Silent Contamination: The State of the Art, Knowledge Gaps, and a Preliminary Risk Assessment of Tire Particles in Urban Parks
Abstract
1. Introduction
2. Materials and Methods
3. Definition and Chemical Composition of Tire and Road Wear Particles
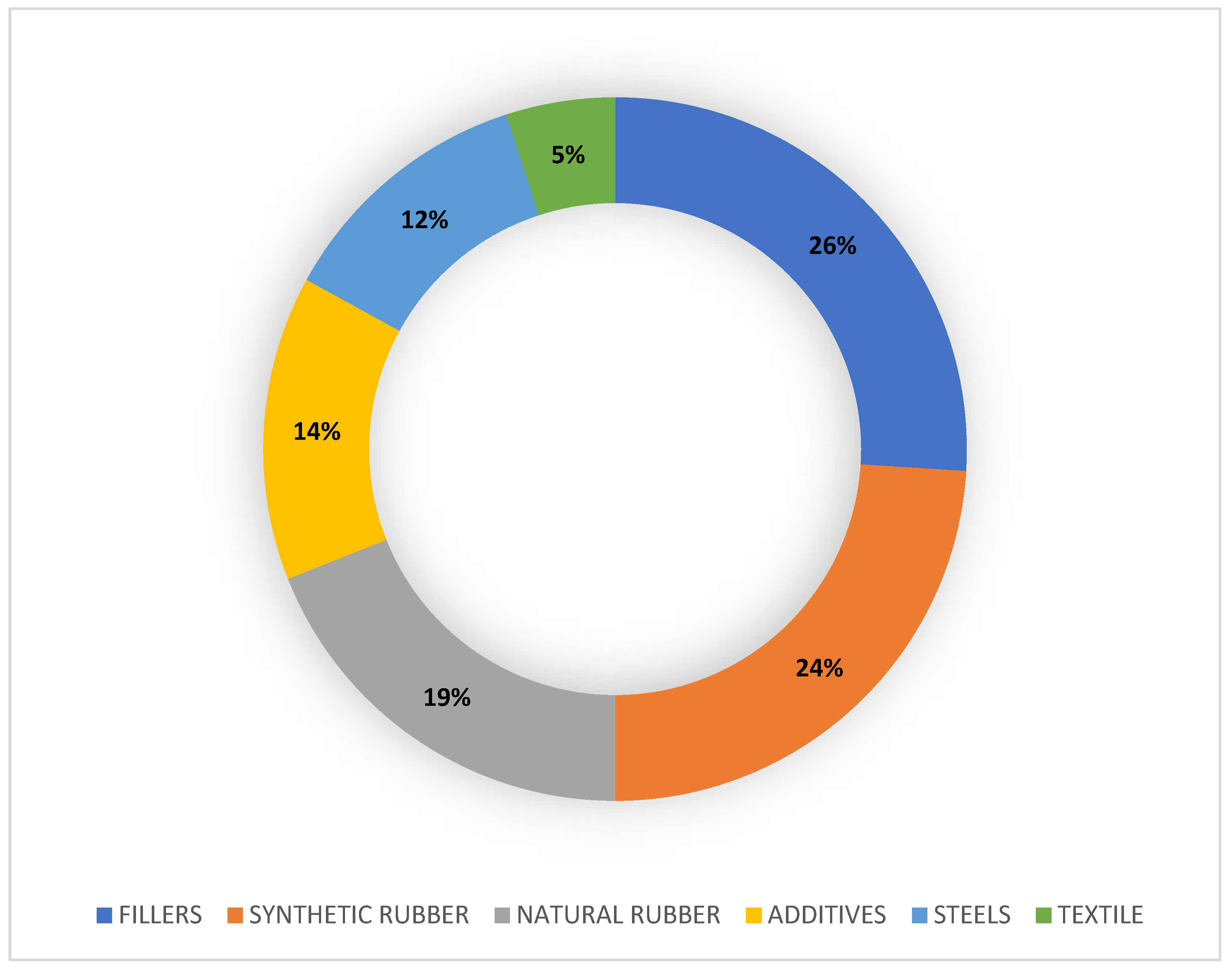
- Rubbers: Components typically consist of blends of styrene-butadiene rubber (SBR), such as polybutadiene (PBD), and isoprene rubber (IR), the forerunner of natural rubber (NR), mixed with carbon black or silica (as a reinforcing agent/filler), oils (as softeners and extenders), and curing chemicals. In the past, tires were made only of natural rubber, such as that extracted from the Brazilian rubber tree Hevea brasiliensis [39]; nowadays, also for ecological reasons, a mixture of natural and synthetic rubbers is used. Such SBRs describe families of synthetic rubbers derived from emulsion-polymerized (E-SBR, more widely used) or solution-polymerized (S-SBR) styrene and butadiene. The styrene/butadiene ratio affects the properties of the rubber: if the styrene content is high, the rubbers are harder and less rubbery. Normally, 23.5% of the rubber consists of styrene and the remaining 76.5% consists of butadiene [40].
- Organic chemicals: Benzoic acid (BZA) and N-nitrosodiphenylamine (NDphA) are burning retarders and slow down the vulcanization process [34]. Diamines and waxes are also used as anti-degradants by oxidizing agents (oxygen or ozone) and by heat [41,42]. Studies on TRWPs leachate confirmed that they are a potential source of benzothiazoles (BTs), as accelerators of vulcanization, and 1-octanethiol (1-OT) [43], phthalates (PTEs), additives, such as bisphenol A (BSA), and polycyclic aromatic hydrocarbons (PAHs), such as benzo-γ-perylene, fluorene, benzo-α-pyrene, benzo-β-fluoranthene, phenanthrene, benzo-κ-fluoranthene, pyrene, anthracene, and fluoranthene [35,44,45,46,47]. Other substances released in leachate are N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine (6-PPD) and its ozonation product 6-PPD-quinone [48], used as stabilizing additives, hexa(methoxymethyl)melamine (HMMM) [49,50,51], and N,N′-diphenylguanidine (DPG), an accelerator of vulcanization [14,52].
- Heavy metals (HM): Trace elements such as Zn, Al, Fe, Cd, Cr, Ni, Hg, and Cu are present on TRWPs [43]; consequently, tire wear contributes to the release of HMs into the environment. About 1% of zinc oxide (ZnO) is used as a catalyst to vulcanize the rubber mixtures, transforming them into highly elastic matter; consequently, TRWPs are considered to be among the main sources of Zn in the environment [53]. Other accelerators are sulfur, sulphenamides, and thiazoles [34].
- Fillers: Most of the components of a tire tread consist of fillers, mainly black carbon (22–40%) finely pulverized by incomplete combustion and added for making the tire resistant to UV rays. In recent years, carbon is sometimes replaced by nanometer glass spheres of silica, which gives the tread strong adhesion and resistance to tearing, heat, and ageing [13].
4. Environmental Fate of TRWPs
4.1. Transport and Deposition Pathways
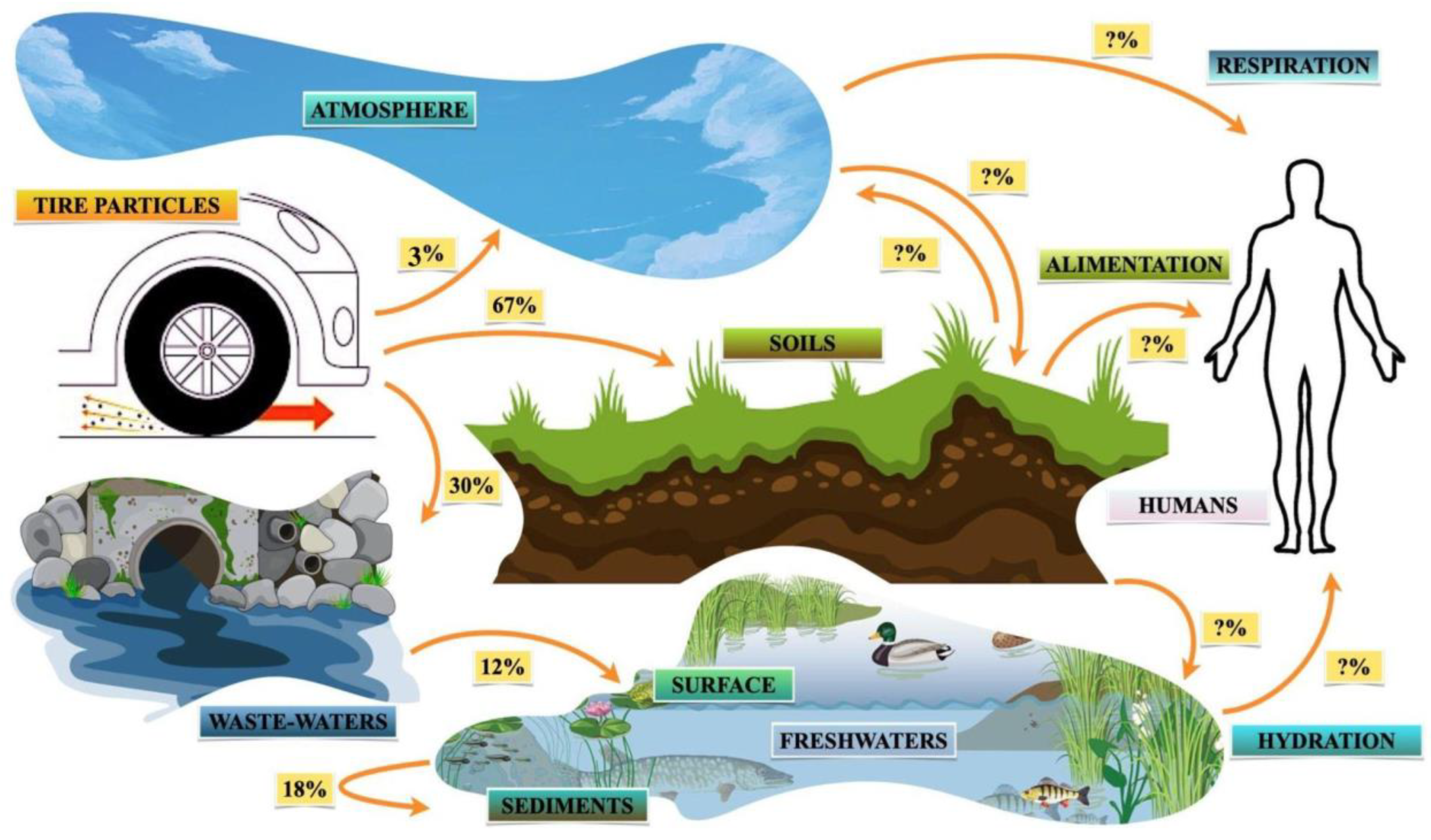
4.1.1. Soil
4.1.2. Air
4.1.3. Sewer Systems, Freshwater, and Sediments
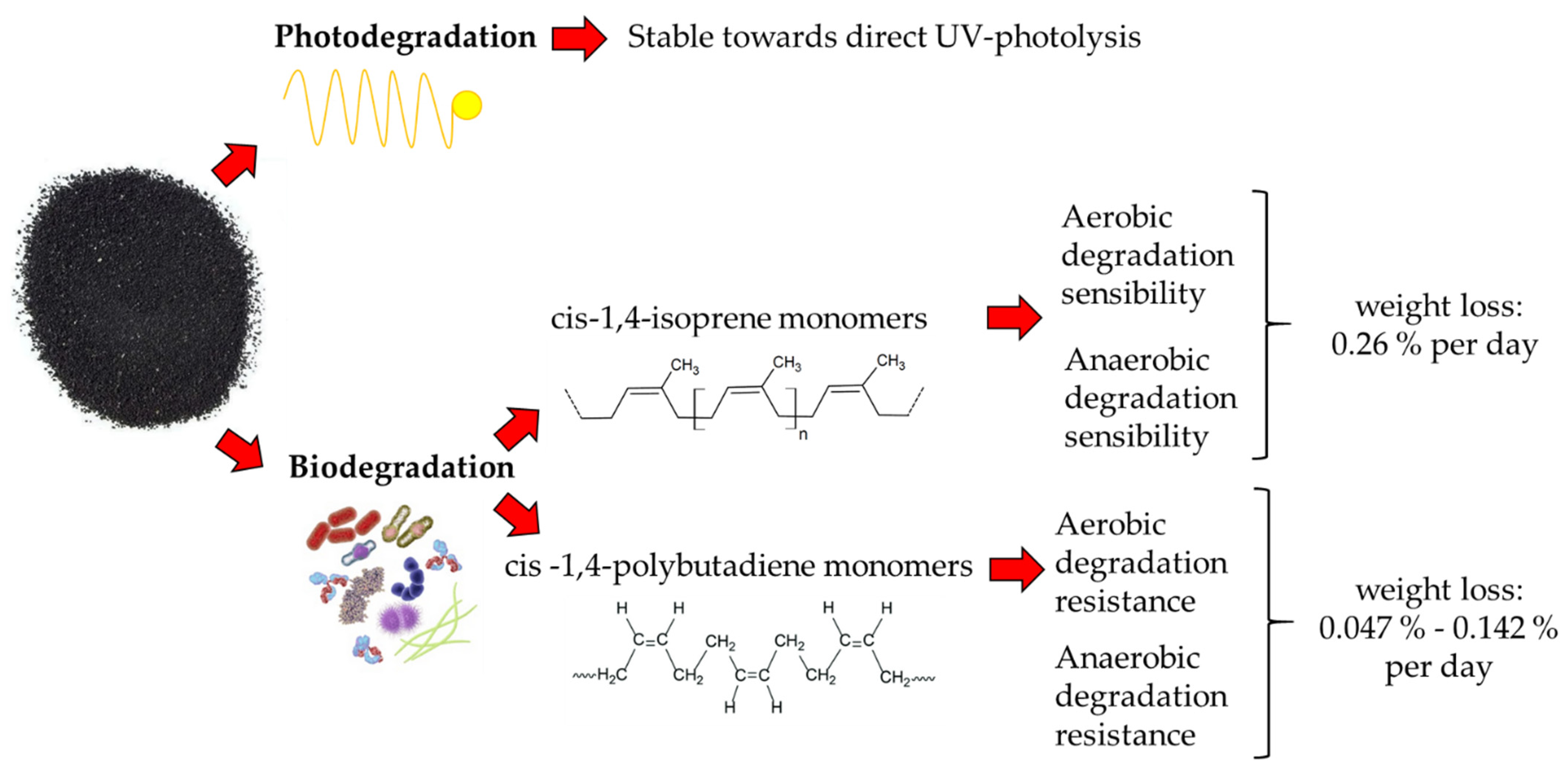
4.2. Degradation Processes
4.3. Environmental Markers of TRWPs
5. Toxicity Effects on Edaphic Fauna and Plants
5.1. Earthworms
5.2. Nematodes
5.3. Springtails
5.4. Woodlice
5.5. Plants
6. Preliminary Risk Assessment and Future Projections for Urban Soils: The ‘Milan Case’
- PECx = predicted environmental concentration of the x contaminant
- EFx = mg of the emission factor of the x contaminant emitted by registered cars
- M = kilograms of urban soil considered.
6.1. TRWP Emission Factors
- EFTRWPs = emission factors of TRWPs (mg x vehicles/y);
- EFv = vehicle-specific emission factors of TRWPs (mg/km);
- Nv = number of vehicles on the road
- d = distance in km/y
6.2. Mass of Considered Soil
- VSOIL = Total volume of Forlanini Park soil potentially impacted by TRWPs
- lFOR = Forlanini Avenue’s length adjacent to the park
- dFOR = Depth of soil potentially contaminated by TRWPs [66]
- Msoil = kg d.w. of the Forlanini soil mass considered
- ρ = kg/m3 of the soil’s density
- VSOIL = m3 of the total volume of Forlanini Park’s soil from Equation (5)
6.3. PEC of TRWPs
7. Discussion
8. Sustainability and Risk Mitigation Measures
9. Conclusions
- (a)
- Greater and deeper attention to soil contamination by TRWPs;
- (b)
- Standardization in the detection of these contaminants, with greater awareness of the choice of environmental markers;
- (c)
- Standardization in toxicity tests, using efficient model organisms sensitive to TRWPs;
- (d)
- Analyze the effects determined not only by the leachate but also by the particles to understand the environmental toxicity of TRWPs;
- (e)
- Carry out interventions at the urban level to reduce both the contact between the tire tread and the asphalt and between the road and the surrounding soil;
- (f)
- Promoting new sustainable tires as an efficient strategy to reduce TRWPs.
Author Contributions
Funding
Conflicts of Interest
References
- Karagulian, F.; Belis, C.A.; Dora, C.F.C.; Prüss-Ustün, A.M.; Bonjour, S.; Adair-Rohani, H.; Amann, M. Contributions to Cities’ Ambient Particulate Matter (PM): A Systematic Review of Local Source Contributions at Global Level. Atmos. Environ. 2015, 120, 475–483. [Google Scholar] [CrossRef]
- Samek, L.; Stegowski, Z.; Styszko, K.; Furman, L.; Zimnoch, M.; Skiba, A.; Kistler, M.; Kasper-Giebl, A.; Rozanski, K.; Konduracka, E. Seasonal Variations of Chemical Composition of PM2.5 Fraction in the Urban Area of Krakow, Poland: PMF Source Attribution. Air Qual. Atmos. Health 2020, 13, 89–96. [Google Scholar] [CrossRef]
- ISO International Organization for Standardization. Plastics—Vocabulary (ISO 472:2013). 2013. Available online: https://www.iso.org/obp/ui/#iso:std:iso:472:ed-4:v1:en (accessed on 28 January 2023).
- Hartmann, N.B.; Huffer, T.; Thompson, R.C.; Hassellov, M.; Verschoor, A.; Daugaard, S.; Rist, T.; Karlsson, N.; Brennholt, M.; Cole, M.P.; et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar]
- Arias, A.H.; Alfonso, M.B.; Girones, L.; Piccolo, M.C.; Marcovecchio, J.E. Synthetic Microfibers and Tyre Wear Particles Pollution in Aquatic Systems: Relevance and Mitigation Strategies. Environ. Pollut. 2021, 295, 118607. [Google Scholar] [PubMed]
- Sundt, P.; Schulze, P.-E.; Syversen, F. Sources of Microplastics-Pollution to the Marine Environment. Mepex Nor. Environ. Agency 2014, 86, 20. [Google Scholar]
- Lassen, C.; Foss Hansen, S.; Magnusson, K.; Norén, F.; Bloch Hartmann, N.I.; Rehne Jensen, P.; Nielsen, T.G.; Brinch, A. Microplastics—Occurrence, Effects and Sources of Releases to the Environment in Denmark Danish; Environmental Protection Agency: Copenhagen, Denmark, 2015; Volume 29, p. 1401. [Google Scholar]
- Magnusson, K.; Eliasson, K.; Fråne, A.; Haikonen, K.; Hultén, J.; Olshammar, M.; Stadmark, J.; Voisin, A. Swedish Sources and Pathways for Microplastics to the Marine Environment. A Review of Existing Data; Report number C 183; Swedish Environmental Protection Agency: Stockholm, Sweden, 2016. [Google Scholar]
- Sherrington, C.; Darrah, C.; Hann, S.; Cole, G.; Corbin, M. Study to Support the Development of Measures to Combat a Range of Marine Litter Sources; Report for European Commission DG Environment; The Danish Environmental Protection Agency: Copenhagen, Denmark, 2016. [Google Scholar]
- Andersson-Sköld, Y.; Johannesson, M.; Gustafsson, M.; Järlskog, I.; Lithner, D.; Polukarova, M.; Strömvall, A.-M. Microplastics from Tyre and Road Wear; Digitala Vetenskapliga Arkivet: Stockholm, Sweden, 2020. [Google Scholar]
- Zhou, Y.; Liu, X.; Wang, J. Characterization of Microplastics and the Association of Heavy Metals with Microplastics in Suburban Soil of Central China. Sci. Total Environ. 2019, 694, 3798. [Google Scholar] [CrossRef]
- Boucher, J.; Friot, D. Primary Microplastics in the Oceans: A Global Evaluation of Sources, Primary Microplastics in the Oceans: A Global Evaluation of Sources; IUCN International Union for Conservation of Nature and Natural Resources (IUCN): Gland, Switzerland, 2017; p. 43. [Google Scholar]
- Kole, J.P.; Löhr, A.J.; van Belleghem, F.G.A.J.; Ragas, A.M.J. Wear and tear of tyres: A stealthy source of microplastics in the environment. Int. J. Environ. Res. Public Health 2017, 14, 1265. [Google Scholar]
- Wagner, S.; Hüffer, T.; Klöckner, P.; Wehrhahn, M.; Hofmann, T.; Reemtsma, T. Tire wear particles in the aquatic environment—A review on generation, analysis, occurrence, fate and effects. Water Res. 2018, 139, 83–100. [Google Scholar] [CrossRef]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef]
- Wik, A.; Lycken, J.; Dave, G. Sediment quality assessment of road runoff detention systems in Sweden and the potential contribution of tire wear. Water Air Soil Pollut. 2008, 194, 301–314. [Google Scholar] [CrossRef]
- Wik, A. Toxic components leaching from tire rubber. Bull. Environ. Contam. Toxicol. 2007, 79, 114–119. [Google Scholar] [CrossRef]
- Wik, A.; Dave, G. Acute toxicity of tire rubber leachates to Daphnia magna—Variability and toxic components. Chemosphere 2006, 64, 1777–1784. [Google Scholar] [CrossRef] [PubMed]
- Benevento, S.; Draper, A. Analysis of tire rubber leachate with a bacterial mutagenesis assay. In Proceedings of the SETAC North America 26th Annual Meeting, Baltimore, MD, USA, 13–17 November 2005; pp. 13–17. [Google Scholar]
- Gualtieri, M.; Andrioletti, M.; Vismara, C.; Milani, M.; Camatini, M. Toxicity of tire debris leachates. Environ. Int. 2005, 31, 723–730. [Google Scholar] [CrossRef]
- Humphrey, D.N.; Katz, L.E. Water-quality effects of tire shreds placed above the water table—Five-year field study. Transp. Res. Rec. 2000, 1714, 18–24. [Google Scholar] [CrossRef]
- Sheehan, P.J.; Warmerdam, J.M.; Ogle, S.; Humphrey, D.N.; Patenaude, S.M. Evaluating the risk to aquatic ecosystems posed by leachate from tire shred fill in roads using toxicity tests, toxicity identification evaluations, and groundwater modeling. Environ. Toxicol. Chem. 2006, 25, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Kreider, M.L.; Panko, J.M.; McAtee, B.L.; Sweet, L.I.; Finley, B.L. Physical and chemical characterization of tire-related particles: Comparison of particles generated using different methodologies. Sci. Total Environ. 2010, 408, 652–659. [Google Scholar] [CrossRef]
- Sommer, F.; Dietze, V.; Baum, A.; Sauer, J.; Gilge, S.; Maschowski, C.; Giere, R. Tire abrasion as a major source of microplastics in the environment. Aerosol Air Qual. Res. 2018, 18, 2014–2028. [Google Scholar] [CrossRef]
- Workek, J.; Badura, X.; Białas, A.; Chwiej, J.; Kawoń, K.; Styszko, K. Pollution from Transport: Detection of Tyre Particles in Environmental Samples. Energies 2022, 15, 2816. [Google Scholar] [CrossRef]
- Alexandrova, O.; Kaloush, K.E.; Allen, J.O. Impact of Asphalt Rubber Friction Course Overlays on Tire Wear Emissions and Air Quality Models for Phoenix, Arizona, Airshed. Transp. Res. Rec. 2007, 2011, 98–106. [Google Scholar] [CrossRef]
- Grigoratos, T.; Martini, G. Brake Wear Particle Emissions: A Review. Environ. Sci. Pollut. Res. 2015, 22, 2491–2504. [Google Scholar] [CrossRef]
- Panko, J.M.; Chu, J.; Kreider, M.L.; Unice, K.M. Measurement of airborne concentrations of tire and road wear particles in urban and rural areas of France, Japan, and the United States. Atmos. Environ. 2013, 72, 192–199. [Google Scholar] [CrossRef]
- Adachi, K.; Tainosho, Y. Characterization of heavy metal particles embedded in tire dust. Environ. Int. 2004, 30, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Kayhanian, M.; McKenzie, E.R.; Leatherbarrow, J.E.; Young, T.M. Characteristics of road sediment fractionated particles captured from paved surfaces, surface run-off and detention basins. Sci. Total Environ. 2012, 439, 172–186. [Google Scholar]
- Rhodes, E.P.; Ren, Z.Y.; Mays, D.C. Zinc leaching from tire crumb rubber. Environ. Sci. Technol. 2012, 46, 12856–12863. [Google Scholar] [CrossRef] [PubMed]
- Baensch-Baltruschat, B.; Kocher, B.; Stock, F.; Reifferscheid, G. Tyre and road wear particles (TRWP)—A review of generation, properties, emissions, human health risk, ecotoxicity, and fate in the environment. Sci. Total Environ. 2020, 733, 137823. [Google Scholar] [CrossRef]
- Park, I.; Kim, H.; Lee, S. Characteristics of tire wear particles generated in a laboratory simulation of tire/road contact conditions. J. Aerosol Sci. 2018, 124, 30–40. [Google Scholar] [CrossRef]
- Wik, A.; Dave, G. Occurrence and effects of tire wear particles in the environment—A critical review and an initial risk assessment. Environ. Pollut. 2009, 157, 1–11. [Google Scholar] [CrossRef]
- Baensch-Baltruschat, B.; Kocher, B.; Kochleus, C.; Stock, F.; Reifferscheid, G. Tyre and road wear particles—A calculation of generation, transport and release to water and soil with special regard to German roads. Sci. Total Environ. 2021, 752, 141939. [Google Scholar] [CrossRef]
- Bertling, J.; Bertling, R.; Hamann, L. Kunststoffe in der Umwelt: Mikro-und Makroplastik. In Ursachen, Mengen, Umweltschicksale, Wirkungen, Lösungsansätze, Empfehlungen. Kurzfassung der Konsortialstudie, Fraunhofer UMSICHT; Fraunhofer-Institut für Umwelt-, Sicherheits- und Energietechnik UMSICHT: Oberhausen, Germany, 2018. [Google Scholar]
- United States Tires Manufactures Association—USTMA 2017. Available online: https://www.ustires.org/whats-tire-0 (accessed on 28 January 2023).
- European Chemical Agency—ECHA, Annex XV Report—Rubber Granules Evaluation—Ver 1.01. 2017. Available online: https://echa.europa.eu/documents/10162/17220/annexes_to_axv_report_rubber+granules_en.pdf/f3cc9f58-8ab3-8e4a-0258-51466817f0fd?t=1488792289876 (accessed on 28 January 2023).
- Onokpise, O.U. Natural rubber, Hevea brasiliensis (Willd. ex A. Juss.) Müll. Arg, germplasm collection in the Amazon Basin, Brazil: A retrospective. Econ. Bot. 2004, 58, 544–555. [Google Scholar] [CrossRef]
- Obrecht, W.; Lambert, J.P.; Happ, M.; Oppenheimer-Stix, C.; Dunn, J.; Krüger, R. Rubber, 4. Emulsion Rubbers. In Ullmann’s Encyclopedia of Industrial Chemistry; Verlag Chemie: Hoboken, NJ, USA, 2012. [Google Scholar]
- Ahlbom, J.; Duus, U. Nya Hjulspår-En Produktstudie av Gummidäck; Report 6/94; Swedish Chemicals Agency: Solna, Sweden, 1994; p. 77. [Google Scholar]
- Barbin, W.W.; Rodgers, M.B. The science of rubber compounding. In Science and Technology of Rubber, 2nd ed.; Mark, J.E., Erman, B., Eirich, F.R., Eds.; Academic Press: San Diego, CA, USA, 1994; pp. 419–469. [Google Scholar]
- Halle, L.L.; Palmqvist, A.; Kampmann, K.; Jensen, A.; Hansen, T.; Khan, F.R. Tire wear particle and leachate exposures from a pristine and road-worn tire to Hyalella azteca: Comparison of chemical content and biological effects. Aquat. Toxicol. 2021, 232, 105769. [Google Scholar]
- Lamprea, K.; Bressy, A.; Mirande-Bret, C.; Caupos, E.; Gromaire, M.C. Alkylphenol and bisphenol A contamination of urban runoff: An evaluation of the emission potentials of various construction materials and automotive supplies. Environ. Sci. Poll. Res. 2018, 25, 21887–21900. [Google Scholar] [CrossRef]
- Celeiro, M.; Dagnac, T.; Llompart, M. Determination of priority and other hazardous substances in football fields of synthetic turf by gas chromatography-mass spectrometry: A health and environmental concern. Chemosphere 2018, 195, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Celeiro, M.; Lamas, J.P.; Garcia-Jares, C.; Dagnac, T.; Ramos, L.; Llompart, M. Investigation of PAH and other hazardous contaminant occurrence in recycled tyre rubber surfaces. case study: Restaurant playground in an indoor shopping center. Int. J. Environ. Anal. Chem. 2014, 94, 1264–1271. [Google Scholar] [CrossRef]
- Krüger, O.; Kalbe, U.; Berger, W.; Nordhau, K.; Christoph, G.; Walzel, H.P. Comparison of batch and column tests for the elution of artificial turf system components. Environ. Sci. Technol. 2012, 46, 13085–13092. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Zhao, H.; Peter, K.T.; Gonzalez, M.; Weel, J.; Wu, C.; Hu, X.; Prat, J.; Mudrock, E.; Heinger, R.; et al. A ubiquitous tire rubber–derived chemical induces acute mortality in coho salmon. Science 2021, 371, 185–189. [Google Scholar] [CrossRef]
- Alhelou, R.; Seiwert, B.; Reemtsma, T. Hexamethoxymethylmelamine—A precursor of persistent and mobile contaminants in municipal wastewater and the water cycle. Water Res. 2019, 165, 114973. [Google Scholar] [CrossRef]
- Johannessen, C.; Helm, P.; Metcalfe, C.D. Detection of selected tire wear compounds in urban receiving waters. Environ. Pollut. 2021, 287, 117659. [Google Scholar] [CrossRef]
- Peter, K.T.; Tian, Z.; Wu, C.; Lin, P.; White, S.; Du, B.; McIntyre, J.K.; Scholz, N.L.; Kolodziej, E.P. Using high-resolution mass spectrometry to identify organic contaminants linked to urban stormwater mortality syndrome in coho Salmon. Environ. Sci. Technol. 2018, 52, 10317–10327. [Google Scholar] [CrossRef]
- Seiwert, B.; Klöckner, P.; Wagner, S.; Reemtsma, T. Source-related smart suspect screening in the aqueous environment: Search for tire-derived persistent and mobile trace organic contaminants in surface waters. Anal. Bioanal. Chem. 2020, 412, 4909–4919. [Google Scholar] [CrossRef]
- Councell, T.B.; Duckenfield, K.U.; Landa, E.R.; Callender, E. Tire-Wear Particles as a Source of Zinc to the Environment. Environ. Sci. Technol. 2004, 38, 4206–4214. [Google Scholar] [CrossRef]
- Hillenbrand, T.; Toussaint, D.; Böhm, E.; Fuchs, S.; Scherer, U.; Kreissig, J.; Kotz, C. Einträge von Kupfer, Zink und Blei in Gewässer und Böden—Analyse der Emissionspfade und möglicher. In Emissionsminderungsmaßnahmen; Umweltbundesamt: Dessau, Germany, 2005. [Google Scholar]
- Knight, L.J.; Parker-Jurd, F.N.F.; Al-SidCheikh, M.; Thompson, R.C. Tyre wear particles: An abundant yet widely unreported microplastic? Environ. Sci. Pollut. Res. Int. 2020, 27, 18345–18354. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Bolan, N.; Tsang, D.C.W.; Sarkar, B.; Bradney, L.; Li, Y. A review of microplastics aggregation in aquatic environments: Influence factors, analytical methods, and environmental implications. J. Hazard. Mater. 2021, 402, 123496. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; He, L.; Peng, S.; Li, M.; Tong, M. Influence of titanium dioxide nanoparticles on the transport and deposition of microplastics in quartz sand. Environ. Pollut. 2019, 253, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Paul-Pont, I.; Hegaret, H.; Moriceau, B.; Lambert, C.; Huvet, A.; Soudant, P. Interactions between polystyrene microplastics and marine phytoplankton lead to species-specific hetero-aggregation. Environ. Pollut. 2017, 228, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, F.F.; Wang, Z.Y.; Cao, X.S.; Xing, B.S. Heteroaggregation of graphene oxide with minerals in aqueous phase. Environ. Sci. Technol. 2015, 49, 2849–2857. [Google Scholar] [CrossRef]
- Sieber, R.; Kawecki, D.; Nowack, B. Dynamic probabilistic material flow analysis of rubber release from tires into the environment. Environ. Pollut. 2020, 258, 113573. [Google Scholar]
- Unice, K.M.; Kreider, M.L.; Panko, J.M. Comparison of tire and road wear particle concentrations in sediment for watersheds in France, Japan, and the United States by quantitative pyrolysis GC/MS analysis. Environ. Sci. Technol. 2013, 47, 8138–8147. [Google Scholar]
- Kim, L.; Lee, T.Y.; Kim, H.; An, Y.J. Toxicity assessment of tire particles released from personal mobilities (bicycles, cars, and electric scooters) on soil organisms. J. Hazard. Mater. 2022, 437, 129362. [Google Scholar]
- Sheng, Y.; Liu, Y.; Wang, K.; Cizdziel, J.C.; Wu, Y.; Zhou, Y. Ecotoxicological effects of micronized car tire wear particles and their heavy metals on the earthworm (Eisenia fetida) in soil. Sci. Total Environ. 2021, 793, 148613. [Google Scholar]
- Kumata, H.; Yamada, J.; Masuda, K.; Takada, H.; Sato, Y.; Sakurai, T.; Fujiwara, K. Benzothiazolamines as tire-derived molecular markers: Sorptive behavior in street runoff and application to source apportioning. Environ. Sci. Technol. 2002, 36, 702–708. [Google Scholar]
- Kumata, H.; Sanada, Y.; Takada, H.; Ueno, T. Historical trends of n-cyclohexyl-2-benzothiazoleamine, 2-(4 morpholinyl)benzothiazole, and other anthropogenic contaminants in the urban reservoir sediment core. Environ. Sci. Technol. 2000, 34, 246–253. [Google Scholar] [CrossRef]
- Fauser, P.; Tjell, J.C.; Mosbaek, H.; Pilegaard, K. Quantification of tire-tread particles using extractable organic zinc as tracer. Rubber Chem. Technol. 1999, 72, 969–977. [Google Scholar] [CrossRef]
- Reddy, C.M.; Quinn, J.G. Environmental chemistry of benzothiazoles derived from rubber. Environ. Sci. Technol. 1997, 31, 2847–2853. [Google Scholar] [CrossRef]
- Selonen, S.; Dolar, A.; Kokalj, A.J.; Sackey, L.N.; Skalar, T.; Fernandes, V.C.; Rede, D.; Delerue-Matos, C.; Hurley, R.; Nizzetto, L.; et al. Exploring the impacts of microplastics and associated chemicals in the terrestrial environment—Exposure of soil invertebrates to tire particles. Environ. Res. 2021, 201, 111495. [Google Scholar] [CrossRef]
- Cadle, S.H.; Williams, R.L. Gas and particle emissions from automobile tires in laboratory and field studies. Rubber Chem. Technol. 1978, 52, 146–158. [Google Scholar] [CrossRef]
- Pierson, W.R.; Brachaczek, W.W. Airborne particulate debris from rubber tires. Rubber Chem. Technol. 1974, 47, 1275–1299. [Google Scholar] [CrossRef]
- Boulter, P. A Review of Emission Factors and Models for Road Vehicle Non-Exhaust Particulate Matter; TRL Limited: Bracknell, UK, 2005. [Google Scholar]
- Cardina, J.A. Particle size determination of tire-tread rubber in atmospheric dust. Rubber Chem. Technol. 1974, 47, 1005–1010. [Google Scholar] [CrossRef]
- Kim, M.G.; Yagawa, K.; Inoue, H.; Lee, Y.K.; Shirai, T. Measurement of tire tread in urban air by pyrolysis—Gas chromatography with flame photometric detection. Atmos. Environ. Part A—Gen. Top. 1990, 24, 1417–1422. [Google Scholar] [CrossRef]
- Panko, J.M.; Hitchcock, K.M.; Fuller, G.W.; Green, D. Evaluation of tire wear contribution to PM2.5 in urban environments. Atmosphere 2019, 10, 99. [Google Scholar] [CrossRef]
- Zeng, E.Y.; Tran, K.; Young, D. Evaluation of potential molecular markers for urban stormwater runoff. Environ. Monit. Assess. 2004, 90, 23–43. [Google Scholar] [CrossRef]
- Ni, H.-G.; Lu, F.-H.; Luo, X.-L.; Tian, H.-Y.; Zeng, E.Y. Occurrence, phase distribution, and mass loadings of benzothiazoles in riverine runoff of the Pearl River Delta, China. Environ. Sci. Technol. 2008, 42, 1892–1897. [Google Scholar] [CrossRef]
- Brownlee, B.G.; Carey, J.H.; MacInnis, G.A.; Pellizzari, I.T. Aquatic environmental chemistry of 2-(thiocyanomethylthio)benzothiazole and related benzothiazoles. Environ. Toxicol. Chem. 1992, 11, 1153–1168. [Google Scholar] [CrossRef]
- Wypych, G. Handbook of UV Degradation and Stabilization, 3rd ed.; Wypych, G., Ed.; Elsevier: Toronto, ON, Canada, 2020; Volume 7, p. 321. [Google Scholar]
- Wagner, S.; Klöckner, P.; Reemtsma, T. Aging of tire and road wear particles in terrestrial and freshwater environments—A review on processes, testing, analysis and impact. Chemosphere 2022, 288, 132467. [Google Scholar] [CrossRef]
- Rose, K.; Steinbüchel, A. Biodegradation of Natural Rubber and Related Compounds: Recent Insights into a Hardly Understood Catabolic Capability of Microorganisms. Appl. Environ. Microbiol. 2005, 71, 2803–2812. [Google Scholar] [CrossRef]
- Tsuchii, A.; Tokiwa, Y. Microbial degradation of tyre rubber particles. Biotechnol. Lett. 2001, 23, 963–969. [Google Scholar] [CrossRef]
- Stevenson, K.; Stallwood, B.; Hart, A.G. Tire rubber recycling and bioremediation: A review. Bioremediation J. 2008, 12, 1–11. [Google Scholar] [CrossRef]
- Spies, R.B.; Andresen, B.D.; Rice, D.W. Benzothiazoles in estuarine sediments as indicators of street runoff. Nature 1987, 327, 697–699. [Google Scholar] [CrossRef]
- Müller, A.; Kocher, B.; Altmann, K.; Braun, U. Determination of tire wear markers in soil samples and their distribution in a roadside soil. Chemosphere 2022, 294, 133653. [Google Scholar] [CrossRef]
- Hjortenkrans, D.S.T.; Bergback, B.G.; Haggerud, A.V. Metal emissions from brake linings and tires: Case studies of Stockholm, Sweden 1995/1998 and 2005. Environ. Sci. Technol. 2007, 41, 5224–5230. [Google Scholar] [CrossRef]
- Davis, A.P.; Shokouhian, M.; Ni, S. Loading estimates of lead, copper, cadmium, and zinc in urban runoff from specific sources. Chemosphere 2001, 44, 997–1009. [Google Scholar] [CrossRef]
- Dolar, A.; Selonen, S.; Van Gestel, C.A.; Perc, V.; Drobne, D.; Kokalj, A.J. Microplastics, chlorpyrifos and their mixtures modulate immune processes in the terrestrial crustacean Porcellio scaber. Sci. Total Environ. 2021, 772, 144900. [Google Scholar] [CrossRef] [PubMed]
- Selonen, S.; Dolar, A.; Kokalj, A.J.; Skalar, T.; Dolcet, L.P.; Hurley, R.; van Gestel, C.A.M. Exploring the impacts of plastics in soil– the effects of polyester textile fibers on soil invertebrates. Sci. Total Environ. 2020, 700, 134451. [Google Scholar] [CrossRef] [PubMed]
- Halle, L.L.; Palmqvist, A.; Kampmann, K.; Khan, F.R. Ecotoxicology of micronized tire rubber: Past, present and future considerations. Sci. Total Environ. 2020, 706, 135694. [Google Scholar] [CrossRef] [PubMed]
- Marwood, C.; McAtee, B.; Kreider, M.; Ogle, R.S.; Finley, B.; Sweet, L.; Panko, J. Acute aquatic toxicity of tire and road wear particles to alga, daphnid, and fish. Ecotoxicology 2011, 20, 2079–2089. [Google Scholar] [CrossRef]
- Turner, A.; Rice, L. Toxicity of tire wear particle leachate to the marine macroalga, Ulva lactuca. Environ. Pollut. 2010, 158, 3650–3654. [Google Scholar] [CrossRef]
- Cunningham, B.; Harper, B.; Brander, S.; Harper, S. Toxicity of micro and nano tire particles and leachate for model freshwater organisms. J. Hazard. Mater. 2022, 429, 128319. [Google Scholar] [CrossRef]
- Carrasco-Navarro, V.; Muñiz-González, A.B.; Sorvari, J.; Martínez-Guitarte, J.L. Altered gene expression in Chironomus riparius (insecta) in response to tire rubber and polystyrene microplastics. Environ. Pollut. 2021, 285, 117462. [Google Scholar] [CrossRef]
- Khan, F.R.; Louise Lynn Halle, L.L.; Palmqvist, A. Acute and long-term toxicity of micronized car tire wear particles to Hyalella azteca. Aquat. Toxicol. 2019, 213, 105216. [Google Scholar] [CrossRef]
- Kim, S.W.; Leifheit, E.F.; Maaß, S.; Rillig, M.C. Time-Dependent Toxicity of Tire Particles on Soil Nematodes. Front. Environ. Sci. 2021, 9, 744668. [Google Scholar] [CrossRef]
- Lackmann, C.; Velki, M.; Šimić, A.; Müller, A.; Braun, U.; Ečimović, S.; Hollert, H. Two types of microplastics (polystyrene-HBCD and car tire abrasion) affect oxidative stress-related biomarkers in earthworm Eisenia andrei in a time-dependent manner. Environ. Int. 2022, 163, 107190. [Google Scholar] [CrossRef]
- Ding, J.; Zhu, D.; Wang, H.T.; Lassen, S.B.; Chen, Q.L.; Li, G.; Lv, M.; Zhu, Y.G. Dysbiosis in the gut microbiota of soil fauna explains the toxicity of tire tread particles. Environ. Sci. Technol. 2020, 54, 7450–7460. [Google Scholar] [CrossRef] [PubMed]
- Selonen, S.; Kokalj, A.J.; Benguedouara, H.; Petroodya, S.S.A.; Dolar, A.; Drobnec, D.; van Gestel, C.A.M. Modulation of chlorpyrifos toxicity to soil arthropods by simultaneous exposure to polyester microfibers or tire particle microplastics. Appl. Soil Ecol. 2023, 181, 104657. [Google Scholar] [CrossRef]
- Dolar, A.; Drobne, D.; Dolenec, M.; Marinšek, M.; Kokalj, A.J. Time-dependent immune response in Porcellio scaber following exposure to microplastics and natural particles. Sci. Total Environ. 2022, 818, 151816. [Google Scholar] [CrossRef] [PubMed]
- Leifheit, E.F.; Kissenerl, H.L.; Faltin, E.; Ryo, M.; Rillig, M.C. Tire abrasion particles negatively affect plant growth even at low concentrations and alter soil biogeochemical cycling. Soil Ecol. Lett. 2022, 4, 409–415. [Google Scholar] [CrossRef]
- Jager, T.; Fleuren, R.H.L.J.; Hogendoorn, E.A.; de Korte, G. Elucidating the Routes of Exposure for Organic Chemicals in the Earthworm, Eisenia andrei (Oligochaeta). Environ. Sci. Technol. 2003, 37, 3399–3404. [Google Scholar] [CrossRef]
- Van Gestel, C.A.M.; Loureiro, S.; Zidar, P. Terrestrial isopods as model organisms in soil ecotoxicology: A review. ZooKeys 2018, 801, 127–162. [Google Scholar] [CrossRef]
- Loureiro, S.; Sampaio, A.; Brandão, A.; Nogueira, A.J.A.; Soares, A.M.V.M. Feeding behavior of the terrestrial isopod Porcellionides pruinosus Brandt, 1833 (Crustacea, Isopoda) in response to changes in food quality and contamination. Sci. Total Environ. 2006, 369, 119–128. [Google Scholar] [CrossRef]
- Unice, K.M.; Weeber, M.P.; Abramson, M.M.; Reid, R.C.D.; van Gils, J.A.G.; Markus, A.A.; Vethaak, A.D.; Panko, J.M. Characterizing export of land-based microplastics to the estuary—Part II: Sensitivity analysis of an integrated geospatial microplastic transport modeling assessment of tire and road wear particles. Sci. Total Environ. 2019, 646, 1650–1659. [Google Scholar] [CrossRef]
- Caetano, A.L.; Marques, C.R.; Gonc¸alves, F.; da Silva, E.F.; Pereira, R. Copper toxicity in a natural reference soil: Ecotoxicological data for the derivation of preliminary soil screening values. Ecotoxicology 2016, 25, 163–177. [Google Scholar] [CrossRef]
- Klöckner, P.; Reemtsma, T.; Eisentraut, P.; Braun, U.; Ruhl, A.S.; Wagner, S. Tire and road wear particles in road environment—Quantification and assessment of particle dynamics by Zn determination after density separation. Chemosphere 2019, 222, 714–721. [Google Scholar] [CrossRef]
- Redondo-Hasselerharm, P.E.; de Ruijter, V.N.; Mintenig, S.M.; Verschoor, A.; Koelmans, A.A. Ingestion and Chronic Effects of Car Tire Tread Particles on Freshwater Benthic Macroinvertebrates. Environ. Sci. Technol. 2018, 52, 13986–13994. [Google Scholar] [CrossRef] [PubMed]
- Asheim, J.; Vike-Jonas, K.; Gonzalez, S.V.; Lierhagen, S.; Venkatraman, V.; Veivåg, I.L.S.; Snilsberg, B.; Flaten, T.P.; Asimakopoulos, A.G. Benzotriazoles, benzothiazoles and trace elements in an urban road setting in Trondheim, Norway: Re-visiting the chemical markers of traffic pollution. Sci. Total Environ. 2019, 649, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Maliszewska-Kordybach, B. Polycyclic aromatic hydrocarbons in agricultural soils in Poland: Preliminary proposals for criteria to evaluate the level of soil contamination. Appl. Geochem. 1996, 11, 121–127. [Google Scholar] [CrossRef]
- Jekel, M. Scientific Report on Tyre and Road Wear Particles, TRWP, in the Aquatic Environment; European Tyre Rubber Manufactures Association (ETRMA): Brussels, Belgium, 2019; pp. 1–35. [Google Scholar]
- Luo, Z.; Zhou, X.; Su, Y.; Wang, H.; Yu, R.; Zhou, S.; Xu, E.G.; Xing, B. Environmental occurrence, fate, impact, and potential solution of tire microplastics: Similarities and differences with tire wear particles. Sci. Total Environ. 2021, 795, 148902. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Xiong, X.; He, M.; Tsang, D.C.; Gupta, J.; Khan, E.; Bolan, N.S. Microplastics as pollutants in agricultural soils. Environ. Pollut. 2020, 265, 114980. [Google Scholar] [CrossRef] [PubMed]
- Verschoor, A.J.; Van Gelderen, A.; Hofstra, U. Fate of recycled tyre granulate used on artificial turf. Environ. Sci. Eur. 2021, 33, 27. [Google Scholar] [CrossRef]
- Araujo-Morera, J.; Lopez-Manchado, M.A.; Verdejo, R.; Hernandez, S.M. Sustainable mobility: The route of tires through the circular economy model. Waste Manag. 2021, 126, 309–322. [Google Scholar] [CrossRef]
- Jomin, T.; Renuka, P. Road to Sustainable Tire Materials: Current State-of-the-Art and Future Prospectives. Environ. Sci. Technol. 2023, 57, 2209–2216. [Google Scholar]
- Ren, X.; Barrera, C.S.; Tardiff, J.L.; Gil, A.; Cornish, K. Liquid Guayule Natural Rubber, a Renewable and Crosslinkable Processing Aid in Natural and Synthetic Rubber Compounds. J. Clean. Prod. 2020, 276, 122933. [Google Scholar] [CrossRef]
- Rodgers, S.; Meng, F.; Poulston, S.; Conradie, A.; McKechnie, J. Renewable Butadiene: A Case for Hybrid Processing via Bio- and Chemo-Catalysis. J. Clean. Prod. 2022, 364, 132614. [Google Scholar] [CrossRef]
- Bartoli, M.; Rosi, L.; Frediani, M.; Undri, A.; Frediani, P. Depolymerization of Polystyrene at Reduced Pressure through a Microwave Assisted Pyrolysis. J. Anal. Appl. Pyrolysis 2015, 113, 281–287. [Google Scholar] [CrossRef]
- Abdulrahman, A.S.; Jabrail, F.H. Treatment of Scrap Tire for Rubber and Carbon Black Recovery. Recycling 2022, 7, 27. [Google Scholar] [CrossRef]

| Compartments | [TRWPs] | References |
|---|---|---|
| Road face | 0.7–210 g/kg 1 | [60,61,62,63] |
| Soil | 0.6–117 g/kg 1 | [62,64,65,66] |
| Sediment | 0.3–155 g/kg 1 | [16,34,60,63] |
| Sewage | 0.3–179 mg/L | [34,60,61,63,71] |
| Freshwater | 0.09–6.4 mg/L | [34,61,72,73] |
| Air | 0.4–11 µg/m3 | [61,62,64,66,68,69,70] |
| Markers | Methods | Specificity | References |
|---|---|---|---|
| SBR | Thermal extraction desorption gas chromatography–mass spectrometry (TED-GC/MS) Fourier transform infrared spectroscopy (FTIR) Raman spectroscopy | High | [25,34,62,65,66,69] |
| BTs | Thermal extraction desorption gas chromatography–mass spectrometry (TED-GC/MS) | Medium | [34,60,61,70,72,78] |
| Zn | Inductively coupled plasma–optical emission spectroscopy (ICP-OES) | Medium/low | [16,34,79,80,81,82,83] |
| Species | TRWPs’ Average Size (μm) | [TRWPs’] Range (mg/kd d.w.) | Exposition Time (Days) | Soil | Physical Parameters | Analyses | Ref. | |
|---|---|---|---|---|---|---|---|---|
| Edaphic fauna | C. elegans | 125 μm | 1–10.000 mg/kg | Acute test: 2 days Chronic test: 12 days | Texture: sand 93.3%, silt 5.0%, and clay 1.7% pH: 6.7 ± 0.2 | Temperature: 20 ± 2 °C WHC: 0.34 ± 0.10 mL/g Photoperiod: in the dark | Endpoint (%): Survival rate, body length, and brood size Chemical analysis: ICP-OES | [95] |
| E. andrei | <600 µm | 1–1000 mg/kg | Acute test: 48 h Chronic test: 28 days | Texture: fine sand 95.5%, silt and clay 4.5% Organic Carbon: 2.5% pH: N.A. | Temperature: 20 ± 2 °C WHC: 50% Photoperiod: N.A. | Endpoint (%): Net response (avoidance test) and brood size Biochemical parameters: Protein content, ROS, GST, CAT, GR, AChE, CES, GPx, and MXR | [96] | |
| E. fetida | 606.25 μm | 10,000–200,000 mg/kg | Chronic test: 14–28 days | Standard soil (OECD 220 guideline): kaolinite clay 20%, quartz sand 70%, and sphagnum peat 10% Organic carbon: 5% Nitrogen: 0.098% pH: 6.9 | Temperature: 20 ± 2 °C WHC: 25% Photoperiod: 12:12 h | Biochemical parameters: SOD, CAT, POD, GSH, and MDA Chemical analysis: ICP-MS, ATR-FTIR, and SEM | [65] | |
| E. crypticus | 443.25 μm | 48–30,000 mg/kg | Chronic test: 21 days | LUFA 2.2 soil: loamy sand Organic carbon: 1.73% Nitrogen: 0.19% pH: 5.6–4.94 | Temperature: 20 °C WHC: 50–60% Photoperiod: 16:8 h | Endpoint (%): Survival, brood size, and gut microbial alteration Chemical analysis: GC-MS and ICP-MS | [68,97] | |
| F. candida | 382.5 µm | 200–15,000 mg/kg | Chronic test: 28 days | LUFA 2.2 soil: loamy sand Organic carbon: 1.73% Nitrogen: 0.19% pH: 5.6–4.94 | Temperature: 20 °C WHC: 50% Photoperiod: 16:8 h | Endpoint (%): Survival, body length, and brood size | [62,68,98] | |
| P. scaber | 141.45 μm | 200–15,000 mg/kg | Chronic test: 21 days | LUFA 2.2 soil: loamy sand Organic carbon: 1.73% Nitrogen: 0.19% pH: 5.6–4.94 | Temperature: 20 °C WHC: 40% Photoperiod: in the dark | Biochemical analysis: AChE and ETS Genetic analysis: Expression of immune-related genes in hemocytes and the digestive gland and hepatopancreas | [98,99] | |
| Plants | A. porrum | 125 μm | 10,000–160,000 mg/kg | Chronic test: 42 days | Albic Luvisol: loamy sand Organic carbon: 1.87% Nitrogen: 0.12% pH: 5.41 | Temperature: 22–18 °C WHC: 40% Photoperiod: 12:12 h | Endpoint (%): Effects on plant growth, change soil pH, and alteration in litter decomposition and respiration | [100] |
| V. radiata | 326.5 μm | 1–10 g/kg | Chronic test: 28 days | Loamy sand organic matter (SOM): 0.9% pH: 5.4 | Temperature: 26 °C WHC: 80% Photoperiod: 16:8 h | Endpoint (%): Growth rate of the shoots and leaves and root length Biochemical analyses: Content of polyphenolic compounds (anthocyanins, chlorophyll, flavonoids, and nitrogen balance index), and photosynthetic activities | [62] | |
| Vehicles | EFTRWPs (mg/km) | ||
|---|---|---|---|
| Urban | Rural | Highway | |
| Car | 1.32 × 102 | 8.5 × 101 | 1.0 × 102 |
| Bus | 4.2 × 102 | 2.7 × 102 | 3.3 × 102 |
| Motorcycle | 6.0 × 101 | 3.9 × 101 | 4.7 × 101 |
| Truck | 6.6 × 102 | 4.2 × 102 | 5.2 × 102 |
| Lorry | 8.5 × 102 | 5.5 × 102 | 6.7 × 102 |
| Substances | PNECSOIL [mg/kg d.w.] | PECSOIL [mg/kg d.w.] | RQ | |
|---|---|---|---|---|
| Tire Particles | 10 4 | 21.62 | 2.16 * | |
| Organic Chemicals (non-PAHs) | Benzothiazole | 0.017 2 | 0.060 | 3.58 * |
| 1-indanone | - | 0.002 | - | |
| 1-octanethiol | - | 0.0008 | - | |
| Organic Chemicals (PAHs) | Pyrene | 1 2 | 0.037 | 0.037 |
| Fluoranthene | 1.5 2 | 0.018 | 0.012 | |
| Phenanthrene | 1.8 2 | 0.009 | 0.005 | |
| Benzo[ghi]perylene | 0.17 2 | 0.004 | 0.024 | |
| Anthracene | 0.13 2 | 0.002 | 0.022 | |
| Acenaphthylene | 0.29 2 | 0.0004 | 0.001 | |
| Benzo(b)fluoranthene | 0.28 2 | 0.006 | 0.022 | |
| Chrysene | 0.55 2 | 0.012 | 0.023 | |
| Indeno(1.2.3-cd)pyrene | 0.13 2 | 0.002 | 0.022 | |
| Fluorene | 1 2 | 0.004 | 0.004 | |
| Naphthalene | 1 2 | 0.0002 | 0.0002 | |
| Benzo[a]anthracene | 0.079 2 | 0.008 | 0.105 | |
| Benzo(a)pyrene | 0.053 2 | 0.004 | 0.075 | |
| Acenaphthene | 0.038 2 | 0.0001 | 0.005 | |
| Benzo(k)fluoranthene | 0.27 2 | 0.001 | 0.007 | |
| Dibenzo(a.h)anthracene | 0.054 2 | 0.001 | 0.034 | |
| Metals | Zn | 83.1 1 | 19.62 | 0.23 |
| Fe | - | 0.37 | - | |
| Al | - | 0.76 | - | |
| Cu | 65 1,3 | 0.42 | 0.006 | |
| Cr | 21.1 1 | 0.06 | 0.003 | |
| Ni | 29.9 1 | 0.06 | 0.002 | |
| Pb | 212 1 | 0.06 | 0.0003 | |
| Hg | 0.022 1 | 0.0003 | 0.018 | |
| Cd | 0.9 1 | 0.001 | 0.002 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Federico, L.; Masseroni, A.; Rizzi, C.; Villa, S. Silent Contamination: The State of the Art, Knowledge Gaps, and a Preliminary Risk Assessment of Tire Particles in Urban Parks. Toxics 2023, 11, 445. https://doi.org/10.3390/toxics11050445
Federico L, Masseroni A, Rizzi C, Villa S. Silent Contamination: The State of the Art, Knowledge Gaps, and a Preliminary Risk Assessment of Tire Particles in Urban Parks. Toxics. 2023; 11(5):445. https://doi.org/10.3390/toxics11050445
Chicago/Turabian StyleFederico, Lorenzo, Andrea Masseroni, Cristiana Rizzi, and Sara Villa. 2023. "Silent Contamination: The State of the Art, Knowledge Gaps, and a Preliminary Risk Assessment of Tire Particles in Urban Parks" Toxics 11, no. 5: 445. https://doi.org/10.3390/toxics11050445
APA StyleFederico, L., Masseroni, A., Rizzi, C., & Villa, S. (2023). Silent Contamination: The State of the Art, Knowledge Gaps, and a Preliminary Risk Assessment of Tire Particles in Urban Parks. Toxics, 11(5), 445. https://doi.org/10.3390/toxics11050445







