Atrazine Exposure and Reproductive Dysfunction through the Hypothalamus-Pituitary-Gonadal (HPG) Axis
Abstract
:1. Introduction
2. The Effects of Atrazine on Female Reproductive Function in Mammalian Models
| Reference | Species | Exposure | Duration | Outcomes |
|---|---|---|---|---|
| In vitro | ||||
| Pogrmic-Majkic et al. [28] | Rat granulosa cells | 0 or 20 μM | 48 h | Increase in progesterone; Increase in progesterone/estradiol ratio; Increase in Star and Cyp11a1; Activation of AKT and CREB |
| Fa et al. [29] | Rat granulosa cells | 0, 10, or 20 μM | 12 or 48 h | Increase in estradiol in 8-Br-cAMP stimulated granulosa cells; Decrease in Cyp11a1 and Lhr expression; Decrease in ovulatory gene expression (Areg, Ereg, PgR) |
| Basini et al. [30] | Swine granulosa cells | 0, 0.1, or 10 μM | 48 h | Decrease in estradiol at 0.1 μM; Increase in progesterone at 10 μM; Increase in VEGF at 0.1 and 10 μM; Increase in NO at 10 μM |
| Tinfo et al. [31] | Granulosa cells and H295R adrenal cortical carcinoma cells | 0 or 10 μM | 24 h | Increased estradiol and aromatase activity after 24 h in granulosa cells; Increased estradiol and estrone in H295R cells; Increase in progesterone in both cell types |
| Holloway et al. [32] | Human granulosa and endometrial stromal cells | 0, 1 nM, or 1 μM | 24 h | Increase in aromatase activity in granulosa cell cultures; No alterations in aromatase protein or proportion of cells expressing aromatase |
| Sanderson et al. [33] | H295R adrenal cortical carcinoma cells | 0–30 μM | 24 h | Increase in CYP19A1 activity; Increase in CYP19 mRNA |
| Gestational | ||||
| Davis et al. [34] | Sprague-Dawley rats | 0, 1, 5, 20, or 100 mg/kg/day | GD 14–21 | Delay in vaginal opening at 100 mg/kg/day; No alterations in mammary gland development on PND 45; No alterations in estrous cyclicity through PND 272 |
| Hovey et al. [35] | Long-Evans rats | 0, 6.5, 50, or 100 mg/kg/day | GD 13–19 | No effects on mammary gland development beyond a transitory response to high doses at PND1 |
| Rayner et al. [36] | Long-Evans rats | 0 or 100 mg/kg/day | GD 13–15 or 15–17 or 17–19 or 13–19 | Delay in vaginal opening in the GD 13–15 treatment group; No alterations in estrous cyclicity from PND 37–67; Decrease in area of mammary gland as soon as PND 4; Delays in development of mammary glands through PND 67; No alterations in serum hormone levels |
| Rayner et al. [37] | Long-Evans rats | 0 or 100 mg/kg/day | GD 15–19 | Delay in age vaginal opening; Delays in mammary gland epithelial development; No alterations in uterine or ovarian weight; No alterations in estrous cyclicity or serum hormone levels |
| Peripubertal | ||||
| Ashby et al. [38] | AP and Sprague-Dawley rats | 0, 10, 30, or 100 mg/kg/day | PND 21 up to PND 46 | Decrease in uterine weight at 100 mg/kg/day at PND 30 and PND 33; Delay in age of vaginal opening |
| Laws et al. [39] | Wistar rats | 0, 12.5, 25, 50, 100, or 200 mg/kg/day | PND 22- PND 41 | Delay in VO in the 50, 100, and 200 mg/kg/day treatment groups; Decreases in pituitary, ovary, uterine, adrenal, and kidney weight in 200 mg/kg/day at PND 41; No alterations in T3, T4, or TSH; Alterations in estrous cycle in first 15 days following VO at 100 mg/kg/day; No alterations in estrous cyclicity from PND 57–149 |
| Adult | ||||
| Foradori et al. [40] | Sprague Dawley and Long-Evans rats | 0.75–100 mg/kg/day oral gavage: 300–1460 ppm diet | 4 days | Decrease in LH surge and area under the curve in Sprague-Dawley rats treated by oral gavage; Decrease in mean number of corpora lutea and number of ova in Sprague-Dawley rats treated by oral gavage; No change with dietary consumption |
| Goldman et al. [41] | Long Evans rats | 0, 10, 30, or 100 mg/kg/day | 1 or 4 days | Decrease in LH surge; Increase in serum progesterone; Increase in Kiss1 mRNA expression |
| Foradori et al. [42] | Wistar rats | 0, 50, 100, or 200 mg/kg/day | 4 days | No alterations in gene expression, peptide levels, or immunoreactivity; Reduction observed in GnRH pulse frequency; Increase in GnRH pulse amplitude |
| Quignot et al. [43] | Sprague-Dawley rats | 0 or 200 mg/kg/day | 14 days | Decrease in uterine and ovary weight; Increase in estrone and estradiol; Increase in ovarian aromatase expression; Increase in estrous cycle; Alterations in steroidogenic genes |
| Taketa et al. [44] | Sprague-Dawley rats | 0 or 300 mg/kg/day | 4 days or 2 weeks | Abnormal estrus cycle (persistent diestrus); Luteal cell hypertrophy and atretic follicles observed; Increase in serum progesterone; Increase in steroidogenic gene SR-B1 |
| Foradori et al. [45] | Wistar rats | 0, 50, 100, or 200 mg/kg/day | 4 days | Reduction in magnitude of LH and FSH surges; Decrease in GnRH neurons; Measures of HPG activation return to normal 4 days after cessation |
| Foradori et al. [46] | Wistar rats | 0, 50, 100, or 200 mg/kg/day | 4 days | Decrease in LH pulse frequency and increase in pulse period and pulse amplitude in 200 mg/kg/day treatment group |
| Shibayama et al. [47] | Sprague-Dawley rats | 0, 3, 30, or 300 mg/kg/day | 2 or 4 weeks | Loss of corpora lutea; Increase in atretic follicles; Swelling of leuteal cells; Prolonged estrous cycle; Decrease in ovarian and uterine weight |
| Juliani et al. [48] | Wistar rats | 0, 0.75, or 400 mg/kg/day | 14 (0.75 mg/kg) or 30 days (400 mg/kg) | No increases in atretic antral follicles, but intensity level of apoptosis in granulosa cells was visibly higher than control in the 0.75 mg/kg/day group; Disorganized granulosa cells, discontinuous zona pellucida, high intensity of apoptosis in atretic antral follicles in the 400 mg/kg/day treatment group |
| McMullin et al. [49] | Sprague-Dawley rats | 0, 30, 100, or 300 mg/kg/day | 5 days | Decrease in LH at 30, 100, and 300 mg/kg/day; No binding to estrogen receptor in vivo |
| Cooper et al. [50] | Sprague-Dawley and Long-Evans rats | 0, 50, 100, 200, or 300 mg/kg/day | 1, 3, or 21 days | Suppression of serum LH and PRL; Increase in pituitary LH |
2.1. Gestational Atrazine Exposure in Females
2.2. Peripubertal Atrazine Exposure in Females
2.3. Adulthood Atrazine Exposure in Females
2.4. Cellular and Genetic Mechanisms of Reproductive Dysfunction in Females
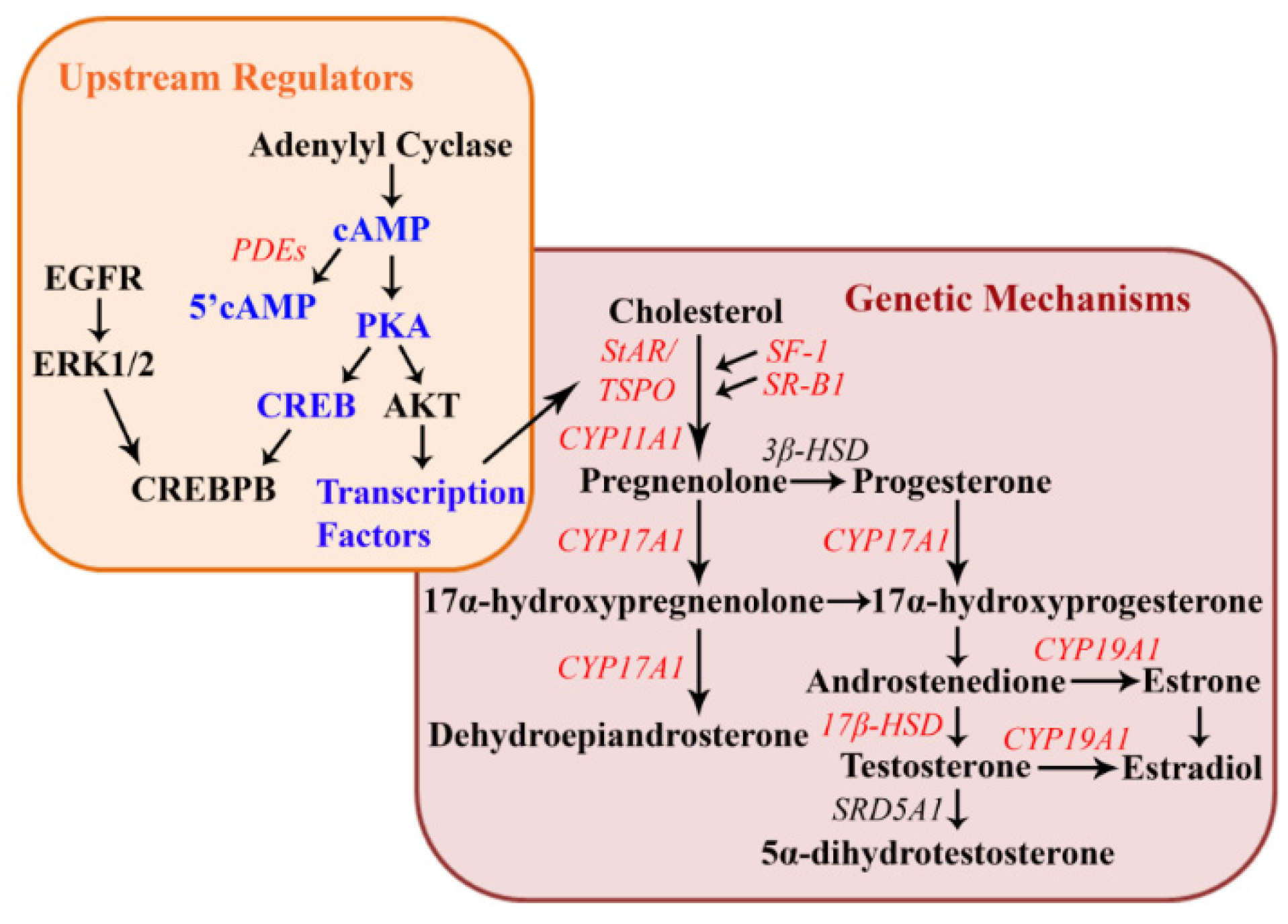
2.5. Tissue Levels of Atrazine in Females
2.6. Conclusions
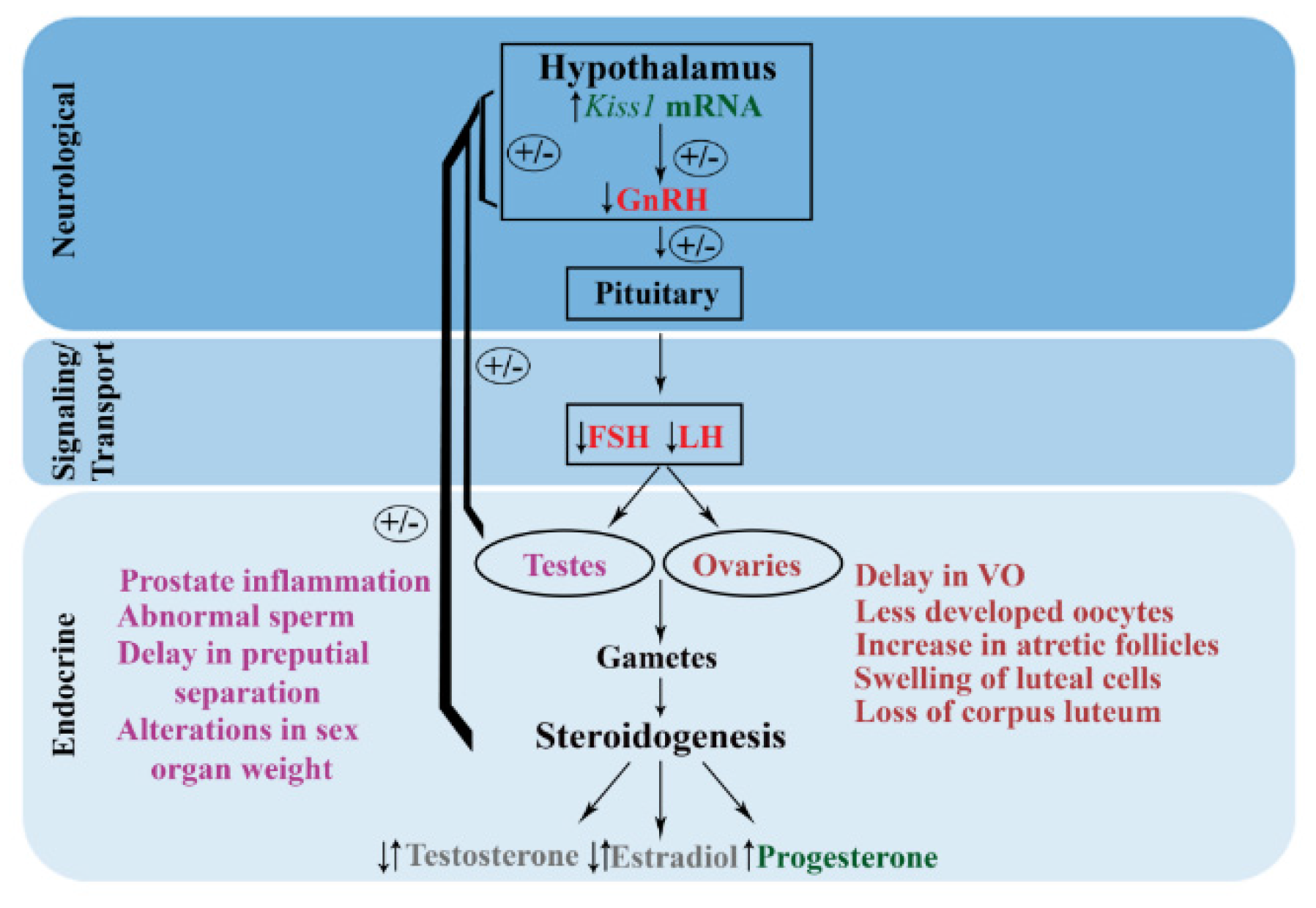
3. The Effects of Atrazine on Male Reproductive Function in Mammalian Models
| Reference | Species | Exposure | Duration | Outcomes |
|---|---|---|---|---|
| In vitro | ||||
| Forgacs et al. [74] | BLTK1 Murine Leydig cells | 1, 3, 10, 30, 100, 300, or 600 μM | 12, 24, or 48 h | Increase in progesterone and testosterone; Alterations in steroidogenic genes |
| Kucka et al. [70] | Sprague-Dawley anterior pituitary and Leydig cells | 10–50 μM | Various time points | Increase in cAMP and prolactin in pituitary cells; Inhibition of PDE4 isoenzymes |
| Abarikwu et al. [75] | Wistar rat Leydig cells | Cytotoxic studies 0.5, 5, 10, 25, or 50 μg/mL | Cytotoxic studies 24–72 h; Gene expression studies 2 h | Decrease in cell viability at 48 and 72 h at 25 and 50 μM; Alterations in steroidogenic gene expression |
| Gestational | ||||
| Stanko et al. [76] | Long-Evans rats | 100 mg/kg/day | GD 15–19 | No body weight alterations; Delay in preputial separation; Increase in testosterone and decrease in estrone at PND 120; Decrease in estradiol at PND 180 |
| Rosenberg et al. [77] | Sprague-Dawley rats | 0, 10, 50, 75, or 100 mg/kg/day | GD14-Parturition | Increase in dead pups at 75 and 100 mg/kg; Increase in preputial separation at 100 mg/kg/day; Decrease in angiogenital distance at 100 mg/kg/day; No gross morphology alterations; Decrease in serum and intratesticular testosterone |
| Peripubertal | ||||
| Jin et al. [78] | ICR mice | 50, 100, or 200 mg/kg/day | 3 weeks | Decrease in body, testes, and liver weight; Decrease in testosterone; Increase in estradiol; Decrease in expression of steroidogenic genes |
| Pogrmic-Majkic et al. [79] | Wistar rats | In vitro: 1–50 μM In vivo: 200 mg/kg | 24 or 72 h | Transient increases in steroidogenic genes after 24 h exposure followed by a decline |
| Pogrmic et al. [80] | Wistar rats | 50 or 200 mg/kg/day | PND 23–50 | Decrease in steroidogenic genes and androgens |
| Friedmann et al. [81] | Rat | 50 mg/kg/day | PND 46–48 (acute); PND 22–48 (chronic) | Decrease in serum and intratesticular testosterone |
| Trentacoste et al. [82] | Sprague-Dawley rats | 0, 100, or 200 mg/kg/day | PND 22–48 | Delay in preputial separation; Decrease in serum LH and testosterone; Decrease in intratesticular testosterone; Decrease in organ weights |
| Stoker et al. [83] | Wistar rats | 0, 12.5, 25, 50, 100,150, or 200 mg/kg | PND 23–53 | Decrease in seminal vesicle and prostate weight; No alterations in testes weight; Delay in preputial separation; No alterations in serum testosterone at PND 53 |
| Adult | ||||
| Song et al. [84] | Sprague-Dawley rats | 38.5, 77, or 154 mg/kg/day | 30 days | Decrease in spermatozoa number; Increase in spermatozoa abnormality; Decrease in serum testosterone; Decrease in expression of inhibin-B; Increase in FSH and LH |
| Jin et al., [85] | ICR mice | 0, 100, or 200 mg/kg/day | 4 × 1 week | Decrease in testicular testosterone; Decrease in relative testes weight at 200 mg/kg; Decrease in steroidogenic gene expression |
| Riffle et al. [86] | Wistar rats | 0, 5, 25, 75, 200 mg/kg/day | 3 Days | Increase in serum progesterone in intact and castrated males at 25 and 200 mg/kg and 25, 75, and 200 mg/kg respectively; Increase in serum corticosterone at 25, 75, and 200 mg/kg in intact and castrated rats; Increase in testosterone and luteinizing hormone in intact males at 25 mg/kg; Alterations in proteins associated with testosterone production |
| Abarikwu et al. [87] | Wistar rats | 0, 120, or 200 mg/kg/day | 7 or 16 Days | Decrease in seminal vesicle and prostate weight at 200 mg/kg/day; Decreased sperm number with increased abnormal sperm at 120 and 200 mg/kg/day |
| Victor-Costa et al. [88] | Wistar rat | 50, 200, or 300 mg/kg/day | 7, 15, or 40 days | Decreased body weight, increased testes and adrenal weight; Decreased testosterone; Increased estradiol; Dilation of seminiferous tubules; Testicular atrophy |
| Developmental Origins of Atrazine Toxicity | ||||
| DeSesso et al. [89] | Wistar rats | 0, 1, 5, 25, or 125 mg/kg/day | GD 6–21 or PND 2–21 | No alterations in spermatid counts in testes, spermatozoa counts in epididymides, or plasma testosterone levels at PND 70 or PND 170; Increase in percentage of abnormal sperm on PND 70 at 125 mg/kg/day; PND exposure showed a reduction in absolute testes and epididymis weights in the 125 mg/kg/day treatment group on PND 70; No effect on plasma testosterone or sperm morphology at PND 70 and PND 170 |
| Fraites et al. [72] | Sprague-Dawley rats | 0, 1, 5, 20, or 100 mg/kg/day | GD 14–21 | No alterations in testosterone production, timing of puberty, play behavior, AGD, or male sex organ weights at any atrazine treatment at PND 59 |
| Rayner et al. [90] | Long-Evans rats | 100 mg/kg/day | GD 15–19 | Delay in preputial separation; Increase in lateral prostate weight at PND 120; No alterations in testes and seminal vesicle weight at PND 120 and PND 220; No alterations in serum testosterone and androstenedione, but a decrease in prolactin at PND 220 |
3.1. Gestational Atrazine Exposure in Males
3.2. Peripubertal Atrazine Exposure in Males
3.3. Adulthood Atrazine Exposure in Males
3.4. Developmental Origins of Atrazine Toxicity
3.5. Cellular and Genetic Mechanisms of Reproductive Dysfunction in Males
3.6. Tissue Levels of Atrazine in Males
3.7. Conclusions
4. The Effects of Atrazine on Reproductive Function in Anuran and Fish Models
4.1. The Effects of Atrazine on Reproductive Function in Anuran Models
4.2. The Effects of Atrazine on Reproductive Function in Fish Models
| Reference | Exposure (μg/L) | Duration | Outcomes |
|---|---|---|---|
| African Clawed Frog (Xenopus laevis) | |||
| Chen et al. [109] | 0 or 100 | Stage 46/47-120 days | No signs of hermaphroditism; Delay of seminiferous tubules; Alterations in 143 and 121 proteins in testes and ovaries, respectively; Proteins involved in cellular and metabolic processes along with apoptosis and tight junctions were altered |
| Hayes et al. [102] | 0 or 2.5 | Hatching-Metamorphosis (stage 66) | Decrease in plasma testosterone; Decrease in size of dermal breeding glands; Decrease in relative number of testicular tubules with mature sperm bundles which disappeared 3 years after metamorphosis; Decrease in mating competition |
| Kloas et al. [107] | 0, 0.01, 0.1, 1, 25, or 100 | 8 dpf–metamorphosis (83 dpf) | No effect on growth or larval development; No effects observed on sex ratios or on gonadal histology |
| DuPreez et al. [106] | 0, 1, 10, or 25 | 96 hpf–2 ypm | No effect of clutch size, hatching rate, or time to metamorphosis in F1 generation; No alterations in sex ratio of offspring (F2); No alterations in testicular morphology (F1 or F2) |
| Oka et al. [108] | 0, 0.1, 1, 10, or 100 | Tadpole (stage 49)–metamorphosis (stage 66) | No effect on metamorphosis, gonad development, or aromatase expression; No alterations in levels of vitellogenin |
| Hayes et al. [101] | 0, 0.1, 0.4, 0.8, 1, or 25 | Larvae-stage 66 | Gonadal malformations; Increase in number of hermaphrodites |
| Coady et al. [105] | 0, 0.1, 1, 10, or 25 | 72 hph-2-3 mpm | Concentrations of 0.1 and 25 μg/L did not alter mortality, metamorphosis, gonad development, or aromatase activity; 1 μg/L elicited a decrease in estradiol |
| Hecker et al. [112] | 0, 10, or 100 | 49 days (Adult) | No alterations in germ cell development, aromatase, or plasma hormone concentrations |
| Hecker et al. [113] | 0, 1, 25, or 250 | 36 days (Adult) | No alterations in testicular aromatase activity, expression of cyp19, gonadosomatic index, or plasma estradiol; A significant decrease in plasma testosterone was observed at 250 μg/L |
| Freeman et al. [103] | 0, 100, 450, or 800 | Stage 47 or 54–1, 2, 3, 4, or 5 weeks | Delayed metamorphosis at 100, 450, and 800 μg/L |
| Carr et al. [104] | 0, 1, 10, or 25 | 48 hph–metamorphosis (stage 66) | No alterations in mortality, larval growth, metamorphosis, or sex ratios |
| Hayes et al. [100] | 0, 0.1–200 | Hatching (stage 48)–trail reabsorption (stage 66) | Hermaphroditism in males; Decrease in testosterone levels at 25 μg/L |
| Leopard Frog (Rana pipiens) | |||
| Hayes et al. [110] | 0, 0.1, or 25 | 2 dph–tail reabsorption (stage 66) | Underdeveloped testes; Sex reversal in some males (testicular oocytes observed) |
| Spotted Marsh Frog (Limnodynastes tasmaniensis) | |||
| Spolyarich et al. [111] | 0, 0.1, 1, 3, or 30 | Gosner stage 28–44 | No alterations in tadpole growth, development, or sex ratios; Testicular oocytes observed in one fish at 3 μg/L atrazine, although not statistically significant |
| Fathead Minnow (Pimephales promelas) | |||
| Tillitt et al. [115] | 0, 0.5, 5, or 50 | Adult exposure (14 or 30 days) | Decrease in total egg production; Reduction in total number of spawning events; No alterations in steroid hormone levels; Testicular oocytes found in the 5 μg/Ltreatment group; Ovaries with lipid accumulation and atretic follicles observed |
| Battelle [116] | 0, 25, or 250 | Adult exposure (21 days) | No alterations in GSI, mean egg production or spawning events; No histological alterations (no atretic follicles; testicular atrophy) |
| Bringolf et al. [117] | 0, 5, or 50 | Adult exposure (21 days) | No alterations in survival, spawning, egg production, relative gonad weight, or in gonad histology |
| Goldfish (Carassius auratus) | |||
| Nadzialek et al. [118] | 0, 100, or 1000 | Adult exposure (56 days) | No alterations in GSI, plasma concentrations of estradiol, or alterations in cyp19a1; Decrease in 11-ketotestosterone after 56 day exposure in the 1000 μg/L treatment |
| Spanò et al. [119] | 0, 100, or 1000 | Adult exposure (21 days) | No alterations in GSI; Decrease in testosterone and 11-ketotestosterone and an increase in estradiol in the 1000 μg/L treatment group; Structural disruption in testis tissue (1000 μg/L); Follicular atresia in ovary tissue in both treatments; No changes in vitellogenin |
| Japanese Medaka (Oryzias latipes) | |||
| Papoulias et al. [120] | 0, 0.5, 5, or 50 | Adult (14 or 38 days) | Decrease in total egg production in all atrazine treatments after day 25 of exposure; No alterations in spawning events, GSI, aromatase protein, or whole body estradiol or testosterone |
| Guppies (Poecilia reticulate) | |||
| Shenoy [121] | 0, 1, or 13.5 | Adult (through gestation) | Atrazine exposed male offspring were less likely to perform mating behaviors (frequency of gonopodium swings and number of forced copulatory events), and performed less frequently than control male |
| Shenoy [122] | 0, 1, or 15 | Adult male (16 weeks) | No change in number of mating attempts; Decrease in number of mating displays; Decrease in number of orange spots |
| Barramundi (Lates calcarifer) | |||
| Kroon et al. [123] | 0, 0.1, 0.5, 5, 50, or 100 | Juvenile (48 h) | No alterations in cyp19b1 in brain tissue; No alterations in VGT; Increase in plasma testosterone at 0.1 and 5 μg/L; Increase in plasma estradiol at 5 ppb |
| Zebrafish (Danio rerio) | |||
| Freeman et al. [124] | 0, 0.3, 3, or 30 | 1–72 hpf | Decrease in spawning events at 30 μg/L treatment; Egg bound and increase in atretic follicles in adult females exposed to 30 μg/L; Increase in ovarian progesterone in adult females exposed to 3 or 30 μg/L; Transcriptomic analysis revealed alterations in genes involved in steroidogenesis in adult female ovary |
| Wirbisky et al. [125] | 0, 0.3, 3, or 30 | 1–72 hpf | Decrease in serotonin metabolite 5-hydroxyindoleacetic acid and serotonin turnover in adult female brain; Transcriptomic analysis revealed alterations in genes throughout the serotonergic pathway in adult female brain |
| Weber et al. [114] | 0, 0.3, 3, or 30 | 1–72 hpf | Significant increase in head length at 72 hpf in all atrazine treatments; Transcriptomic data included genes altered in neuroendocrine and reproductive system function, cell cycle, and carcinogenesis |
| Corvi et al. [126] | 0, 0.1, 1, or 10 μM | 17 dpf–130 dpf | No alterations in sex ratio; No alterations in gonad development |
| Suzawa and Ingraham [127] | 0, 0.1, 1, or 10 μM | 17 dpf–6 mpf | Increase in expression of cyp19a1 in all concentrations; Skewed sex ratio (increased number of females); Activation of MAPK pathway, SF-1, and PI3K pathway; Phosphorylation of Akt/PKB; Increase in cAMP in JEG3 cells; Upregulation in StAR and cyp11a1 |
| Kazeto et al. [128] | 4.6, 46, or 460 nM | 17 dpf–20 dpf | No alterations in cyp19a1 or cyp19a2 at 4.6, 4.6, or 460 nM atrazine |
4.3. Cellular and Genetic Mechanisms of Reproductive Dysfunction in Anuran and Fish Models
5. Epidemiological Studies with Atrazine
| Reference | Results |
|---|---|
| Chevrier et al. [27] | Quantified levels of atrazine and/or atrazine mercapturate in urine of 5.5% of 579 pregnant females. These levels were associated with fetal growth restriction and small head circumference. No association with congenital malformations. |
| Cragin et al. [130] | Study compared females in Illinois and Vermont and found women who lived in Illinois had a higher rate of menstrual cycle irregularity, longer follicular phases, and decreased levels of menstrual cycle biomarker. |
| Bakke et al. [131] | Urine samples collected from farm families in Iowa (United States) found higher levels of urinary atrazine in farmers as compared to controls. High levels of atrazine correlated to recent field application. |
| Ochoa-Acuña et al. [18] | Study revealed higher atrazine levels in drinking water during the third trimester and entire pregnancy was associated with babies born small for their gestational age. |
| Winchester et al. [132] | Elevated levels of pesticide exposure (atrazine) from April to July were associated with higher rates of birth defects. |
| Curwin et al. [133] | Urine samples collected from farm and non-farm families in Iowa (United States) found higher levels of urinary atrazine in farm families. |
| Barr et al. [17] | Urine samples collected from individuals in Georgia (United States). Atrazine, atrazine mercapturate, and other atrazine metabolites (DACT, DEA, DIA) were found in urine samples with DACT being the most prevalent. |
| Mattix et al. [134] | Prevalence of abdominal wall defects was higher in Indiana (United States) as compared to the national rate. Comparison demonstrated a positive correlation between abdominal wall defects and atrazine exposure. |
| Munger et al. [135] | Iowa study found greater risk of intrauterine growth retardation with higher levels of atrazine in drinking water. |
| Villanova et al. [136] | Atrazine levels in water from the district of Finistère in West Brittany, France, were not associated with lower birth weight or babies born small for gestational age. Results suggest association with prematurity. |
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Plant, T.M. The hypothalamo-pituitary-gonadal axis. J. Endocrinol. 2015, 226, 14–54. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Kuang, L.; Han, B.; Li, Q.; Chen, Y.; Zhu, C.; Chen, Y.; Xia, F.; Cang, Z.; Zhu, C.; et al. Follicle-stimulating hormone associates with prediabetes and diabetes in postmenopausal women. Acta Diabetol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-M.; Yang, W.-X. Molecular regulation of hypothalamus-pituitary-gonads axis in males. Gene 2014, 551, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Vadakkadath, M.S.; Atwood, C.S. The role of hypothalamic-pituitary-gonadal hormones in the normal structure and functioning of the brain. Cell Mol. Life Sci. 2005, 62, 257–270. [Google Scholar]
- Roy, J.R.; Chakraborty, S.; Chakraborty, T.R. Estrogen-like endocrine disrupting chemicals affecting puberty in humans—A review. Med. Sci. Monit. 2009, 15, 137–145. [Google Scholar]
- Swedenborg, E.; Rüegg, J.; Mäkelä, S.; Pongratz, I. Endocrine disrupting chemicals: Mechanisms of action and involvement in metabolic disorders. J. Mol. Endocrinol. 2009, 43, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Danjou, A.M.; Fervers, B.; Boutron-Ruault, M.C.; Philip, T.; Clavel-Chapelon, F.; Dossus, L. Estimated dietary dioxin exposure and breast cancer risk among women from the French E3N prospective cohort. Breast. Cancer. Res. 2015, 17, 17–39. [Google Scholar] [CrossRef] [PubMed]
- Swan, S.H.; Kruse, R.L.; Liu, F.; Barr, D.B.; Drobnis, E.Z.; Redmon, J.B.; Wang, C.; Brazil, C.; Overstreet, J.W. Study for Future Families Research Group. Semen quality in relation to biomarkers of pesticide exposure. Environ. Health. Perspect. 2003, 111, 1478–1484. [Google Scholar] [CrossRef] [PubMed]
- Vafeiadi, M.; Georgiou, V.; Chalkiadaki, G.; Rantakokko, P.; Kiviranta, H.; Karachaliou, M.; Fthenou, E.; Venihaki, M.; Sarri, K.; Vassilaki, M.; et al. Association of Prenatal Exposure to Persistent Organic Pollutants with Obesity and Cardiometabolic Traits in Early Childhood: The Rhea Mother-Child Cohort (Crete, Greece). Environ. Health. Perspect. 2015. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Wu, Q.; Sakthivel, S.; Pavithran, P.V.; Vasukutty, J.R.; Kannan, K. Urinary levels of endocrine-disrupting chemicals, including bisphenols, bisphenol a diglycidyl ethers, benzophenones, parabens, and triclosan in obese and non-obese Indian children. Environ. Res. 2015, 137, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.A.; Gluckman, P.D. Early developmental conditioning of later health and disease: Physiology or pathophysiology. Physiol. Rev. 2014, 94, 1027–1076. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.D.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors. Endocrinology 2006, 147, S43–S49. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Bosagna, C.; Settles, M.; Lucker, B.; Skinner, M.K. Epigenetic transgenerational actions of vinclozolin on promoter regions of the sperm epigenome. PLoS ONE 2010. [Google Scholar] [CrossRef] [PubMed]
- Fudvoye, J.; Bourguignon, J.-P.; Parent, A.-S. Endocrine Disrupting Chemicals and Human Growth Maturation: A focus on early critical windows of exposure. Vitam. Horm. 2014, 94, 1–25. [Google Scholar] [PubMed]
- Vandenberg, L.N. Low-dose effects of hormones and endocrine disruptors. Vitam. Horm. 2012, 94, 129–165. [Google Scholar]
- Barr, D.B.; Panuwet, P.; Nguyen, J.V.; Udunka, S.; Needham, L.L. Assessing exposure to atrazine and its metabolites using biomonitoring. Environ. Health. Perspect. 2007, 115, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Acuña, H.; Frankenberger, J.; Hahn, L.; Carbajo, C. Drinking-water herbicide exposure in Indiana and prevalence of small-for-gestational-age and preterm delivery. Environ. Health. Perspect. 2009, 117, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, J.C.; Wetzel, L.T.; Stevens, J.T.; Simpkins, J.W. The mammary tumor response in triazine-treated female rats: A threshold-mediated interaction with strain and species-specific reproductive senescence. Steroids 1999, 64, 672–678. [Google Scholar] [CrossRef]
- Solomon, K.R.; Carr, J.A.; du Preez, L.H.; Giesy, J.P.; Kendall, R.J.; Smith, E.E.; van der Kraak, G.J. Effects of atrazine on fish, amphibians, and aquatic reptiles: A critical review. Crit. Rev. Toxicol. 2008, 38, 721–772. [Google Scholar] [CrossRef] [PubMed]
- Rinsky, J.L.; Hopenhayn, C.; Golla, V.; Browning, S.; Bush, H.M. Atrazine exposure in public drinking water and preterm birth. Public. Health. Rep. 2012, 127, 72–80. [Google Scholar] [PubMed]
- Rohr, J.R.; McCoy, K.A. A qualitative meta-analysis reveals consistent effects of atrazine on freshwater fish and amphibians. Environ. Health. Perspect. 2010, 118, 20–32. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (U.S. EPA). EPA 816-F-02-013. In List of Contaminants and Their MCLs; U.S. Environmental Protection Agency: Washington, DC, USA, 2002. [Google Scholar]
- Vonberg, D.; Vanderborght, J.; Cremer, N.; Pütz, T.; Herbst, M.; Vereecken, H. 20 years of long term atrazine monitoring in a shallow aquifer in western Germany. Water. Res. 2014, 50, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Jablonowski, N.D.; Schäffer, A.; Burauel, P. Still present after all these years: Persistence plus potential toxicity raise questions about the use of atrazine. Environ. Sci. Pollut. Res. 2011, 18, 328–331. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking Water. WHO: Geneva, Switzerland, 2011; Chapter 12; pp. 307–442. [Google Scholar]
- Chevrier, C.; Limon, G.; Monfort, C.; Rouget, F.; Garlantezec, R.; Petit, C.; Durand, G.; Cordier, S. Urinary biomarkers of prenatal atrazine exposure and adverse birth outcomes in the PELAGIE birth cohort. Environ. Health. Perspect. 2011, 119, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Pogrmic-Majkic, K.; Samardzija, D.; Fa, S.; Hrubik, J.; Glisic, B.; Kaisarevic, S.; Andric, N. Atrazine enhances progesterone production through activation of multiple signaling pathways in FSH-stimulated rat granulosa cells: Evidence for premature luteinization. Biol. Reprod. 2014, 91, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fa, S.; Pogrmic-Majkic, K.; Samardzija, D.; Glisic, B.; Kaisarevic, S.; Kovacevic, R.; Andric, N. Involvement of ERK1/2 signaling pathway in atrazine action on FSH-stimulated LHR and CYP19A1 expression in rat granulosa cells. Toxicol. Appl. Pharmacol. 2013, 270, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Basini, G.; Bianchi, F.; Bussolati, S.; Baioni, L.; Ramoni, R.; Grolli, S.; Conti, V.; Bianchi, F.; Grasselli, F. Atrazine disrupts steroidogenesis, VEGF and NO production in swine granulosa cells. Ecotoxicol. Environ. Saf. 2011, 85, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Tinfo, N.S.; Hotchkiss, M.G.; Buckalew, A.R.; Zorrilla, L.M.; Cooper, R.L.; Laws, S.C. Understanding the effects of atrazine on steroidogenesis in rat granulosa and H295R adrenal cortical carcinoma cells. Reprod. Toxicol. 2011, 31, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Holloway, A.C.; Anger, D.A.; Crankshaw, D.J.; Wu, M.; Foster, W.G. Atrazine-induced changes in aromatase activity in estrogen sensitive target tissues. J. Appl. Toxicol. 2008, 28, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, J.T.; Seinen, W.; Giesy, J.P.; van den Berg, M. 2-chloro-S-triazine herbicides induce aromatase (CYP19) activity in H295R human adrenocortical carcinoma cells: A novel mechanism for estrogenicity? Toxicol. Sci. 2000, 54, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.K.; Murr, A.S.; Best, D.S.; Fraites, M.J.; Zorrilla, L.M.; Narotsky, M.G.; Stoker, T.E.; Goldman, J.M.; Cooper, R.L. The effects of prenatal exposure to atrazine on pubertal and postnatal reproductive indices in the female rat. Reprod. Toxicol. 2011, 32, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Hovey, R.C.; Coder, P.S.; Wolf, J.C.; Sielken, R.L., Jr.; Tisdel, M.O.; Breckenridge, C.B. Quantitative assessment of mammary gland development in female Long Evans rats following in utero exposure to atrazine. Toxicol. Sci. 2011, 119, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Rayner, J.L.; Enoch, R.R.; Fenton, S.E. Adverse effects of prenatal exposure to atrazine during a critical period of mammary gland growth. Toxicol. Sci. 2005, 87, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Rayner, J.L.; Wood, C.; Fenton, S.E. Exposure parameters necessary for delayed puberty and mammary gland development in Long-Evans rats exposed in utero to atrazine. Toxicol. Appl. Pharmacol. 2004, 195, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Ashby, J.; Tinwell, H.; Stevens, J.; Pastoor, T.; Brechenridge, C.B. The effects of atrazine on the sexual maturation of female rats. Regul. Toxicol. Pharmacol. 2002, 35, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Laws, S.C.; Ferrell, J.M.; Stoker, T.E.; Schmid, J.; Cooper, R.L. The effects of atrazine on female Wistar rats: An evaluation of the protocol for assessing pubertal development and thyroid function. Toxicol. Sci. 2000, 58, 366–367. [Google Scholar] [CrossRef] [PubMed]
- Foradori, C.D.; Sawhney, C.P.; Tisdel, M.; Yi, K.D.; Simpkins, J.W.; Handa, R.J.; Breckenridge, C.B. The effect of atrazine administered by gavage or in diet on the LH surge and reproductive performance in intact female Sprague-Dawley and Long Evans rats. Birth Defects Res. B Dev. Reprod. Toxicol. 2014, 101, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Goldman, J.M.; Davis, L.K.; Murr, A.S.; Cooper, R.L. Atrazine-induced elevation or attenuation of the LH surge in the ovariectomized, estrogen-primed female rat: Role of adrenal progesterone. Reproduction 2013, 146, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Foradori, C.D.; Zimmerman, A.D.; Hinds, L.R.; Zuloaga, K.L.; Breckenridge, C.B.; Handa, R.J. Atrazine inhibits pulsatile gonadotropin-releasing hormone (GnRH) release without altering GnRH messenger RNA or protein levels in the female rat. Biol. Reprod. 2013, 88, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Quignot, N.; Arnaud, M.; Robidel, F.; Lecomte, A.; Tournier, M.; Cren-Olivé, C.; Barouki, R.; Lemazurier, E. Characterization of endocrine-disrupting chemicals based on hormonal balance disruption in male and female adult rats. Reprod. Toxicol. 2012, 33, 339–352. [Google Scholar] [CrossRef] [PubMed]
- Taketa, Y.; Yoshida, M.; Inoue, K.; Takahashi, M.; Sakamoto, Y.; Watanabe, G.; Taya, K.; Yamate, J.; Nishikawa, A. Differential stimulation pathways of progesterone secretion from newly formed corpora lutea in rats treated with ethylene glycol monomethyl ether, sulpiride, or atrazine. Toxicol. Sci. 2011, 121, 267–287. [Google Scholar] [CrossRef] [PubMed]
- Foradori, C.D.; Hinds, L.R.; Hanneman, W.H.; Handa, R.J. Effects of atrazine and its withdrawal on gonadotropin-releasing hormonee neuroendocrine function in the adult female Wistar rat. Biol. Reprod. 2009, 81, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Foradori, C.D.; Hinds, L.R.; Hanneman, W.H.; Legare, M.E.; Clay, C.M.; Handa, R.J. Atrazine inhibits pulsatile luteinizing hormone release without altering pituitary sensitivity to a gonadotropin-releasing hormone receptor agonist in female Wistar rats. Biol. Reprod. 2009, 81, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Shibayama, H.; Kotera, T.; Shinoda, Y.; Hanada, T.; Kajihara, T.; Ueda, M.; Tamura, H.; Ishibashi, S.; Yamashita, Y.; Ochi, S. Collaborative work on evaluation of ovarian toxicity. 14) Two- or four-week repeated-dose studies and fertility study of atrazine in female rats. Toxicol. Sci. 2009, 34, 147–155. [Google Scholar] [CrossRef]
- Juliani, C.C.; Silva-Zacarin, E.C.M.; Santos, D.C.; Boer, P.A. Effects of atrazine on female Wistar rats: Morphological alterations in ovarian follicles and immunocytochemical labeling of 90 kDa heat shock protein. Micron 2008, 39, 607–616. [Google Scholar] [CrossRef] [PubMed]
- McMullin, T.; Andersen, M.E.; Nagahara, A.; Lund, T.D.; Pak, T.; Handa, R.J.; Hanneman, W.H. Evidence that atrazine and diaminochlorotriazine inhibit the estrogen/progesterone induced surge of luteinizing hormone in female Sprague-Dawley rats without changing estrogen receptor action. Toxicol. Sci. 2004, 79, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.L.; Stoker, T.L.; Tyrey, L.; Goldman, J.M.; McElroy, W.K. Atrazine disrupts the hypothalamic control of pituitary-ovarian function. Toxicol. Sci. 2000, 53, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Wetzel, L.T.; Luempert, L.G., 3rd; Breckenridge, C.B.; Tisdel, M.O.; Stevens, J.T.; Thakur, A.K.; Extrom, P.J.; Eldridge, J.C. Chronic effects of atrazine on estrus and mammary tumor formation in female Sprague-Dawley and Fischer 344 rats. J. Toxicol. Environ. Health 1994, 43, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.T.; Breckenridge, C.B.; Wetzel, L.T.; Gillis, J.H.; Luempert, L.G., 3rd; Eldridge, J.C. Hypothesis for mammary tumorigenesis in Sprague-Dawley rats exposed to certain triazine herbicides. J. Toxicol. Environ. Health. 1994, 43, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.T.; Breckenridge, C.B.; Wetzel, L. A risk characterization for atrazine: Oncogenicity profile. J. Toxicol. Environ. Health. Part. A 1999, 56, 69–109. [Google Scholar] [CrossRef] [PubMed]
- Pintér, A.; Török, G.; Börzsönyi, M.; Surján, A.; Csík, M.; Kelecsényi, Z.; Kocsis, Z. Long-term carcinogenicity bioassay of the herbicide atrazine in F344 rats. Neoplasma 1990, 37, 533–544. [Google Scholar] [CrossRef]
- Thakur, A.K.; Wetzel, L.T.; Voelker, R.W.; Wakefield, A.E. Results of a two-year oncogenicity study in Fischer 344 rats with atrazine. In Triazine Herbicides: Risk Assessment; Ballantine, L.G., MacFarland, J.E., Hackett, D.S., Eds.; American Chemical Society Symposium Series: Washington, DC, USA, 1998; Chapter 30; pp. 348–398. [Google Scholar]
- Simpkins, J.W.; Swenberg, J.A.; Weiss, N.; Brusick, D.; Eldridge, J.C.; Stevens, J.T.; Handa, R.J.; Hovey, R.C.; Plant, T.M.; Pastoor, T.P.; et al. Atrazine and Breast Cancer: A framework assessment of the toxicological and epidemiological evidence. Toxicol. Sci. 2011, 123, 441–459. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.L.; Laws, S.C.; Das, P.C.; Narotsky, M.G.; Goldman, J.M.; Tyrey, E.L.; Stoker, T.E. Atrazine and reproductive function: Mode and mechanism of action studies. Birth Defects Res. B Dev. Reprod Toxicol. 2007, 80, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, J.C.; Wetzel, L.T.; Tyrey, L. Estrous cycle patterns of Sprague-Dawley rats during acute and chronic atrazine administration. Reprod. Toxicol. 1999, 13, 491–499. [Google Scholar] [CrossRef]
- Gojmerac, T.; Kniewald, J. Atrazine biodegeneration in rats: A model for mammalian metabolism. Bull. Environ. Contam. Toxicol. 1989, 43, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Das, P.C.; McElroy, W.K.; Cooper, R.L. Differential modulation of catecholamines by chlorotriazine herbicides in pheochromocytoma (PC12) cells in vitro. Toxicol. Sci. 2000, 56, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Dodd, C.A.; Xiao, S.; Krishna, S.; Ye, X.; Filipov, N.M. Gestational and lactational exposure to atrazine via the drinking water causes specific behavioral deficits an selectively alters monoaminergic systems in C57BL/6 mouse dams, juvenile and adult offspring. Toxicol. Sci. 2014, 141, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Shafer, T.J.; Ward, T.R.; Meacham, C.A.; Cooper, R.L. Effects of chlorotriazine herbicide, cyanazine, on GABAA receptors in cortical tissue from rat brain. Toxiocology 1999, 142, 57–68. [Google Scholar] [CrossRef]
- Gottsch, M.L.; Cunningham, M.J.; Smith, J.T.; Popa, S.M.; Acohido, B.V.; Crowley, W.F.; Seminara, S.; Clifton, D.K.; Steiner, R.A. A role for kisspeptins in the regulation of gonadotropin secretion in the mouse. Endocrinology 2004, 145, 4073–4077. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.M.; Castellano, J.M.; Fernández-Fernández, R.; Tovar, S.; Roa, J.; Mayen, A.; Nogueiras, R.; Vazquez, M.J.; Barreiro, M.L.; Magni, P.; et al. Characterization of the potent luteinizing hormone-releasing activity of KiSS-1 peptide, the natural ligand of GPR54. Endocrinology 2005, 146, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Gojmerac, T.; Kartal, B.; Ćurić, S.; Žurić, M.; Kušević, S.; Cventi, Z. Serum biochemical changes associated with cystic ovarian degeneration in pigs after atrazine treatment. Toxicol. Lett. 1996, 85, 9–15. [Google Scholar] [CrossRef]
- Hirshfield, A.N. Overview of ovarian follicular development: Considerations for the toxicologist. Environ. Mol. Mutagen. 1997, 29, 10–15. [Google Scholar] [CrossRef]
- Fraites, M.J.P.; Cooper, R.L.; Buckalew, A.; Jayaraman, S.; Mills, L.; Laws, S.C. Characterization of the hypothalamic-pitutiary-adrenal axis response to atrazine and metabolites in the female rat. Toxicol. Sci. 2009, 121, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Telfer, E.; Gosden, R.G.; Faddy, M.J. Impact of exogenous progesterone on ovarian follicular dynamics and function in mice. J. Reprod. Fertil. 1991, 93, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.Y.; Liu, Z.; Shimada, M.; Sterneck, E.; Johnson, P.F.; Hedrick, S.M.; Richards, J.S. MAPK3/1 (ERK1/2) in ovarian granulosa cells are essential for female fertility. Science. 2009, 324, 938–941. [Google Scholar] [CrossRef] [PubMed]
- Kucka, M.; Pogrmic-Majkic, K.; Fa, S.; Stojilkovic, S.S.; Kovacevic, R. Atrazine acts as an endocrine disrupter by inhibiting cAMP-specific phosphodiesterase-4. Toxicol. Appl. Pharmacol. 2012, 265, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Roberge, M.; Hakk, H.; Larsen, G. Atrazine is a competitive inhibitor of phosphodiesterase but does not affect the estrogen receptor. Toxicol. Lett. 2004, 154, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Fraites, M.J.; Narotsky, M.G.; Best, D.S.; Stoker, T.E.; Davis, L.K.; Goldman, J.M.; Hotchkiss, M.G.; Klinefelter, G.R.; Kamel, A.; Qian, Y.; et al. Gestational atrazine exposure: Effects on male reproductive development and metabolite distribution in the dam, fetus, and neonate. Reprod. Toxicol. 2011, 32, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Quignot, N.; Tournier, M.; Pouech, C.; Cren-Olive, C.; Barouki, R.; Lemazurier, E. Quantification of steroids and endocrine disrupting chemicals in rat ovaries by LC-MS/MS for reproductive toxicology assessment. Anal. Bioanal. Chem. 2012, 403, 1629–1640. [Google Scholar] [CrossRef] [PubMed]
- Forgacs, A.L.; D’Souza, M.L.; Huhtaniemi, I.T.; Rahman, N.A.; Zacharewski, T.R. Triazine herbicides and their chlorometabolites alter steroidogenesis in BLTK1 murine Leydig cells. Toxicol. Sci. 2013, 134, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Abarikwu, S.O.; Farombi, E.O.; Kashyap, M.P.; Pant, A.B. Atrazine induces transcriptional changes in marker genes associated with steroidgogenesis in primary cultures of rat Leydig cells. Toxicol. Vitr. 2011, 25, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Stanko, J.P.; Enoch, R.R.; Rayner, J.L.; Davis, C.C.; Wolf, D.C.; Malarkey, D.E.; Fenton, S.E. Effects of prenatal exposure to a low dose atrazine metabolite mixture on pubertal timing and prostate development of male Long-Evans rats. Reprod. Toxicol. 2010, 30, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, B.G.; Chen, H.; Folmer, J.; Liu, J.; Papadopoulos, V.; Zirkin, B.R. Gestational Exposure to atrazine: Effects on the postnatal development of male offspring. J. Androl. 2008, 29, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wang, L.; Fu, Z. Oral exposure to atrazine modulates hormone synthesis and the transcription of steroidogeneic genes in male peripubertal mice. Gen. Comp. Endocrinol. 2013, 184, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Pogrmic-Majkic, K.; Fa, S.; Dakic, V.; Kaisarevic, S.; Kovacevic, R. Upregulation of peripubertal rat Leydig cell steroidogenesis following 24 h in vitro and in vivo exposure to atrazine. Toxicol. Sci. 2010, 118, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Pogrmic, K.; Fa, S.; Dakic, V.; Kaisarevic, S.; Kovacevic, R. Atrazine oral exposure of peripubertal male rats downregulates steroidogenesis gene expression in Leydig cells. Toxcol. Sci. 2009, 111, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Friedmann, A.S. Atrazine inhibition of testosterone production in rat males following peripubertal exposure. Reprod. Toxicol. 2002, 16, 275–279. [Google Scholar] [CrossRef]
- Trentacoste, S.V.; Friedmann, A.S.; Youker, R.T.; Breckenridge, C.B.; Zirkein, B.R. Atrazine effects on testosterone levels and androgen-dependent reproductive orangs in peripubertal male rats. J. Androl. 2001, 22, 142–148. [Google Scholar] [PubMed]
- Stoker, T.E.; Laws, S.C.; Guidici, D.L.; Cooper, R.L. The effect of atrazine on puberty in male Wistar rats: An evaluation in the protocol for the assessment of pubertal development and thyroid function. Toxicol. Sci. 2000, 58, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Jia, Z.C.; Chen, J.Y.; Hu, J.X.; Zhang, L.S. Toxic effects of atrazine on reproductive system of male rats. Biomed. Environ. Sci. 2014, 27, 281–288. [Google Scholar] [PubMed]
- Jin, Y.; Wang, L.; Chen, G.; Lin, X.; Miao, W.; Fu, Z. Exposure of mice to atrazine and its metabolite diaminochlorotriazine elicits oxidative stress and endocrine disruption. Environ. Toxciol. Pharmacol. 2014, 37, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Riffle, B.W.; Klinefelter, G.R.; Cooper, R.L.; Winnik, W.M.; Swank, A.; Jayaraman, S.; Suarez, J.; Best, D.; Laws, S.C. Novel molecular events associated with altered steroidogenesis induced by exposure to atrazine in the intact and castrate male rat. Reprod. Toxicol. 2014, 47, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Abarikwu, S.O.; Adesiyan, A.C.; Oyeloja, T.O.; Oyeyemi, M.O.; Farombi, E.O. Changes in sperm characteristics and induction of oxidative stress in the testis and epididymis of experimental rats by a herbicide, atrazine. Arch. Environ. Contam. Toxicol. 2010, 58, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Victor-Costa, A.B.; Bandeira, S.M.; Oliveira, A.G.; Mahecha, G.A.; Oliveira, C.A. Changes in testicular morphology and steroidogenesis in adult rats exposed to Atrazine. Reprod. Toxicol. 2010, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- De Sesso, J.M.; Scialli, A.R.; White, T.E.K.; Breckenridge, C.B. Multigeneration reproduction and male development toxicity studies on atrazine in rats. Birth Defects Res. B Dev. Reprod. Toxicol. 2014, 101, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Rayner, J.L.; Enoch, R.R.; Wolf, D.C.; Fenton, S.E. Atrazine-induced reproductive tract alterations after transplancental and/or lactational exposure in male Long-Evans rats. Toxicol. Appl. Pharmacol. 2007, 218, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Barouki, R.; Gluckman, P.D.; Grandjean, P.; Hanson, M.; Heindel, J.J. Developmental origins of non-communicable disease: Implications for research and public health. Environ. Health. 2012, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ho, S.M. Epigenetics meets endocrinology. J. Mol. Endocrinol. 2011, 46, 11–32. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in utero and early-life conditions on adult health and disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Jirtle, R.L.; Skinner, M.K. Environmental epigenomics and disease susceptibility. Nat. Rev. Genet. 2007, 8, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Stoker, T.E.; Robinette, C.L.; Cooper, R.L. Maternal exposure to atrazine during lactation suppresses suckling-induced prolactin release and results in prostatitis in the adult offspring. Toxicol. Sci. 1999, 52, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, S.R.; Ma, Y.J.; Lee, B.T.; Prevot, V. Glia-to-neuron signaling and the neuroendocrine control of female puberty. Recent. Prog. Horm. Res. 2000, 55, 197–223. [Google Scholar] [CrossRef] [PubMed]
- Manna, P.R.; Chandrala, S.P.; Jo, Y.; Stocco, D.M. cAMP-independent signaling regulates steroidogenesis in mouse Leydig cells in the absence of StAR phosphorylation. J. Mol. Endocrinol. 2006, 37, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Manna, P.R.; Jo, Y.; Stocco, D.M. Regulation of Leydig cell steroidogenesis by extracellular signal-related kinase 1/2: Role of protein kinase A and protein kinase C signaling. J. Endocrinol. 2007, 193, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Ross, M.K.; Jones, T.L.; Filipov, N.M. Disposition of the herbicide 2-chloro-4-(ethylamino)-6-(isopropylamino)-S-triazine (atrazine) and its major metabolites in mice: A liquid chromatography/mass spectrometry analysis of urine, plasma, and tissue levels. Drug Metab. Dispos. 2009, 37, 776–786. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.B.; Collins, A.; Lee, M.; Mendoza, M.; Noriega, N.; Stuart, A.A.; Vonk, A. Hermaphroditic, demasculinized frogs after exposure to the herbicide atrazine at low ecologically relevant doses. Proc. Natl. Acad. Sci. USA 2002, 99, 5476–5480. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.B.; Stuart, A.A.; Mendoza, M.; Collins, A.; Noriega, N.; Vonk, A.; Johnston, G.; Liu, R.; Kpodzo, D. Characterization of atrazine-induced gonadal malformations in African clawed frogs (Xenopus laevis) and comparisons with effects of an androgen antagonist (cyproterone acetate) and exogenous estrogen (17β-estradiol): Support for the demasculinization/feminization hypothesis. Environ. Health Perspect. 2006, 114, 134–141. [Google Scholar] [PubMed]
- Hayes, T.B.; Khoury, V.; Narayan, A.; Nazir, M.; Park, A.; Brown, T.; Adame, L.; Chan, E.; Buchholz, D.; Stueve, T.; et al. Atrazine induces complete feminization and chemical castration in male African clawed frogs (Xenopus laevis). Proc. Natl. Acad. Sci. USA 2010, 107, 4612–4617. [Google Scholar] [CrossRef] [PubMed]
- Freeman, J.L.; Rayburn, A.L. Developmental impact of atrazine on metamorphing Xenopus laevis as revealed by nuclear analysis and morphology. Enivron. Toxicol. Chem. 2005, 24, 1648–1653. [Google Scholar] [CrossRef]
- Carr, J.A.; Gentles, A.; Smith, E.E.; Goleman, W.L.; Urquidi, L.J.; Thuett, K.; Kendall, R.J.; Giesy, J.P.; Gross, T.S.; Solomon, K.R.; et al. Response of larval Xenopus laevis to atrazine: Assessment of growth, metamorphosis, and gonadal and laryngeal morphology. Environ. Toxicol. Chem. 2003, 22, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Coady, K.K.; Murphy, M.B.; Villeneuve, D.L.; Hecker, M.; Jones, P.D.; Carr, J.A.; Solomon, K.R.; Smith, E.E.; van der Kraak, G.; Kendall, R.J.; et al. Effects of atrazine on metamorphosis, growth, laryngeal and gonadal development, aromatase activity, and sex steroid concentrations in Xenopus laevis. Ecotoxicol. Environ. Saf. 2005, 62, 160–173. [Google Scholar] [CrossRef] [PubMed]
- Du Preez, L.H.; Kunene, N.; Everson, G.J.; Carr, J.A.; Giesy, J.P.; Gross, T.S.; Hosmer, A.J.; Kendall, R.J.; Smith, E.E.; Solomon, K.R.; et al. Reproduction, larval growth, and reproductive development in African clawed frogs (Xenopus laevis) exposed to atrazine. Chemosphere 2008, 71, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Kloas, W.; Lutz, I.; Springer, T.; Krueger, H.; Wolf, J.; Holden, L.; Hosmer, A. Does atrazine influence larval development and sexual differentiation in Xenopus laevis? Toxicol. Sci. 2009, 107, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Oka, T.; Tooi, O.; Mitsui, N.; Miyahara, M.; Ohnishi, Y.; Takase, M.; Kashiwagi, A.; Shinkai, T.; Santo, N.; Iguchi, T. Effect of atrazine on metamorphosis and sexual differentiation in Xenopus laevis. Aquat. Tox. 2008, 87, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, J.; Zhu, H.; Ding, J.; Peng, Y. Proteomics analysis of Xenopus laevis gonad tissue following chronic exposure to atrazine. Environ. Toxicol. Chem. 2015, 34, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.; Haston, K.; Tsui, M.; Hoang, A.; Haeffele, C.; Vonk, A. Atrazine-induced hermaphroditism at 0.1 ppb in American leopard frogs (Rana pipiens): Laboratory and field evidence. Environ. Health Perspect. 2003, 111, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Spolyarich, N.; Hyne, R.; Wilson, S.; Palmer, C.; Byrne, M. Growth, development and sex ratios of Spotted Marsh Frog (Limnodynastes tasmaniensis) larvae exposed to atrazine and a herbicide mixture. Chemosphere 2010, 78, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Kim, W.J.; Park, J.W.; Murphy, M.B.; Villeneuve, D.; Coady, K.K.; Jones, P.D.; Solomon, K.R.; van der Kraak, G.; Carr, J.A.; et al. Plasma concentrations of estradiol and testosterone, gonadal aromatase activity and ultrastructure of the testis in Xenopus laevis exposed to estradiol or atrazine. Aquat. Toxicol. 2005, 72, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Park, J.W.; Murphy, M.B.; Jones, P.D.; Solomon, K.R.; van der Kraak, G.; Carr, J.A.; Smith, E.E.; du Preez, L.; Kendall, R.J.; et al. Effects of atrazine on CYP19 gene expression and aromatase activity in testes and on plasma sex steroid concentrations of male African clawed frogs (Xenopus laevis). Toxicol. Sci. 2005, 86, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Weber, G.J.; Sepúlveda, M.S.; Peterson, S.M.; Lewis, S.L.; Freeman, J.L. Transcriptome alterations following developmental atrazine exposure in zebrafish are associated with disruption of neuroendocrine and reproductive system function, cell cycle, and carcinogenesis. Toxicol. Sci. 2013, 132, 458–466. [Google Scholar] [PubMed]
- Tillitt, D.E.; Papoulias, D.M.; Whyte, J.J.; Richter, C.A. Atrazine reduces reproduction in fathead minnow (Pimephales promelas). Aquat. Toxicol. 2010, 99, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Battelle Corporation, 2005. Multi-chemical evaluation of the short-term reproduction assay with the fathead minnow. In Draft Final Report to U.S. Environmental Protection Agency; U.S. Environmental Protection Agency: Columbus, OH, Contract No. 68-W-01-023. Available online: http://www.epa.gov/scipoly/oscpendo/docs/edmvac/draftfishrepromulti-chemical.pdf (accessed on 15 June 2015).
- Bringolf, R.B.; Belden, J.B. Summerfelt, R.C. Effects of atrazine on fathead minnow in a short-term reproductive assay. Environ. Toxicol. Chem. 2004, 23, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Nadzialek, S.; Spanò, L.; Mandiki, S.N.; Kestemont, P. High doses of atrazine do not disrupt activity and expression of aromatase in female gonads of juvenile goldfish (Carassius auratus L.). Ecotoxicology 2008, 17, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Spanò, L.; Tyler, C.R.; van Aerle, R.; Devos, P.; Mandiki, S.N.; Silvestre, F.; Thomé, J.P.; Kestemont, P. Effects of atrazine on sex steroid dynamics, plasma vitellogenin concentration and gonad development in adult goldfish (Carassius auratus). Aquat. Toxicol. 2004, 10, 369–379. [Google Scholar] [CrossRef]
- Papoulais, D.M.; Tillitt, D.E.; Talykina, M.G.; Whyte, J.J.; Richter, C.A. Atrazine reduces reproduction in Japanese medaka. Aquat. Toxicol. 2014, 154, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, K. Environmentally realistic exposure to the herbicide atrazine alters some sexually selected traits in male guppies. PLoS ONE 2012, 7, e30611. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, K. Prenatal exposure to low doses of atrazine affects mating behaviors in male guppies. Horm. Behav. 2014, 66, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Kroon, F.J.; Hook, S.E.; Jones, D.; Metcalfe, S.; Osborn, H.L. Effects of atrazine on endocrinology and physiology in juvenile barramundi, Lates calcarifer (Bloch). Environ. Toxicol. Chem. 2014, 33, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Freeman, J.L.; Wirbisky, S.E.; Weber, G.J.; Sepúlveda, M.S. A developmental origin of adult reproductive dysfunction in the zebrafish associated with an embryonic exposure to the herbicide atrazine. In Proceedings of PPTOX IV: Environmental stressors in disease and implications for human health, Boston, MA, USA, 26–29 October 2014.
- Wirbisky, S.E.; Weber, G.J.; Sepúlveda, M.S.; Xiao, C.; Cannon, J.R.; Freeman, J.L. Developmental origins of neurotransmitter and transcriptome alterations in adult female zebrafish exposed to atrazine during embryogenesis. Toxicology 2015, 333, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Corvi, M.M.; Stanley, K.A.; Peterson, T.S.; Kent, M.L.; Feist, S.W.; la Du, J.K.; Volz, D.C.; Hosmer, A.J.; Tanguay, R.L. Investigating the impact of chronic atrazine exposure on sexual development in zebrafish. Birth Defects Res. B Dev. Reprod. Toxicol. 2012, 95, 276–288. [Google Scholar] [CrossRef] [PubMed]
- Suzawa, M.; Ingraham, H.A. The herbicide atrazine activates endocrine gene networks via non-steroidal NR5A nuclear receptors in fish and mammalian cells. PLoS ONE 2008. [Google Scholar] [CrossRef] [PubMed]
- Kazeto, Y.; Place, A.R.; Trant, J.M. Effects of endocrine disrupting chemicals on the expression of CYP19 genes in zebrafish (Danio. rerio) juveniles. Aquat. Toxicol. 2004, 69, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.; Lower, N. The impact of two pesticides on olfactory-mediated endocrine function in mature male Atlantic salmon (Salmo salar L.) parr. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 129, 269–276. [Google Scholar] [CrossRef]
- Cragin, L.A.; Kesner, J.S.; Bachand, A.M.; Barr, D.B.; Meadows, J.W.; Krieg, E.F.; Reif, J.S. Menstrual cycle characteristics and reproductive hormone levels in women exposed to atrazine in drinking water. Environ. Res. 2011, 111, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Bakke, B.; de Roos, A.J.; Barr, D.B.; Stewart, P.A.; Blair, A.; Freeman, L.B.; Lynch, C.F.; Allen, R.H.; Alavanja, M.C.; Vermeulen, R. Exposure to atrazine and selected non-persistent pesticides among corn farmers during a growing season. J. Exp. Sci. Environ. Epidemiol. 2009, 19, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Winchester, P.D.; Huskins, J.; Ying, J. Agrichemicals in surface water and birth defects in the United States. Acta Paediatr. 2009, 98, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Curwin, B.D.; Hein, M.J.; Sanderson, W.T.; Striley, C.; Heederik, D.; Kromhout, H.; Reynolds, S.J.; Alavanja, M.C. Urinary pesticide concentrations among children, mothers and fathers living in farm and non-farm households in Iowa. Ann. Occup. Hyg. 2007, 51, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Mattix, K.D.; Winchester, P.D.; Scherer, L.R. Incidence of abdominal wall defects is related to surface water atrazine and nitrate levels. J. Pediatr. Surg. 2007, 42, 947–949. [Google Scholar] [CrossRef] [PubMed]
- Munger, R.; Isacson, P.; Hu, S.; Burns, T.; Hanson, J.; Lynch, C.F.; Cherryholms, K.; van Drope, P.; Hausler, W.J. Intrauterine growth retardation in Iowa communities with herbicide-contaminated drinking water supplies. Environ. Health Perspect. 1997, 105, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, C.M.; Durand, G.; Coutté, M.B.; Chevrier, C.; Cordier, S. Atrazine in municipal drinking water and risk of low birth weight, preterm delivery, and small-for-gestational-age status. Occup. Environ. Med. 2005, 62, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Belloni, V.; Dessì-Fulgheri, F.; Zaccaroni, M.; Di Consiglio, E.; De Angelis, G.; Testai, E.; Santochirico, M.; Alleva, E.; Santucci, D. Early exposure to low doses of atrazine affects behavior in juvenile and adult CD1 mice. Toxicology. 2011, 279, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.; Mandel, J.S.; DeSesso, J.M.; Scialli, A.R. Atrazine and pregnancy outcomes: A systemic review of epidemiologic evidence. Birth Defects Res. B Dev. Reprod. Toxicol. 2014, 101, 1–22. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wirbisky, S.E.; Freeman, J.L. Atrazine Exposure and Reproductive Dysfunction through the Hypothalamus-Pituitary-Gonadal (HPG) Axis. Toxics 2015, 3, 414-450. https://doi.org/10.3390/toxics3040414
Wirbisky SE, Freeman JL. Atrazine Exposure and Reproductive Dysfunction through the Hypothalamus-Pituitary-Gonadal (HPG) Axis. Toxics. 2015; 3(4):414-450. https://doi.org/10.3390/toxics3040414
Chicago/Turabian StyleWirbisky, Sara E., and Jennifer L. Freeman. 2015. "Atrazine Exposure and Reproductive Dysfunction through the Hypothalamus-Pituitary-Gonadal (HPG) Axis" Toxics 3, no. 4: 414-450. https://doi.org/10.3390/toxics3040414
APA StyleWirbisky, S. E., & Freeman, J. L. (2015). Atrazine Exposure and Reproductive Dysfunction through the Hypothalamus-Pituitary-Gonadal (HPG) Axis. Toxics, 3(4), 414-450. https://doi.org/10.3390/toxics3040414






