Auricular Neuromodulation: The Emerging Concept beyond the Stimulation of Vagus and Trigeminal Nerves
Abstract
1. Neuromodulation and Neurostimulation
- Specificity: stimulation can be targeted to particular areas avoiding the insurgence of systemic side-effects, typical of traditional drug therapies;
- Safety: neurostimulation techniques are generally well-tolerated and almost devoid of dangerous side effects;
- Flexibility: the treatment can be interrupted at any time.
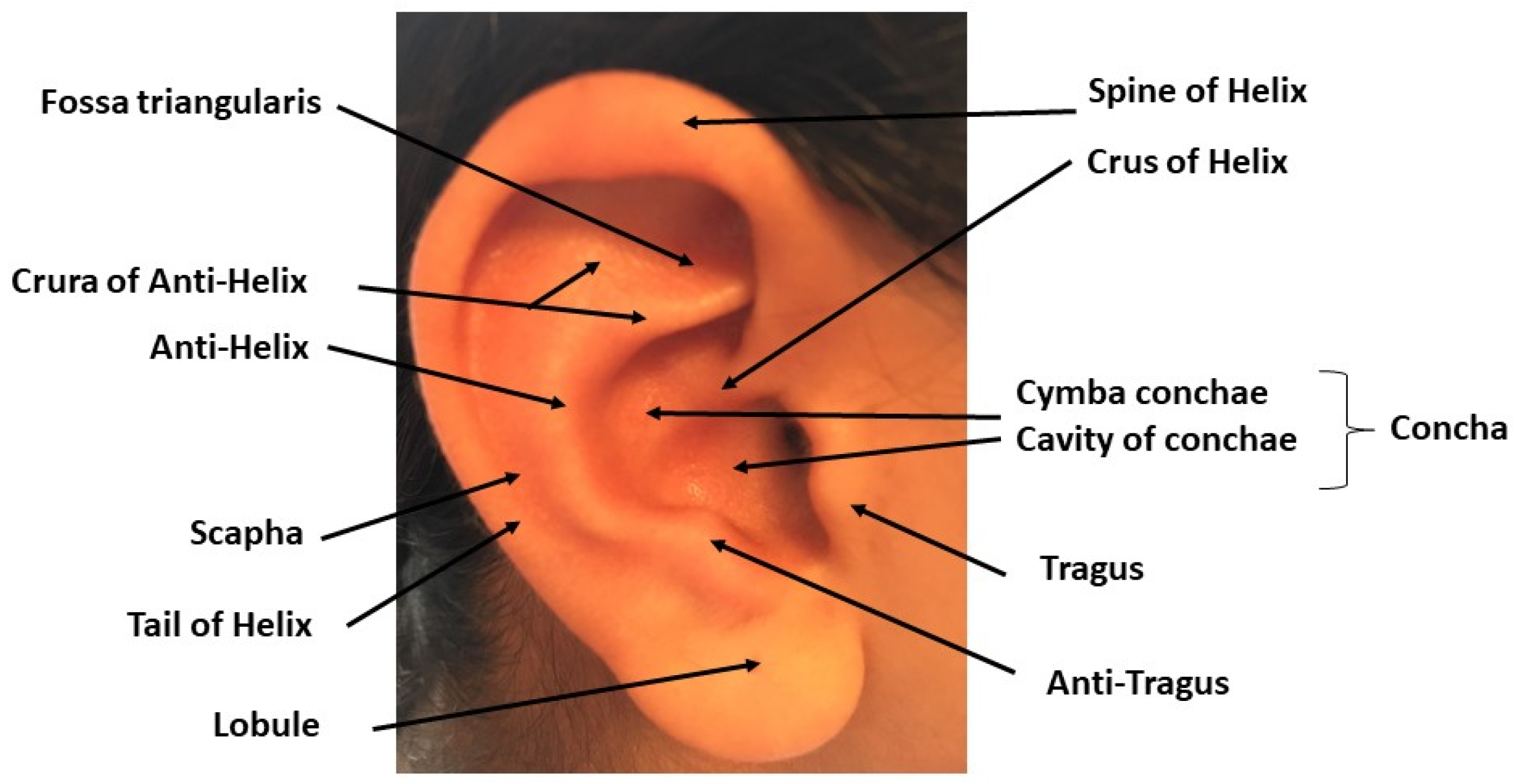
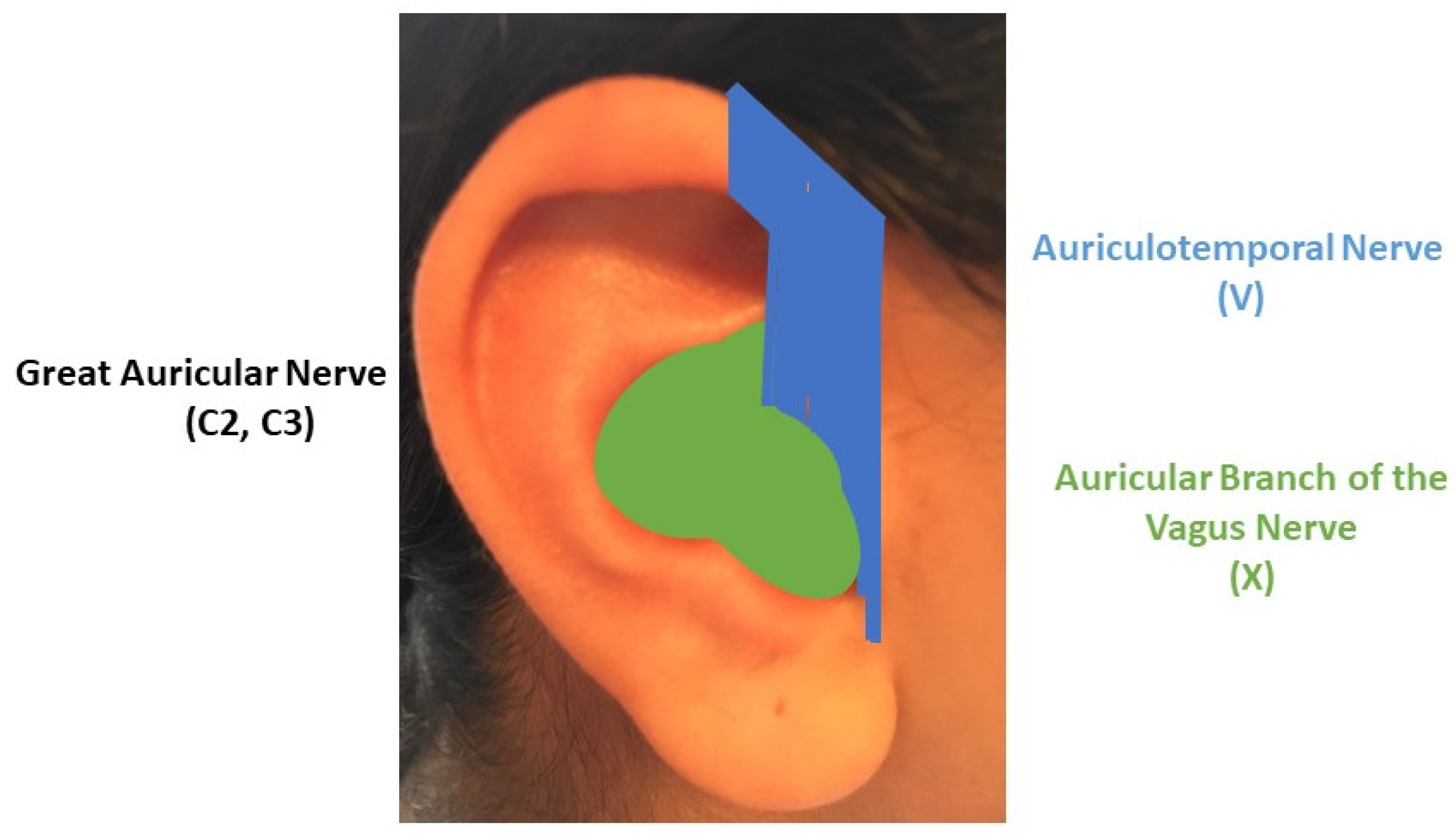
2. TNS, tVNS and Auricular Stimulation: Possible Sites and Mechanisms of Action in the Central Nervous System
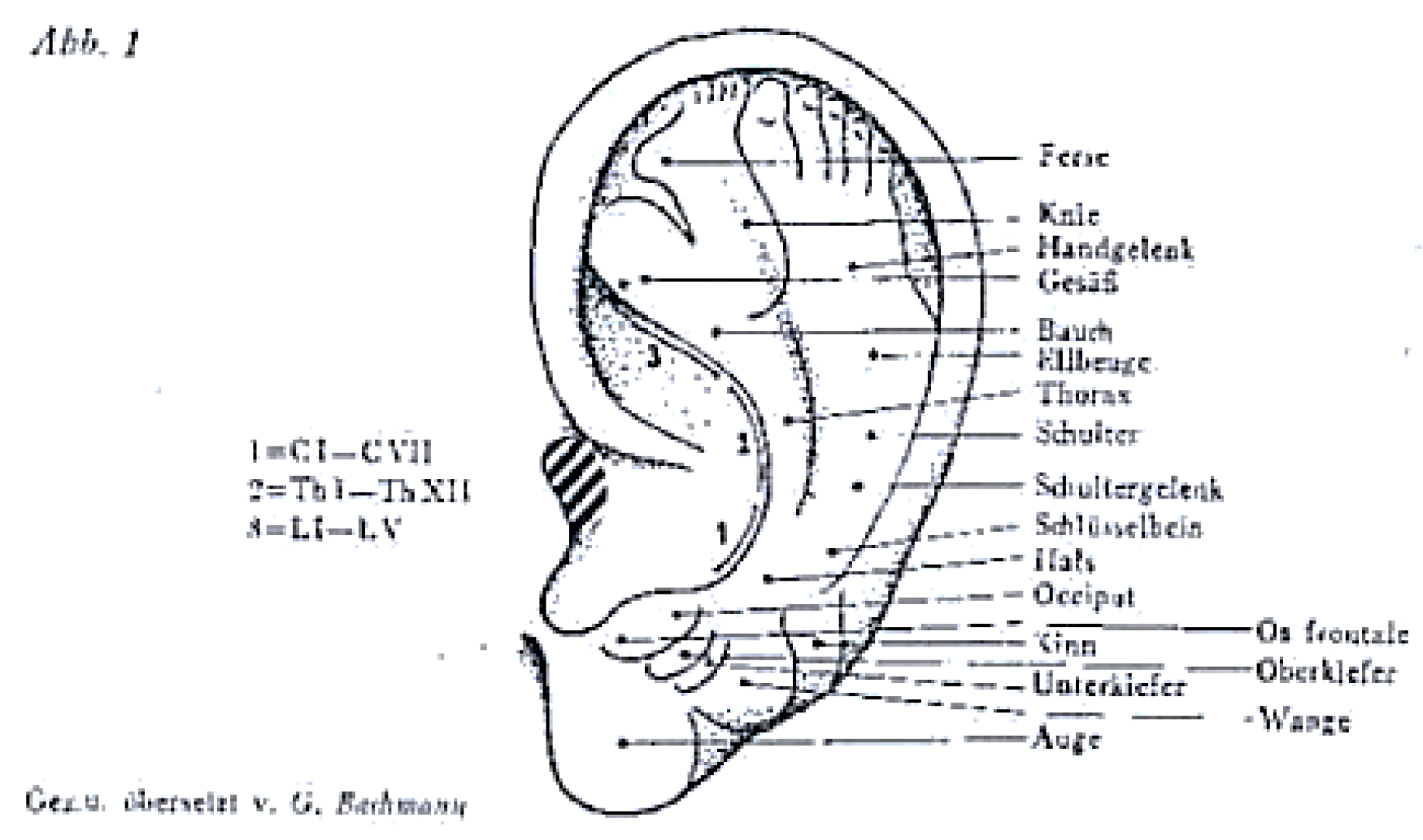
3. The “Next Frontier” of Neuromodulation of the Outer Ear: The Cervical Plexus Supply Area
4. Concluding Remarks
Acknowledgments
Conflicts of Interest
Abbreviations
| ABVN | Auricular Branch of the Vagus Nerve |
| ATN | Auricular Temporal Nerve |
| BSAA | Brain Stem Auricular Acupoint |
| CNS | central nervous system |
| FDA | Food and Drug Administration |
| fMRI | functional Magnetic Resonance Imaging |
| GAN | Great Auricular Nerve |
| TAA | Thumb Auricular Acupoint |
| TENS | transcutaneous electrical nerve stimulation |
| TNS | trigeminal nerve stimulation |
| tVNS | transcutaneous Vagus Nerve Stimulation |
| VNS | Vagus Nerve Stimulation |
| WFCMS | World Federation of Chinese Medicine Societies |
| WHO | World Health Organization |
References
- Katz, P.S.; Calin, R.J. Neuromodulation. In Encyclopedia of Neuroscience; Springer: Berlin/Heidelberg, Germany, 2009; pp. 497–503. [Google Scholar]
- Shiozawa, P.; da Silva, M.E.; de Carvalho, T.C.; Cordeiro, Q.; Brunoni, A.R.; Fregni, F. Transcutaneous vagus and trigeminal nerve stimulation for neuropsychiatric disorders: A systematic review. Arq. Neuropsiquiatr. 2014, 72, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Goroszeniuk, T.; Pang, D. Peripheral neuromodulation: A review. Curr. Pain Headache Rep. 2014, 18, 412. [Google Scholar] [CrossRef] [PubMed]
- Lopez, H.S.; Brown, A.M. Neuromodulation. Curr. Opin. Neurobiol. 1992, 2, 317–322. [Google Scholar] [CrossRef]
- Nadim, F.; Bucher, D. Neuromodulation of neurons and synapses. Curr. Opin. Neurobiol. 2014, 29, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Marder, E.; Thirumalai, V. Cellular, synaptic and network effects of neuromodulation. Neural Netw. 2002, 15, 479–493. [Google Scholar] [CrossRef]
- Marder, E. Neuromodulation of neuronal circuits: Back to the future. Neuron 2012, 76, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wokke, M.E.; Talsma, L.J.; Vissers, M.E. Biasing neural network dynamics using non-invasive brain stimulation. Front. Syst. Neurosci. 2015, 8, 246. [Google Scholar] [CrossRef] [PubMed]
- Katz, P.S. Intrinsic and extrinsic neuromodulation of motor circuits. Curr. Opin. Neurobiol. 1995, 5, 799–808. [Google Scholar] [CrossRef]
- Katz, P.S.; Frost, W.N. Intrinsic neuromodulation: Altering neuronal circuits from within. Trends Neurosci. 1996, 19, 54–61. [Google Scholar] [CrossRef]
- Rise, M.T. Instrumentation for neuromodulation. Arch. Med. Res. 2000, 31, 237–247. [Google Scholar] [CrossRef]
- Borovikova, L.V.; Ivanova, S.; Zhang, M.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.; Abumrad, N.; Eaton, J.W.; Tracey, K.J. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 2000, 405, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Ballina, M.; Tracey, K.J. The neurology of the immune system: Neural reflexes regulate immunity. Neuron 2009, 64, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Tracey, K.J. Reflex control of immunity. Nat. Rev. Immunol. 2009, 9, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Edwards, C.A.; Kouzani, A.; Lee, K.H.; Ross, E.K. Neurostimulation Devices for the Treatment of Neurologic Disorders. Mayo Clin. Proc. 2017, 92, 1427–1444. [Google Scholar] [CrossRef] [PubMed]
- Moseley, B.D.; DeGiorgio, C.M. Refractory status epilepticus treated with trigeminal nerve stimulation. Epilepsy Res. 2013, 108, 600–603. [Google Scholar] [CrossRef] [PubMed]
- Cook, I.A.; Espinoza, R.; Leuchter, A.F. Neuromodulation for depression: Invasive and noninvasive (deep brain stimulation, transcranial magnetic stimulation, trigeminal nerve stimulation). Neurosurg. Clin. N. Am. 2013, 25, 103–116. [Google Scholar] [CrossRef] [PubMed]
- DeGiorgio, C.M.; Murray, D.; Markovic, D.; Whitehurst, T. Trigeminal nerve stimulation for epilepsy: Long-term feasibility and efficacy. Neurology 2009, 72, 936–938. [Google Scholar] [CrossRef] [PubMed]
- DeGiorgio, C.M.; Soss, J.; Cook, I.A.; Markovic, D.; Gornbein, J.; Murray, D.; Oviedo, S.; Gordon, S.; Corralle-Leyva, G.; Kealey, C.P.; et al. Randomized controlled trial of trigeminal nerve stimulation for drug-resistant epilepsy. Neurology 2013, 80, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Soss, J.; Heck, C.; Murray, D.; Markovic, D.; Oviedo, S.; Corrale-Leyva, G.; Gordon, S.; Kealey, C.; DeGiorgio, C. A prospective long-term study of external trigeminal nerve stimulation for drug-resistant epilepsy. Epilepsy Behav. 2014, 42, 44–47. [Google Scholar] [CrossRef] [PubMed]
- DeGiorgio, C.M.; Shewmon, A.; Murray, D.; Whitehurst, T. Pilot study of trigeminal nerve stimulation (TNS) for epilepsy: A proof-of-concept trial. Epilepsia 2006, 47, 1213–1215. [Google Scholar] [CrossRef] [PubMed]
- DeGiorgio, C.M.; Shewmon, D.A.; Whitehurst, T. Trigeminal nerve stimulation for epilepsy. Neurology 2003, 61, 421–422. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, T.; Bajwa, Z.; Lantz, G.; Lee, S.; Burstein, R. Implanted auriculotemporal nerve stimulator for the treatment of refractory chronic migraine. Headache 2010, 50, 1064–1069. [Google Scholar] [CrossRef] [PubMed]
- Chou, D.E.; Gross, G.J.; Casadei, C.H.; Yugrakh, M.S. External Trigeminal Nerve Stimulation for the Acute Treatment of Migraine: Open-Label Trial on Safety and Efficacy. Neuromodulation 2017, 20, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Riederer, F.; Penning, S.; Schoenen, J. Transcutaneous Supraorbital Nerve Stimulation (t-SNS) with the Cefaly® Device for Migraine Prevention: A Review of the Available Data. Pain Ther. 2015, 4, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Schoenen, J.E. Migraine prevention with a supraorbital transcutaneous stimulator: A randomized controlled trial. Neurology 2013, 80, 697. [Google Scholar] [CrossRef] [PubMed]
- Cook, I.A.; Kealey, C.P.; DeGiorgio, C.M. The potential use of trigeminal nerve stimulation in the treatment of epilepsy. Ther. Deliv. 2015, 6, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Shiozawa, P.; Cordeiro, Q. Trigeminal nerve stimulation for clinical trials in neuropsychiatry: The issue of skin integrity. J. ECT 2015, 31, e29–e30. [Google Scholar] [CrossRef] [PubMed]
- Schrader, L.M.; Cook, I.A.; Miller, P.R.; Maremont, E.R.; DeGiorgio, C.M. Trigeminal nerve stimulation in major depressive disorder: First proof of concept in an open pilot trial. Epilepsy Behav. 2011, 22, 475–478. [Google Scholar] [CrossRef] [PubMed]
- Pop, J.; Murray, D.; Markovic, D.; DeGiorgio, C.M. Acute and long-term safety of external trigeminal nerve stimulation for drug-resistant epilepsy. Epilepsy Behav. 2011, 22, 574–576. [Google Scholar] [CrossRef] [PubMed]
- Fanselow, E.E. Central mechanisms of cranial nerve stimulation for epilepsy. Surg. Neurol. Int. 2012, 3, S247–S254. [Google Scholar] [CrossRef] [PubMed]
- Mercante, B.; Pilurzi, G.; Ginatempo, F.; Manca, A.; Follesa, P.; Tolu, E.; Deriu, F. Trigeminal nerve stimulation modulates brainstem more than cortical excitability in healthy humans. Exp. Brain Res. 2015, 233, 3301–3311. [Google Scholar] [CrossRef] [PubMed]
- Pilurzi, G.; Mercante, B.; Ginatempo, F.; Follesa, P.; Tolu, E.; Deriu, F. Transcutaneous trigeminal nerve stimulation induces a long-term depression-like plasticity of the human blink reflex. Exp. Brain Res. 2015, 234, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Piquet, M.; Balestra, C.; Sava, S.L.; Schoenen, J.E. Supraorbital transcutaneous neurostimulation has sedative effects in healthy subjects. BMC Neurol. 2011, 11, 135. [Google Scholar] [CrossRef] [PubMed]
- Ginatempo, F.; Pirina, P.; Melis, F.; Deriu, F. Short-term trigeminal neuromodulation does not alter sleep latency in healthy subjects: A pilot study. Neurol. Sci. 2018, 39, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Mercante, B.; Enrico, P.; Floris, G.; Quartu, M.; Boi, M.; Serra, M.P.; Follesa, P.; Deriu, F. Trigeminal nerve stimulation induces Fos immunoreactivity in selected brain regions, increases hippocampal cell proliferation and reduces seizure severity in rats. Neuroscience 2017, 361, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-Q.; Zhu, L.-J.; Wang, X.-H.; Zuo, J.; He, H.-Y.; Tian, M.-M.; Wang, L.; Liang, G.-L.; Wang, Y. Chronic Trigeminal Nerve Stimulation Protects Against Seizures, Cognitive Impairments, Hippocampal Apoptosis, and Inflammatory Responses in Epileptic Rats. J. Mol. Neurosci. 2016, 59, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Revesz, D.; Tjernstrom, M.; Ben-Menachem, E.; Thorlin, T. Effects of vagus nerve stimulation on rat hippocampal progenitor proliferation. Exp. Neurol. 2008, 214, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Ventureyra, E.C.G. Transcutaneous vagus nerve stimulation for partial onset seizure therapy. Child Nerv. Syst. 2000, 16, 101–102. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, P.; López, M.; Larraya, I.; Chamorro, J.; Cobo, J.L.; Ordóñez, S.; Vega, J.A. Innervation of the Human Cavum Conchae and Auditory Canal: Anatomical Basis for Transcutaneous Auricular Nerve Stimulation. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Kreuzer, P.M.; Landgrebe, M.; Resch, M.; Husser, O.; Schecklmann, M.; Geisreiter, F.; Poeppl, T.B.; Prasser, S.J.; Hajak, G.; Rupprecht, R.; et al. Feasibility, safety and efficacy of transcutaneous vagus nerve stimulation in chronic tinnitus: An open pilot study. Brain Stimul. 2014, 7, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Hösl, K.; Kiess, O.; Schanze, A.; Kornhuber, J.; Forster, C. BOLD fMRI deactivation of limbic and temporal brain structures and mood enhancing effect by transcutaneous vagus nerve stimulation. J. Neural Transm. (Vienna) 2007, 114, 1485–1493. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.X.; He, W.; Jing, X.H.; Liu, J.L.; Ben, H.; Liu, K.; Zhu, B. Transcutaneous auricular vagus nerve stimulation protects endotoxemic rat from lipopolysaccharide-induced inflammation. Evid. Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Kiess, O.; Hösl, K.; Terekhin, P.; Kornhuber, J.; Forster, C. CNS BOLD fMRI effects of sham-controlled transcutaneous electrical nerve stimulation in the left outer auditory canal—A pilot study. Brain Stimul. 2013, 6, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Hein, E.; Nowak, M.; Kiess, O.; Biermann, T.; Bayerlein, K.; Kornhuber, J.; Kraus, T. Auricular transcutaneous electrical nerve stimulation in depressed patients: A randomized controlled pilot study. J. Neural Transm. (Vienna) 2012, 120, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Laqua, R.; Leutzow, B.; Wendt, M.; Usichenko, T. Transcutaneous vagal nerve stimulation may elicit anti- and pro-nociceptive effects under experimentally-induced pain—A crossover placebo-controlled investigation. Auton. Neurosci. 2014, 185, 120–122. [Google Scholar] [CrossRef] [PubMed]
- Rong, P.-J.; Fang, J.-L.; Wang, L.-P.; Meng, H.; Liu, J.; Ma, Y.-G.; Ben, H.; Li, L.; Liu, R.P.; Huang, Z.X.; et al. Transcutaneous vagus nerve stimulation for the treatment of depression: A study protocol for a double blinded randomized clinical trial. BMC Complement. Altern. Med. 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Assenza, G.; Campana, C.; Colicchio, G.; Tombini, M.; Assenza, F.; Di Pino, G.; Di Lazzaro, V. Transcutaneous and invasive vagal nerve stimulations engage the same neural pathways: In-vivo human evidence. Brain Stimul. 2017, 10, 853–854. [Google Scholar] [CrossRef] [PubMed]
- Rong, P.; Liu, A.; Zhang, J.; Wang, Y.; Yang, A.; Li, L.; Liu, R.; He, W.; Liu, H.; Huang, F.; et al. An alternative therapy for drug-resistant epilepsy: Transcutaneous auricular vagus nerve stimulation. Chin. Med. J. 2014, 127, 300–304. [Google Scholar] [PubMed]
- Deriu, F. Anatomo-physiological basis for auricular stimulation. Med. Acupunct. 2018, 30, 1–10. [Google Scholar]
- Peuker, E.T.; Filler, T.J. The nerve supply of the human auricle. Clin. Anat. 2002, 15, 35–37. [Google Scholar] [CrossRef] [PubMed]
- Yakunina, N.; Kim, S.S.; Nam, E.-C. Optimization of Transcutaneous Vagus Nerve Stimulation Using Functional MRI. Neuromodulation 2016, 20, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Polak, T.; Markulin, F.; Ehlis, A.-C.; Langer, J.B.M.; Ringel, T.M.; Fallgatter, A.J. Far field potentials from brain stem after transcutaneous vagus nerve stimulation: Optimization of stimulation and recording parameters. J. Neural Transm. (Vienna) 2009, 116, 1237–1242. [Google Scholar] [CrossRef] [PubMed]
- Fallgatter, A.J.; Neuhauser, B.; Herrmann, M.J.; Ehlis, A.-C.; Wagener, A.; Scheuerpflug, P.; Reiners, K.; Riederer, P. Far field potentials from the brain stem after transcutaneous vagus nerve stimulation. J. Neural Transm. (Vienna) 2003, 110, 1437–1443. [Google Scholar] [CrossRef] [PubMed]
- Frangos, E.; Ellrich, J.; Komisaruk, B.R. Non-invasive Access to the Vagus Nerve Central Projections via Electrical Stimulation of the External Ear: fMRI Evidence in Humans. Brain Stimul. 2014, 8, 624–636. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, J.T.; Mifflin, S.W.; Gould, G.G.; Frazer, A. Induction of c-Fos and DeltaFosB immunoreactivity in rat brain by Vagal nerve stimulation. Neuropsychopharmacology 2007, 33, 1884–1895. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Kong, J. Transcutaneous Vagus Nerve Stimulation: A Promising Method for Treatment of Autism Spectrum Disorders. Front. Neurosci. 2017, 10, 609. [Google Scholar] [CrossRef] [PubMed]
- Lerman, I.; Hauger, R.; Sorkin, L.; Proudfoot, J.; Davis, B.; Huang, A.; Lam, K.; Simon, B.; Baker, D.G. Noninvasive Transcutaneous Vagus Nerve Stimulation Decreases Whole Blood Culture-Derived Cytokines and Chemokines: A Randomized, Blinded, Healthy Control Pilot Trial. Neuromodulation 2016, 19, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Ay, I.; Napadow, V.; Ay, H. Electrical stimulation of the vagus nerve dermatome in the external ear is protective in rat cerebral ischemia. Brain Stimul. 2014, 8, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhang, L.; He, G.; Tan, X.; Jin, X.; Li, C. Transcutaneous auricular vagus nerve stimulation regulates expression of growth differentiation factor 11 and activin-like kinase 5 in cerebral ischemia/reperfusion rats. J. Neurol. Sci. 2016, 369, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Hays, S.A.; Ruiz, A.; Bethea, T.; Khodaparast, N.; Carmel, J.B.; Rennaker, R.L.; Kilgard, M.P. Vagus nerve stimulation during rehabilitative training enhances recovery of forelimb function after ischemic stroke in aged rats. Neurobiol. Aging 2016, 43, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Hays, S.A.; Khodaparast, N.; Hulsey, D.R.; Ruiz, A.; Sloan, A.M.; Rennaker, R.L.; Kilgard, M.P. Vagus nerve stimulation during rehabilitative training improves functional recovery after intracerebral hemorrhage. Stroke 2014, 45, 3097–3100. [Google Scholar] [CrossRef] [PubMed]
- Greif, R.; Laciny, S.; Mokhtarani, M.; Doufas, A.G.; Bakhshandeh, M.; Dorfer, L.; Sessler, D.I. Transcutaneous electrical stimulation of an auricular acupuncture point decreases anesthetic requirement. Anesthesiology. Am. Soc. Anesthesiol. 2002, 96, 306–312. [Google Scholar] [CrossRef]
- Yeo, S.; Kim, K.S.; Lim, S. Randomised clinical trial of five ear acupuncture points for the treatment of overweight people. Acupunct. Med. 2013, 32, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Badran, B.W.; Dowdle, L.T.; Mithoefer, O.J.; LaBate, N.T.; Coatsworth, J.; Brown, J.C.; DeVries, W.H.; Austelle, C.W.; McTeague, L.M.; George, M.S. Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation (taVNS) via electrical stimulation of the tragus: A concurrent taVNS/fMRI study and review. Brain Stimul. 2017. [Google Scholar] [CrossRef]
- Nogier, P. Acupuncture of the ear pavilion. Dtsch. Z. Akupunkt. 1957, Band VI, 25–35. [Google Scholar]
- Bourdiol, R.J. Elements of Auriculotherapy; Maisonneuve: Moulins-les-Metz, France, 1982. [Google Scholar]
- Oleson, T. Auriculotherapy Manual: Chinese and Western Systems of Ear Acupuncture; Health Care Alternatives: Los Angeles, CA, USA, 1998. [Google Scholar]
- Nogier, P. Handbook to of Auriculotherapy; Maisonneuve: Moulins-les-Metz, France, 1981. [Google Scholar]
- Alimi, D.; Geissmann, A.; Gardeur, D. Auricular acupuncture stimulation measured on fuctional magnetic resonance imaging. Med. Acupunct. 2007, 13, 18–21. [Google Scholar]
- Liboni, W.; Romoli, M.; Allais, G.; Mana, O.; Giacobbe, M.D.; Pugliese, A.M.; Bellacicco, F.; Terzi, M.G.; Schiapparelli, P.; Benedetto, C. The fMRI for the auricular acupuncture validation: Experimental protocol. Presented at the Acti XXII Congresso Nationale Soc Ital Rifles Ago Auri (SIRAA), Asti, Italy, 16–17 November 2007. [Google Scholar]
- Alimi, D.; Chelly, J.E. New Universal Nomenclature in Auriculotherapy. J. Altern. Complement. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Rabischong, P.; Terral, C. Scientific Basis of Auriculotherapy: State of the Art. Med. Acupunct. 2014, 26, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Usichenko, T.; Hacker, H.; Lotze, M. Transcutaneous auricular vagal nerve stimulation (taVNS) might be a mechanism behind the analgesic effects of auricular acupuncture. Brain Stimul. 2017, 10, 1042–1044. [Google Scholar] [CrossRef] [PubMed]
- Napadow, V.; Edwards, R.R.; Cahalan, C.M.; Mensing, G.; Greenbaum, S.; Valovska, A.; Li, A.; Kim, J.; Maeda, Y.; Park, K.; et al. Evoked pain analgesia in chronic pelvic pain patients using respiratory-gated auricular vagal afferent nerve stimulation. Pain Med. 2012, 13, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Capone, F.; Assenza, G.; Di Pino, G.; Musumeci, G.; Ranieri, F.; Florio, L.; Barbato, C.; Di Lazzaro, V. The effect of transcutaneous vagus nerve stimulation on cortical excitability. J. Neural Transm. (Vienna) 2014, 122, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Antonino, D.; Teixeira, A.L.; Maia-Lopes, P.M.; Souza, M.C.; Sabino-Carvalho, J.L.; Murray, A.R.; Deuchars, J.; Vianna, L.C. Non-invasive vagus nerve stimulation acutely improves spontaneous cardiac baroreflex sensitivity in healthy young men: A randomized placebo-controlled trial. Brain Stimul. 2017, 10, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Shu, J.; Liu, R.-Y.; Huang, X.-F. Efficacy of ear-point stimulation on experimentally induced seizure. Acupunct. Electrother. Res. 2005, 30, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Huang, H.-M.; Xu, Y.-J.; Lu, R.-L.; Zhou, X.-H.; Zhou, C. Controlled study of auricular point taping and pressing therapy for treatment of vascular dementia. Zhongguo Zhen Jiu 2009, 29, 95–97. [Google Scholar] [PubMed]
- Romoli, M.; Allais, G.; Airola, G.; Benedetto, C.; Mana, O.; Giacobbe, M.; Pugliese, A.M.; Battistella, G.; Fornari, E. Ear acupuncture and fMRI: A pilot study for assessing the specificity of auricular points. Neurol. Sci. 2014, 35 (Suppl. 1), 189–193. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-T.; Lin, Y.-W.; Tang, N.-Y.; Cheng, C.-Y.; Hsieh, C.-L. Electric stimulation of the ears ameliorated learning and memory impairment in rats with cerebral ischemia-reperfusion injury. Sci. Rep. 2016, 6, 20381. [Google Scholar] [CrossRef] [PubMed]
- Van Leusden, J.W.R.; Sellaro, R.; Colzato, L.S. Transcutaneous Vagal Nerve Stimulation (tVNS): A new neuromodulation tool in healthy humans? Front. Psychol. 2015, 6, 102. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mercante, B.; Deriu, F.; Rangon, C.-M. Auricular Neuromodulation: The Emerging Concept beyond the Stimulation of Vagus and Trigeminal Nerves. Medicines 2018, 5, 10. https://doi.org/10.3390/medicines5010010
Mercante B, Deriu F, Rangon C-M. Auricular Neuromodulation: The Emerging Concept beyond the Stimulation of Vagus and Trigeminal Nerves. Medicines. 2018; 5(1):10. https://doi.org/10.3390/medicines5010010
Chicago/Turabian StyleMercante, Beniamina, Franca Deriu, and Claire-Marie Rangon. 2018. "Auricular Neuromodulation: The Emerging Concept beyond the Stimulation of Vagus and Trigeminal Nerves" Medicines 5, no. 1: 10. https://doi.org/10.3390/medicines5010010
APA StyleMercante, B., Deriu, F., & Rangon, C.-M. (2018). Auricular Neuromodulation: The Emerging Concept beyond the Stimulation of Vagus and Trigeminal Nerves. Medicines, 5(1), 10. https://doi.org/10.3390/medicines5010010





