The Chemotaxonomy of Common Sage (Salvia officinalis) Based on the Volatile Constituents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Essential Oils
2.2. Gas Chromatography-Mass Spectrometry
2.3. Quantitative Gas Chromatography
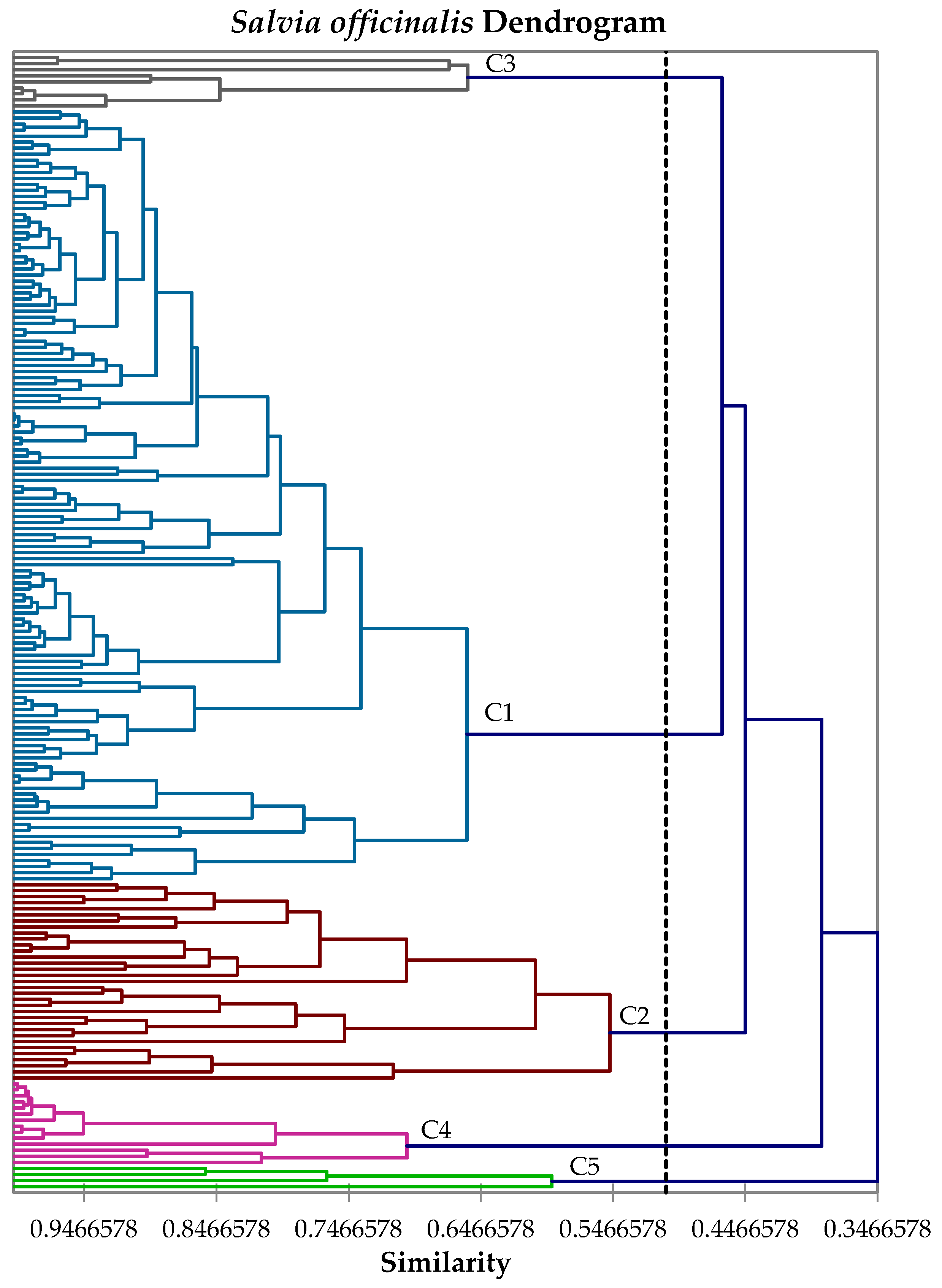
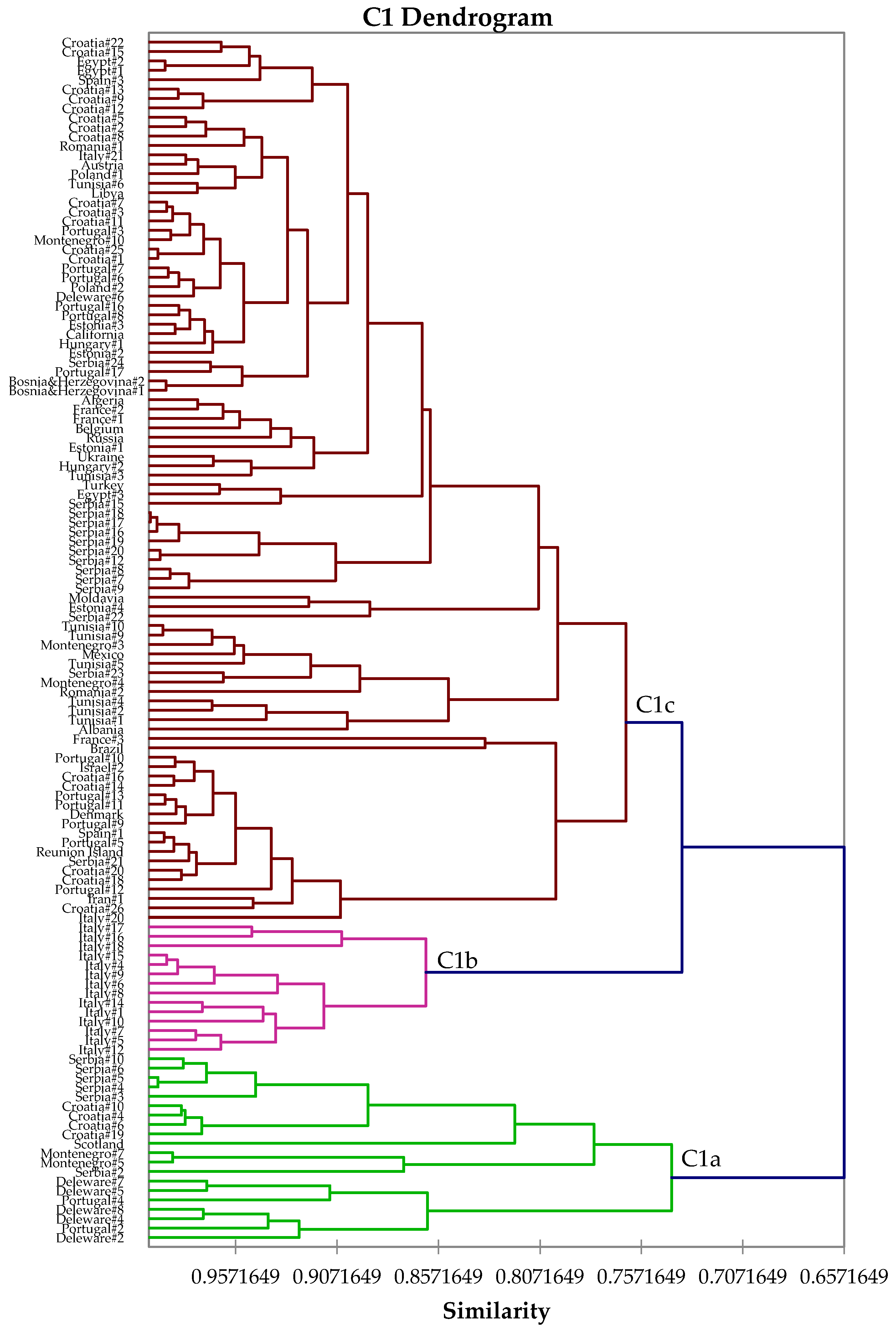
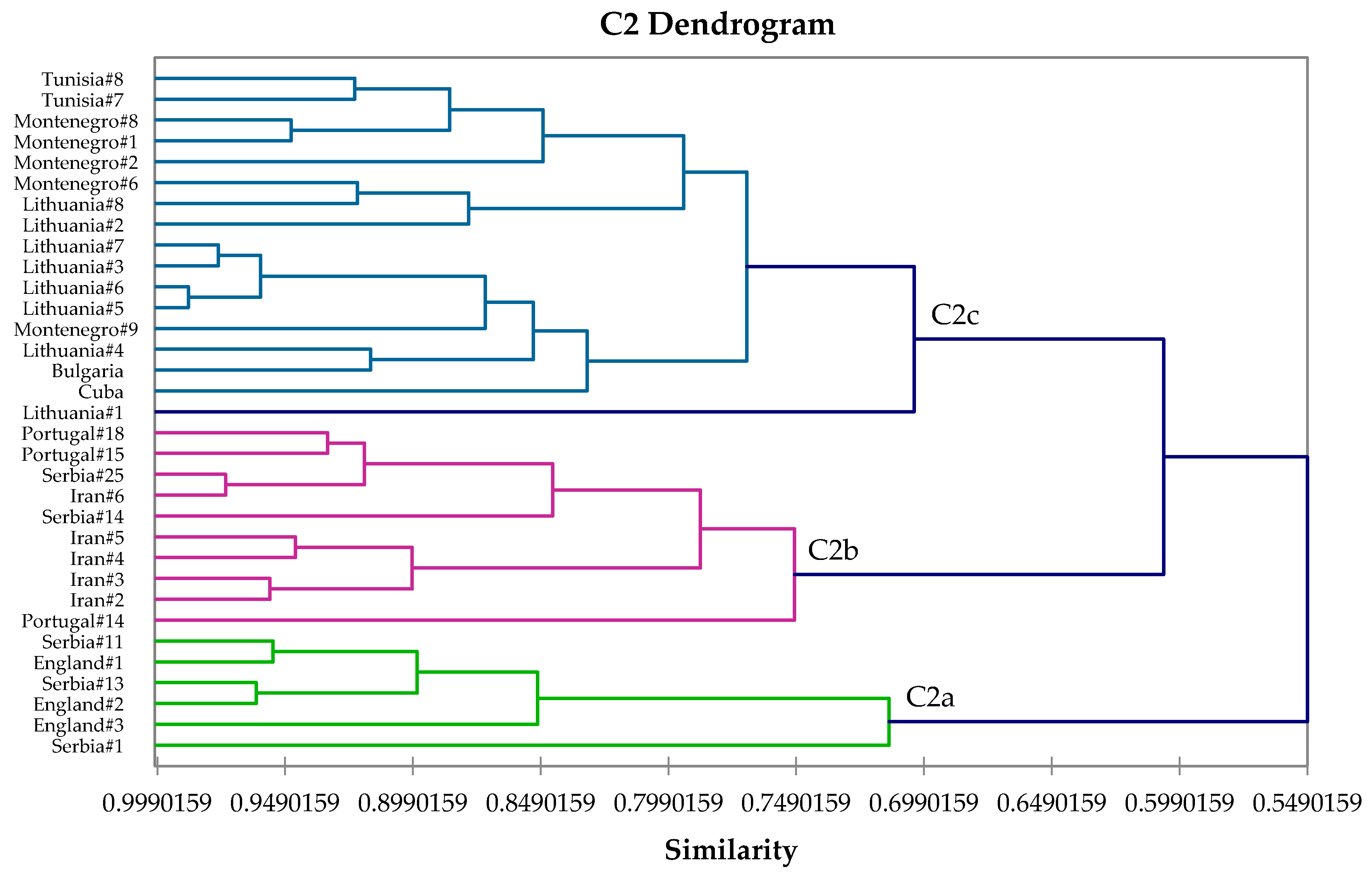
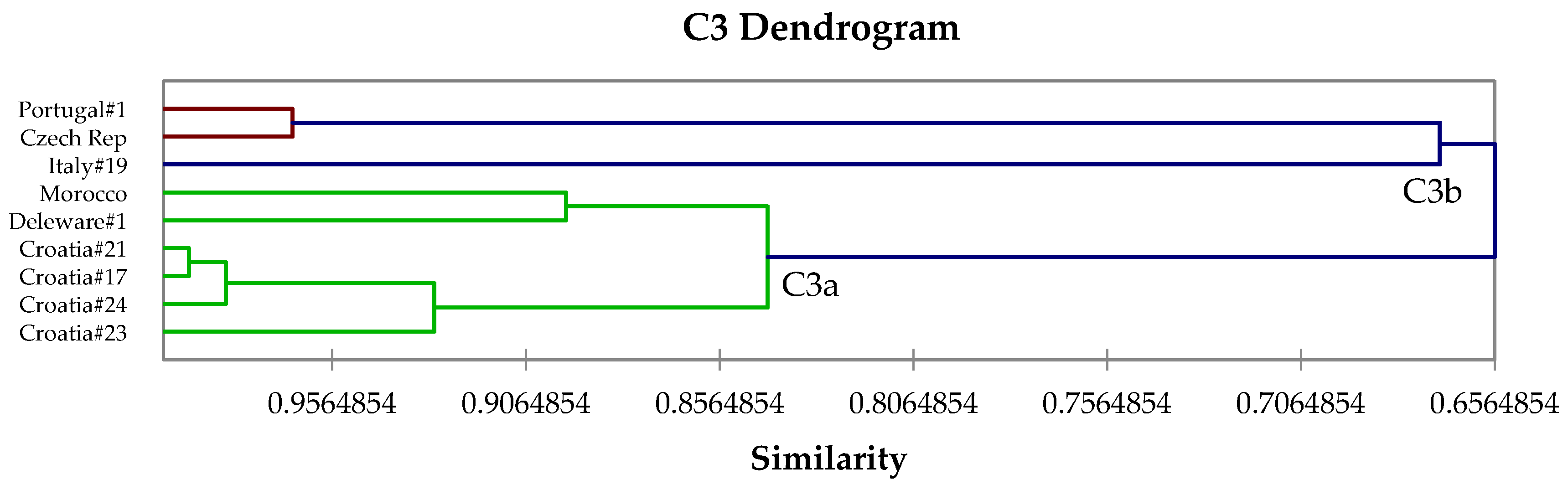
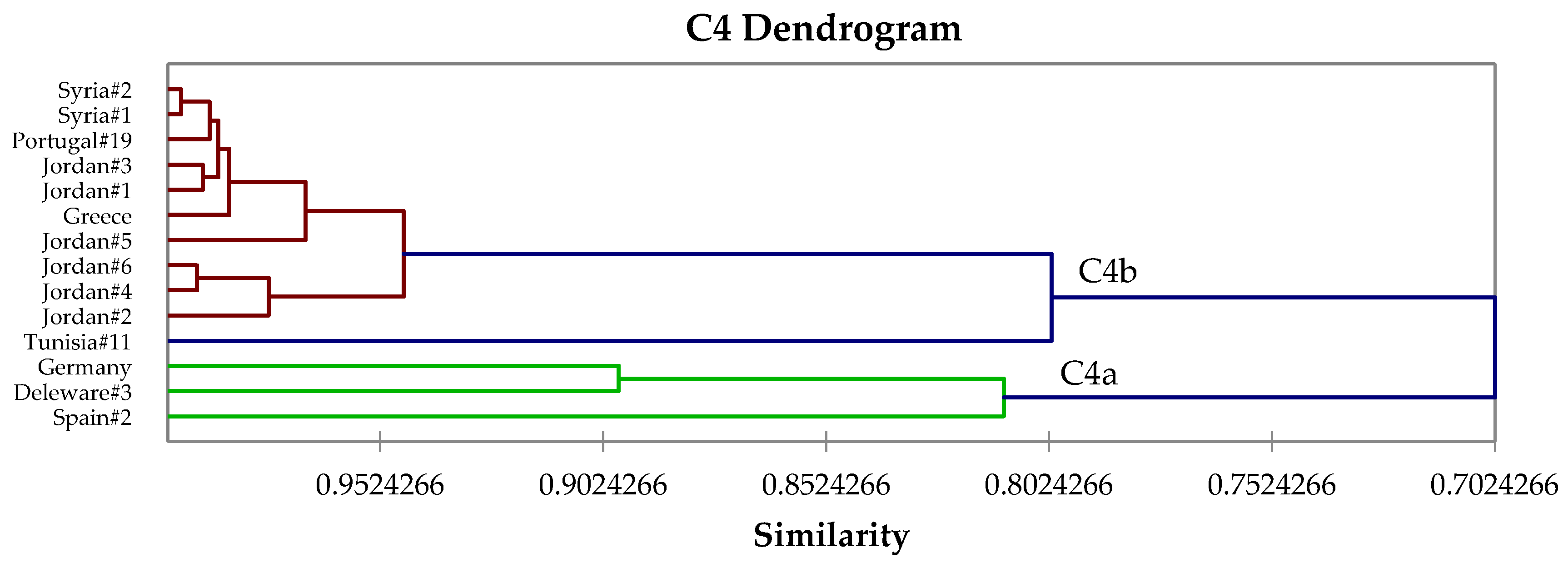
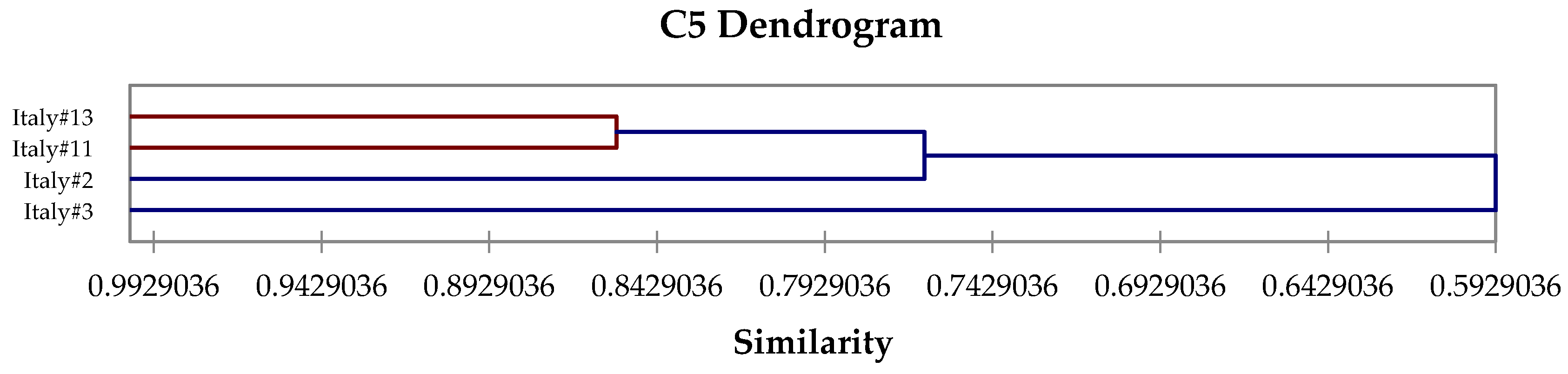
2.4. Hierarchical Cluster Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Lewis, W.H.; Elvin-Lewis, M.P.F. Medical Botany-Plants Affecting Man’s Health; John Wiley & Sons Ltd.: New York, NY, USA, 1977. [Google Scholar]
- Bruneton, J. Pharmacognosy, 2nd ed.; Intercept Ltd.: London, UK, 1999. [Google Scholar]
- Moerman, D.E. Native American Ethnobotany; Timber Press Inc.: Portland, OR, USA, 1998. [Google Scholar]
- Lachenmeier, D.W.; Emmert, J.; Kuballa, T.; Sartor, G. Thujone-Cause of absinthism? Forensic Sci. Int. 2006, 158, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Höld, K.M.; Sirisoma, N.S.; Ikeda, T.; Narahashi, T.; Casida, J.E. α-Thujone (the active component of absinthe): γ-Aminobutyric acid type A receptor modulation and metabolic detoxification. Proc. Natl. Acad. Sci. USA 2000, 97, 3826–3831. [Google Scholar] [CrossRef] [PubMed]
- Höld, K.M.; Sirisoma, N.S.; Casida, J.E. Detoxification of α- and β-thujones (the active ingredients of absinthe): Site specificity and species differences in cytochrome P450 oxidation in vitro and in vivo. Chem. Res. Toxicol. 2001, 14, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Pelkonen, O.; Abass, K.; Wiesner, J. Thujone and thujone-containing herbal medicinal and botanical products: Toxicological assessment. Regulat. Toxicol. Pharmacol. 2013, 65, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Waidyanatha, S.; Johnson, J.D.; Hong, S.P.; Robinson, V.G.; Gibbs, S.; Graves, S.W.; Hooth, M.J.; Smith, C.S. Toxicokinetics of α-thujone following intravenous and gavage administration of α-thujone or α- and β-thujone mixture in male and female F344/N rats and B6C3F1 mice. Toxicol. Appl. Pharmacol. 2013, 271, 216–228. [Google Scholar] [CrossRef] [PubMed]
- National Toxicology Program. NTP Technical Report on the Toxicology and Carcinogenesis Studies of α,β-Thujone in F344/N Rats and B6C3F1 Mice; Research Triangle Park: North Carolina, NC, USA, 2011. [Google Scholar]
- Lachenmeier, D.W.; Uebelacker, M. Risk assessment of thujone in foods and medicines containing sage and wormwood-Evidence for a need of regulatory changes? Regulat. Toxicol. Pharmacol. 2010, 58, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Meschler, J.P.; Howlett, A.C. Thujone exhibits low affinity for cannabinoid receptors but fails to evoke cannabimimetic responses. Pharmacol. Biochem. Behav. 1999, 62, 473–480. [Google Scholar] [CrossRef]
- Deiml, T.; Haseneder, R.; Zieglgänsberger, W.; Rammes, G.; Eisensamer, B.; Rupprecht, R.; Hapfelmeier, G. α-Thujone reduces 5-HT3 receptor activity by an effect on the agonist-induced desensitization. Neuropharmacol. 2004, 46, 192–201. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Abu-Darwish, M.S.; Cabral, C.; Ferreira, I.V.; Gonçalves, M.J.; Cavaleiro, C.; Cruz, M.T.; Al-Bdour, T.H.; Salgueiro, L. Essential oil of common sage (Salvia officinalis L.) from Jordan: Assessment of safety in mammalian cells and its antifungal and anti-inflammatory potential. BioMed. Res. Int. 2013, 2013, ID538940. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.; Shaabani, M. Essential oil composition, phenolic content, antioxidant and antimicrobial activity in Salvia officinalis L. cultivated in Iran. Adv. Environ. Biol. 2012, 6, 221–226. [Google Scholar]
- Avato, P.; Fortunato, I.M.; Ruta, C.; D’Elia, R. Glandular hairs and essential oils in micropropagated plants of Salvia officinalis L. Plant Sci. 2005, 169, 29–36. [Google Scholar] [CrossRef]
- Awen, B.Z.; Unnithan, C.R.; Ravi, S.; Kermagy, A.; Prabhu, V.; Hemlal, H. Chemical composition of Salvia officinalis essential oil of Libya. J. Essent. Oil Bear. Plants 2011, 14, 89–94. [Google Scholar] [CrossRef]
- Aziz, E.E.; Sabry, R.M.; Ahmed, S.S. Plant growth and essential oil production of sage (Salvia officinalis L.) and curly-leafed parsley (Petroselinum crispum ssp. crispum L.) cultivated under salt stress conditions. World Appl. Sci. J. 2013, 28, 785–796. [Google Scholar]
- Bayrak, A.; Akgül, A. Composition of essential oils from Turkish Salvia species. Phytochemistry 1987, 26, 846–847. [Google Scholar] [CrossRef]
- Bettaieb, I.; Zakhama, N.; Wannes, W.A.; Kchouk, M.E.; Marzouk, B. Water deficit effects on Salvia officinalis fatty acids and essential oils composition. Sci. Hortic. 2009, 120, 271–275. [Google Scholar] [CrossRef]
- Bouajaj, S.; Benyamna, A.; Bouamama, H.; Romane, A.; Falconieri, D.; Piras, A.; Marongiu, B. Antibacterial, allelopathic and antioxidant activities of essential oil of Salvia officinalis L. growing wild in the Atlas Mountains of Morocco. Nat. Prod. Res. 2013, 27, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Bouaziz, M.; Yangui, T.; Sayadi, S.; Dhouib, A. Disinfectant properties of essential oils from Salvia officinalis L. cultivated in Tunisia. Food Chem. Toxicol. 2009, 47, 2755–2760. [Google Scholar] [CrossRef] [PubMed]
- Bozin, B.; Mimica-Dukić, N.; Samojilik, I.; Jovin, E. Antimicrobial and antioxidant properties of rosemary and sage (Rosmarinus officinalis L. and Salvia officinalis L.) essential oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef] [PubMed]
- Carta, C.; Moretti, M.D.L.; Peana, A.T. Activity of the oil of Salvia officinalis L. against Botrytis cinerea. J. Essent. Oil Res. 1996, 8, 399–404. [Google Scholar] [CrossRef]
- Chalchat, J.C.; Michet, A.; Pasquier, B. Study of clones of Salvia officinalis L. Yields and chemical composition of essential oil. Flavour Fragr. J. 1998, 13, 68–70. [Google Scholar] [CrossRef]
- Couladis, M.; Tzakou, O.; Mimica-Dukić, N.; Jančić, R.; Stojanović, D. Essential oil of Salvia officinalis L. from Serbia and Montenegro. Flavour Fragr. J. 2002, 17, 119–126. [Google Scholar] [CrossRef]
- Damjanovic-Vratnica, B.; Ðakov, T.; Šukovic, D.; Damjanovic, J. Chemical composition and antimicrobial activity of essential oil of wild-growing Salvia officinalis L. from Montenegro. J. Essent. Oil Bear. Plants 2008, 11, 79–89. [Google Scholar] [CrossRef]
- Longaray Delamare, A.P.; Moschen-Pistorello, I.T.; Artico, L.; Atti-Serafini, L.; Echeverrigaray, S. Antibacterial activity of the essential oils of Salvia officinalis L. and Salvia triloba L. cultivated in South Brazil. Food Chem. 2007, 100, 603–608. [Google Scholar] [CrossRef]
- Dob, T.; Berramdane, T.; Dahmane, D.; Benabdelkader, T.; Chelghoum, C. Chemical composition of the essential oil of Salvia officinalis L. from Algeria. Chem. Nat. Comp. 2007, 43, 491–494. [Google Scholar] [CrossRef]
- Edris, A.E.; Jirovetz, L.; Buchbauer, G.; Denkova, Z.; Stoyanova, A.; Slavchev, A. Chemical composition, antimicrobial activities and olfactive evaluation of a Salvia officinalis L. (sage) essential oil from Egypt. J. Essent. Oil Res. 2007, 19, 186–189. [Google Scholar] [CrossRef]
- El Hadri, A.; Gómez del Río, M.Á.; Sanz, J.; González Coloma, A.; Idaomar, M.; Ribas Ozonas, B.; Benedí González, J.; Sánchez Reus, M.I. Cytotoxic activity of α-humulene and trans-caryophyllene from Salvia officinalis in animal and human tumor cells. Anal. Real Acad. Nac. Farm. 2010, 76, 343–356. [Google Scholar]
- Farhat, M.B.; Jordán, M.J.; Chaouech-Hamada, R.; Landoulsi, A.; Sotomayor, J.A. Variations in essential oil, phenolic compounds, and antioxidant activity of Tunisian cultivated Salvia officinalis L. J. Agric. Food Chem. 2009, 57, 10349–10356. [Google Scholar] [CrossRef] [PubMed]
- Fellah, S.; Diouf, P.N.; Petrissans, M.; Perrin, D.; Romdhane, M.; Abderrabba, M. Chemical composition and antioxidant properties of Salvia officinalis L. oil from two culture sites in Tunisia. J. Essent. Oil Res. 2006, 18, 553–556. [Google Scholar] [CrossRef]
- Geneva, M.P.; Stancheva, I.V.; Boychinova, M.M.; Mincheva, N.H.; Yonova, P.A. Effects of foliar fertilization and arbuscular mycorrhizal colonization on Salvia officinalis L. growth, antioxidant capacity, and essential oil composition. J. Sci. Food Agric. 2010, 90, 696–702. [Google Scholar] [PubMed]
- Guillen, M.D.; Cabo, N.; Burillo, J. Characterisation of the essential oils of some cultivated aromatic plants of industrial interest. J. Sci. Food Agric. 1996, 70, 359–363. [Google Scholar] [CrossRef]
- Hayouni, E.A.; Chraief, I.; Abedrabba, M.; Bouix, M.; Leveau, J.Y.; Mohammed, H.; Hamdi, M. Tunisian Salvia officinalis L. and Schinus molle L. essential oils: Their chemical compositions and their preservative effects against Salmonella inoculated in minced beef meat. Int. J. Food Microbiol. 2008, 125, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Jug-Dujaković, M.; Ristić, M.; Pljevljakušić, D.; Dajić-Stevanović, Z.; Liber, Z.; Hančević, K.; Radić, T.; Šatović, Z. High diversity of indigenous populations of Dalmatian sage (Salvia officinalis L.) in essential-oil composition. Chem. Biodivers. 2012, 9, 2309–2323. [Google Scholar] [CrossRef] [PubMed]
- Khalil, R.; Li, Z.G. Antimicrobial activity of essential oil of Salvia officinalis L. collected in Syria. Afr. J. Biotechnol. 2011, 10, 8397–8402. [Google Scholar]
- Knezevic-Vukcevic, J.; Vukovic-Gacic, B.; Stevic, T.; Stanojevic, J.; Nikolic, B.; Simic, D. Antimutagenic effect of essential oil of sage (Salvia officinalis L.) and its fractions against UV-induced mutations in bacterial and yeast cells. Arch. Biol. Sci. 2005, 57, 163–172. [Google Scholar] [CrossRef]
- Laborda, R.; Manzano, I.; Gamón, M.; Gavidia, I.; Pérez-Bermúdez, P.; Boluda, R. Effects of Rosmarinus officinalis and Salvia officinalis essential oils on Tetranychus urticae Koch (Acari: Tetranychidae). Ind. Crops Prod. 2013, 48, 106–110. [Google Scholar] [CrossRef]
- Lakušić, B.S.; Ristić, M.S.; Slavkovska, V.N.; Stojanović, D.L.J.; Lakušić, D.V. Variations in essential oil yields and compositions of Salvia officinalis (Lamiaceae) at different developmental stages. Bot. Serb. 2013, 37, 127–139. [Google Scholar]
- Länger, R.; Mechtler, C.; Jurenitsch, J. Composition of the essential oils of commercial samples of Salvia officinalis L. and S. fruticosa Miller: A comparison of oils obtained by extraction and steam distillation. Phytochem. Anal. 1996, 7, 289–293. [Google Scholar] [CrossRef]
- Lima, C.F.; Carvalho, F.; Fernandes, E.; Bastos, M.L.; Santos-Gomes, P.C.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Evaluation of toxic/protective effects of the essential oil of Salvia officinalis on freshly isolated rat hepatocytes. Toxicol. Vitro 2004, 18, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Maksimović, M.; Vidic, D.; Miloš, M.; Edita Šolić, M.; Abadžić, S.; Siljak-Yakovlev, S. Effect of the environmental conditions on essential oil profile in two Dinaric Salvia species: S. brachyodon Vandas and S. officinalis L. Biochem. Syst. Ecol. 2007, 35, 473–478. [Google Scholar] [CrossRef]
- Maric, S.; Maksimovic, M.; Milos, M. The impact of the locality altitudes and stages of development on the volatile constituents of Salvia officinalis L. from Bosnia and Herzegovina. J. Essent. Oil Res. 2006, 18, 178–180. [Google Scholar] [CrossRef]
- Miguel, G.; Cruz, C.; Faleiro, M.L.; Simões, M.T.F.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Salvia officinalis L. essential oils: Effect of hydrodistillation time on the chemical composition, antioxidant and antimicrobial activities. Nat. Prod. Res. 2011, 25, 526–541. [Google Scholar] [CrossRef] [PubMed]
- Milhau, G.; Valentin, A.; Benoit, F.; Mallié, M.; Bastide, J.M.; Pélissier, Y.; Bessiere, J.M. In vitro antimalarial activity of eight essential oils. J. Essent. Oil Res. 1997, 9, 329–333. [Google Scholar] [CrossRef]
- Mirjalili, M.H.; Salehi, P.; Sonboli, A.; Vala, M.M. Essential oil variation of sage (Salvia officinalis L.) aerial parts during its phenological cycle. Chem. Nat. Comp. 2006, 42, 19–23. [Google Scholar] [CrossRef]
- Pace, L.; Piccaglia, R. Characterization of the essential oil of a wild Italian endemic sage: Salvia officinalis L. var. angustifolia Ten. (Labiatae). J. Essent. Oil Res. 1995, 7, 443–446. [Google Scholar] [CrossRef]
- Pino, J.A.; Estarrón, M.; Fuentes, V. Essential oil of sage (Salvia officinalis L.) grown in Cuba. J. Essent. Oil Res. 1997, 9, 221–222. [Google Scholar] [CrossRef]
- Pinto, E.; Salgueiro, L.R.; Cavaleiro, C.; Palmeira, A.; Gonçalves, M.J. In vitro susceptibility of some species of yeasts and filamentous fungi to essential oils of Salvia officinalis. Ind. Crops Prod. 2007, 26, 135–141. [Google Scholar] [CrossRef]
- Raal, A.; Orav, A.; Arak, E. Composition of the essential oil of Salvia officinalis L. from various European countries. Nat. Prod. Res. 2007, 21, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Radulescu, V.; Chiliment, S.; Oprea, E. Capillary gas chromatography-mass spectrometry of volatile and semi-volatile compounds of Salvia officinalis. J. Chromatogr. A 2004, 1027, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Formisano, C.; Rigano, D.; Senatore, F.; Delfine, S.; Cardile, V.; Rosselli, S.; Bruno, M. Chemical composition and anticancer activity of essential oils of Mediterranean sage (Salvia officinalis L.) grown in different environmental conditions. Food Chem. Toxicol. 2013, 55, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Santos-Gomes, P.C.; Fernandes-Ferreira, M. Organ- and season-dependent variation in the essential oil composition of Salvia officinalis L. cultivated at two different sites. J. Agric. Food Chem. 2001, 49, 2908–2916. [Google Scholar] [CrossRef] [PubMed]
- Savelev, S.U.; Okello, E.J.; Perry, E.K. Butyryl- and acetyl-cholinesterase inhibitory activities in essential oils of Salvia species and their constituents. Phytother. Res. 2004, 18, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Seidler-Łożykowska, K.; Mordalski, R.; Król, D.; Bocianowski, J.; Karpińska, E. Yield and quality of sage herb (Salvia officinalis L.) from organic cultivation. Biol. Agric. Hortic. 2015, 31, 53–60. [Google Scholar] [CrossRef]
- Sellami, I.H.; Rebey, I.B.; Sriti, J.; Rahali, F.Z.; Limam, F.; Marzouk, B. Drying sage (Salvia officinalis L.) plants and its effects on content, chemical composition, and radical scavenging activity of the essential oil. Food Bioprocess Technol. 2012, 5, 2978–2989. [Google Scholar] [CrossRef]
- Sharopov, F.S.; Satyal, P.; Setzer, W.N.; Wink, M. Chemical compositions of the essential oils of three Salvia species cultivated in Germany. Am. J. Essent. Oils Nat. Prod. 2015, 3, 26–29. [Google Scholar]
- Taarit, M.B.; Msaada, K.; Hosni, K.; Marzouk, B. Changes in fatty acid and essential oil composition of sage (Salvia officinalis L.) leaves under NaCl stress. Food Chem. 2010, 119, 951–956. [Google Scholar] [CrossRef]
- Tucker, A.O.; Maciarello, M.J. Essential oils of cultivars of Dalmatian sage (Salvia officinalis L.). J. Essent. Oil Res. 1990, 2, 139–144. [Google Scholar] [CrossRef]
- Velickovic, D.T.; Ristic, M.S.; Randjelovic, N.V.; Smelcerovic, A.A. Chemical composition and antimicrobial characteristic of the essential oils obtained from the flower, leaf and stem of Salvia officinalis L. originating from southeast Serbia. J. Essent. Oil Res. 2002, 14, 453–458. [Google Scholar] [CrossRef]
- Venskutonis, P. Effect of drying on the volatile constituents of thyme (Thymus vulgaris L.) and sage (Salvia officinalis L.). Food Chem. 1997, 59, 219–227. [Google Scholar] [CrossRef]
- Vera, R.R.; Chane-Ming, J.; Fraisse, D.J. Chemical composition of the essential oil of sage (Salvia officinalis L.) from Reunion Island. J. Essent. Oil Res. 1999, 11, 399–402. [Google Scholar] [CrossRef]
- Zutic, I.; Putievsky, E.; Dudai, N. Influence of harvest dynamics and cut height on yield components of sage (Salvia officinalis L.). J. Herbs Spices Med. Plants 2003, 10, 49–61. [Google Scholar] [CrossRef]
- Bernotienė, G.; Nivinskienė, O.; Butkienė, R.; Mockutė, D. Essential oil composition variability in sage (Salvia officinalis L.). Chemija 2007, 18, 38–43. [Google Scholar]
- Jirovetz, L.; Buchbauer, G.; Denkova, Z.; Slavchev, A.; Stoyanova, A.; Schmidt, E. Chemical composition, antimicrobial activities and odor descriptions of various Salvia sp. and Thuja sp. essential oils. Ernährung/Nutrition 2006, 30, 152–159. [Google Scholar]
| RI a | RI b | Compound | Percent Composition c | ||
|---|---|---|---|---|---|
| Albania d | Mexico e | California f | |||
| 847 | 856 | (Z)-Salvene | 0.2 | 0.3 | 0.3 |
| 855 | 866 | (E)-Salvene | tr g | tr | 0.1 |
| 921 | 926 | Tricyclene | 0.2 | 0.1 | 0.2 |
| 926 | 930 | α-Thujene | 0.3 | 0.5 | 0.5 |
| 932 | 939 | α-Pinene | 5.0 | 2.4 | 5.2 |
| 945 | 954 | Camphene | 5.2 | 3.5 | 5.3 |
| 973 | 975 | Sabinene | 0.1 | 0.6 | - |
| 980 | 979 | β-Pinene | 4.1 | 2.6 | 1.3 |
| 981 | 979 | 1-Octen-3-ol | tr | 0.1 | 0.1 |
| 989 | 990 | Myrcene | 2.8 | 4.5 | 1.2 |
| 1000 | 1002 | α-Phellandrene | 0.1 | tr | - |
| 1018 | 1017 | α-Terpinene | 0.5 | tr | 0.2 |
| 1022 | 1024 | p-Cymene | 0.6 | 0.2 | 1.3 |
| 1029 | 1029 | Limonene | 1.5 | 1.4 | 2.2 |
| 1034 | 1031 | 1,8-Cineole | 26.9 | 15.5 | 11.9 |
| 1038 | 1037 | (Z)-β-Ocimene | 0.1 | 0.1 | - |
| 1042 | 1042 | Benzene acetaldehyde | - | tr | - |
| 1049 | 1050 | (E)-β-Ocimene | - | tr | - |
| 1059 | 1059 | γ-Terpinene | 0.7 | 0.7 | 0.4 |
| 1070 | 1070 | cis-Sabinene hydrate | 0.1 | 0.4 | - |
| 1086 | 1088 | Terpinolene | 0.2 | 0.2 | 0.3 |
| 1090 | 1091 | p-Cymenene | tr | - | - |
| 1100 | 1096 | Linalool | 0.3 | tr | 0.3 |
| 1103 | 1098 | trans-Sabinene hydrate | - | 0.4 | - |
| 1108 | 1102 | α-Thujone | 17.2 | 18.8 | 27.4 |
| 1118 | 1114 | β-Thujone | 3.8 | 4.4 | 6.0 |
| 1122 | 1127 | Chrysanthenone | tr | - | - |
| 1137 | 1138 | 3-iso-Thujanol | tr | - | - |
| 1147 | 1146 | Camphor | 12.8 | 14.9 | 21.4 |
| 1149 | 1151 | neo-iso-3-Thujanol | tr | - | - |
| 1161 | 1162 | trans-Pinocamphone | 0.1 | - | - |
| 1168 | 1168 | 3-Thujanol | 0.2 | - | - |
| 1169 | 1169 | Borneol | 1.2 | 1.0 | 1.7 |
| 1170 | 1166 | δ-Terpineol | 0.4 | 0.2 | - |
| 1180 | 1177 | Terpinen-4-ol | 0.5 | 0.6 | 0.4 |
| 1186 | 1188 | α-Terpineol | 1.1 | 0.4 | 0.4 |
| 1236 | 1237 | Ascaridole | - | 0.2 | - |
| 1254 | 1257 | Linalyl acetate | 0.2 | - | - |
| 1286 | 1288 | Bornyl acetate | 1.1 | 0.5 | 1.8 |
| 1294 | 1290 | trans-Sabinyl acetate | 0.1 | tr | 0.2 |
| 1337 | 1320 | 2,3-Pinanediol | tr | - | - |
| 1346 | 1249 | α-Terpinyl acetate | 0.6 | - | - |
| 1375 | 1376 | α-Copaene | 0.1 | - | - |
| 1419 | 1419 | β-Caryophyllene | 4.9 | 3.4 | 3.5 |
| 1432 | --- | 6-Oxobornyl acetate | tr | - | - |
| 1434 | 1433 | α-Maaliene | 0.1 | - | - |
| 1439 | 1441 | Aromadendrene | 0.4 | 0.2 | - |
| 1446 | 1444 | Myltayl-4(12)-ene | tr | - | - |
| 1448 | --- | 5-Oxobornyl acetate | 0.1 | - | - |
| 1453 | 1454 | α-Humulene | 3.1 | 5.7 | 4.4 |
| 1460 | 1460 | allo-Aromadendrene | - | 0.1 | 0.1 |
| 1467 | 1466 | 9-epi-β-Caryophyllene | 0.1 | - | - |
| 1476 | 1476 | trans-Cadina 1(6)-4-diene | 0.1 | - | - |
| 1482 | 1485 | Germacrene D | - | 0.1 | - |
| 1487 | 1483 | Guaia-1(10)-11-diene | 0.1 | - | - |
| 1496 | 1496 | Viridiflorene | 0.3 | - | 0.2 |
| 1497 | 1500 | Bicyclogermacrene | - | 0.1 | - |
| 1511 | 1523 | δ-Amorphene | 0.1 | - | - |
| 1517 | 1523 | δ-Cadinene | 0.1 | - | - |
| 1579 | 1578 | Spathulenol | - | 0.1 | - |
| 1583 | 1583 | Caryophyllene oxide | 0.1 | 0.2 | - |
| 1591 | 1592 | Viridiflorol | 2.0 | 7.4 | 1.5 |
| 1609 | 1608 | Humulene epoxide II | 0.2 | 0.3 | 0.2 |
| 1636 | 1640 | Caryophylla-4(12),8(13)-dien-5α-ol | 0.1 | - | - |
| 2056 | 2057 | Manool | 0.2 | 8.2 | - |
| Monoterpene Hydrocarbons | 21.5 | 17.0 | 18.5 | ||
| Oxygenated Monoterpenoids | 66.5 | 57.3 | 71.5 | ||
| Sesquiterpene Hydrocarbons | 9.4 | 9.5 | 8.2 | ||
| Oxygenated Sesquiterpenoids | 2.4 | 8.0 | 1.7 | ||
| Others | 0.2 | 8.2 | 0.1 | ||
| Total Identified | 100 | 100 | 100 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Craft, J.D.; Satyal, P.; Setzer, W.N. The Chemotaxonomy of Common Sage (Salvia officinalis) Based on the Volatile Constituents. Medicines 2017, 4, 47. https://doi.org/10.3390/medicines4030047
Craft JD, Satyal P, Setzer WN. The Chemotaxonomy of Common Sage (Salvia officinalis) Based on the Volatile Constituents. Medicines. 2017; 4(3):47. https://doi.org/10.3390/medicines4030047
Chicago/Turabian StyleCraft, Jonathan D., Prabodh Satyal, and William N. Setzer. 2017. "The Chemotaxonomy of Common Sage (Salvia officinalis) Based on the Volatile Constituents" Medicines 4, no. 3: 47. https://doi.org/10.3390/medicines4030047
APA StyleCraft, J. D., Satyal, P., & Setzer, W. N. (2017). The Chemotaxonomy of Common Sage (Salvia officinalis) Based on the Volatile Constituents. Medicines, 4(3), 47. https://doi.org/10.3390/medicines4030047








