Pharmacogenetics and Forensic Toxicology: A New Step towards a Multidisciplinary Approach
Abstract
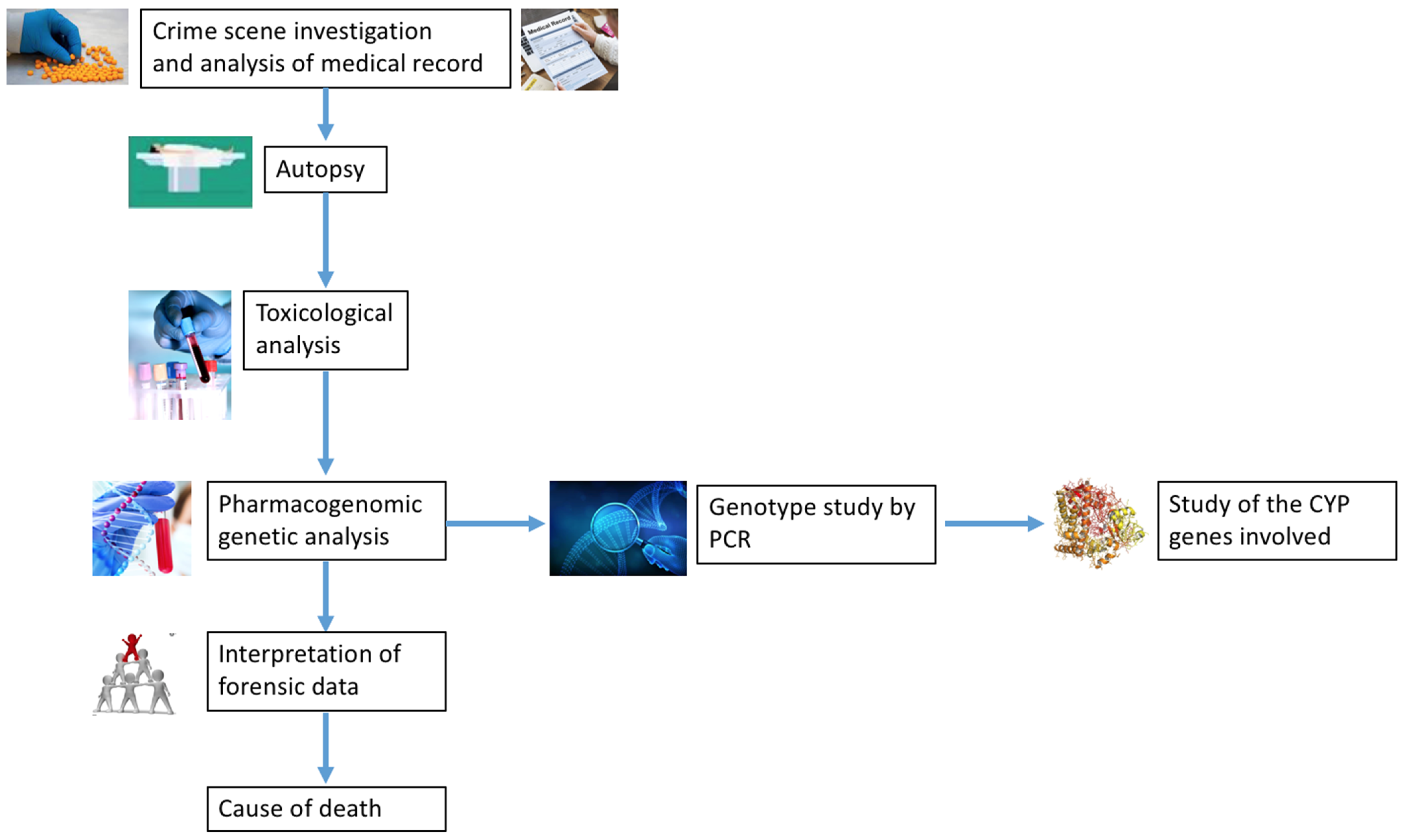
1. Introduction
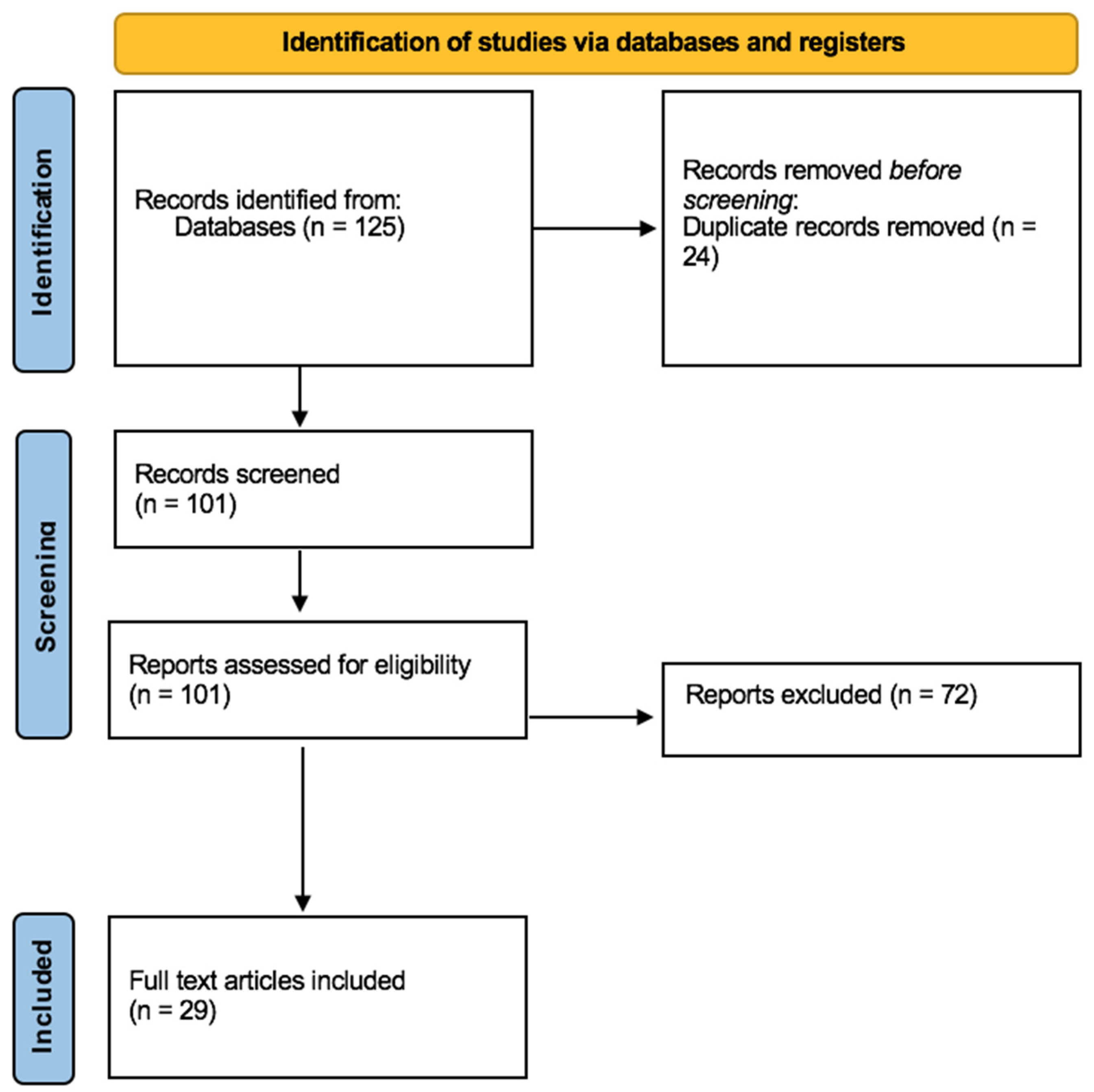
2. Materials and Methods
2.1. Inclusion and Exclusion Criteria
2.2. Quality Assessment and Data Extraction
2.3. Characteristics of Eligible Studies
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferrara, M.; Sessa, F.; Rendine, M.; Spagnolo, L.; De Simone, S.; Riezzo, I.; Maglietta, F. A multidisciplinary approach is mandatory to solve complex crimes: A case report. Egypt. J. Forensic Sci. 2019, 9, 1–7. [Google Scholar] [CrossRef]
- Ledda, C.; Pomara, C.; Bracci, M.; Mangano, D.; Ricceri, V.; Musumeci, A.; Rapisarda, V. Natural carcinogenic fiber and pleural plaques assessment in a general population: A cross-sectional study. Environ. Res. 2016, 150, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Spagnolo, L.; Bertozzi, G.; Ferrara, M.; Fiore, C.; Sessa, F. From crime scene investigation to the identification of the cause of death: A muscle-popping case report. Rom. J. Leg Med. 2018, 26, 340–343. [Google Scholar]
- Filetti, V.; Di Mizio, G.; Rendine, M.; Fortarezza, P.; Ricci, P.; Pomara, C.; Sessa, F. Volatile organic compounds: Instrumental and canine detections link an individual to the crime scene. Egypt. J. Forensic Sci. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Montana, A.; Rapisarda, V.; Esposito, M.; Amico, F.; Cocimano, G.; Nunno, N.D.; Ledda, C.; Salerno, M. A Rare Case of Suicide by Ingestion of Phorate: A Case Report and a Review of the Literature. Healthcare 2021, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Maiellaro, A.; Perna, A.; Giugliano, P.; Esposito, M.; Vacchiano, G. Sudden Death from Primary Cerebral Melanoma: Clinical Signs and Pathological Observations. Healthcare 2021, 9, 341. [Google Scholar] [CrossRef] [PubMed]
- Pomara, C.; Fiore, C.; D’Errico, S.; Riezzo, I.; Fineschi, V. Calcium oxalate crystals in acute ethylene glycol poisoning: A confocal laser scanning microscope study in a fatal case. Clin. Toxicol. 2008, 46, 322–324. [Google Scholar] [CrossRef]
- Orban, G.; Bombardi, C.; Marino Gammazza, A.; Colangeli, R.; Pierucci, M.; Pomara, C.; Pessia, M.; Bucchieri, F.; Benigno, A.; Smolders, I.; et al. Role(s) of the 5-HT2C receptor in the development of maximal dentate activation in the hippocampus of anesthetized rats. CNS Neurosci. Ther. 2014, 20, 651–661. [Google Scholar] [CrossRef]
- Sessa, F.; Franco, S.; Picciocchi, E.; Geraci, D.; Chisari, M.G.; Marsala, G.; Salerno, M. Addictions substance free during lifespan. Acta Med. Mediterr. 2018, 34, 2081–2087. [Google Scholar] [CrossRef]
- Rapisarda, V.; Loreto, C.; Castorina, S.; Romano, G.; Garozzo, S.F.; Musumeci, A.; Migliore, M.; Avola, R.; Cinà, D.; Pomara, C.; et al. Occupational exposure to fluoro-edenite and prevalence of anti-nuclear autoantibodies. Future Oncol. 2018, 14, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Ledda, C.; Iavicoli, I.; Bracci, M.; Avola, R.; Senia, P.; Santarelli, L.; Pomara, C.; Rapisarda, V. Serum lipid, lipoprotein and apolipoprotein profiles in workers exposed to low arsenic levels: Lipid profiles and occupational arsenic exposure. Toxicol Lett. 2018, 282, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Fineschi, V.; Neri, M.; Di Donato, S.; Pomara, C.; Riezzo, I.; Turillazzi, E. An immunohistochemical study in a fatality due to ovarian hyperstimulation syndrome. Int. J. Leg. Med. 2006, 120, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Montana, A.; Liberto, A.; Filetti, V.; Nunno, N.D.; Amico, F.; Salerno, M.; Loreto, C.; Sessa, F. Anaphylactic Death: A New Forensic Workflow for Diagnosis. Healthcare 2021, 9, 117. [Google Scholar] [CrossRef] [PubMed]
- Monda, V.; Salerno, M.; Sessa, F.; Bernardini, R.; Valenzano, A.; Marsala, G.; Zammit, C.; Avola, R.; Carotenuto, M.; Messina, G.; et al. Functional Changes of Orexinergic Reaction to Psychoactive Substances. Mol. Neurobiol. 2018, 55, 6362–6368. [Google Scholar] [CrossRef] [PubMed]
- Sessa, F.; Salerno, M.; Pomara, C. Autopsy Tool in Unknown Diseases: The Experience with Coronaviruses (SARS-CoV, MERS-CoV, SARS-CoV-2). Medicina 2021, 57, 309. [Google Scholar] [CrossRef] [PubMed]
- Spear, B.B.; Heath-Chiozzi, M.; Huff, J. Clinical application of pharmacogenetics. Trends Mol. Med. 2001, 7, 201–204. [Google Scholar] [CrossRef]
- Pirmohamed, M. Pharmacogenetics: Past, present and future. Drug Discov. Today 2011, 16, 852–861. [Google Scholar] [CrossRef]
- Roses, A.D. Pharmacogenetics and the practice of medicine. Nature 2000, 405, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Wolf, C.R.; Smith, G.; Smith, R.L. Pharmacogenetics. BMJ 2000, 320, 987–990. [Google Scholar] [CrossRef]
- Kupiec, T.C.; Raj, V.; Vu, N. Pharmacogenomics for the forensic toxicologist. J. Anal. Toxicol. 2006, 30, 65–72. [Google Scholar] [CrossRef]
- Lötsch, J.; Skarke, C.; Liefhold, J.; Geisslinger, G. Genetic predictors of the clinical response to opioid analgesics: Clinical utility and future perspectives. Clin. Pharmacokinet. 2004, 43, 983–1013. [Google Scholar] [CrossRef]
- Tamminga, W.J.; Wemer, J.; Oosterhuis, B.; De Zeeuw, R.A.; De Leij, L.F.; Jonkman, J.H. The prevalence of CYP2D6 and CYP2C19 genotypes in a population of healthy Dutch volunteers. Eur. J. Clin. Pharmacol. 2001, 57, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Poolsup, N.; Li Wan Po, A.; Knight, T.L. Pharmacogenetics and psychopharmacotherapy. J. Clin. Pharm. Ther. 2000, 25, 197–220. [Google Scholar] [CrossRef] [PubMed]
- Druid, H.; Holmgren, P. A compilation of fatal and control concentrations of drugs in postmortem femoral blood. J. Forensic Sci. 1997, 42, 79–87. [Google Scholar] [CrossRef]
- Koski, A.; Ojanperä, I.; Sistonen, J.; Vuori, E.; Sajantila, A. A fatal doxepin poisoning associated with a defective CYP2D6 genotype. Am. J. Forensic Med. Pathol. 2007, 28, 259–261. [Google Scholar] [CrossRef]
- Chiurillo, M.A.; Grimán, P.; Morán, Y.; Camargo, M.E.; Ramírez, J.L. Analysis of CYP2D6 gene variation in Venezuelan population: Implications for forensic toxicology. Forensic Sci. Int. Genet. Suppl. Ser. 2009, 2, 483–484. [Google Scholar] [CrossRef]
- Shen, M.; Shi, Y.; Xiang, P. CYP3A4 and CYP2C19 genetic polymorphisms and zolpidem metabolism in the Chinese Han population: A pilot study. Forensic Sci. Int. 2013, 227, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Woodall, K.L.; Solbeck, P.; Ross, C.J.; Carleton, B.C.; Hayden, M.R.; Koren, G.; Madadi, P. Codeine-related deaths: The role of pharmacogenetics and drug interactions. Forensic Sci. Int. 2014, 239, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Ciszkowski, C.; Madadi, P.; Phillips, M.S.; Lauwers, A.E.; Koren, G. Codeine, ultrarapid-metabolism genotype, and postoperative death. New Engl. J. Med. 2009, 361, 827–828. [Google Scholar] [CrossRef] [PubMed]
- Høiseth, G.; Majid, U.; Mørland, J.; Bramness, J.G.; Molden, E. CYP2C19 genetics in fatal carisoprodol intoxications. Eur. J. Clin. Pharmacol. 2012, 68, 1561–1565. [Google Scholar] [CrossRef] [PubMed]
- Thieme, D.; Rolf, B.; Sachs, H.; Schmid, D. Correlation of inter-individual variations of amitriptyline metabolism examined in hairs with CYP2C19 and CYP2D6 polymorphisms. Int. J. Legal. Med. 2008, 122, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Koski, A.; Sistonen, J.; Ojanperä, I.; Gergov, M.; Vuori, E.; Sajantila, A. CYP2D6 and CYP2C19 genotypes and amitriptyline metabolite ratios in a series of medicolegal autopsies. Forensic Sci. Int. 2006, 10, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Tawfik Khattab, A.M.; Zayed, A.A.; Ahmed, A.; AbdelAal, A.G.; Mekdad, A.A. The role of PON1 and CYP2D6 genes in susceptibility to organophosphorus chronic intoxication in Egyptian patients. Neurotoxicology 2016, 53, 102–107. [Google Scholar] [CrossRef]
- Neukamm, M.A.; Vogt, S.; Hermanns-Clausen, M.; Naue, J.; Thierauf, A.; Auwärter, V. Fatal doxepin intoxication—suicide or slow gradual intoxication? Forensic Sci. Int. 2013, 227, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Madadi, P.; Hildebrandt, D.; Gong, I.Y.; Schwarz, U.I.; Ciszkowski, C.; Ross, C.J.; Sistonen, J.; Carleton, B.C.; Hayden, M.R.; Lauwers, A.E.; et al. Fatal hydrocodone overdose in a child: Pharmacogenetics and drug interactions. Pediatrics 2010, 126, 986–989. [Google Scholar] [CrossRef]
- Launiainen, T.; Rasanen, I.; Vuori, E.; Ojanperä, I. Fatal venlafaxine poisonings are associated with a high prevalence of drug interactions. Int. J. Leg. Med. 2011, 125, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Riccardi, L.N.; Lanzellotto, R.; Falconi, M.; Ceccardi, S.; Bini, C.; Pelotti, S. Development of a tetraplex PCR assay for CYP2D6 genotyping in degraded DNA samples. J. Forensic Sci. 2014, 59, 690–695. [Google Scholar] [CrossRef]
- Jakobsson, G.; Larsson, R.; Pellè, L.; Kronstrand, R.; Gréen, H. Oxycodone findings and CYP2D6 function in postmortem cases. Forensic Sci. Int. Genet. 2021, 53, 102510. [Google Scholar] [CrossRef]
- Vevelstad, M.; Øiestad, E.L.; Bremer, S.; Bogen, I.L.; Zackrisson, A.L.; Arnestad, M. Is toxicity of PMMA (paramethoxymethamphetamine) associated with cytochrome P450 pharmacogenetics? Forensic Sci. Int. 2016, 261, 137–147. [Google Scholar] [CrossRef]
- Wu, A.H.; Kearney, T. Lack of impairment due to confirmed codeine use prior to a motor vehicle accident: Role of pharmacogenomics. J. Forensic Leg. Med. 2013, 20, 1024–1027. [Google Scholar] [CrossRef]
- Bastami, S.; Haage, P.; Kronstrand, R.; Kugelberg, F.C.; Zackrisson, A.L.; Uppugunduri, S. Pharmacogenetic aspects of tramadol pharmacokinetics and pharmacodynamics after a single oral dose. Forensic Sci. Int. 2014, 238, 125–132. [Google Scholar] [CrossRef]
- Levo, A.; Koski, A.; Ojanperä, I.; Vuori, E.; Sajantila, A. Post-mortem SNP analysis of CYP2D6 gene reveals correlation between genotype and opioid drug (tramadol) metabolite ratios in blood. Forensic Sci. Int. 2003, 135, 9–15. [Google Scholar] [CrossRef]
- Koren, G.; Cairns, J.; Chitayat, D.; Gaedigk, A.; Leeder, S.J. Pharmacogenetics of morphine poisoning in a breastfed neonate of a codeine-prescribed mother. Lancet 2006, 368, 704. [Google Scholar] [CrossRef]
- Piatkov, I.; Jones, T.; Van Vuuren, R.J. Suicide cases and venlafaxine. Acta Neuropsychiatr 2011, 23, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Rahikainen, A.L.; Palo, J.U.; Haukka, J.; Sajantila, A. Post-mortem analysis of suicide victims shows ABCB1 haplotype 1236T-2677T-3435T as a candidate predisposing factor behind adverse drug reactions in females. Pharm. Genom. 2018, 28, 99–106. [Google Scholar] [CrossRef]
- Drevin, G.; Picard, N.; Jousset, N.; Briet, M.; Abbara, C. Pitfalls and challenges associated with phenoconversion in forensic toxcicology. Forensic Sci. Int. Genet. 2021, 51, 102433. [Google Scholar] [CrossRef] [PubMed]
- Neuvonen, A.M.; Palo, J.U.; Sajantila, A. Post-mortem ABCB1 genotyping reveals an elevated toxicity for female digoxin users. Int. J. Legal Med. 2011, 125, 265–269. [Google Scholar] [CrossRef]
- Fonseca, S.; Amorim, A.; Costa, H.A.; Franco, J.; Porto, M.J.; Santos, J.C.; Dias, M. Sequencing CYP2D6 for the detection of poor-metabolizers in post-mortem blood samples with tramadol. Forensic Sci. Int. 2016, 265, 153–159. [Google Scholar] [CrossRef][Green Version]
- Buchard, A.; Linnet, K.; Johansen, S.S.; Munkholm, J.; Fregerslev, M.; Morling, N. Postmortem blood concentrations of R- and S-enantiomers of methadone and EDDP in drug users: Influence of co-medication and p-glycoprotein genotype. J. Forensic Sci. 2010, 55, 457–463. [Google Scholar] [CrossRef]
- Karlsson, L.; Zackrisson, A.L.; Josefsson, M.; Carlsson, B.; Green, H.; Kugelberg, F.C. Influence of CYP2D6 and CYP2C19 genotypes on venlafaxine metabolic ratios and stereoselective metabolism in forensic autopsy cases. Pharm. J. 2015, 15, 165–171. [Google Scholar] [CrossRef]
- Andresen, H.; Augustin, C.; Streichert, T. Toxicogenetics—Cytochrome P450 microarray analysis in forensic cases focusing on morphine/codeine and diazepam. Int. J. Leg. Med. 2013, 127, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Jannetto, P.J.; Wong, S.H.; Gock, S.B.; Laleli-Sahin, E.; Schur, B.C.; Jentzen, J.M. Pharmacogenomics as molecular autopsy for postmortem forensic toxicology: Genotyping cytochrome P450 2D6 for oxycodone cases. J. Anal. Toxicol. 2002, 26, 438–447. [Google Scholar] [CrossRef]
- Mamdani, F.; Berlim, M.T.; Beaulieu, M.M.; Labbe, A.; Merette, C.; Turecki, G. Gene expression biomarkers of response to citalopram treatment in major depressive disorder. Transl. Psychiatry 2011, 1, 13. [Google Scholar] [CrossRef] [PubMed]
- Skadrić, I.; Stojković, O. Defining screening panel of functional variants of CYP1A1, CYP2C9, CYP2C19, CYP2D6, and CYP3A4 genes in Serbian population. Int. J. Leg. Med. 2020, 134, 433–439. [Google Scholar] [CrossRef]
- Turillazzi, E.; La Rocca, G.; Anzalone, R.; Corrao, S.; Neri, M.; Pomara, C.; Riezzo, I.; Karch, S.B.; Fineschi, V. Heterozygous nonsense SCN5A mutation W822X explains a simultaneous sudden infant death syndrome. Virchows Arch. 2008, 453, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Salerno, M.; Villano, I.; Nicolosi, D.; Longhitano, L.; Loreto, C.; Lovino, A.; Sessa, F.; Polito, A.N.; Monda, V.; Chieffi, S.; et al. Modafinil and orexin system: Interactions and medico-legal considerations. Front. Biosci. 2019, 24, 564–575. [Google Scholar] [CrossRef]
- Pitsiladis, Y.P.; Tanaka, M.; Eynon, N.; Bouchard, C.; North, K.N.; Williams, A.G.; Collins, M.; Moran, C.N.; Britton, S.L.; Fuku, N.; et al. Athlome Project Consortium. Athlome Project Consortium: A concerted effort to discover genomic and other "omic" markers of athletic performance. Physiol. Genomics 2016, 48, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Ingelman-Sundberg, M.; Sim, S.C.; Gomez, A.; Rodriguez-Antona, C. Influence of cytochrome P450 polymorphisms on drug therapies: Pharmacogenetic, pharmacoepigenetic and clinical aspects. Pharmacol. Ther. 2007, 116, 496–526. [Google Scholar] [CrossRef] [PubMed]
- Boyle, J.; Stock, C.J. CYP2D6 Expression in Veterans Experiencing Opioid Overdose: A Postmortem Review. Pharmgenomics Pers. Med. 2020, 13, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Snider, N.T.; Sikora, M.J.; Sridar, C.; Feuerstein, T.J.; Rae, J.M.; Hollenberg, P.F. The endocannabinoid anandamide is a substrate for the human polymorphic cytochrome P450 2D6. J. Pharmacol. Exp. Ther. 2008, 327, 538–545. [Google Scholar] [CrossRef]
- Sridar, C.; Snider, N.T.; Hollenberg, P.F. Anandamide oxidation by wild-type and polymorphically expressed CYP2B6 and CYP2D6. Drug Metab. Dispos. 2011, 39, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Sessa, F.; Salerno, M.; Di Mizio, G.; Bertozzi, G.; Messina, G.; Tomaiuolo, B.; Pisanelli, D.; Maglietta, F.; Ricci, P.; Pomara, C. Anabolic Androgenic Steroids: Searching New Molecular Biomarkers. Front. Pharmacol. 2018, 9, 1321. [Google Scholar] [CrossRef] [PubMed]
- Albano, G.D.; Sessa, F.; Messina, A.; Monda, V.; Bertozzi, G.; Maglietta, F.; Giugliano, P.; Vacchiano, G.; Gabriella, M.; Salerno, M. AAS and organs damage: A focus on Nandrolone effects. Acta Med. Mediterr. 2017, 6, 939–946. [Google Scholar]
- Lucas, D.; Goulitquer, S.; Marienhagen, J.; Fer, M.; Dreano, Y.; Schwaneberg, U.; Amet, Y.; Corcos, L. Stereoselective epoxidation of the last double bond of polyunsaturated fatty acids by human cytochromes P450. J. Lipid Res. 2010, 51, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Honda, A.; Miyazaki, T.; Ikegami, T.; Iwamoto, J.; Maeda, T.; Hirayama, T.; Saito, Y.; Teramoto, T.; Matsuzaki, Y. Cholesterol 25-hydroxylation activity of CYP3A. J. Lipid Res. 2011, 52, 1509–1516. [Google Scholar] [CrossRef]
- Chen, H.; Howald, W.N.; Juchau, M.R. Biosynthesis of all-trans-retinoic acid from all-trans-retinol: Catalysis of all-trans-retinol oxidation by human P-450 cytochromes. Drug Metab. Dispos. 2000, 28, 315–322. [Google Scholar]
- Wendt, F.R.; Sajantila, A.; Moura-Neto, R.S.; Woerner, A.E.; Budowle, B. Full-gene haplotypes refine CYP2D6 metabolizer phenotype inferences. Int. J. Leg. Med. 2018, 132, 1007–1024. [Google Scholar] [CrossRef]
- Nebert, D.W. Polymorphisms in drug-metabolizing enzymes: What is their clinical relevance and why do they exist? Am. J. Hum. Genet. 1997, 60, 265–271. [Google Scholar] [PubMed]
- Ingelman-Sundberg, M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): Clinical consequences, evolutionary aspects and functional diversity. Pharm. J. 2005, 5, 6–13. [Google Scholar] [CrossRef]
- Mercadante, S. Opioid metabolism and clinical aspects. Eur. J. Pharmacol. 2015, 769, 71–78. [Google Scholar] [CrossRef]
- Siegle, I.; Fritz, P.; Eckhardt, K.; Zanger, U.M.; Eichelbaum, M. Cellular localization and regional distribution of CYP2D6 mRNA and protein expression in human brain. Pharmacogenetics 2001, 11, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Cocimano, G.; Ministrieri, F.; Rosi, G.L.; Nunno, N.D.; Messina, G.; Sessa, F.; Salerno, M. Smart drugs and neuroenhancement: What do we know? Front. Biosci. 2021, 26, 347–359. [Google Scholar] [CrossRef]
- Bertozzi, G.; Sessa, F.; Albano, G.D.; Sani, G.; Maglietta, F.; Roshan, M.H.K.; Volti, G.L.; Bernardini, R.; Avola, R.; Pomara, C.; et al. The Role of Anabolic Androgenic Steroids in Disruption of the Physiological Function in Discrete Areas of the Central Nervous System. Mol. Neurobiol. 2018, 55, 5548–5556. [Google Scholar] [CrossRef] [PubMed]
- Kornhuber, J.; Bleich, S. Actions of psychotropic drugs beyond their primary targets at the synaptic cleft. Eur. Arch. Psychiatry Clin. Neurosci. 2006, 256, 265–267. [Google Scholar] [CrossRef]
- Maeda, K.; Sugiyama, Y. Impact of genetic polymorphisms of transporters on the pharmacokinetic, pharmacodynamic and toxicological properties of anionic drugs. Drug Metab. Pharm. 2008, 23, 223–235. [Google Scholar] [CrossRef]
- Esposito, M.; Licciardello, G.; Privitera, F.; Iannuzzi, S.; Liberto, A.; Sessa, F.; Salerno, M. Forensic Post-Mortem Investigation in AAS Abusers: Investigative Diagnostic Protocol. A Systematic Review. Diagnostics 2021, 11, 1307. [Google Scholar] [CrossRef] [PubMed]
- Tzang, R.F.; Hong, C.J.; Liou, Y.J.; Yu, Y.W.; Chen, T.J.; Tsai, S.J. Association study of p11 gene with major depressive disorder, suicidal behaviors and treatment response. Neurosci. Lett. 2008, 447, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Budowle, B.; Van Daal, A. Extracting evidence from forensic DNA analyses: Future molecular biology directions. Biotechniques 2009, 46, 342–350. [Google Scholar] [CrossRef]
- Turillazzi, E.; Baroldi, G.; Silver, M.D.; Parolini, M.; Pomara, C.; Fineschi, V. A systematic study of a myocardial lesion: Colliquative myocytolysis. Int. J. Cardiol. 2005, 104, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, Y.P.; Rennert, H. Pharmacogenomics and the future of toxicology testing. Clin. Lab. Med. 2012, 32, 509–523. [Google Scholar] [CrossRef]
- Wyman, J.F. Principles and procedures in forensic toxicology. Clin. Lab. Med. 2012, 32, 493–507. [Google Scholar] [CrossRef] [PubMed]
- Sajantila, A.; Palo, J.U.; Ojanperä, I.; Davis, C.; Budowle, B. Pharmacogenetics in medico-legal context. Forensic Sci. Int. 2010, 203, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Sessa, F.; Esposito, M.; Messina, G.; Di Mizio, G.; Di Nunno, N.; Salerno, M. Sudden Death in Adults: A Practical Flow Chart for Pathologist Guidance. Healthcare 2021, 9, 870. [Google Scholar] [CrossRef] [PubMed]
- Musshoff, F.; Stamer, U.M.; Madea, B. Pharmacogenetics and forensic toxicology. Forensic Sci. Int. 2010, 203, 53–62. [Google Scholar] [CrossRef]
- Salerno, M.; Cascio, O.; Bertozzi, G.; Sessa, F.; Messina, A.; Monda, V.; Cipolloni, L.; Biondi, A.; Daniele, A.; Pomara, C. Anabolic androgenic steroids and carcinogenicity focusing on Leydig cell: A literature review. Oncotarget 2018, 9, 19415–19426. [Google Scholar] [CrossRef] [PubMed]
- Bafunno, V.; Bury, L.; Tiscia, G.L.; Fierro, T.; Favuzzi, G.; Caliandro, R.; Sessa, F.; Grandone, E.; Margaglione, M.; Gresele, P. A novel congenital dysprothrombinemia leading to defective prothrombin maturation. Thromb. Res. 2014, 134, 1135–1141. [Google Scholar] [CrossRef]
- Caenazzo, L.; Tozzo, P. The Future of Biobanking: What Is Next? BioTech 2020, 9, 23. [Google Scholar] [CrossRef]
- Rebsamen, M.C.; Desmeules, J.; Daali, Y.; Chiappe, A.; Diemand, A.; Rey, C.; Chabert, J.; Dayer, P.; Hochstrasser, D.; Rossier, M.F. The AmpliChip CYP450 test: Cytochrome P450 2D6 genotype assessment and phenotype prediction. Pharm. J. 2009, 9, 34–41. [Google Scholar] [CrossRef]
- Shimizu, M.; Sawaya, R.; Kishimoto, I.; Yamazaki, H. Genotyping of wild-type cytochrome P450 2A6 and whole-gene deletion using human blood samples and a multiplex real-time polymerase chain reaction method with dual-labeled probes. Clin. Chim. Acta 2015, 441, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Stamer, U.M.; Bayerer, B.; Wolf, S.; Hoeft, A. Rapid and reliable method for cytochrome P450 2D6 genotyping. Clin. Chem. 2002, 48, 1412–1417. [Google Scholar] [CrossRef]
- Muller, B.; Zopf, K.; Bachofer, J.; Steimer, W. Optimized strategy for rapid cytochrome P450 2D6 genotyping by real-time long PCR. Clin. Chem. 2003, 49, 624–1631. [Google Scholar] [CrossRef][Green Version]
- Alaeddini, R. Forensic implications of PCR inhibition—A review. Scie Int. Genet. 2012, 6, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Zubakov, D.; Boersma, A.W.; Choi, Y.; van Kuijk, P.F.; Wiemer, E.A.; Kayser, M. MicroRNA markers for forensic body fluid identification obtained from microarray screening and quantitative RT-PCR confirmation. Int. J. Leg. Med. 2010, 124, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Qiao, W.; Yang, Y.; Sebra, R.; Mendiratta, G.; Gaedigk, A.; Desnick, R.J.; Scott, S.A. Long-read single molecule real-time full gene sequencing of cytochrome P450-2D6. Hum. Mutat. 2016, 37, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.L.; Chung, Y.K.; Tan, H.C. Cost-effective real-time reverse transcriptase PCR (RT-PCR) to screen for Dengue virus followed by rapid single-tube multiplex RT-PCR for serotyping of the virus. J. Clin. Microbiol. 2007, 45, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Rouet, F.; Ekouevi, D.K.; Chaix, M.L.; Burgard, M.; Inwoley, A.; Tony, T.D.A.; Danel, C.; Anglaret, X.; Leroy, V.; Msellati, P.; et al. Transfer and evaluation of an automated, low-cost real-time reverse transcription-PCR test for diagnosis and monitoring of human immunodeficiency virus type 1 infection in a West African resource-limited setting. J. Clin. Microbiol. 2005, 43, 2709–2717. [Google Scholar] [CrossRef] [PubMed]
- Pomara, C.; Salerno, M.; Sessa, F.; Esposito, M.; Barchitta, M.; Ledda, C.; Grassi, P.; Liberto, A.; Mattaliano, A.R.; Rapisarda, V.; et al. Safe Management Strategies in Clinical Forensic Autopsies of Confirmed COVID-19 Cases. Diagnostics 2021, 11, 457. [Google Scholar] [CrossRef] [PubMed]
- Musso, N.; Falzone, L.; Stracquadanio, S.; Bongiorno, D.; Salerno, M.; Esposito, M.; Sessa, F.; Libra, M.; Stefani, S.; Pomara, C. Post-Mortem Detection of SARS-CoV-2 RNA in Long-Buried Lung Samples. Diagnostics 2021, 11, 1158. [Google Scholar] [CrossRef] [PubMed]
- Pomara, C.; Sessa, F.; Galante, D.; Pace, L.; Fasanella, A.; Di Nunno, N.; Esposito, M.; Salerno, M. Do We Really Need Hazard Prevention at the Expense of Safeguarding Death Dignity in COVID-19? Diagnostics 2021, 11, 1913. [Google Scholar] [CrossRef]
- Pomara, C.; Sessa, F.; Ciaccio, M.; Dieli, F.; Esposito, M.; Giammanco, G.M.; Garozzo, S.F.; Giarratano, A.; Prati, D.; Rappa, F.; et al. COVID-19 Vaccine and Death: Causality Algorithm According to the WHO Eligibility Diagnosis. Diagnostics 2021, 11, 955. [Google Scholar] [CrossRef] [PubMed]
- Warner, M.; Trinidad, J.P.; Bastian, B.A.; Miniño, A.M.; Hedegaard, H. Drugs most frequently involved in drug overdose deaths: United States. Natl. Vital Stat. Rep. 2016, 65, 1–15. [Google Scholar] [PubMed]
- Hedegaard, H.; Bastian, B.A.; Trinidad, J.P.; Spencer, M.; Warner, M. Drugs most frequently involved in drug overdose deaths: United States. Natl. Vital Stat. Rep. 2018, 67, 1–14. [Google Scholar] [PubMed]
- Hedegaard, H.; Miniño, A.M.; Warner, M. Urban–rural differences in drug overdose death rates, by sex, age, and type of drugs onvolved. Natl. Vital Stat. Rep. 2019, 345, 1–8. [Google Scholar]
- Jones, C.M.; Mack, K.A.; Paulozzi, L.J. Pharmaceutical overdose deaths, united states, 2010. JAMA 2013, 309, 657–659. [Google Scholar] [CrossRef] [PubMed]
- Caenazzo, L.; Tozzo, P.; Dierickx, K. Incidental findings in forensics: Are we sure that it is a question easy to deal with? Int. J. Leg. Med. 2021, 135, 591–592. [Google Scholar] [CrossRef]
- Calcaterra, S.; Glanz, J.; Binswanger, I.A. National trends in pharmaceutical opioid related overdose deaths compared to other substance related overdose deaths: 1999–2009. Drug Alcohol. Depend. 2013, 131, 263–270. [Google Scholar] [CrossRef]
- Cryer, B. NSAID-associated deaths: The rise and fall of NSAID-associated GI mortality. Am. J. Gastroenterol. 2005, 100, 1694–1695. [Google Scholar] [CrossRef] [PubMed]
- Goldkind, L.; Laine, L. A systematic review of NSAIDs withdrawn from the market due to hepatotoxicity: Lessons learned from the bromfenac experience. Pharmacoepidemiol. Drug Saf. 2006, 15, 213–220. [Google Scholar] [CrossRef]
- Bafunno, V.; Santacroce, R.; Chetta, M.; D’Andrea, G.; Pisanelli, D.; Sessa, F.; Trotta, T.; Tagariello, G.; Peyvandi, F.; Margaglione, M. Polymorphisms in genes involved in autoimmune disease and the risk of FVIII inhibitor development in Italian patients with haemophilia A. Haemophilia 2010, 16, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Santacroce, R.; Santoro, R.; Sessa, F.; Iannaccaro, P.; Sarno, M.; Longo, V.; Gallone, A.; Vecchione, G.; Muleo, G.; Margaglione, M. Screening of mutations of hemophilia A in 40 Italian patients: A novel G-to-A mutation in intron 10 of the F8 gene as a putative cause of mild hemophilia A in southern Italy. Blood Coagul. Fibrinolysis. 2008, 19, 197–202. [Google Scholar] [CrossRef]
- Patanè, F.G.; Esposito, M.; Musumeci, A.G.; Palermo, M.; Torrisi, M.; Salerno, M.; Montana, A. Multidisciplinary Approach to Suspected Sudden Death Caused by Arteriovenous Malformation Rupture: A Case Report. Medicina 2021, 57, 644. [Google Scholar] [CrossRef]
- Bonn, D. Adverse drug reactions remain a major cause of death. Lancet 1998, 351, 1183. [Google Scholar] [CrossRef]
- Sessa, F.; Polito, R.; Monda, V.; Scarinci, A.; Salerno, M.; Carotenuto, M.; Cibelli, G.; Valenzano, A.; Campanozzi, A.; Mollica, M.P.; et al. Effects of a Plastic-Free Lifestyle on Urinary Bisphenol A Levels in School-Aged Children of Southern Italy: A Pilot Study. Front. Public Health 2021, 9, 626070. [Google Scholar] [CrossRef] [PubMed]
- Bafunno, V.; Santacroce, R.; Chetta, M.; Peyvandi, F.; Sessa, F.; Chinni, E.; Longo, V.; Margaglione, M. Polymorphic miRNA-mediated gene contribution to inhibitor development in haemophilia A. Haemophilia 2012, 18, 1003–1007. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, K.; Argo, A.; Borroni, C.; Catalano, D.; Dell’acqua, L.; Farè, F.; Procaccianti, P.; Roda, G.; Gambaro, V. Development and validation of a reliable method for studying the distribution pattern for opiates metabolites in brain. J. Pharm. Biomed. Anal. 2013, 73, 125–130. [Google Scholar] [CrossRef] [PubMed]
| Drugs of Toxicological Interest and Their Metabolizing Enzymes [22,23,24] | |
|---|---|
| Gene Involved in Drug Metabolization | Drugs Metabolized |
| CYP2C19 | Amitriptylineim, Ipramine, Diazepam, Citalopram, Carisoprodol, several Anticonvulsants |
| CYP2D6 | Amphetamines, Codeine, Oxycodone, Hydrocodone, Methadone, Tramadol, some Antipsychotics, some Tricyclic Antidepressants, Steroids |
| CYP3A4 | Benzodiazepines, Fentanyl, Methadone, Buprenorphine, Hypnotics, some Antipsychotics, some Tricyclic Antidepressants |
| CYP2E1 | Ethanol, some Anaesthetics, Paracetamol, Verapamil |
| Reference | Study Design | Number of Cases | Type of Drugs | Type of Sample | Toxicological Method | Genotypic Method | Gene Study | Main Findings about the Genetic Investigation in Order to Define the Cause of Death (* Indicates the Haplotype) |
|---|---|---|---|---|---|---|---|---|
| Kupiec T.C. [20] | Case report | Death of a 30-year-old male | Methadone and benzodiazepines | Blood | LC–MS | Quantitative real-time PCR | CYP3A4 (benzodiazepines)- CYP2D6 (Methadone) | The subject had a CYP2D6 polymorphism that increased its toxicity combined with benzodiazepines interaction resulting in death. |
| Koski A. [25] | Original article | 11 deceased people | Doxepin (tricyclic antidepressants) | Blood | GC–MS | Quantitative real-time PCR | CYP2D6 | In cases of indeterminate death after drug administration, cytochrome genotyping is a very useful method for establishing the cause of death. Completely non-functional CYP2D6 cytochromes (* 3/* 4) with poor drug metabolism were found in the present study. |
| Chiurillo M.A [26] | Original article | 110 healthy individuals | EDTA | Blood | GC–MS | PCR tetra-primer | CYP2D6 | Analysis of CYP2D6 * 3, * 4, * 5, * 6, and 10 * alleles of 110 Velezuelan, unrelated healthy individuals. Allele frequencies obtained for * 4 (15.9%) and * 10 (16.4%) had a narrow therapeutic window and showed increased toxicity. |
| Shen M. [27] | Original article | 300 healthy individuals | Zolpidem (sedative and hypnotic drug) | Blood | UPLC–MS/MS | PCR | CYP2D6, CYP2C19, CYP2C9, CYP3A4, CYP1A2 | The *18 CYP3A4 and *2 CYP2C19 alleles are associated with poor Zolpidem metabolism with increased toxicity. Genetic factors play a crucial role in drug metabolism with implications in forensic toxicology. |
| Lam J. [28] | Original article | 68 deceased people | Codeine | Blood | GC–MS | PCR- QiaSymphony DNA purification system | CYP2D6 | Individuals carrying the ABCB1 1236T variant of CYP2D6 had higher codeine concentrations than the general population with increased toxicity. |
| Ciszkowski C. [29] | Case report | Death of a healthy 2-year-old boy | Codeine, Acetaminophen | Blood | GC–MS | PCR | CYP2D6 | In the present case, cytochrome P-450 2D6 (CYP2D6) was genotyped and revealed the functional duplication of the CYP2D6 allele, resulting in an ultrafast metabolizer phenotype. The codeine prescribed was within the recommended range; however, the increased conversion of codeine to morphine resulted in a toxic buildup of morphine. |
| Høiseth G. [30] | Original article | 75 Healthy individuals | Carisoprodol (muscle relaxant) | Blood | GC–MS | TaqMan-based realtime PCR assays | CYP2C19 | This study found no evidence of an association between CYP2C19 genetics and the mortality risk of carisoprodol. |
| Thieme D. [31] | Original article | 23 Healthy individuals | Amitriptyline, Nortriptyline (Tricyclic Antidepressant) | Hair | LC–MS | TaqMan-based realtime PCR assays | CYP2D6, CYP2C19 | The genotypes of CYP2C19 (alleles *2, *3, and *4) and CYP2D6 (*3, *4, and *6) were examined and a substantial change in metabolites was seen from the wild-type variant. |
| Koski A. [32] | Original article | 202 deceased people | Amitriptyline (Tricyclic Antidepressant) | Blood | LC–MS | Long PCR reactions | CYP2D6, CYP2C19 | Pharmacogenetic analysis of CYP2D6 and CYP2C19 genotypes is of great help in cases of subjects who die after drug administration. |
| Tawfik Khattab A.M. [33] | Original article | 187 Healthy individuals | Organophosphorus drugs | Blood | GC–MS | Long PCR reactions | CYP2D6, CYP2C19 | In some populations, some genetic polyforms are very frequent, so it is important to consider this in pharmacogenetics. |
| Neukamm M.A. [34] | Case report | 52-year-old female | Doxepin (Tricyclic Antidepressants) | Blood | LC–MS | PCR | CYP2D6 | The death of the woman was due to a cytochrome polymorphism and an interaction with other drugs rather than an overdose. |
| Madadi P [35] | Case report | Child aged 5 years 9 months | Clarithromycin (antibiotic) and valproic acid (antiepileptic) | Blood | GC | TaqMan-based realtime PCR assays | CYP2D6 | The child had a particular genetic polymorphism of cytochrome CYP2D6 (CYP2D6 * 2A/* 41). Co-administration of clarithromycin and valproic acid prevented drug elimination and increased bioavailability. It is important to analyze pharmacogenetics, pharmacokinetics, and drug interactions in similar cases in this case. |
| Launiainen T. [36] | Original article | 123 deceased people | Venlafaxine (serotonin and norepinephrine reuptake inhibitors—SSNRIs) | Blood | GC–MS | Long PCR reactions | CYP2D6 | This study found no evidence of an association between CYP2D6 genetics and the mortality risk of venlafaxine. |
| Riccardi L.N. [37] | Original article | 32 deceased people | Central nervous system drugs | 8 post-mortem saliva, 8 blood, 5 spleen samples, and 10 buccal swabs from a population study, and 1 paraffine-embedded tissue | GC–MS | Tetraplex PCR | CYP2D6 | Excellent method for evaluating CYP2D6 and its implications in the forensic field. |
| Jakobsson G. [38] | Original article | 174 deceased people | Oxycodone (opioid) | Blood | LC–MS | Digital droplet PCR | CYP2D6 | In toxicology, not only the concentration of metabolites in the blood is important to assess drug toxicity, but also the genotyping of cytochromes. A fast or slow phenotype changes the toxicity of a drug. A pharmacogenetic study should always be performed. |
| Vevelstad M. [39] | Original article | 29 deceased people | Paramethoxymethamphetamine (PMMA) | Blood | LC–MS | Quantitative real-time PCR | CYP2D6 | In some, CYP2D6 concentrations of PMMA were higher than in others, with metabolites at a lower blood concentration. |
| Wu A.H. [40] | Case report | Young woman | Codeine | Blood | LC–MS | Quantitative real-time PCR | CYP2D6, UGT2B7, CYP3A4 | In this case report, a young woman had taken codeine and caused a car accident. The young woman initially went to jail. However, through a pharmacogenetic study, it was possible to state that she was not in a state of acute intoxication so this element provided evidence for her release. |
| Bastami S. [41] | Original article | 20 Healthy individuals | Tramadol (opioid) | Blood | HPLC | Quantitative real-time PCR | CYP2D6 | The study of the CYP2D6 genotype helps to establish with greater certainty the metabolic relationship of tramadol with its metabolite to evaluate the time of drug intake. |
| Levo A. [42] | Original article | 33 deceased people (11 males and 22 females) | Tramadol (opioid) | Blood | LC–MS | PCR | CYP2D6 | The genetic variation of drug metabolizing enzymes is substantial and can be studied in forensic toxicology. Furthermore, genetic factors play a dominant role in the metabolism of individual drugs. |
| Koren G. [43] | Case report | Male infant | Codeine and Paracetamol | Blood | GC–MS | PCR | CYP2D6 | The CYP2D6 *2A allele with CYP2D6 *2 × 2 gene duplication (ultra-rapid metaboliser) resulted in increased morphine formation from codeine, resulting in drowsiness and the death of the child. |
| Piatkov I. [44] | Original article | 10 suicide cases | Venlafaxine (selective serotonin and norepinephrine reuptake inhibitors-SSNRIs) | Blood | GC–MS | Quantitative real-time PCR | CYP2D6, CYP2C19, CYP2C9 | Venlafaxine, which caused neurotoxicity and suicide in these people, was related to the particular functional genetic polymorphisms of cytochrome P450, especially CYP2C19 *17. |
| Rahikainen A.L. [45] | Original article | 349 suicide cases | Citalopram (antidepressant of the selective serotonin reuptake inhibitor class) | Blood, | LC–MS | PCR | P-gp | Genetic variation in efflux transporter and permeability glycoprotein (P-gp) in women taking citalopram was associated with completed violent suicides and even violent suicide attempts. |
| Drevin G. [46] | Case report | 35-year-old man | Ethanol, Morphine, Antidepressants | Blood | GC–MS | Taqman real-time PCR analyses | CYP2D6, CYP2C19 | Cytochrome genotyping is a very useful method for establishing the cause of death. |
| Neuvonen A.M. [47] | Original article | 112 deceased people | Digoxin (heart failure) | Blood | HPLC | Quantitative real-time PCR | P-gp ABCB1gene | There is a link between ABCB1 polymorphisms and increased mortality, suggesting genotyping analysis prior to digoxin treatment. |
| Fonseca S. [48] | Original article | 100 deceased people | Tramadol (opioid) | Blood | GC–MS | Quantitative real-time PCR | CYP2D6 | Poor metabolizers have very low metabolic capacity and higher metabolic ratios. The metabolism of tramadol is correlated with the phenotype of the metabolizer and it is essential to know the phenotype in cases of opioid-related deaths. |
| Buchard A. [49] | Original article | 90 deceased people | Methadone (opioid) | Blood | GC–MS | Quantitative real-time PCR | P-gp MDR1gene | This study found no evidence of an association between P-glycoprotein MDR1 gene genetics and the mortality risk of methadone. |
| Karlsson L. [50] | Original article | 94 deceased people | Venlafaxine (selective serotonin and norepinephrine reuptake inhibitors-SSNRIs) | Blood | GC–MS | Quantitative real-time PCR | CYP2C19, CYP2D6 | The CYP2D6 genotype influences the O-demethylation of venlafaxine, while CYP2C19 influences the N-demethylation of venlafaxine, which affects the toxicity of this drug. |
| Andresen H. [51] | Original article | 11 deceased people | Morphine and codeine | Blood | GC–MS | Quantitative real-time PCR | CYP2D6 | Pharmacogenetic evaluation in forensic toxicology is essential, as the genetic polymorphisms of cytochromes play a key role in drug toxicity. |
| Jannetto P.J. [52] | Original article | 25 deceased people | Fentanyl | Blood | GC–MS | Quantitative real-time PCR | CYP3A4, CYP3A5 | Post-mortem data are evidence that CYP3A4 and CYP3A5 are involved in the metabolism of fentanyl. In particular, homozygous CYP3AS * 3 causes an altered metabolism of fentanyl with an increase in its concentration in the blood. Pharmacogenetics and molecular autopsy are crucial in these deaths. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Nunno, N.; Esposito, M.; Argo, A.; Salerno, M.; Sessa, F. Pharmacogenetics and Forensic Toxicology: A New Step towards a Multidisciplinary Approach. Toxics 2021, 9, 292. https://doi.org/10.3390/toxics9110292
Di Nunno N, Esposito M, Argo A, Salerno M, Sessa F. Pharmacogenetics and Forensic Toxicology: A New Step towards a Multidisciplinary Approach. Toxics. 2021; 9(11):292. https://doi.org/10.3390/toxics9110292
Chicago/Turabian StyleDi Nunno, Nunzio, Massimiliano Esposito, Antonina Argo, Monica Salerno, and Francesco Sessa. 2021. "Pharmacogenetics and Forensic Toxicology: A New Step towards a Multidisciplinary Approach" Toxics 9, no. 11: 292. https://doi.org/10.3390/toxics9110292
APA StyleDi Nunno, N., Esposito, M., Argo, A., Salerno, M., & Sessa, F. (2021). Pharmacogenetics and Forensic Toxicology: A New Step towards a Multidisciplinary Approach. Toxics, 9(11), 292. https://doi.org/10.3390/toxics9110292










