Pattern of Paracetamol Poisoning: Influence on Outcome and Complications
Abstract
1. Introduction
2. Materials and Methods
2.1. Criteria for Liver Transplantation
2.2. Biomarkers
2.3. Statistical Analysis
2.4. Ethics and Consent
3. Results
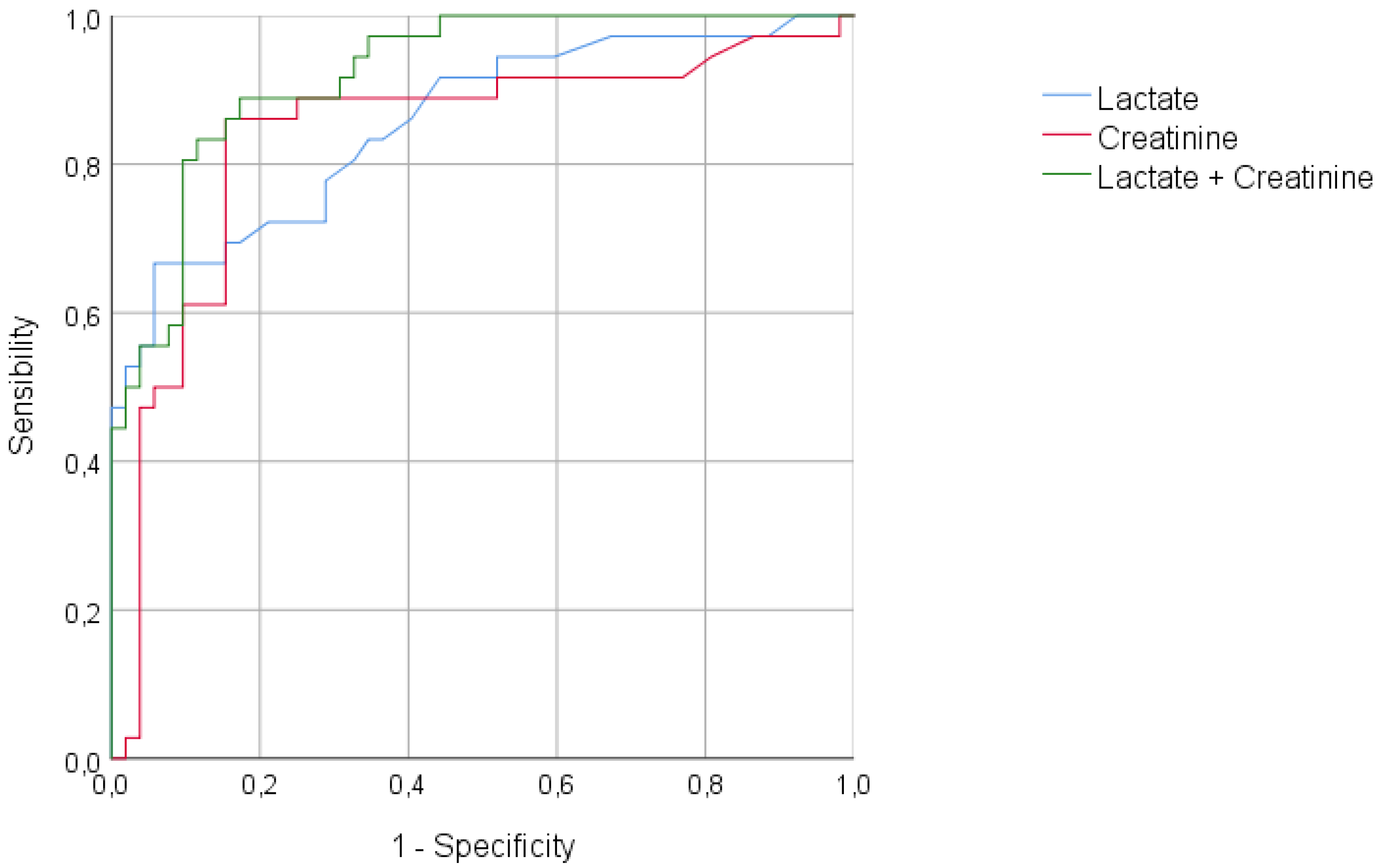
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Serper, M.; Wolf, M.S.; Parikh, N.A.; Tillman, H.; Lee, W.M.; Ganger, D.R. Risk Factors, Clinical Presentation, and Outcomes in Overdose with Acetaminophen Alone or With Combination Products: Results from the Acute Liver Failure Study Group. J. Clin. Gastroenterol. 2016, 50, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Caparrotta, T.M.; Antoine, D.J.; Dear, J.W. Are some people at increased risk of paracetamol-induced liver injury? A critical review of the literature. Eur. J. Clin. Pharmacol. 2018, 74, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Craig, D.G.; Bates, C.M.; Davidson, J.S.; Martin, K.G.; Hayes, P.C.; Simpson, K.J. Staggered overdose pattern and delay to hospital presentation are associated with adverse outcomes following paracetamol-induced hepatotoxicity. Br. J. Clin. Pharmacol. 2012, 73, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Rumack, B.H.; Matthew, H. Acetaminophen poisoning and toxicity. Pediatrics 1975, 55, 871–876. [Google Scholar] [PubMed]
- Bernal, W.; Donaldson, N.; Wyncoll, D.; Wendon, J. Blood lactate as an early predictor of outcome in paracetamol-induced acute liver failure: A cohort study. Lancet 2002, 359, 558–563. [Google Scholar] [CrossRef]
- Riordan, S.M.; Williams, R. Blood lactate and outcome of paracetamol-induced acute liver failure. Lancet 2002, 360, 573. [Google Scholar] [CrossRef]
- Smilkstein, M.J.; Knapp, G.L.; Kulig, K.W.; Rumack, B.H. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N. Engl. J. Med. 1988, 319, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Rumack, B.H. Acetaminophen hepatotoxicity: The first 35 years. J. Toxicol. Clin. Toxicol. 2002, 40, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Doyon, S.; Klein-Schwartz, W. Hepatotoxicity despite early administration of intravenous N-acetylcysteine for acute acetaminophen overdose. Acad. Emerg. Med. 2009, 16, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Prescott, L.F.; Illingworth, R.N.; Critchley, J.A.; Stewart, M.J.; Adam, R.D.; Proudfoot, A.T. Intravenous N-acetylcysteine: The treatment of choice for paracetamol poisoning. Br. Med. J. 1979, 2, 1097–1100. [Google Scholar] [CrossRef] [PubMed]
- Smilkstein, M.J.; Bronstein, A.C.; Linden, C.; Augenstein, W.L.; Kulig, K.W.; Rumack, B.H. Acetaminophen overdose: A 48-hour intravenous N-acetylcysteine treatment protocol. Ann. Emerg. Med. 1991, 20, 1064–1068. [Google Scholar] [CrossRef]
- Bateman, D.N.; Dear, J.W.; Carroll, R.; Pettie, J.; Yamamoto, T.; Elamin, M.E.; Peart, L.; Dow, M.; Coyle, J.; Gray, A.; et al. Impact of reducing the threshold for acetylcysteine treatment in acute paracetamol poisoning: The recent United Kingdom experience. Clin. Toxicol. 2014, 52, 868–872. [Google Scholar] [CrossRef] [PubMed]
- Cairney, D.G.; Beckwith, H.K.; Al-Hourani, K.; Eddleston, M.; Bateman, D.N.; Dear, J.W. Plasma paracetamol concentration at hospital presentation has a dose-dependent relationship with liver injury despite prompt treatment with intravenous acetylcysteine. Clin. Toxicol. 2016, 54, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.; Sivilotti, M.L.; Dargan, P.I.; Wood, D.M.; Greene, S.L. External validation of the paracetamol-aminotransferase multiplication product to predict hepatotoxicity from paracetamol overdose. Clin. Toxicol. 2015, 53, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Laskey, D.; Neavyn, M. APAP × AT as a hepatotoxicity predictor in patients with acetaminophen ingestions of chronic, subacute, or unknown time. Clin. Toxicol. 2015, 53, 677–678. [Google Scholar]
- Nacca, N.E.; Hodgman, M. APAP × AT in staggered, chronic and time unknown acetaminophen overdoses. Clin. Toxicol. 2015, 53, 391. [Google Scholar]
- Sivilotti, M.L.A.; Green, T.J.; Langmann, C.; Yarema, M.; Juurlink, D.; Johnson, D. Multiplying the serum aminotransferase by the acetaminophen concentration to predict toxicity following overdose. Clin. Toxicol. 2010, 48, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Al-Hourani, K.; Mansi, R.; Pettie, J.; Dow, M.; Bateman, D.N.; Dear, J.W. The predictive value of hospital admission serum alanine transaminase activity in patients treated for paracetamol overdose. Int. J. Med. 2013, 106, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Curtis, R.M.; Sivilotti, M.L. A descriptive analysis of aspartate and alanine aminotransferase rise and fall following acetaminophen overdose. Clin. Toxicol. 2015, 53, 849–855. [Google Scholar] [CrossRef] [PubMed]
- McGovern, A.J.; Vitkovitsky, I.V.; Jones, D.L.; Mullins, M.E. Can AST/ALT ratio indicate recovery after acute paracetamol poisoning? Clin. Toxicol. 2015, 53, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Gray, T.A.; Buckley, B.M.; Vale, J.A. Hyperlactataemia and metabolic acidosis following paracetamol overdose. Q. J. Med. 1987, 65, 811–821. [Google Scholar] [PubMed]
- Landry, D.W.; Oliver, J.A. The pathogenesis of vasodilatory shock. N. Engl. J. Med. 2001, 345, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Bradberry, S.M.; Hart, M.; Bareford, D.; Jones, A.F.; Vale, J.A. Factor V and factor VIII: V ratio as prognostic indicators in paracetamol poisoning. Lancet 1995, 346, 646. [Google Scholar] [CrossRef]
- Pereira, L.M.; Langley, P.G.; Hayllar, K.M.; Tredger, J.M.; Williams, R. Coagulation factor V and VIII/V ratio as predictors of outcome in paracetamol induced fulminant hepatic failure: Relation to other prognostic indicators. Gut 1992, 33, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Kucera, O.; Al-Dury, S.; Lotkova, H.; Rousar, T.; Rychtrmoc, D.; Cervin-kova, Z. Steatotic rat hepatocytes in primary culture are more susceptible to the acute toxic effect of acetaminophen. Physiol. Res. 2012, 61 (Suppl. 2), S93–S101. [Google Scholar]
- Nguyen, G.C.; Sam, J.; Thuluvath, P.J. Hepatitis C is a predictor of acute liver injury among hospitalizations for acetaminophen overdose in the United States: A nationwide analysis. Hepatology 2008, 48, 1336–1341. [Google Scholar] [CrossRef] [PubMed]
- Radosevich, J.J.; Patanwala, A.E.; Erstad, B.L. Hepatotoxicity in obese versus non-obese patients with acetaminophen poisoning who are treated with intravenous N-Acetylcysteine. Am. J. Ther. 2016, 23, e714–e719. [Google Scholar] [CrossRef] [PubMed]
- Rumack, B.H.; Peterson, R.C.; Koch, G.G.; Amara, I.A. Acetaminophen overdose. 662 cases with evaluation of oral acetylcysteine treatment. Arch. Intern. Med. 1981, 141, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Read, R.B.; Tredger, J.M.; Williams, R. Analysis of factors responsible for continuing mortality after paracetamol overdose. Hum. Toxicol. 1986, 5, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Denison, H.; Kaczynski, J.; Wallerstedt, S. Paracetamol medication and alcohol abuse: A dangerous combination for the liver andthe kidney. Scand. J. Gastroenterol. 1987, 22, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Makin, A.; Williams, R. Paracetamol hepatotoxicity and alcohol consumption in deliberate and accidental overdose. Int. J. Med. 2000, 93, 341–349. [Google Scholar] [CrossRef]
- Court, M.H.; Peter, I.; Hazarika, S.; Vasiadi, M.; Greenblatt, D.J.; Lee, W.M.; Acute Liver Failure Study Group. Candidate gene polymorphisms in patients with acetaminophen-induced acute liver failure. Drug Metab. Dispos. 2014, 42, 28–32. [Google Scholar] [CrossRef] [PubMed]
| Variables | Measure | Single Ingestion (n = 55) | Multiple Ingestion (n = 35) | p-Value |
|---|---|---|---|---|
| Age (years) | mean, SD | 33 ± 14 | 45 ± 12 | 0.001 |
| Gender (M/F) | n (%) | 20 M, 35 F | 17 M, 18 F | 0.25 |
| Outcome | ||||
| ICU length of stay (days) | mean, SD | 5.1 ± 1 | 6.9 ± 1.2 | 0.23 |
| Mortality | n (%) | 2 (3.6%) | 7 (20%) | 0.01 |
| Predisposing factors | ||||
| Steatosis (Y/N) | n (%) | 17 (31%) | 20 (57%) | 0.016 |
| Ethanol abuse (Y/N) | n (%) | 23 (42%) | 26 (74%) | 0.04 |
| Obesity (Y/N) | n (%) | 4 (7%) | 2 (6%) | 0.73 |
| Interventions–complications | ||||
| Timing to NAC (h) | n (%) | 12 h: 21 (38%) | 11 (31%) | 0.89 |
| (missing = 6) | 12–24 h: 19 (35%) | 13 (37%) | ||
| 24–48 h: 4 (7%) | 4 (11%) | |||
| >48 h: 7 (13%) | 5 (14%) | |||
| CRRT (Y/N) | n (%) | 12 (22%) | 13 (37%) | 0.12 |
| Vasopressors (Y/N) | n (%) | 8 (15%) | 11 (31%) | 0.06 |
| Mechanical ventilation (Y/N) | n (%) | 13 (24%) | 14 (40%) | 0.26 |
| Infection (Y/N) | n (%) | 11 (20%) | 12 (34%) | 0.14 |
| Indications for transplantation | ||||
| Encephalopathy | n (%) | 31 (56%) grade 0 | 14 (40%) | 0.34 |
| Grade (West-Haven) | 14 (25%) grade 1 | 10 (28%) | ||
| 1 (1.8%) grade 2 | 3 (9%) | |||
| 3 (5.4%) grade 3 | 4(11%) | |||
| 5 (9%) grade 4 | 4 (11%) | |||
| Criteria for transplantation (Y/N) | n (%) | 13 (24%) | 9 (26%) | 0.82 |
| Transplantation (Y/N) | n (%) | 5 (9%) | 2 (6%) | 0.7 |
| Biological markers | ||||
| Peak AST (IU/L) | median (P25–P75) | 9409 (5369–13,183) | 10,176 (6644–15,285) | 0.26 |
| Peak ALT (IU/L) | median (P25–P75) | 6250 (3110–9225) | 7269 (5391–10,905) | 0.14 |
| Arterial pH | median (P25–P75) | 7.38 (7.34–7.41) | 7.35 (7.25–7.40) | 0.006 |
| Bilirubin max (mg/dL) | median (P25–P75) | 9.7 (4.8–14) | 12.4 (4.9–16) | 0.047 |
| Creatinine max (mg/dL) | median (P25–P75) | 0.8 (0.6–1.5) | 2.18 (0.8–4.4) | 0.007 |
| Lactate max (mmol/L) | median (P25–P75) | 2.7 (1.8–6.5) | 3.3 (1.6–8.3) | 0.032 |
| Factor min V (%) | median (P25–P75) | 13 (4–26) | 18 (9–48) | 0.36 |
| Ammonia (µg/dL) | median (P25–P75) | 182 (126–302) | 186 (138–354) | 0.61 |
| AST/ALT ratio >2 | n (%) | 4 (7) | 9 (26) | 0.017 |
| t½ AST (h) | median (P25–P75) | 39.6 (34.8–45.6) | 38.4 (32.4–46.8) | 0.58 |
| t½ ALT (h) | median (P25–P75) | 15.6 (14.4–20.4) | 18 (14.4–25.2) | 0.23 |
| ny × AT (mg/L × IU/L) | median (P25–P75) | 42,476 (23,746–93,680) | 112,083 (34,926–270,910) | 0.4 |
| Time to peak ALT (h) | median (P25–P75) | 60 (48–78) | 42 (24–51) | 0.006 |
| AUC AST (IU·h·L–1) | median (P25–P75) | 358,404 (230,076–527,568) | 379,842 (230,521–726,090) | 0.73 |
| AUC ALT (IU·h·L–1) | median (P25–P75) | 458,160 (287,336–583,344) | 267,012 (115,720–470,274) | 0.04 |
| Variables (Median) | Time to NAC 12 h | Time to NAC 12–24 h | Time to NAC 24–48 h | Time to NAC >48 h | p-Value |
|---|---|---|---|---|---|
| (n = 32) | (n = 32) | (n = 8) | (n = 12) | ||
| Bilirubin max (mg/dL) | 8.5 | 4.7 | 5 | 6.3 | 0.86 |
| Peak AST (IU/L) | 13,255 | 10,150 | 11,975 | 10,176 | 0.47 |
| Peak ALT (IU/L) | 7520 | 699 | 10,075 | 7050 | 0.16 |
| APAP × AT (mg/L × IU/L) | 48,538 | 58,658 | 29,520 | 70,403 | 0.96 |
| Factor V min V (%) | 15 | 12 | 13 | 41 | 0.1 |
| Peak ammonia (µg/dL) | 227 | 185 | 225 | 202 | 0.9 |
| Arterial pH min | 7.36 | 7.37 | 7.4 | 7.35 | 0.44 |
| Lactate max (mmol/L) | 3.5 | 3.3 | 2 | 2.9 | 0.78 |
| Creatinine max (mg/dL) | 1.04 | 1.43 | 1.13 | 1.85 | 0.36 |
| Need for vasopressors | 11 (34%) | 8 (25%) | 1 (12%) | 0 | 0.1 |
| CRRT | 8 (25%) | 10 (31%) | 2 (25%) | 4 (33%) | 0.92 |
| Mechanical ventilation | 11 (34%) | 11 (34%) | 1 (12%) | 5 (41%) | 0.58 |
| Infection | 11 (34%) | 5 (15%) | 3 (37%) | 4 (33%) | 0.33 |
| Mortality | 6 (18%) | 3 (9%) | 0 | 0 | 0.2 |
| Variables | Good Outcome (n = 54) | Bad Outcome (n = 36) | p-Value |
|---|---|---|---|
| Age (year) | 36 ± 11 | 42 ± 16 | 0.005 |
| Peak AST (IU/L) | 10430 ± 5227 | 12385 ± 7000 | 0.045 |
| Peak ALT (IU/L) | 8022 ± 3683 | 8109 ± 4782 | 0.81 |
| Creatinine max (mg/dL) | 2.2 (0.6–1.1) | 3.2 (1.5–5.2) | <0.001 |
| Bilirubin max (mg/dL) | 3.6 (2.4–6.4) | 7.5 (4.5–13.5) | <0.001 |
| Factor V min (%) | 17 (12–33) | 10 (4–33) | 0.021 |
| Ammonia max (µg/dL) | 161 (113–335) | 264 (175–370) | 0.001 |
| Arterial pH min | 7.4 (7.37–7.42) | 7.32 (7.24–7.35) | <0.001 |
| Lactate max (mmol/L) | 2 (1.4–3) | 6.8 (2.9–14.5) | <0.001 |
| AST/ALT ratio >2 | 5 (9) | 9 (25) | 0.044 |
| t½ AST (h) | 42 (36–48) | 34.8 (27.6–39.6) | 0.005 |
| t½ ALT (h) | 15.2 (14–18.3) | 18.3 (16–25.2) | 0.001 |
| APAP × AT (mg/L × IU/L) | 31,414 (14,214–69,556) | 79,990 (41,184–368,301) | 0.001 |
| Time to peak ALT (h) | 60 (48–72) | 60 (36–72) | 0.11 |
| AUC AST (IU·h·L–1) | 354,324 (223,708–525,993) | 388,512 (247,092–591,492) | 0.11 |
| AUC ALT (IU·h·L–1) | 469,674 (290,662–539,460) | 409,464 (191,958–654,102) | 0.78 |
| Variables | Odds Ratio (95CI) | p-Value | Adjusted Odds Ratio (95CI) | p-Value |
|---|---|---|---|---|
| Lactate max | 1.6 (1.26–2.2) | <0.001 | 1.74 (1.09–2.77) | 0.02 |
| Bilirubin max | 1.61 (1.05–1.28) | 0.002 | ||
| Creatinine max | 1.71 (1.29–2.27) | <0.001 | 1.45 (1.05–1.97) | 0.025 |
| Single ingestion | 0.36 (0.14–0.86) | 0.022 | ||
| Age | 1.05 (1.01–1.08) | 0.007 | ||
| Gender (M) | 1.17 (0.49–2.76) | 0.72 | ||
| Factor V min | 0.98 (0.96–1.01) | 0.11 | ||
| Ammonia max | 1.01 (1.002–1.01) | 0.003 | 1.01 (0.99–1.02) | 0.085 |
| Alcohol | 1.21 (0.51–2.82) | 0.66 | ||
| Steatosis | 2.10 (0.88–4.87) | 0.1 | ||
| LOG peak AST * | 3.76 (0.96–14.7) | 0.06 | ||
| LOG peak ALT * | 1.14 (0.99–3.34) | 0.81 | ||
| AST/ALT >2 | 3.26 (0.99–10.7) | 0.05 | ||
| t½ AST | 0.56 (0.34–0.93) | 0.026 | ||
| t½ ALT | 3.89 (1.45–10.4) | 0.007 | ||
| LOG APAP * | 3.98 (1.77–8.92) | 0.001 | ||
| Time to peak ALT | 0.98 (0.96–1.004) | 0.12 | ||
| LOG AUC AST * | 1.6 (0.71–3.56) | 0.25 | ||
| LOG AUC ALT * | 1.04 (0.52–2.09) | 0.91 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castanares-Zapatero, D.; Dinant, V.; Ruggiano, I.; Willem, H.; Laterre, P.-F.; Hantson, P. Pattern of Paracetamol Poisoning: Influence on Outcome and Complications. Toxics 2018, 6, 58. https://doi.org/10.3390/toxics6040058
Castanares-Zapatero D, Dinant V, Ruggiano I, Willem H, Laterre P-F, Hantson P. Pattern of Paracetamol Poisoning: Influence on Outcome and Complications. Toxics. 2018; 6(4):58. https://doi.org/10.3390/toxics6040058
Chicago/Turabian StyleCastanares-Zapatero, Diego, Valérie Dinant, Ilaria Ruggiano, Harold Willem, Pierre-François Laterre, and Philippe Hantson. 2018. "Pattern of Paracetamol Poisoning: Influence on Outcome and Complications" Toxics 6, no. 4: 58. https://doi.org/10.3390/toxics6040058
APA StyleCastanares-Zapatero, D., Dinant, V., Ruggiano, I., Willem, H., Laterre, P.-F., & Hantson, P. (2018). Pattern of Paracetamol Poisoning: Influence on Outcome and Complications. Toxics, 6(4), 58. https://doi.org/10.3390/toxics6040058





