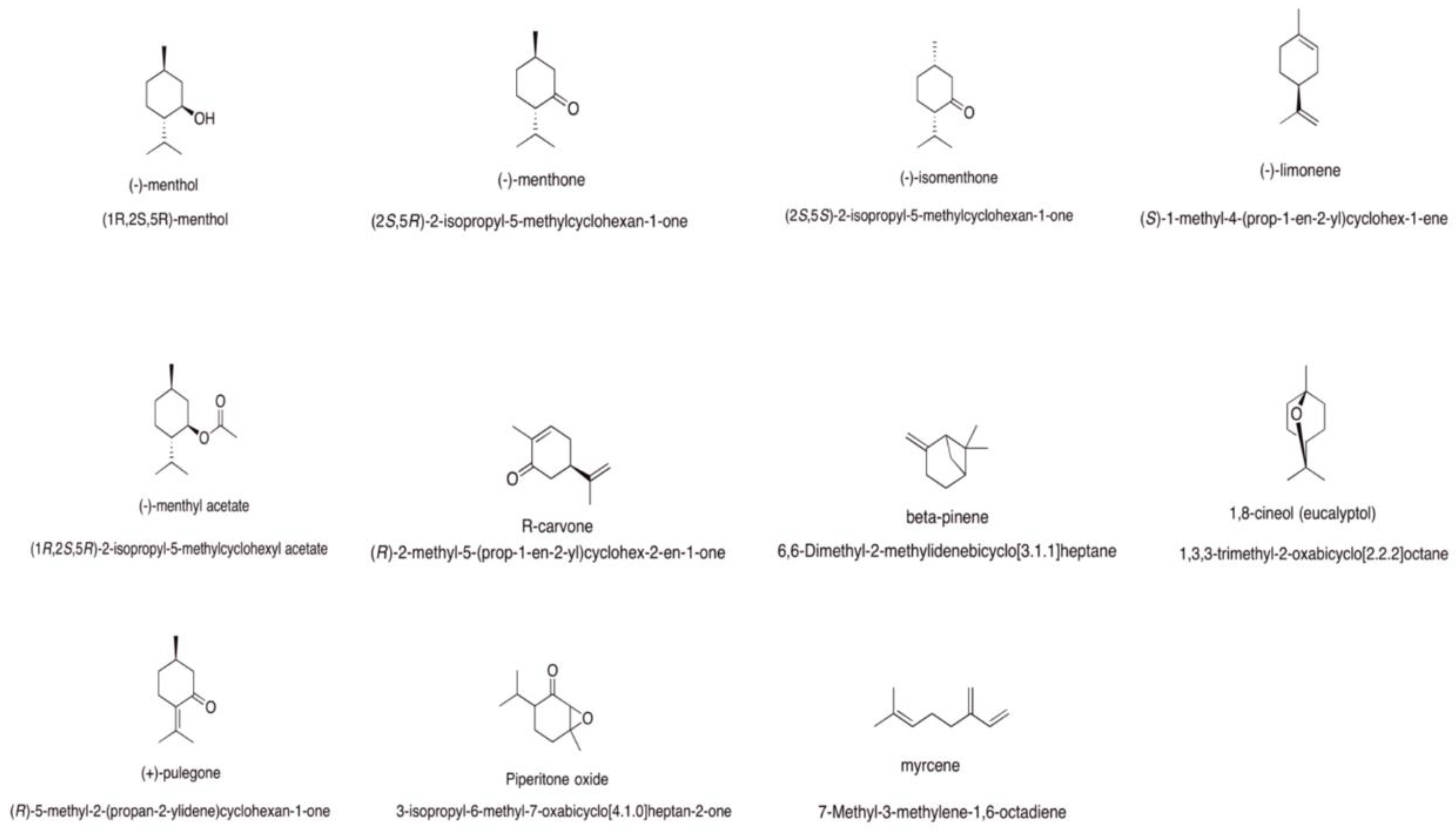
Mentha spp. Essential Oils
2. Antioxidant Properties
3. Antifungal Properties
4. Antibiofilm Properties
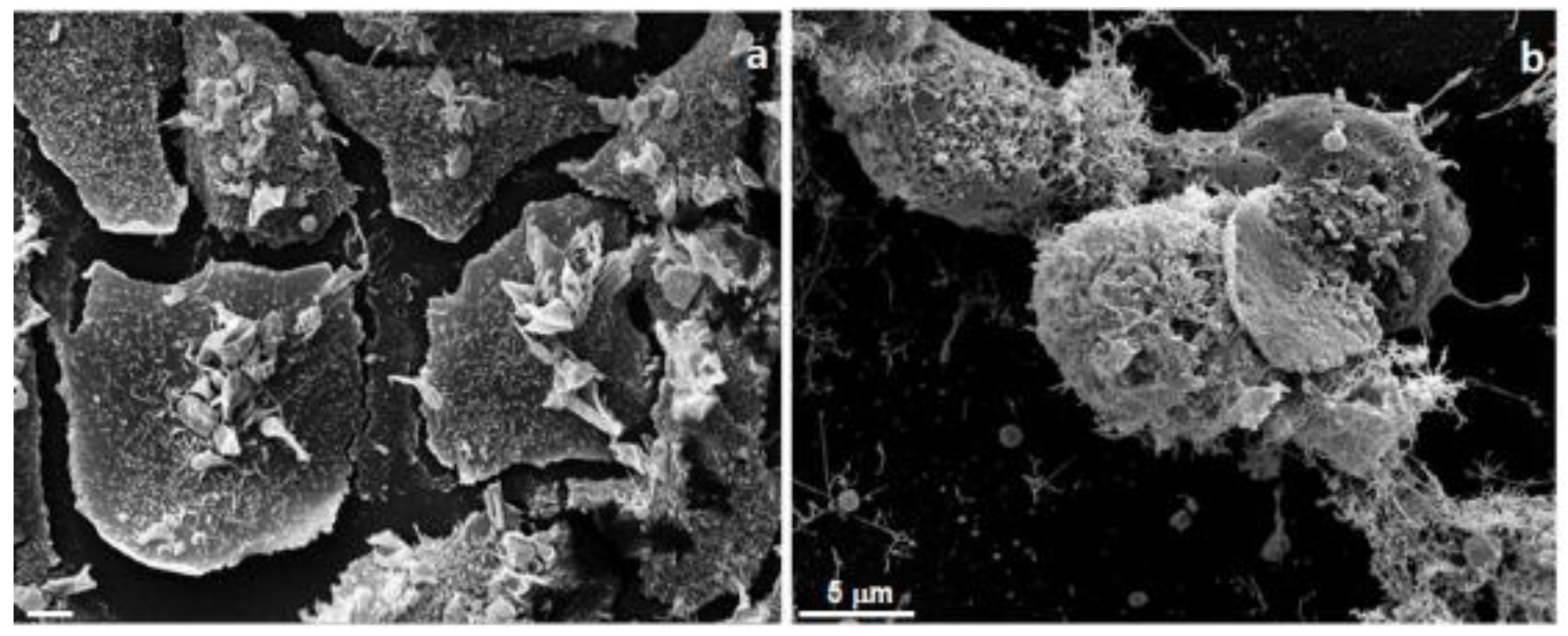
5. Cytotoxic Activity
6. Side Effects and Toxicity
7. Conclusions
Funding
Conflicts of Interest
References
- Prabuseenivasan, S.; Jayakumar, M.; Ignacimuthu, S. In vitro antibacterial activity of some plant essential oils. BMC Complement. Altern. Med. 2006, 6, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Aburjai, T.; Natsheh, F.M. Plants used in cosmetics. Phytother. Res. 2003, 17, 987–1000. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.J. Antimicrobial properties of plant secondary metabolites. Proc. Nutr. Soc. 2004, 63, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Nakatsu, T.; Lupo, A.T.; Chinn, J.W.; Kang, R.K.L. Studies in Natural Products Chemistry; Elsevier: New York, NY, USA, 2000; Volume 21, pp. 571–631. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Edris, A. Pharmaceutical and therapeutic potentials of essential oils and their individuals’ volatile constituents. A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef] [PubMed]
- Garzoli, S.; Pirolli, A.; Vavala, E.; Di Sotto, A.; Sartorelli, G.; Bozovic, M.; Angiolella, L.; Mazzanti, G.; Pepi, F.; Ragno, R. Multidisciplinary approach to determine the optimal time and period to extract the essential oil from Mentha suaveolens ehrh. Molecules 2015, 20, 9640–9655. [Google Scholar] [CrossRef] [PubMed]
- Nieto, G.; Ros, G.; Castillo, J. Antioxidant and Antimicrobial Properties of Rosemary (Rosmarinus officinalis, L.): A Review. Medicines 2018, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Tucker, A.O.; Naczi, R.F.C. Mentha: An overview of its classification and relationships. In Mint: The Genus Mentha: Medicinal and Aromatic Plants-Industrial Profiles; Lawrence, B.M., Ed.; CRC Press: Boca Raton, FL, USA, 2006; p. 3. [Google Scholar]
- Sakkas, H.; Papadopoulou, C. Antimicrobial Activity of Basil, Oregano, and Thyme Essential Oils. J. Microbiol. Biotechnol. 2017, 27, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, I.T.A.; Furlanetti, V.F.; Anibal, P.C.; Duarte, M.C.T.; Höfling, J.F. Potential pharmacological and toxicological basis of the essential oil from Mentha spp. Rev. Ciênc. Farm. Básica Apl. 2009, 30, 235–239. [Google Scholar]
- Chávez-González, M.L.; Rodríguez-Herrera, R.; Aguilar, C.N. Essential oils: A natural alternative to combat antibiotics resistance antibiotic resistance in mechanisms and new antimicrobial approaches. In Antibiotic Resistance; Kateryna, K., Mahendra, R., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 227–237. [Google Scholar]
- Sharma, V.; Hussain, S.; Gupta, M.; Saxena, A. In vitro anticancer activity of extracts of Mentha spp. against human cancer cells. Indian J. Biochem. Biophys. 2014, 51, 416–419. [Google Scholar] [PubMed]
- Amabeoku, G.J.; Erasmus, S.J.; Ojewole, J.A.; Mukinda, J.T. Antipyretic and antinociceptive properties of Mentha longifolia Huds. (Lamiaceae) leaf aqueous extract in rats and mice. Meth. Find. Exp. Clin. Pharmacol. 2009, 31, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Yadegarinia, D.; Gachkar, L.; Rezaei, M.B.; Taghizadeh, M.; Astaneh, S.A.; Rasooli, I. Biochemical activities of Iranian Mentha piperita L. and Myrtus communis L. essential oils. Phytochemistry 2006, 67, 1249–1255. [Google Scholar] [CrossRef] [PubMed]
- Gonzales-Burgos, E.; Gomez-Serranillos, M.P. Terpene compounds in nature. A review of their potential antioxidant activity. Curr. Med. Chem. 2012, 19, 5319–5341. [Google Scholar] [CrossRef]
- Kaufmann, H.; Dorhoi, A. Molecular determinants in phagocyte-bacteria interactions. Immunity 2016, 44, 476–491. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Cunningham-Bussel, A. Beyond oxidative stress: An immunologist’s guide to reactive oxygen species. Nat. Rev. Immunol. 2013, 13, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Dawidowicz, A.L.; Olszowy, M. Does antioxidant properties of the main component of essential oil reflect its antioxidant properties? The comparison of antioxidant properties of essential oils and their main components. Nat. Prod. Res. 2014, 28, 1952–1963. [Google Scholar] [CrossRef] [PubMed]
- Pisoschi, A.M.; Negulescu, G.P. Methods for total antioxidant activity determination: A review. Biochem. Anal. Biochem. 2011, 1. [Google Scholar] [CrossRef]
- Olszowy, M.; Dawidowicz, A.L. Essential oils as antioxidants: Their evaluation by DPPH, ABTS, FRAP, CUPRAC, and β-carotene bleaching methods. Monatsh. Chem. 2016, 147, 2083–2091. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Byrne, D.H. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Marc, F.; Davin, A.; Deglène-Benbrahim, L.; Ferrand, C.; Baccaunaud, M.; Fritsch, P. Studies of several analytical methods for antioxidant potential evaluation in food. Med. Sci. 2004, 20, 458–463. [Google Scholar]
- Pellegrini, N.; Serafini, M.; Colombi, B.; Del Rio, D.; Salvatore, S.; Bianchi, M.; Brighenti, F. Total antioxidant capacity of plant foods, beverages and oils consumed in Italy assessed by three different in vitro assays. J. Nutr. 2003, 133, 2812–2819. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Yin, J.J.; Charles, D.; Zhou, K.; Moore, J.; Yu, L.L. Total phenolic contents, chelating capacities, and radical-scavenging properties of black peppercorn, nutmeg, rosehip, cinnamon and oregano leaf. Food Chem. 2007, 100, 990–997. [Google Scholar] [CrossRef]
- Amorati, R.; Foti, M.C.; Valgimigli, L. Antioxidant activity of essential oils. J. Agric. Food Chem. 2013, 61, 10835–10847. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.; Bail, S.; Buchbauer, G.; Stoilova, I.; Atanasova, T.; Stoyanova, A.; Krastanov, A.; Jirovetz, L. Chemical composition, olfactory evaluation and antioxidant effects of essential oil from Mentha x piperita. Nat. Prod. Commun. 2009, 4, 1107–1112. [Google Scholar] [PubMed]
- Yang, S.A.; Jeon, S.K.; Lee, E.J.; Shim, C.H.; Lee, I.S. Comparative study of the chemical composition and antioxidant activity of six essential oils and their components. Nat. Prod. Res. 2010, 24, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Wang, H.; Wang, J.; Zhou, L.; Yang, P. Chemical composition and anti-inflammatory, cytotoxic and antioxidant activities of essential oil from leaves of Mentha piperita grown in China. PLoS ONE 2014, 9, e114767. [Google Scholar] [CrossRef] [PubMed]
- da Silva Ramos, R.; Lobato Rodrigues, A.B.; Ferreira Farias, A.L.; Simões, R.C.; Pinheiro, M.T.; dos Anios Ferreira, R.M.; Costa Barbosa, L.M.; Picanço Souto, R.N.; Fernandes, J.B.; da Silvas Santos, L.; et al. Chemical composition and in vitro antioxidant, cytotoxic, antimicrobial, and larvicidal activities of the essential oil of Mentha piperita L. (Lamiaceae). Sci. World J. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, M.; Ricci, A.; Serio, A.; Chaves-López, C.; Mazzarrino, G.; D’Amato, S.; Lo Sterzo, C.; Paparella, A. Characterization of Essential Oils Obtained from Abruzzo Autochthonous Plants: Antioxidant and Antimicrobial Activities Assessment for Food Application. Foods 2018, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Dorman, H.; Koşar, M.; Başer, K.; Hiltunen, R. Phenolic profile and antioxidant evaluation of Mentha x piperita L. (peppermint) extracts. Nat. Prod. Commun. 2009, 4, 535–542. [Google Scholar] [PubMed]
- Sroka, Z.; Fecka, I.; Cisowski, W. Antiradical and Anti-H2O2 properties of polyphenolic compounds from anaqueous peppermint extract. Z. Naturforsch. C 2005, 60, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, P.; Cardoso, T.; Ferreira, F.; Fernandes-Ferreira, M.; Piper, P.; Sousa, M.J. Mentha piperita essential oil induces apoptosis in yeast associated with both cytosolic and mitochondrial ROS-mediated damage. FEMS Yeast Res. 2014, 14, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- Kamkar, A.; Javan, A.J.; Asadi, F.; Kamalinejad, M. The antioxidative effect of Iranian Mentha pulegium extracts and essential oil in sunflower oil. Food Chem. Toxicol. 2010, 48, 1796–1800. [Google Scholar] [CrossRef] [PubMed]
- Cherrat, L.; Espina, L.; Bakkali, M.; Pagan, R.; Laglaoui, A. Chemical composition, antioxidant and antimicrobial propertie of Mentha pulegium, Lavandula stoechas and Satureja calamintha Scheele essential oils and an evaluation of their bactericidal effect in combined processes. Innov. Food Sci. Emerg. Technol. 2014, 22, 221–229. [Google Scholar] [CrossRef]
- Abdelli, M.; Moghrani, H.; Aboun, A.; Maachi, R. Algerian Mentha pulegium L. leaves essential oil: Chemical composition, antimicrobial, insecticidal and antioxidant activities. Ind. Crops Prod. 2016, 94, 197–205. [Google Scholar] [CrossRef]
- Bouyahya, A.; Et-Touys, A.; Bakri, Y.; Talbaui, A.; Fellah, H.; Abrini, J.; Dakka, N. Chemical composition of Mentha pulegium and Rosmarinus officinalis essential oils and their antileishmanial, antibacterial and antioxidant activities. Microb. Pathog. 2017, 111, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Snoussi, M.; Noumi, E.; Trabelsi, N.; Flamini, G.; Papetti, A.; De Feo, V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules 2015, 20, 14402–14424. [Google Scholar] [CrossRef] [PubMed]
- Bardaweel, S.K.; Bakchiche, B.; AL-Salamat, H.A.; Rezzoug, M.; Gherib, A.; Flamini, G. Chemical composition, antioxidant, antimicrobial and Antiproliferative activities of essential oil of Mentha spicata L. (Lamiaceae) from Algerian Saharan atlas. BMC Complement. Altern. Med. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Eissa, T.F.; González-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. Compositional analysis and in vitro protective activity against oxidative stress of essential oils from egyptian plants used in traditional medicine. Nat. Prod. Commun. 2014, 9, 1377–1382. [Google Scholar] [PubMed]
- El-Askary, H.I.; El-Kashoury, E.A.; Kandil, Z.A.; Salem, M.A.; Ezzat, S.M. Biological activity and standardization of the ethanolic extract of the aerial parts of Mentha suaveolens Ehrh. World J. Pharm. Pharm. Sci. 2014, 3, 223–241. [Google Scholar]
- Ferreira, A.; Proenc, C.; Serralheiro, M.L.M.; Ara´ujo, M.E.M. The in vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from Portugal. J. Ethnopharmacol. 2006, 108, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Sitzmann, J.; Habegger, R.; Schnitzler, W.H.; Grassmann, J. Comparative analysis of antioxidant activities of fourteen Mentha essential oils and their components. Chem. Biodivers. 2014, 11, 1978–1989. [Google Scholar] [CrossRef] [PubMed]
- Spagnoletti, A.; Guerrini, A.; Tacchini, M.; Vinciguerra, V.; Leone, C.; Maresca, I.; Simonetti, G.; Sacchetti, G.; Angiolella, L. Chemical Composition and Bio-efficacy of Essential Oils from Italian Aromatic Plants: M. suaveolens, C. capitatus, O. hirtum and R. officinalis. Nat. Prod. Commun. 2016, 11, 1517–1520. [Google Scholar]
- Abad, M.J.; Ansuategui, M.; Bermejo, P. Active antifungal substances from natural sources. Arkivoc 2007, 7, 6–145. [Google Scholar]
- Pfaller, M.A. Antifungal drug resistance: Mechanisms, epidemiology, and consequences for treatment. Am. J. Med. 2012, 125, S3–S13. [Google Scholar] [CrossRef] [PubMed]
- Weitzman, I.; Summerbell, R.C. The Dermatophytes. Clin. Microbiol. Rev. 1995, 8, 240–259. [Google Scholar] [CrossRef] [PubMed]
- Mimica-Dukić, N.; Bozin, B.; Soković, M.; Mihajlović, B.; Matavulj, M. Antimicrobial and antioxidant activities of three Mentha species essential oils. Planta. Med. 2003, 69, 413–419. [Google Scholar] [PubMed]
- Tampieri, M.P.; Galuppi, R.; Macchioni, F.; Carelle, M.S.; Falcioni, L.; Cioni, P.L.; Morelli, I. The inhibition of Candida albicans by selected essential oils and their major components. Mycopathologia 2005, 159, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Samber, N.; Khan, A.; Varma, A.; Manzoor, N. Synergistic anti-candidal activity and mode of action of Mentha piperita essential oil and its major components. Pharm. Biol. 2015, 53, 1496–1504. [Google Scholar] [CrossRef] [PubMed]
- Freire, J.C.P.; Júnior, J.K.O.; Silva, D.F.; de Sousa, J.P.; Guerra, F.Q.S.; de Oliveira Lima, E. Antifungal Activity of Essential Oils against Candida albicans Strains Isolated from Users of Dental Prostheses. Evid. Based Complement. Alternat. Med. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Soković, M.D.; Vukojević, J.; Marin, P.D.; Brkić, D.D.; Vajs, V.; van Griensven, L.J. Chemical composition of essential oils of Thymus and Mentha species and their antifungal activities. Molecules 2009, 14, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Saharkhiz, M.J.; Motamedi, M.; Zomorodian, K.; Pakshir, K.; Miri, R.; Hemyari, K. Chemical composition, antifungal and antibiofilm activities of the essential oil of Mentha piperita L. ISRN Pharm. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Rajkowska, K.; Otlewska, A.; Kunicka-Styczyńska, A.; Krajewska, A. Candida albicans Impairments Induced by Peppermint and Clove Oils at Sub-Inhibitory Concentrations. Int. J. Mol. Sci. 2017, 18, 1307. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.Y.; Abd El-Salam, M.M. Anti-dermatophyte efficacy and environmental safety of some essential oils commercial and in vitro extracted pure and combined against four keratinophilic pathogenic fungi. Environ. Health Prev. Med. 2015, 20, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Ebani, V.V.; Najar, B.; Bertelloni, F.; Pistelli, L.; Mancianti, F.; Nardoni, S. Chemical composition and in vitro antimicrobial efficacy of sixteen essential oils against Escherichia coli and Aspergillus fumigatus. Vet. Sci. 2018, 5, 62. [Google Scholar] [CrossRef] [PubMed]
- Powers, C.N.; Osier, J.L.; McFeeters, R.L.; Brazell, C.B.; Olsen, E.L.; Moriarity, D.M.; Satyal, P.; Setzer, W.N. Antifungal and cytotoxic activities of sixty commercially-available essential oils. Molecules 2018, 23, 1549. [Google Scholar] [CrossRef] [PubMed]
- Adam, K.; Sivropoulou, A.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antifungal Activities of Origanum vulgare subsp. hirtum, Mentha spicata, Lavandula angustifolia, and Salvia fruticosa Essential Oils against Human Pathogenic Fungi. J. Agric. Food Chem. 1998, 46, 1739−1745. [Google Scholar] [CrossRef]
- Khoury, M.; Stien, D.; Eparvier, V.; Ouaini, N.; El Beyrouthy, M. Report on the medicinal use of eleven lamiaceae species in Lebanon and Rationalization of their antimicrobial potential by examination of the chemical composition and antimicrobial activity of their essential oils. Evid. Based Complement. Alternat. Med. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Nardoni, S.; Giovanelli, S.; Pistelli, L.; Mugnaini, L.; Profili, G.; Pisseri, F.; Mancianti, F. In Vitro Activity of Twenty Commercially Available, Plant-Derived Essential Oils against Selected Dermatophyte Species. Nat. Prod. Commun. 2015, 10, 1473–1478. [Google Scholar] [PubMed]
- Ertaş, A.; Gören, A.C.; Haşimi, N.; Tolan, V.; Kola, U. Evaluation of Antioxidant, Cholinesterase Inhibitory and Antimicrobial Properties of Mentha longifolia subsp. noeana and Its Secondary Metabolites. Rec. Nat. Prod. 2015, 9, 105–115. [Google Scholar]
- Mahboubi, M.; Haghi, G. Antimicrobial activity and chemical composition of Mentha pulegium L. essential oil. J. Ethnopharmacol. 2008, 119, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, M.J.; Vincente, A.M.; Cavaleiro, C.; Salgueiro, L. Composition and antifungal activity of the essential oil of Mentha cervina from Portugal. Nat. Prod. Res. 2007, 21, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Oumzil, H.; Ghoulami, S.; Rhajaoui, M.; Ilidrissi, A.; Fkih-Tetouani, S.; Faid, M.; Benjouad, A. Antibacterial and antifungal activity of essential oils of Mentha suaveolens. Phytother. Res. 2002, 16, 727–731. [Google Scholar] [CrossRef] [PubMed]
- Angiolella, L.; Vavala, E.; Sivric, S.; D’Auria, F.D.; Ragno, R. In vitro activity of Mentha suaveolens essential oil against Cryptococcus neoformans and dermatophytes. Int. J. Essent. Oil Ther. 2010, 4, 35–36. [Google Scholar]
- Pietrella, D.; Angiolella, L.; Vavala, E.; Rachini, A.; Mondello, F.; Ragno, R.; Bistoni, F.; Vecchiarelli, A. Beneficial effect of Mentha suaveolens essential oil in the treatment of vaginal candidiasis assessed by real-time monitoring of infection. BMC Complement. Altern. Med. 2011, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Stringaro, A.; Vavala, E.; Colone, M.; Pepi, F.; Mignogna, G.; Garzoli, S.; Cecchetti, S.; Ragno, R.; Angiolella, L. Effects of Mentha suaveolens essential oil alone or in combination with other drugs in Candida albicans. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Scazzocchio, F.; Garzoli, S.; Conti, C.; Leone, C.; Renaioli, C.; Pepi, F.; Angiolella, L. Properties and limits of some essential oils: chemical characterization, antimicrobial activity, interaction with antibiotics and cytotoxicity. Nat. Prod. Res. 2016, 30, 1909–1918. [Google Scholar] [CrossRef] [PubMed]
- Douglas, L.J. Candida biofilms and their role in infection. Trends Microbiol. 2003, 11, 30–36. [Google Scholar] [CrossRef]
- Ramage, G.; Rajendran, R.; Sherry, L.; Williams, C. Fungal biofilm resistance. Int. J. Microbiol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Rodrigues, C.F.; Araújo, D.; Rodrigues, M.E.; Henriques, M. Candida species Biofilms’ antifungal resistance. J. Fungi 2017, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Cu’ellar-Cruz, M.; Lopez-Romero, E.; Villagomez-Castro, J.C.; Ruiz-Baca, E. Candida species: new insights into biofilm formation. Future Microbiol. 2012, 7, 755–771. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Lal, P.; Pruthi, V. Prevention of Candida albicans biofilm by plant oils. Mycopathologia 2008, 165, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Busato de Feiria, S.N.; de Laet Santana, P.; Boni, G.C.; Anibal, P.C.; Gomes Boriollo, M.F.; Figueira, G.M.; de Oliveira Sousa, I.M.; Pereira, B.; Foglio, M.A.; Höfling, J.F. Essential oil composition of Mentha spp. extracted seasonally and their effects against Candida yeast growth and biofilm formation. Adv. Med. Plant Res. 2016, 4, 106–115. [Google Scholar]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential oils and their constituents as anticancer agents: A mechanistic view. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Vtorushin, S.V.; Khristenko, K.Y.; Zavyalova, M.V.; Perelmuter, V.M.; Litviakov, N.V.; Denisov, E.V.; Dulesova, A.Y.; Cherdyntseva, N.V. The phenomenon of multidrug resistance in the treatment of malignant tumors. Exp. Oncol. 2014, 36, 144–156. [Google Scholar] [PubMed]
- Lesgards, J.F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer Activities of Essential Oils Constituents and Synergy with Conventional Therapies: A Review. Phytother. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; So, I.; Chun, J.N.; Jeon, J.H. The antitumor effects of geraniol: Modulation of cancer hallmark pathways. Int. J. Oncol. 2016, 48, 1772–1782. [Google Scholar]
- Carnesecchi, S.; Bras-Gonçalves, R.; Bradaia, A.; Zeisel, M.; Gossé, F.; Poupon, M.F.; Raul, F. Geraniol, a component of plant essential oils, modulates DNA synthesis and potentiates 5-fluorouracil efficacy on human colon tumor xenografts. Cancer Lett. 2004, 215, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Sun, J. D-Limonene: Safety and clinical applications. Altern. Med. Rev. 2007, 12, 259–264. [Google Scholar] [PubMed]
- Slamenova, D.; Horvathova, E.; Sramkova, M.; Marsalkova, L. DNA-protective effects of two components of essential plant oils carvacrol and thymol on mammalian cells cultured in vitro. Neoplasma 2007, 54, 108–112. [Google Scholar] [PubMed]
- Brahmi, F.; Hadj-Ahmed, S.; Zarrouk, A.; Bezine, M.; Nury, T.; Madani, K.; Chibane, M.; Vejux, A.; Andreoletti, P.; Boulekbache-Makhlouf, L.; et al. Evidence of biological activity of Mentha species extracts on apoptotic and autophagic targets on murine RAW264.7 and human U937 monocytic cells. Pharm. Biol. 2017, 55, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Hajighasemi, F.; Hashemi, V.; Khoshzaban, F. Cytotoxic effect of Mentha spicata aqueous extract on cancerous cell lines in vitro. J. Med. Plants Res. 2011, 5, 5142–5147. [Google Scholar]
- Abdel-Hameed, E.-S.S.; Salman, M.S.; Fadl, M.A.; Elkhateeb, A.; Hassan, M.M. Chemical composition and biological activity of Mentha longifolia L. essential oil growing in taif, KSA extracted by hydrodistillation, solvent free microwave and microwave hydrodistillation. J. Essent. Oil-Bear. Plants 2018, 21, 1–14. [Google Scholar] [CrossRef]
- Mangone, C.; Colone, M.; Calcabrini, A.; Maggi, F.; Iannarelli, R.; Rasoanaivo, P.; Nicoletti, M.; Angiolella, L.; Stringaro, A. Antitumoral activities of three essential oils. Natural 2015, 146, 73. [Google Scholar]
- Colone, M.; Calcabrini, A.; Unfer, V.; Stringaro, A. Contribution of electron microscopy to study in vitro inositol effects on human spermatozoa. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4–9. [Google Scholar] [PubMed]
- Condello, M.; Caraglia, M.; Castellano, M.; Arancia, G.; Meschini, S. Structural and functional alterations of cellular components as revealed by electron microscopy. Microsc. Res. Tech. 2013, 76, 1057–1069. [Google Scholar] [CrossRef] [PubMed]
- Soylu, E.M.; Soylu, S.; Kurt, S. Antimicrobial activities of the essential oils of various plants against tomato late blight disease agent Phytophthora infestans. Mycopathologia 2006, 161, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Santoro, G.F.; das Graças Cardoso, M.; Guimarães, L.G.; Mendonça, L.Z.; Soares, M.J. Trypanosoma cruzi: activity of essential oils from Achillea millefolium L., Syzygium aromaticum L. and Ocimum basilicum L. on epimastigotes and trypomastigotes. Exp. Parasitol. 2007, 116, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Santoro, G.F.; das Graças Cardoso, M.; Guimarães, L.G.; Salgado, A.P.; Menna-Barreto, R.F.; Soares, M.J. Effect of oregano (Origanum vulgare L.) and thyme (Thymus vulgaris L.) essential oils on Trypanosoma cruzi (Protozoa: Kinetoplastida) growth and ultrastructure. Parasitol. Res. 2007, 100, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Giordani, C.; Molinari, A.; Toccacieli, L.; Calcabrini, A.; Stringaro, A.; Chistolini, P.; Arancia, G.; Diociaiuti, M. Interaction of Tea Tee Oil with model and cellular membranes. J. Med. Chem. 2006, 49, 4581–4588. [Google Scholar] [CrossRef] [PubMed]
- Maggi, F.; Randriana, R.F.; Rasoanaivo, P.; Nicoletti, M.; Quassinti, L.; Bramucci, M.; Lupidi, G.; Petrelli, D.; Vitali, A.; Papa, F.; et al. Chemical Composition and in vitro Biological Activities of the Essential Oil of Vepris macrophylla (Baker) I.Verd. Endemic to Madagascar. Chem. Biodivers. 2013, 10, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.; Schlegel, J. Mitochondrial calcium release induced by prooxidants. Toxicol. Lett. 1993, 67, 119–127. [Google Scholar] [CrossRef]
- Novgorodov, S.A.; Gudz, T.I. Permeability transition pore of the inner mitochondrial membrane can operate in two open states with different selectivities. J. Bioenerg. Biomembr. 1996, 28, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Vercesi, A.E.; Kowaltowski, A.J.; Grijalba, M.T.; Meinicke, A.R.; Castilho, R.F. The role of reactive oxygen species in mitochondrial permeability transition. Biosci. Rep. 1997, 17, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.S. Mitochondrial membrane permeabilization: The sine qua non for cell death. Bioessays 2006, 28, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Vigan, M. Essential oils: Renewal of interest and toxicity. Eur. J. Dermatol. 2010, 20, 685–692. [Google Scholar] [PubMed]
- Amin, G.; Sourmaghi, M.H.; Jaafari, S.; Hadjagaee, R.; Yazdinezhad, A. Influence of phenological stages and method of distillation on Iranian cultivated Bay leaves volatile oil. Pak. J. Biol. Sci. 2007, 10, 2895–2899. [Google Scholar] [PubMed]
- Freire, C.M.; Marques, M.O.; Costa, M. Effects of seasonal variation on the central nervous system activity of Ocimum gratissimum L. essential oil. J. Ethnopharmacol. 2006, 105, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Nair, B. Final report on the safety assessment of Mentha piperita (Peppermint) Oil, Mentha piperita (Peppermint) leaf extract, Mentha piperita (Peppermint) leaf, and Mentha piperita (Peppermint) leaf water. Int. J. Toxicol. 2001, 20, 61–73. [Google Scholar] [PubMed]
- Melzer, J.; Rösch, W.; Reichling, J.; Brignoli, R.; Saller, R. Meta-analysis: Phytotherapy of functional dyspepsia with the herbal drug preparation STW 5 (Iberogast). Aliment. Pharmacol. Ther. 2004, 20, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health Benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef] [PubMed]
| EOs | DPPH Activity | ABTS Activity | References |
|---|---|---|---|
| M. piperita | 860 μg/mL | - | [28] |
| 57.9 ± 1.34% | 80.6 ± 1.45% | [29] | |
| 600 μg/mL | - | [30] | |
| 540 μg/mL | - | [31] | |
| 11.289 ± 0.514 μg/g | 0.154 ± 0.006 mmol/g | [32] | |
| M. pulegium | 14736 ± 156 μg/mL | - | [36] |
| 30.38 ± 0.8% | - | [37] | |
| 69.60 μg/mL | - | [38] | |
| 321.41 ± 2.53 μg/mL | - | [39] | |
| M. spicata | 3 μg/mL | - | [40] |
| 3450 ± 172.5 μg/mL | 40.2 ± 0.2 μg/mL | [41] | |
| M. longifolia | 57.4 μg/mL | - | [42] |
| M. suaveolens | 31 μg/mL | - | [43] |
| 52.4 ± 2.5% | - | [44] |
| Mentha spp. | Candida spp. | Dermathophytes | Aspergillus spp. | References | |||
|---|---|---|---|---|---|---|---|
| MIC 1 μg/mL | DDA 2 mm | MIC μg/mL | DDA mm | MIC μg/mL | DDA mm | ||
| M. piperita | 7120 | - | 3560 | - | - | - | [50] |
| 44.5 | - | - | - | - | - | [51] | |
| 445 | 90 | - | - | [16] | |||
| 225 | - | - | - | - | [52] | ||
| 256 | - | - | - | - | - | [53] | |
| - | - | −1335–2670 | - | 1335–2670 | - | [54] | |
| 1068–3560 | - | - | - | −445–3560 | - | [55] | |
| 890 | - | - | - | - | - | [56] | |
| - | - | 890- | - | - | - | [57] | |
| - | - | - | - | No activity | - | [58] | |
| 625 | - | - | - | 625 | - | [59] | |
| M. spicata | - | - | 0.25 | 25 to 40 | - | - | [60] |
| - | - | 890–2225 | - | 890–2225 | - | [54] | |
| - | - | 512 | - | - | - | [61] | |
| - | - | 1780–2670 | - | - | - | [62] | |
| 625 | - | - | - | 313 | - | [59] | |
| M. longifolia | 7120 | - | - | - | - | - | [50] |
| 3.9 | 2.5 | - | - | - | - | [63] | |
| M. pulegium | 890 | 16 | - | - | 222.5 | 10 | [64] |
| 1112.5 | 19 | - | - | - | - | [38] | |
| M. cervinia | 1112.5 to 2225 | - | 1112.5–2225 | - | 1112.5–2225 | - | [65] |
| M. suaveolens | 0.03 to 0.24 | - | - | - | - | - | [66] |
| - | - | 53.4–111.25 | - | - | - | [67] | |
| 4 | - | - | - | 6.8 | - | [43] | |
| 390 to 780 | - | - | - | - | - | [68] | |
| 780 | - | - | - | - | - | [69] | |
| 760 to 1560 | - | - | - | - | - | [46] | |
| 780 | 17 to 35 | - | - | - | - | [70] | |
| Compound | MIC (μg/mL) |
|---|---|
| 1,8 cineole | 2760 to 7360 |
| Menthol | 220 to 1320 |
| Limonene | 500 to 1000 |
| Carvone | 240 to 960 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stringaro, A.; Colone, M.; Angiolella, L. Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils. Medicines 2018, 5, 112. https://doi.org/10.3390/medicines5040112
Stringaro A, Colone M, Angiolella L. Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils. Medicines. 2018; 5(4):112. https://doi.org/10.3390/medicines5040112
Chicago/Turabian StyleStringaro, Annarita, Marisa Colone, and Letizia Angiolella. 2018. "Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils" Medicines 5, no. 4: 112. https://doi.org/10.3390/medicines5040112
APA StyleStringaro, A., Colone, M., & Angiolella, L. (2018). Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils. Medicines, 5(4), 112. https://doi.org/10.3390/medicines5040112







