Perinatal Lead Exposure Promotes Sex-Specific Epigenetic Programming of Disease-Relevant Pathways in Mouse Heart
Abstract
1. Introduction
2. Materials and Methods
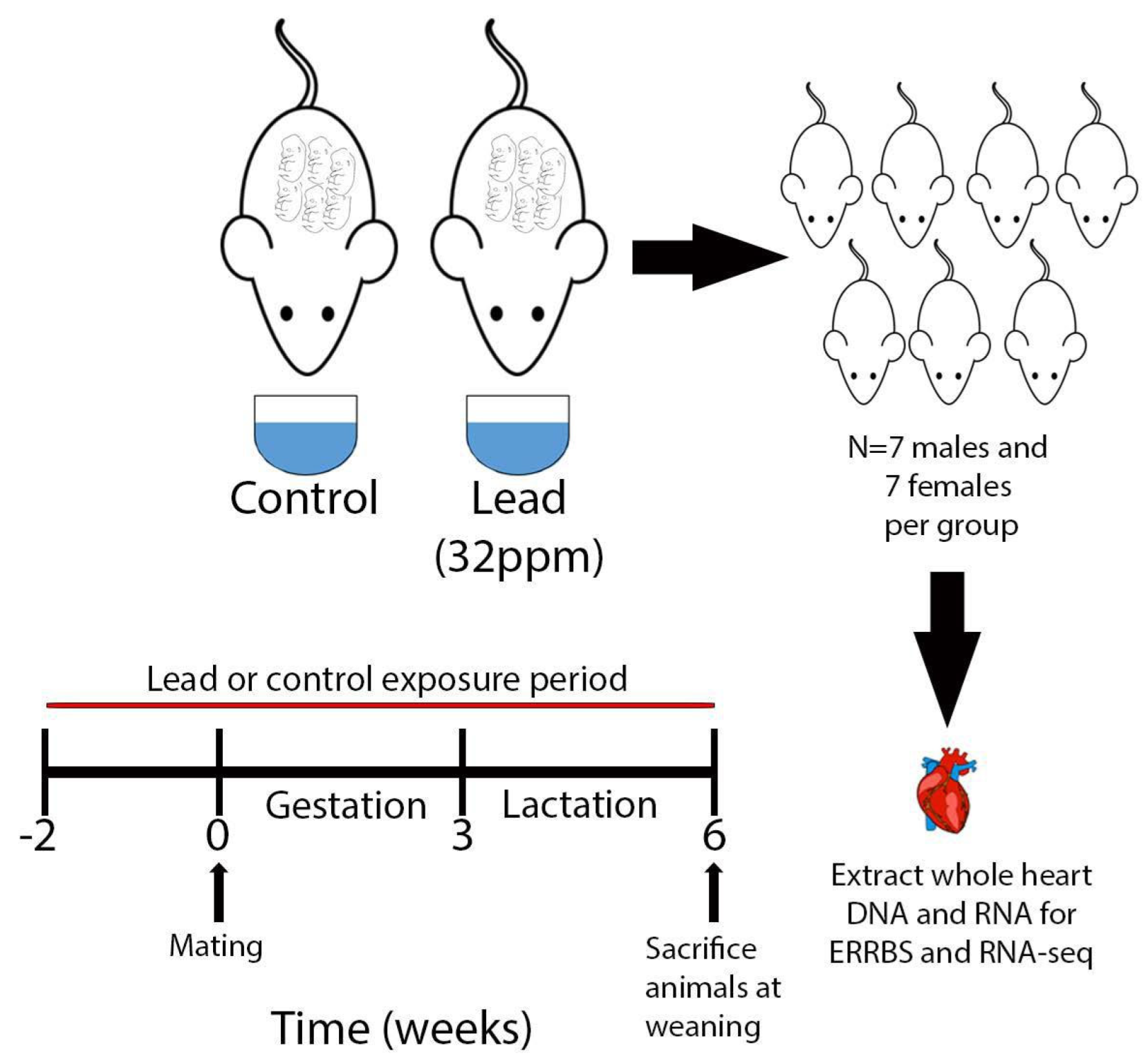
2.1. Animals and Study Paradigm
2.2. Tissue Collection and Extraction of RNA and DNA
2.3. Gene Expression Analysis
2.4. Enhanced Reduced Representation Bisulfite Sequencing
2.5. Bioinformatics Analysis of ERRBS Data
2.6. Gene Set Enrichment Analysis of RNA-seq and ERRBS Data
2.7. Analysis of Overlap in DNA Methylation between Sexes
2.8. Validating a Potential Pb-Biomarker in a Human Study
3. Results
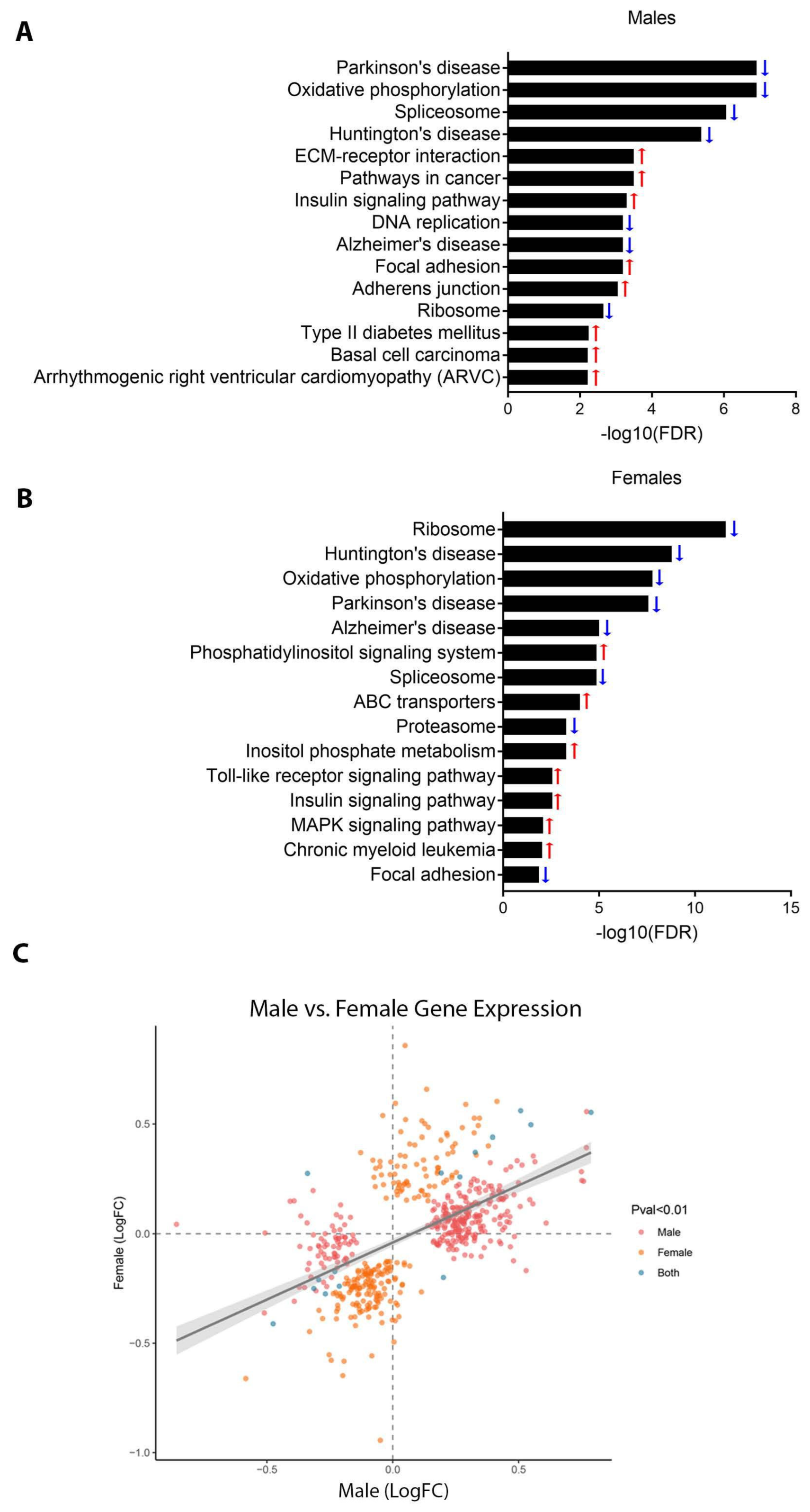
3.1. Effect of Perinatal Pb Exposure on Gene Expression
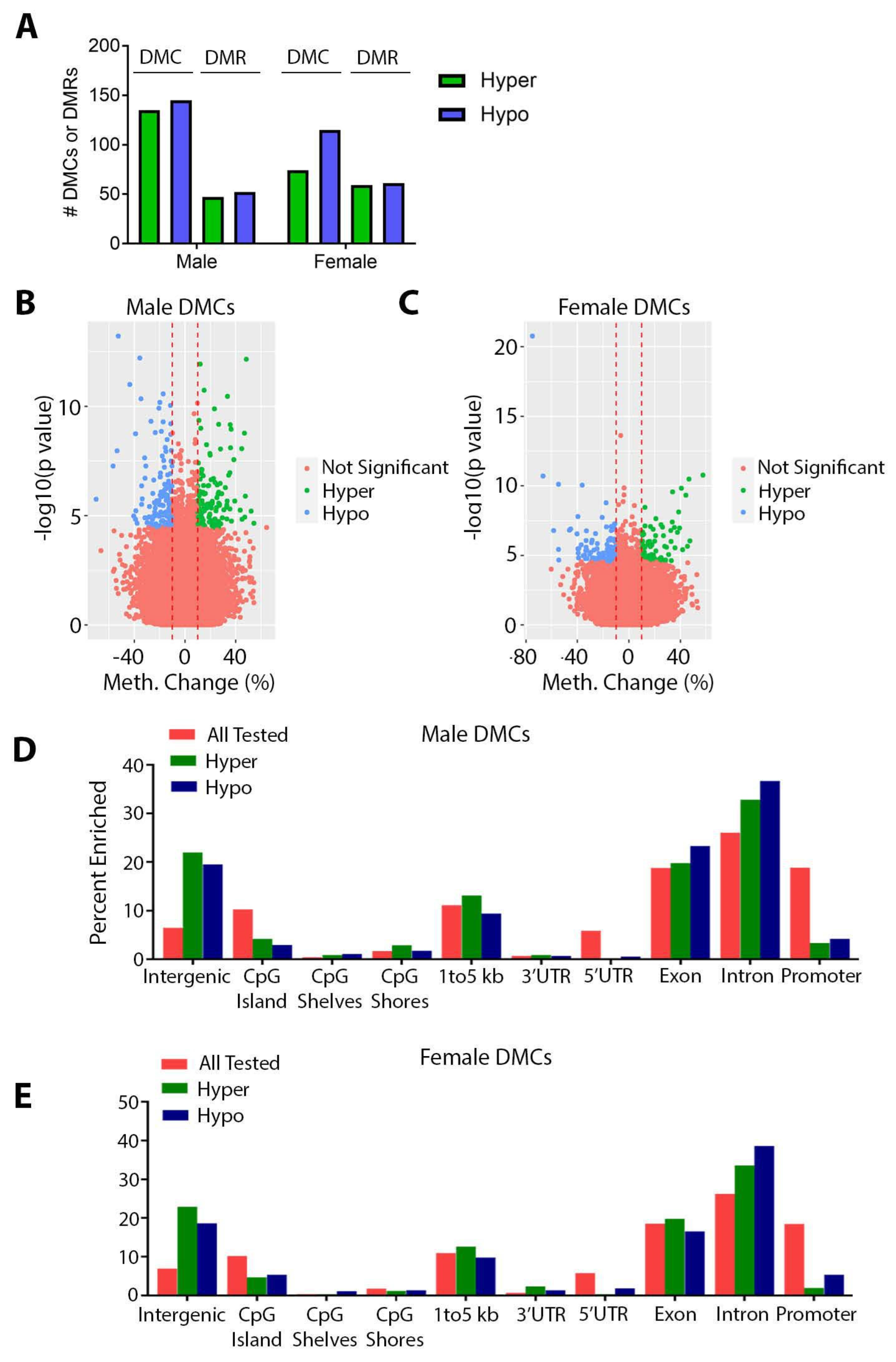
3.2. Effects of Perinatal Pb Exposure on DNA Methylation
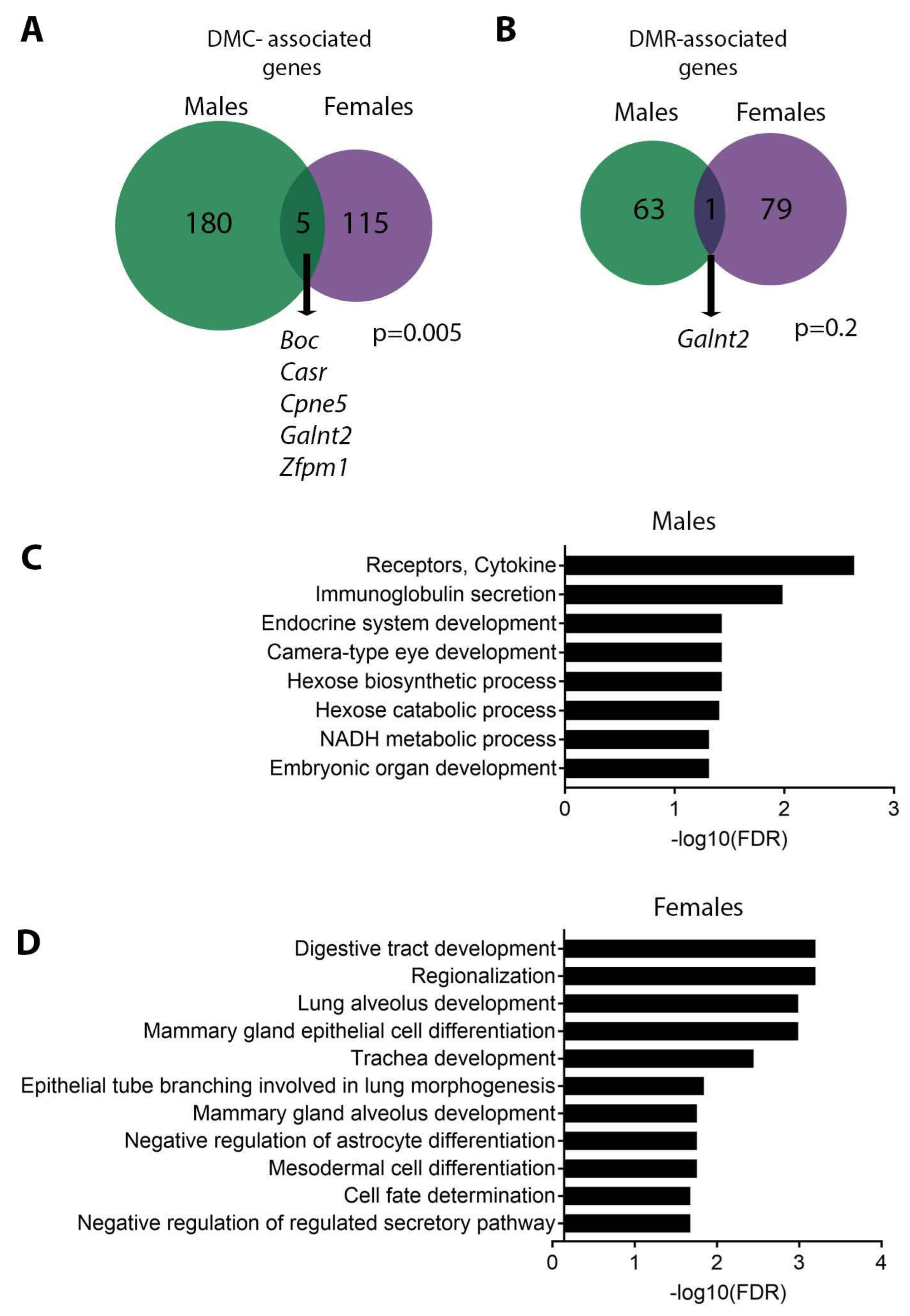
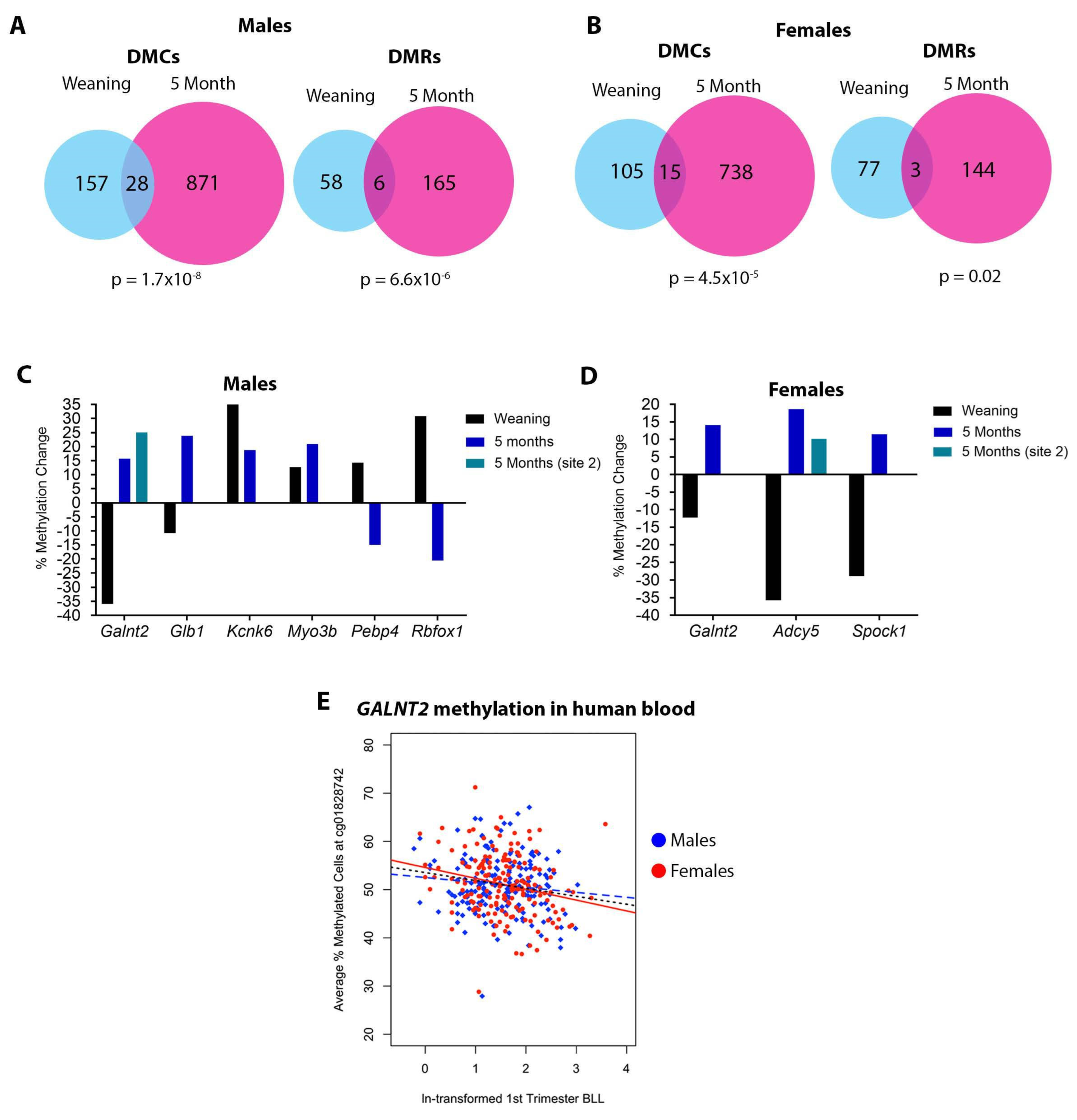
3.3. Sex Specificity of Pb-Induced Changes in DNA Methylation
3.4. DNA Methylation at Weaning vs. 5 Months of Age
3.5. Effects of Gestational Pb Exposure on GALNT2 Methylation in a Human Cohort
4. Discussion
4.1. Pb Effects on Body Weight
4.2. Effects of Pb on Gene Expression Pathways
4.3. Effects of Pb on DNA Methylation
4.4. Pb Exposure and Galnt2 Methylation
4.5. Limitations of the Study
| Gene | Differential Methylation in Males or Females? | Link to Cardiovascular Development or Disease | Reference |
|---|---|---|---|
| Galnt2 | Both | Regulation of HDL cholesterol; promoter hypermethylation associated with coronary heart disease; DNA methylation at GALNT2 associated with sex differences in cardiometabolic diseases | [94,95,96,105] |
| Glb1 | Males | Mutation of GLB1 associated with cardiomyopathy in humans | [106] |
| Kcnk6 | Males | Channel encoded by this gene functions in ventricular repolarization; Gene deficiency in mice leads to pulmonary hypertension | [107,108] |
| Myo3b | Males | Expressed in cardiac mesoderm stage of cardiac differentiation | [109] |
| Pebp4 | Males | Expressed in adventitial layer of coronary arteries | [110] |
| Rbfox1 | Males | Functions in cardiac gene splicing and is down-regulated in heart failure; copy number variants associated with stress cardiomyopathy; gene variants associated with lower blood pressure | [111,112,113,114] |
| Adcy5 | Females | Adenylyl cyclase 5 exacerbates oxidative stress and cardiomyopathy in response to chronic adrenergic stimulation; knockout of Adcy5 in mice protects against aging-induced cardiomyopathy; enhances myocardial contractility and function during exercise | [115,116,117] |
| Spock1 | Females | Plays a role in calcification of the vasculature; and in collagen deposition during cardiac fibrosis | [118,119] |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chowdhury, R.; Ramond, A.; O’Keeffe, L.M.; Shahzad, S.; Kunutsor, S.K.; Muka, T.; Gregson, J.; Willeit, P.; Warnakula, S.; Khan, H.; et al. Environmental toxic metal contaminants and risk of cardiovascular disease: Systematic review and meta-analysis. BMJ 2018, 362, k3310. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.M.; Entringer, S.; Buss, C.; Wadhwa, P.D. Developmental Origins of Health and Disease: Environmental Exposures. Semin. Reprod. Med. 2009, 27, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.P. The origins of the developmental origins theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Thornburg, K.L. The programming of cardiovascular disease. J. Dev. Orig. Health Dis. 2015, 6, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, A.P. The Key Role of Epigenetics in Human Disease Prevention and Mitigation. N. Engl. J. Med. 2018, 378, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Pepin, M.E.; Drakos, S.; Ha, C.-M.; Tristani-Firouzi, M.; Selzman, C.H.; Fang, J.C.; Wende, A.R.; Wever-Pinzon, O. DNA methylation reprograms cardiac metabolic gene expression in end-stage human heart failure. Am. J. Physiol. Circ. Physiol. 2019, 317, H674–H684. [Google Scholar] [CrossRef]
- Perera, B.; Faulk, C.; Svoboda, L.K.; Goodrich, J.M.; Dolinoy, D.C. The role of environmental exposures and the epigenome in health and disease. Environ. Mol. Mutagen. 2020, 61, 176–192. [Google Scholar] [CrossRef]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef]
- Gilsbach, R.; Preissl, S.; Grüning, B.; Schnick, T.; Burger, L.; Benes, V.; Würch, A.; Bönisch, U.; Günther, S.; Backofen, R.; et al. Dynamic DNA methylation orchestrates cardiomyocyte development, maturation and disease. Nat. Commun. 2014, 5, 5288. [Google Scholar] [CrossRef]
- Tabish, A.M.; Arif, M.; Song, T.; Elbeck, Z.; Becker, R.C.; Knöll, R.; Sadayappan, S. Association of intronic DNA methylation and hydroxymethylation alterations in the epigenetic etiology of dilated cardiomyopathy. Am. J. Physiol. Circ. Physiol. 2019, 317, H168–H180. [Google Scholar] [CrossRef]
- Ciccarone, F.; Castelli, S.; Ioannilli, L.; Ciriolo, M.R. High Dietary Fat Intake Affects DNA Methylation/Hydroxymethylation in Mouse Heart: Epigenetic Hints for Obesity-Related Cardiac Dysfunction. Mol. Nutr. Food Res. 2019, 63, e1800970. [Google Scholar] [CrossRef]
- Murray, R.; Bryant, J.; Titcombe, P.; Barton, S.J.; Inskip, H.; Harvey, N.C.; Cooper, C.; Lillycrop, K.; Hanson, M.; Godfrey, K.M. DNA methylation at birth within the promoter of ANRIL predicts markers of cardiovascular risk at 9 years. Clin. Epigenet. 2016, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Lund, G.; Andersson, L.; Lauria, M.; Lindholm, M.; Fraga, M.F.; Villar-Garea, A.; Ballestar, E.; Esteller, M.; Zaina, S. DNA methylation polymorphisms precede any histological sign of atherosclerosis in mice lacking apolipo-protein E. J. Biol. Chem. 2004, 279, 29147–29154. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Agha, G.; Baccarelli, A.A. The Role of DNA Methylation in Cardiovascular Risk and Disease: Methodological Aspects, Study Design, and Data Analysis for Epidemiological Studies. Circ. Res. 2016, 118, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Levin, R.; Vieira, C.L.Z.; Rosenbaum, M.H.; Bischoff, K.; Mordarski, D.C.; Brown, M.J. The urban lead (Pb) burden in humans, animals and the natural environment. Environ. Res. 2021, 193, 110377. [Google Scholar] [CrossRef]
- Obeng-Gyasi, E. Sources of lead exposure in various countries. Rev. Environ. Health 2019, 34, 25–34. [Google Scholar] [CrossRef]
- Lanphear, B.P.; Rauch, S.; Auinger, P.; Allen, R.W.; Hornung, R.W. Low-level lead exposure and mortality in US adults: A population-based cohort study. Lancet Public Health 2018, 3, e177–e184. [Google Scholar] [CrossRef]
- Kiełtucki, J.; Dobrakowski, M.; Pawlas, N.; Sredniawa, B.; Boroń, M.; Kasperczyk, S. The analysis of QT interval and repolarization morphology of the heart in chronic exposure to lead. Hum. Exp. Toxicol. 2017, 36, 1081–1086. [Google Scholar] [CrossRef]
- Basha, D.C.; Basha, S.S.; Reddy, G.R. Lead-induced cardiac and hematological alterations in aging Wistar male rats: Alleviating effects of nutrient metal mixture. Biogerontology 2012, 13, 359–368. [Google Scholar] [CrossRef]
- Afridi, H.I.; Kazi, T.G.; Kazi, N.; Kandhro, G.A.; Baig, J.A.; Jamali, M.K.; Arain, M.B.; Shah, A.Q.; Shah, F.; Khan, S.; et al. Association of Environmental Toxic Elements in Biological Samples of Myocardial Infarction Patients at Different Stages. Biol. Trace Element Res. 2011, 141, 26–40. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Guallar, E.; Silbergeld, E.K.; Rothenberg, S.J. Lead Exposure and Cardiovascular Disease—A Systematic Review. Environ. Health Perspect. 2007, 115, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Huo, X.; Chen, G.; Luo, X.; Xu, X. Lead (Pb) exposure and heart failure risk. Environ. Sci. Pollut. Res. 2021, 28, 28833–28847. [Google Scholar] [CrossRef] [PubMed]
- Shvachiy, L.; Geraldes, V.; Amaro-Leal, Â.; Rocha, I. Intermittent low-level lead exposure provokes anxiety, hypertension, autonomic dysfunction and neuroinflammation. Neurotoxicology 2018, 69, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Davuljigari, C.B.; Gottipolu, R.R. Late-life Cardiac Injury in Rats following Early Life Exposure to Lead: Reversal Effect of Nutrient Metal Mixture. Cardiovasc. Toxicol. 2020, 20, 249–260. [Google Scholar] [CrossRef]
- Hejtmancik, M., Jr.; Williams, B.J. Time and level of perinatal lead exposure for development of norepinephrine cardio-toxicity. Res. Commun. Chem. Pathol. Pharm. 1979, 24, 367–376. [Google Scholar]
- Williams, B.J.; Hejtmancik, M.R., Jr.; Abreu, M. Cardiac effects of lead. Fed. Proc. 1983, 42, 2989–2993. [Google Scholar]
- Yin, J.; Wang, A.-P.; Li, W.-F.; Shi, R.; Jin, H.-T.; Wei, J.-F. Sensitive biomarkers identification for differentiating Cd and Pb induced toxicity on zebrafish embryos. Environ. Toxicol. Pharmacol. 2017, 56, 340–349. [Google Scholar] [CrossRef]
- Ou, Y.; Bloom, M.S.; Nie, Z.; Han, F.; Mai, J.; Chen, J.; Lin, S.; Liu, X.; Zhuang, J. Associations between toxic and essential trace elements in maternal blood and fetal congenital heart defects. Environ. Int. 2017, 106, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Huo, X.; Zhang, S.; Cheng, Z.; Huang, Y.; Xu, X. Relations of blood lead levels to echocardiographic left ventricular structure and function in preschool chil-dren. Chemosphere 2021, 268, 128793. [Google Scholar] [CrossRef]
- Farzan, S.F.; Howe, C.G.; Chen, Y.; Gilbert-Diamond, D.; Cottingham, K.L.; Jackson, B.P.; Weinstein, A.R.; Karagas, M.R. Prenatal lead exposure and elevated blood pressure in children. Environ. Int. 2018, 121, 1289–1296. [Google Scholar] [CrossRef]
- Vaziri, N.D. Mechanisms of lead-induced hypertension and cardiovascular disease. Am. J. Physiol. Circ. Physiol. 2008, 295, H454–H465. [Google Scholar] [CrossRef] [PubMed]
- Regitz-Zagrosek, V.; Kararigas, G. Mechanistic Pathways of Sex Differences in Cardiovascular Disease. Physiol. Rev. 2017, 97, 1–37. [Google Scholar] [CrossRef]
- Ebert, S.N.; Liu, X.-K.; Woosley, R.L. Female Gender as a Risk Factor for Drug-Induced Cardiac Arrhythmias: Evaluation of Clinical and Experimental Evidence. J. Women’s Health 1998, 7, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, M.; Varma, G.; Adams, B.; Anderson, D.W.; Schneider, J.S.; Cory-Slechta, D.A. Developmental Lead Exposure and Prenatal Stress Result in Sex-Specific Reprograming of Adult Stress Physiology and Epigenetic Profiles in Brain. Toxicol. Sci. 2018, 163, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Faulk, C.; Barks, A.; Liu, K.; Goodrich, J.M.; Dolinoy, D.C. Early-life lead exposure results in dose- and sex-specific effects on weight and epigenetic gene regulation in weanling mice. Epigenomics 2013, 5, 487–500. [Google Scholar] [CrossRef]
- Aljahdali, A.A.; Peterson, K.E.; Cantoral, A.; Ruiz-Narvaez, E.; Tellez-Rojo, M.M.; Kim, H.M.; Hébert, J.R.; Wirth, M.D.; Torres-Olascoaga, L.A.; Shivappa, N.; et al. Diet Quality Scores and Cardiometabolic Risk Factors in Mexican Children and Adolescents: A Lon-gitudinal Analysis. Nutrients 2022, 14, 896. [Google Scholar] [CrossRef]
- Liu, Y.; Ettinger, A.S.; Téllez-Rojo, M.; Sánchez, B.N.; Zhang, Z.; Cantoral, A.; Hu, H.; Peterson, K.E. Prenatal Lead Exposure, Type 2 Diabetes, and Cardiometabolic Risk Factors in Mexican Children at Age 10–18 Years. J. Clin. Endocrinol. Metab. 2020, 105, 210–218. [Google Scholar] [CrossRef]
- Wang, T.; Pehrsson, E.C.; Purushotham, D.; Li, D.; Zhuo, X.; Zhang, B.; Lawson, H.A.; Province, M.A.; Krapp, C.; Lan, Y.; et al. The NIEHS TaRGET II Consortium and environmental epigenomics. Nat. Biotechnol. 2018, 36, 225–227. [Google Scholar] [CrossRef]
- Svoboda, L.K.; Neier, K.; Wang, K.; Cavalcante, R.G.; Rygiel, C.A.; Tsai, Z.; Jones, T.R.; Liu, S.; Goodrich, J.M.; Lalancette, C.; et al. Tissue and sex-specific programming of DNA methylation by perinatal lead exposure: Implications for environmental epigenetics studies. Epigenetics 2020, 16, 1102–1122. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Annest, J.L.; Roberts, J.; Murphy, R.S. National estimates of blood lead levels: United States, 1976–1980: Association with selected demographic and socioeconomic factors. N. Engl. J. Med. 1982, 307, 573–579. [Google Scholar] [CrossRef]
- Leech, T.G.; Adams, E.A.; Weathers, T.D.; Staten, L.K.; Filippelli, G.M. Inequitable Chronic Lead Exposure: A Dual Legacy of Social and Environmental Injustice. Fam. Community Health 2016, 39, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.; Peng, L.; Xu, X.; Zheng, L.; Qiu, B.; Qi, Z.; Zhang, B.; Han, D.; Piao, Z. Elevated Blood Lead Levels of Children in Guiyu, an Electronic Waste Recycling Town in China. Environ. Health Perspect. 2007, 115, 1113–1117. [Google Scholar] [CrossRef] [PubMed]
- Caravanos, J.; Dowling, R.; Dra, M.M.T.-R.; Cantoral, A.; Kobrosly, R.; Estrada, D.; Orjuela, M.; Gualtero, S.; Msc, B.E.; Rivera, A.; et al. Blood Lead Levels in Mexico and Pediatric Burden of Disease Implications. Ann. Glob. Health 2014, 80, 269–277. [Google Scholar] [CrossRef]
- Svoboda, L.; Wang, K.; Jones, T.; Colacino, J.; Sartor, M.; Dolinoy, D. Sex-Specific Alterations in Cardiac DNA Methylation in Adult Mice by Perinatal Lead Exposure. Int. J. Environ. Res. Public Health 2021, 18, 577. [Google Scholar] [CrossRef] [PubMed]
- Trim Galore. Available online: https://www.bioinformatics.babraham.ac.uk/projects/trim_galore/ (accessed on 19 November 2019).
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. Babraham Bioinformatics; Babraham Institute: Cambridge, UK, 2010; Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 19 November 2019).
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. EdgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Patil, S.; Sartor, M.A. RNA-Enrich: A cut-off free functional enrichment testing method for RNA-seq with improved detection power. Bioinformatics 2016, 32, 1100–1102. [Google Scholar] [CrossRef] [PubMed]
- Garrett-Bakelman, F.E.; Sheridan, C.K.; Kacmarczyk, T.J.; Ishii, J.; Betel, D.; Alonso, A.; Mason, C.E.; Figueroa, M.E.; Melnick, A.M. Enhanced Reduced Representation Bisulfite Sequencing for Assessment of DNA Methylation at Base Pair Resolution. J. Vis. Exp. 2015, 10, e52246. [Google Scholar] [CrossRef]
- Akalin, A.; Garrett-Bakelman, F.; Kormaksson, M.; Busuttil, J.; Zhang, L.; Khrebtukova, I.; Milne, T.; Huang, Y.; Biswas, R.; Hess, J.; et al. Base-Pair Resolution DNA Methylation Sequencing Reveals Profoundly Divergent Epigenetic Landscapes in Acute Myeloid Leukemia. PLoS Genet. 2012, 8, e1002781. [Google Scholar] [CrossRef]
- Park, Y.; Figueroa, M.E.; Rozek, L.S.; Sartor, M.A. MethylSig: A whole genome DNA methylation analysis pipeline. Bioinformatics 2014, 30, 2414–2422. [Google Scholar] [CrossRef]
- Cavalcante, R.G.; Sartor, M.A. Annotatr: Genomic regions in context. Bioinformatics 2017, 33, 2381–2383. [Google Scholar] [CrossRef] [PubMed]
- Sartor, M.A.; Leikauf, G.; Medvedovic, M. LRpath: A logistic regression approach for identifying enriched biological groups in gene expression data. Bioinformatics 2009, 25, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.T.; Cavalcante, R.G.; Lee, C.; Qin, T.; Patil, S.; Wang, S.; Tsai, Z.T.Y.; Boyle, A.P.; Sartor, M.A. Poly-Enrich: Count-based methods for gene set enrichment testing with genomic regions. NAR Genom. Bioinform. 2020, 2, lqaa006. [Google Scholar] [CrossRef]
- Dolinoy, D.C.; Weidman, J.R.; Jirtle, R.L. Epigenetic gene regulation: Linking early developmental environment to adult disease. Reprod. Toxicol. 2007, 23, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Perng, W.; Tamayo-Ortiz, M.; Tang, L.; Sánchez, B.N.; Cantoral, A.; Meeker, J.D.; Dolinoy, D.C.; Roberts, E.F.; Martinez-Mier, E.A.; Lamadrid-Figueroa, H.; et al. Early Life Exposure in Mexico to ENvironmental Toxicants (ELEMENT) Project. BMJ Open 2019, 9, e030427. [Google Scholar] [CrossRef]
- Jansen, E.C.; Dolinoy, D.; Peterson, K.E.; O’Brien, L.M.; Chervin, R.D.; Cantoral, A.; Tellez-Rojo, M.M.; Solano-Gonzalez, M.; Goodrich, J. Adolescent sleep timing and dietary patterns in relation to DNA methylation of core circadian genes: A pilot study of Mexican youth. Epigenetics 2021, 16, 894–907. [Google Scholar] [CrossRef]
- Rygiel, C.A.; Goodrich, J.M.; Solano-González, M.; Mercado-García, A.; Hu, H.; Téllez-Rojo, M.M.; Peterson, K.E.; Dolinoy, D.C. Prenatal Lead (Pb) Exposure and Peripheral Blood DNA Methylation (5mC) and Hydroxymethylation (5hmC) in Mexican Adolescents from the ELEMENT Birth Cohort. Environ. Health Perspect. 2021, 129, 067002. [Google Scholar] [CrossRef]
- Moran, S.; Arribas, C.; Esteller, M. Validation of a DNA methylation microarray for 850,000 CpG sites of the human genome enriched in enhancer sequences. Epigenomics 2016, 8, 389–399. [Google Scholar] [CrossRef]
- Ait-Aissa, K.; Blaszak, S.C.; Beutner, G.; Tsaih, S.-W.; Morgan, G.; Santos, J.H.; Flister, M.J.; Joyce, D.L.; Camara, A.K.S.; Gutterman, D.D.; et al. Mitochondrial Oxidative Phosphorylation defect in the Heart of Subjects with Coronary Artery Disease. Sci. Rep. 2019, 9, 7623. [Google Scholar] [CrossRef]
- Riehle, C.; Abel, E.D. Insulin Signaling and Heart Failure. Circ. Res. 2016, 118, 1151–1169. [Google Scholar] [CrossRef]
- Samarel, A.M. Focal adhesion signaling in heart failure. Pflug. Arch. 2014, 466, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.; Henley, T.; Rossi, S.; Costello, M.J.; Polacheck, W.; Griffith, B.E.; Bressan, M. Adherens junction engagement regulates functional patterning of the cardiac pacemaker cell lineage. Dev. Cell 2021, 56, 1498–1511.e7. [Google Scholar] [CrossRef] [PubMed]
- Bartnik, M.; Norhammar, A.; Rydén, L. Hyperglycaemia and cardiovascular disease. J. Intern. Med. 2007, 262, 145–156. [Google Scholar] [CrossRef]
- Ghigo, A.; Li, M. Phosphoinositide 3-kinase: Friend and foe in cardiovascular disease. Front. Pharmacol. 2015, 6, 169. [Google Scholar] [CrossRef] [PubMed]
- Couture, L.; Nash, J.A.; Turgeon, J. The ATP-Binding Cassette Transporters and Their Implication in Drug Disposition: A Special Look at the Heart. Pharmacol. Rev. 2006, 58, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Gilda, J.E.; Gomes, A.V. Proteasome dysfunction in cardiomyopathies. J. Physiol. 2017, 595, 4051–4071. [Google Scholar] [CrossRef]
- Bagchi, A.K.; Akolkar, G.; Mandal, S.; Ayyappan, P.; Yang, X.; Singal, P.K. Toll-like receptor 2 dominance over Toll-like receptor 4 in stressful conditions for its detrimental role in the heart. Am. J. Physiol. Circ. Physiol. 2017, 312, H1238–H1247. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Y.; Cao, Z.; Wang, M.; Liu, X.; Gao, T.; Hu, Q.; Yuan, W.; Lin, L. Up-regulated TLR 4 in cardiomyocytes exacerbates heart failure after long-term myocardial infarction. J. Cell. Mol. Med. 2015, 19, 2728–2740. [Google Scholar] [CrossRef]
- Deegan, D.F.; Karbalaei, R.; Madzo, J.; Kulathinal, R.J.; Engel, N. The developmental origins of sex-biased expression in cardiac development. Biol. Sex Differ. 2019, 10, 46. [Google Scholar] [CrossRef]
- Sharma, S.; De Carvalho, D.; Jeong, S.; Jones, P.A.; Liang, G. Nucleosomes Containing Methylated DNA Stabilize DNA Methyltransferases 3A/3B and Ensure Faithful Epigenetic Inheritance. PLoS Genet. 2011, 7, e1001286. [Google Scholar] [CrossRef]
- Nasab, H.; Rajabi, S.; Eghbalian, M.; Malakootian, M.; Hashemi, M.; Mahmoudi-Moghaddam, H. Association of As, Pb, Cr, and Zn urinary heavy metals levels with predictive indicators of cardiovascular disease and obesity in children and adolescents. Chemosphere 2022, 294, 133664. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wang, N.; Nie, X.; Zhao, L.; Li, Q.; Cang, Z.; Chen, C.; Lu, M.; Cheng, J.; Zhai, H.; et al. Lead Exposure Induces Weight Gain in Adult Rats, Accompanied by DNA Hypermethylation. PLoS ONE 2017, 12, e0169958. [Google Scholar] [CrossRef]
- Faulk, C.; Barks, A.; Sánchez, B.N.; Zhang, Z.; Anderson, O.S.; Peterson, K.E.; Dolinoy, D.C. Perinatal Lead (Pb) Exposure Results in Sex-Specific Effects on Food Intake, Fat, Weight, and Insulin Response across the Murine Life-Course. PLoS ONE 2014, 9, e104273. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wen, X.; Faulk, C.; Boehnke, K.; Zhang, H.; Dolinoy, D.C.; Xi, C. Perinatal Lead Exposure Alters Gut Microbiota Composition and Results in Sex-specific Bodyweight Increases in Adult Mice. Toxicol. Sci. 2016, 151, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.D.; Oh, H.; Jo, W.H.; Hoang, N.H.M.; Kim, M.-S. Mixtures modeling identifies heavy metals and pyrethroid insecticide metabolites associated with obesity. Environ. Sci. Pollut. Res. 2022, 29, 20379–20397. [Google Scholar] [CrossRef]
- Scinicariello, F.; Portier, C.J.; Mevissen, M.; Buser, M.C. Blood lead level association with lower body weight in NHANES 1999–2006. Toxicol. Appl. Pharm. 2013, 273, 516–523. [Google Scholar] [CrossRef]
- Zhu, P.; Hawkins, J.; Linthicum, W.H.; Wang, M.; Li, N.; Zhou, N.; Wen, Q.; Timme-Laragy, A.; Song, X.; Sun, Y. Heavy Metal Exposure Leads to Rapid Changes in Cellular Biophysical Properties. ACS Biomater. Sci. Eng. 2020, 6, 1965–1976. [Google Scholar] [CrossRef]
- Yang, L.; Li, X.; Jiang, A.; Li, X.; Chang, W.; Chen, J.; Ye, F. Metformin alleviates lead-induced mitochondrial fragmentation via AMPK/Nrf2 activation in SH-SY5Y cells. Redox Biol. 2020, 36, 101626. [Google Scholar] [CrossRef]
- Yun, S.; Wu, Y.; Niu, R.; Feng, C.; Wang, J. Effects of lead exposure on brain glucose metabolism and insulin signaling pathway in the hippocampus of rats. Toxicol. Lett. 2019, 310, 23–30. [Google Scholar] [CrossRef]
- Chibowska, K.; Baranowska-Bosiacka, I.; Falkowska, A.; Gutowska, I.; Goschorska, M.; Chlubek, D. Effect of Lead (Pb) on Inflammatory Processes in the Brain. Int. J. Mol. Sci. 2016, 17, 2140. [Google Scholar] [CrossRef]
- Leff, T.; Stemmer, P.; Tyrrell, J.; Jog, R. Diabetes and Exposure to Environmental Lead (Pb). Toxics 2018, 6, 54. [Google Scholar] [CrossRef] [PubMed]
- Luna, A.L.; Acosta-Saavedra, L.C.; Martínez, M.; Torres-Avilés, N.; Gómez, R.; Calderón-Aranda, E.S. TLR4 is a target of environmentally relevant concentration of lead. Toxicol. Lett. 2012, 214, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-Y.; Bovet, L.; Maeshima, M.; Martinoia, E.; Lee, Y. The ABC transporter AtPDR8 is a cadmium extrusion pump conferring heavy metal resistance. Plant J. 2007, 50, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Zhaolai, G.; Xinqi, Y.; Linyang, L.; Ming, Z.; Jie, Y.; Hong, T. Changqun Duan Genome-Wide Analysis of the ATP-Binding Cassette (ABC) Transporter Family in Zea mays L. and Its Re-sponse to Heavy Metal Stresses. Int. J. Mol. Sci. 2022, 23, 2109. [Google Scholar]
- Critchley, B.J.; Isalan, M.; Mielcarek, M. Neuro-Cardio Mechanisms in Huntington’s Disease and Other Neurodegenerative Disorders. Front. Physiol. 2018, 9, 559. [Google Scholar] [CrossRef]
- Joers, V.; Emborg, M.E. Modeling and imaging cardiac sympathetic neurodegeneration in Parkinson’s disease. Am. J. Nucl. Med. Mol. Imaging 2014, 4, 125–159. [Google Scholar]
- Yang, M.; Li, C.; Zhang, Y.; Ren, J. Interrelationship between Alzheimer’s disease and cardiac dysfunction: The brain-heart continuum? Acta. Biochim. Biophys. Sin. 2020, 52, 1–8. [Google Scholar] [CrossRef]
- Appleton, A.A.; Jackson, B.P.; Karagas, M.; Marsit, C.J. Prenatal exposure to neurotoxic metals is associated with increased placental glucocorticoid receptor DNA methylation. Epigenetics 2017, 12, 607–615. [Google Scholar] [CrossRef]
- Sen, A.; Heredia, N.; Senut, M.-C.; Hess, M.; Land, S.; Qu, W.; Hollacher, K.; Dereski, M.O.; Ruden, D.M. Early life lead exposure causes gender-specific changes in the DNA methylation profile of DNA extracted from dried blood spots. Epigenomics 2015, 7, 379–393. [Google Scholar] [CrossRef]
- Shah, S.; McRae, A.F.; Marioni, R.E.; Harris, S.E.; Gibson, J.; Henders, A.K.; Redmond, P.; Cox, S.R.; Pattie, A.; Corley, J.; et al. Genetic and environmental exposures constrain epigenetic drift over the human life course. Genome Res. 2014, 24, 1725–1733. [Google Scholar] [CrossRef]
- Kochmanski, J.; Montrose, L.; Goodrich, J.M.; Dolinoy, D.C. Environmental Deflection: The Impact of Toxicant Exposures on the Aging Epigenome. Toxicol. Sci. 2017, 156, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Khetarpal, S.A.; Schjoldager, K.T.; Christoffersen, C.; Raghavan, A.; Edmondson, A.C.; Reutter, H.M.; Ahmed, B.; Ouazzani, R.; Peloso, G.M.; Vitali, C.; et al. Loss of Function of GALNT2 Lowers High-Density Lipoproteins in Humans, Nonhuman Primates, and Rodents. Cell Metab. 2016, 24, 234–245. [Google Scholar] [CrossRef]
- Peng, P.; Wang, L.; Yang, X.; Huang, X.; Ba, Y.; Chen, X.; Guo, J.; Lian, J.; Zhou, J. A Preliminary Study of the Relationship between Promoter Methylation of the ABCG1, GALNT2 and HMGCR Genes and Coronary Heart Disease. PLoS ONE 2014, 9, e102265. [Google Scholar] [CrossRef] [PubMed]
- Asllanaj, E.; Zhang, X.; Ochoa-Rosales, C.; Nano, J.; Bramer, W.M.; Portilla-Fernandez, E.; Braun, K.V.; Gonzalez-Jaramillo, V.; Ahrens, W.; Ikram, A.; et al. Sexually dimorphic DNA-methylation in cardiometabolic health: A systematic review. Maturitas 2020, 135, 6–26. [Google Scholar] [CrossRef] [PubMed]
- Buhari, O.; Dayyab, F.; Igbinoba, O.; Atanda, A.; Medhane, F.; Faillace, R. The association between heavy metal and serum cholesterol levels in the US population: National Health and Nutrition Examination Survey 2009–2012. Hum. Exp. Toxicol. 2019, 39, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Markunas, C.A.; Xu, Z.; Harlid, S.; Wade, P.A.; Lie, R.T.; Taylor, J.A.; Wilcox, A.J. Identification of DNA methylation changes in newborns related to maternal smoking during pregnancy. Environ. Health Perspect. 2014, 122, 1147–1153. [Google Scholar] [CrossRef]
- Everson, T.; Punshon, T.; Jackson, B.P.; Hao, K.; Lambertini, L.; Chen, J.; Karagas, M.R.; Marsit, C.J. Cadmium-Associated Differential Methylation throughout the Placental Genome: Epigenome-Wide Association Study of Two U.S. Birth Cohorts. Environ. Health Perspect. 2018, 126, 017010. [Google Scholar] [CrossRef]
- Weng, D.Y.; Chen, J.; Taslim, C.; Hsu, P.-C.; Marian, C.; David, S.P.; Loffredo, C.A.; Shields, P.G. Persistent alterations of gene expression profiling of human peripheral blood mononuclear cells from smokers. Mol. Carcinog. 2016, 55, 1424–1437. [Google Scholar] [CrossRef] [PubMed]
- Litviňuková, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Worth, C.L.; Lindberg, E.L.; Kanda, M.; Polanski, K.; Heinig, M.; Lee, M.; et al. Cells of the adult human heart. Nature 2020, 588, 466–472. [Google Scholar] [CrossRef]
- Bakulski, K.M.; Dou, J.F.; Thompson, R.C.; Lee, C.; Middleton, L.Y.; Perera, B.P.U.; Ferris, S.P.; Jones, T.R.; Neier, K.; Zhou, X.; et al. Single-Cell Analysis of the Gene Expression Effects of Developmental Lead (Pb) Exposure on the Mouse Hippocampus. Toxicol. Sci. 2020, 176, 396–409. [Google Scholar] [CrossRef]
- Yu, J.; Chen, L.; Gu, W.; Liu, S.; Wu, B. Heterogeneity effects of nanoplastics and lead on zebrafish intestinal cells identified by single-cell sequencing. Chemosphere 2022, 289, 133133. [Google Scholar] [CrossRef] [PubMed]
- Greco, C.M.; Kunderfranco, P.; Rubino, M.; Larcher, V.; Carullo, P.; Anselmo, A.; Kurz, K.; Carell, T.; Angius, A.; Latronico, M.; et al. DNA hydroxymethylation controls cardiomyocyte gene expression in development and hypertrophy. Nat. Commun. 2016, 7, 12418. [Google Scholar] [CrossRef] [PubMed]
- Teslovich, T.M.; Musunuru, K.; Smith, A.V.; Edmondson, A.C.; Stylianou, I.M.; Koseki, M.; Pirruccello, J.P.; Ripatti, S.; Chasman, D.I.; Willer, C.J.; et al. Biological, clinical and population relevance of 95 loci for blood lipids. Nature 2010, 466, 707–713. [Google Scholar] [CrossRef]
- Morrone, A.; Bardelli, T.; Donati, M.A.; Giorgi, M.; Di Rocco, M.; Gatti, R.; Parini, R.; Ricci, R.; Taddeucci, G.; d’Azzo, A.; et al. Beta-galactosidase gene mutations affecting the lysosomal enzyme and the elastin-binding protein in GM1-gangliosidosis patients with cardiac involvement. Hum. Mutat. 2000, 15, 354–366. [Google Scholar] [CrossRef]
- Chai, S.; Wan, X.; Nassal, D.M.; Liu, H.; Moravec, C.S.; Ramirez-Navarro, A.; Deschênes, I. Contribution of two-pore K(+) channels to cardiac ventricular action potential revealed using human iPSC-derived cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H1144–H1153. [Google Scholar] [CrossRef] [PubMed]
- Pandit, L.M.; Lloyd, E.E.; Reynolds, J.O.; Lawrence, W.S.; Reynolds, C.; Wehrens, X.; Bryan, R.M. TWIK-2 Channel Deficiency Leads to Pulmonary Hypertension through a Rho-Kinase–Mediated Process. Hypertension 2014, 64, 1260–1265. [Google Scholar] [CrossRef]
- Liu, Q.; Jiang, C.; Xu, J.; Zhao, M.T.; Van Bortle, K.; Cheng, X.; Wang, G.; Chang, H.Y.; Wu, J.C.; Snyder, M.P. Genome-Wide Temporal Profiling of Transcriptome and Open Chromatin of Early Cardiomyocyte Differen-tiation Derived From hiPSCs and hESCs. Circ. Res. 2017, 121, 376–391. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, S.; Yan, P.; Ren, J.; Song, M.; Li, J.; Lei, J.; Pan, H.; Wang, S.; Ma, X.; et al. A single-cell transcriptomic landscape of primate arterial aging. Nat. Commun. 2020, 11, 2202. [Google Scholar] [CrossRef]
- Gao, C.; Ren, S.; Lee, J.-H.; Qiu, J.; Chapski, D.; Rau, C.D.; Zhou, Y.; Abdellatif, M.; Nakano, A.; Vondriska, T.M.; et al. RBFox1-mediated RNA splicing regulates cardiac hypertrophy and heart failure. J. Clin. Investig. 2016, 126, 195–206. [Google Scholar] [CrossRef]
- Frese, K.S.; Meder, B.; Keller, A.; Just, S.; Haas, J.; Vogel, B.; Fischer, S.; Backes, C.; Matzas, M.; Köhler, D.; et al. RNA splicing regulated by RBFOX1 is essential for cardiac function in zebrafish. J. Cell Sci. 2015, 128, 3030–3040. [Google Scholar] [CrossRef]
- Lacey, C.J.; Doudney, K.; Bridgman, P.G.; George, P.M.; Mulder, R.T.; Zarifeh, J.J.; Kimber, B.; Cadzow, M.J.; Black, M.A.; Merriman, T.R.; et al. Copy number variants implicate cardiac function and development pathways in earthquake-induced stress cardiomyopathy. Sci. Rep. 2018, 8, 7548. [Google Scholar] [CrossRef] [PubMed]
- He, K.Y.; Wang, H.; Cade, B.E.; Nandakumar, P.; Giri, A.; Ware, E.B.; Haessler, J.; Liang, J.; Smith, J.A.; Franceschini, N.; et al. Rare variants in fox-1 homolog A (RBFOX1) are associated with lower blood pressure. PLoS Genet. 2017, 13, e1006678. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.; Yan, L.; Gao, S.; Hu, C.L.; Ge, H.; Davidow, A.; Park, M.; Bravo, C.; Iwatsubo, K.; Ishikawa, Y.; et al. Type 5 adenylyl cyclase increases oxidative stress by transcriptional regulation of manganese superoxide dismutase via the SIRT1/FoxO3a pathway. Circulation 2013, 127, 1692–1701. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Vatner, D.E.; O’Connor, J.P.; Ivessa, A.; Ge, H.; Chen, W.; Hirotani, S.; Ishikawa, Y.; Sadoshima, J.; Vatner, S.F. Type 5 Adenylyl Cyclase Disruption Increases Longevity and Protects Against Stress. Cell 2007, 130, 247–258. [Google Scholar] [CrossRef]
- Esposito, G.; Perrino, C.; Ozaki, T.; Takaoka, H.; Defer, N.; Petretta, M.P.; De Angelis, M.C.; Mao, L.; Hanoune, J.; Rockman, H.A.; et al. Increased myocardial contractility and enhanced exercise function in transgenic mice overexpressing either adenylyl cyclase 5 or 8. Basic Res. Cardiol. 2008, 103, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Ciceri, P.; Elli, F.; Cappelletti, L.; Tosi, D.; Savi, F.; Bulfamante, G.; Cozzolino, M. Osteonectin (SPARC) Expression in Vascular Calcification: In Vitro and Ex Vivo Studies. Calcif. Tissue Res. 2016, 99, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, A.D. The role of secreted protein acidic and rich in cysteine (SPARC) in cardiac repair and fibrosis: Does ex-pression of SPARC by macrophages influence outcomes? J. Mol. Cell. Cardiol. 2016, 93, 156–161. [Google Scholar] [CrossRef]

| Condition | Total | # Hypermethylated (% Total) | # Hypomethylated (% Total) | Total Tested |
|---|---|---|---|---|
| Female Pb | 189 | 74 | 115 | 672,230 |
| Male Pb | 280 | 135 | 145 | 730,731 |
| Condition | Total | # Hypermethylated (% Total) | # Hypomethylated (% Total) | Total Tested |
|---|---|---|---|---|
| Female Pb | 120 | 59 | 61 | 137,035 |
| Male Pb | 99 | 47 | 52 | 134,644 |
| Gene | % Change DNA Meth. (M) | FDR (M) | Genomic Annot. (M) | Chromosomal Position (M) | % Change DNA Meth. (F) | FDR (F) | Genomic Annot. (F) | Chromosomal Position (F) |
|---|---|---|---|---|---|---|---|---|
| Boc | 25.1 | 6.99 × 10−3 | Exon | 44496428 | −16.96 | 0.010 | Exon | 44496335 |
| Casr | 17.14 | 0.014 | Intron | 36530044 | −19.68 | 0.030 | Intron | 36530087 |
| Cpne5 | −25.34 | 0.042 | 1 to 5 kb | 29241524 | −58.46 | 2.26 × 10−3 | 1 to 5 kb | 29241568 |
| Galnt2 | −15.39 | 0.037 | Intron | 122767052 | −15.76 | 6.63 × 10−3 | Intron | 122335932 |
| 27.41 | 0.011 | Intron | 122336116 | −10.77 | 1.39 × 10−3 | Intron | 121128632 | |
| 11.12 | 0.012 | Intron | 121567725 | |||||
| 13.59 | 0.050 | Intron | 122789211 | |||||
| 39.47 | 1.89 × 10−3 | Intron | 122698947 | |||||
| Zfpm1 | 27.41 | 0.011 | Exon | 122336116 | −15.76 | 6.63 × 10−3 | Exon | 122335932 |
| Gene | % Change DNA Methylation (M) | FDR (M) | Genomic Annotation (M) | Chromosomal Start Position (M) | % Change DNA Methylation (F) | FDR (F) | Genomic Annotation (F) | Chromosomal Position (F) |
|---|---|---|---|---|---|---|---|---|
| Galnt2 | −35.94 | 0.001 | Intron | 120754001 | −12.23 | 0.038 | Intron | 123893001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Svoboda, L.K.; Wang, K.; Goodrich, J.M.; Jones, T.R.; Colacino, J.A.; Peterson, K.E.; Tellez-Rojo, M.M.; Sartor, M.A.; Dolinoy, D.C. Perinatal Lead Exposure Promotes Sex-Specific Epigenetic Programming of Disease-Relevant Pathways in Mouse Heart. Toxics 2023, 11, 85. https://doi.org/10.3390/toxics11010085
Svoboda LK, Wang K, Goodrich JM, Jones TR, Colacino JA, Peterson KE, Tellez-Rojo MM, Sartor MA, Dolinoy DC. Perinatal Lead Exposure Promotes Sex-Specific Epigenetic Programming of Disease-Relevant Pathways in Mouse Heart. Toxics. 2023; 11(1):85. https://doi.org/10.3390/toxics11010085
Chicago/Turabian StyleSvoboda, Laurie K., Kai Wang, Jaclyn M. Goodrich, Tamara R. Jones, Justin A. Colacino, Karen E. Peterson, Martha M. Tellez-Rojo, Maureen A. Sartor, and Dana C. Dolinoy. 2023. "Perinatal Lead Exposure Promotes Sex-Specific Epigenetic Programming of Disease-Relevant Pathways in Mouse Heart" Toxics 11, no. 1: 85. https://doi.org/10.3390/toxics11010085
APA StyleSvoboda, L. K., Wang, K., Goodrich, J. M., Jones, T. R., Colacino, J. A., Peterson, K. E., Tellez-Rojo, M. M., Sartor, M. A., & Dolinoy, D. C. (2023). Perinatal Lead Exposure Promotes Sex-Specific Epigenetic Programming of Disease-Relevant Pathways in Mouse Heart. Toxics, 11(1), 85. https://doi.org/10.3390/toxics11010085










