Pressurized Solvent Extraction with Ethyl Acetate and Liquid Chromatography—Tandem Mass Spectrometry for the Analysis of Selected Conazole Fungicides in Matcha
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
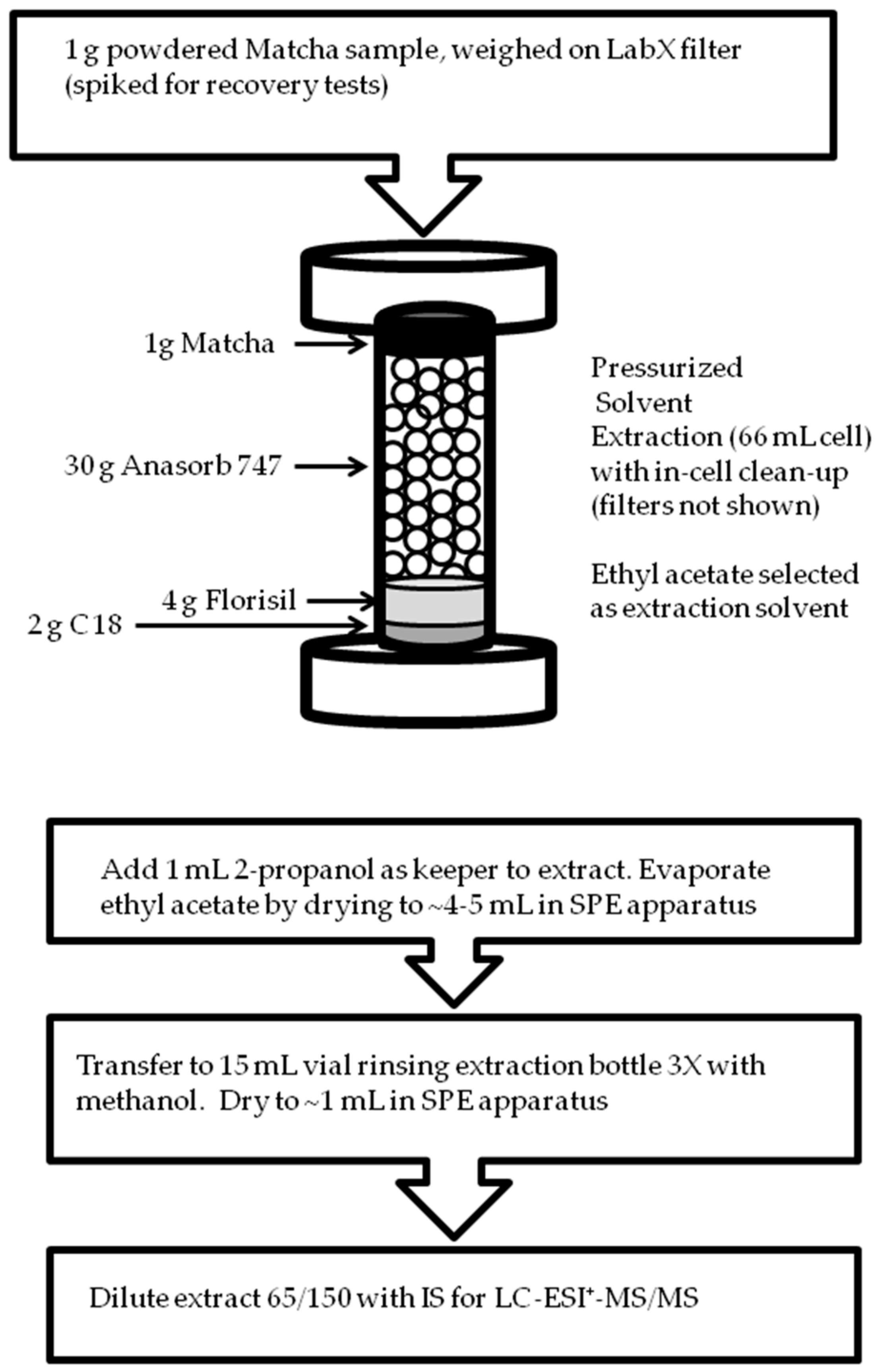
2.2. Sample Preparation
2.3. LC-ESI+-MS/MS Analysis
3. Results
3.1. Cell Design for Pressurized Solvent Extraction of Matcha
3.2. Modifications to the LC-ESI+-MS/MS Analysis
3.3. Method Detection Limits and Calibration
3.4. Matrix Effects and Recoveries of Pressurized Solvent Extraction
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Raina-Fulton, R.; Behdarvandan, A.; Mohamad, A.A. The challenges of fungicide analyses using gas chromatography and liquid chromatography-mass spectrometry methods. Austin Environ. Sci. 2018, 3, 1031. [Google Scholar]
- Raina-Fulton, R.; Aborkhees, G.; Behdarvandan, A. Analysis of herbicide and/or pesticide residues in dietary botanical supplements. Encycl. Anal. Chem. 2018, A9603, 1–126. [Google Scholar] [CrossRef]
- Xue, J.; Li, H.; Liu, F.; Xue, J.; Chen, X.; Zhan, J. Transfer of difenoconazole and azoxystrobin residues from chrysanthemum flow tea to its infusion. Food Addit. Contam. Part A 2014, 31, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Raina, R.; Smith, E. Detection of azole fungicides in atmospheric samples collected in the Canadian prairies. J. AOAC Int. 2012, 95, 1350–1356. [Google Scholar] [CrossRef] [PubMed]
- Pihlström, T.; Blomkvist, G.; Friman, P.; Pagard, U.; Österdahl, B. Analysis of pesticide residues in fruit and vegetables with ethyl acetate extraction using gas and liquid chromatography with tandem mass spectrometric detection. Anal. Bioanal. Chem. 2007, 389, 1773–1789. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, Y.; Matsuoka, T.; Mitsuhashi, T. Multi-residue screening method of acidic pesticides in agricultural products by liquid chromatography/time of flight mass spectrometry. J. Pestic. Sci. 2009, 34, 265–272. [Google Scholar] [CrossRef]
- Zayats, M.F.; Leschev, S.M.; Zayats, M.A. An improved extraction method of rapeseed oil sample preparation for the subsequent determination in it of azole class fungicides by gas chromatography. Anal. Chem. Res. 2015, 3, 37–45. [Google Scholar] [CrossRef][Green Version]
- Grimalt, S.; DeHouck, P. Review of analytical methods for the determination of pesticide residues in grapes. J. Chromatogr. A 2016, 1433, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, M.R.; Oulkar, D.P.; Shabeer, A.T.P.; Banerjee, K. Quantitative screening of agrochemical residues in fruits and vegetables by buffered ethyl acetate extraction and LC-MS/MS. J. Agric. Food Chem. 2015, 63, 4449–4456. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Yoshida, Y.; Kiso, M.; Nagashima, H. Simultaneous analysis of individual catechins and caffeine in green tea. J. Chromatogr. A 1996, 749, 295–299. [Google Scholar] [CrossRef]
- Weiss, D.J.; Anderton, C.R. Determination of catechins in matcha green tea by micellar electrokinetic chromatography. J. Chromatogr. A 2003, 1011, 173–180. [Google Scholar] [CrossRef]
- El-Aty, A.M.A.; Choi, J.; Rahman, M.; Kim, S.; Tosun, A.; Shim, J. Residues and contaminants in tea and tea infusions: A review. Food Addit. Contam. Part A 2014, 31, 1794–1804. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Nemoto, S.; Matsuda, R. Simultaneous determination of pesticide residues in tea by LC-MS/MS-Modification of Japanese official multiresidue method. Jpn. J. Food Chem. Saf. 2014, 21, 27–36. [Google Scholar] [CrossRef]
- Saito, S.; Nemoto, S.; Teshima, R. Multiresidue determination of pesticides in tea by gas chromatography-tandem mass spectrometry. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2015, 50, 760–776. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lopez, S.; Hayward, D.G.; Park, H.Y.; Wong, J.W.; Kim, S.S.; Wan, J.; Reddy, R.M.; Quinn, D.J.; Steinger, D. Determination of multiresidue pesticides in botanical dietary supplements using gas chromatography-triple-quadrupole mass spectrometry (GC-MS/MS). J. Agric. Food Chem. 2016, 64, 6125–6132. [Google Scholar] [CrossRef] [PubMed]
- Villanueva Bermejo, D.; Ibáñez, E.; Reglero, G.; Turner, C.; Fornari, T.; Rodriguez-Meizoso, I. High catechins/low caffeine powder from green tea leaves by pressurized liquid extraction and supercritical antisolvent precipitation. Sep. Purif. Technol. 2015, 148, 49–56. [Google Scholar] [CrossRef]
- Raina-Fulton, R. Determination of neonicotinoid insecticides and strobilurin fungicides in particle phase atmospheric samples by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2015, 63, 5152–5162. [Google Scholar] [CrossRef] [PubMed]
- Chitescu, C.L.; Oosterink, E.; de Jong, J.; Stolker, A.A.M. Ultrasonic or accelerated solvent extraction followed by U-HPLC-high mass accuracy MS for screening of pharmaceuticals and fungicides in soil and plant samples. Talanta 2012, 88, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bao, X.; Zhao, S.; Wu, J.; Han, A.; Ye, Q. Analysis of multi-pesticide residues in the foods of animal origin by GC-MS coupled with accelerated solvent extraction and gel permeation chromatography cleanup. Food Chem. 2011, 126, 646–654. [Google Scholar] [CrossRef]
- Bakirci, G.T.; Hişil, Y. Fast and simple extraction of pesticide residues in selected fruits and vegetables using tetrafluoroethane and toluene followed by ultrahigh-performance liquid chromatography/tandem mass spectrometry. Food Chem. 2012, 135, 1901–1913. [Google Scholar] [CrossRef] [PubMed]
- Karcher, S.; Kornmüller, A.; Jekel, M. Screening of commercial sorbents for the removal of reactive dyes. Dyes Pigments 2001, 51, 111–125. [Google Scholar] [CrossRef]
- Gałuszka, A.; Migaszewski, Z.M.; Konieczka, P.; Namieśnik, J. Analytical Eco-Scale for assessing the greenness of analytical procedures. Trends Anal. Chem. 2012, 37, 61–72. [Google Scholar] [CrossRef]
- Płotka-Wasylka, J. A new tool for the evaluation of the analytical procedure: Green Analytical Procedure Index. Talanta 2018, 181, 204–209. [Google Scholar] [CrossRef] [PubMed]
| Target Analyte | Retention Time (min) | Quantitative SRM, Confirmation SRM (Cone Voltage, Collision Energy) | Method Detection Limits with Matrix Matched Standards (mg/kg) |
|---|---|---|---|
| Benzotriazole | 8.24 | 120→65 (40,17), 120→92 (40,17) | 0.0006 |
| Sulfathiazole | 8.46 | 256→155 (20,15), 256→92 (20,25) | 0.002 |
| Imazamox | 9.09 | 306→261 (40,20), 306→217 (40,20) | 0.010 |
| Sulfamethizole | 9.34 | 271→156 (20,15), 271→92 (20,25) | 0.002 |
| Tebuthiuron (thiadiazolylurea herbicide) | 10.48 | 229→172 (25,15), 229→116 (25,25) | 0.001 |
| Tricyclazole (benzothiazole fungicide) | 12.06 | 190→163 (35,20), 190→136 (35,25) | 0.002 |
| Sulfentrazone | 15.36 | 387→307 (35,20), 389→309 (35,20) | 0.010 |
| Imazalil | 15.8 | 297→159 (20,25), 297→201 (20,20) | 0.010 |
| Thioconazole | 15.93 | 391→130 (20,20), 391→360 (20,10) | 0.010 |
| Azaconazole | 16.71 | 300→159 (30,25), 300→231 (30,15) | 0.001 |
| Triadimenol | 18.19 | 296→70 (15,15), 298→70 (15,15), 296→99 (15,10) | 0.002 |
| Paclobutrazol (plant growth regulator with triazole moiety) | 18.58 | 294→70 (30,20), 295→70 (25,20), 296→70 (15,15) | 0.010 |
| Triticonazole | 19.39 | 318→70 (20,15), 320→70 (20,20) | 0.010 |
| Cyproconazole | 19.76 | 292→70 (30,15), 294→70(30,20) | 0.002 |
| Hexaconazole | 20.58 | 314→70 (25,20), 316→70(25,20) | 0.010 |
| Uniconazole (uniconazole-P) | 20.94 | 292→70 (30,15), 294→70 (30,20) | 0.010 |
| Etaconazole | 21.58 | 328→159 (30,25), 330→161 (30,25), 328→187 (30,30) | 0.001 |
| Prochloraz | 21.61 | 376→70 (15,25),378→70 (15,25), 376→308 (15,15) | 0.010 |
| Myclobutanil | 21.73 | 289→70 (25,15), 291→70 (25,15) | 0.010 |
| Triadimefon | 21.73 | 295→70 (25,20), 297→70 (25,20) | 0.002 |
| Prothioconazole (analyzed as prothioconazole-desthio) | 21.75 | 314→70 (25,20), 312→70 (25,20), 312→125 (25,20) | 0.005 |
| Tebuconazole | 21.94 | 308.5→70 (35,20), 310.5→70 (35,20), 308.5→125 (35,20) | 0.001 |
| Bromuconazole | 22.01 | 376→159 (30,25), 378→159 (30,25) | 0.010 |
| Penconazole | 22.12 | 284→70 (25,15), 284→159 (25,15) | 0.010 |
| Metconazole | 22.15 | 321→70 (30,20), 323→70 (30,20) | 0.010 |
| Diniconazole | 22.46 | 326→70(35,25), 328→70(35,25), 326→159 (35,20) | 0.0006 |
| Epoxiconazole | 22.46 | 330→121 (25,20), 332→121 (25,20), 330→123 (25,20) | 0.010 |
| Tetraconazole | 22.46 | 372→159 (30,25), 372→70 (30,25) | 0.010 |
| Biteranol | 22.73 | 338→99 (15,15), 338→269 (15,15) | 0.002 |
| Propiconazole | 22.73 | 342→159 (30,25), 342→69 (30,25) | 0.010 |
| Flusilazole | 22.94 | 316→165 (30,25), 316→248 (30,15) | 0.0006 |
| Fenbuconazole | 23.12 | 337→70 (30,20), 337→125 (30,20) | 0.001 |
| Tebufenozide (insecticide) | 23.12 | 353→133 (12,17), 353→297 (12,17) | 0.002 |
| Difenoconazole | 23.64 | 406→251 (30,25), 408→253 (30,25) | 0.010 |
| Etoxazole | 25.02 | 360→57 (35,25), 360→141 (35,30), 360→177.5 (35,20) | 0.010 |
| Propiconazole-phenyld3 (internal standard) | 22.80 | 347→164 (50,25), 349→166 (50,25), 347→69 (50,25), 349→69 (50,25) | NA |
| Target Analyte (SRM) | Recovery, Spiked at 0.01 mg/kg 1 (Average ±SD, n = 4 ) | % Matrix Effect | Detected Concentration in Matcha (mg/kg) 2 |
|---|---|---|---|
| Selected Analytes with MS Signal Suppression or Soft Enhancement | |||
| Tebuthiuron (229→172) | 80.7 ± 4.70 | −19% | ND |
| Sulfentrazone (387→307) | 64.0 ± 18.3 | −35% | ND |
| Triadimenol (296→70) | 109.5 ± 11.7 | −32% | ND |
| Paclobutanil (295→70) | 51.8 ± 14.0 | −38% | ND |
| Cyproconazole (292→70) | 69.3 ± 12.9 | −37% | ND |
| Uniconazole (292→70) | 23.9 ± 12.9 | −29% | ND |
| Myclobutanil (291→70) | 84.9 ± 38.3 | −53% | ND |
| Triadimenfon (295→70) | 96.1 ± 34.6 | −44% | ND |
| Hexaconazole (314→70) | 12.1 ± 19.8 | −75% | ND |
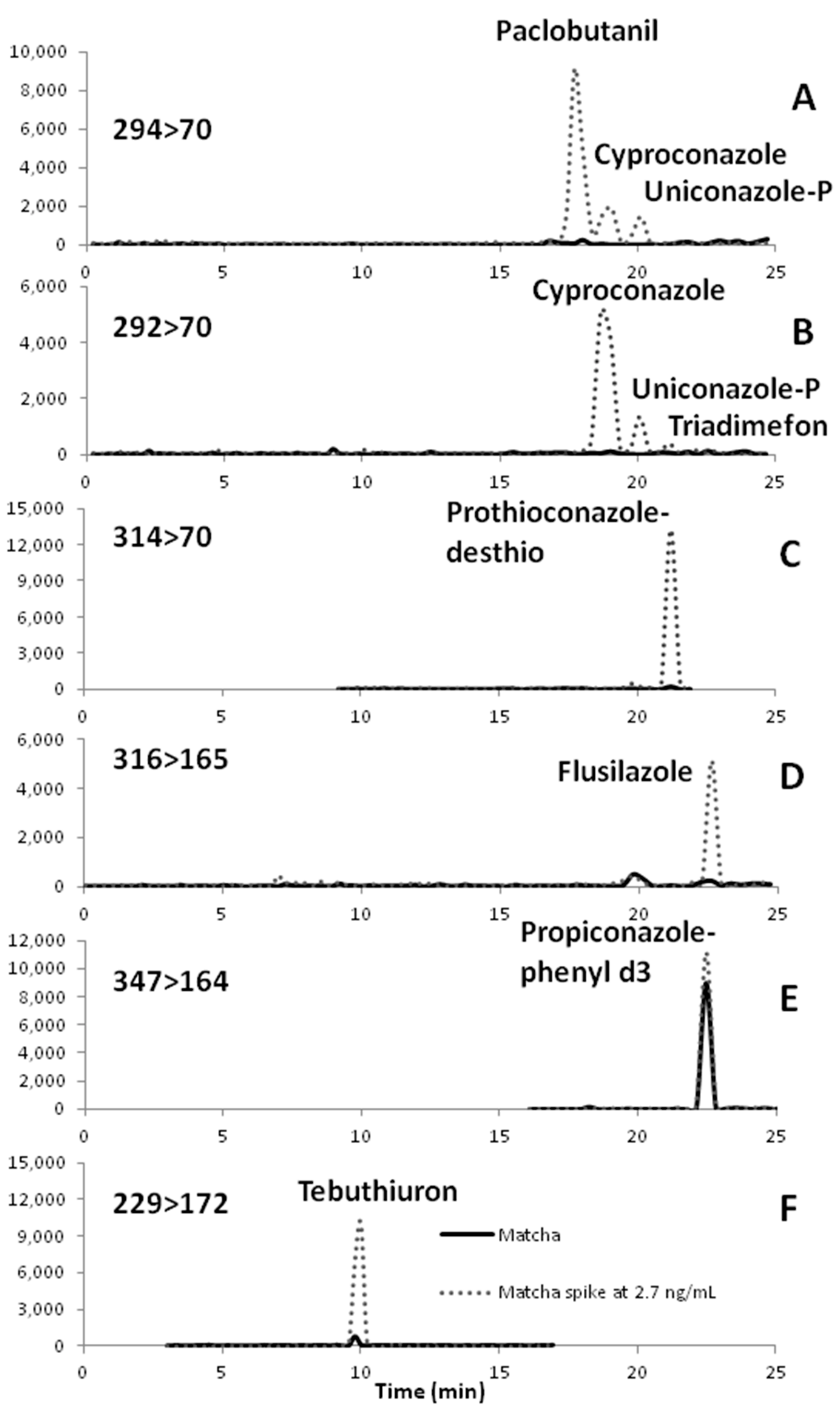
| Prothioconazole-desthio (314→70) | 69.2 ± 29.2 | −54% | 0.0035 |
| Flusilazole (316→165) | 40.4 ± 28.9 | 6% | 0.0024 |
| Propiconazole (342→159) | 41.9 ± 25.0 | −54% | ND |
| Etaconazole (330→161) | 49.1 ± 11.3 | −5% | ND |
| Azaconazole (300→159) | 32.3 ± 11.0 | −33% | ND |
| Difenconazole (406→251) | 20.4 ± 87.9 | −47% | ND |
| Analytes with Severe Signal Enhancement | |||
| Diniconazole (326→70) | 21.5 ± 5.40 | 102% | ND |
| Fenbuconazole (337→70) | 29.9 ± 8.10 | 81% | ND |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raina-Fulton, R.; Mohamad, A.A. Pressurized Solvent Extraction with Ethyl Acetate and Liquid Chromatography—Tandem Mass Spectrometry for the Analysis of Selected Conazole Fungicides in Matcha. Toxics 2018, 6, 64. https://doi.org/10.3390/toxics6040064
Raina-Fulton R, Mohamad AA. Pressurized Solvent Extraction with Ethyl Acetate and Liquid Chromatography—Tandem Mass Spectrometry for the Analysis of Selected Conazole Fungicides in Matcha. Toxics. 2018; 6(4):64. https://doi.org/10.3390/toxics6040064
Chicago/Turabian StyleRaina-Fulton, Renata, and Aisha A. Mohamad. 2018. "Pressurized Solvent Extraction with Ethyl Acetate and Liquid Chromatography—Tandem Mass Spectrometry for the Analysis of Selected Conazole Fungicides in Matcha" Toxics 6, no. 4: 64. https://doi.org/10.3390/toxics6040064
APA StyleRaina-Fulton, R., & Mohamad, A. A. (2018). Pressurized Solvent Extraction with Ethyl Acetate and Liquid Chromatography—Tandem Mass Spectrometry for the Analysis of Selected Conazole Fungicides in Matcha. Toxics, 6(4), 64. https://doi.org/10.3390/toxics6040064




