Releases of Fire-Derived Contaminants from Polymer Pipes Made of Polyvinyl Chloride
Abstract
:1. Introduction
2. Materials and Methods
2.1. Infrared Spectrocopy and Thermogravimetry Analysis of PVC Pipe
2.2. Analysis of Leachates from Burned PVC Pipes by GC-MS
2.3. Analysis of Emissions from Burning PVC Pipes by GC-MS
2.4. Analysis of Emissions from Burning PVC Pipes by Infrared Spectrometry
3. Results
3.1. Thermogravimetric Characterization of PVC Pipes
3.2. Leachate Analysis for Polymer Pipes
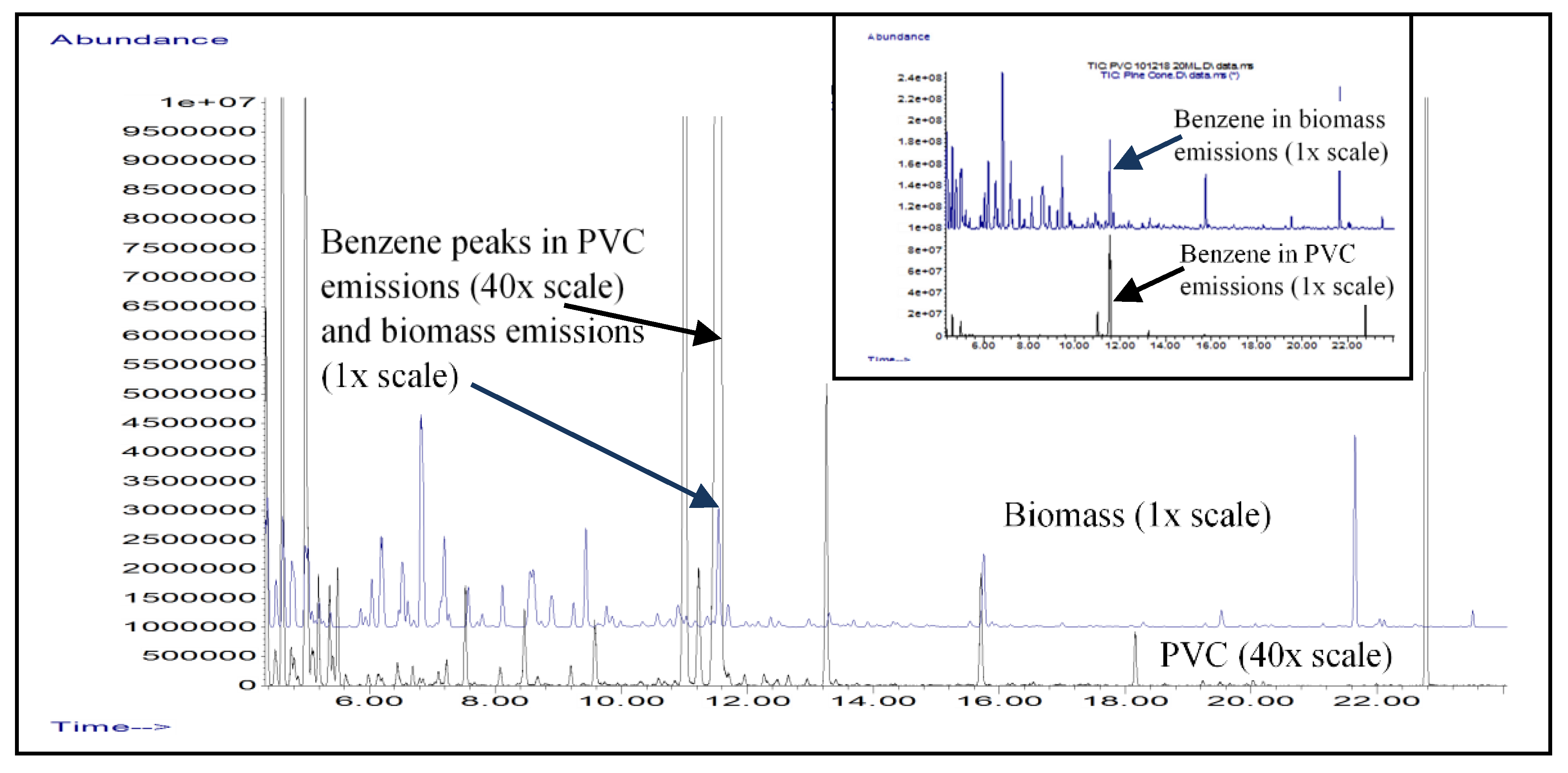
3.3. Emission Analysis of Burning Polymer Pipes
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Verma, R.; Vinoda, K.S.; Papireddy, M.; Gowda, A.N.S. Toxic Pollutants from Plastic Waste—A Review. Procedia Environ. Sci. 2016, 35, 701–708. [Google Scholar] [CrossRef]
- Valavanidis, A.; Iliopoulos, N.; Gotsis, G.; Fiotakis, K. Persistent free radicals, heavy metals and PAHs generated in particulate soot emissions and residue ash from controlled combustion of common types of plastic. J. Hazard. Mater. 2008, 156, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, D., Jr.; Quintiere, J.G. Material fire properties and predictions for thermoplastics. Fire Saf. J. 1996, 26, 241–268. [Google Scholar] [CrossRef]
- The Sacramento Bee. Rare Toxic Cocktail from Camp Fire is Poisoning Paradise Water. Available online: https://www.sacbee.com/news/local/environment/article228969259.html (accessed on 18 August 2019).
- North Bay Business Journal. Santa Rosa: Fountaingrove Water System Needs $43M Replacement after Sonoma County Wildfires. Available online: http://www.northbaybusinessjournal.com/northbay/sonomacounty/8147434-181/sonoma-santa-rosa-fountaingrove (accessed on 30 July 2019).
- American Chemical Society. California Wildfires Caused Unexpected Benzene Contamination of Drinking Water. Available online: https://cen.acs.org/environment/water/California-wildfires-caused-unexpected-benzene/96/i26 (accessed on 18 August 2019).
- Saskatchewan Ministry of Environment. Health and Environmental Effects of Burning Waste Plastics. Available online: http://www.saskh2o.ca/PDF/epb433.pdf (accessed on 18 August 2019).
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- Tabb, M.M.; Blumberg, B. New modes of action for endocrine-disrupting chemicals. Mol. Endocrinol. 2006, 20, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Wolff, M.S.; Engel, S.M.; Berkowitz, G.S.; Ye, X.; Silva, M.J.; Zhu, C.; Wetmur, J.; Calafat, A.M. Prenatal phenol and phthalate exposures and birth outcomes. Environ. Health Perspect. 2008, 116, 1092–1097. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J. Role of exposure to environmental chemicals in the developmental basis of disease and dysfunction. Reprod. Toxicol. 2007, 23, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Gollapalli, R.; Blinder, A.; Patel, M. Identification of leachable impurities in an ophthalmic drug product originating from a polymer additive Irganox 1010 using mass spectroscopy. J. Pharm. Biomed. Anal. 2018, 152, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Lagace, L.; Charron, C.; Sadiki, M. Analysis of plastic residues in maple sap and syrup collected from tubing systems sanitized with isopropyl alcohol. Heliyon 2017, 3, e00306. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry. Toxic Substances Portal-2-Butoxyethanol and 2-Butoxyethanol Acetate. Available online: https://www.atsdr.cdc.gov/phs/phs.asp?id=345&tid=61 (accessed on 18 August 2019).
- Moldovan, Z.; Marincas, O.; Povar, I.; Lupascu, T.; Longree, P.; Rota, J.S.; Singer, H.; Alder, A.C. Environmental exposure of anthropogenic micropollutants in the Prut River at the Romanian-Moldavian border: A snapshot in the lower Danube river basin. Environ. Sci. Pollut. Res. 2018, 25, 31040–31050. [Google Scholar] [CrossRef] [PubMed]
- Loschner, D.; Rapp, T.; Schlosser, F.U.; Schuster, R.; Stottmeister, E.; Zander, S. Experience with the application of the draft European Standard prEN 15768 to the identification of leachable organic substances from materials in contact with drinking water by GC-MS. Anal. Methods 2011, 3, 2547–2556. [Google Scholar] [CrossRef]
- Wiley Online Library. Toxicological Evaluation No. 114: Data for 2-Ethylhexanol. Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1002/3527600418.mb10476kske0020 (accessed on 18 August 2019).
- Busse, M.D.; Hubbert, K.R.; Fiddler, G.O.; Shestak, C.J.; Powers, R.F. Lethal soil temperatures during burning of masticated forest residues. Int. J. Wildland Fire 2005, 14, 267–276. [Google Scholar] [CrossRef]
- City of Santa Rosa Water. Technical Memorandum No. 1 Post-Fire Water Quality Investigation: Analysis of Cause of Water Contamination. Available online: https://srcity.org/DocumentCenter/View/19837/Post-Fire-Water-Quality-Investigation-Analysis-of-Cause-of-Water-Contamination (accessed on 3 November 2019).
- Quinn, A.L.; Regan, J.M.; Tobin, J.M.; Marinik, B.J.; McMahon, J.M.; McNett, D.A.; Sushynski, C.M.; Crofoot, S.D.; Jean, P.A.; Plotzke, K.P. In Vitro and In Vivo Evaluation of the Estrogenic, Androgenic, and Progestagenic Potential of Two Cyclic Siloxanes. Toxicol. Sci. 2007, 96, 145–153. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Rhodes-Brower, S.; Miller, M.R.; Munson, A.E.; Germolec, D.R.; Walker, V.R.; Korach, K.S.; Meade, B.J. Octamethylcyclotetrasiloxane exhibits estrogenic activity in mice via ERalpha. Toxicol. Appl. Pharmacol. 2003, 192, 254–261. [Google Scholar] [CrossRef]
- Skjevrak, I.; Due, A.; Gjerstad, K.O.; Herikstad, H. Volatile organic components migrating from plastic pipes (HDPE, PEX and PVC) into drinking water. Water Res. 2003, 37, 1912–1920. [Google Scholar] [CrossRef]
- World Health Organization. IARC Monographs on the Identification of Carcinogenic Hazards to Humans. Available online: https://monographs.iarc.fr/agents-classified-by-the-iarc/ (accessed on 20 August 2019).
- King, W.D.; Marrett, L.D. Case-control study of bladder cancer and chlorination by-products in treated water (Ontario, Canada). Cancer Causes Control 1996, 7, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Ratnayaka, D.D.; Brandt, M.J.; Johnson, K.M. Water Supply, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2009; Available online: https://www.sciencedirect.com/topics/earth-and-planetary-sciences/asbestos-cement (accessed on 24 August 2019).
- EPA Report on Permeation and Leaching Prepared by American Water Works Association (AWWA). November 2002. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/permeationandleaching.pdf (accessed on 24 August 2019).
- Hugget, C.; Levin, B.C. Toxicity of the Pyrolysis and Combustion Products of Poly (Vinyl Chlorides): A Literature Assessment. Fire Mater. 1987, 11, 131–142. [Google Scholar] [CrossRef]
- Orecchio, S.; Fiore, M.; Barreca, S.; Vara, G. Volatile Profiles of Emissions from Different Activities Analyzed Using Canister Samplers and Gas Chromatography-Mass Spectrometry (GC/MS) Analysis: A Case Study. Int. J. Environ. Res. Public Health 2017, 14, 195. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Buekens, A.; Jiang, X.; Li, X. Dioxins and polyvinylchloride in combustion and fires. Waste Manag. Res. 2015, 33, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, T.; Negoro, R.; Muto, A.; Sakata, Y. Prevention of chlorinated hydrocarbons formation during pyrolysis of PVC or PVDC mixed plastics. Green Chem. 2006, 8, 697–700. [Google Scholar] [CrossRef]
- Al Robaidi, A.; Mousa, A.; Massadeh, S.; Al Rawabdeh, I.; Anagreh, N. The Potential of Silane Coated Calcium Carbonate on Mechanical Properties of Rigid PVC Composites for Pipe Manufacturing. Mater. Sci. Appl. 2011, 2, 481–485. [Google Scholar] [CrossRef]
- Guermazi, N.; Haddar, N.; Elleuch, K.; Ayedi, H.F. Effect of filler addition and weathering conditions on the performance of PVC/CaCO3 composites. Polym. Compos. 2015, 37, 2171–2183. [Google Scholar] [CrossRef]
- Whelton, A.J.; Nguyen, T. Contaminant Migration from Polymeric Pipes Used in Buried Potable Water Distribution Systems: A Review. Crit. Rev. Env. Sci. Tec. 2013, 43, 679–751. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Nor, N.H.M.; Hermsena, E.; Kooi, M.; Mintenig, S.M.; France, J.D. Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res. 2019, 155, 410–422. [Google Scholar] [CrossRef] [PubMed]
| Identified Compounds | Burned PVC | PVC |
|---|---|---|
| Tetradecane | 27.8 | 21.6 |
| Hexadecane | 11.6 | 13.6 |
| Octadecane | 1.1 | 1.2 |
| Docosane | 9.0 | 5.4 |
| 2-Butoxyethanol * | 2.7 | 2.8 |
| Irganox 1010 constituent ** | 3.3 | 3.8 |
| 2-Ethyl-1-hexanol | 14.0 | 11.6 |
| Diethyl phthalate | 3.1 | 2.4 |
| Diisooctyl phthalate | <MDL *** | 3.0 |
| GC Peak (min) | Compounds | Level (ppbv) | PEL * (ppmv) | STEL * (ppmv) | Cancer Target Risk (CTR) ** = 1 × 10−6 (mg/m3; ppbv) | Non-cancer Hazard Index (NCHI) = 1 (mg/m3; ppbv) |
|---|---|---|---|---|---|---|
| 4.61 | Chloromethane | 3007 | 50 | 100 | N.A.*** | 9.4 × 101; 45.5 |
| 4.87 | Chloroethene | 25 | N.A. | N.A. | 1.7 × 10−1; 0.0665 | 1.0 × 102; 39.1 |
| 5.00 | 1-Butene | 4885 | N.A. | N.A. | N.A. | N.A. |
| 5.02 | 1,3-Butadiene | 2377 | 1 | 5 | 9.4 × 10−2; 0.0425 | 2.1; 0.95 |
| 5.49 | Ethyl Chloride | 506 | 100 | N.A. | N.A. | 1.0 × 104; 3789 |
| 7.10 | Methylene chloride | 10 | 200 | 300 | N.A. | 5.2 × 103; 1763 |
| 7.42 | Carbon disulfide | 4 | N.A. | N.A. | N.A. | 7.3 × 102; 207 |
| 11.63 | Benzene | 10,519 | 1 | 5 | 3.6 × 10−1 (0.113 ppbv) | 3.1 × 101; 9.7 |
| 18.63 | Chlorobenzene | 10 | 10 | N.A. | N.A. | 5.2 × 101; 11.3 |
| 19.23 | Ethylbenzene | 11 | 5 | 30 | 1.1 (0.253 ppbv) | 1.0 × 103; 230 |
| 20.19 | o-Xylene | 11 | 100 | 150 | N.A. | 1.0 × 102; 23.0 |
| Carbon monoxide CO | Ethylene C2H4 | Acetylene C2H2 | Hydrogen Chloride HCl | Formaldehyde HCHO | Octamethyl-cyclotetra- siloxane C8H24O4Si4 | |
|---|---|---|---|---|---|---|
| Mean ± 1sd * | 33.8 ± 2.1 | 3.1 ± 0.3 | 2.0 ± 0.1 | 2.3 ± 0.1 | 0.049 ± 0.001 | 0.462 ± 0.051 |
| Solubility ** | 27.6 mg/L | 2.9 mg/L | Very low | 720 g/L | 40 g/L | 56.2 μg/L |
| Vap. P. ** | >760 torr | >760 torr | >760 torr | >760 torr | >760 torr | 0.934 torr |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chong, N.S.; Abdulramoni, S.; Patterson, D.; Brown, H. Releases of Fire-Derived Contaminants from Polymer Pipes Made of Polyvinyl Chloride. Toxics 2019, 7, 57. https://doi.org/10.3390/toxics7040057
Chong NS, Abdulramoni S, Patterson D, Brown H. Releases of Fire-Derived Contaminants from Polymer Pipes Made of Polyvinyl Chloride. Toxics. 2019; 7(4):57. https://doi.org/10.3390/toxics7040057
Chicago/Turabian StyleChong, Ngee Sing, Saidi Abdulramoni, Dwight Patterson, and Heather Brown. 2019. "Releases of Fire-Derived Contaminants from Polymer Pipes Made of Polyvinyl Chloride" Toxics 7, no. 4: 57. https://doi.org/10.3390/toxics7040057
APA StyleChong, N. S., Abdulramoni, S., Patterson, D., & Brown, H. (2019). Releases of Fire-Derived Contaminants from Polymer Pipes Made of Polyvinyl Chloride. Toxics, 7(4), 57. https://doi.org/10.3390/toxics7040057





