Use of References in Responses from Scandinavian Drug Information Centres
Abstract
1. Introduction
2. Materials and Methods
2.1. Study setting
2.2. Analysis of references
2.3. Statistics
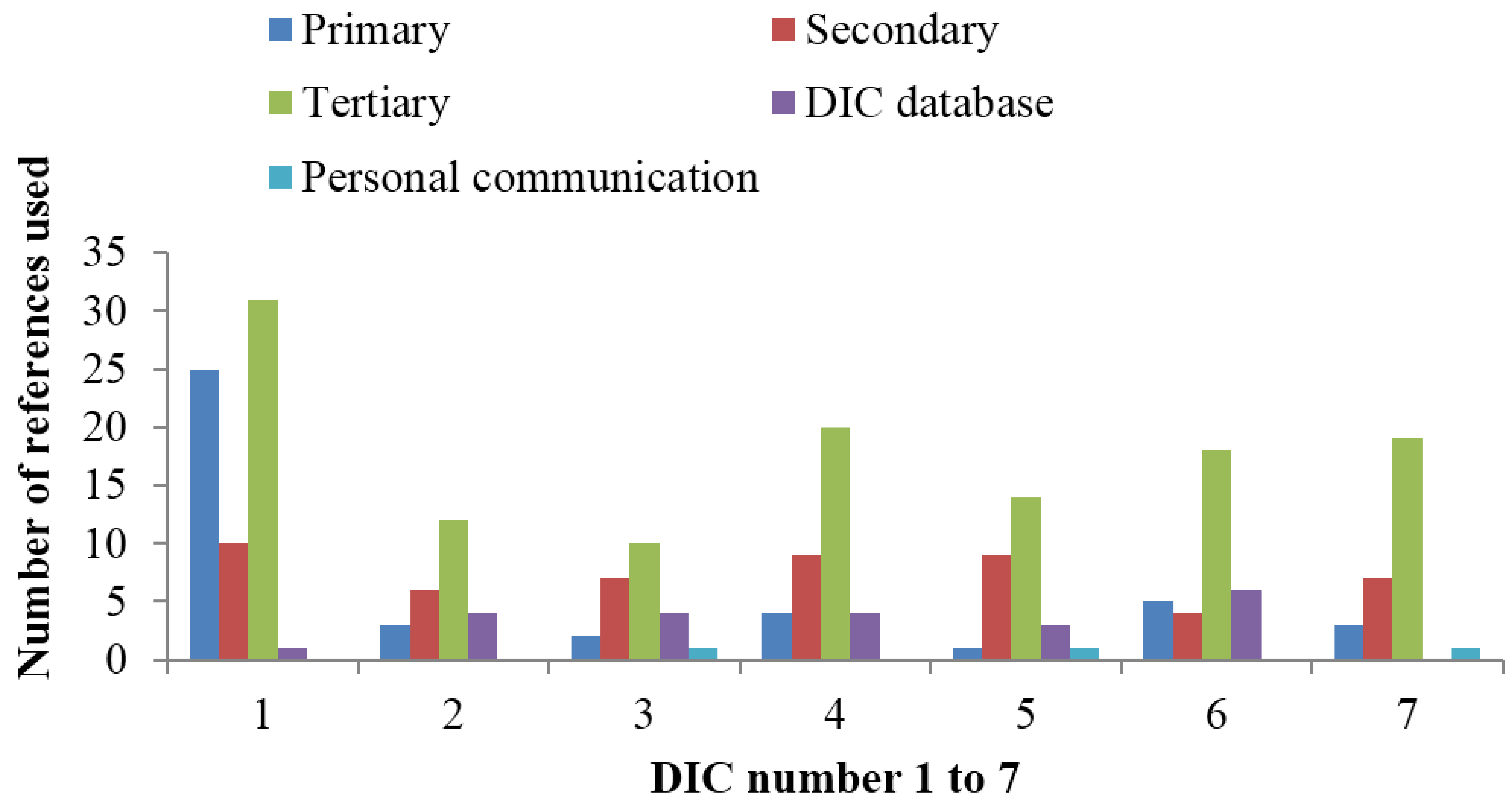
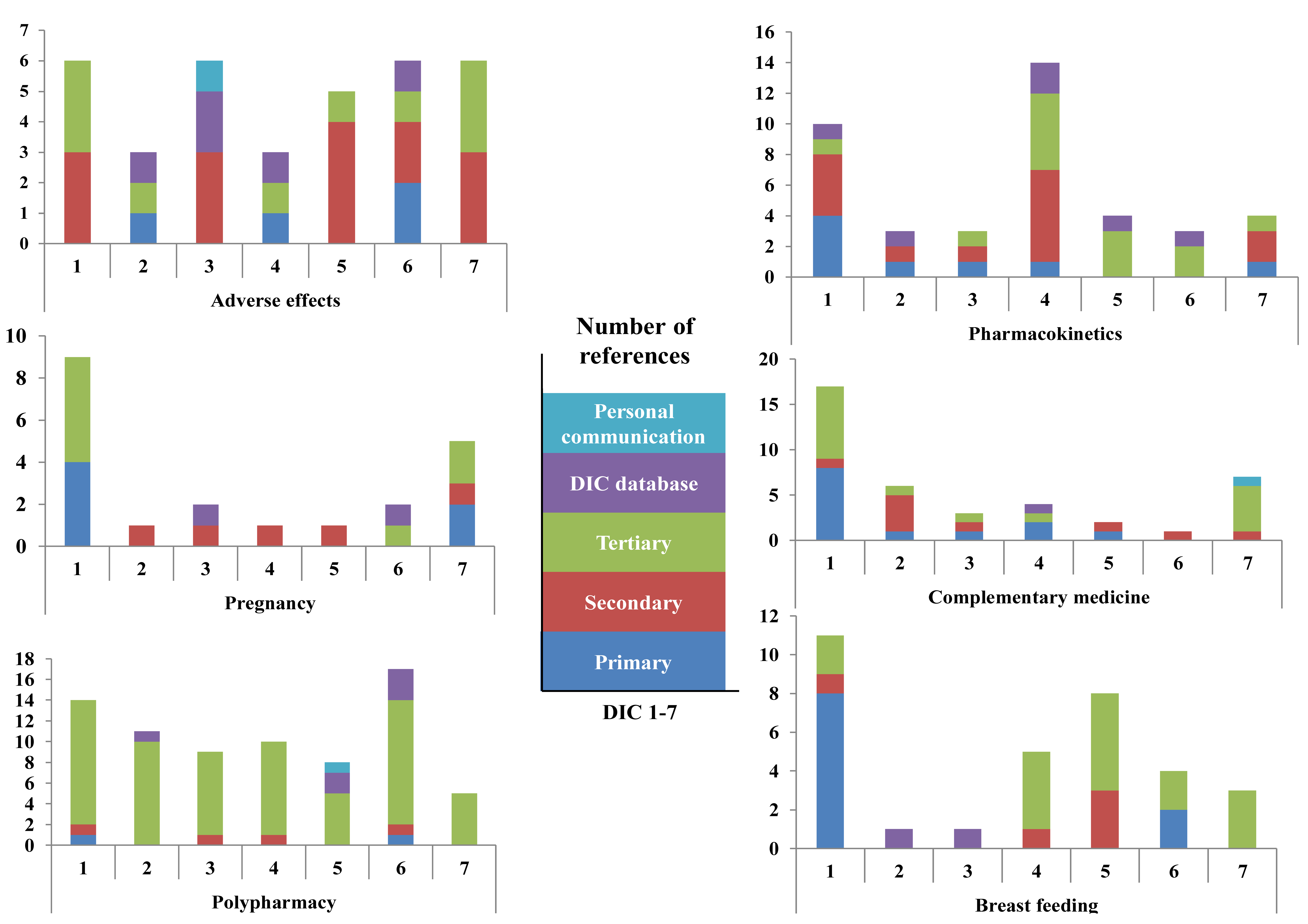
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hedegaard, U.; Damkier, P. Problem-oriented drug information: physicians’ expectations and impact on clinical practice. Eur. J. Clin. Pharmacol. 2009, 65, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Schjøtt, J.; Reppe, L.A.; Roland, P.D.; Westergren, T. A question-answer pair (QAP) database integrated with websites to answer complex questions submitted to the Regional Medicines Information and Pharmacovigilance Centres in Norway (RELIS): A descriptive study. BMJ Open 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Alvan, G.; Andersson, M.L.; Asplund, A.B.; Böttiger, Y.; Elwin, C.E.; Gustafsson, L.L.; Öhman, B.; Tornqvist, E. The continuing challenge of providing drug information services to diminish the knowledge--practice gap in medical practice. Eur. J. Clin. Pharmacol. 2013, 69, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Reppe, L.A.; Spigset, O.; Schjøtt, J. Drug information services today: Current role and future perspectives in rational drug therapy. Clin. Ther. 2016, 38, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Tröger, U.; Meyer, F.P. The regional drug-therapy consultation service centre-a conception that has been serving patients and physicians alike for 30 years in Magdeburg (Germany). Eur. J. Clin. Pharmacol. 2000, 55, 707–711. [Google Scholar]
- Scala, D.; Bracco, A.; Cozzolino, S.; Cristinziano, A.; De Marino, C.; Di Martino, A.; Gonzalez, E.; Mancini, A.; Romagnuolo, F.; Zeuli, L. Italian drug information centres: Benchmark report. Pharm. World Sci. 2001, 23, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.M.; Schilit, S.; Nathan, J.P.; Zerilli, T.; McGuire, H. Update on the status of 89 drug information centers in the United States. Am. J. Health Syst. Pharm. 2009, 66, 1718–1722. [Google Scholar] [CrossRef] [PubMed]
- Thürmann, P.A. Clinical pharmacology in everyday clinical care. Eur. J. Clin. Pharmacol. 2013, 69, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P.A. The mass production of redundant, misleading and conflicted systematic reviews and meta-analyses. Milbank Q. 2016, 94, 485–514. [Google Scholar] [CrossRef] [PubMed]
- Gurevitch, J.; Koricheva, J.; Nakagawa, S.; Stewart, G. Meta-analysis and the science of research synthesis. Nature 2018, 555, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Björk, B.C. Predatory’ open access: A longitudinal study of article volumes and market characteristics. BMC Med. 2015, 13, 230. [Google Scholar] [CrossRef] [PubMed]
- Bolshete, P. Analysis of thirteen predatory publishers: A trap for eager-to-publish researchers. Curr. Med. Res. Opin. 2018, 34, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Clauson, K.A.; Marsh, W.A.; Polen, H.H.; Seamon, M.J.; Ortiz, B.I. Clinical decision support tools: Analysis of online drug information databases. BMC Med. Inf. Decis. Making 2007, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Gaebelein, C.J.; Gleason, B.L. Contemporary Drug Information: An Evidence-Based Approach; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008; pp. 1–370. [Google Scholar]
- International Pharmaceutical Federation (FIP). Requirements for Drug Information Centres, 2005. Available online: http://www.cff.org.br/userfiles/file/cebrim/RequirementsforDrugInformationCentres%202005%20 final.pdf (accessed on 23 May 2018).
- American Society of Health-System Pharmacists (ASHP). ASHP Guidelines on the Pharmacist’s role in Providing Drug Information, 2014. Available online: https://www.ashp.org/-/media/assets/ policy-guidelines/docs/guidelines/pharmacists-role-providing-drug-information.ashx?la=en&hash=1A4238FCD F07FEC6D1F3F1205D2027D41F23AE20 (accessed on 28 June 2018).
- Reppe, L.A.; Lydersen, S.; Schjøtt, J.; Damkier, P.; Christensen, H.R.; Kampmann, J.P.; Böttiger, Y.; Spigset, O. Relationship between time consumption and quality of responses to drug-related queries: A Study from seven drug information centers in Scandinavia. Clin. Ther. 2016, 38, 1738–1749. [Google Scholar] [CrossRef] [PubMed]
- Reppe, L.A.; Spigset, O.; Kampmann, J.P.; Damkier, P.; Christensen, H.R.; Böttiger, Y.; Schjøtt, J. Quality assessment of structure and language elements of written responses given by seven Scandinavian drug information centres. Eur. J. Clin. Pharmacol. 2017, 73, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, S.L.; Dodson, T.B.; Assael, L.A.; Landesberg, R.; Marx, R.E.; Mehrotra, B. American association of oral and maxillofacial surgeons position paper on bisphosphonate-related osteonecrosis of the jaws--2009 update. J. Oral. Maxillofacial Surg. 2009, 67, 2–12. [Google Scholar] [CrossRef]
- The American Association of Oral and Maxillofacial Surgeons (AAOMS). Available online: https://www.aaoms.org/ (accessed on 23 May 2018).
- Llerena, A.; Öhman, B.; Alván, G. References used in a drug information centre. Eur. J. Clin. Pharmacol. 1995, 49, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Moorman, K.L.; Macdonald, E.A.; Trovato, A.; Tak, C.R. Assessment and use of drug information references in Utah pharmacies. Pharm. Pract. 2017, 15, 839. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.O.; McGuiness, S.M.; Bourne, P.E. Pharmacy Informatics; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2010; pp. 1–289. ISBN 9781420071757. [Google Scholar]
- Reppe, L.A.; Spigset, O.; Schjøtt, J. Which factors predict the time spent answering queries to a drug information centre? Pharm. World Sci. 2010, 32, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Lyrvall, H. Problemorienterad läkemedelsinformation—en möjlighet att förbättra sjukvårdens kvalitet [in Swedish]. Ph.D. Thesis, Karolinska Institutet, Stockholm, Sweden, 1994. [Google Scholar]
- Reppe, L.A.; Spigset, O.; Böttiger, Y.; Christensen, H.R.; Kampmann, J.P.; Damkier, P.; Lydersen, S.; Schjøtt, J. Factors associated with time consumption when answering drug-related queries to Scandinavian drug information centres: A multi-centre study. Eur. J. Clin. Pharmacol. 2014, 70, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Schjøtt, J.; Pomp, E.; Gedde-Dahl, A. Quality and impact of problem-oriented drug information: A method to change clinical practice among physicians? Eur. J. Clin. Pharmacol. 2002, 57, 897–902. [Google Scholar] [CrossRef] [PubMed]
- National Health Service (NHS), UK Medicines Information (UKMi), Specialist Pharmacy Service (2017) Guide to writing medicines Q & As. Available online: https://www.sps.nhs.uk/articles/guide- to-writing-medicines-qas (accessed on 28 June 2018).
- Widnes, S.F.; Schjøtt, J. Risk perception regarding drug use in pregnancy. Am. J. Obstet. Gynecol. 2017, 216, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Jahnsen, J.A.; Widnes, S.F.; Schjøtt, J. Analysis of questions about use of drugs in breastfeeding to Norwegian drug information centres. Int. Breastfeeding J. 2018, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Natural Medicines. Available online: https://naturalmedicines.therapeuticresearch.com (accessed on 23 May 2018).
- Schjøtt, J.; Erdal, H. Questions about complementary and alternative medicine to the regional medicines information and pharmacovigilance centres in Norway (RELIS): A descriptive pilot study. BMC Complement. Altern. Med. 2014, 14, 56. [Google Scholar] [CrossRef] [PubMed]
- Kongsholm, G.G.; Nielsen, A.K.; Damkier, P. Drug interaction databases in medical literature: transparency of ownership, funding, classification algorithms, level of documentation, and staff qualifications. A systematic review. Eur. J. Clin. Pharmacol. 2015, 71, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Schjøtt, J. Benefits of a national network of drug information centres: RELIS. Eur. J. Clin. Pharmacol. 2017, 73, 125–126. [Google Scholar] [CrossRef] [PubMed]
| Category | Query | Requested Response Time | References | ||
|---|---|---|---|---|---|
| Total | Mean | Range | |||
| Adverse effects | A female patient presents with deep, infected pockets close to the jaw bone, and needs to have these rinsed every fourth to fifth week for the next six months. The patient uses alendronate 70 mg once weekly. Should alendronate be discontinued during treatment? | Within a week | 35 | 5.0 | 3–6 |
| Pharmacokinetics | A male patient with formerly performed gastric bypass needs treatment for Helicobacter pylori infection because of symptomatic ulcus. The GP wants to start treatment with pantoprazole 40 mg once daily, metronidazole 400 mg twice daily, and amoxicillin 750 mg twice daily. Will the absorption of the drugs be reduced, and are dosage adjustments necessary? | Within 2 days | 41 | 5.9 | 3–14 |
| Pregnancy | A pregnant women manifests with moderate depression (MADRS1 score 29), and there is indication for treatment with an antidepressant. What antidepressant is the first choice of drug during pregnancy? | Within the next day | 21 | 3.0 | 1–9 |
| Complementary medicine | A GP has registered an increasing use of Ginkgo biloba in nursing care homes and home nursing services. He (she) has also registered that ginkgo might increase bleeding time. What documentation exists on this topic, and what is the relevance for concomitant use of, for example, warfarin, acetylsalicylic acid, clopidogrel, and enoxaparin? | None | 40 | 5.7 | 1–17 |
| Polypharmacy | A male patient, 75 years old, has gradually developed impaired cognition for the last five to six months (MMS2 score 18 at examination). He has essential hypertension treated with atenolol 100 mg once daily and losartan/hydrochlorothiazide 100/12.5 mg once daily. His blood pressure was 130/90 mmHg at the latest appointment. He also uses simvastatin 40 mg and acetylsalisylic acid 160 mg once daily. He uses paroxetine 40 mg in the morning for anxiety/depression, diazepam 5 mg as needed, and promethazine/propiomazine3 50 mg at night for sleep. He also uses tolterodine 2.8/4 mg4 once daily for overactive bladder (dosage increased from 1.4/2 mg4 three months ago). The patient does not smoke. Can any of these drugs, or drug interactions, increase the risk of impaired cognition? | None | 74 | 10.6 | 5–17 |
| Breast feeding | A female patient, 13 weeks post partum, presents with active ulcerative colitis. She has earlier been treated with sulfasalazine 500 mg × 3 (discontinued during pregnancy). Can she use sulfasalazine while breast feeding? | None | 33 | 4.7 | 1–11 |
| Total | 244 | 5.8 | 1–17 | ||
| Type of reference | Total n (%) | Type of query and number of references | |||||
|---|---|---|---|---|---|---|---|
| Adverse effects | Pharmacokinetics | Pregnancy | Complementary medicine | Polypharmacy | Breast feeding | ||
| Primary (original studies) | 43 (17.6) | 4 | 8 | 6 | 13 | 2 | 10 |
| Secondary (reviews) | 52 (21.3) | 15 | 14 | 5 | 9 | 4 | 5 |
| Tertiary (drug monographs, handbooks, etc.) | 124 (50.8) | 10 | 13 | 8 | 16 | 61 | 16 |
| DIC database | 22 (9.0) | 5 | 6 | 2 | 1 | 6 | 2 |
| Personal communication | 3 (1.2) | 1 | 0 | 0 | 1 | 1 | 0 |
| Total | 244 (100.0) | 35 | 41 | 21 | 40 | 74 | 33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schjøtt, J.; Böttiger, Y.; Damkier, P.; Reppe, L.A.; Kampmann, J.P.; Christensen, H.R.; Spigset, O. Use of References in Responses from Scandinavian Drug Information Centres. Medicines 2018, 5, 66. https://doi.org/10.3390/medicines5030066
Schjøtt J, Böttiger Y, Damkier P, Reppe LA, Kampmann JP, Christensen HR, Spigset O. Use of References in Responses from Scandinavian Drug Information Centres. Medicines. 2018; 5(3):66. https://doi.org/10.3390/medicines5030066
Chicago/Turabian StyleSchjøtt, Jan, Ylva Böttiger, Per Damkier, Linda Amundstuen Reppe, Jens Peter Kampmann, Hanne Rolighed Christensen, and Olav Spigset. 2018. "Use of References in Responses from Scandinavian Drug Information Centres" Medicines 5, no. 3: 66. https://doi.org/10.3390/medicines5030066
APA StyleSchjøtt, J., Böttiger, Y., Damkier, P., Reppe, L. A., Kampmann, J. P., Christensen, H. R., & Spigset, O. (2018). Use of References in Responses from Scandinavian Drug Information Centres. Medicines, 5(3), 66. https://doi.org/10.3390/medicines5030066





