Ecological Effects of Benzyl Chloride on Different Korean Aquatic Indigenous Species Using an Artificial Stream Mesocosm Simulating a Chemical Spill
Abstract
1. Introduction
2. Materials and Methods
2.1. Artificial Stream Mesocosm System
2.2. Test Organisms
2.3. Test Chemical and Exposure Scenario
2.4. Artificial Stream Mesocosm Study
2.4.1. Test Chemical Exposure
2.4.2. Observation of Test Organisms
- Periphyton algae
- Crustacea
- Fish
- Chironomidae and freshwater red worm
2.4.3. Water Parameters and Chemical Analysis
2.5. Animal Welfare and Institutional Animal Care and Use Committee
2.6. Statistical Analyses
2.7. Estimation of Hazard Concentration
3. Results and Discussion
3.1. Benzyl Chloride Concentration in Water
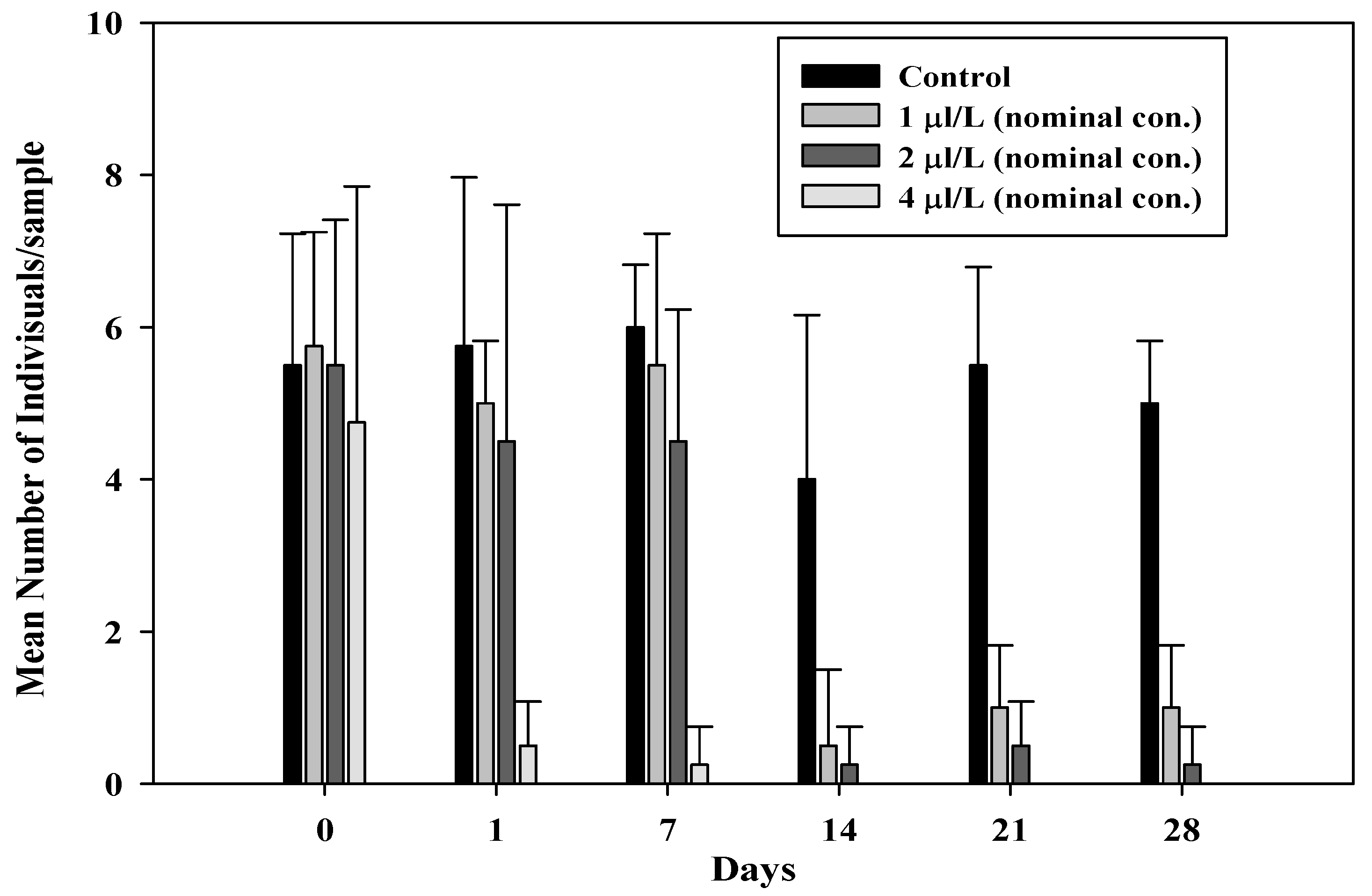
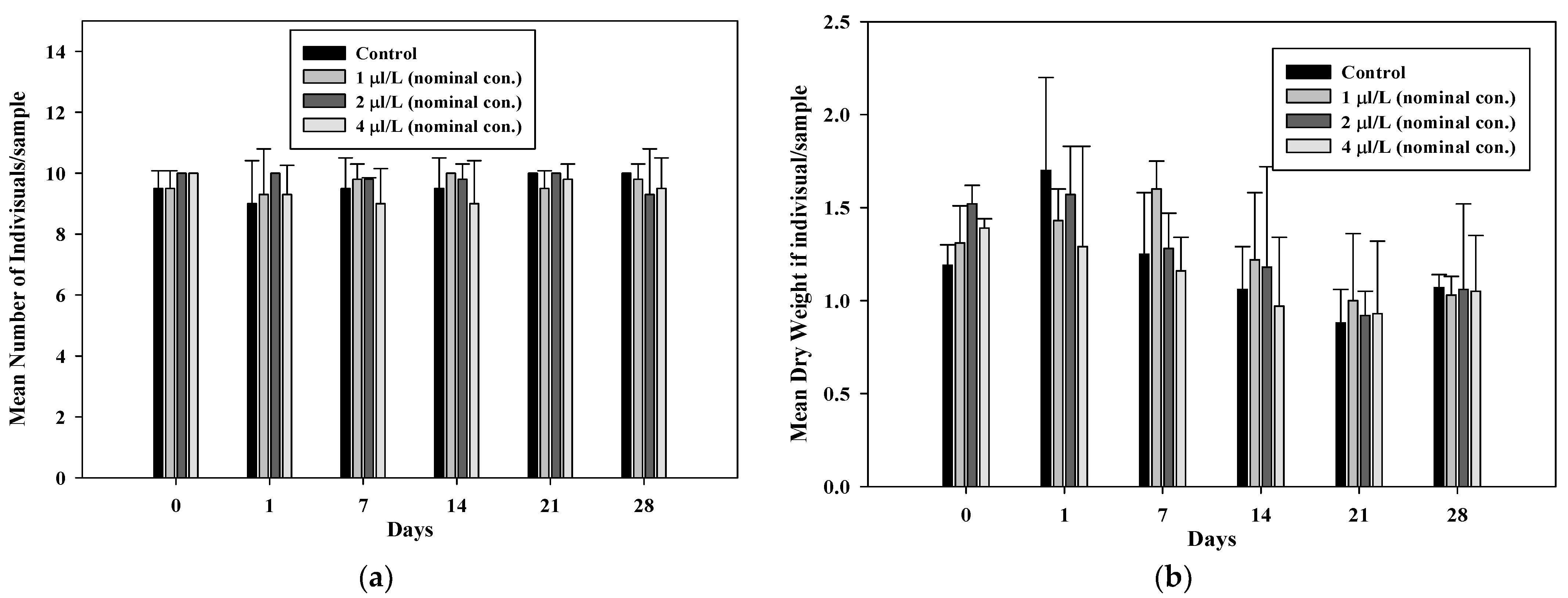
3.2. Effect of Test Organisms
- Periphyton algae
- Crustacea
- Fish
- Chironomidae and freshwater red worm
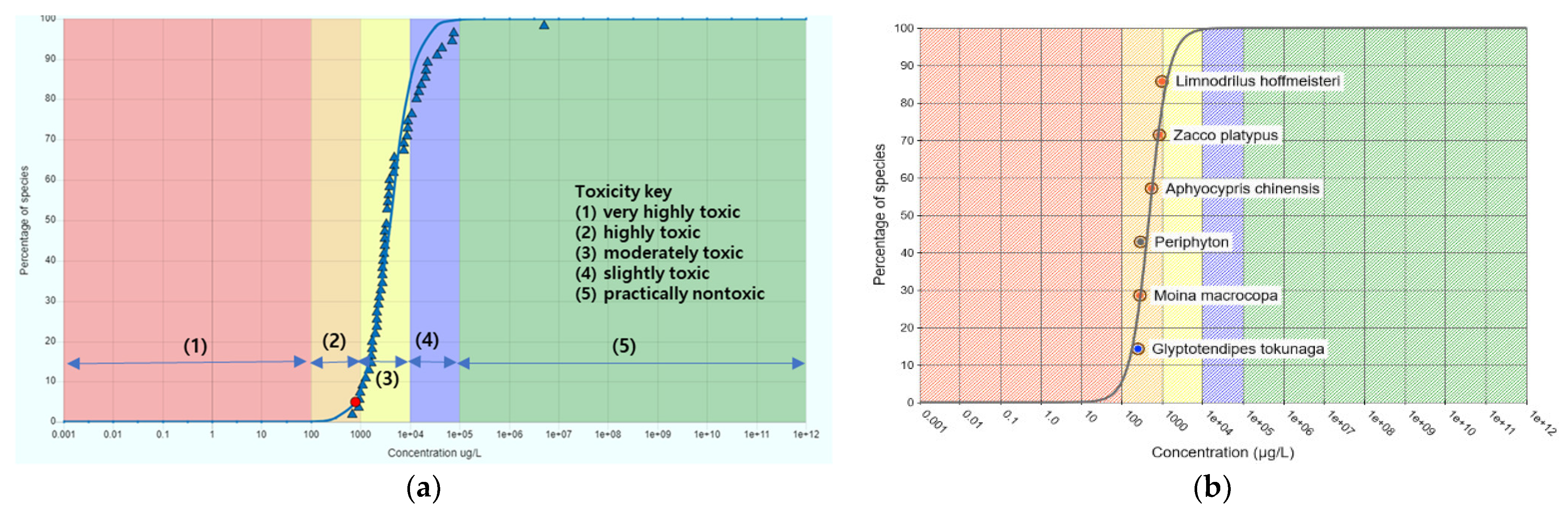
3.3. Estimation of Hazard Concentration
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- U.S. EPA. Health And Environmental Effects Profile for Benzyl Chloride; EPA/600/X-86/148 (NTIS PB88219449); U.S. Environmental Protection Agency: Washington, DC, USA, 1986.
- HIS Markit, Chemical Economics Handbook, Benzyl Chloride. 2018. Available online: https://ihsmarkit.com/products/benzyl-chloride-chemical-economics-handbook.html (accessed on 12 March 2021).
- Huh, D.-A.; Huh, E.-H.; Byeon, S.-H.; Sohn, J.-R.; Moon, K.W. Development of accident probability index using surrogate indicators of chemical accidents in chemical plants. Int. J. Environ. Res. Public Health 2019, 16, 3271. [Google Scholar] [CrossRef]
- FACTS Hazardous Materials Accidents Knowledge Base. Available online: http://www.factsonline.nl/accidents/%205405/91738_BENZYL%20CHLORIDE/chemical-accidents-with-benzyl-chloride (accessed on 11 January 2021).
- You, J.; Chung, Y.-J. Case Analysis of the Harmful Chemical Substances’ Spill. Fire Sci. Eng. 2014, 28, 90–98. [Google Scholar] [CrossRef]
- National Institute of Chemical Safety, Chemical Substance Information System. Available online: https://icis.me.go.kr/main.do (accessed on 12 March 2021).
- Lee, K.; Kwon, H.-M.; Cho, S.; Kim, J.; Moon, I. Improvements of safety management system in Korean chemical industry after a large chemical accident. J. Loss Prev. Process Ind. 2016, 42, 6–13. [Google Scholar] [CrossRef]
- Chae, Y.; Cui, R.; Moon, J.; An, Y.-J. Ecological hazard assessment of methyl ethyl ketone using the species sensitivity distribution approach in a soil ecosystem. J. Hazard. Mater. 2018, 360, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.J.; Park, S.H.; Lim, H.B.; Lee, C.M. Development on Health Risk Assessment Method for Multi-Media Exposure of Hazardous Chemical by Chemical Accident. Int. J. Environ. Res. Public Health 2020, 17, 3385. [Google Scholar] [CrossRef] [PubMed]
- McReady, D.; Williams, J.B. Risk Assessment for a Chemical Spill into a River. J. Risk Anal. Crisis Response 2013, 3, 116–126. [Google Scholar] [CrossRef][Green Version]
- Lamberti, G.A.; Steinman, A.D. Research in Artificial Streams: Applications, Uses, and Abuses. J. N. Am. Benthol. Soc. 1993, 12, 313–384. [Google Scholar] [CrossRef]
- Solomon, K.R. Overview of Recent Developments in Ecotoxicological Risk Assessment. Risk Anal. 1996, 16, 627–633. [Google Scholar] [CrossRef]
- Boyle, T.P.; Fairchild, J.F. The role of mesocosm studies in ecological risk analysis. Ecol. Appl. 1997, 7, 1099–1102. [Google Scholar] [CrossRef]
- Clements, W.H.; Carlisle, D.M.; Courtney, L.A.; Harrahy, E.A. Integrating observational and experimental approaches to demonstrate causation in stream biomonitoring studies. Environ. Toxicol. Chem. 2002, 21, 1138–1146. [Google Scholar] [CrossRef]
- Giddings, J.M.; Solomon, K.R.; Maund, S.J. Probabilistic risk assessment of cotton pyrethroids: II. Aquatic mesocosm and field studies. Environ. Toxicol. Chem. 2001, 20, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Connolly, N.M.; Pearson, R.G. The effect of fine sedimentation on tropical stream macroinvertebrate assemblages: A comparison using flow-through artificial stream channels and recirculating mesocosms. Hydrobiologia 2007, 592, 423–438. [Google Scholar] [CrossRef]
- Quinlan, E.L.; Nietch, C.T.; Blocksom, K.; Lazorchak, J.M.; Batt, A.L.; Griffiths, R.; Klemm, D.J. Temporal Dynamics of Periphyton Exposed to Tetracycline in Stream Mesocosms. Environ. Sci. Technol. 2011, 45, 10684–10690. [Google Scholar] [CrossRef]
- Schmitz, J.; Stahlschmidt, P.; Brühl, C.A. Assessing the Risk of Herbicides to Terrestrial Non-target Plants using Higher-tier Studies. Hum. Ecol. Risk Assess. Int. J. 2015, 21, 2137–2154. [Google Scholar] [CrossRef]
- Beuter, L.-K.; Dören, L.; Hommen, U.; Kotthoff, M.; Schäfers, C.; Ebke, K.P. Testing effects of pesticides on macroinvertebrate communities in outdoor stream mesocosms using carbaryl as example test item. Environ. Sci. Eur. 2019, 31, 5. [Google Scholar] [CrossRef]
- Miller, J.L.; Schmidt, T.S.; Van Metre, P.C.; Mahler, B.J.; Sandstrom, M.W.; Nowell, L.H.; Carlisle, D.M.; Moran, P.W. Common insecticide disrupts aquatic communities: A mesocosm-to-field ecological risk assessment of fipronil and its degradates in U.S. streams. Sci. Adv. 2020, 6, eabc1299. [Google Scholar] [CrossRef] [PubMed]
- Reilly, T.J. The Use of Mesocosms in Marine Oil Spill Ecological Research and Development. Pure Appl. Chem. 1999, 71, 153–160. [Google Scholar] [CrossRef]
- Silva, D.P.; Duarte, G.; Villela, H.D.; Santos, H.; Rosado, P.M.; Rosado, J.G.; Rosado, A.S.; Ferreira, E.M.; Soriano, A.U.; Peixoto, R.S. Adaptable mesocosm facility to study oil spill impacts on corals. Ecol. Evol. 2019, 9, 5172–5185. [Google Scholar] [CrossRef] [PubMed]
- Morales-McDevitt, M.E.; Shi, D.; Knap, A.H.; Quigg, A.; Sweet, S.T.; Sericano, J.L.; Wade, T.L. Mesocosm experiments to better understand hydrocarbon half-lives for oil and oil dispersant mixtures. PLoS ONE 2020, 15, e0228554. [Google Scholar] [CrossRef] [PubMed]
- Aljaibachi, R.; Laird, W.B.; Stevens, F.; Callaghan, A. Impacts of polystyrene microplastics on Daphnia magna: A laboratory and a mesocosm study. Sci. Total Environ. 2020, 705, 135800. [Google Scholar] [CrossRef]
- Lott, C.; Eich, A.; Unger, B.; Makarow, D.; Battagliarin, G.; Schlegel, K.; Lasut, M.T.; Weber, M. Field and mesocosm methods to test biodegradable plastic film under marine conditions. PLoS ONE 2020, 15, e0236579. [Google Scholar] [CrossRef] [PubMed]
- McCready, D.; Williams, J.B. A simplified approach to evaluate human and aquatic exposure to a chemical spilled in a river. J. Hazard. Mater. 2011, 193, 225–232. [Google Scholar] [CrossRef]
- Chapman, P.M.; Fairbrother, A.; Brown, D. A critical evaluation of safety (uncertainty) factors for ecological risk assessment. Environ. Toxicol. Chem. 1998, 17, 99–108. [Google Scholar] [CrossRef]
- Bejarano, A.C.; Farr, J.K. Development of short, acute exposure hazard estimates: A tool for assessing the effects of chemical spills in aquatic environments. Environ. Toxicol. Chem. 2013, 32, 1918–1927. [Google Scholar] [CrossRef]
- OECD. Guidance Document on Simulated Freshwater Lentic Field Tests (Outdoor Microcosms and Mesocosms) OECD Environment Health and Safety Publications Series on Testing and Assessment No. 53; OECD: Paris, France, 2006. [Google Scholar]
- Kim, B.S.; Park, Y.G.; Park, G.H.; Jung, M.H.; Yu, A.S.; Yang, Y.J.; Shin, J.S.; Kim, J.H.; Yun, S.M.; Ahn, Y.J. Comparative toxicity of some pesticides on reproduction of Korean native freshwater Cladocerans, Moina macrocopa and Daphnia sp. Korean J. Pestic. Sci. 2007, 11, 246–253. [Google Scholar]
- Benider, A.; Tifnouti, A.; Pourriot, R. Growth of Moina macrocopa (Straus 1820) (Crustacea, Cladocera): Influence of trophic conditions, population density and temperature. Hydrobiologia 2002, 468, 1–11. [Google Scholar] [CrossRef]
- Nandini, S.; Sarma, S. Reproductive strategies of Moina (Cladocera) in relation to their habitat. Limnetica 2019, 38, 137–145. [Google Scholar] [CrossRef]
- Cho, H.; Yoo, J.; Han, Y.; Han, T.; Kim, S.; Jung, J. Toxicity Monitoring of River Sediments in the Geum River Basin using Daphnia magna and Moina macrocopa. J. Korean Soc. Water Environ. 2007, 26, 1000–1007. [Google Scholar]
- Yoon, J.-D.; Kim, J.-H.; Park, S.-H.; Jang, M.-H. The Distribution and Diversity of Freshwater Fishes in Korean Peninsula. Korean J. Ecol. Environ. 2018, 51, 71–85. [Google Scholar] [CrossRef]
- Kim, J.-H.; Yeom, D.-H.; Kim, W.-K.; An, K.-G. Regional Ecological Health or Risk Assessments of Stream Ecosystems Using Biomarkers and Bioindicators of Target Species (Pale Chub). Water Air Soil Pollut. 2016, 227, 469. [Google Scholar] [CrossRef]
- Park, C.-B.; Kim, G.-E.; Kim, D.-W.; Kim, S.; Yeom, D.-H. Biomonitoring the effects of urban-stream waters on the health status of pale chub (Zacco platypus): A comparative analysis of biological indexes and biomarker levels. Ecotoxicol. Environ. Saf. 2021, 208, 111452. [Google Scholar] [CrossRef] [PubMed]
- Baek, M.J.; Yoon, T.J.; Bae, Y.J. Development of Glyptotendipes tokunagai (Diptera: Chironomidae) Under Different Temperature Conditions. Environ. Entomol. 2012, 41, 950–958. [Google Scholar] [CrossRef]
- NIH. PubChem-Benzyl Chloride [WWW Document]. Natl. Libr. Med. 2004. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Benzyl-chloride (accessed on 26 July 2021).
- NICS (National Institute of Chemical Safety). Technical Guidelines on Accident Scenario Selection; NICS: Deajeon, Korea, 2014. [Google Scholar]
- Arar, J.E. EPA Method 446.0 In vitro determination of chlorophylls a, b, c 1c and pheopigments in marine and freshwater algae by visible spectrophotometry, Revision 1.2. In EPA/600/R-97/072; Methods for the Determination of Chemical Substances in Marine and Estuarine; 446.0-1; EPA(Environmental Protection Agency): Washington, DC, USA, 1997; pp. 1–22. [Google Scholar]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem. Physiol. Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Noh, W.; Park, S.; Lee, S.-J.; Ryu, B.-G.; Kim, J. The potential of a natural biopolymeric flocculant, ε-poly-l-lysine, for harvesting Chlorella ellipsoidea and its sustainability perspectives for cost and toxicity. Bioprocess. Biosyst. Eng. 2019, 42, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Froese, R. Cube law, condition factor and weight-length relationships: History, meta-analysis and recommendations. J. Appl. Ichthyol. 2006, 22, 241–253. [Google Scholar] [CrossRef]
- EPA. National Recommended Water Quality Criteria-Aquatic Life Criteria Table [WWW Document]. 2021. Available online: https://www.epa.gov/wqc/national-recommended-water-quality-criteria-aquatic-life-criteria-table (accessed on 22 July 2021).
- Bejarano, A.C.; Farr, J.K.; Jenne, P.; Chu, V.; Hielscher, A. The Chemical Aquatic Fate and Effects database (CAFE), a tool that supports assessments of chemical spills in aquatic environments. Environ. Toxicol. Chem. 2016, 35, 1576–1586. [Google Scholar] [CrossRef]
- Smit, C.E. Environmental Risk Limits for Benzyl Chloride and Benzylidene Chloride; National Institute for Public Health and the Environment (RIVM) Report 601714016; RIVM: Bilthoven, The Netherlands, 2010.
- Swann, R.L.; Laskowski, D.A.; McCall, P.J.; Kuy, K.; Dishburger, H.J. A rapid method for the estimation of the environmental parameters octanol/water partition coefficient, soil sorption constant, water to air ratio, and water solubility. Rev. Environ. Contam. Toxicol. 1983, 85, 17–28. [Google Scholar] [CrossRef]
- Environment Canada, Health Canada. Screening Assessment for the Challenge Benzene, (Chloromethyl)-(Benzyl Chloride); Chemical Abstracts Service Registry Number 100-44-7; Government of Canada: Vancouver, BC, Canada, 2009.
- Zou, T.; Zhang, J. A New Fluorescence Quantum Yield Efficiency Retrieval Method to Simulate Chlorophyll Fluorescence under Natural Conditions. Remote Sens. 2020, 12, 4053. [Google Scholar] [CrossRef]
- EPA (US Environmental Protection Agency). ECOTOX Knowledgebase [WWW Document]. 2021. Available online: https://cfpub.epa.gov/ecotox/quick_query.htm (accessed on 19 August 2021).



| Section | Size (cm) (Width × Length × Depth) | Substrate |
|---|---|---|
| Head tank | 50 × 100 × 100 | Water (80 cm depth) |
| Upper Riffle section | 30 × 200 × 10 (with slope approximately 4.3%) | Stone/tile (0.5 cm depth for tile, approximately 10 cm depth for stone) |
| Lower Run section | 30 × 200 × 10 | Gravel (2–5 cm depth) |
| Pool section | 30 × 100 × 40 | Water (30 cm depth) |
| Lower section | 40 × 200 × 20 | Sand/sediment (10 cm depth) |
| Tail tank | 40 × 100 × 50 | Water (30 cm depth) |
| Section | Test Species | Age | Acclimation Period | Number of Individuals | Endpoint | Study Parameter |
|---|---|---|---|---|---|---|
| Upper Riffle section | Periphyton algae (species not classified) | 30 days growth on tiles | 30 d | - | Growth | Chlorophyll-a, dry cell weight, quantum yield |
| dower Run section | Moina macrocopa | Neonates (<24 h) | - | 20 individuals (5/subreplicate, 4 subrep.) | Survival, fecundity | Mortality of parents, Number of living offsprings |
| Pool section | Zacco platypus | Approximately 2–3 months (4–5 cm in length) | 14 d | 50 individuals | Survival, behavior, growth | Mortality, behavior observation, length and weight |
| Lower section | Glyptotendipes tokunagai, Limnodrilus hoffmeisteri | 2nd instar larvae (G. tokunagai), adults (L. hoffmeisteri) | 1 d (G. tokunagai) 7 d (L. hoffmeisteri) | 240 individuals | Survival | Number of survived organisms |
| Tail tank | Aphyocypris chinensis | Approximately 2–3 months | 14 d | 50 individuals | Survival, behavior, growth | Mortality, behavior observation, length and weight |
| Released to | Fraction of Distribution to Each Medium (% of Total) | |||
|---|---|---|---|---|
| Air | Water | Soil | Sediment | |
| Water (100%) | 7.9 | 90 | 0.16 | 1.6 |
| Section | Mean Measured Concentration (±SD) (μL/L) | ||
|---|---|---|---|
| 1 µL/L | 2 µL/L | 4 µL/L | |
| Head Tank | 0.29 ± 0.17 | 0.58 ± 0.32 | 1.01 ± 0.08 |
| Upper Riffle section (Stone/tile) | 0.29 ± 0.17 | 0.41 ± 0.41 | 0.76 ± 0.33 |
| Lower Run section (Gravel) | 0.28 ± 0.15 | 0.52 ± 0.33 | 0.98 ± 0.04 |
| Pool section | 0.27 ± 0.14 | 0.45 ± 0.29 | 0.85 ± 0.09 |
| Lower section (Sand/sediment) | 0.25 ± 0.13 | 0.39 ± 0.26 | 0.74 ± 0.13 |
| Tail tank | 0.19 ± 0.13 | 0.27 ± 0.22 | 0.54 ± 0.23 |
| Nominal Concentration (µL/L) | a Survival Rate (±SD) (%) | a Total No. of Offspring | Average Offspring/Surviving Females/Day (±SD) |
|---|---|---|---|
| Control | 75.0 ± 25.0 | 159 | 1.03 ± 0.51 |
| 1 | 60.0 ± 22.4 | 142 | 1.04 ± 0.71 |
| 2 | 50.0 ± 25.0 | 104 * | 1.16 ± 0.31 |
| 4 | 5.0 ± 11.2 * | 38 * | 0.59 ± 0.45 |
| Nominal Concentration (µL/L) | Survival Rate (%) | Condition Factor (K) (±SD) | ||
|---|---|---|---|---|
| A. chinensis | Z. platypus | A. chinensis | Z. platypus | |
| Control | 100 | 97.5 | 1.09 ± 0.15 | 1.05 ± 0.26 |
| 1 | 100 | 100 | 1.10 ± 0.16 | 0.92 ± 0.18 |
| 2 | 97.5 | 100 | 1.09 ± 0.23 | 0.92 ± 0.19 |
| 4 | 100 | 82.5 | 1.05 ± 0.19 | 0.95 ± 0.21 |
| NOEC (μL/L) | |||||
|---|---|---|---|---|---|
| G. tokunaga | M. macrocopa | Periphyton Algae | A. chinensis | Z. platypus | L. hoffmeisteri |
| 0.25 | 0.28 | 0.29 | 0.54 | 0.85 | 0.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.-Y.; Park, S.-H.; Kim, D.-W.; Noh, W.; Lee, S.-J.; Jeong, H.-J.; Park, J.-B.; Gwak, Y.-J.; Park, J.-W.; Yeom, D.-H. Ecological Effects of Benzyl Chloride on Different Korean Aquatic Indigenous Species Using an Artificial Stream Mesocosm Simulating a Chemical Spill. Toxics 2021, 9, 347. https://doi.org/10.3390/toxics9120347
Kim S-Y, Park S-H, Kim D-W, Noh W, Lee S-J, Jeong H-J, Park J-B, Gwak Y-J, Park J-W, Yeom D-H. Ecological Effects of Benzyl Chloride on Different Korean Aquatic Indigenous Species Using an Artificial Stream Mesocosm Simulating a Chemical Spill. Toxics. 2021; 9(12):347. https://doi.org/10.3390/toxics9120347
Chicago/Turabian StyleKim, Soo-Yeon, Seong-Hwan Park, Dae-Wook Kim, Won Noh, Sang-Jun Lee, Hee-Jin Jeong, Jong-Bin Park, Yeong-Ji Gwak, Jin-Woo Park, and Dong-Hyuk Yeom. 2021. "Ecological Effects of Benzyl Chloride on Different Korean Aquatic Indigenous Species Using an Artificial Stream Mesocosm Simulating a Chemical Spill" Toxics 9, no. 12: 347. https://doi.org/10.3390/toxics9120347
APA StyleKim, S.-Y., Park, S.-H., Kim, D.-W., Noh, W., Lee, S.-J., Jeong, H.-J., Park, J.-B., Gwak, Y.-J., Park, J.-W., & Yeom, D.-H. (2021). Ecological Effects of Benzyl Chloride on Different Korean Aquatic Indigenous Species Using an Artificial Stream Mesocosm Simulating a Chemical Spill. Toxics, 9(12), 347. https://doi.org/10.3390/toxics9120347





