Sub-Micromolar Methylmercury Exposure Promotes Premature Differentiation of Murine Embryonic Neural Precursor at the Expense of Their Proliferation
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Ethics
2.2. Cell Culture Procedures and Experimental Treatments
2.3. Immunocytochemistry
2.4. Microscopy and Quantification
2.5. Statistics
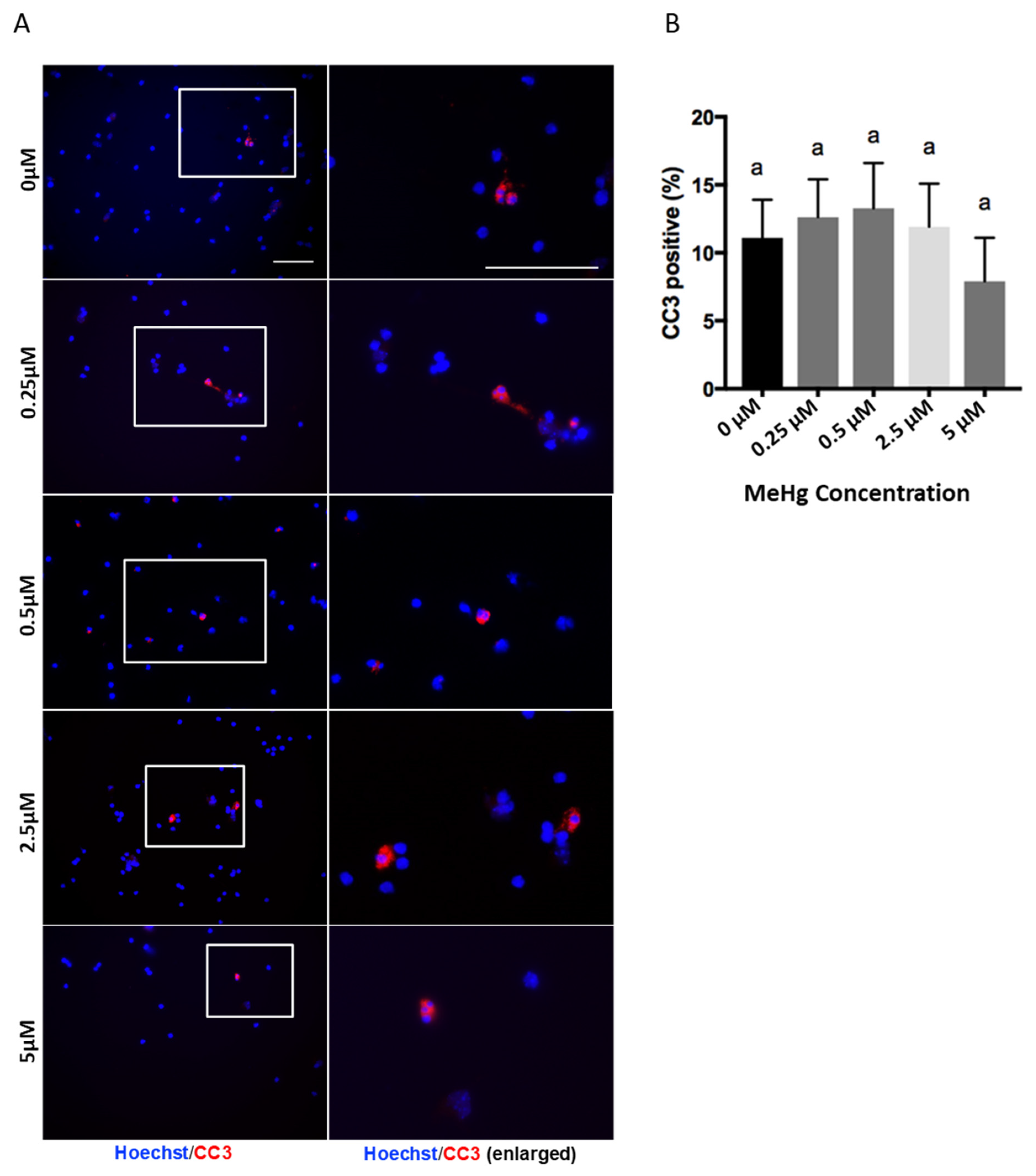
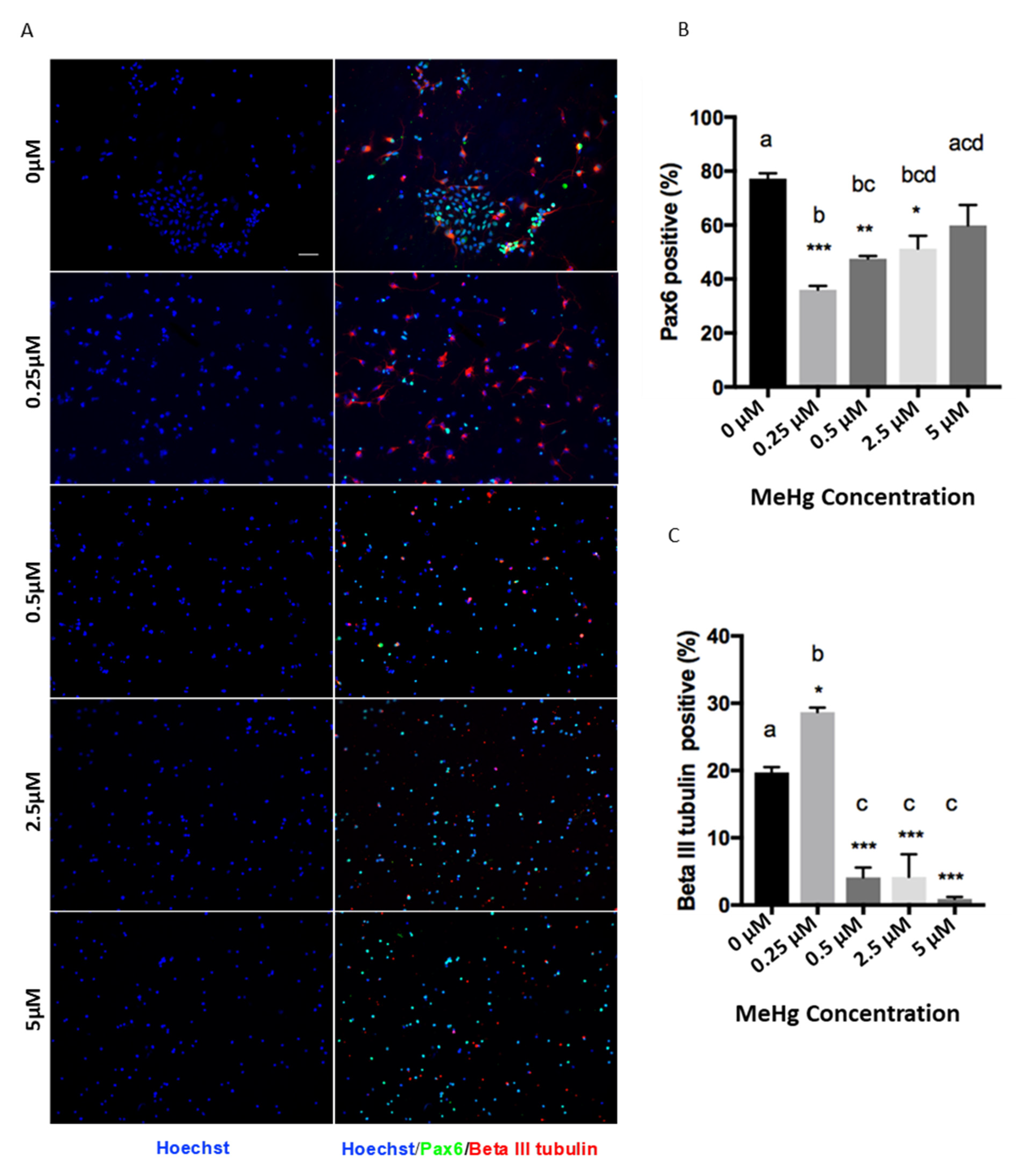
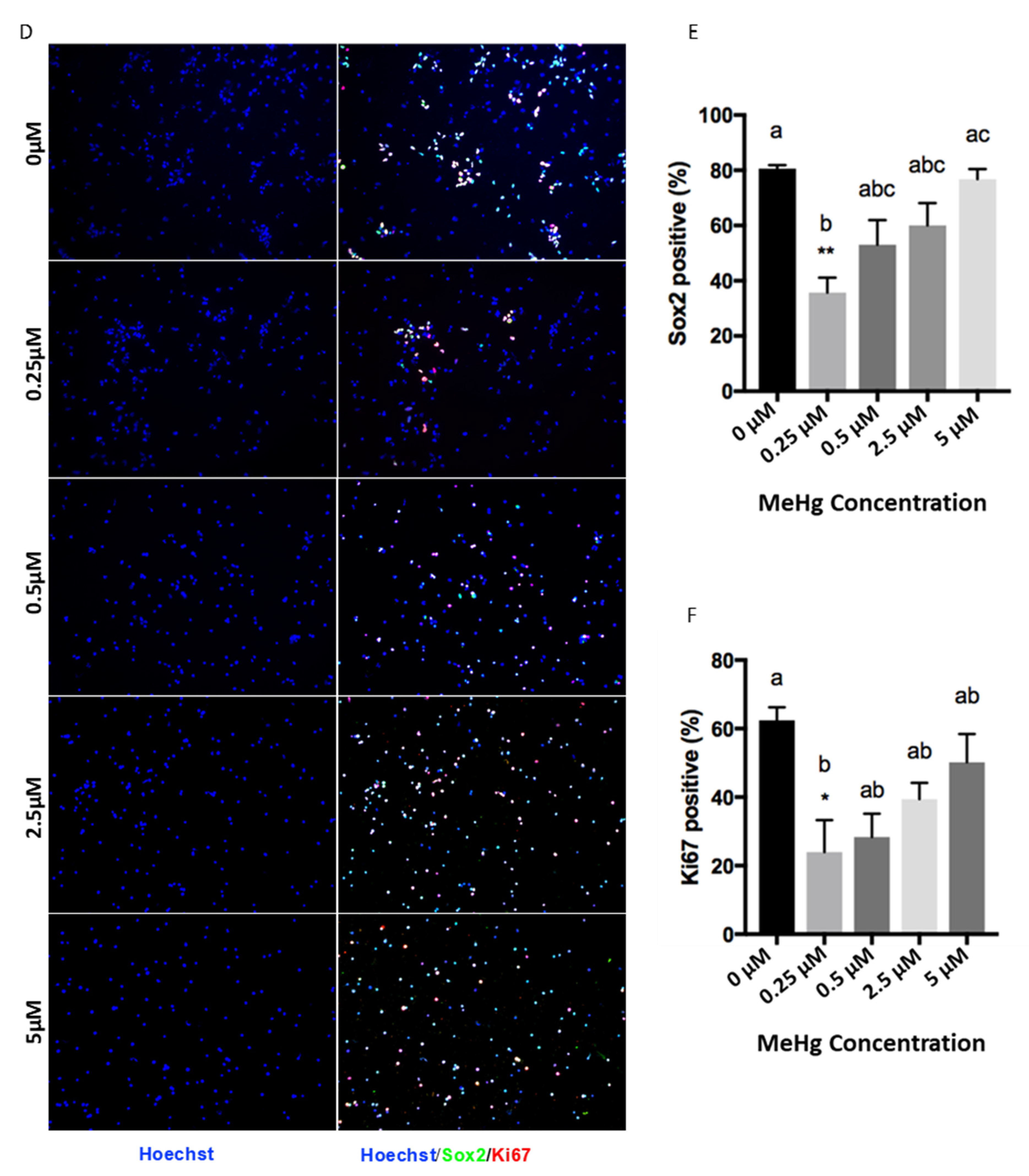
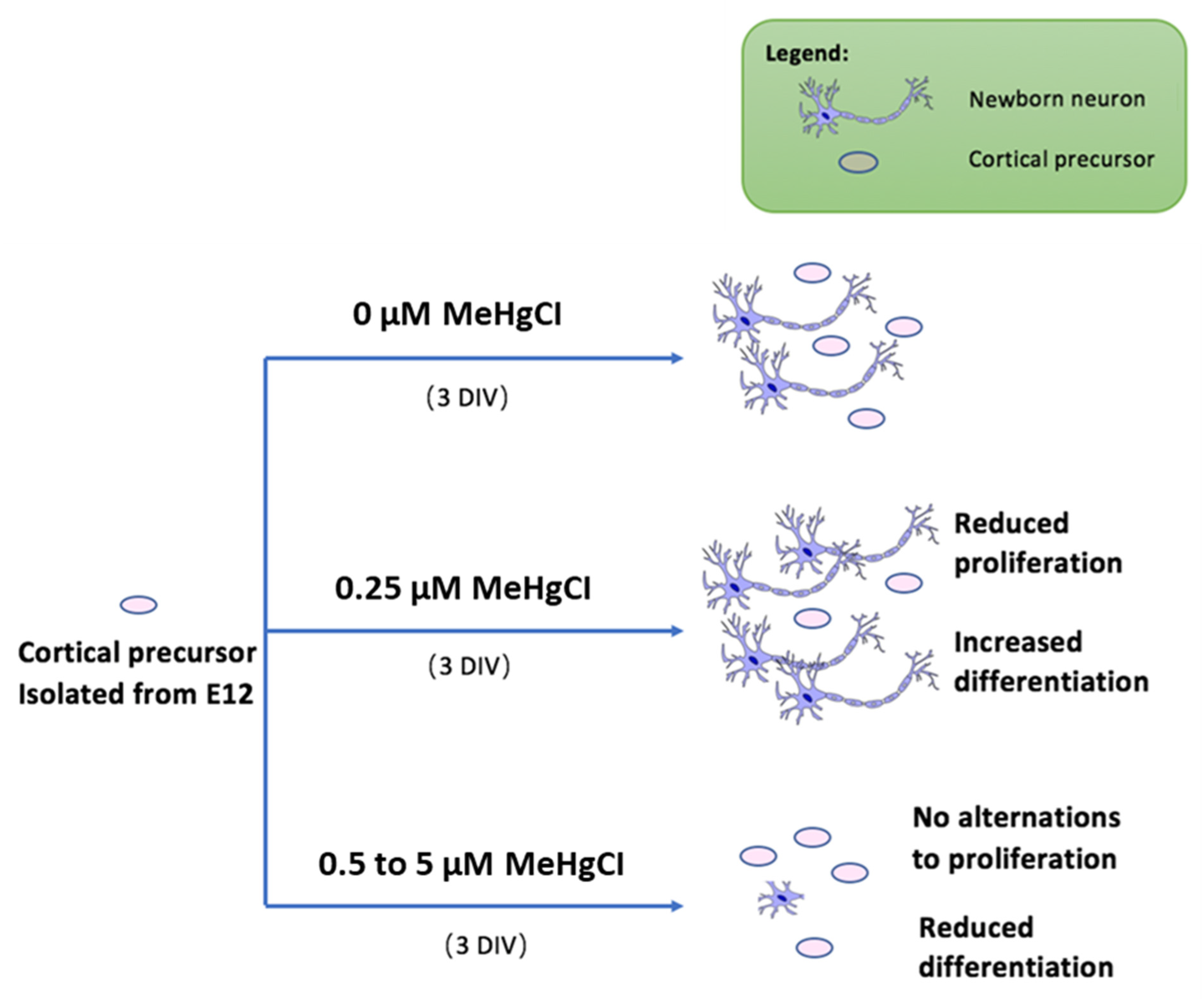
3. Results
3.1. Cell Viability
3.2. Effects of Cortical Precursors on Proliferation and Differentiation
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mergler, D.; Anderson, H.A.; Chan, L.H.M.; Mahaffey, K.R.; Murray, M.; Sakamoto, M.; Stern, A.H. Methylmercury exposure and health effects in humans: A worldwide concern. AMBIO J. Hum. Environ. 2007, 36, 3–11. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Mason, R.P.; Chan, H.M.; Jacob, D.J.; Pirrone, N. Mercury as a global pollutant: Sources, pathways, and effects. Environ. Sci. Technol. 2013, 47, 4967–4983. [Google Scholar] [CrossRef] [PubMed]
- Ha, E.; Basu, N.; Bose-O’Reilly, S.; Dórea, J.G.; McSorley, E.; Sakamoto, M.; Chan, H.M. Current progress on understanding the impact of mercury on human health. Environ. Res. 2017, 152, 419–433. [Google Scholar] [CrossRef] [PubMed]
- Amin Zaki, L.; Elhassani, S.; Majeed, M.A.; Clarkson, T.W.; Doherty, R.A.; Greenwood, M.R.; Giovanoli-Jakubczak, T. Perinatal Methylmercury Poisoning in Iraq. Am. J. Dis. Child. 1976, 130, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Koya, G.; Takeuchi, T. Fetal Minamata disease: A neuropathological study of two cases of intrauterine intoxication by a methyl mercury compound. J. Neuropathol. Exp. Neurol. 1965, 24, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Debes, F.; Budtz-Jørgensen, E.; Weihe, P.; White, R.F.; Grandjean, P. Impact of prenatal methylmercury exposure on neurobehavioral function at age 14 years. Neurotoxicol. Teratol. 2006, 28, 536–547. [Google Scholar] [CrossRef] [PubMed]
- Yorifuji, T.; Tsuda, T.; Inoue, S.; Takao, S.; Harada, M. Long-term exposure to methylmercury and psychiatric symptoms in residents of Minamata, Japan. Environ. Int. 2011, 37, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Onishchenko, N.; Tamm, C.; Vahter, M.; Hökfelt, T.; Johnson, J.A.; Johnson, D.A.; Ceccatelli, S. Developmental exposure to methylmercury alters learning and induces depression-like behavior in male mice. Toxicol. Sci. 2007, 97, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Onishchenko, N.; Karpova, N.; Sabri, F.; Castrén, E.; Ceccatelli, S. Long-lasting depression-like behavior and epigenetic changes of BDNF gene expression induced by perinatal exposure to methylmercury. J. Neurochem. 2008, 106, 1378–1387. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, L.; Tomasini, M.C.; Tanganelli, S.; Mazza, R.; Coluccia, A.; Carratù, M.R.; Antonelli, T. Developmental exposure to methylmercury elicits early cell death in the cerebral cortex and long-term memory deficits in the rat. Int. J. Dev. Neurosci. 2009, 27, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Yan, C.; Tian, Y.; Yuan, X.; Shen, X. Effects of low level of methylmercury on proliferation of cortical progenitor cells. Brain Res. 2010, 1359, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Tamm, C.; Duckworth, J.; Hermanson, O.; Ceccatelli, S. High susceptibility of neural stem cells to methylmercury toxicity: Effects on cell survival and neuronal differentiation. J. Neurochem. 2006, 97, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Theunissen, P.T.; Pennings, J.L.A.; Robinson, J.F.; Claessen, S.M.H.; Kleinjans, J.C.S.; Piersma, A.H. Time-response evaluation by transcriptomics of methylmercury effects on neural differentiation of murine embryonic stem cells. Toxicol Sci. 2011, 122, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Burke, K.; Cheng, Y.; Li, B.; Petrov, A.; Joshi, P.; Berman, R.F.; Reuhl, K.R.; DiCicco-Bloom, E. Methylmercury elicits rapid inhibition of cell proliferation in the developing brain and decreases cell cycle regulator, cyclin E. Neurotoxicology 2006, 27, 970–981. [Google Scholar] [CrossRef] [PubMed]
- Silveira, P.P.; Portella, A.K.; Goldani, M.Z.; Barbieri, M.A. Developmental origins of health and disease (DOHaD). J. Pediatr. 2007, 83, 494–504. [Google Scholar] [CrossRef]
- Gillman, M.W.; Barker, D.; Bier, D.; Cagampang, F.; Challis, J.; Fall, C.; Thornburg, K.L. Meeting Report on the 3rd International Congress on Developmental Origins of Health and Disease (DOHaD). Pediatr. Res. 2007, 61, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, M.; Usuki, F. Low concentrations of methylmercury inhibit neural progenitor cell proliferation associated with up-regulation of glycogen synthase kinase 3β and subsequent degradation of cyclin E in rats. Toxicol Appl Pharmacol. 2015, 288, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Tamm, C.; Duckworth, J.K.; Hermanson, O.; Ceccatelli, S. Methylmercury inhibits differentiation of rat neural stem cells via Notch signaling. NeuroReport 2008, 19, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Bose, R.; Onishchenko, N.; Edoff, K.; Lang, A.M.J.; Ceccatelli, S. Inherited effects of low-dose exposure to methylmercury in neural stem cells. Toxicol. Sci. 2012, 130, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Hanao, N.; Nishiyama, K.; Kadota, Y.; Inoue, M.; Sato, M.; Suzuki, S. Differential effects of cobalt and mercury on lipid metabolism in the white adipose tissue of high-fat diet-induced obesity mice. Toxicol. Appl. Pharmacol. 2012, 258, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.W.; Tobita, M.; Takahashi, T.; Kuge, S.; Kita, K.; Naganuma, A. siRNA-mediated AMPKalpha1 subunit gene PRKAA1 silencing enhances methylmercury toxicity in HEK293 cells. J. Toxicol. Sci. 2010, 35, 601–604. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gallagher, D.; DeVito, L.; Cancino, I.; Tsui, D.; He, L.; Keller, G.M.; Frankland, P.W.; Kaplan, D.R.; Miller, F.D. Metformin activates atypical PKC-CBP pathway to promote neurogenesis and enhance spatial memory formation. Cell Stem Cell 2012, 11, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Fatt, M.; Hsu, K.; He, L.; Wondisford, F.; Miller, F.D.; Kaplan, D.R.; Wang, J. Metformin acts on two different molecular pathways to enhance adult neural precursor proliferation/self-renewal and differentiation. Stem Cell Reports 2015, 5, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, M.; Zhang, Y.; Lopez-Toledano, M.; Clarke, A.; Deth, R. Differential neurogenic effects of casein-derived opioid peptides on neuronal stem cells: Implications for redox-based epigenetic changes. J. Nutr. Biochem. 2017, 37, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Juliandi, B.; Tanemura, K.; Igarashi, K.; Tominaga, T.; Furukawa, Y.; Otsuka, M.; Moriyama, N.; Ikegami, D.; Abematsu, M.; Sanosaka, T.; et al. Reduced Adult Hippocampal Neurogenesis and Cognitive Impairments following Prenatal Treatment of the Antiepileptic Drug Valproic Acid. Stem Cell Reports 2015, 5, 996–1009. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Norman, A.A.; Woodard, C.L.; Yang, G.; Gauthier-Fisher, A.; Fujitani, M.; Vessey, J.P.; Cancino, G.I.; Sachewsky, N.; Woltjen, K.; et al. Transient maternal IL-6 mediates long-lasting changes in neural stem cell pools by deregulating an endogenous self-renewal pathway. Cell Stem Cell 2013, 13, 564–576. [Google Scholar] [CrossRef] [PubMed]
- Lapham, L.W.; Cernichiari, E.; Cox, C.; Myers, G.J.; Baggs, R.B.; Brewer, R.; Shamlaye, C.F.; Davidson, P.W.; Clarkson, T.W. An analysis of autopsy brain tissue from infants prenatally exposed to methylmercury. Neurotoxicology 1995, 16, 689–704. [Google Scholar] [PubMed]
- Sakamoto, M.; Chan, H.M.; Domingo, J.L.; Koriyama, C.; Murata, K. Placental transfer and levels of mercury, selenium, vitamin E, and docosahexaenoic acid in maternal and umbilical cord blood. Environ. Int. 2018, 111, 309–315. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, X.; Wang, J.; Chan, H.M. Sub-Micromolar Methylmercury Exposure Promotes Premature Differentiation of Murine Embryonic Neural Precursor at the Expense of Their Proliferation. Toxics 2018, 6, 61. https://doi.org/10.3390/toxics6040061
Yuan X, Wang J, Chan HM. Sub-Micromolar Methylmercury Exposure Promotes Premature Differentiation of Murine Embryonic Neural Precursor at the Expense of Their Proliferation. Toxics. 2018; 6(4):61. https://doi.org/10.3390/toxics6040061
Chicago/Turabian StyleYuan, Xiaoyang, Jing Wang, and Hing Man Chan. 2018. "Sub-Micromolar Methylmercury Exposure Promotes Premature Differentiation of Murine Embryonic Neural Precursor at the Expense of Their Proliferation" Toxics 6, no. 4: 61. https://doi.org/10.3390/toxics6040061
APA StyleYuan, X., Wang, J., & Chan, H. M. (2018). Sub-Micromolar Methylmercury Exposure Promotes Premature Differentiation of Murine Embryonic Neural Precursor at the Expense of Their Proliferation. Toxics, 6(4), 61. https://doi.org/10.3390/toxics6040061




