Human and Veterinary Antibiotics Used in Portugal—A Ranking for Ecosurveillance
Abstract
:1. Introduction
2. Materials and Methods
2.1. Antimicrobial Consumption
2.2. Key Pharmacokinetic Features and Occurrence of the Most Representative Antibiotics
2.3. Antibiotic Ranking Approach
3. Results
3.1. Antimicrobial Consumption
| Active substance | Human Medicine | Veterinary Medicine | Target species [25] | Amount [2010 + 2011] (tonnes) | ||||
|---|---|---|---|---|---|---|---|---|
| Amount (tonnes) | Administration route [24] | Amount (tonnes) | Administration route [25] | |||||
| [2010] | [2011] | [2010] | [2011] | |||||
| Aminoglycosides | ||||||||
| Apramycin | --- | --- | --- | 1.020 | 0.761 | Oral use (drinking water) | Porcine; bovine; poultry; | 1.781 |
| Dihydrostreptomycin (Streptomycin) +H | --- | --- | --- | 0.526 | 0.425 | Injection use | Porcine; bovine; ovine; caprine; equidae; rabbit; canine; feline; | 0.951 |
| Gentamicin+H | 0.004 | 0.000 | Injection use | 0.068 | 0.035 | Injection use | Porcine; bovine; | 0.107 |
| Neomycin +H | 0.001 | 0.000 | Oral use (tablet); Injection use | 0.274 | 1.903 | In-feed use a) | Porcine; poultry; | 2.178 |
| Netilmycin +H | 0.004 | 0.001 | Injection use | --- | --- | --- | --- | 0.005 |
| Paromomycin +H | 0.000 | 0.000 | --- | 0.00 | 0.326 | Intramammary use | Bovine | 0.326 |
| Spectinomycin | --- | --- | --- | 0.019 | 0.028 | Injection use Oral use (drinking water) | Porcine; bovine; ovine; caprine; equidae; canine; feline;Porcine; poultry; | 0.047 |
| Total amount | 0.009 | 0.001 | 1.021 | 3.478 | ||||
| Amphenicols | ||||||||
| Florfenicol | --- | --- | --- | 1.491 | 1.487 | Injection use In-feed use | Porcine; BovinePorcine | 2.978 |
| Cloramphenicol +H | 0.000 | 0.000 | 0.000 | 0.000 | 0.00 | |||
| Total amount | 1.491 | 1.487 | ||||||
| Antituberculosis | ||||||||
| Etambutol | 0.003 | 0.004 | Oral use (tablet) | --- | --- | --- | --- | 0.007 |
| Pirazynamide | 0.001 | 0.002 | Oral use (tablet) | --- | --- | --- | --- | 0.003 |
| Rifampicin | 0.041 | 0.043 | Oral use (capsule); lyophilisate for infusion | --- | --- | --- | --- | 0.084 |
| Total amount | 0.045 | 0.049 | ||||||
| Cephalosporins | ||||||||
| Cefaclor +H | 0.780 | 0.550 | Oral use (capsule, Granules) | --- | --- | --- | --- | 1.330 |
| Cefadroxil +H | 0.463 | 0.374 | Oral use (capsule) | 0.003 | 0.004 | Injection use | Bovine; equidae; canine; | 0.844 |
| Cefalexin | --- | --- | --- | 0.481 | 0.220 | Injection use Intramammary use | Bovine; feline; canine;Bovine | 0.701 |
| Cefalonium | --- | --- | --- | 0.009 | 0.011 | Intramammary use | Bovine | 0.020 |
| Cephapirin | --- | --- | --- | 0.005 | 0.003 | Intramammary use | Bovine | 0.008 |
| Cefatrizine +H | 0.766 | 0.622 | Oral use (capsule; tablet; liquid suspension) | --- | --- | --- | --- | 1.388 |
| Cefazolin +H | 0.00 | 0.00 | --- | 0.004 | 0.003 | Intramammary use | Bovine | 0.007 |
| Cefditoren | 0.052 | 0.033 | Oral use (tablet ) | --- | --- | --- | --- | 0.085 |
| Cefeprozil | 0.344 | 0.086 | Oral use (tablet ) | --- | --- | --- | --- | 0.430 |
| Cefixime +H | 0.452 | 0.408 | Oral use (tablet; liquid suspension) | --- | --- | --- | --- | 0.860 |
| Cefonicide | 0.004 | 0.004 | Injection use | 0.008 | ||||
| Cefoperazone | --- | --- | --- | 0.012 | 0.010 | Intramammary use | Bovine | 0.022 |
| Cefquinome | --- | --- | --- | 0.099 | 0.064 | Injection use Intramammary use | Porcine; bovine; equidae; Bovine; | 0.163 |
| Cefodizime | 0.004 | 0.004 | Injection use | --- | --- | --- | --- | 0.008 |
| Cefotaxime | 0.005 | 0.00 | Injection use | --- | --- | --- | --- | 0.005 |
| Cefovecin | --- | --- | --- | 0.002 | 0.002 | Injection use | Canine; feline; | 0.004 |
| Cefradine +H | 0.927 | 0.890 | Oral use (capsule) | --- | --- | --- | --- | 1.817 |
| Cefuroxime +H | 1.477 | 1.473 | Oral use (tablet); Injection use | --- | --- | --- | --- | 2.950 |
| Ceftiofur | --- | --- | --- | 0.198 | 0.252 | Injection use | Porcine; bovine; | 0.450 |
| Ceftriaxone +H | 0.000 | 0.123 | Injection use | --- | --- | --- | --- | 0.123 |
| Total amount | 5.278 | 4.571 | 0.813 | 0.569 | ||||
| Macrolides | ||||||||
| Azithromycin +H | 1.375 | 1.466 | Oral use (tablet ) | --- | --- | --- | --- | 2.841 |
| Clarithromycin +H | 3.365 | 3.274 | Oral use (tablet ) | --- | --- | --- | --- | 6.639 |
| Erythromycin +H | 0.462 | 0.414 | Oral use (tablet ) | 0.013 | 0.017 | Injection use | Bovine | 0.906 |
| Gamithromycin | --- | --- | --- | 0.020 | 0.021 | Injection use | Bovine | 0.041 |
| Roxithromycin | 0.042 | 0.050 | Oral use (tablet ) | --- | --- | --- | --- | 0.092 |
| Spiramycin | 0.648 | 0.563 | Oral use (capsule ) Injection use | 0.278 | 0.279 | Injection use | Porcine; bovine; ovine; caprine | 1.768 |
| Telithromycin | 0.018 | ----- | Oral use (tablet ) | --- | --- | --- | --- | 0.018 |
| Tildipirosin | --- | --- | --- | 0.000 | 0.001 | Injection use | Porcine; bovine; | 0.001 |
| Tilmicosin | --- | --- | --- | 3.288 | 5.071 | Injection use Oral use (drinking water) | BovinePorcine; bovine; poultry | 8.359 |
| Tylosin | --- | --- | --- | 9.906 | 16.302 | Injection use Oral use (drinking water) In-feed use | Porcine; bovine; ovine; caprine; Porcine; bovine; poultry;Porcine; bovine; poultry; | 26.208 |
| Tulathromycin | --- | --- | --- | 0.014 | 0.054 | Injection use | Porcine; bovine | 0.068 |
| Total amount | 5.910 | 5.767 | 13.519 | 21.745 | ||||
| Lincosamides | ||||||||
| Clindamycin | 0.175 | 0.159 | Oral use (capsule) Injection use | 0.001 | 0.001 | Capsule | Canine | 0.336 |
| Lincomycin | 0.004 | 0.005 | Oral use (capsule) | 1.713 | 16.731 | Injection use Oral use (drinking water) In-feed use | Porcine; bovine; ovine; caprine; equidae; canine; feline Porcine; PoultryPorcine | 18.453 |
| Total amount | 0.179 | 0.164 | 1.714 | 16.732 | ||||
| Monobactams | ||||||||
| Aztreonam | 0.015 | 0.008 | Injection use | --- | --- | --- | --- | 0.023 |
| Total amount | 0.015 | 0.008 | ||||||
| Penicilins | ||||||||
| Amoxicillin +H | 49.603 | 50.901 | Oral use (tablet, liquid suspension) | 24.868 | 21.227 | Injection use Oral use (drinking water) Oral use (tablet) | Porcine; bovine; canine; feline; Porcine Canine | 146.599 |
| Ampicillin +H | 0.186 | 0.276 | Injection use | 4.557 | 2.894 | Injection use Oral use (drinking water) d) | Porcine; bovine; ovine; equidae; canine; feline Bovine; ovine | 7.913 |
| Benzylpenicillin +H | 0.131 | 0.131 | Injection use b,C) | 4.909 | 2,184 | Injection use e) Intramammary use f) | Porcine; bovine; ovine; equidae; canine; feline; Bovine | 7.355 |
| Cloxacillin +H | --- | --- | --- | 0.359 | 0.071 | Intramammary use g) | Bovine; ovine; caprine; | 0.430 |
| Dicloxacillin +H | 0.012 | 0.008 | Oral use (tablet ) | --- | --- | --- | 0.020 | |
| Flucloxacillin +H | 4.130 | 3.596 | Oral use (capsule ) | --- | --- | --- | --- | 7.726 |
| Phenoxymethylpenicillin +H | 0.000 | 0.000 | --- | 0.144 | 0.021 | Oral use (drinking water) | Poultry | 0.165 |
| Pivmecillinam | 0.013 | 0.008 | Oral use (tablet ) | --- | --- | --- | --- | 0.021 |
| Total amount | 54.075 | 54.92 | 34.837 | 26.397 | ||||
| Polymyxins | ||||||||
| Colistin +H | 0 | 0 | --- | 15.408 | 8.013 | Injection use Oral use (drinking water) In-feed use | Porcine; bovine; canine Porcine; bovine; ovine; poultry Porcine; poultry; rabbit | 23.421 |
| Total amount | 15.408 | 8.013 | ||||||
| Polypeptides | ||||||||
| Bacitracin +H | 0.000 | 0.000 | --- | 0.000 | 1.710 | In-feed use | Porcine; bovine; poultry; rabbit | 1.710 |
| Total amount | 1.710 | |||||||
| Quinolones | ||||||||
| Ciprofloxacin | 5.963 | 10.940 | Oral use (tablet) | --- | --- | --- | --- | 16.903 |
| Danofloxacin | --- | --- | --- | 0.043 | 0.049 | Injection use | Bovine | 0.092 |
| Difloxacin | --- | --- | --- | 0.001 | 0.000 | Oral use (tablet) | Canine | 0.001 |
| Enrofloxacin | --- | --- | --- | 5.644 | 8.386 | Injection use Oral use (tablet) Oral use (drinking water) | Porcine; bovine; canine; feline; Canine; feline Porcine; poultry; rabbit | 14.03 |
| Flumequine | --- | --- | --- | 0.675 | 0.470 | Oral use (drinking water) | Bovine; poultry | 1.145 |
| Levofloxacin +H | 0.804 | 0.832 | Oral use (tablet) | --- | --- | --- | --- | 1.636 |
| Lomefloxacin | 0.005 | 0.004 | Oral use (tablet) | --- | --- | --- | --- | 0.009 |
| Marbofloxacin | --- | --- | --- | 0.040 | 0.117 | Injection use | Porcine; bovine; canine; feline | 0.157 |
| Moxifloxacin +H | 0.224 | 0.202 | Oral use (tablet) | --- | --- | --- | --- | 0.426 |
| Norfloxacin +H | 0.748 | 0.678 | Oral use (tablet) | --- | --- | --- | --- | 1.426 |
| Ofloxacin +H | 0.140 | 0.116 | Oral use (tablet) | --- | --- | --- | --- | 0.256 |
| Oxolinic acid | --- | --- | --- | 0.003 | 0.000 | Oral use (pellet ) | Fish | 0.003 |
| Pradofloxacin | --- | --- | --- | 0.000 | 0.001 | Oral use (tablet, liquid suspension) | Canine; feline | 0.001 |
| Prulifloxacin | 0.726 | 0.626 | Oral use (tablet) | --- | --- | --- | --- | 1.352 |
| Total amount | 8.61 | 13.398 | 6.406 | 9.023 | ||||
| Sulfonamides and Trimethoprim | ||||||||
| Sulfadiazine +H | 0.005 | 0.008 | Oral use (tablet) | 10.478 | 4.428 | Injection use Oral use (drinking water) In-feed use Oral use (paste) | Porcine; bovine; ovine; equidae Porcine; poultry Porcine Equidae | 14.919 |
| Sulfaguanidine | --- | --- | --- | 0.017 | 0.026 | --- | --- | 0.043 |
| Sulfadoxine | --- | --- | --- | 0.030 | 0.036 | Injection use | Porcine; bovine; ovine; equidae; canine; feline | 0.066 |
| Sulfaquinoxaline | --- | --- | --- | 0.022 | 0.028 | Oral use (drinking water) | Rabbits | 0.050 |
| Sulfamethoxazole +H | 3.971 | 3.764 | Oral use (tablet) | --- | --- | --- | --- | 7.735 |
| Trimethoprim +H | 0.794 | 0.753 | Oral use (tablet) h) | 1.545 | 3.040 | Injection use i) | Porcine; bovine; ovine; equidae; canine; feline | 6.131 |
| Total amount | 4.77 | 4.523 | 12.092 | 7.558 | ||||
| Tetracyclines | ||||||||
| Doxycycline +H | 0.242 | 0.247 | Oral use (capsule) | 28.115 | 28.273 | Oral use (drinking water) In-feed use Oral use (tablet) | Porcine; bovine; poultryPorcineCanine; feline | 56.877 |
| Minocycline | 0.260 | 0.256 | Oral use (tablet ) | 0.516 | ||||
| Oxitetracycline | --- | --- | --- | 46.539 | 13.329 | Injection use Oral use (drinking water) Intrauterine use Cutaneous use (spray) Nebulisation use | Porcine; bovine; ovine; equidae; canine; feline; Porcine; bovine; ovine; caprine; Porcine; bovine; ovine; caprine; equidae Porcine; bovine; ovine; caprine; equidae Porcine; bovine; ovine; caprine; equidae; poultry; rabit; canine; feline | 59.868 |
| Tetracycline +H | 0.000 | 0.000 | --- | 2.001 | 2.86 | Oral use (drinking water) j) | Porcine; bovine; ovine; caprine; poultry | 4.861 |
| Total amount | 0.502 | 0.503 | 76.655 | 44.462 | ||||
| Pleuromutilins | ||||||||
| Tiamulin | --- | --- | --- | 14.659 | 16.302 | Injection use Oral use (drinking water) In-feed use | Porcine Porcine; poultry; Porcine; rabbit; | 30.961 |
| Valnemulin | --- | --- | --- | 0.113 | 5.075 | In-feed use | Porcine | 5.188 |
| Total amount | 14.772 | 21.377 | ||||||
| Other antibiotic | ||||||||
| Fosfomycin | 1.613 | 1.748 | Oral use | --- | --- | --- | --- | 3.361 |
| Fusidic acid | 0.303 | 0.279 | Oral use (granule) | --- | --- | --- | --- | 0.582 |
| Rifaximin | --- | --- | --- | 0.012 | 0.011 | Cutaneous use (spray) Intramammary use | Bovine Bovine | 0.023 |
| Total amount | 1.916 | 2.027 | 0.012 | 0.011 | ||||
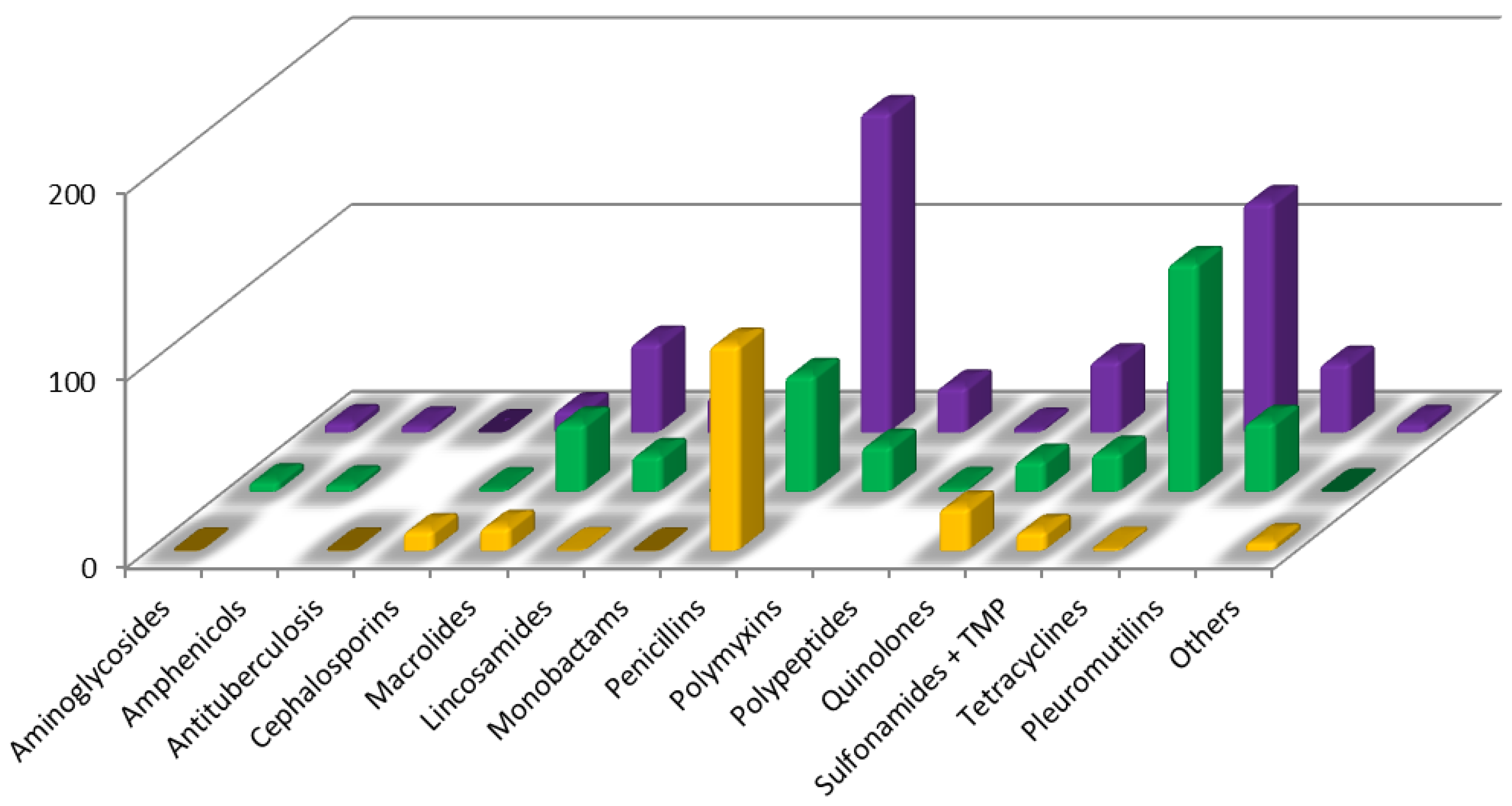

3.2. Key Pharmacokinetic Features and Occurrence of the Most Representative Antibiotics
3.3. Antibiotic Ranking Approach
| Key Features | |
|---|---|
| AMINOGLYCOSIDES | Characterization: Aminoglycosides consumption in human medicine is mostly reserved to hospital care (e.g., gentamicin, streptomycin, neomycin, paromomycin, tobramycin). In veterinary medicine aminoglycosides are mainly used in drinking water (e.g., apramycin) and in feed (e.g., neomycin) for the treatment of bacterial gastrointestinal infections manly in calves, pigs and poultry. PK: Due to their highly polar and cationic nature aminoglycosides are poorly absorbed from intestinal tract and undergo negligible biotransformation when administered parenterally. Its pharmacokinetic profile is similar for humans and animal species. After oral administration the absorption of neomycin in calves was minimal (1%–11%) and about 90% was recovered in faeces (70%–80% as the parent compound). The urinary recovery of paromomycin (aminosidine) ranges from 80%–100% of total administered dose [26,27]. Environment: Fate and behavior studies in environment as well as monitoring results on their occurrence in surface or wastewater were not found. However, aminoglycosides are of special interest because of their potential impact on the spread and maintenance of antimicrobial resistance. It has been demonstrated that bacteria (enterococci) isolates from environmental samples (groundwater, wastewater) were resistant against gentamicin and streptomycin [28]. |
| AMPHENICOLS | Characterization: The most used active substance for this group is florfenicol, which is administered to farm animals by intramuscular (IM) route or in feed, mainly in cattle and porcine. PK: Cattle have similar kinetic profile as pigs. Florfenicol is absorbed completely and rapidly though the gastrointestinal (GI) tract in pigs, however oral bioavailability reported in broiler chickens is lower (55%–71%). After administration in farm animals four metabolites were identified (florfenicol-amine, florfenicol alcohol, florfenicol oxamid acid and monocloride florfenicol) but the only relevant microbiologically active residue is florfenicol. The main elimination route of florfenicol is urine, with 45%–60% of a dose excreted as parent drug, 11%–17% as florfenicol-amine, <10% was eliminated as florfenicol oxamid acid and 1% as florfenicol alcohol [29,30,31,32]. Environment: Fate and behavior studies in environment as well as monitoring results on their occurrence in soil, surface or wastewater are scarce. However, considering an open environmental assessment for the veterinary medicinal product Nuflor™, florfenicol showed an average biotransformation in soil of 13 days and in water greater than 28 days (d). The degradation by hydrolysis or photolysis (direct or indirect) was not significant and the degree of soil sorption was also considered low [33]. According to Subbiah et al. [34] residual florfenicol in soil can exert selective effect on bacteria (E. coli). In occurrence studies florfenicol was not detected. |
| CEPHALOSPORINS | Characterization: Cephalosporines are mainly used in Portugal in human medicine (Table 1) available mostly as tablets, granules or capsules to treat urinary tract infections due their high concentration in urine. In veterinary species they are frequently used for intramammary treatment of clinical mastitis in lactating cattle (Table 1), although ceftiofur is also available for administration by intramuscular route in porcine and bovine. PK: The pharmacokinetic profile of cephalosporins may vary between molecules and between species. The major route of excretion is renal filtration with the exception of cephalothin, cephapirin, and cefotaxime (which are deacetylated) and ceftriaxone and cefoperazone which are eliminated by bile. In some instances, about 90% of active substance is excreted unchanged [35,36]. Environment: Cephalosporines are subject to hydrolytic cleavage of the β-lactam ring under alkaline conditions being less sensitive to hydrolysis than penicillins. Similar to penicillins and tetracyclines, they can be complexed with cations and accumulate in sewage sludge and sediments [37], what may explain the bacterial resistance (E. coli) against cephalosporins observed in sewage treatment plants (e.g. cefuroxime) [38]. Cephalosporins are degraded with half-lives <5 d in the water sediment. However the degradation rate in the sediment was substance specific, for instance, cefradine was degraded in 0.87 t½ (d) and cefuroxime in 2.6 t½ (d). Abiotic processes seem to play an important role in the elimination of cephalosporins in surface water. The degradation of cephalosporins in environment is higher at higher pH values [39]. Hydrolysis of ceftiofur in soils was accelerated by increasing pH; the t½ values at pH 5.7 and 9 were ± 100, 8 and 4 days respectively, at 22 °C [40,41]. |
| MACROLIDES | Characterization: In human medicine the most predominant macrolides were azithromycin, clarithromycin. Tylosin and tilmicosin are solely used in veterinary medicine mainly in drinking water and incorporated in feed. Spiramycin and erythromycin are used for both human and animal practice. PK: Macrolides are quite lipophilic molecules that undergo extensive hepatic metabolism. Metabolism of erithromycin is via hepatic microsomal enzymes and only 2%–5% of orally administered erythromycin is excreted in active form in the urine. Clarithromycin is eliminated by renal and non-renal mechanisms and metabolized to several metabolites. In urine the amount of clarithromycin excreted unchanged ranges from 20%–40%, and 10%–15% of the dose is excreted as 14-hydroxyclarithromycin. Azithromycin undergoes some hepatic metabolism to inactive metabolites, but biliary excretion is the major route of elimination [42,43,44]. Oral bioavailability of tylosin is low with 22.5% in pigs and 30%–34% in broiler chickens. Tylosin is metabolized by liver and excreted mainly via faeces with only 6% of the parent compound being excreted by urine. Several compounds (tylosin A, tylosin Factor B, C and D and metabolite) may quantitatively differ between formulations and species. Tilmicosin has slow and low absorption (22%). After administration of a single oral dose of tilmicosin to pigs about 80% is excreted in faeces and 15% in urine. Similarly, in sheep, 72% are excreted in faeces and 13% in urine [45,46,47]. Environment: Their extensive administration in farm animals, could lead to contamination of manure and the subsequent contamination of soils whenever used as fertilizer. Schuesener et al. [48] studied the persistence of erythromycin, tylosin and roxithromycin in soil. Degradation half-lives of 20 days and 8 days were determined for erythromycin and tylosin respectively, however the concentration of roxithromycin remained unchanged after 120 days. Considering this study erythromycin and tylosin are not persistent in soil. Also in manure tylosin and tilmicosin were not detected being rapidly degraded. Both active substances are weak bases that are unstable under alkaline and acid conditions but are relatively stable at neutral conditions. The same review concluded that tylosin sorption in sediment also increased by the raising in ionic strength and is very immobile in sandy loam and loam sediments [48]. Therefore, their hydrolysis depends of the physical-chemical properties of manure and sludge. It has been reported that tylosin and erythromycin does not modify the ribosomal mechanism responsible for resistance of soil environmental bacteria [48,49,50]. In environment, erythromycin is transformed in dehydro-erythromycin. Azithromycin, clarithromycin and roxithromycin are degraded via direct photolysis. Azithromycin under photolysis turns into other photo products which exhibit a lower antibiotic activity than the parent compound [51,52]. Loads of spiramycin, dehydro-erythromycin and roxithromycin in treated wastewater were generally lower than in untreated water, because they can bind to sludge and will be released later [53]. |
| LINCOSAMIDES | Characterization: Lincosamides are basic compounds with high lipid solubility which include lincomycin and clindamycin. Both substances are used in human and veterinary medicine. Lincomycin is administered mainly in hospital sector and in drinking water or in feed for animals destined to human consumption. In humans lincomycin is used orally but has currently been largely replaced by clindamycin. PK: Lincosamides are well absorbed from intestines of non-herbivores and are eliminated mainly by hepatic metabolism (e.g., S-oxidation, de-methylation) although about 20% is excreted as unchanged form by urine. Lincomycin oral absorption in swine was in the range of 20%–50%, similarly to humans. The main component of the urine in human is the unchanged lincomycin (4%–17%). In pigs, about 11%–21% was excreted into the urine: 50% as unchanged lincomycin, and trace amounts of N-demethyl lincomycin. Chickens treated orally showed that excreta contained 80% lincomycin, ≤10% lincomycin sulfoxide, ≤5%N-demethyl lincomycin [42,54,55]. Clindamycin metabolites are much like those described for lincomycin. The bile is the major excretion route of clindamycin. About 28% excreted by the liver in the glucuronide form, 28% as clindamycin sulfoxide, and 9% as N-demethyl clindamycin [42]. Environment: The pKa of these substances is about 7.6, indicating that they will exist partially in the cation form which could absorb more strongly to soils containing organic carbon and clay than their neutral counterparts. No behavior studies in environment could be found for these substances. Studies demonstrated that clindamycin degraded in the sediment with a half-life of 1.0–1.6 days [56,57]. |
| PENICILLINS | Characterization: Penicillins are widely used in human health care and hospital sector such as in veterinary medicine in target animals. Amoxicillin is the most frequently administered active substance from this class (~147 tonnes 2010 and 2011). PK: Amoxicillin is usually administered by oral route in humans and animals due to its good absorption from the GI tract. This good absorption is attributed to its free amino group and zwitterionic ionisation at physiological pH, as amoxicillin exhibit low lipophilicity. Large interspecies differences in oral bioavailability of amoxicillin have been reported ranging from 10% in horses to 25%–30% in pigs and calves, while 63% in chickens and 75% to 80% in humans. Renal excretion (tubular secretion) is the primary elimination route as for most β-lactam antibiotics and their metabolites. Penicillin G, Penicillin V, ticarciline and aminopenicillins are metabolized to some extent by hydrolysis of the β-lactam ring. The two major metabolites found are amoxicilloic acid (AMA) and amoxicillindiketopiperazine-2',5'-dione (DIKETO) [58,59,60,61]. Environment: Penicillins have a similar environmental behavior as cephalosporins. They are highly polar substances and consequently would not be expected to sorb readily to soil components. Due to the chemically unstable β-lactam ring they are susceptible to hydrolysis and therefore easily degraded [62]. However, they have been occasionally detected in the environment. |
| PLEUROMUTILINS | Characterization: The most used active substances for this group are tiamulin and valnemulin, both consumed solely in veterinary medicine. PK: Tiamulin undergoes extensive metabolism in the liver and no antimicrobial activity was observed for 67% of the metabolites. Metabolite 8-α-hydroxymutilin was identified as residue marker. In dogs 33% of tiamulin dose was excreted in the urine [63,64,65]. Valnemulin is also excreted rapidly, mostly via the bile and faeces (around 87% of the total dose). In pigs valnemulin is extensively metabolized and excretion of parent molecule and metabolites occurs mainly via bile (73%–95% of the daily dose of total was recovered from the faeces). In rabbits, valnemulin is extensively metabolized with the same metabolites being found as in pigs [66,67,68]. Environment: Schuesener et al., studied the persistence of tiamulin in soil. Degradation half-lives of 16 days were determined. Considering this study tiamulin could not be seen as persistent in soil. However, in manure tiamulin was detected for about 180 days, showing no degradation. Thus, the soil will be contaminated with tiamulin if the manure is used as fertilizer [48]. |
| POLYMYXINS | Characterization: The polymyxins are a group of N-monoacetylated decapeptides that includes polymyxin B and polymyxin E (colistin). These peptides are considered as basic surface-active cationic detergents with an ability to intercalate in phospholipid which results in cell death (bactericidal properties). Despite being available for clinical use since the 1950s, the increasing prevalence of multidrug resistance worldwide has led to the resurgence of its use. PK: Polymyxins are not absorbed from the GI tract. Therefore systemic therapy in humans requires that a parenteral preparation (either polymyxin B sulfate or colistin methanesulphonate) be injected. Polymyxins are slowly excreted unchanged by glomerular filtration in urine. On the basis of the findings in earlier studies, approximately 60% of the dose of polymyxin B was recovered in urine [42,55,69]. Although it has been used regularly in veterinary medicine for decades for the treatment of GI infections, its use is now being questioned given the ever growing need for antimicrobials for the treatment of multidrug-resistant infection in humans. Colistin sulphate is poorly absorbed after oral administration and following parenteral administration excretion is via urine with no detectable residues found in faeces [70,71]. Environment: One study demonstrated the resistance of Staphylococcus aureus isolates from medical hospital, veterinary hospital, and slaughterhouse waste effluents against colistin [72]. |
| QUINOLONES | Characterization: The most predominant active substances are: ciprofloxacin, levofloxacin, moxifloxacin, norfloxacin, ofloxacin and prulifloxacin solely aplicated in human medicine and enrofloxacin and flumequine administered in food animals, mainly in drinking water. Interestingly ciprofloxacin although not authorized for animal use is an important active metabolite of enrofloxacin. PK: The majority of fluoroquinolones are primarily excreted in urine as the parent compound, by glomerular filtration or tubular secretion. However urinary excretion of these quinolones may be highly variable presenting 7% to 90% as fraction of unchanged drug excreted in urine. Ciprofloxacin and levofloxacin, the most representative fluoroquinolones for human use, are mainly eliminated by kidneys presenting 33%–60% and 61%–83% of urinary excretion, respectively. Only 15% of a ciprofloxacin dose has been reported to be recovered in feces. In contrast, moxifloxacin exhibit very little renal elimination. Only 20% of a moxifloxacin dose was recovered unchanged in the urine. After an oral dose 25% of a moxifloxacin dose was recovered as unchanged drug in feces partially. Furthermore, prulifloxacin is a prodrug of the active metabolite ulifloxacin. The proportion of ofloxacin dose excreted in urine is about 70% [73,74,75,76]. Enrofloxacin is metabolized to ciprofloxacin via deethylation of the ethyl group on the piperazine ring. About 13%–60% of enrofloxacin is metabolized into ciprofloxacin (20%–50%) and smaller amounts (around 10%) of oxygenated or dealkylated ciprofloxacin and hydrolylated enrofloxacin. Although in pigs there were only small traces, in cattle, the proportion of ciprofloxacin metabolized from enrofloxacin in plasma has been measured as high as 25%. Therefore around 90% of the applied enrofloxacin is excreted into environment in the active form either as parent compound or its active metabolite ciprofloxacin [42,77]. In addition, oral bioavailability in broiler chickens is 57% for norfloxacin, 64% for enrofloxacin and 69% for ciprofloxacin [60]. Environment: Except for prulifloxacin, quinolones exhibit large chemical stability, do not degrade with increased temperature and do not undertake hydrolysis. Fluoroquinolones, despite the low octanol-water coefficient values, bind strongly to sludge, probably through metal complexes interactions, influencing their solubility. Ofloxacin, compared to other fluoroquinolones, adsorbs strongly to different types of soils and is not easily released. A recent study demonstrated that their degradation could be enhanced under nitrifying conditions. Prulifloxacin showed extensive degradation in hydrolytic and oxidative processes, being stable to thermal and photolytic stress conditions [78,79,80]. |
| SULPHONAMIDES + TRIMETHOPRIM | Characterization: The sulfonamides have been applied in composite drugs with trimethoprim (TMP). PK : The treated humans and animals excrete the sulphonamides as parent compound or as acetylated derivatives. All sulfonamides (except the enteric compounds oral use) are excreted by kidneys. Orally used sulfonamides are primarily excreted via feces, with little or no drug being absorbed systemically. Acetylation is the major pathway of sulfonamides in most species, and apparently acetylated metabolites are the major urinary metabolites in cattle, sheep and swine. Glucuronide conjugation and aromatic hydroxylation are two additional metabolic pathways by which sulfonamides are metabolized in animals. In calves and cows, sulfadiazine is acetylated to a great degree with lower concentration of 4-hydroxysulfadiazine and with no glucuronides or 5-hydroxy derivatives detected in this species [81,82,83,84,85,86]. In pigs, two main metabolites were reported N-acetylsulfadiazine and 4-hydroxysulfadiazine and two minor metabolites identified as N-formylsulfadiazine and N-acetyl-4-hydroxysulfadiazine. Sulfadiazine accounted for 44% of the 96% excreted [87]. TMP is rapidly absorbed after oral administration, being the oral bioavailability reported as 79% in chickens and 73% in pig [83,84,85]. The greater part of the dose is excreted as unchanged drug and around 10%–20% of a dose is metabolized and metabolites are mainly excreted in the urine as conjugates. The fraction of TMP dose excreted as unchanged compound in humans is 69% ± 17 % while in veterinary use reported values varies from 2% in goat, 3% in cow, 10% in horses, 16% in pigs and 20% in dogs [55,88]. Environment: Sulphonamides are polar and hydrophilic, being transferred into the aquatic environment. They are considered the most mobile of antibiotics. They exhibit weak sorption to soil and so does not easily adsorb onto activated carbon. In environment the most predominant sulphonamide was sulfamethoxazole . In the waste water could be found the parent compound and its metabolite N4-acetylsulfamethoxazole, which it is assumed to be retransformed to the antibiotic itself by abiotic processes. After sulphametazine acetylation, the acetylated compound undergoes conjugation to form the glucuronide of N-4-acetylated sulfamethazine. Sulfamethazine metabolite, glucuronide of N-4-acetylated sulfamethazine is converted back to the parent drug in liquid manure. The abiotic degradation (indirect photolysis) found to be the main elimination pathway. They are classified as photo- and thermally stable substances at the degradation half-life (DT50) > 1 year [81,89,90,91]. TMP was also found to be susceptible to indirect photolysis in wastewater effluents. However is highly stable to direct photolysis and very stable photo-transformation products are generated during this process [92]. |
| TETRACICLYNES | Characterization: Doxycycline has gained importance due of its higher bioavailability relative to other tetracyclines. In veterinary medicine this broad-spectrum antibacterials are mainly used in drinking water and in food for animals. PK: Biovailability vary between species and molecules. In pigs, oxytetracycline, tetracycline and chlortetracycline biovailability is very low: 3%, 18%, 11% pigs, respectively [93]. In broiler chickens and pigs oral bioavailability of doxycycline was 41%, 48%, respectively, which was lower than that found in humans (90%–95%) [60,94]. The primary route of elimination for most tetracyclines is the kidney (with doxycycline being an important exception) although they are also excreted by bile into intestines, where they are partially reabsorbed via enterohepatic recirculation. Minoclycline is an exception and is significantly metabolized in the liver with low renal clearance. The tetracyclines (except minocycline and doxycycline) are excreted unchanged in the urine by glomerular filtration. About 10%–35% of a dose of oxytetracycline is excreted in active form in the urine and 20%–60% of a dose of tetracycline was reported. In most domestic animals biotransformation of the tetracyclines seems to be limited, and generally about one-third of a given dose is excreted unchanged. Sheep excrete nearly 21% of an oral dose of oxytetracycline, and young bulls excrete about 17%–75% of chlortetracycline as parent compound. Doxycycline and minocycline may be more extensively biotransformed than other tetracyclines (up to 40% of a given dose). Generally 50%–80% of a given dose is recoverable from the urine. Intestinal (biliary) elimination is always significant, it is commonly ~10%–20%, even with parenteral administration; for doxycycline and its metabolites, and minoclycline, this is the major route of excretion [43,95,96]. Environment: The tetracyclines just like penicillins, readily precipitate as complex forms with cations, as calcium and magnesium and were accumulated in sludge or sediment. Studies have shown that tetracyclines can absorb strongly to different kind of soil (loam, clay), sediment, being considered immobile, like enrofloxacin, ciprofloxacin and ofloxacin [97]. Szatmári et al., found degradation half-lives of 66.5 days for doxycycline in soil fertilized with liquid manure. Smaller values have been reported for oxytetracicline and for tetracycline: about 17 days and 22 days respectively. This difference indicates a higher adsorption to the soil particles of doxycycline. Temperature, UV and pH might contribute to the degradation of tetracyclines [98,99]. |
| BACITRACIN | Characterization: Bacitracin zinc is an antibiotic from the group of peptides widely used in veterinary medicine mainly incorporate in feed of farm animals (1.7 tonnes for 2012). PK: After oral application bacitracin is hardly absorbed from gastrointestinal tract and approximately 95% of an oral dose is excreted via faeces (only 3% or less via urine). It is metabolized to amino acids and smaller peptides via the main metabolite desamidobacitracin (inactive). Main metabolites in faeces are bacitracin A, B1, B2, F, desamidobacitracin and catabolic peptides [100]. Environment: Given its low stability constant, bacitracin zinc would be expected to dissociate into zinc ions and free bacitracin under environmental conditions [81]. |
| OTHER: HOSPITAL USE | Characterization: Imipenem, linezolide, piperacillin and vancomycin are miscellaneous antibacterial mostly used in hospital settings for serious infections. Imipenem is a carbapenem antibiotic with a broad spectrum of activity particularly useful for critically ill patients with septic complications due to unidentified bacteria or bacteria resistant to many other antibiotics. Linezolid belongs to a new class of antibacterial agents, the oxazolidinones, which are active against a variety of gram-positive pathogenic bacteria, including methicillin-resistant strains of Staphylococcus aureus. Piperacillin is an extended-spectrum penicillin which combines a ureidopenicillin and a beta-lactamase inhibitor. Vancomycin primarily administered via the intravenous route in the critical care setting in human medicine. In this setting, vancomycin has been mainly used to treat infections where MRSA is the presumed cause of infection. PK: Imipenem is rapidly metabolized by the renal brush border enzyme dehydropeptidase I. It is therefore co-administered as a one-to-one combination with the enzyme inhibitor cilastatin, which results in the excretion of about 70% of unchanged imipenem into the urine [101]. Linezolid and the two main metabolites have been reported to be predominantly eliminated by the kidneys. Its urinary recovery (% of dose) after 24 hours was much as 51.3% [102,103]. Both piperacillin and tazobactam undergo hepatic metabolism; two metabolites formed by beta-lactam ring cleavage contribute little (10%) to the overall elimination of piperacillin and about 26% of an metabolite of tazobactam was found in healthy volunteers. The recovery of piperacillin and tazobactam in urine over 12 h was 42.5% and 60.0% of the corresponding administered doses, respectively. Biliary route may also play important role piperacillin elimination [104]. Due to its poor absorption after oral administration vancomycin may also be used in some GI conditions (e.g., Clostridium difficile diarrhea). With normal renal function, about 90% of intravenous vancomycin is eliminated unchanged in the urine during the first 24 h by glomerular filtration. By intravenous route no vancomycin was observed in faeces [105]. Environment: Data about the fate and degradation in environment was not found. Vancomycin was the only one detected in wastewater and surface water (see Table 3). |
| Active substance | Marketing authorization in Portugal | Environmental concentration levels per sphere (ng/L or µg/kg) | European country [Reference] | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hum. Med. | Vet. Med. | Wastewater influent | Wastewater effluent | Surface water | Groundwater | Drinking water | Sediment/sludge | Soil | Manure/Dung | ||
| AMPHENICOLS | |||||||||||
| Florfenicol | X | n.d. | EU [106] | ||||||||
| CEPHALOSPORINS | |||||||||||
| Cephalexin | X | 283 | n.d. | n.d. | Serbia [106] | ||||||
| MACROLIDES | |||||||||||
| Azithromycin | X | 101.0 | France [26] | ||||||||
| Clarithromycin | X | 319.0 | 374 | 20.3 | 0.16 | Italy [27,110,111] | |||||
| Erythromycin | X | X | 12.0 | 52.0 | 15.9 | 0.18 | Italy [27,110] | ||||
| Roxithromycin | X | 0.56 | Germany [29] | ||||||||
| Spiramycin | X | X | 603.0 | 454.0 | 74.20 | 0.66 | Italy [27,28] | ||||
| Tilmicosin | X | 0.46 | 0.35 HS | 2.5 | n.d. | Italy [27,107] | |||||
| Tylosin | X | n.d. | n.d. HS | 2.77 | n.d. | Italy [27,28,107] | |||||
| LINCOSAMIDES | |||||||||||
| Lincomycin | X | X | 9.7 | 6.1 | 248.9 | n.d. | Italy [27,28] | ||||
| Clindamycine | X | X | 277 | EU [106] | |||||||
| PENICILLINS | |||||||||||
| Amoxicillin | X | X | 18.0 | n.d. | 5.7 | n.d. | Italy [27] | ||||
| Ampicillin | X | X | 252 | Portugal [116] | |||||||
| Benzylpenicillin | X | 127 | Portugal [116] | ||||||||
| Phenoxymethylpenicillin | X * | 122 | EU [106] | ||||||||
| Cloxacillin | X | n.d. | Germany [30] | ||||||||
| Dicloxacillin | X | n.d. | Germany [30] | ||||||||
| QUINOLONES | |||||||||||
| Ciprofloxacin | X | 443 | 7.3 3 | 4.6 | Spain [31,109] | ||||||
| Enrofloxacin | X | 447.1 | 211.5 HS | 102.5 | Portugal [28,117,118] | ||||||
| Norfloxacin | X | 37 | n.d. | Portugal [28] | |||||||
| Ofloxacin | X | 1903 | 2.7 | 3.3 | Spain [31,115] | ||||||
| Danofloxacin | X | n.d. | n.d. HS | Italy [107] | |||||||
| Flumequine | X | 25.7 | EU [106] | ||||||||
| SULFONAMIDES AND TRIMETHOPRIM | |||||||||||
| Sulfadiazine | X | X | 4.33 | Germany [29] | |||||||
| Sulfamethoxazole | X | 11.60 | 0.40 | 0.48 | 410 | Germany [29,30,120] | |||||
| Sulfadoxine | X | n.d. | EU [106] | ||||||||
| Trimethoprim | X | X | 0.32 | Germany [30] | |||||||
| TETRACYCLINES | |||||||||||
| Oxitetracycline | n.d. | 136 | Germany [30,129] | ||||||||
| Doxycycline | X | X | 6.7 HS | Sweden [119] | |||||||
| Tetracycline | X | n.d. | Germany [30] | ||||||||
| OTHERS | |||||||||||
| Vancomycin H | X | 41 | 40 | 4.8 | n.d. | Italy [107] | |||||
| Rank 1 | Amount (tonnes) | % Exc. Unchanged form | Occ. | Rank 2 | Amount (tonnes) | % Exc. Unchanged form | Occ. | Rank 3 | Amount (tonnes) | % Exc. Unchanged form | Occ. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ciprofloxacin | 16.90 | 60 | + | Oxytetracycline | 59.87 | 35 | + | Amoxicillin | 146.6 | 90 | + |
4. Discussion
5. Conclusions
Abbreviations
| DDD | defined daily dose |
| DGAV | Portuguese National Authority for Animal Health |
| EMA | European Medicines Agency |
| ESVAC | European Surveillance of Veterinary Antimicrobial Consumption |
| EU | European Union |
| H | ospital exclusive use |
| HS | Hospital samples |
| INFARMED | Portuguese National Authority of Medicines and Health Products |
| n.d. | not detectable |
| n.f. | not found in our literature review |
| PEC | Predicted Environmental Concentration |
| STPs | sewage treatment plants |
| TMP | trimethoprim |
| WHO | World Health Organization |
| WWTPs | Wastewater treatment plants |
| + | found |
| +H | also hospital use |
| % Exc. | Unchanged form refers to the proportion of the molecule excreted through urine as parent compound |
Author Contributions
Conflicts of Interest
References
- Neu, H.C.; Gootz, T.D. Medical Microbiology; Samuel Baron: University of Texas Medical Branch at Galveston, TX. USA, 1996. Chapter 11. 4th ed. Available online: http://www.ncbi.nlm.nih.gov/books/NBK7986/ (accessed on 1 April 2014).
- Aminov, R.I. A brief history of the antibiotic era: Lessons learned and challenges for the future. Front. Microbiol. 2010, 1, 1–7. [Google Scholar]
- Alanis, A.J. Resistance to antibiotics: Are we in the post-antibiotic era? Arch. Med. Res. 2005, 36, 697–705. [Google Scholar]
- Kalghatgi, S.; Spina, C.S.; Costello, J.C.; Liesa, M.; Morones-Ramirez, J.R.; Slomovic, S.; Molina, A.; Shirihai, O.S.; Collins, J.J. Bactericidal Antibiotics Induce Mitochondrial Dysfunction and Oxidative Damage in Mammalian Cells. Sci. Transl. Med. 2013, 192, 192–85. [Google Scholar]
- Seifrtová, M.; Nováková, L.; Lino, C.; Pena, A.; Solich, P. An overview of analytical methodologies for the determination of antibiotics in environmental waters. Anal. Chim. Acta 2009, 649, 158–179. [Google Scholar]
- Ding, C.; He, J. Effect of antibiotics in the environment on microbial populations. Appl. Microbiol. Biotechnol. 2010, 87, 925–941. [Google Scholar]
- Iglesias, A.; Nebot, C.; Miranda, J.M.; Vázquez, B.I.; Abuín, C.M.F.; Cepeda, A. Determination of the Presence of Three Antimicrobials in Surface Water Collected from Urban and Rural Areas. Antibiotics 2013, 2, 46–57. [Google Scholar]
- Sukul, P.; Spiteller, M. Fluoroquinolone Antibiotics in the Environment. Rev. Environ. Contam. Toxicol. 2007, 191, 131–162. [Google Scholar]
- Meisel, M.L.; Costa, M.C.; Pena, A. Regulatory approach on environmental risk assessment. Risk management recommendations, reasonable and prudent alternatives. Ecotoxicology 2009, 18, 1176–1181. [Google Scholar]
- Boxall, A.B.A.; Fogg, L.; Blackwell, P.A.; Kay, P.; Pemberton, E.J. Review of Veterinary Medicines in the Environment. Available online: http://www.environment-agency.gov.uk (accessed on 1 April 2014).
- Kemper, N. Veterinary antibiotics in the aquatic and terrestrial environment. Ecol. Indic. 2008, 8, 1–13. [Google Scholar]
- De Garcia, S.O.; Pinto, P.P.; Encina, P.G.; Mata, R.I. Consumption and occurrence of pharmaceutical and personal care products in the aquatic environment in Spain. Sci. Total Environ. 2013, 444, 451–465. [Google Scholar]
- Lindberg, R. Determination of Antibiotics in the Swedish Environment with Emphasis on Sewage Treatment Plants. Ph.D. Thesis, Umeå University, Umeå, Sweden, 2006. [Google Scholar]
- Silva, L.J.G.; Lino, C.M.; Meisel, L.; Barceló, D.; Pena, A. Ecopharmacovigilance. In The Handbook of Environmental Chemistry, 1st ed.; Barcelo, D., Ed.; Springer-Verlag: Berlin, Germany; Heidelberg GmbH & Co. KG: New York, NY, USA, 2012; pp. 213–242. [Google Scholar]
- EC. Regulation (EU) Nº 1235/2010 (3) of the European Parliament and of the council of 15 December 2010 amending, as regards pharmacovigilance of medicinal products for human use, Regulation (EC) No 726/2004 laying down Community procedures for the authorisation and supervision of medicinal products for human and veterinary use and establishing a European Medicines Agency, and Regulation (EC) No 1394/2007 on advanced therapy medicinal products. Off. J. Eur. Union 2010, L348, 1–16. [Google Scholar]
- Moulin, G.; Cavalié, P.; Pellanne, I.; Chevance, A.; Millemann, Y.; Colin, P.; Chauvin, C. A comparison of antimicrobial usage in human and veterinary medicine in France from 1999 to 2005. J. Antimicrob. Chemother. 2008, 62, 617–625. [Google Scholar]
- Roos, V.; Gunnarsson, L.; Fick, J.; Larsson, D.G; Rudén, C. Prioritising pharmaceuticals for environmental risk assessment. Sci. Total Environ. 2012, 421–422, 102–110. [Google Scholar]
- De Souza, S.M.; Vasconcelos, E.C.; Dziedzic, M.; de Oliveira, C.M. Environmental risk assessment of antibiotics: An intensive care unit analysis. Chemosphere 2009, 7, 962–967. [Google Scholar]
- Direcção Geral de Veterinária (DGV). Relatório Nacional de Monitorização do Consumo de Antimicrobianos; DGV: Lisboa, Portugal, 2010. [Google Scholar]
- Direcção Geral de Veterinária (DGV). Relatório Nacional de Monitorização do Consumo de Antimicrobianos; DGV: Lisboa, Portugal, 2011. [Google Scholar]
- European Medicines agency (EMA). Revised ESVAC Reflection Paper on Collecting Data on Consumption of Antimicrobial Agents Per Animal Species, on Technical Units of Measurement and Indicators for Reporting Consumption of Antimicrobial Agents in Animals; EMA/286416/2012-Rev.1; Veterinary Medicines Division: London, UK, 2013. [Google Scholar]
- Medicines Statistics Reports; Market Monitorisation; 2010–2011; Infarmed–Portugal.
- Besse, J.P.; Kausch, C.B.; Garric, J. Exposure Assessment of Pharmaceuticals and Their Metabolites in the Aquatic Environment: Application to the French Situation and Preliminary Prioritization. J. Hum. Ecol. Risk Assess. 2008, 4, 665–695. [Google Scholar]
- Infomed—Data Base for Human Medicinal Products. Available online: http://www.infarmed.pt/infomed/inicio.php (accessed on 1 April 2014).
- Apifarma symposium—Data Base for Veterinary Medicinal Products. Available online: https://www.apifarma.pt/simposiumvet/Paginas/Pesquisaavancada.aspx (accessed on 1 April 2014).
- European Medicines Agency (EMA). European Public MRL Assessment Report (EPMAR)—Neomycin (including framyacetin) (all producing species), Committee for Medicinal Products for Veterinary Use: Cannary Wharf, London, UK, 2013.
- European Medicines Agency (EMA). Committee for Medicinal Products for Veterinary Use. Aminosidine Summary Report; Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1996. [Google Scholar]
- Rice, E.W.; Messer, J.W.; Johnson, C.H.; Reasoner, D.J. Occurrence of High-Level Aminoglycoside Resistance in Environmental Isolates of Enterococci. Appl. Environ. Microbiol. 1995, 61, 374–376. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products. Florfenicol (extension to pigs)—Summary Report (4). Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1999. [Google Scholar]
- European Medicines Agency (EMA). Florfenicol summary report (1); Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK. Available online: http://www.ema.europa.eu/docs/en_GB/ document_library/Maximum_Residue_Limits_-_Report/2009/11/WC500014274.pdf (accessed on 26 February 2014).
- Shen, J.; Wu, X.; Hu, D.; Jiang, H. Pharmacokinetics of florfenicol in healthy and Escherichia coli-infected broiler chickens. Res. Vet. Sci. 2002, 73, 137–140. [Google Scholar]
- Liu, J.; Fung, K.F.; Chen, Z.; Zeng, Z.; Zhang, J. Pharmacokinetics of florfenicol in healthy pigs and in pigs experimentally infected with Actinobacillus pleuropneumoniae. Antimicrob. Agents Chemother. 2003, 47, 820–823. [Google Scholar]
- Environmental Assessment for the Use of Nuflor® Injectable Solution in Cattle. 1996. Available online: http://www.fda.gov/ucm/groups/fdagov-public/@fdagov-av-gen/documents/document/ucm072358.pdf (accessed on 15 February 2014).
- Subbiah, M.; Mitchell, S.M.; Ullman, J.L.; Call, D.R. Lactams and Florfenicol Antibiotics Remain Bioactive in Soils while Ciprofloxacin, Neomycin, and Tetracycline Are Neutralize. Appl. Environ. Microbiol. 2011, 77, 7255–7260. [Google Scholar]
- European Medicines Agency (EMA); Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products. Ceftiofur—Summary Report; Cannary Wharf, London, UK. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Maximum_Residue_Limits__Report/2009/11/WC500011904.pdf (accessed on 14 February 2014).
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products, Cefquinome—Summary Report (extension to pigs); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1999. [Google Scholar]
- Christian, T.; Schneider, R.J.; Färber, H.A.; Skutlarek, D.; Meyer, M.T.; Goldbach, H.E. Determination of antibiotic residues in manure, soil, and surface water. Acta Hydrochim. Hydrobiol. 2003, 31, 36–44. [Google Scholar]
- Kuemmerer, K. Resistance in the environment. J. Antimicrob. Chemother. 2004, 54, 311–320. [Google Scholar]
- Jiang, M.; Wang, L.; Ji, R. Biotic and abiotic degradation of four cephalosporin antibiotics in a lake surface water and sediment. Chemosphere 2010, 80, 1399–1405. [Google Scholar]
- Gilbertson, T.J.; Hornish, R.E.; Jaglan, P.S.; Koshy, K.T.; Nappier, J.L.; Stahl, G.L.; Cazers, A.R.; Nappier, J.M.; Marc, F.; Kubicek, M.F. Environmental fate of ceftiofur sodium, a cephalosporin antibiotic. Role of animal excreta in its decomposition. J. Agric. Food Chem. 1990, 38, 890–894. [Google Scholar]
- Wang, P.; Yuan, T.; Hu, J.; Tan, Y. Determination of cephalosporin antibiotics in water samples by optimised solid phase extraction and high performance liquid chromatography with ultraviolet detector. Int. J. Environ. Anal. Chem. 2011, 91, 1267–1281. [Google Scholar]
- Papich, M.G.; Riviere, J.E. Chloramphenicol and derivatives, macrolides, lincosamides and miscellaneous antimicrobials. In Veterinary Pharmacology and Therapeutics, 8th ed.; Adams, H.R., Ed.; Iowa State University Press: Ames, IA, USA, 2001; pp. 868–897. [Google Scholar]
- Chambers, H.F. Antimicrobial agents. In Goodman & Gilman’s the Pharmacological Basis of Therapeutics, 10th ed.; Hardman, J.G., Limbird, L.E., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 1239–1271. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Erythomycin—Summary Report (2); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2000. [Google Scholar]
- Kowalski, C.; Roliński, Z.; Zań, R.; Wawron, W. Pharmacokinetics of tylosin in broiler chickens. Pol. J. Vet. Sci. 2002, 5, 127–130. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Tylosin—Summary Report (3); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1997. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Tilmicosin—(extension to chicken) Summary Report (2); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1997. [Google Scholar]
- Schluesener, M.P.; Bester, K. Persistence of antibiotics such as macrolides, tiamulin and salinomycin in soil. Environ. Pollut. 2006, 143, 565–571. [Google Scholar]
- Zhou, Z.; Raskin, L.; Zilles, J.L. Effects of Swine Manure on Macrolide, Lincosamide, and Streptogramin B Antimicrobial Resistance in Soils. Appl. Environ. Microbiol. 2010, 76, 2218–2224. [Google Scholar]
- Aust, M-O.; Godlinski, F.; Travis, G.R.; Hao, X.; McAllister, T.A.; Leinweber, P.; Thiele-Bruhn, S. Distribution of sulfamethazine, chlortetracycline and tylosin in manure and soil of Canadian feedlots after subtherapeutic use in cattle. Environ. Pollut. 2008, 156, 1243–1251. [Google Scholar]
- Tong, L.; Eichhorn, P.; Pérez, S.; Wanga, X.; Barceló, D. Photodegradation of azithromycin in various aqueous systems under simulated and natural solar radiation: Kinetics and identification of photoproducts. Chemosphere 2011, 83, 340–348. [Google Scholar]
- Vione, D.; Feitosa-Felizzola, J.; Minero, C.; Chiron, S. Phototransformation of selected human-used macrolides in surface water: Kinetics, model predictions and degradation pathways. Water Res. 2009, 43, 1959–1967. [Google Scholar]
- Göbel, A.; McArdell, C.S.; Joss, A.; Siegrist, H.; Giger, W. Giger W. Fate of sulfonamides, macrolides, and trimethoprim in different wastewater treatment technologies. Sci. Total Environ. 2007, 372, 361–371. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Lincomycin—Summary report (1); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1998. [Google Scholar]
- Guiguère, S. Lincosamides, pleuromutilins and, Streptogramins. In Antimicrobial Therapy in Veterinary Medicine, 4th ed.; Giguère, S., Prescott, J.F., Baggot, J.D., Walker, R.D., Dowling, P.M., Eds.; Blackwell Publishing: Ames, IA, USA, 2006; pp. 179–190. [Google Scholar]
- Wang, C.; Teppen, B.J.; Boyd, S.A.; Li, H. Sorption of Lincomycin at Low Concentrations from Water by Soils. Soil Sci. Soc. Am. J. 2012, 76, 1222–1228. [Google Scholar]
- Chenxi, W.; Alison, L.; Spongberg, A.L.; Witter, J.D. Determination of the persistence of pharmaceuticals in biosolids using liquid-chromatography tandem mass spectrometry. Chemosphere 2008, 73, 511–518. [Google Scholar]
- Vaden, S.L.; Riviere, J.E. Penicillin and related B-lactam antibiotics. In Veterinary Pharmacology and Therapeutics, 8th Ed.; Adams, H.R., Ed.; Iowa State University Press: Ames, IA, USA, 2001; pp. 818–827. [Google Scholar]
- Reyns, T.; De Boever, S.; Schauvliege, S.; Gasthuys, F.; Meissonnier, G.; Oswald, I.; de Backer, P.; Croubels, S. Influence of administration route on the biotransformation of amoxicillin in the pig. J. Vet. Pharmacol. Ther. 2008, 32, 241–248. [Google Scholar]
- Anadon, A.; Martínez-Larranaga, M.R. Residues of antimicrobial drugs and feed additives in animal products: Regulatory aspects. Livest. Prod. Sci. 1999, 59, 183–198. [Google Scholar]
- Li, M.; Anderson, G.D.; Phillips, B.R.; Kong, W.; Shen, D.D.; Wang, J. Interactions of amoxicillin and cefaclor with human renal organic anion and peptide transporters. Drug Metab. Dispos. 2006, 34, 547–555. [Google Scholar]
- European Medicines Agency (EMA). Reflection paper on use of pleuromutilins in food-producing animals in the European Union: Development of resistance and impact on human and animal health; Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2012. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Tiamulin—Summary Report (2) (extension to rabbits); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2000. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Tiamulin—Summary Report (1); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1999. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Tiamulin—Summary Report (3), The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2008.
- European Medicines Agency (EMA); Committee for Medicinal Products for Veterinary Use. European Public MRL Assessment Report (EPMAR)—Valnemulin (rabbits), 2010; [EMA/CVMP/137196/2009].
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Valnemulin—Summary Report (1); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1998.
- Valnemulin—Summary of Product Characteristics. Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/veterinary/medicines/000042/vet_med_000116.jsp&mid=WC0b01ac058008d7a8 (accessed on 28 February 2014).
- Abdelraouf, K.; He, J.; Ledesma, K.R.; Hu, M.; Tam, V.H. Pharmacokinetics and renal disposition of polymyxin B in an animal model. Antimicrob. Agents Chemother. 2012, 56, 5724–5727. [Google Scholar]
- European Medicines Agency (EMA). Use of colistin products in animals within the European Union: Development of resistance and possible impact on human and animal health, The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2013.
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Colistin—Summary Report, The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1995.
- Ahaduzzaman, Md.; Hassan, M.M.; Alam, M.; Islam, S.K.M.A.; Uddin, I. Antimicrobial Resistance Pattern against Staphylococcus aureus in Environmental Effluents. Res. J. Vet. Pract. 2014, 2, 13–16. [Google Scholar]
- Sousa, J.; Alves, G.; Fortuna, A.; Falcão, A. Third- and Fourth-generation Fluoroquinolone Antibacterials: A Systematic Review of Safety and Toxicity Profiles. Curr. Drug Saf. 2014. [Google Scholar] [CrossRef]
- Aminimanizani, A.; Beringer, P.; Jelliffe, R. Comparative pharmacokinetics and pharmacodynamics of the newer fluoroquinolone antibacterials. Clin. Pharmacokinet. 2001, 40, 169–187. [Google Scholar]
- Zhanel, G.G.; Noreddin, A.M. Pharmacokinetics and pharmacodynamics of the new fluoroquinolones: Focus on respiratory infections. Curr. Opin. Pharmacol. 2001, 1, 459–463. [Google Scholar]
- Immanuel, C.; Hemanthkumar, A.K.; Gurumurthy, P.; Venkatesan, P. Dose related pharmacokinetics of ofloxacin in healthy volunteers. Int. J. Tuberc. Lung Dis. 2002, 6, 1017–1022. [Google Scholar]
- Slana, M.; Dolenc, M.S. Environmental Risk Assessment of antimicrobials applied in veterinary medicine-A field study and laboratory approach. Environ. Toxicol. Pharmacol. 2013, 35, 131–141. [Google Scholar]
- Jerker, F.; Soedersrtroem, H.; Linderberg, R.H.; Phan, C.; Tysklind, M.; Larsson, D.G.J. Contamination of surface, ground, and drinking water from pharmaceutical production. Environ. Toxicol. Chem. 2009, 28, 2522–2527. [Google Scholar]
- Dorival-García, N.; Zafra-Gómez, A.; Navalón, A.; González-López, J.; Hontoria, E.; Vílchez, J.L. Removal and degradation characteristics of quinolone antibiotics in laboratory-scale activated sludge reactors under aerobic, nitrifying and anoxic conditions. J. Environ. Manag. 2013, 120, 75–83. [Google Scholar]
- Raju, B.; Ramesh, M.; Srinivas, R.; Raju, S.S.; Venkateswarlu, Y. Removal and degradation characteristics of quinolone antibiotics in laboratory-scale activated sludge reactors under aerobic, nitrifying and anoxic conditions. Identification and characterization of stressed degradation products of prulifloxacin using LC-ESI-MS/Q-TOF, MSn experiments: Development of a validated specific stability-indicating LC-MS method. J. Pharm. Biomed. Anal. 2011, 56, 560–568. [Google Scholar]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar]
- Sittig, S.; Kasteel, R.; Groeneweg, J.; Hofmann, D.; Thiele, B.; Köppchen, S.; Vereecken, H. Dynamics of transformation of the veterinary antibiotic sulfadiazine in two soils. Chemosphere 2014, 95, 470–477. [Google Scholar]
- Baert, K.; De Baere, A.; Croubles, D.; Gasthuys, F.; De Backer, C. Pharmacokinetics and bioavailability of sulfadiazine and trimethoprim (trimazin 30%) after oral adminstration in non-fasted young pigs. J. Vet. Pharmacol. Ther. 2001, 24, 295–298. [Google Scholar]
- Baert, K.; De Beare, S.; Croubles, S.; De Backer, P. Pharmacokinetics and oral bioavailability of sulfadiazine and trimethoprim in broiler chickens. Vet. Res. Commun. 2003, 27, 301–309. [Google Scholar]
- Abu-Basha, E.A.; Gehring, R.; Hantash, T.M.; Al-Shunnaq, A.F.; Idkaidek, N.M. Parmacokinetics and bioavailability of sulfadiazine and trimethoprim following intravenous, intramuscular and oral administration in ostriches (Struthio camelus). J. Vet. Pharmacol. Ther. 2009, 32, 258–263. [Google Scholar]
- Spoo, J.W.; Riviere, J.E. Sulphonamides. In Veterinary Pharmacology and Therapeutics, 8th ed.; Adams, H.R., Ed.; Iowa State University Press: Ames, IA, USA, 2001; pp. 796–817. [Google Scholar]
- Lamshöft, M.; Sukul, P.; Zühlke, S.; Spiteller, M. Metabolism of 14C-labelled and non-labelled sulfadiazine after administration to pigs. Anal. Bioanal. Chem. 2007, 388, 1733–1745. [Google Scholar]
- Straub, J.O. An Environmental Risk Assessment for Human—Use Trimethoprim in European Surface Waters. Antibiotics 2013, 2, 115–162. [Google Scholar]
- Göbel, A.; McArdell, C.S.; Joss, A.; Siegrist, H.; Giger, W. Fate of sulfonamides, macrolides, and trimethoprim in different wastewater treatment technologies. Sci. Total Environ. 2007, 372, 361–371. [Google Scholar]
- Zhang, Y.; Xu, J.; Zhong, Z.; Guo, C.; Li, L.; He, Y.; Fan, W.; Chen, Y. Degradation of sulfonamides antibiotics in lake water and sediment. Environ. Sci. Pollut. Res. 2013, 20, 2372–2380. [Google Scholar]
- Baran, W.; Adamek, E.; Ziemiańska, J.; Sobczak, A. Effects of the presence of sulfonamides in the environment and their influence on human health. J. Hazard. Mater. 2011, 196, 1–15. [Google Scholar]
- Sirtori, C.; Aguera, A.; Gernjak, W.; Malato, S. Effect of water-matrix composition on trimetoprim solar photodegradation kinetics and pathways. Water Res. 2010, 44, 2735–2744. [Google Scholar]
- Nielsen, P.; Gyrd-Hansen, N.B. Bioavailability of oxytetracycline, tetracycline and chlortetracycline after oral administration to fed and fasted pigs. J. Vet. Pharmacol. Ther. 1996, 19, 305–311. [Google Scholar]
- Anadon, A.; Martinez-Larranaga, M.R.; Diaz, M.J.; Bringas, P.; Fernandez, M.C.; Fernandez-Cruz, M.L.; Iturbe, J.; Martinez, M.A. Pharmacokinetics of doxycycline in broiler chickens. Avian Pathoogy 1994, 23, 79–90. [Google Scholar]
- European Medicines Agency (EMA). Oxitetracycline, tetracycline, chlortetracycline—Summary report (1); Committee for Veterinary Medicinal Products/The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 1999.
- Monforts, M.H.M.M. The Trigger in the Environmental Risk Assessment for (veterinary) Medicines in the European Union: A Critical Appraisal 2005, RIVM report 601500002/2005.
- Tolls, J. Sorption of veterinary pharmaceuticals in soils: A review. Environ. Sci. Technol. 2001, 35, 3397–3406. [Google Scholar]
- Szatmári, I.; Barcza, T.; Koermoeczy, P.S.; Laczay, P. Ecotoxicological assessment of doxicycline in soil. J. Environ. Sci. Health Part B 2012, 47, 129–135. [Google Scholar]
- Wu, X.; Wei, Y.; Zheng, J.; Zhao, X.; Zhong, W. The behavior of tetracyclines and their degradation products during swine manure composting. Bioresour. Technol. 2011, 102, 5924–5931. [Google Scholar]
- European Medicines Agency (EMA). Committee for Veterinary Medicinal Products Bacitracin (extension to rabbits): Summary report (3); The European Agency for the Evaluation of Medicinal Products: Cannary Wharf, London, UK, 2002.
- Tegeder, I.; Bremer, F.; Oelkers, R.; Schobel, H.; Schüttler, J.; Brune, K.; Geisslinger, G. Pharmacokinetics of imipenem-cilastatin in critically ill patients undergoing continuous venovenous hemofiltration. J. Antimicrob. Agents Chemother. 1997, 41, 2640–2645. [Google Scholar]
- Burkhardt, O.; Borner, K.; von der Höh, N.; Köppe, P.; Pletz, M.W.; Nord, C.E.; Lode, H. Single- and multiple-dose pharmacokinetics of linezolid and co-amoxiclav in healthy human volunteers. J. Antimicrob. Chemother. 2002, 50, 707–712. [Google Scholar]
- Slatter, J.G.; Stalker, K.; Feenstra, K.L.; Welshman, I.R.; Bruss, J.B.; Sams, J.P.; Johnson, M.G.; Sanders, P.E.; Hauer, M.J.; Faqerness, P.E.; et al. Pharmacokinetics, metabolism, and excretion of linezolid following an oral dose of [14C]linezolid to healthy human subjects. Drug Metab. Dispos. 2001, 29, 1136–1145. [Google Scholar]
- Westphal, J.F.; Brogard, J.M.; Caro-Sampara, F.; Adloff, M.; Blicklé, J.F.; Monteil, H.; Jehl, F. Assessment of biliary excretion of piperacillin-tazobactam in humans. Antimicrob. Agents Chemother. 1997, 41, 1636–1640. [Google Scholar]
- Cunha, B.A. Vancomycin revisited: A reappraisal of clinical use. Crit. Care Clin. 2008, 24, 393–420. [Google Scholar]
- Loos, R.; Carvalho, R.; António, D.C.; Comero, S.; Locoro, G.; Tavazzi, S.; Paracchini, B.; Ghiani, M.; Lettieri, T.; Blaha, L.; et al. EU-wide monitoring survey on emerging polar organic contaminants in wastewater treatment plant effluents. Water Res. 2013, 47, 6475–6487. [Google Scholar]
- Verlicchi, P.; Al Aukidy, M.; Galletti, A.; Petrović, M.; Barceló, D. Hospital effluent: Investigation of the concentrations and distribution of pharmaceuticals and environmental risk assessment. Sci. Total Environ. 2012, 430, 109–118. [Google Scholar]
- Santos, L.H.M.L.M.; Gros, M.; Rodriguez-Mozaz, S.; Delerue-Matos, C.; Pena, A.; Barceló, D.; Conceição, M.B.S.M. Montenegro. Contribution of hospital effluents to the load of pharmaceuticals in urban wastewaters: Identification of ecologically relevant pharmaceuticals. Sci. Total Environ. 2013, 461–462, 302–316. [Google Scholar]
- López-Serna, R.; Jurado, A.; Vázquez-Suñé, E.; Carrera, J.; Petrović, M.; Barceló, D. Occurrence of 95 pharmaceuticals and transformation products in urban groundwaters underlying the metropolis of Barcelona, Spain. Environ. Pollut. 2013, 174, 305–315. [Google Scholar]
- Calamari, D.; Zuccato, E.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Strategic survey of therapeutic drugs in the Rivers Po and Lambro in Northern Italy. Environ. Sci. Technol. 2003, 37, 1241–1248. [Google Scholar]
- Al Aukidy, M.; Verlicchi, P.; Jelić, A.; Petrović, M.; Barcelò, D. Monitoring release of pharmaceutical compounds: Occurrence and environmental risk assessment of two WWTP effluents and their receiving bodies in the Po Valley, Italy. Sci. Total Environ. 2012, 438, 15–25. [Google Scholar]
- Weigel, S.; Kallenborn, R.; Huhnerfuss, H. Simultaneous solid-phase extraction of acidic, neutral an.d. basic pharmaceuticals from aqueous samples at ambient (neutral) pH and their determination by gas chromatography-mass spectrometry. J. Chromatogr. A 2004, 1023, 183–195. [Google Scholar]
- Petrović, M.; Škrbić, B.; Živančev, J.; Ferrando-Climent, L.; Barcelo, D. Determination of 81 pharmaceutical drugs by high performance liquid chromatography coupled to mass spectrometry with hybrid triple quadrupole–linear ion trap in different types of water in Serbia. Serbia. Sci. Total Environ. 2014, 468–469, 415–428. [Google Scholar]
- Gómez, M.; Petrović, M.; Fernández-Alba, A.R.; Barceló, D. Determination of pharmaceuticals of various therapeutic classes by solid-phase extraction and liquid chromatography-tandem mass spectrometry analysis in hospital effluent wastewaters. J. Chromatogr. A 2006, 1114, 224–233. [Google Scholar]
- López-Roldán, R.; Alda, M.L.; Gros, M.; Petrović, M.; Martín-Alonso, J.; Barceló, D. Advanced monitoring of pharmaceuticals and estrogens in the Llobregat River basin (Spain) by liquid chromatography–triple quadrupole-tandem mass spectrometry in combination with ultra-performance liquid chromatography–time of flight-mass spectrometry. Chemosphere 2010, 80, 1337–1344. [Google Scholar]
- Salgado, R.; Marques, R.; Noronha, J.P.; Mexia, J.T.; Carvalho, G.; Oehmen, A.; Reis, M.A.M. Assessing the diurnal variability of pharmaceutical and personal care products in a full-scale activated sludge plant. Environ. Pollut. 2011, 159, 2359–2367. [Google Scholar]
- Seifrtova, M.; Pena, A.; Lino, C.M.; Solich, P. Determination of fluoroquinolone antibiotics in hospital and municipal wastewaters in Coimbra by liquid chromatography with a monolithic column and fluorescence detection. Anal. Bioanal. Chem. 2008, 391, 799–805. [Google Scholar]
- Pena, A.; Chmielova, D.; Lino, C.M.; Solich, P. Determination of fluorquinolone antibiotics in surface waters from Mon.d.ego River by high performance liquid chromatography using amonolithic column. J. Sep. Sci. 2007, 30, 2924–2928. [Google Scholar]
- Lindberg, R.; Jarnheimer, P.; Olsen, B.; Johansson, M.; Tysklind, M. Determination of antibiotic substances in hospital sewage water using solid phase extraction and liquid chromatography/mass spectrometry and group analogue internal standards. Chemosphere 2004, 57, 1479–1488. [Google Scholar]
- Sachera, F.; Langea, F.T.; Braucha, H.-J.; Blankenhornb, I. Pharmaceuticals in groundwaters. Analytical methods and results of a monitoring program in Baden-Württemberg, Germany. J. Chromatogr. A 2001, 938, 199–210. [Google Scholar]
- Golet, E.V.; Alder, A.C.; Hartmann, A.; Ternes, T.A.; Giger, W. Trace Determination of Fluoroquinolone Antibacterial Agents in Urban Wastewater by Solid-Phase Extraction and Liquid Chromatography with Fluorescence Detection. Anal. Chem. 2001, 73, 3632–3638. [Google Scholar]
- Golet, E.M.; Alder, A.; Giger, W. Environmental Exposure and Risk Assessment of Fluoroquinolone Antibacterial Agents in Wastewater and River Water of the Glatt Valley Watershed, Switzerland. Environ. Sci. Technol. 2002, 36, 3645–3651. [Google Scholar]
- Martínez-Carballo, E.; González-Barreiro, C.; Scharf, S.; Gans, O. Environmental monitoring study of selected veterinary antibiotics in animal manure and soils in Austria. Environ. Pollut. 2007, 148, 570–579. [Google Scholar]
- Vulliet, E.; Cren-Olivé, C. Screening of pharmaceuticals and hormones at the regional scale, in surface and groundwaters intended to human consumption. Environ. Pollut. 2011, 159, 2929–2934. [Google Scholar]
- Perret, D.; Gentili, A.; Marchese, S.; Greco, A.; Curini, R. Sulphonamide residues in Italian surface and drinking waters: a small scale reconnaissance. Chromatographia 2006, 63, 225–232. [Google Scholar]
- Madureira, T.V.; Barreiro, J.C.; Rocha, M.J.; Rocha, E.; Cass, Q.B.; Tiritan, M.T. Spatiotemporal distribution of pharmaceuticals in the Douro River estuary (Portugal). Sci. Total Environ. 2010, 408, 5513–5520. [Google Scholar]
- Haller, M.Y.; Müller, S.R.; McArdell, C.S.; Alder, A.C.; Suter, M.J.-F. Quantification of veterinary antibiotics (sulfonamides and trimethoprim) in animal manure by liquid chromatography–mass spectrometry. J. Chromatogr. A 2002, 952, 111–120. [Google Scholar]
- Bendz, D.; Paxéus, N.A.; Ginn, T.R.; Logec, F.J. Occurrence and fate of pharmaceutically active compounds in the environment, a case study: Höje River in Sweden. J. Hazard. Mater. 2005, 122, 195–204. [Google Scholar]
- Winckler, C.; Engels, H.; Hund-Rinke, K.; Luckow, T.; Simon, M.; Steffens, G. Verhalten von Tetracyclinen und anderen Veterinärantibiotika in Wirtschaftsdünger und Boden UFOPLAN 20073 248. 2003.
- Infarmed (Autoridade Nacional do Medicamento e Produtos de Saúde). Relatório do departamento dos medicamentos veterinários 2000–2007: o medicamento veterinário—abordagem analítica, Infarmed: Lisboa, Portugal, 2007.
- European Medicines Agency (EMA). Guideline on the Environmental Risk Assessment of Medical Products for Human Use—EMEA/CHMP/SWP/4447/00 corr 1*, Veterinary Medicines Division: London, UK.
- European Medicines Agency (EMA). Guideline on Environmental impact Assessment (EIAS) for Veterinary Medical Products—Phase I—CVMP/VICH/592/98-Final, London, UK, 2000.
- Mompelat, S.; le bot, B.; Thomas, O. Occurrence and fate of pharmaceutical products and by-products, from resource to drinking water. Environ. Int. 2009, 35, 803–814. [Google Scholar]
- Toutain, P.L.; Bousquét-Melou, A. Bioavailability and its assessment. J. Vet. Pharmacol. Ther. 2004, 27, 455–466. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Almeida, A.; Duarte, S.; Nunes, R.; Rocha, H.; Pena, A.; Meisel, L. Human and Veterinary Antibiotics Used in Portugal—A Ranking for Ecosurveillance. Toxics 2014, 2, 188-225. https://doi.org/10.3390/toxics2020188
Almeida A, Duarte S, Nunes R, Rocha H, Pena A, Meisel L. Human and Veterinary Antibiotics Used in Portugal—A Ranking for Ecosurveillance. Toxics. 2014; 2(2):188-225. https://doi.org/10.3390/toxics2020188
Chicago/Turabian StyleAlmeida, Anabela, Sofia Duarte, Ricardo Nunes, Humberto Rocha, Angelina Pena, and Leonor Meisel. 2014. "Human and Veterinary Antibiotics Used in Portugal—A Ranking for Ecosurveillance" Toxics 2, no. 2: 188-225. https://doi.org/10.3390/toxics2020188
APA StyleAlmeida, A., Duarte, S., Nunes, R., Rocha, H., Pena, A., & Meisel, L. (2014). Human and Veterinary Antibiotics Used in Portugal—A Ranking for Ecosurveillance. Toxics, 2(2), 188-225. https://doi.org/10.3390/toxics2020188







