Do Surface Charges on Polymeric Filters and Airborne Particles Control the Removal of Nanoscale Aerosols by Polymeric Facial Masks?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
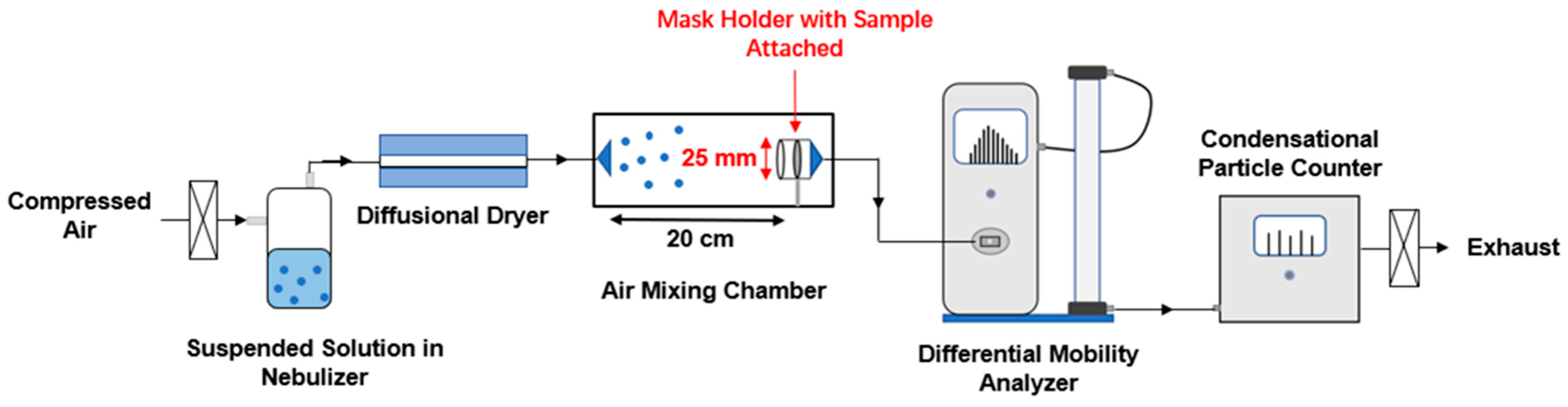
2.2. Filtration Efficiency Test
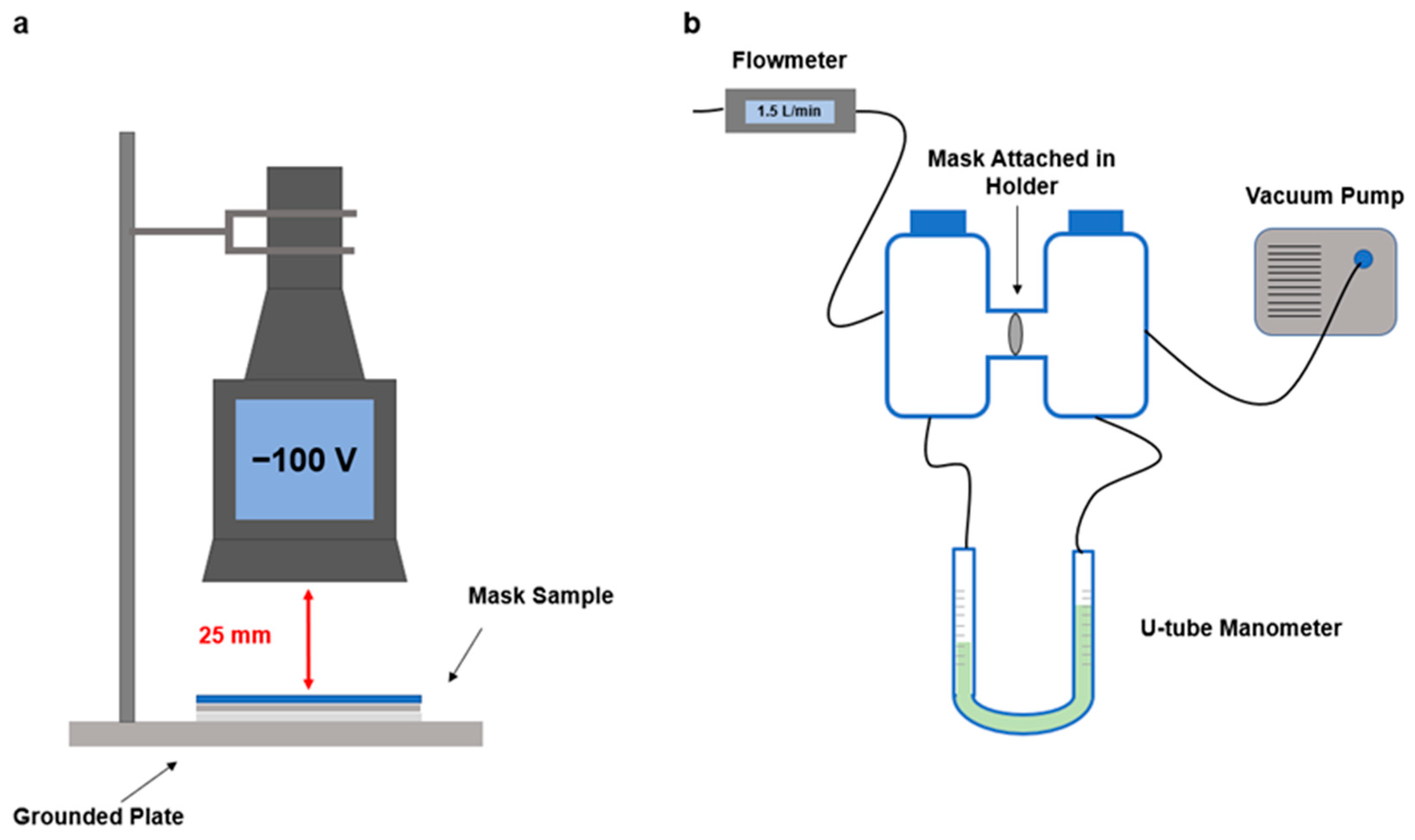
2.3. Mask Surface Potential
2.4. Particle Surface Charge
2.5. Pressure Drop Measurement across Filters
3. Results and Discussion
3.1. Mask Characterization Results: Surface Potential, Filtration Efficiency and Pressure Drop
3.2. Particle Surface Charge Characterization Results
3.3. Impact of Mask Surface Potential (Ψf) on Filtration Efficiency of PSL Particles
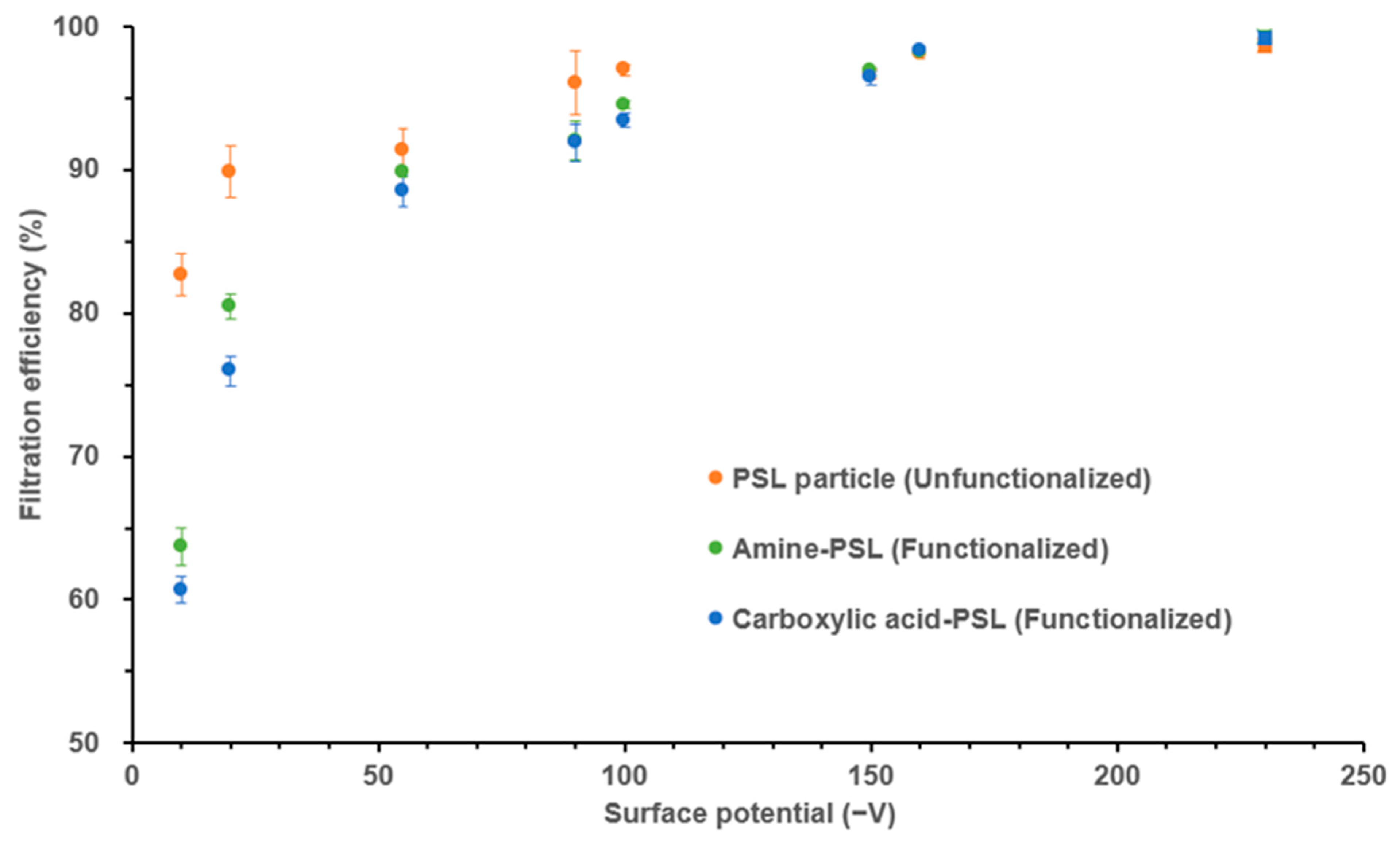
3.4. Impact of Aerosol Particle Surface Potential (Ψp) on Filter Filtration Efficiency
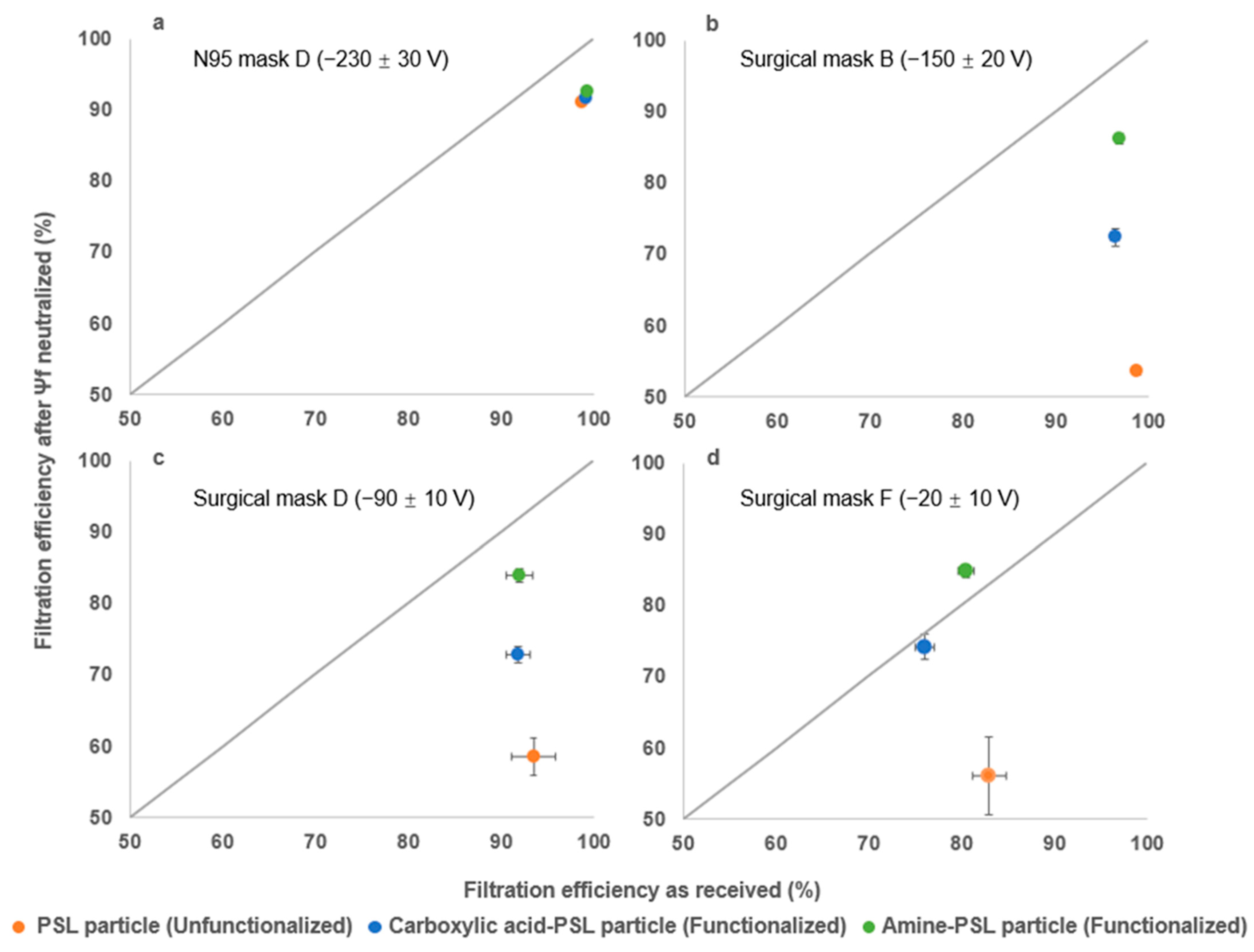
3.5. Impact of Removing Mask Surface Charge on Filtration Efficiency
3.6. Impact of Humidity on Filtration Efficiency
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tang, S.; Mao, Y.; Jones, R.M.; Tan, Q.; Ji, J.S.; Li, N.; Shen, J.; Lv, Y.; Pan, L.; Ding, P.; et al. Aerosol Transmission of SARS-CoV-2? Evidence, Prevention and Control. Environ. Int. 2020, 144, 106039. [Google Scholar] [CrossRef]
- Gu, Z.; Han, J.; Zhang, L.; Wang, H.; Luo, X.; Meng, X.; Zhang, Y.; Niu, X.; Lan, Y.; Wu, S.; et al. Unanswered Questions on the Airborne Transmission of COVID-19. Environ. Chem. Lett. 2023, 21, 725–739. [Google Scholar] [CrossRef]
- Peng, Z.; Rojas, A.L.P.; Kropff, E.; Bahnfleth, W.; Buonanno, G.; Dancer, S.J.; Kurnitski, J.; Li, Y.; Loomans, M.G.L.C.; Marr, L.C.; et al. Practical Indicators for Risk of Airborne Transmission in Shared Indoor Environments and Their Application to COVID-19 Outbreaks. Environ. Sci. Technol. 2022, 56, 1125–1137. [Google Scholar] [CrossRef]
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B.; et al. Tracking Changes in SARS-CoV-2 Spike: Evidence That D614G Increases Infectivity of the COVID-19 Virus. Cell 2020, 182, 812–827.e19. [Google Scholar] [CrossRef]
- Morawska, L.; Milton, D.K. It Is Time to Address Airborne Transmission of Coronavirus Disease 2019 (COVID-19). Clin. Infect. Dis. 2020, 71, 2311–2313. [Google Scholar] [CrossRef]
- Morawska, L.; Tang, J.W.; Bahnfleth, W.; Bluyssen, P.M.; Boerstra, A.; Buonanno, G.; Cao, J.; Dancer, S.; Floto, A.; Franchimon, F.; et al. How Can Airborne Transmission of COVID-19 Indoors Be Minimised? Environ. Int. 2020, 142, 105832. [Google Scholar] [CrossRef]
- Aydin, O.; Emon, B.; Cheng, S.; Hong, L.; Chamorro, L.P.; Saif, M.T.A. Performance of Fabrics for Home-Made Masks against the Spread of COVID-19 through Droplets: A Quantitative Mechanistic Study. Extrem. Mech. Lett. 2020, 40, 100924. [Google Scholar] [CrossRef]
- Zangmeister, C.D.; Radney, J.G.; Vicenzi, E.P.; Weaver, J.L. Filtration Efficiencies of Nanoscale Aerosol by Cloth Mask Materials Used to Slow the Spread of SARS-CoV-2. ACS Nano 2020, 14, 9188–9200. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Ullah, A.; Lee, J.; Jeong, Y.; Hashmi, M.; Zhu, C.; Joo, K.I.; Cha, H.J.; Kim, I.S. Reusability Comparison of Melt-Blown vs. Nanofiber Face Mask Filters for Use in the Coronavirus Pandemic. ACS Appl. Nano Mater. 2020, 3, 7231–7241. [Google Scholar] [CrossRef] [PubMed]
- Konda, A.; Prakash, A.; Moss, G.A.; Schmoldt, M.; Grant, G.D.; Guha, S. Aerosol Filtration Efficiency of Common Fabrics Used in Respiratory Cloth Masks. ACS Nano 2020, 14, 6339–6347. [Google Scholar] [CrossRef] [PubMed]
- Bisiaux, M.M.; Edwards, R.; Heyvaert, A.C.; Thomas, J.M.; Fitzgerald, B.; Susfalk, R.B.; Schladow, S.G.; Thaw, M. Stormwater and Fire as Sources of Black Carbon Nanoparticles to Lake Tahoe. Environ. Sci. Technol. 2011, 45, 2065–2071. [Google Scholar] [CrossRef]
- Zhang, R.; Khalizov, A.; Wang, L.; Hu, M.; Xu, W. Nucleation and Growth of Nanoparticles in the Atmosphere. Chem. Rev. 2012, 112, 1957–2011. [Google Scholar] [CrossRef]
- Caudillo, L.; Rörup, B.; Heinritzi, M.; Marie, G.; Simon, M.; Wagner, A.C.; Müller, T.; Granzin, M.; Amorim, A.; Ataei, F.; et al. Chemical Composition of Nanoparticles from α-Pinene Nucleation and the Influence of Isoprene and Relative Humidity at Low Temperature. Atmos. Chem. Phys. 2021, 21, 17099–17114. [Google Scholar] [CrossRef]
- Mishra, S.; Tripathi, S.N.; Kanawade, V.P.; Haslett, S.L.; Dada, L.; Ciarelli, G.; Kumar, V.; Singh, A.; Bhattu, D.; Rastogi, N.; et al. Rapid Night-Time Nanoparticle Growth in Delhi Driven by Biomass-Burning Emissions. Nat. Geosci. 2023, 16, 224–230. [Google Scholar] [CrossRef]
- Bessa, M.J.; Brandão, F.; Viana, M.; Gomes, J.F.; Monfort, E.; Cassee, F.R.; Fraga, S.; Teixeira, J.P. Nanoparticle Exposure and Hazard in the Ceramic Industry: An Overview of Potential Sources, Toxicity and Health Effects. Environ. Res. 2020, 184, 109297. [Google Scholar] [CrossRef]
- Kittelson, D.B.; Watts, W.F.; Johnson, J.P. Nanoparticle Emissions on Minnesota Highways. Atmos. Environ. 2004, 38, 9–19. [Google Scholar] [CrossRef]
- Ermolin, M.S.; Fedotov, P.S.; Malik, N.A.; Karandashev, V.K. Nanoparticles of Volcanic Ash as a Carrier for Toxic Elements on the Global Scale. Chemosphere 2018, 200, 16–22. [Google Scholar] [CrossRef]
- O’Dowd, C.D.; Smith, M.H.; Consterdine, I.E.; Lowe, J.A. Marine Aerosol, Sea-Salt, and the Marine Sulphur Cycle: A Short Review. Atmos. Environ. 1997, 31, 73–80. [Google Scholar] [CrossRef]
- Karl, M.; Leck, C.; Coz, E.; Heintzenberg, J. Marine Nanogels as a Source of Atmospheric Nanoparticles in the High Arctic. Geophys. Res. Lett. 2013, 40, 3738–3743. [Google Scholar] [CrossRef]
- Lin, K.; Marr, L.C. Humidity-Dependent Decay of Viruses, but Not Bacteria, in Aerosols and Droplets Follows Disinfection Kinetics. Environ. Sci. Technol. 2020, 54, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.; Daher, N.; Kam, W.; Shafer, M.M.; Ning, Z.; Schauer, J.J.; Sioutas, C. Spatial and Temporal Variation of Chemical Composition and Mass Closure of Ambient Coarse Particulate Matter (PM10-2.5) in the Los Angeles Area. Atmos. Environ. 2011, 45, 2651–2662. [Google Scholar] [CrossRef]
- Wang, C.C.; Prather, K.A.; Sznitman, J.; Jimenez, J.L.; Lakdawala, S.S.; Tufekci, Z.; Marr, L.C. Airborne Transmission of Respiratory Viruses. Science 2021, 373, eabd9149. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Liu, C.; Hsu, P.C.; Zhang, C.; Liu, N.; Zhang, J.; Lee, H.R.; Lu, Y.; Qiu, Y.; Chu, S.; et al. Nanofiber Air Filters with High-Temperature Stability for Efficient PM2.5 Removal from the Pollution Sources. Nano Lett. 2016, 16, 3642–3649. [Google Scholar] [CrossRef] [PubMed]
- Kadiiska, M.B.; Mason, R.P.; Dreher, K.L.; Costa, D.L.; Ghio, A.J. In Vivo Evidence of Free Radical Formation in the Rat Lung after Exposure to an Emission Source Air Pollution Particle. Chem. Res. Toxicol. 1997, 10, 1104–1108. [Google Scholar] [CrossRef] [PubMed]
- Maher, B.A.; Ahmed, I.A.M.; Karloukovski, V.; MacLaren, D.A.; Foulds, P.G.; Allsop, D.; Mann, D.M.A.; Torres-Jardón, R.; Calderon-Garciduenas, L. Magnetite Pollution Nanoparticles in the Human Brain. Proc. Natl. Acad. Sci. USA 2016, 113, 10797–10801. [Google Scholar] [CrossRef]
- NIOSH TEB-APR-STP-0059; Determination of Particulate Filter Efficiency Level for N95 Series Filter against Liquid Particulates for Non-Powered, Air-Purifying Respirators Standard Testing Procedure (STP). National Institute for Occupational Safety and Health National Personal Protective Technology Laboratory: Washington, DC, USA, 2019; pp. 1–9.
- Yue, Y.; Wang, J.; He, W.; Guo, Y.; Gao, H.; Liu, J. Evaluation of Regeneration Processes for Filtering Facepiece Respirators in Terms of the Bacteria Inactivation Efficiency and Influences on Filtration Performance. ACS Nano 2020, 14, 13161–13171. [Google Scholar] [CrossRef]
- Kilic, A.; Shim, E.; Pourdeyhimi, B. Electrostatic Capture Efficiency Enhancement of Polypropylene Electret Filters with Barium Titanate. Aerosol Sci. Technol. 2015, 49, 666–673. [Google Scholar] [CrossRef]
- Liao, L.; Xiao, W.; Zhao, M.; Yu, X.; Wang, H.; Wang, Q.; Chu, S.; Cui, Y. Can N95 Respirators Be Reused after Disinfection? How Many Times? ACS Nano 2020, 14, 6348–6356. [Google Scholar] [CrossRef]
- Sanchez, A.L.; Hubbard, J.A.; Dellinger, J.G.; Servantes, B.L. Experimental Study of Electrostatic Aerosol Filtration at Moderate Filter Face Velocity. Aerosol Sci. Technol. 2013, 47, 606–615. [Google Scholar] [CrossRef]
- Morais, F.G.; Sakano, V.K.; de Lima, L.N.; Franco, M.A.; Reis, D.C.; Zanchetta, L.M.; Jorge, F.; Landulfo, E.; Catalani, L.H.; Barbosa, H.M.J.; et al. Filtration Efficiency of a Large Set of COVID-19 Face Masks Commonly Used in Brazil. Aerosol Sci. Technol. 2021, 55, 1028–1041. [Google Scholar] [CrossRef]
- Rengasamy, S.; Miller, A.; Eimer, B.C.; Shaffer, R.E. Filtration Performance of FDA-Cleared Surgical Masks. J. Int. Soc. Respir. Prot. 2020, 26, 54–70. [Google Scholar]
- Tcharkhtchi, A.; Abbasnezhad, N.; Zarbini Seydani, M.; Zirak, N.; Farzaneh, S.; Shirinbayan, M. An Overview of Filtration Efficiency through the Masks: Mechanisms of the Aerosols Penetration. Bioact. Mater. 2021, 6, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Hinds, W.C.; Zhu, Y. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles; John Wiley & Sons: Hoboken, NJ, USA, 2022; ISBN 1119494044. [Google Scholar]
- Lorenz, M.; Malangré, D.; Du, F.; Baune, M.; Thöming, J.; Pesch, G.R. High-Throughput Dielectrophoretic Filtration of Sub-Micron and Micro Particles in Macroscopic Porous Materials. Anal. Bioanal. Chem. 2020, 412, 3903–3914. [Google Scholar] [CrossRef] [PubMed]
- Kilic, A.; Russell, S.; Shim, E.; Pourdeyhimi, B. The Charging and Stability of Electret Filters; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; ISBN 9780081005828. [Google Scholar]
- Rashid, T.U.; Sharmeen, S.; Biswas, S. Effectiveness of N95 Masks against SARS-CoV-2: Performance Efficiency, Concerns, and Future Directions. ACS Chem. Heal. Saf. 2022, 29, 135–164. [Google Scholar] [CrossRef] [PubMed]
- Ou, Q.; Pei, C.; Chan Kim, S.; Abell, E.; Pui, D.Y.H. Evaluation of Decontamination Methods for Commercial and Alternative Respirator and Mask Materials—View from Filtration Aspect. J. Aerosol Sci. 2020, 150, 105609. [Google Scholar] [CrossRef] [PubMed]
- Kerner, M.; Schmidt, K.; Schumacher, S.; Puderbach, V.; Asbach, C.; Antonyuk, S. Evaluation of Electrostatic Properties of Electret Filters for Aerosol Deposition. Sep. Purif. Technol. 2020, 239, 116548. [Google Scholar] [CrossRef]
- Nifuku, M.; Zhou, Y.; Kisiel, A.; Kobayashi, T.; Katoh, H. Charging Characteristics for Electret Filter Materials. J. Electrostat. 2001, 51–52, 200–205. [Google Scholar] [CrossRef]
- Xiao, H.; Song, Y.; Chen, G. Correlation between Charge Decay and Solvent Effect for Melt-Blown Polypropylene Electret Filter Fabrics. J. Electrostat. 2014, 72, 311–314. [Google Scholar] [CrossRef]
- Romay, F.J.; Liu, B.Y.H.; Chae, S.J. Experimental Study of Electrostatic Capture Mechanisms in Commercial Electret Filters. Aerosol Sci. Technol. 1998, 28, 224–234. [Google Scholar] [CrossRef]
- Yim, W.; Cheng, D.; Patel, S.H.; Kou, R.; Meng, Y.S.; Jokerst, J.V. KN95 and N95 Respirators Retain Filtration Efficiency despite a Loss of Dipole Charge during Decontamination. ACS Appl. Mater. Interfaces 2020, 12, 54473–54480. [Google Scholar] [CrossRef]
- Mahdavi, A.; Haghighat, F.; Bahloul, A.; Brochot, C.; Ostiguy, C. Particle Loading Time and Humidity Effects on the Efficiency of an N95 Filtering Facepiece Respirator Model under Constant and Inhalation Cyclic Flows. Ann. Occup. Hyg. 2015, 59, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Drewnick, F.; Pikmann, J.; Fachinger, F.; Moormann, L.; Sprang, F.; Borrmann, S. Aerosol Filtration Efficiency of Household Materials for Homemade Face Masks: Influence of Material Properties, Particle Size, Particle Electrical Charge, Face Velocity, and Leaks. Aerosol Sci. Technol. 2021, 55, 63–79. [Google Scholar] [CrossRef]
- Zazzera, L.; Mader, B.; Ellefson, M.; Eldridge, J.; Loper, S.; Zabasajja, J.; Qian, J. Comparison of Ceria Nanoparticle Concentrations in Effluent from Chemical Mechanical Polishing of Silicon Dioxide. Environ. Sci. Technol. 2014, 48, 13427–13433. [Google Scholar] [CrossRef]
- Tabti, B.; Mekideche, M.R.; Plopeanu, M.C.; Dumitran, L.M.; Herous, L.; Dascalescu, L. Corona-Charging and Charge-Decay Characteristics of Nonwoven Filter Media. IEEE Trans. Ind. Appl. 2010, 46, 634–640. [Google Scholar] [CrossRef]
- Noh, K.C.; Park, J.H.; Jung, Y.K.; Yi, S.; Hwang, J. Characteristics of Submicron-Sized Aerosol Filtration and Pressure Drop of an Electret Filter Installed in an Air Diffuser in a Residential Apartment Unit. Aerosol Air Qual. Res. 2011, 11, 80–89. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Gunji, Y.; Okahisa, S.; Kusdianto, K.; Gen, M.; Sato, S.; Lenggoro, I.W. Preliminary Study on the Measurement of the Electrostatic Charging State of PM2.5 Collected on Filter Media. Asian J. Atmos. Environ. 2015, 9, 137–145. [Google Scholar] [CrossRef]
- Speed, D.; Westerhoff, P.; Sierra-Alvarez, R.; Draper, R.; Pantano, P.; Aravamudhan, S.; Chen, K.L.; Hristovski, K.; Herckes, P.; Bi, X.; et al. Physical, Chemical, and in Vitro Toxicological Characterization of Nanoparticles in Chemical Mechanical Planarization Suspensions Used in the Semiconductor Industry: Towards Environmental Health and Safety Assessments. Environ. Sci. Nano 2015, 2, 227–244. [Google Scholar] [CrossRef]
- NanoBrook Inc. Instruction Manual for the NanoBrook Series Particle/Protein Size and Zeta Potential Analyzer, Ver. 1.3DC; NanoBrook Inc.: Long Island, NY, USA, 2023. [Google Scholar]
- Laengert, S.E.; Kwon, Y.J.; Corbin, J.C.; Sipkens, T.A.; Morkus, P.; LaRue, R.J.; Latulippe, D.R.; Clase, C.M.; de Lannoy, C.-F. Aerosol Charge Neutralization and Its Impact on Particle Filtration Efficiency of Common Face Mask Materials. J. Aerosol Sci. 2023, 173, 106189. [Google Scholar] [CrossRef]
- Varanges, V.; Caglar, B.; Lebaupin, Y.; Batt, T.; He, W.; Wang, J.; Rossi, R.M.; Richner, G.; Delaloye, J.R.; Michaud, V. On the Durability of Surgical Masks after Simulated Handling and Wear. Sci. Rep. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Fuchs, N.A. On the Stationary Charge Distribution on Aerosol Particles in a Bipolar Ionic Atmosphere. Geofis. Pura E Appl. 1963, 56, 185–193. [Google Scholar] [CrossRef]
- Wiedensohler, A. An Approximation of the Bipolar Charge Distribution for Particles in the Submicron Size Range. J. Aerosol Sci. 1988, 19, 387–389. [Google Scholar] [CrossRef]
- Jiang, J.; Kim, C.; Wang, X.; Stolzenburg, M.R.; Kaufman, S.L.; Qi, C.; Sem, G.J.; Sakurai, H.; Hama, N.; Mcmurry, P.H. Aerosol Charge Fractions Downstream of Six Bipolar Chargers: Effects of Ion Source, Source Activity, and Flowrate. Aerosol Sci. Technol. 2014, 48, 1207–1216. [Google Scholar] [CrossRef]
- Kim, J.; Hinestroza, J.P.; Jasper, W.; Barker, R.L. Effect of Solvent Exposure on the Filtration Performance of Electrostatically Charged Polypropylene Filter Media. Text. Res. J. 2009, 79, 343–350. [Google Scholar] [CrossRef]
- Vaaraslahti, K.; Laitinen, A.; Keskinen, J. Spray Charging of Droplets in a Wet Scrubber. J. Air Waste Manag. Assoc. 2002, 52, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Mainelis, G.; Willeke, K.; Baron, P.; Reponen, T.; Grinshpun, S.A.; Górny, R.L.; Trakumas, S. Electrical Charges on Airborne Microorganisms. J. Aerosol Sci. 2001, 32, 1087–1110. [Google Scholar] [CrossRef]
- Herous, L.; Nemamcha, M.; Remadnia, M.; Dascalescu, L. Factors That Influence the Surface Potential Decay on a Thin Film of Polyethylene Terephthalate (PET). J. Electrostat. 2009, 67, 198–202. [Google Scholar] [CrossRef]
- de Lima Burgo, T.A.; Rezende, C.A.; Bertazzo, S.; Galembeck, A.; Galembeck, F. Electric Potential Decay on Polyethylene: Role of Atmospheric Water on Electric Charge Build-up and Dissipation. J. Electrostat. 2011, 69, 401–409. [Google Scholar] [CrossRef]
- Guardiola, J.; Rojo, V.; Ramos, G. Influence of Particle Size, Fluidization Velocity and Relative Humidity on Fluidized Bed Electrostatics. J. Electrostat. 1996, 37, 1–20. [Google Scholar] [CrossRef]
- Zhao, M.; Liao, L.; Xiao, W.; Yu, X.; Wang, H.; Wang, Q.; Lin, Y.L.; Kilinc-Balci, F.S.; Price, A.; Chu, L.; et al. Household Materials Selection for Homemade Cloth Face Coverings and Their Filtration Efficiency Enhancement with Triboelectric Charging. Nano Lett. 2020, 20, 5544–5552. [Google Scholar] [CrossRef]
- Zangmeister, C.D.; Radney, J.G.; Staymates, M.E.; Vicenzi, E.P.; Weaver, J.L. Hydration of Hydrophilic Cloth Face Masks Enhances the Filtration of Nanoparticles. ACS Appl. Nano Mater. 2021, 4, 2694–2701. [Google Scholar] [CrossRef]


| Mask Type | Sample Number | Surface Potential (Ψf, V) | Filtration Efficiency (%) | Pressure Drop (mmH2O/cm2) |
|---|---|---|---|---|
| N95 mask | N95 mask A | −800 ± 108 | 99.2 ± 0.3 | 9 |
| N95 mask B | −539 ± 85 | 99.5 ± 0.2 | 7 | |
| N95 mask C | −440 ± 53 | 99.1 ± 0.2 | 7 | |
| N95 mask D | −230 ± 30 | 99.4 ± 0.2 | 6.5 | |
| Surgical mask | Surgical mask A | −160 ± 18 | 98.1 ± 0.3 | 8 |
| Surgical mask B | −150 ± 20 | 96.7 ± 0.4 | 5 | |
| Surgical mask C | −100 ± 12 | 97.7 ± 0.4 | 4.5 | |
| Surgical mask D | −90 ± 10 | 96.1 ± 2.3 | 7.5 | |
| Surgical mask E | −55 ± 5 | 91.4 ± 1.4 | 3 | |
| Surgical mask F | −20 ± 10 | 90.9 ± 1.8 | 4.5 | |
| Surgical mask G | −10 ± 5 | 82.7 ± 1.4 | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Ersan, M.S.; Westerhoff, P.; Herckes, P. Do Surface Charges on Polymeric Filters and Airborne Particles Control the Removal of Nanoscale Aerosols by Polymeric Facial Masks? Toxics 2024, 12, 3. https://doi.org/10.3390/toxics12010003
Zhang Z, Ersan MS, Westerhoff P, Herckes P. Do Surface Charges on Polymeric Filters and Airborne Particles Control the Removal of Nanoscale Aerosols by Polymeric Facial Masks? Toxics. 2024; 12(1):3. https://doi.org/10.3390/toxics12010003
Chicago/Turabian StyleZhang, Zhaobo, Mahmut S. Ersan, Paul Westerhoff, and Pierre Herckes. 2024. "Do Surface Charges on Polymeric Filters and Airborne Particles Control the Removal of Nanoscale Aerosols by Polymeric Facial Masks?" Toxics 12, no. 1: 3. https://doi.org/10.3390/toxics12010003
APA StyleZhang, Z., Ersan, M. S., Westerhoff, P., & Herckes, P. (2024). Do Surface Charges on Polymeric Filters and Airborne Particles Control the Removal of Nanoscale Aerosols by Polymeric Facial Masks? Toxics, 12(1), 3. https://doi.org/10.3390/toxics12010003










