Metal Levels in Blood of Three Species of Shorebirds during Stopover on Delaware Bay Reflect Levels in Their Food, Horseshoe Crab Eggs
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
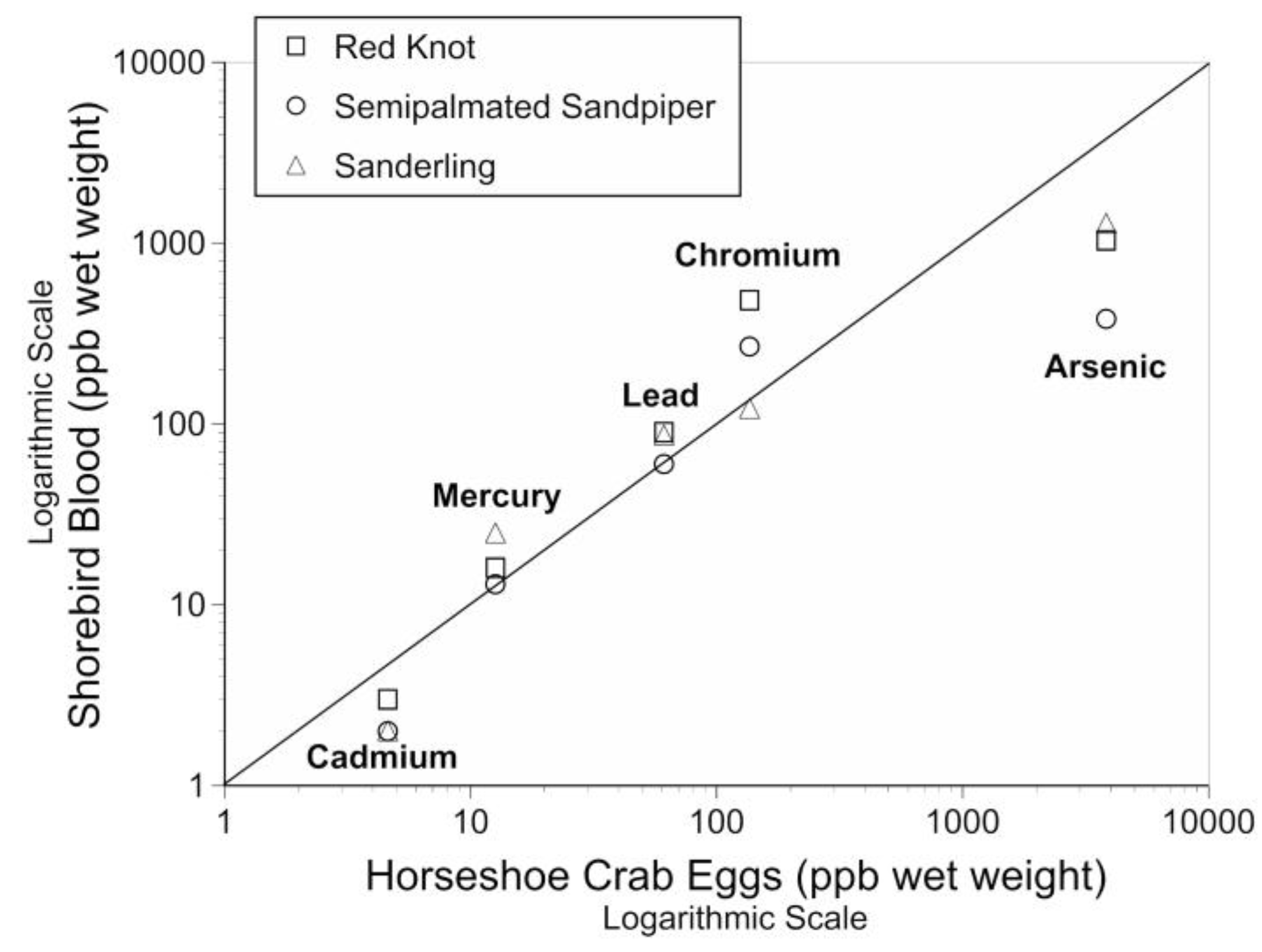
4.1. Relationship between Metals in Prey and in Shorebird Blood
4.2. Species Differences
4.3. Ratio of Metal Levels in Blood/Feathers for Shorebirds
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nygard, T.; Lie, E.; Roy, N.; Steinnes, E. Metal dynamics in an Antarctic food chain. Mar. Pollut. Bull. 2001, 42, 598–602. [Google Scholar] [CrossRef]
- Kim, J.; Koo, T.J. The use of feathers to monitor heavy metal contamination in herons, Korea. Arch. Environ. Contam. Toxicol. 2007, 53, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.; Gochfeld, M. Habitat, Population Dynamics, and Metal Levels in Colonial Waterbirds: A Food Chain Approach; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Furness, R.W.; Rainbow, P.S. Heavy Metals in the Marine Environment; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Burger, J.; Gochfeld, M. Effects of chemicals and pollution on seabirds. In Biology of Marine Birds; Schreiber, B.A., Ed.; CRC Press: Boca Raton, FL, USA, 2001; pp. 485–576. [Google Scholar]
- Fitzgerald, W.F.; Engstrom, D.R.; Lamborg, C.J.; Tseng, C.M.; Balcom, P.H.; Hamerschmidt, C.R. Modern and historic atmospheric mercury fluxes in northern Alaska: Global sources and Arctic depletion. Environ. Sci. Technol. 2005, 39, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Hammerschmidt, C.F.; Fitzgerald, W.F.; Lamborg, C.H.; Balcom, P.H.; Tseng, C.M. Biogeochemical cycling of methylmercury in lakes and tundra watersheds of Arctic Alaska. Environ. Technol. Sci. 2006, 40, 1204–1211. [Google Scholar] [CrossRef]
- Burger, J. Shorebirds as marine animals. In Behavior of Marine Animals; Burger, J., Olla, E.J., Eds.; Plenum Press: New York, NY, USA, 1984; Volume 5, pp. 17–81. [Google Scholar]
- Myers, J.P.; Morrison, R.I.G.; Antas, P.Z.; Harrington, B.A.; Lovejoy, T.E.; Sallaberry, M.; Senner, S.E.; Tarak, A. Conservation strategy for migratory species. Am. Sci. 1987, 75, 19–26. [Google Scholar]
- Niles, L.J.; Sitters, H.P.; Dey, A.D.; Arce, N.; Atkinson, P.W.; Baker, A.J.; Bennett, K.A.; Buchanan, J.; Carmona, R.; Harrington, B.A.; et al. Status of the Red Knot, Calidris canutus rufa, in the Western Hemisphere. Stud. Avian Biol. 2008, 36, 1–185. [Google Scholar]
- Niles, L.J.; Bart, J.; Sitters, H.P.; Dey, A.D.; Clark, K.E.; Aktinson, P.W.; Baker, A.J.; Bennett, K.A.; Kalasz, K.S.; Clark, N.A.; et al. Effects of horseshoe crab harvest in Delaware Bay on Red Knots: Are harvest restrictions working? BioScience 2009, 59, 153–164. [Google Scholar] [CrossRef]
- Burger, J.; Niles, L.J.; Porter, R.; Dey, A.; Koch, S.; Gordon, D. Migration and overwintering of Red Knots (Calidris canutus rufa) along the Atlantic coast of the United States. Condor 2012, 114, 302–313. [Google Scholar]
- Morrison, R.E.G.; Aubrey, Y.; Butler, R.W.; Beyersbergen, G.W.; Donaldson, G.M.; Gratto-Trevor, C.L.; Hicklin, P.W.; Johnson, V.H.; Ross, R.K. Declines in North American shorebird populations. Wader Study Group Bull. 2001, 94, 37–42. [Google Scholar]
- Mizrahi, D.; Peters, K. Relationship between sandpipers and horseshoe crabs in Delaware Bay: A synthesis. In Biology and Conservation of Horseshoe Crabs; Tanacredi, J.T., Botton, M.L., Smith, D.R., Eds.; Springer: New York, NY, USA, 2009; pp. 65–88. [Google Scholar]
- Simmons, R.E.; Kolberg, H.; Brady, R.; Erni, B. Declines in migrant shorebird populations from a winter-quarter perspective. Conserv. Biol. 2015, 29, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Piersma, T.; Wiersma, P.; Gills, J. The many unknowns about plovers and sandpipers of the world: Introduction to a wealth of research opportunities highly relevant for shorebird conservation. Wader Study Group Bull. 1997, 82, 23–33. [Google Scholar]
- Andres, B.A.; Smith, P.A.; Morrison, R.G.; Gratto-Trevor, E.L.; Brown, S.C.; Friis, C.A. Population estimates of North American shorebirds. Wader Study Group Bull. 2012, 119, 178–194. [Google Scholar]
- Brown, S.; Gratto-trevor, C.; Porter, R.; Weiser, E.L.; Mizrahi, D.; Bentzen, R.; Boldenow, M.; Clay, R.; Freeman, S.; Girous, A.-A.; et al. Migratory connectivity of semipalmated sandpipers and implications for conservation. Condor 2017, 119, 207–224. [Google Scholar] [CrossRef]
- Baker, A.J.; Gonzalex, P.; Morrison, R.I.G.; Harrington, B.A. Red Knot (Calidris canutus). In The Birds of North America; Poole, A., Ed.; Online Cornell Lab of Ornithology: Ithaca, NY, USA, 2013. [Google Scholar]
- Western Hemisphere Shorebird Reserve Network (WHSRN) Western Hemisphere Shorebird Reserve Network List of Sites. Available online: http://www.whsrn.org/sites/map-sites/sites-western-hemisphere-shorebird-reserve-network (accessed on 23 August 2017).
- Clark, K.E.; Niles, L.J.; Burger, J. Abundance and distribution of migrant shorebirds in Delaware Bay. Condor 1993, 95, 694–705. [Google Scholar] [CrossRef]
- Tsipoura, N.; Burger, J. Shorebird diet during spring migration stop-over on Delaware Bay. Condor 1999, 101, 635–644. [Google Scholar] [CrossRef]
- Brown, S.; Hickey, C.; Harrington, B.; Gill, R. United States Shorebird Conservation Plan; Manomet Center for Conservation Sciences: Manomet, MA, USA, 2001. [Google Scholar]
- Walls, E.A.; Berkson, J.; Smith, S.A. The horseshoe crab, Limulus Polyphemus: 200 million years of existence, 100 years of study. Rev. Fish Sci. 2002, 10, 39–73. [Google Scholar] [CrossRef]
- Shuster, C.N., Jr.; Botton, M.L. A contribution to the population biology of horseshoe crabs, Limulus polyphemus (L.), in Delaware Bay. Estuaries 1985, 8, 363–372. [Google Scholar] [CrossRef]
- Botton, M.L.; Loveland, R.E.; Jacobsen, T.R. Site selection by migratory shorebirds in Delaware Bay, and its relationship to beach characteristics and abundance of horseshoe crab (Limulus polyphemus) eggs. Auk 1994, 111, 605–616. [Google Scholar]
- Botton, M.L.; Loveland, R.E. Updating the life history of the Atlantic horseshoe crab, Limulus polyphemus. Jersey Shorel. 2001, 20, 6–9. [Google Scholar]
- Dunne, P.; Sibley, D.; Sutton, C.; Wander, W. Aerial surveys in Delaware Bay: Confirming an enormous spring staging area for shorebirds. Wader Study Group Bull. 1982, 35, 32–33. [Google Scholar]
- Burger, J.; Niles, L. Shorebirds, stakeholders, and competing claims to the beach and intertidal habitat in Delaware Bay, New Jersey, USA. Nat. Sci. 2017, 9, 181–205. [Google Scholar] [CrossRef]
- Kraemer, G.; Michels, S. History of horseshoe crab harvest in Delaware Bay. In Biology and Conservation of Horseshoe Crabs; Tanacredi, J.T., Botton, M.L.A., Smith, D.R., Eds.; Springer: New York, NY, USA, 2009; pp. 299–313. [Google Scholar]
- McGowan, C.P.; Hines, J.E.; Nichols, J.D.; Lyons, J.E.; Smith, J.E.; Smith, D.R.; Kalasz, K.S.; Niles, L.J.; Dey, A.D.; Clark, N.A.; et al. Demographic consequences of migratory stopover: Linking red knot survival to horseshoe crab spawning abundance. Ecosphere 2011, 2, 11–22. [Google Scholar] [CrossRef]
- U.S. Fish & Wildlife Service (USFWS). Migratory Bird Treaty Act Protected Species (10.13 Lists) as of December 2, 2017. USFWS. Available online: https://www.fws.gov/birds/management/managed-species/migratory-bird-treaty-act-protected-species.php (accessed on 23 August 2017).
- U.S. Fish & Wildlife Service (USFWS). Programmatic Biological Assessment for Structural Shellfish Aquaculture Projects for Red Knot (Calidris canutus rufa); USFWS: Lower Delaware Bay, NJ, USA, 2016.
- Baker, A.J.; Gonzalez, P.M.; Piersma, T.; Niles, L.J.; de Lima, I.; Nascimento, S.; Atkinson, P.W.; Collins, P.; Clark, N.A.; Minton, C.D.T.; et al. Rapid population decline in red knots: Fitness consequences of refuelling rates and late arrival in Delaware Bay. Proc. R. Soc. Lond. 2004, 271, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Morrison, R.I.G.; Davidson, N.C.; Wilson, J.R. Survival of the fattest body stores on migration and survival in Red Knot, Calidris canutus islandica. J. Avian Biol. 2007, 38, 479–487. [Google Scholar] [CrossRef]
- Burger, J.; Gochfeld, M.; Niles, L.; Dey, A.; Jeitner, C.; Pittfield, T.; Tsipoura, N. Metals in tissues of migrant semipalmated sandpipers (Calidris pusilla) from Delaware Bay, New Jersey. Environ. Res. 2014, 133, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.; Tsipoura, N.; Niles, L.J.; Gochfeld, M.; Dey, A.; Mizrahi, D. Mercury, lead, cadmium, arsenic, chromium and selenium in feathers of shorebirds during migrating through Delaware Bay, New Jersey: Comparing the 1990s and 2011/2012. Toxics 2015, 3, 63–74. [Google Scholar] [CrossRef]
- Tsipoura, N.; Burger, J.; Niles, L.; Dey, A.; Gochfeld, M.; Peck, M.; Mizrahi, D. Metal levels in shorebird feathers and blood during migration through Delaware Bay. Arch. Environ. Contam. Toxicol. 2017, 72, 562–574. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.; Tsipoura, N. Metals in horseshoe crab eggs from Delaware Bay, USA: Temporal patterns from 1993–2012. Environ. Monit. Assess. 2014, 186, 6947–6958. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, M.; Norman, D. Effects of waterborne mercury on terrestrial wildlife at Clear Lake: Evaluation and testing of a predictive model. Environ. Toxicol. Chem. 1998, 17, 214–227. [Google Scholar] [CrossRef]
- SAS. Statistical Analysis System; SAS Institute: Cary, NC, USA, 2005. [Google Scholar]
- Monteiro, L.R.; Furness, R.W. Seabirds as monitors of mercury in the marine environment. Water Air Soil Pollut. 1995, 80, 851–870. [Google Scholar] [CrossRef]
- Evers, D.C.; Burgess, N.M.; Champous, L.; Hoskins, B.; Major, A.; Goodale, W.M.; Robert, J.T.; Poppenga, R.; Daigle, T. Patterns and interpretation of mercury exposure in freshwater avian communities in northeastern North America. Ecotoxicology 2005, 14, 193–221. [Google Scholar] [CrossRef]
- Burger, J.; Dixon, C.; Shukla, T.; Tsipoura, N.; Gochfeld, M. Metal levels in horseshoe crabs (Limulus polyphemus) from Maine to Florida. Environ. Res. 2002, 90, 227–236. [Google Scholar] [CrossRef]
- Mizrahi, D.S. Importance of soft-sediment habitat to migrating shorebirds staging in Delaware Bay during spring migration. In Final Report to National Fish and Wildlife Foundation; National Fish and Wildlife Foundation: Washington, DC, USA, 2013. [Google Scholar]
- Eisler, R. Mercury hazards to fish, wildlife and invertebrates: A synoptic review. In U.S. Fish Wildlife Service Report 85; U.S. Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1987. [Google Scholar]
- Eisler, R. Lead hazards to fish, wildlife and invertebrates: A synoptic review. In U.S. Fish Wildlife Service Report 85; U.S. Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1988. [Google Scholar]
- Burger, J. Metals in avian feathers: Bioindicators of environmental pollution. Rev. Environ. Toxicol. 1993, 5, 197–306. [Google Scholar]
- Furness, R.W.; Muirhead, S.J.; Woodburn, M. Using bird feathers to measure mercury in the environment: Relationship between mercury content and moult. Mar. Pollut. Bull. 1986, 17, 27–30. [Google Scholar] [CrossRef]
- Frederick, P.C.; Hylton, B.; Health, J.A.; Spalding, M.G. A historical record of mercury contamination in Florida, USA as inferred from avian feather tissue. Environ. Toxicol. Chem. 2004, 23, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, P.W.; Baker, A.J.; Bevan, R.M.; Clark, N.A.; Cole, K.B.; Gonzalez, P.M.; Newton, J.; Niles, L.J.; Robinson, R.A. Unravelling the migration and moult strategies of a long-distance migrant using stable isotopes: Red Knot Calidris canutus movement in the Americas. Ibis 2005, 147, 738–749. [Google Scholar] [CrossRef]
- Lourenco, P.M.; Piersma, T. Migration distance and breeding latitude correlate with the scheduling of pre-alternate body moult: A comparison among migratory waders. J. Ornithol. 2015, 156, 657–665. [Google Scholar] [CrossRef]
| Arsenic | Cadmium | Chromium | Lead | Mercury | |
|---|---|---|---|---|---|
| Red Knot | |||||
| Blood (n = 30) | 1036 ± 188 | 2.9 ± 0.7 | 484 ± 62 | 90 ± 12 | 16 ± 3.1 |
| Feather (n = 30) | 446 ± 42 | 17 ± 2.4 | 578 ± 83 | 484 ± 67 | 576 ± 105 |
| Ratio | 2.32 | 0.17 | 0.84 | 0.19 | 0.03 |
| Sanderling | |||||
| Blood (n = 20) | 1288 ± 193 | 2 ± 0.7 | 122 ± 21 | 87 ± 14 | 25 ± 5.3 |
| Feather (n = 20) | 311 ± 64 | 10.5 ± 2.6 | 463 ± 63 | 367 ± 52 | 730 ± 109 |
| Ratio | 4.14 | 0.19 | 0.26 | 0.24 | 0.03 |
| Semipalmated sandpiper | |||||
| Blood (n = 38) | 381 ± 45 | 1.8 ± 0.5 | 268 ± 52 | 59.8 ± 11 | 12.7 ± 3.3 |
| Feather (n = 30) | 842 ± 101 | 14.2 ± 2.7 | 524 ± 64 | 411 ± 46 | 428 ± 58 |
| Ratio | 0.45 | 0.13 | 0.51 | 0.15 | 0.03 |
| Interspecific Differences (X2) | |||||
| Blood | 31.9 (<0.0001) | 6.7 (0.04) | 27.9 (<0.0001) | 5.0 (0.08) | 3.0 (NS) |
| Feathers | 25.4 (<0.0001) | 2.9 (NS) | 0.7 (NS) | 0.8 (NS) | 5.1 (0.08) |
| Ratio | 37.9 (<0.0001) | 1.4 (NS) | 13.8 (0.001) | 3.4 (NS) | 0.09 (NS) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burger, J.; Tsipoura, N.; Gochfeld, M. Metal Levels in Blood of Three Species of Shorebirds during Stopover on Delaware Bay Reflect Levels in Their Food, Horseshoe Crab Eggs. Toxics 2017, 5, 20. https://doi.org/10.3390/toxics5030020
Burger J, Tsipoura N, Gochfeld M. Metal Levels in Blood of Three Species of Shorebirds during Stopover on Delaware Bay Reflect Levels in Their Food, Horseshoe Crab Eggs. Toxics. 2017; 5(3):20. https://doi.org/10.3390/toxics5030020
Chicago/Turabian StyleBurger, Joanna, Nellie Tsipoura, and Michael Gochfeld. 2017. "Metal Levels in Blood of Three Species of Shorebirds during Stopover on Delaware Bay Reflect Levels in Their Food, Horseshoe Crab Eggs" Toxics 5, no. 3: 20. https://doi.org/10.3390/toxics5030020
APA StyleBurger, J., Tsipoura, N., & Gochfeld, M. (2017). Metal Levels in Blood of Three Species of Shorebirds during Stopover on Delaware Bay Reflect Levels in Their Food, Horseshoe Crab Eggs. Toxics, 5(3), 20. https://doi.org/10.3390/toxics5030020







