An Overview of Bortezomib-Induced Neurotoxicity
Abstract
:1. Introduction
2. Incidence and Risk Factors
3. Signs of BIPN
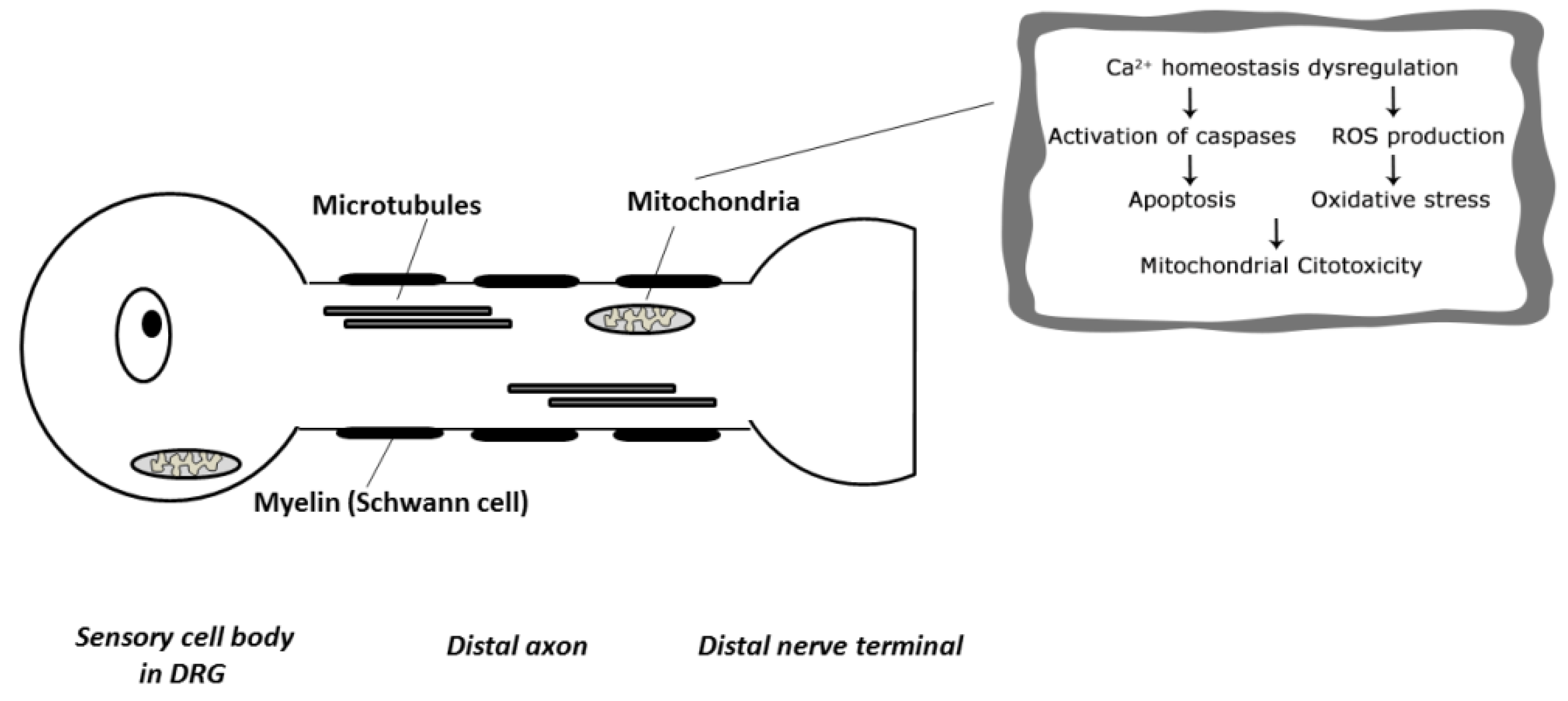
4. Pathogenesis of Neurotoxicity
5. Management of Neurotoxicity
6. Conclusions
Acknowledgements
Conflicts of Interest
References
- Adams, J. The development of proteasome inhibitors as anticancer drugs. Cancer Cell 2004, 5, 417–421. [Google Scholar] [CrossRef]
- Altun, M.; Galardy, P.J.; Shringarpure, R.; Hideshima, T.; LeBlanc, R.; Anderson, K.C.; Ploegh, H.L.; Kessler, B.M. Effects of PS-341 on the activity and composition of proteasomes in multiple myeloma cells. Cancer Res. 2005, 65, 7896–7901. [Google Scholar] [PubMed]
- Schiff, D.; Wen, P.Y.; van den Bent, M.J. Neurological adverse effects caused by cytotoxic and targeted therapies. Nat. Rev. Clin. Oncol. 2009, 6, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Roussel, M.; Moreau, P.; Huynh, A.; Mary, J.Y.; Danho, C.; Caillot, D.; Hulin, C.; Fruchart, C.; Marit, G.; Pégourié, B.; et al. Bortezomib and high-dose melphalan as conditioning regimen before autologous stem cell transplantation in patients with de novo multiple myeloma: A phase 2 study of the Intergroupe Francophone du Myelome (IFM). Blood 2010, 115, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.V. Multiple myeloma: 2014 Update on diagnosis, risk-stratification, and management. Am. J. Hematol. 2014, 89, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Grosicki, S.; Barchnicka, A.; Jurczyszyn, A.; Grosicka, A. Bortezomib for the treatment of multiple myeloma. Expert Rev. Hematol. 2014, 7, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Mai, E.K.; Bertsch, U.; Dürig, J.; Kunz, C.; Haenel, M.; Blau, I.W.; Munder, M.; Jauch, A.; Schurich, B.; Hielscher, T.; et al. Phase III trial of bortezomib, cyclophosphamide, dexamethasone (VCD) versus bortezomib, doxorubicin, dexamethasone (PAd) in newly-diagnosed myeloma. Leukemia 2015. [Google Scholar] [CrossRef] [PubMed]
- McConkey, D.J.; Zhu, K. Mechanisms of proteasome inhibitor action and resistance in cancer. Drug Resist. Updat. 2008, 11, 164–179. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Hideshima, T.; Mitsiades, C.; Anderson, K. Proteasome inhibition in hematologic malignancies. Ann. Med. 2004, 36, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Badros, A.; Goloubeva, O.; Dalal, J.S.; Can, I.; Thompson, J.; Rapoport, A.P.; Heyman, M.; Akpek, G.; Fenton, R.G. Neurotoxicity of bortezomib therapy in multiple myeloma: A single-center experience and review of the literature. Cancer 2007, 110, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Stratogianni, A.; Tosch, M.; Schlemmer, H.; Weis, J.; Katona, I.; Isenmann, S.; Haensch, C.A. Bortezomib-induced severe autonomic neuropathy. Clin. Auton. Res. 2012, 2, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Mateos, M.V.; Richardson, P.G.; Schlag, R.; Khuageva, N.K.; Shpilberg, O.; Kropff, M.; Spicka, I.; Palumbo, A.; Wu, K.L.; et al. Risk factors for, and reversibility of, peripheral neuropathy associated with bortezomib-melphalan-prednisone in newly diagnosed patients with multiple myeloma: Subanalysis of the phase 3 VISTA study. Eur. J. Haematol. 2011, 86, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Beijers, A.J.; Jongen, J.L.; Vreugdenhil, G. Chemotherapy-induced neurotoxicity: The value of neuroprotective strategies. Neth. J. Med. 2012, 7, 18–25. [Google Scholar]
- Richardson, P.G.; Barlogie, B.; Berenson, J.; Singhal, S.; Jagannath, S.; Irwin, D.; Rajkumar, S.V.; Srkalovic, G.; Alsina, M.; Alexanian, R.; et al. A phase 2 study of bortezomib in relapsed, refractory myeloma. N. Engl. J. Med. 2003, 26, 2609–2617. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Xie, W.; Mitsiades, C.; Chanan-Khan, A.A.; Lonial, S.; Hassoun, H.; Avigan, D.E.; Oaklander, A.L.; Kuter, D.J.; Wen, P.Y.; et al. Single-agent bortezomib in previously untreated multiple myeloma: Efficacy, characterization of peripheral neuropathy, and molecular correlations with response and neuropathy. J. Clin. Oncol. 2009, 20, 3518–3525. [Google Scholar] [CrossRef] [PubMed]
- Cavaletti, G.; Jakubowiak, A.J. Peripheral neuropathy during bortezomib treatment of multiple myeloma: A review of recent studies. Leuk. Lymphoma 2010, 51, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Bilińska, M.; Usnarska-Zubkiewicz, L.; Pokryszko-Dragan, A. Bortezomib-induced painful neuropathy in patients with multiple myeloma. Contemp. Oncol. (Pozn.) 2013, 17, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Lanzani, F.; Mattavelli, L.; Frigeni, B.; Rossini, F.; Cammarota, S.; Petrò, D.; Jann, S.; Cavaletti, G. Role of a pre-existing neuropathy on the course of bortezomib-induced peripheral neurotoxicity. J. Peripher. Nerv. Syst. 2008, 13, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Bruna, J.; Alé, A.; Velasco, R.; Jaramillo, J.; Navarro, X.; Udina, E. Evaluation of pre-existing neuropathy and bortezomib retreatment as risk factors to develop severe neuropathy in a mouse model. J. Peripher. Nerv. Syst. 2011, 1, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Kang, D.; Lee, J.Y.; Kim, K.; Kim, S.J. Impact of dose modification on intravenous bortezomib-induced peripheral neuropathy in multiple myeloma patients. Support. Care Cancer 2014, 22, 2669–2675. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; Coiteux, V.; Hulin, C.; Leleu, X.; van de Velde, H.; Acharya, M.; Harousseau, J.L. Prospective comparison of subcutaneous versus intravenous administration of bortezomib in patients with multiple myeloma. Haematologica 2008, 93, 1908–1911. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; Pylypenko, H.; Grosicki, S.; Karamanesht, I.; Leleu, X.; Grishunina, M.; Rekhtman, G.; Masliak, Z.; Robak, T.; Shubina, A.; et al. Subcutaneous versus intravenous administration of bortezomib in patients with relapsed multiple myeloma: A randomised, phase 3, non-inferiority study. Lancet Oncol. 2011, 12, 431–440, Erratum in: Lancet Oncol. 2011, 12, 522. [Google Scholar] [CrossRef]
- Arnulf, B.; Pylypenko, H.; Grosicki, S.; Karamanesht, I.; Leleu, X.; van de Velde, H.; Feng, H.; Cakana, A.; Deraedt, W.; Moreau, P. Updated survival analysis of a randomized phase III study of subcutaneous versus intravenous bortezomib in patients with relapsed multiple myeloma. Haematologica 2012, 97, 1925–1928. [Google Scholar] [CrossRef] [PubMed]
- Argyriou, A.A.; Cavaletti, G.; Bruna, J.; Kyritsis, A.P.; Kalofonos, H.P. Bortezomib-induced peripheral neurotoxicity: An update. Arch. Toxicol. 2014, 88, 1669–1679. [Google Scholar] [CrossRef] [PubMed]
- Favis, R.; Sun, Y.; van de Velde, H.; Broderick, E.; Levey, L.; Meyers, M.; Mulligan, G.; Harousseau, J.L.; Richardson, P.G.; Ricci, D.S. Genetic variation associated with bortezomib-induced peripheral neuropathy. Pharmacogenet. Genomics. 2011, 21, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Corthals, S.L.; Kuiper, R.; Johnson, D.C.; Sonneveld, P.; Hajek, R.; van der Holt, B.; Magrangeas, F.; Goldschmidt, H.; Morgan, G.J.; Avet-Loiseau, H. Genetic factors underlying the risk of bortezomib induced peripheral neuropathy in multiple myeloma patients. Haematologica 2011, 9, 1728–1732. [Google Scholar] [CrossRef] [PubMed]
- Broyl, A.; Corthals, S.L.; Jongen, J.L.; van der Holt, B.; Kuiper, R.; de Knegt, Y.; van Duin, M.; el Jarari, L.; Bertsch, U.; Lokhorst, H.M.; et al. Mechanisms of peripheral neuropathy associated with bortezomib and vincristine in patients with newly diagnosed multiple myeloma: A prospective analysis of data from the HOVON-65/GMMG-HD4 trial. Lancet Oncol. 2010, 1, 1057–1065. [Google Scholar] [CrossRef]
- Cata, J.P.; Weng, H.R.; Burton, A.W.; Villareal, H.; Giralt, S.; Dougherty, P.M. Quantitative sensory findings in patients with bortezomib-induced pain. J. Pain 2007, 8, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Argyriou, A.A.; Iconomou, G.; Kalofonos, H.P. Bortezomib-induced peripheral neuropathy in multiple myeloma: A comprehensive review of the literature. Blood 2008, 112, 1593–1599, Erratum in Blood. 2009, 113, 4478. [Google Scholar] [CrossRef] [PubMed]
- Cavaletti, G.; Nobile-Orazio, E. Bortezomib-induced peripheral neurotoxicity: Still far from a painless gain. Haematologica 2007, 92, 1308–1310. [Google Scholar] [CrossRef] [PubMed]
- Ravaglia, S.; Corso, A.; Piccolo, G.; Lozza, A.; Alfonsi, E.; Mangiacavalli, S.; Varettoni, M.; Zappasodi, P.; Moglia, A.; Lazzarino, M.; et al. Immune-mediated neuropathies in myeloma patients treated with bortezomib. Clin. Neurophysiol. 2008, 119, 2507–2512. [Google Scholar] [CrossRef] [PubMed]
- Gilardini, A.; Avila, R.L.; Oggioni, N.; Rodriguez-Menendez, V.; Bossi, M.; Canta, A.; Cavaletti, G.; Kirschner, D.A. Myelin structure is unaltered in chemotherapy-induced peripheral neuropathy. Neurotoxicology 2012, 33, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Meregalli, C.; Chiorazzi, A.; Carozzi, V.A.; Canta, A.; Sala, B.; Colombo, M.; Oggioni, N.; Ceresa, C.; Foudah, D.; La Russa, F.; et al. Evaluation of tubulin polymerization and chronic inhibition of proteasome as citotoxicity mechanisms in bortezomib-induced peripheral neuropathy. Cell Cycle 2014, 13, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Cavaletti, G.; Gilardini, A.; Canta, A.; Rigamonti, L.; Rodriguez-Menendez, V.; Ceresa, C.; Marmiroli, P.; Bossi, M.; Oggioni, N.; D’Incalci, M.; et al. Bortezomib-induced peripheral neurotoxicity: A neurophysiological and pathological study in the rat. Exp. Neurol. 2007, 204, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Meregalli, C.; Canta, A.; Carozzi, V.A.; Chiorazzi, A.; Oggioni, N.; Gilardini, A.; Ceresa, C.; Avezza, F.; Crippa, L.; Marmiroli, P.; et al. Bortezomib-induced painful neuropathy in rats: A behavioral, neurophysiological and pathological study in rats. Eur. J. Pain 2010, 14, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Carozzi, V.A.; Canta, A.; Oggioni, N.; Sala, B.; Chiorazzi, A.; Meregalli, C.; Bossi, M.; Marmiroli, P.; Cavaletti, G. Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp. Neurol. 2010, 226, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Bruna, J.; Udina, E.; Alé, A.; Vilches, J.J.; Vynckier, A.; Monbaliu, J.; Silverman, L.; Navarro, X. Neurophysiological, histological and immunohistochemical characterization of bortezomib-induced neuropathy in mice. Exp. Neurol. 2010, 223, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Siau, C.; Xiao, W.; Bennett, G.J. Paclitaxel- and vincristine-evoked painful peripheral neuropathies: Loss of epidermal innervation and activation of Langerhans cells. Exp. Neurol. 2006, 201, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Krøigård, T.; Schrøder, H.D.; Qvortrup, C.; Eckhoff, L.; Pfeiffer, P.; Gaist, D.; Sindrup, S.H. Characterization and diagnostic evaluation of chronic polyneuropathies induced by oxaliplatin and docetaxel comparing skin biopsy to quantitative sensory testing and nerve conduction studies. Eur. J. Neurol. 2014, 21, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Casafont, I.; Berciano, M.T.; Lafarga, M. Bortezomib induces the formation of nuclear poly(A) RNA granules enriched in Sam68 and PABPN1 in sensory ganglia neurons. Neurotox. Res. 2010, 17, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Palanca, A.; Casafont, I.; Berciano, M.T.; Lafarga, M. Proteasome inhibition induces DNA damage and reorganizes nuclear architecture and protein synthesis machinery in sensory ganglion neurons. Cell. Mol. Life Sci. 2014, 71, 1961–1975. [Google Scholar] [CrossRef] [PubMed]
- Alé, A.; Bruna, J.; Herrando, M.; Navarro, X.; Udina, E. Toxic effects of bortezomib on primary sensory neurons and schwann cells of adult mice. Neurotox. Res. 2015, 2, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Poruchynsky, M.S.; Sackett, D.L.; Robey, R.W.; Ward, Y.; Annunziata, C.; Fojo, T. Proteasome inhibitors increase tubulin polymerization and stabilization in tissue culture cells: A possible mechanism contributing to peripheral neuropathy and cellular toxicity following proteasome inhibition. Cell Cycle 2008, 7, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Staff, N.P.; Podratz, J.L.; Grassner, L.; Bader, M.; Paz, J.; Knight, A.M.; Loprinzi, C.L.; Trushina, E.; Windebank, A.J. Bortezomib alters microtubule polymerization and axonal transport in rat dorsal root ganglion neurons. Neurotoxicology 2013, 39, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Landowski, T.H.; Megli, C.J.; Nullmeyer, K.D.; Lynch, R.M.; Dorr, R.T. Mitochondrial-mediated disregulation of Ca2+ is a critical determinant of Velcade (PS-341/bortezomib) cytotoxicity in myeloma cell lines. Cancer Res. 2005, 65, 3828–3836. [Google Scholar] [CrossRef] [PubMed]
- Siau, C.; Bennett, G.J. Dysregulation of cellular calcium homeostasis in chemotherapy-evoked painful peripheral neuropathy. Anesth. Analg. 2006, 102, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Xiao, W.H.; Bennett, G.J. Mitotoxicity and bortezomib-induced chronic painful peripheral neuropathy. Exp. Neurol. 2012, 238, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.T.; Liu, Z.G.; Yang, W.; Liao, A.J.; Zhang, R.; Wu, B.; Wang, H.H.; Yao, K.; Li, Y.C. Study on mechanism of bortezomib inducing peripheral neuropathy and the reversing effect of reduced glutathione. Zhonghua Xue Ye Xue Za Zhi 2011, 32, 107–111. [Google Scholar] [PubMed]
- Pérez-Galán, P.; Roué, G.; Villamor, N.; Montserrat, E.; Campo, E.; Colomer, D. The proteasome inhibitor bortezomib induces apoptosis in mantle-cell lymphoma through generation of ROS and Noxa activation independent of p53 status. Blood 2006, 107, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Rahmani, M.; Dent, P.; Grant, S. The hierarchical relationship between MAPK signaling and ROS generation in human leukemia cells undergoing apoptosis in response to the proteasome inhibitor Bortezomib. Exp. Cell Res. 2004, 295, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.H.; Liebes, L.; Zou, Y.; Perez-Soler, R. Reactive oxygen species generation and mitochondrial dysfunction in the apoptotic response to Bortezomib, a novel proteasome inhibitor, in human H460 non-small cell lung cancer cells. J. Biol. Chem. 2003, 278, 33714–33723. [Google Scholar] [CrossRef] [PubMed]
- Montagut, C.; Rovira, A.; Albanell, J. The proteasome: A novel target for anticancer therapy. Clin. Transl. Oncol. 2006, 8, 313–317. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Klionsky, D.J. Regulation mechanisms and signaling pathways of autophagy. Annu. Rev. Genet. 2009, 43, 67–93. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, G.; Materazzi, S.; Fusi, C.; Altomare, A.; Aldini, G.; Lodovici, M.; Patacchini, R.; Geppetti, P.; Nassini, R. Novel therapeutic strategy to prevent chemotherapy-induced persistent sensory neuropathy byTRPA1 blockade. Cancer Res. 2013, 73, 3120–3131. [Google Scholar] [CrossRef] [PubMed]
- Mangiacavalli, S.; Corso, A.; de Amici, M.; Varettoni, M.; Alfonsi, E.; Lozza, A.; Lazzarino, M. Emergent T-helper 2 profile with high interleukin-6 levels correlates with the appearance of bortezomib-induced neuropathic pain. Br. J. Haematol. 2010, 149, 916–918. [Google Scholar] [CrossRef] [PubMed]
- Chiorazzi, A.; Canta, A.; Meregalli, C.; Carozzi, V.; Sala, B.; Oggioni, N.; Monbaliu, J.; van de Velde, H.; Cavaletti, G. Antibody against tumor necrosis factor-α reduces bortezomib-induced allodynia in a rat model. Anticancer Res. 2013, 33, 5453–5459. [Google Scholar] [PubMed]
- Kassem, L.A.; Yassin, N.A. Role of erythropoeitin in prevention of chemotherapy-induced peripheral neuropathy. Pak. J. Biol. Sci. 2010, 13, 577–587. [Google Scholar] [PubMed]
- Ceresa, C.; Avan, A.; Giovannetti, E.; Geldof, A.A.; Avan, A.; Cavaletti, G.; Peters, G.J. Characterization of and protection from neurotoxicity induced by oxaliplatin, bortezomib and epothilone-B. Anticancer Res. 2014, 3, 517–523. [Google Scholar]
- Cavaletti, G.; Alberti, P.; Frigeni, B.; Piatti, M.; Susani, E. Chemotherapy-induced neuropathy. Curr. Treat. Options Neurol. 2011, 13, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, J.; Kolesar, J.M. Prevention and treatment of chemotherapy-induced peripheral neuropathy. Am. J. Health Syst. Pharm. 2014, 71, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Janes, K.; Doyle, T.; Bryant, L.; Esposito, E.; Cuzzocrea, S.; Ryerse, J.; Bennett, G.J.; Salvemini, D. Bioenergetic deficits in peripheral nerve sensory axons during chemotherapy-induced neuropathic pain resulting from peroxynitrite-mediated post-translational nitration of mitochondrial superoxide dismutase. Pain 2013, 154, 2432–2440. [Google Scholar] [CrossRef] [PubMed]
- Alé, A.; Bruna, J.; Morell, M.; van de Velde, H.; Monbaliu, J.; Navarro, X.; Udina, E. Treatment with anti-TNF alpha protects against the neuropathy induced by the proteasome inhibitor bortezomib in a mouse model. Exp. Neurol. 2014, 253, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Tsukaguchi, M.; Shibano, M.; Matsuura, A.; Mukai, S. The protective effects of lafutidine for bortezomib induced peripheral neuropathy. J. Blood Med. 2013, 4, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Nakano, A.; Abe, M.; Oda, A.; Amou, H.; Hiasa, M.; Nakamura, S.; Miki, H.; Harada, T.; Fujii, S.; Kagawa, K.; et al. Delayed treatment with vitamin C and N-acetyl-l-cysteine protects Schwann cells without compromising the anti-myeloma activity of bortezomib. Int. J. Hematol. 2011, 93, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.M.; Pang, H.; Cirrincione, C.; Fleishman, S.; Paskett, E.D.; Ahles, T.; Bressler, L.R.; Fadul, C.E.; Knox, C.; Le-Lindqwister, N.; et al. Effect of duloxetine on pain, function, and quality of life among patients with chemotherapy-induced painful peripheral neuropathy: A randomized clinical trial. JAMA 2013, 3, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meregalli, C. An Overview of Bortezomib-Induced Neurotoxicity. Toxics 2015, 3, 294-303. https://doi.org/10.3390/toxics3030294
Meregalli C. An Overview of Bortezomib-Induced Neurotoxicity. Toxics. 2015; 3(3):294-303. https://doi.org/10.3390/toxics3030294
Chicago/Turabian StyleMeregalli, Cristina. 2015. "An Overview of Bortezomib-Induced Neurotoxicity" Toxics 3, no. 3: 294-303. https://doi.org/10.3390/toxics3030294
APA StyleMeregalli, C. (2015). An Overview of Bortezomib-Induced Neurotoxicity. Toxics, 3(3), 294-303. https://doi.org/10.3390/toxics3030294




