Utilizing Multiple Behavioral Endpoints to Identify Negative Control Chemicals in a Larval Zebrafish Behavior Assay
Abstract
1. Introduction
2. Materials and Methods
2.1. Zebrafish Husbandry, Spawning, and Embryo Collection
2.2. Chemicals and Treatment
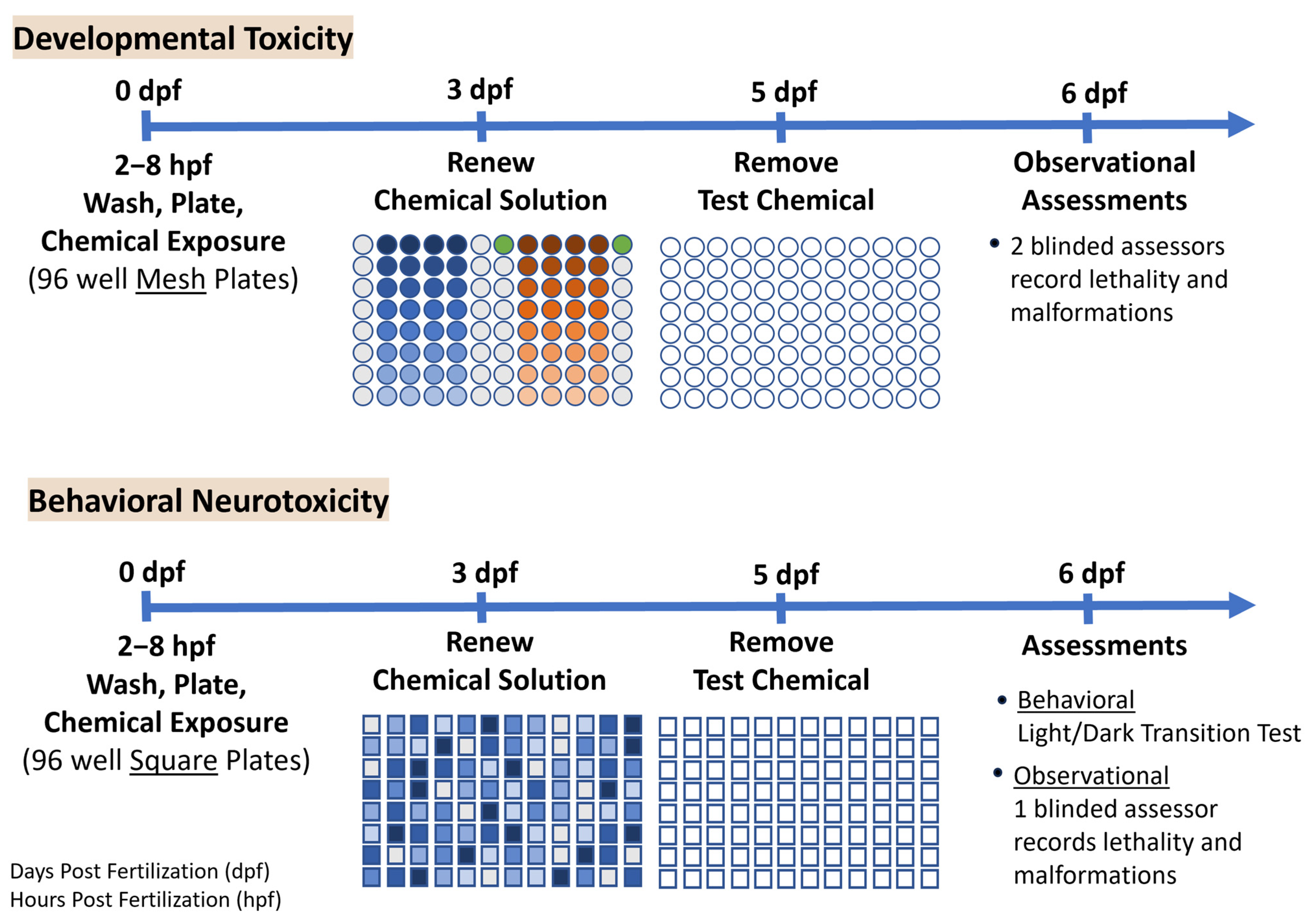
2.3. Developmental Toxicity Experimental Design
2.4. Behavioral Neurotoxicity Experimental Design
2.5. Data Analysis
2.5.1. Statistical Analysis
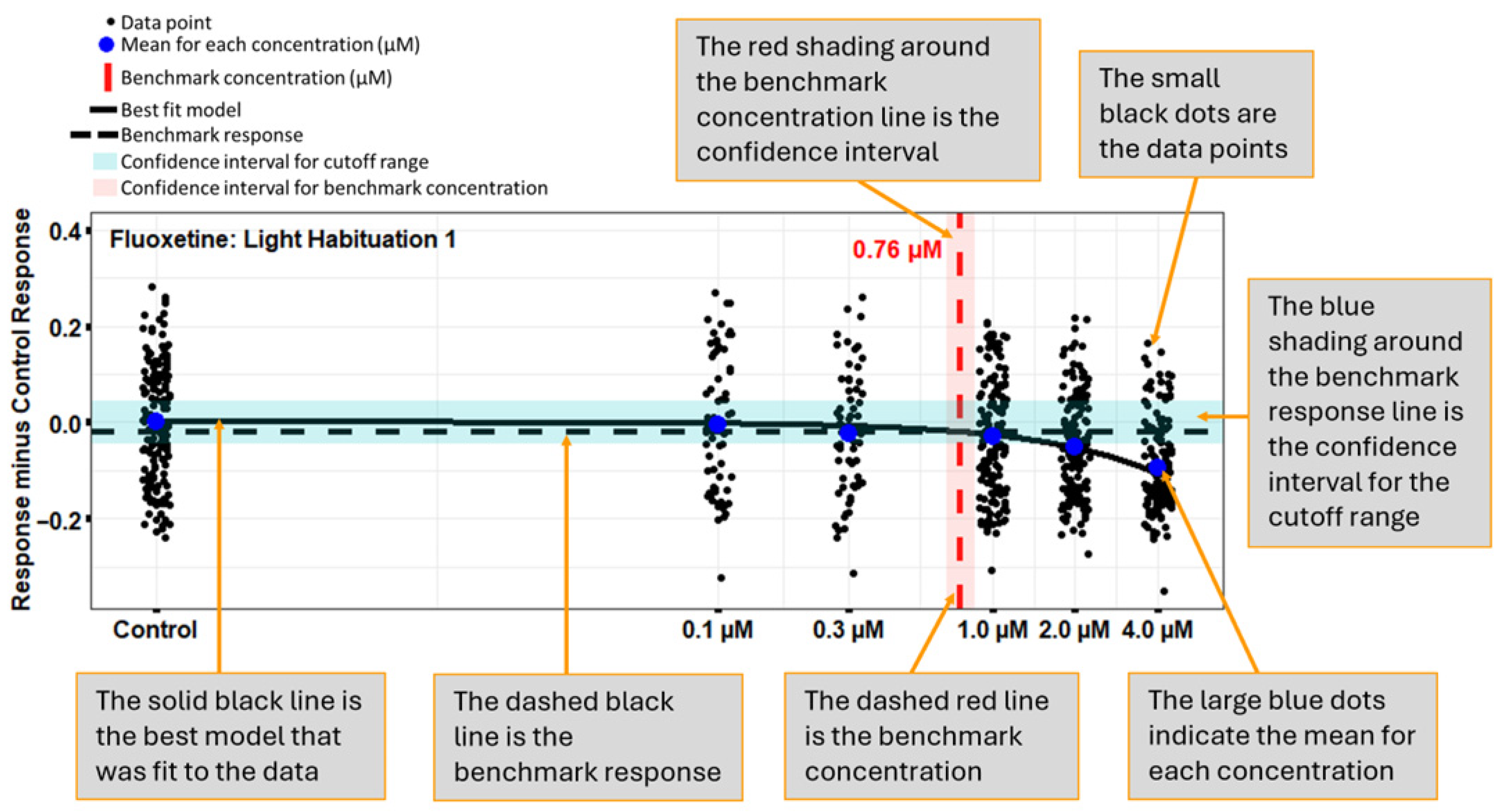
2.5.2. Concentration–Response Modeling (13-Endpoint Analysis)
3. Results
3.1. Fluoxetine: Positive Control
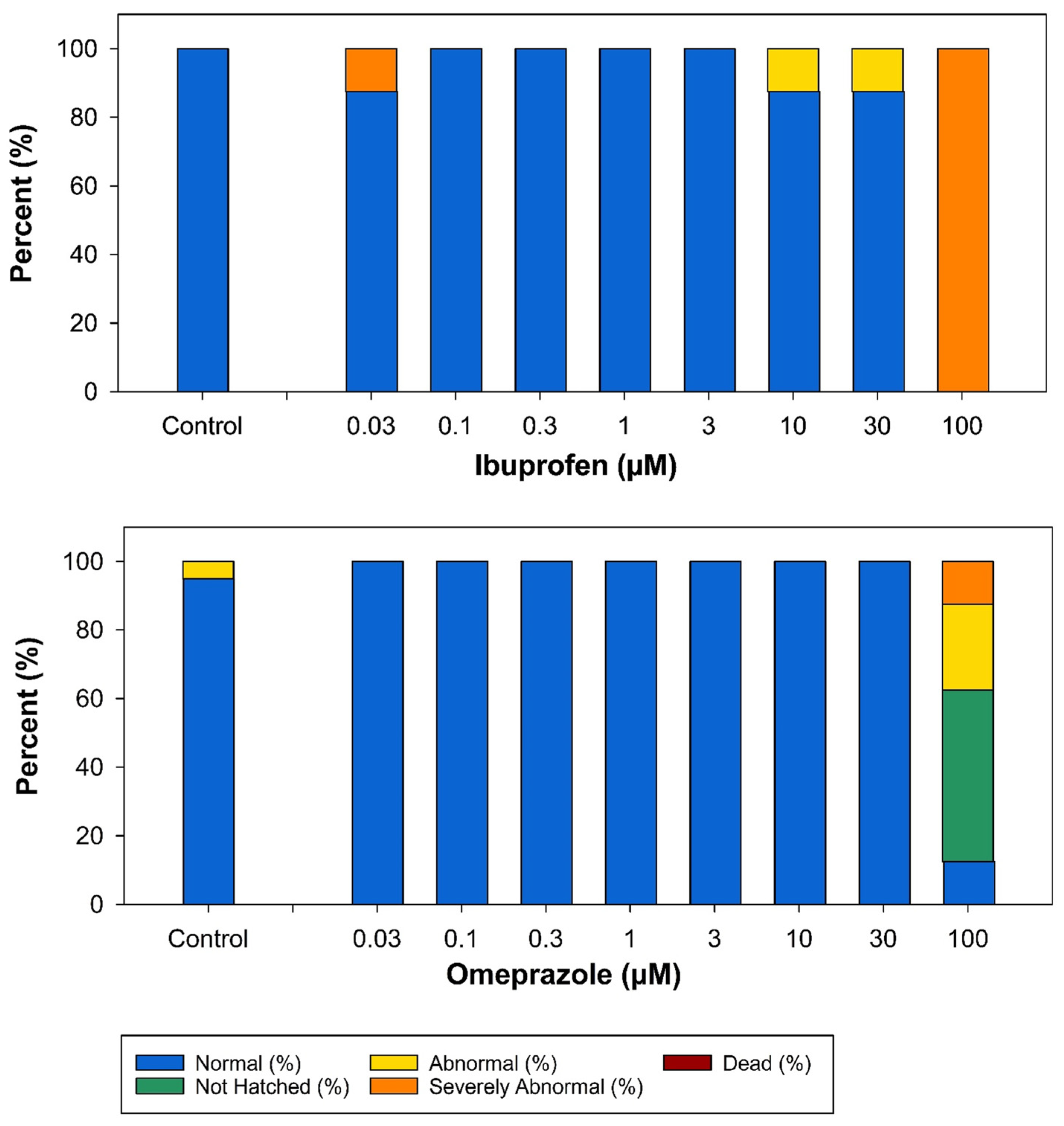
3.2. Developmental Toxicity Experiments
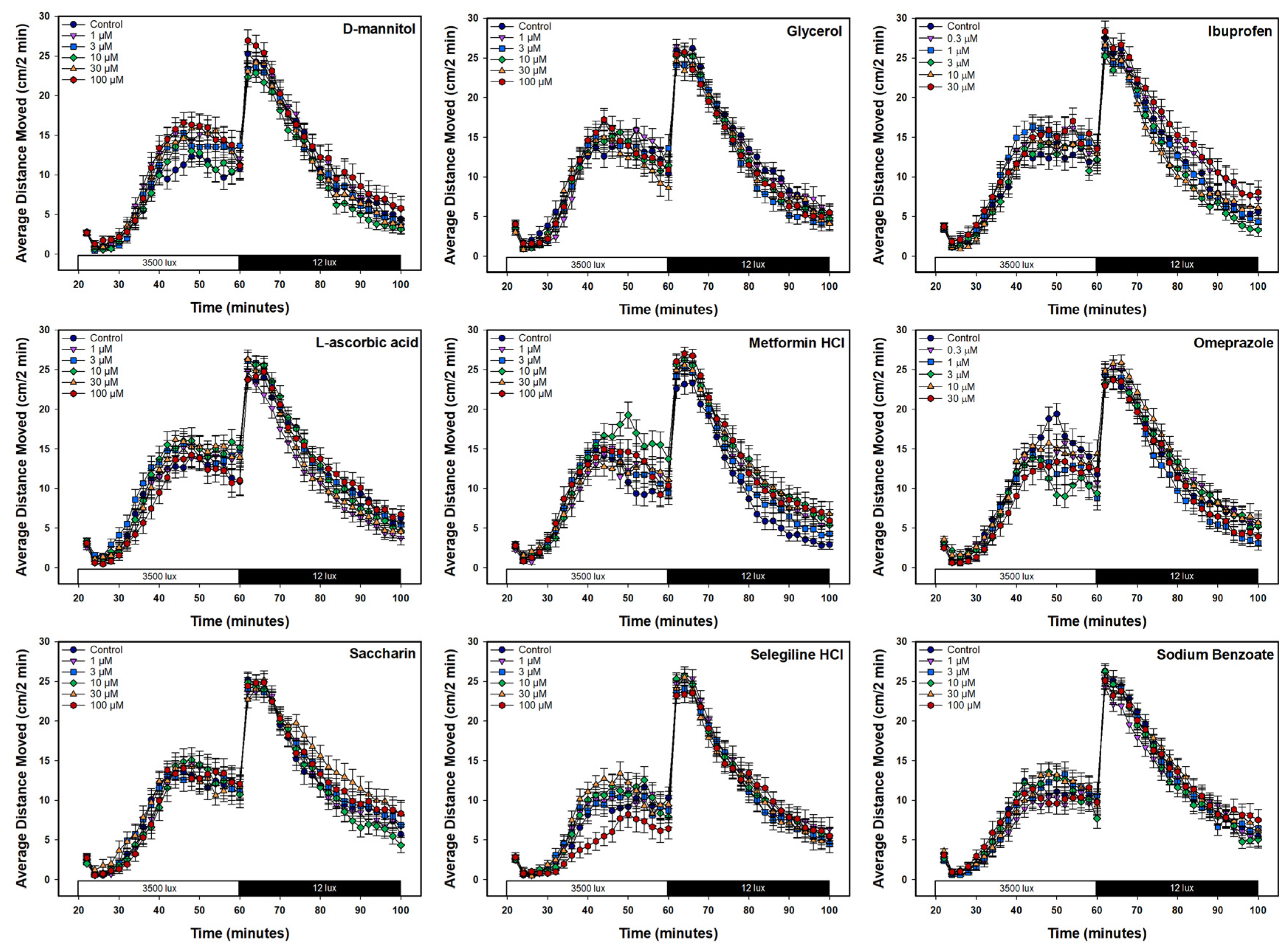
3.3. Behavioral Neurotoxicity Experiments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAALAC | American Association for Accreditation of Laboratory Animal Care |
| ANOVA | Analysis of Variance |
| AUC_r | Dark–Light Area Under the Curve |
| avgSD | Average Speed in the Dark |
| avgSL | Average Speed in the Light |
| avgST | Total Average Speed |
| AZ | Arizona |
| BMC | Benchmark Concentration |
| C | Centigrade |
| CA | California |
| CAS# | Chemical Abstracts Service Number |
| cm | Centimeter |
| D.C. | District of Columbia |
| dpf | Days Post Fertilization |
| DMSO | Dimethyl Sulfoxide |
| DTXSID | Distributed Structure Searchable Toxicity Substance Identifier |
| EPA | Environmental Protection Agency |
| FL | Florida |
| hbt1L | Light Habituation 1 |
| hbt1D | Dark Habituation 1 |
| hbt2L | Light Habituation 2 |
| hbt2D | Dark Habituation 2 |
| hpf | Hours Post Fertilization |
| L | Liter |
| LogKow | Octanol–Water Partition Coefficient |
| LSD | Fishers Least Significant Difference |
| lux | Luminous Flux per Unit Area |
| µM | Micromolar |
| µL | Microliter |
| µS | Micro Siemens |
| mM | Millimolar |
| mL | Milliliters |
| MA | Massachusetts |
| ME | Maine |
| MD | Maryland |
| MO | Missouri |
| n | Number of Independent Observations |
| NC | North Carolina |
| NH | New Hampshire |
| OECD | Organisation for Economic Co-operation and Development |
| PA | Pennsylvania |
| ppm | Parts per Million |
| RoAD | Dark Maximum–Minimum |
| RoAL | Light Maximum–Minimum |
| RTP | Research Triangle Park |
| strtlA | Startle Acceleration |
| strtlAavg | Adjusted Startle |
| strtlF | Startle Fold Change |
| TSCA | Toxic Substances Control Act |
| USA | United States of America |
| UT | Utah |
| VA | Virginia |
References
- Bellinger, D.C. An overview of environmental chemical exposures and neurodevelopmental impairments in children. Pediatr. Med. 2018, 1, 9. [Google Scholar] [CrossRef]
- Dórea, J.G. Exposure to environmental neurotoxic substances and neurodevelopment in children from Latin America and the Caribbean. Environ. Res. 2021, 192, 110199. [Google Scholar] [CrossRef]
- Huffling, K.; McLaughlin, J. Pediatric Chemical Exposure: Opportunities for Prevention. J. Pediatr. Health Care 2022, 36, 27–33. [Google Scholar] [CrossRef]
- Shahin, S.; Medley, E.A.; Naidu, M.; Trasande, L.; Ghassabian, A. Exposure to organophosphate esters and maternal-child health. Environ. Res. 2024, 252, 118955. [Google Scholar] [CrossRef] [PubMed]
- Crofton, K.M. Compendium of Regulatory-Type Studies That Were Conducted to Assess the Potential Developmental Neurotoxicity of Chemicals Draft Report; EFSA Study Report Contract Number EOI/EFSA/2022/01; EFSA: Parma, Italy, 2024. [Google Scholar]
- Miodovnik, A. Environmental neurotoxicants and developing brain. Mt. Sinai J. Med. 2011, 78, 58–77. [Google Scholar] [CrossRef] [PubMed]
- Ton, C.; Lin, Y.; Willett, C. Zebrafish as a model for developmental neurotoxicity testing. Birth Defects Res. A Clin. Mol. Teratol. 2006, 76, 553–567. [Google Scholar] [CrossRef]
- d’Amora, M.; Giordani, S. The Utility of Zebrafish as a Model for Screening Developmental Neurotoxicity. Front. Neurosci. 2018, 12, 976. [Google Scholar] [CrossRef]
- Hill, B.N.; Coldsnow, K.D.; Hunter, D.L.; Hedge, J.M.; Korest, D.; Jarema, K.A.; Padilla, S. Assessment of larval zebrafish locomotor activity for developmental neurotoxicity screening. In Experimental Neurotoxicology Methods; Humana Press: Totowa, NJ, USA, 2021; pp. 327–351. [Google Scholar]
- Gaballah, S.; Swank, A.; Sobus, J.R.; Howey, X.M.; Schmid, J.; Catron, T.; McCord, J.; Hines, E.; Strynar, M.; Tal, T. Evaluation of Developmental Toxicity, Developmental Neurotoxicity, and Tissue Dose in Zebrafish Exposed to GenX and Other PFAS. Environ. Health Perspect. 2020, 128, 47005. [Google Scholar] [CrossRef]
- Jarema, K.A.; Hunter, D.L.; Hill, B.N.; Olin, J.K.; Britton, K.N.; Waalkes, M.R.; Padilla, S. Developmental Neurotoxicity and Behavioral Screening in Larval Zebrafish with a Comparison to Other Published Results. Toxics 2022, 10, 256. [Google Scholar] [CrossRef]
- Yang, Q.; Deng, P.; Xing, D.; Liu, H.; Shi, F.; Hu, L.; Zou, X.; Nie, H.; Zuo, J.; Zhuang, Z.; et al. Developmental Neurotoxicity of Difenoconazole in Zebrafish Embryos. Toxics 2023, 11, 353. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Liu, Y.; Xuan, X.; Xu, Z.; Gao, P.; Jin, Z.; Hong, H.; Sun, H. Dihalogenated nitrophenols exposure induces developmental neurotoxicity in zebrafish embryo. Ecotoxicol. Environ. Saf. 2024, 277, 116359. [Google Scholar] [CrossRef]
- Nishimura, Y.; Murakami, S.; Ashikawa, Y.; Sasagawa, S.; Umemoto, N.; Shimada, Y.; Tanaka, T. Zebrafish as a systems toxicology model for developmental neurotoxicity testing. Congenit. Anom. 2015, 55, 1–16. [Google Scholar] [CrossRef]
- Planchart, A.; Mattingly, C.J.; Allen, D.; Ceger, P.; Casey, W.; Hinton, D.; Kanungo, J.; Kullman, S.W.; Tal, T.; Bondesson, M.; et al. Advancing toxicology research using in vivo high throughput toxicology with small fish models. ALTEX 2016, 33, 435–452. [Google Scholar] [CrossRef]
- Hughes, S.; Hessel, E.V.S. Zebrafish and nematodes as whole organism models to measure developmental neurotoxicity. Crit. Rev. Toxicol. 2024, 54, 330–343. [Google Scholar] [CrossRef] [PubMed]
- Field, H.A.; Ober, E.A.; Roeser, T.; Stainier, D.Y. Formation of the digestive system in zebrafish. I. Liver morphogenesis. Dev. Biol. 2003, 253, 279–290. [Google Scholar] [CrossRef]
- Tao, T.; Peng, J. Liver development in zebrafish (Danio rerio). J. Genet. Genom. 2009, 36, 325–334. [Google Scholar] [CrossRef]
- Katoch, S.; Patial, V. Zebrafish: An emerging model system to study liver diseases and related drug discovery. J. Appl. Toxicol. 2021, 41, 33–51. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef]
- Walter, K.M.; Miller, G.W.; Chen, X.; Yaghoobi, B.; Puschner, B.; Lein, P.J. Effects of thyroid hormone disruption on the ontogenetic expression of thyroid hormone signaling genes in developing zebrafish (Danio rerio). Gen. Comp. Endocrinol. 2019, 272, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Blanton, M.L.; Specker, J.L. The hypothalamic-pituitary-thyroid (HPT) axis in fish and its role in fish development and reproduction. Crit. Rev. Toxicol. 2007, 37, 97–115. [Google Scholar] [CrossRef]
- Alsop, D.; Vijayan, M. The zebrafish stress axis: Molecular fallout from the teleost-specific genome duplication event. Gen. Comp. Endocrinol. 2009, 161, 62–66. [Google Scholar] [CrossRef]
- Alsop, D.; Vijayan, M.M. Development of the corticosteroid stress axis and receptor expression in zebrafish. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R711–R719. [Google Scholar] [CrossRef]
- Porazzi, P.; Calebiro, D.; Benato, F.; Tiso, N.; Persani, L. Thyroid gland development and function in the zebrafish model. Mol. Cell. Endocrinol. 2009, 312, 14–23. [Google Scholar] [CrossRef]
- Heijlen, M.; Houbrechts, A.M.; Darras, V.M. Zebrafish as a model to study peripheral thyroid hormone metabolism in vertebrate development. Gen. Comp. Endocrinol. 2013, 188, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.D.; Cerutti, D.T. Behavioral Neuroscience of Zebrafish. In Methods of Behavior Analysis in Neuroscience, 2nd ed.; Buccafusco, J.J., Ed.; CRC Press/Taylor & Francis Group: Boca Raton, FL, USA, 2009; Chapter 15. [Google Scholar]
- de Esch, C.; Slieker, R.; Wolterbeek, A.; Woutersen, R.; de Groot, D. Zebrafish as potential model for developmental neurotoxicity testing: A mini review. Neurotoxicol. Teratol. 2012, 34, 545–553. [Google Scholar] [CrossRef]
- Legradi, J.; el Abdellaoui, N.; van Pomeren, M.; Legler, J. Comparability of behavioural assays using zebrafish larvae to assess neurotoxicity. Environ. Sci. Pollut. Res. Int. 2015, 22, 16277–16289. [Google Scholar] [CrossRef] [PubMed]
- Kiper, K.G.; Freeman, J.L. Zebrafish as a Tool to Assess Developmental Neurotoxicity. In Cell Culture Techniques; Aschner, M., Costa, L., Eds.; Springer: New York, NY, USA, 2019; pp. 169–193. [Google Scholar]
- Martin, M.M.; Baker, N.C.; Boyes, W.K.; Carstens, K.E.; Culbreth, M.E.; Gilbert, M.E.; Harrill, J.A.; Nyffeler, J.; Padilla, S.; Friedman, K.P.; et al. An expert-driven literature review of “negative” chemicals for developmental neurotoxicity (DNT) in vitro assay evaluation. Neurotoxicol. Teratol. 2022, 93, 107117. [Google Scholar] [CrossRef] [PubMed]
- Haigis, A.-C.; Ottermanns, R.; Schiwy, A.; Hollert, H.; Legradi, J. Getting more out of the zebrafish light dark transition test. Chemosphere 2022, 295, 133863. [Google Scholar] [CrossRef]
- Nasiadka, A.; Clark, M.D. Zebrafish breeding in the laboratory environment. ILAR J. 2012, 53, 161–168. [Google Scholar] [CrossRef]
- Westerfield, M. The Zebrafish Book; A Guide for the Laboratory Use of Zebrafish (Danio rerio), 4th ed.; University of Oregon Press: Eugene, OR, USA, 2007. [Google Scholar]
- Hedge, J.M.; Hunter, D.L.; Sanders, E.; Jarema, K.A.; Olin, J.K.; Britton, K.N.; Lowery, M.; Knapp, B.R.; Padilla, S.; Hill, B.N. Influence of methylene blue or dimethyl sulfoxide on larval zebrafish development and behavior. Zebrafish 2023, 20, 132–145. [Google Scholar] [CrossRef]
- MacPhail, R.C.; Brooks, J.; Hunter, D.L.; Padnos, B.; Irons, T.D.; Padilla, S. Locomotion in larval zebrafish: Influence of time of day, lighting and ethanol. Neurotoxicology 2009, 30, 52–58. [Google Scholar] [CrossRef]
- Dach, K.; Yaghoobi, B.; Schmuck, M.R.; Carty, D.R.; Morales, K.M.; Lein, P.J. Teratological and Behavioral Screening of the National Toxicology Program 91-Compound Library in Zebrafish (Danio rerio). Toxicol. Sci. 2019, 167, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Hagstrom, D.; Truong, L.; Zhang, S.; Tanguay, R.; Collins, E.S. Comparative Analysis of Zebrafish and Planarian Model Systems for Developmental Neurotoxicity Screens Using an 87-Compound Library. Toxicol. Sci. 2019, 167, 15–25. [Google Scholar] [CrossRef]
- Quevedo, C.; Behl, M.; Ryan, K.; Paules, R.S.; Alday, A.; Muriana, A.; Alzualde, A. Detection and prioritization of developmentally neurotoxic and/or neurotoxic compounds using zebrafish. Toxicol. Sci. 2019, 168, 225–240. [Google Scholar] [CrossRef]
- American Veterinary Medical Association (AVMA). AVMA Guidelines for the Euthanasia of Animals: 2020 Edition; Panel on Euthanasia; American Veterinary Medical Association (AVMA): Schaumburg, IL, USA, 2020; Available online: https://www.avma.org/resources-tools/avma-policies/avma-guidelines-euthanasia-animals (accessed on 28 November 2023).
- Rowson, Z.; Paul Friedman, K.; Setzer, R.W.; Padilla, S.; Carstens, K.E. A data analysis approach for locomotor behavioral responses in developmentally exposed zebrafish. NAM J. 2025, 1, 100033. [Google Scholar] [CrossRef]
- Box, G.E.P.; Cox, D.R. An analysis of transformations. J. R. Stat. Soc. Ser. B Methodol. 1964, 26, 211–243. [Google Scholar] [CrossRef]
- Sheffield, T.; Brown, J.; Davidson, S.; Friedman, K.P.; Judson, R. tcplfit2: An R-language general purpose concentration-response modeling package. Bioinformatics 2022, 38, 1157–1158. [Google Scholar] [CrossRef]
- Feshuk, M.; Kolaczkowski, L.; Dunham, K.; Davidson-Fritz, S.E.; Carstens, K.E.; Brown, J.; Judson, R.S.; Paul Friedman, K. The ToxCast pipeline: Updates to curve-fitting approaches and database structure. Front. Toxicol. 2023, 5, 1275980. [Google Scholar] [CrossRef] [PubMed]
- Britton, K.N.; Judson, R.S.; Hill, B.N.; Jarema, K.A.; Olin, J.K.; Knapp, B.R.; Lowery, M.; Feshuk, M.; Brown, J.; Padilla, S. Using Zebrafish to Screen Developmental Toxicity of Per- and Polyfluoroalkyl Substances (PFAS). Toxics 2024, 12, 501. [Google Scholar] [CrossRef]
- David, A.; Pancharatna, K. Developmental anomalies induced by a non-selective COX inhibitor (ibuprofen) in zebrafish (Danio rerio). Environ. Toxicol. Pharmacol. 2009, 27, 390–395. [Google Scholar] [CrossRef]
- Xia, L.; Zheng, L.; Zhou, J.L. Effects of ibuprofen, diclofenac and paracetamol on hatch and motor behavior in developing zebrafish (Danio rerio). Chemosphere 2017, 182, 416–425. [Google Scholar] [CrossRef]
- Sánchez-Aceves, L.M.; Perez-Alvarez, I.; Gomez-Olivan, L.M.; Islas-Flores, H.; Barcelo, D. Developmental alterations, teratogenic effects, and oxidative disruption induced by ibuprofen, aluminum, and their binary mixture on Danio rerio. Environ. Pollut. 2021, 291, 118078. [Google Scholar] [CrossRef]
- Chabchoubi, I.B.; Bouchhima, R.A.; Louhichi, N.; Baanannou, A.; Masmoudi, S.; Hentati, O. Short-term effects of various non-steroidal anti-inflammatory drugs (NSAIDs) on Danio rerio embryos. MethodsX 2023, 10, 102215. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.A.; Lopes, I.; Domingues, I.; Silva, D.; Blasco, J.; Pereira, J.L.; Araujo, C.V.M. Behavioural, developmental and biochemical effects in zebrafish caused by ibuprofen, irgarol and terbuthylazine. Chemosphere 2023, 344, 140373. [Google Scholar] [CrossRef] [PubMed]
- Junaid, M.; Wang, Y.; Hamid, N.; Deng, S.; Li, W.G.; Pei, D.S. Prioritizing selected PPCPs on the basis of environmental and toxicogenetic concerns: A toxicity estimation to confirmation approach. J. Hazard. Mater. 2019, 380, 120828. [Google Scholar] [CrossRef]
- Deeti, S.; O’Farrell, S.; Kennedy, B.N. Early safety assessment of human oculotoxic drugs using the zebrafish visualmotor response. J. Pharmacol. Toxicol. Methods 2014, 69, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.; Bai, Y.; Kreuder, F.; Saaristo, M.; Kaslin, J.; Wlodkowic, D. Sensory-Motor Perturbations in Larval Zebrafish (Danio rerio) Induced by Exposure to Low Levels of Neuroactive Micropollutants during Development. Int. J. Mol. Sci. 2022, 23, 8990. [Google Scholar] [CrossRef]
- Selderslaghs, I.W.; Hooyberghs, J.; Blust, R.; Witters, H.E. Assessment of the developmental neurotoxicity of compounds by measuring locomotor activity in zebrafish embryos and larvae. Neurotoxicol. Teratol. 2013, 37, 44–56. [Google Scholar] [CrossRef]
- Baek, S.H.; Lee, S.H. Omeprazole inhibits melanin biosynthesis in melan-a cells and zebrafish. Exp. Dermatol. 2016, 25, 239–241. [Google Scholar] [CrossRef]
- Hoyberghs, J.; Bars, C.; Ayuso, M.; Van Ginneken, C.; Foubert, K.; Van Cruchten, S. DMSO Concentrations up to 1% are Safe to be Used in the Zebrafish Embryo Developmental Toxicity Assay. Front. Toxicol. 2021, 3, 804033. [Google Scholar] [CrossRef]
- Luis, A.I.S.; Campos, E.V.R.; Oliveira, J.L.; Vallim, J.H.; Proenca, P.L.F.; Castanha, R.F.; de Castro, V.; Fraceto, L.F. Ecotoxicity evaluation of polymeric nanoparticles loaded with ascorbic acid for fish nutrition in aquaculture. J. Nanobiotechnol. 2021, 19, 163. [Google Scholar] [CrossRef]
- Hagedorn, M.; Kleinhans, F.W.; Wildt, D.E.; Rall, W.F. Chill sensitivity and cryoprotectant permeability of dechorionated zebrafish embryos, Brachydanio rerio. Cryobiology 1997, 34, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Tsay, H.J.; Wang, Y.H.; Chen, W.L.; Huang, M.Y.; Chen, Y.H. Treatment with sodium benzoate leads to malformation of zebrafish larvae. Neurotoxicol. Teratol. 2007, 29, 562–569. [Google Scholar] [CrossRef]
- Chen, Q.; Huang, N.N.; Huang, J.T.; Chen, S.; Fan, J.; Li, C.; Xie, F.K. Sodium benzoate exposure downregulates the expression of tyrosine hydroxylase and dopamine transporter in dopaminergic neurons in developing zebrafish. Birth Defects Res. B Dev. Reprod. Toxicol. 2009, 86, 85–91. [Google Scholar] [CrossRef]
- Maes, J.; Verlooy, L.; Buenafe, O.E.; de Witte, P.A.; Esguerra, C.V.; Crawford, A.D. Evaluation of 14 organic solvents and carriers for screening applications in zebrafish embryos and larvae. PLoS ONE 2012, 7, e43850. [Google Scholar] [CrossRef]
- Gaur, H.; Purushothaman, S.; Pullaguri, N.; Bhargava, Y.; Bhargava, A. Sodium benzoate induced developmental defects, oxidative stress and anxiety-like behaviour in zebrafish larva. Biochem. Biophys. Res. Commun. 2018, 502, 364–369. [Google Scholar] [CrossRef]
- Duy-Thanh, D.; Bich-Ngoc, N.; Van den Bossche, F.; Lai-Thanh, N.; Muller, M. Discovering Novel Bioactivities of Controversial Food Additives by Means of Simple Zebrafish Embryotoxicity (ZET) Assays. Toxics 2022, 11, 8. [Google Scholar] [CrossRef]
- Hill, A.J.; Bello, S.M.; Prasch, A.L.; Peterson, R.E.; Heideman, W. Water permeability and TCDD-induced edema in zebrafish early-life stages. Toxicol. Sci. 2004, 78, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Selderslaghs, I.W.; Van Rompay, A.R.; De Coen, W.; Witters, H.E. Development of a screening assay to identify teratogenic and embryotoxic chemicals using the zebrafish embryo. Reprod. Toxicol. 2009, 28, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Hermsen, S.A.; Pronk, T.E.; van den Brandhof, E.J.; van der Ven, L.T.; Piersma, A.H. Transcriptomic analysis in the developing zebrafish embryo after compound exposure: Individual gene expression and pathway regulation. Toxicol. Appl. Pharmacol. 2013, 272, 161–171. [Google Scholar] [CrossRef]
- Verstraelen, S.; Peers, B.; Maho, W.; Hollanders, K.; Remy, S.; Berckmans, P.; Covaci, A.; Witters, H. Phenotypic and biomarker evaluation of zebrafish larvae as an alternative model to predict mammalian hepatotoxicity. J. Appl. Toxicol. 2016, 36, 1194–1206. [Google Scholar] [CrossRef]
- Saputra, F.; Lai, Y.H.; Fernandez, R.A.T.; Macabeo, A.P.G.; Lai, H.T.; Huang, J.C.; Hsiao, C.D. Acute and Sub-Chronic Exposure to Artificial Sweeteners at the Highest Environmentally Relevant Concentration Induce Less Cardiovascular Physiology Alterations in Zebrafish Larvae. Biology 2021, 10, 548. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, J.; Cheng, V.; Reddam, A.; Avila-Barnard, S.; Volz, D.C. Triphenyl phosphate-induced pericardial edema is associated with elevated epidermal ionocytes within zebrafish embryos. Environ. Toxicol. Pharmacol. 2022, 89, 103776. [Google Scholar] [CrossRef]
- Wiegand, J.; Avila-Barnard, S.; Nemarugommula, C.; Lyons, D.; Zhang, S.; Stapleton, H.M.; Volz, D.C. Triphenyl phosphate-induced pericardial edema in zebrafish embryos is dependent on the ionic strength of exposure media. Environ. Int. 2023, 172, 107757. [Google Scholar] [CrossRef] [PubMed]
- Elizalde-Velazquez, G.A.; Gomez-Olivan, L.M.; Garcia-Medina, S.; Islas-Flores, H.; Hernandez-Navarro, M.D.; Galar-Martinez, M. Antidiabetic drug metformin disrupts the embryogenesis in zebrafish through an oxidative stress mechanism. Chemosphere 2021, 285, 131213. [Google Scholar] [CrossRef]
- Elizalde-Velazquez, G.A.; Gomez-Olivan, L.M.; Garcia-Medina, S.; Hernandez-Diaz, M.; Islas-Flores, H.; Galar-Martinez, M.; Garcia-Medina, A.L.; Chanona-Perez, J.J.; Hernandez-Varela, J.D. Polystyrene microplastics mitigate the embryotoxic damage of metformin and guanylurea in Danio rerio. Sci. Total. Environ. 2022, 852, 158503. [Google Scholar] [CrossRef] [PubMed]
- Barros, S.; Alves, N.; Pinheiro, M.; Ribeiro, M.; Morais, H.; Montes, R.; Rodil, R.; Quintana, J.B.; Coimbra, A.M.; Santos, M.M.; et al. Are Fish Populations at Risk? Metformin Disrupts Zebrafish Development and Reproductive Processes at Chronic Environmentally Relevant Concentrations. Environ. Sci. Technol. 2023, 57, 1049–1059. [Google Scholar] [CrossRef]
- Crago, J.; Klaper, R. Place-based screening of mixtures of dominant emerging contaminants measured in Lake Michigan using zebrafish embryo gene expression assay. Chemosphere 2018, 193, 1226–1234. [Google Scholar] [CrossRef]
- Banerjee, P.; Dutta, S.; Pal, R. Dysregulation of Wnt-Signaling and a Candidate Set of miRNAs Underlie the Effect of Metformin on Neural Crest Cell Development. Stem Cells 2016, 34, 334–345. [Google Scholar] [CrossRef][Green Version]
- Godoy, A.A.; Domingues, I.; Arsenia Nogueira, A.J.; Kummrow, F. Ecotoxicological effects, water quality standards and risk assessment for the anti-diabetic metformin. Environ. Pollut. 2018, 243, 534–542. [Google Scholar] [CrossRef]
- Barbieri, F.; Bosio, A.G.; Pattarozzi, A.; Tonelli, M.; Bajetto, A.; Verduci, I.; Cianci, F.; Cannavale, G.; Palloni, L.M.G.; Francesconi, V.; et al. Chloride intracellular channel 1 activity is not required for glioblastoma development but its inhibition dictates glioma stem cell responsivity to novel biguanide derivatives. J. Exp. Clin. Cancer Res. 2022, 41, 53. [Google Scholar] [CrossRef] [PubMed]
- Sallinen, V.; Sundvik, M.; Reenila, I.; Peitsaro, N.; Khrustalyov, D.; Anichtchik, O.; Toleikyte, G.; Kaslin, J.; Panula, P. Hyperserotonergic phenotype after monoamine oxidase inhibition in larval zebrafish. J. Neurochem. 2009, 109, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Faria, M.; Prats, E.; Bellot, M.; Gomez-Canela, C.; Raldua, D. Pharmacological Modulation of Serotonin Levels in Zebrafish Larvae: Lessons for Identifying Environmental Neurotoxicants Targeting the Serotonergic System. Toxics 2021, 9, 118. [Google Scholar] [CrossRef] [PubMed]



| Chemical | CAS # | DTXSID * | Molecular Weight (g/mole) | Octanol-Water Partition (LogKow) Predicted Median * | Octanol-Water Partition (LogKow) Predicted Range * | Purity (%) |
|---|---|---|---|---|---|---|
| D-Mannitol | 69-65-8 | DTXSID1023235 | 182.2 | −3.15 | −4.67 to −2.38 | ≥99.9 |
| Fluoxetine | 54910-89-3 | DTXSID7023067 | 309.3 | 4.16 | 4.05 to 4.65 | ≥98.0 |
| Glycerol | 56-81-5 | DTXSID9020663 | 92.1 | −1.79 | −2.32 to −1.65 | ≥99.0 |
| Ibuprofen | 15687-27-1 | DTXSID5020732 | 206.3 | 3.73 | 3.37 to 3.79 | 99.8 |
| L-Ascorbic acid | 50-81-7 | DTXSID5020106 | 176.1 | −2.15 | −3.85 to −1.85 | 99.2 |
| Metformin hydrochloride | 1115-70-4 | DTXSID9037246 | 165.6 | −1.12 | −2.31 to 0.32 | 99.9 |
| Omeprazole | 73590-58-6 | DTXSID6021080 | 345.4 | 2.20 | 2.09 to 3.40 | 99.9 |
| Saccharin | 81-07-2 | DTXSID5021251 | 183.2 | 0.81 | 0.45 to 0.91 | 99.6 |
| Selegiline hydrochloride | 14611-52-0 | DTXSID9044584 | 223.8 | 2.88 | 0.19 to 2.95 | 99.8 |
| Sodium benzoate | 532-32-1 | DTXSID1020140 | 144.1 | 0.16 | −2.27 to 1.90 | 99.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knapp, B.R.; Hunter, D.L.; Olin, J.K.; Padilla, S.; Jarema, K.A. Utilizing Multiple Behavioral Endpoints to Identify Negative Control Chemicals in a Larval Zebrafish Behavior Assay. Toxics 2025, 13, 727. https://doi.org/10.3390/toxics13090727
Knapp BR, Hunter DL, Olin JK, Padilla S, Jarema KA. Utilizing Multiple Behavioral Endpoints to Identify Negative Control Chemicals in a Larval Zebrafish Behavior Assay. Toxics. 2025; 13(9):727. https://doi.org/10.3390/toxics13090727
Chicago/Turabian StyleKnapp, Bridget R., Deborah L. Hunter, Jeanene K. Olin, Stephanie Padilla, and Kimberly A. Jarema. 2025. "Utilizing Multiple Behavioral Endpoints to Identify Negative Control Chemicals in a Larval Zebrafish Behavior Assay" Toxics 13, no. 9: 727. https://doi.org/10.3390/toxics13090727
APA StyleKnapp, B. R., Hunter, D. L., Olin, J. K., Padilla, S., & Jarema, K. A. (2025). Utilizing Multiple Behavioral Endpoints to Identify Negative Control Chemicals in a Larval Zebrafish Behavior Assay. Toxics, 13(9), 727. https://doi.org/10.3390/toxics13090727







