Synthetic Progestins in Waste and Surface Waters: Concentrations, Impacts and Ecological Risk
Abstract
1. Introduction
2. Classification and Properties of the Most Prominent PGs in Aquatic Environments
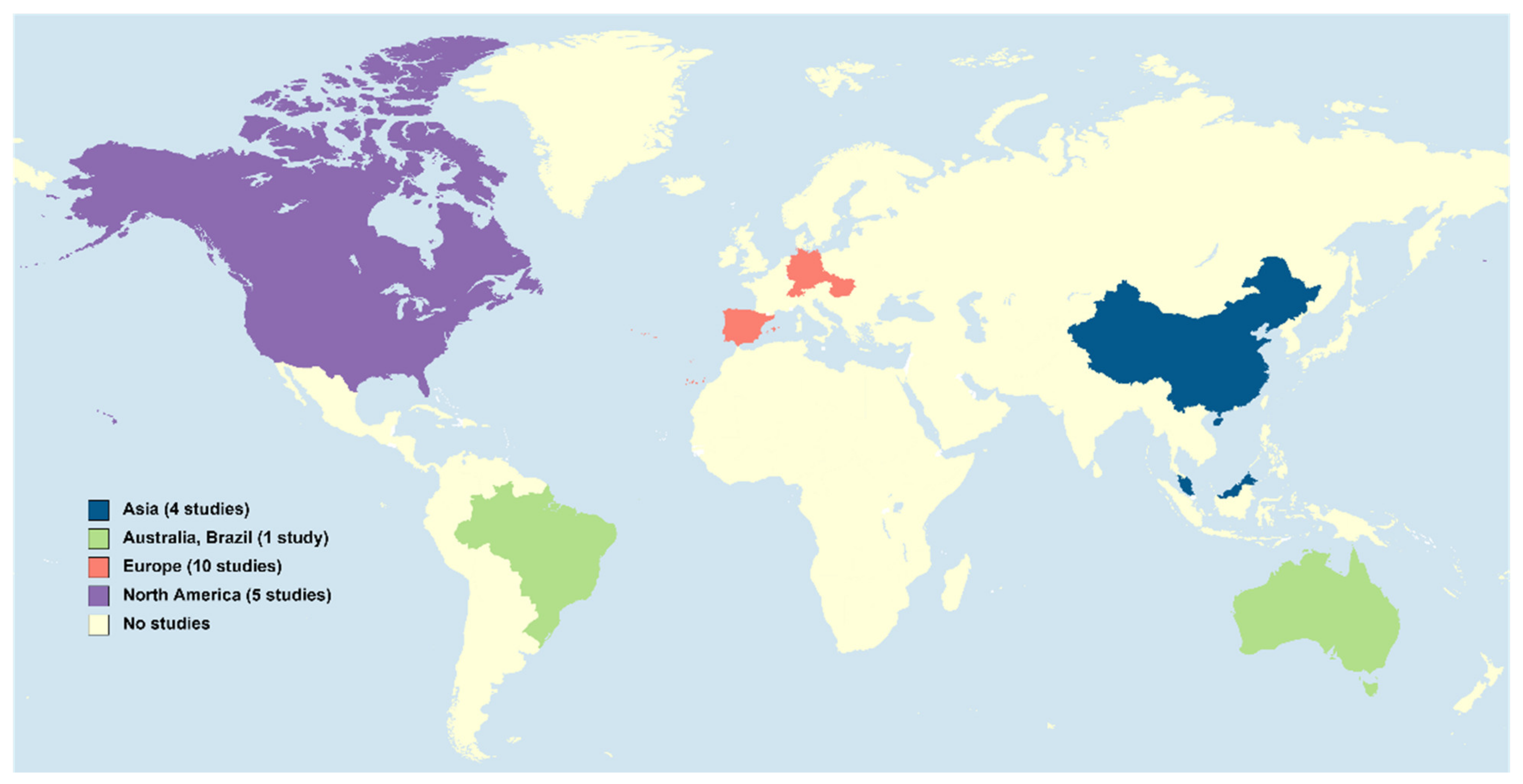
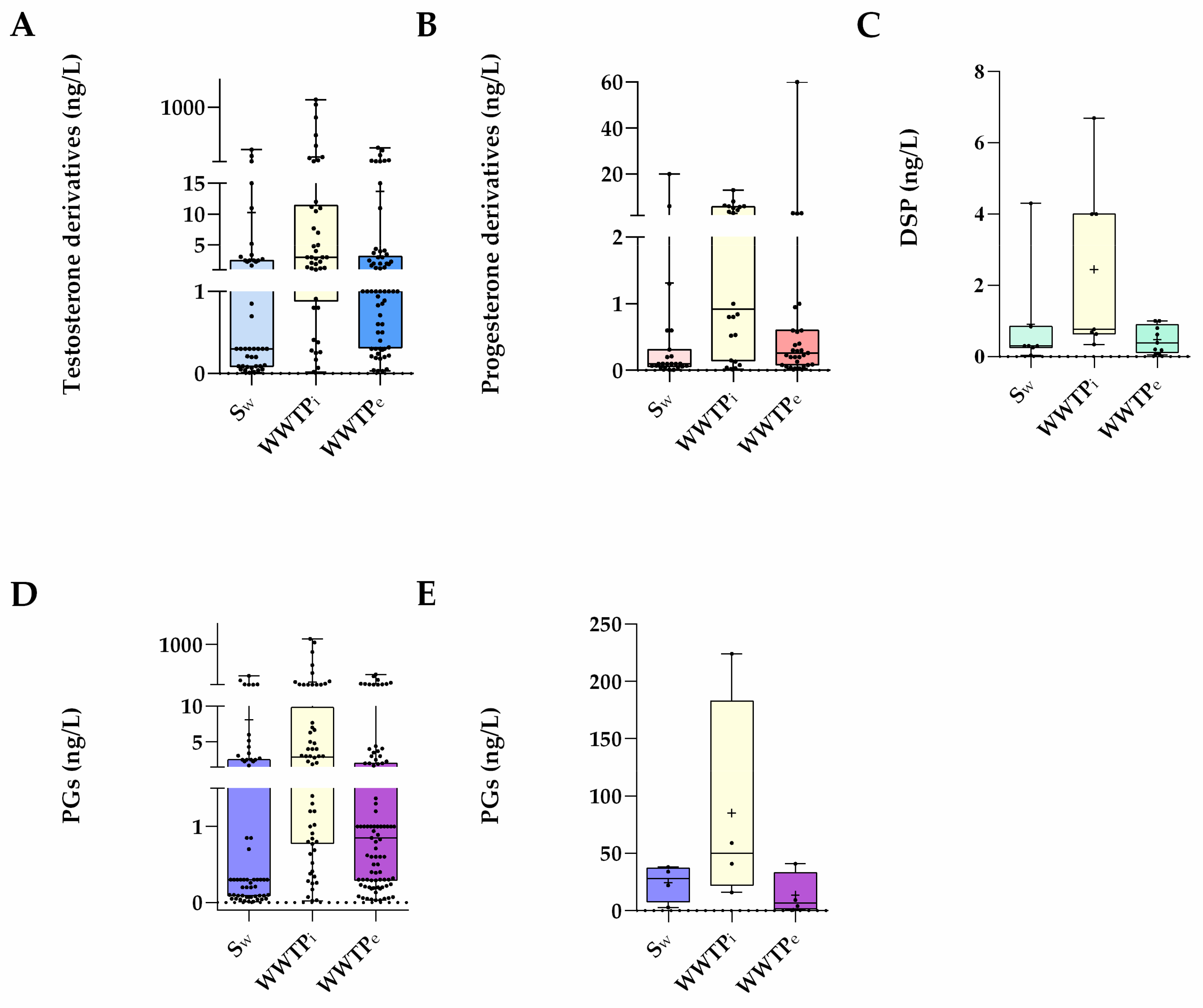
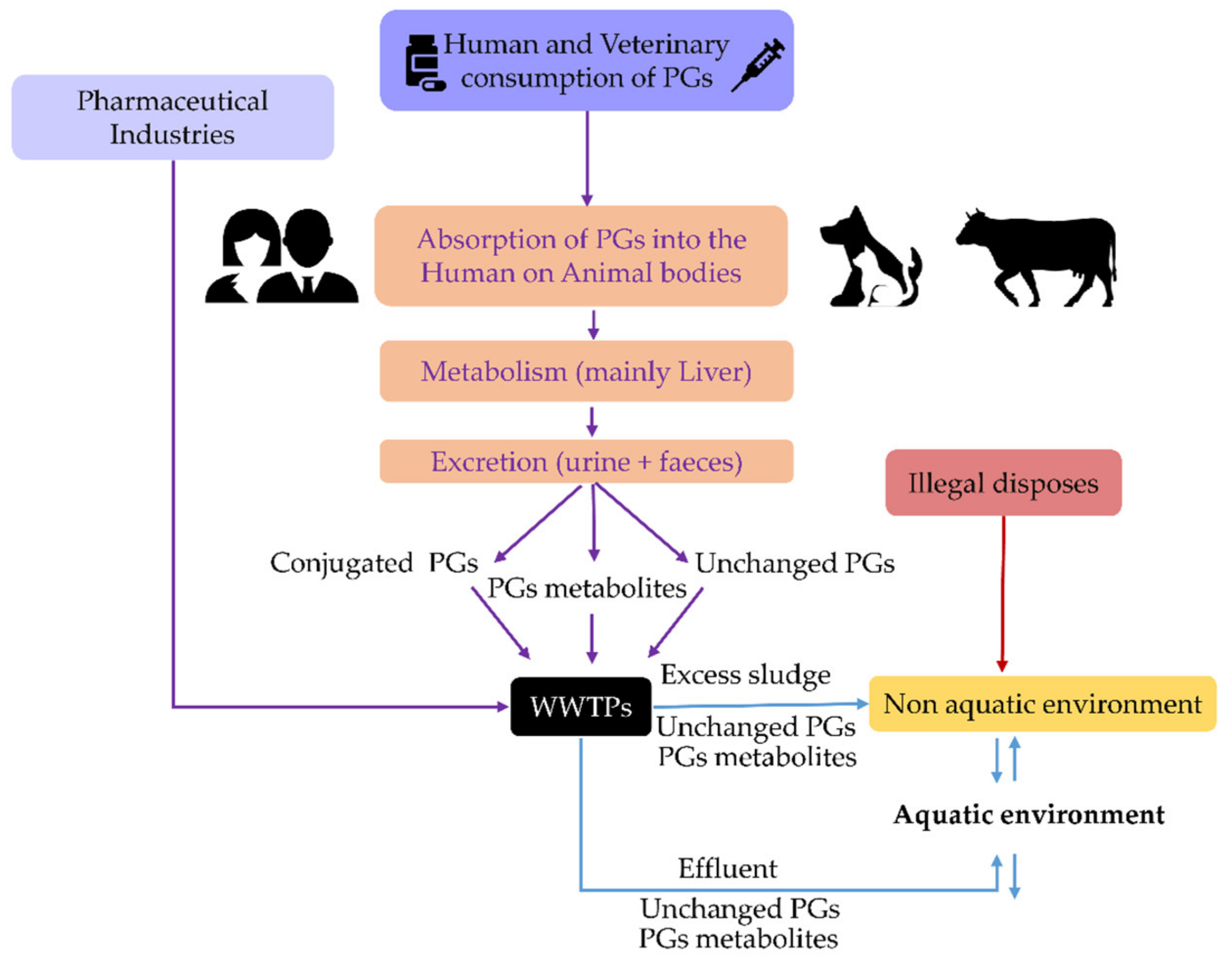
3. Waste and Surface Waters Concentrations of Synthetic Progestins
| Testosterone derivatives (Gonanes) | PGs | QM | Sw (ng/L) | WWTPi (ng/L) | WWTPe (ng/L) | Local (Country) | References |
| GES | (1) | 0.2 | 3 | 1 | Basel and canton Zürich WWTPs (Switzerland). | [31] | |
| (2) | <0.05 | <0.38–7.7 | <0.29–0.71 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.64 | <0.41–7.0 | <0.19–<3.5 | Several WWTPs (Czech and Slovak Republics) | [35] | ||
| (1) | <0.3 | n.e. | <1.0 | Several WWTPs and rivers (Germany). | [33] | ||
| (3) | <0.2 | <3.0 | <1.0 | Jona River and WWTPs (Switzerland). | [36] | ||
| (4) | <21.5 | <21.5 | <21.5 | Five WWTPs (Portugal). | [37] | ||
| LNG | (1) | <2.5–117 | 493–811 | 32–39 | Langat River Basin (Malaysia). | [38] | |
| (5) | <2.5 | n.e. | <2.5 | Southeast Queensland (Australia). | [39] | ||
| (6) | 0.85–3.40 | n.e. | n.e. | Lake Balaton (Hungry). | [40] | ||
| (7) | <15 | n.e. | <15 | Two WWTPs in Quebec (Canada). | [41] | ||
| (2) | <0.08 | <0.26–<2.1 | <0.22–<0.83 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.09 | <0.07–<1.2 | <0.03–<0.32 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | <0.05–<0.7 | n.e. | <0.3–<1.0 | Several WWTPs and rivers (Germany). | [33] | ||
| (1) | ND | ND–38.4 | ND–20.1 | Several WWTPs, Quebec (Canada). | [42] | ||
| (8) | <2.5 | <5–299 ± 17 | <3.0 | Québec and Ontario (Canada). | [43] | ||
| (4) | n.e. | 2.81 | 1.37 | 21 WWTPs (China). | [34] | ||
| (4) | n.e. | n.e. | <1.0 | Several WWTPs effluents (Germany). | [44] | ||
| NET | (4) | n.e. | n.e. | <2.0 | Gran Canaria (Spain) | [45] | |
| (4) | n.e. | 11.2 | 1.92 | 21 WWTPs (China). | [34] | ||
| ENG | (2) | <0.07 | <0.28–<1.4 | <0.21–<0.89 | Blanice River and WWTPs (Czech Republic). | [32] | |
| (2) | <0.09 | <0.25–<1.2 | <0.18–<0.94 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | <0.3 | n.e. | <0.5 | Several WWTPs and rivers (Germany). | [33] | ||
| (4) | n.e. | n.e. | <1.2 | Several WWTPs effluents (Germany). | [44] | ||
| Testosterone derivatives (Estranes) | NTD | (1) | <2.5–230 | 1048–1137 | 218–265 | Langat River Basin (Malaysia). | [38] |
| (4) | n.e. | n.e. | <2.0 | Gran Canaria (Spain). | [45] | ||
| (9) | ND–5.20 | 1.02–94.7 Av. = 25.7 | ND–1.68 Av. = 1.25 | Four WWTPs, Shanghai (China). | [46] | ||
| (5) | <0.21–3.1 | n.e. | n.e. | Freshwater aquaculture (China). | [47] | ||
| (1) | <0.3 | <3 | <0.6 | Basel and canton Zürich WWTPs (Switzerland). | [31] | ||
| (7) | <11 | n.e. | <11 | Two WWTPs in Quebec (Canada). | [41] | ||
| (2) | <0.04 | <0.02–<0.17 | <0.03–0.85 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.01 | <0.02–<0.91 | <0.02–<4.1 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | n.e. | n.e. | <0.40 | Pharmaceutical manufacturing facility discharges (USA). | [48] | ||
| (3) | <0.3 | <3 | <0.6 | Jona River and several WWTPs (Switzerland). | [36] | ||
| (1) | <0.1–<0.3 | n.e. | <1.0 | Several WWTPs and rivers (Germany). | [33] | ||
| (8) | 1.7 ± 0.05–2.7 ± 0.17 | <4.8 | 2 ± 0.2–132 ± 2.2 | Québec and Ontario (Canada). | [43] | ||
| (10) | <2.3 | <2.3 | <2.3 | Basque Country (Spain). | [49] | ||
| (1) | ND | ND–78.8 | ND–31.8 | Several WWTPs, Quebec (Canada). | [42] | ||
| (4) | n.e. | 4.02 | 0.20 | 21 WWTPs (China). | [34] | ||
| (4) | n.e. | n.e. | <1.0 | Several WWTPs effluents (Germany). | [44] | ||
| NTDA | (4) | n.e. | 10.5 | 0.24 | 21 WWTPs (China). | [34] | |
| (1) | <0.3 | n.e. | <0.5 | Several WWTPs and rivers (Germany). | [33] | ||
| (4) | n.e. | n.e. | <1.0 | Several WWTPs (Germany). | [44] | ||
| DIE | (1) | <0.3 | <0.8 | <0.3 | Basel and canton Zürich WWTPs (Switzerland). | [31] | |
| (3) | <0.3 | <0.8 | <0.3 | Jona River and several WWTPs (Switzerland). | [36] | ||
| (2) | <0.09 | 1.9–11.0 | <0.05–1.0 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.04 | 1.3–12 | <0.04–<4.0 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | <0.02–2.3 | n.e. | 1.3–4.4 | Several WWTPs and rivers (Germany). | [33] | ||
| (4) | n.e. | n.e. | 0.3–3.7 | Several WWTPs effluents (Germany). | [44] | ||
Progesterone derivatives | NOMAC | (2) | <0.07 | <0.08–3.6 | <0.03–0.26 | Blanice River and WWTPs (Czech Republic). | [32] |
| MEP | (5) | <0.07–1.3 | n.e. | n.e. | Freshwater aquaculture (China). | [47] | |
| (1) | <0.6 | <6 | <3 | Basel and canton Zürich WWTPs (Switzerland). | [31] | ||
| (3) | <0.6 | <6 | <3 | Jona River and several WWTPs (Switzerland). | [36] | ||
| (2) | <0.06 | <0.02–<0.13 | <0.03–0.23 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (1) | ND | ND–5.7 | ND–2.9 | Several WWTPs, Quebec (Canada). | [42] | ||
| (2) | <0.04 | <0.01–<0.53 | <0.01–0.95 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | <0.05 | n.e. | <0.08 | Several WWTPs and rivers (Germany). | [33] | ||
| MPA | (5) | <0.21–0.31 | n.e. | n.e. | Freshwater aquaculture (China). | [47] | |
| (1) | <0.1 | <0.8 | <0.2 | Basel and canton Zürich WWTPs (Switzerland). | [31] | ||
| (2) | <0.1 | <0.15–4.4 | <0.09–0.58 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.01 | <0.04–8.1 | <0.04–0.38 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (3) | <0.1 | <0.8–5.3 | <0.2 | Jona River and several WWTPs (Switzerland). | [36] | ||
| (1) | <0.05–0.1 | n.e. | <0.08–<0.3 | Several WWTPs and rivers (Germany). | [33] | ||
| (4) | n.e. | 3.09 | 0.23 | 21 WWTPs (China). | [34] | ||
| (4) | n.e. | n.e. | <0.6 | Several WWTPs effluents (Germany). | [44] | ||
| MGA | (4) | n.e. | n.e. | <60 | Gran Canaria (Spain). | [45] | |
| (1) | <0.1 | <1 | <0.6 | Basel and canton Zürich WWTPs (Switzerland). | [31] | ||
| (2) | <0.01 | 0.52–13.0 | 0.13–1.0 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (1) | <0.05–<0.2 | n.e. | <0.06–<0.3 | Several WWTPs and rivers (Germany). | [33] | ||
| (2) | <0.07 | <0.03–<6.3 | <0.06–0.4 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (7) | <6–<20 | n.e. | n.e. | Water bodies in Santa Maria (Brazil). | [50] | ||
| (4) | n.e. | 0.84 | 0.29 | 21 WWTPs (China). | [34] | ||
| Spironolactone derivative | DSP | (6) | 0.26–4.30 | n.e. | n.e. | Lake Balaton (Hungry). | [40] |
| (1) | <0.3 | <4 | <1 | Basel and canton Zürich WWTPs (Switzerland). | [31] | ||
| (2) | <0.85 | 0.64–0.77 | <0.18–<0.62 | Blanice River and WWTPs (Czech Republic). | [32] | ||
| (2) | <0.04 | 0.34–6.7 | <0.07–<0.29 | Several WWTPs (Czech and Slovak Republics). | [35] | ||
| (3) | <0.3 | <4 | <1 | Jona River and several WWTPs (Switzerland). | [36] | ||
| (1) | <0.3 | n.e. | <0.05 | Several WWTPs and rivers (Germany). | [33] | ||
| (4) | n.e. | 0.69 | 0.39 | 21 WWTPs (China). | [34] | ||
| (4) | n.e. | n.e. | <0.8 | Several WWTPs effluents (Germany). | [44] |
4. Biological Effects of PGs in Aquatic Organisms, Particularly Fishes
5. Bioconcentration Factors and Predicted Effect Concentrations of PGs in Fish Plasma
6. Evaluation of Risk Quotients (RQs) for PGs
7. Final Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Wilson, N.J.; Harris, L.M.; Nelson, J.; Shah, S.H. Re-theorizing politics in water governance. Water 2019, 11, 1470. [Google Scholar] [CrossRef]
- Cosgrove, W.J.; Loucks, D.P. Water management: Current and future challenges and research directions. Water Resour. Res. 2015, 51, 4823–4839. [Google Scholar] [CrossRef]
- EU Water Initiative (EUWI). Water Scarcity Management in the Context of WFD. 2006, SCG Agenda Point 8b (WGB/15160506/25d). Available online: https://ec.europa.eu/environment/water/quantity/pdf/comm_droughts/8a_1.pdf (accessed on 27 December 2021).
- EU. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Communities 2000, L327, 0001–0073. [Google Scholar]
- Arnold, K.E.; Brown, A.R.; Ankley, G.T.; Sumpter, J.P. Medicating the environment: Assessing risks of pharmaceuticals to wildlife and ecosystems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130569. [Google Scholar] [CrossRef]
- Kumar, V.; Johnson, A.C.; Trubiroha, A.; Tumová, J.; Ihara, M.; Grabic, R.; Kloas, W.; Tanaka, H.; Kroupová, H.K. The Challenge presented by progestins in ecotoxicological research: A critical review. Environ. Sci. Technol. 2015, 49, 2625–2638. [Google Scholar] [CrossRef]
- Fent, K. Progestins as endocrine disrupters in aquatic ecosystems: Concentrations, effects and risk assessment. Environ. Int. 2015, 84, 115–130. [Google Scholar] [CrossRef]
- Stanczyk, F.Z.; Hapgood, J.P.; Winer, S.; Mishell, D.R., Jr. Progestogens used in postmenopausal hormone therapy: Differences in their pharmacological properties, intracellular actions, and clinical effects. Endocr. Rev. 2013, 34, 171–208. [Google Scholar] [CrossRef]
- Gambrell, R.D., Jr.; Bagnell, C.A.; Greenblatt, R.B. Role of estrogens and progesterone in the etiology and prevention of endometrial cancer: Review. Am. J. Obstet. Gynecol. 1983, 146, 696–707. [Google Scholar] [CrossRef]
- Lobo, R.A. The role of progestins in hormone replacement therapy. Am. J. Obstet. Gynecol. 1992, 166, 1997–2004. [Google Scholar] [CrossRef]
- Chuong, C.J.; Brenner, P.F. Management of abnormal uterine bleeding. Am. J. Obstet. Gynecol. 1996, 175, 787–792. [Google Scholar] [CrossRef]
- Kornek, G.V.; Schenk, T.; Ludwig, H.; Hejna, M.; Scheithauer, W. Placebo-controlled trial of medroxy—Progesterone acetate in gastrointestinal malignancies and cachexia. Onc. Res. Treat. 1996, 19, 164–168. [Google Scholar] [CrossRef]
- EU. The European agency for the evaluation of medicinal products. Eval. Med. Insp. 1999, 33, 969–978. [Google Scholar]
- Apgar, B.S.; Greenberg, G. Using progestins in clinical practice. Am. Fam. Physician 2000, 62, 1839–1846. [Google Scholar]
- Sitruk-Ware, R. New progestagens for contraceptive use. Hum. Reprod. Update 2006, 12, 169–178. [Google Scholar] [CrossRef]
- Edgren, R.A.; Stanczyk, F.Z. Nomenclature of the gonane progestins. Contraception 1999, 60, 313. [Google Scholar] [CrossRef]
- Mathur, R.; Levin, O.; Azziz, R. Use of ethinylestradiol/drospirenone combination in patients with the polycystic ovary syndrome. Ther. Clin. Risk Manag. 2008, 4, 487–492. [Google Scholar] [CrossRef]
- Hohmann, H.; Creinin, M.D. The contraceptive implant. Clin. Obstet. Gynecol. 2007, 50, 907–917. [Google Scholar] [CrossRef]
- Sitruk-Ware, R. Pharmacological profile of progestins. Maturitas 2004, 47, 277–283. [Google Scholar] [CrossRef]
- Katsuki, Y.; Sasagawa, S.; Takano, Y.; Shibutani, Y.; Aoki, D.; Udagawa, Y.; Nozawa, S. Animal studies on the endocrinological profile of dienogest, a novel synthetic steroid. Drugs Exp. Clin. Res. 1997, 23, 45–62. [Google Scholar]
- Bullock, L.P.; Bardin, C.W. Androgenic, synandrogenic, and antiandrogenic actions of progestins. Ann. N. Y. Acad. Sci. 1977, 286, 321–330. [Google Scholar] [CrossRef]
- Seksel, K. Chapter 7—Behavior-modifying drugs. In Small Animal Clinical Pharmacology, 2nd ed.; Maddison, J.E., Page, S.W., Church, D.B., Eds.; W.B. Saunders: Edinburgh, UK, 2008; pp. 126–147. [Google Scholar]
- Moore, N.L.; Hanson, A.R.; Ebrahimie, E.; Hickey, T.E.; Tilley, W.D. Anti-proliferative transcriptional effects of medroxyprogesterone acetate in estrogen receptor positive breast cancer cells are predominantly mediated by the progesterone receptor. J. Steroid Biochem. Mol. Biol. 2020, 199, 105548. [Google Scholar] [CrossRef]
- Thomas, C.P.; Liu, K.Z.; Vats, H.S. Medroxyprogesterone acetate binds the glucocorticoid receptor to stimulate α-ENaC and sgk1 expression in renal collecting duct epithelia. Am. J. Physiol. Renal. Physiol. 2006, 290, F306–F312. [Google Scholar] [CrossRef][Green Version]
- Murphy, S.J.; Littleton-Kearney, M.T.; McCullough, L.D.; Hurn, P.D. Sex, hormones and the endothelium. In Advances in Molecular and Cell Biology; Elsevier: Amsterdam, The Netherlands, 2004; Volume 34, pp. 71–84. [Google Scholar]
- Sitruk-Ware, R. Pharmacology of different progestogens: The special case of drospirenone. Climacteric 2005, 8, 4–12. [Google Scholar] [CrossRef]
- Schindler, A.E.; Campagnoli, C.; Druckmann, R.; Huber, J.; Pasqualini, J.R.; Schweppe, K.W.; Thijssen, J.H.H. Reprint of Classification and pharmacology of progestins. Maturitas 2008, 61, 171–180. [Google Scholar] [CrossRef]
- Fraser, I.S. Non-contraceptive health benefits of intrauterine hormonal systems. Contraception 2010, 82, 396–403. [Google Scholar] [CrossRef]
- Regidor, P.-A. The clinical relevance of progestogens in hormonal contraception: Present status and future developments. Oncotarget 2018, 9, 34628–34638. [Google Scholar] [CrossRef]
- Allen, R.H.; Kaunitz, A.M.; Hickey, M. Chapter 18—Hormonal Contraception. In Williams Textbook of Endocrinology, 13th ed.; Melmed, S., Polonsky, K.S., Larsen, P.R., Kronenberg, H.M., Eds.; Elsevier: Philadelphia, PA, USA, 2016; pp. 664–693. [Google Scholar]
- Zhang, K.; Zhao, Y.; Fent, K. Occurrence and ecotoxicological effects of free, conjugated, and halogenated steroids including 17α-hydroxypregnanolone and pregnanediol in Swiss. Environ. Sci. Technol. 2017, 51, 6498–6506. [Google Scholar] [CrossRef]
- Golovko, O.; Šauer, P.; Fedorova, G.; Kroupová, H.K.; Grabic, R. Determination of progestogens in surface and waste water using SPE extraction and LC-APCI/APPI-HRPS. Sci. Total Environ. 2018, 621, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Weizel, A.; Schlüsener, M.P.; Dierkes, G.; Ternes, T.A. Occurrence of glucocorticoids, mineralocorticoids, and progestogens in various treated wastewater, rivers, and streams. Environ. Sci. Technol. 2018, 52, 5296–5307. [Google Scholar] [CrossRef]
- Yu, Q.; Geng, J.; Zong, X.; Zhang, Y.; Xu, K.; Hu, H.; Deng, Y.; Zhao, F.; Ren, H. Occurrence and removal of progestagens in municipal wastewater treatment plants from different regions in China. Sci. Total Environ. 2019, 668, 1191–1199. [Google Scholar] [CrossRef]
- Šauer, P.; Stará, A.; Golovko, O.; Valentová, O.; Bořík, A.; Grabic, R.; Kroupová, H.K. Two synthetic progestins and natural progesterone are responsible for most of the progestagenic activities in municipal wastewater treatment plant effluents in the Czech and Slovak republics. Water Res. 2018, 137, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Fent, K. Determination of two progestin metabolites (17α-hydroxypregnanolone and pregnanediol) and different classes of steroids (androgens, estrogens, corticosteroids, progestins) in rivers and wastewaters by high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS). Sci. Total Environ. 2018, 610–611, 1164–1172. [Google Scholar] [CrossRef]
- Silva, S.; Cardoso, V.V.; Duarte, L.; Carneiro, R.N.; Almeida, C.M.M. Characterization of five Portuguese wastewater treatment plants: Removal efficiency of pharmaceutical active compounds through conventional treatment processes and environmental risk. Appl. Sci. 2021, 11, 7388. [Google Scholar] [CrossRef]
- Tan, E.S.S.; Ho, Y.B.; Zakaria, M.P.; Latif, P.A.; Saari, N. Simultaneous extraction and determination of pharmaceuticals and personal care products (PPCPs) in river water and sewage by solid-phase extraction and liquid chromatography-tandem mass spectrometry. Int. J. Environ. Anal. Chem. 2015, 95, 816–832. [Google Scholar] [CrossRef]
- King, O.C.; van de Merwe, J.P.; McDonald, J.A.; Leusch, F.D.L. Concentrations of levonorgestrel and ethinylestradiol in wastewater effluents: Is the progestin also cause for concern? Environ. Toxicol. Chem. 2016, 35, 1378–1385. [Google Scholar] [CrossRef]
- Avar, P.; Maasz, G.; Takacs, P.; Lovas, S.; Zrinyi, Z.; Svigruha, R.; Takatsy, A.; Toth, L.G.; Pirger, Z. HPLC-MS/MS analysis of steroid hormones in environmental water samples. Drug Test. Anal. 2016, 8, 123–127. [Google Scholar] [CrossRef]
- Comtois-Marotte, S.; Chappuis, T.; Vo Duy, S.; Gilbert, N.; Lajeunesse, A.; Taktek, S.; Desrosiers, M.; Veilleux, É.; Sauvé, S. Analysis of emerging contaminants in water and solid samples using high resolution mass spectrometry with a Q Exactive orbital ion trap and estrogenic activity with YES-assay. Chemosphere 2017, 166, 400–411. [Google Scholar] [CrossRef]
- Yarahmadi, H.; Duy, S.V.; Hachad, M.; Dorner, S.; Sauvé, S.; Prévost, M. Seasonal variations of steroid hormones released by wastewater treatment plants to river water and sediments: Distribution between particulate and dissolved phases. Sci. Total Environ. 2018, 635, 144–155. [Google Scholar] [CrossRef]
- Goeury, K.; Vo Duy, S.; Munoz, G.; Prévost, M.; Sauvé, S. Analysis of Environmental Protection Agency priority endocrine disruptor hormones and bisphenol A in tap, surface and wastewater by online concentration liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2019, 1591, 87–98. [Google Scholar] [CrossRef]
- Weizel, A.; Schlüsener, M.P.; Dierkes, G.; Wick, A.; Ternes, T.A. Fate and behavior of progestogens in activated sludge treatment: Kinetics and transformation products. Water Res. 2021, 188, 116515. [Google Scholar] [CrossRef]
- Guedes-Alonso, R.; Ciofi, L.; Sosa-Ferrera, Z.; Santana-Rodríguez, J.J.; Bubba, M.d.; Kabir, A.; Furton, K.G. Determination of androgens and progestogens in environmental and biological samples using fabric phase sorptive extraction coupled to ultra-high performance liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2016, 1437, 116–126. [Google Scholar] [CrossRef]
- Xu, G.; Ma, S.; Tang, L.; Sun, R.; Xiang, J.; Xu, B.; Bao, Y.; Wu, M. Occurrence, fate, and risk assessment of selected endocrine disrupting chemicals in wastewater treatment plants and receiving river of Shanghai, China. Environ. Sci. Pollut. Res. Int. 2016, 23, 25442–25450. [Google Scholar] [CrossRef]
- Liu, S.; Xu, X.R.; Qi, Z.H.; Chen, H.; Hao, Q.W.; Hu, Y.X.; Zhao, J.L.; Ying, G.G. Steroid bioaccumulation profiles in typical freshwater aquaculture environments of South China and their human health risks via fish consumption. Environ. Pollut. 2017, 228, 72–81. [Google Scholar] [CrossRef]
- Scott, T.-M.; Phillips, P.J.; Kolpin, D.W.; Colella, K.M.; Furlong, E.T.; Foreman, W.T.; Gray, J.L. Pharmaceutical manufacturing facility discharges can substantially increase the pharmaceutical load to US wastewaters. Sci. Total Environ. 2018, 636, 69–79. [Google Scholar] [CrossRef]
- Miossec, C.; Lanceleur, L.; Monperrus, M. Multi-residue analysis of 44 pharmaceutical compounds in environmental water samples by solid-phase extraction coupled to liquid chromatography-tandem mass spectrometry. J. Sep. Sci. 2019, 42, 1853–1866. [Google Scholar] [CrossRef]
- Pivetta, G.G.; Gastaldini, M.D.C. Presence of emerging contaminants in urban water bodies in southern Brazil. J. Water Health 2019, 17, 329–337. [Google Scholar] [CrossRef]
- Besse, J.-P.; Garric, J. Progestagens for human use, exposure and hazard assessment for the aquatic environment. Environ. Pollut. 2009, 157, 3485–3494. [Google Scholar] [CrossRef]
- Kasambala, H.R.; Rwiza, M.J.; Mdegela, R.H. Levels and distribution of progesterone in receiving waters and wastewaters of a growing urban area. Water Sci. Technol. 2019, 80, 1107–1117. [Google Scholar] [CrossRef]
- Gomes, R.L.; Scrimshaw, M.D.; Lester, J.N. Fate of conjugated natural and synthetic steroid estrogens in crude sewage and activated sludge batch studies. Environ. Sci. Technol. 2009, 43, 3612–3618. [Google Scholar] [CrossRef]
- Blair, B.; Nikolaus, A.; Hedman, C.; Klaper, R.; Grundl, T. Evaluating the degradation, sorption, and negative mass balances of pharmaceuticals and personal care products during wastewater treatment. Chemosphere 2015, 134, 395–401. [Google Scholar] [CrossRef]
- Ojoghoro, J.O.; Scrimshaw, M.D.; Sumpter, J.P. Steroid hormones in the aquatic environment. Sci. Total Environ. 2021, 792, 148306. [Google Scholar] [CrossRef]
- Mills, L.J.; Chichester, C. Review of evidence: Are endocrine-disrupting chemicals in the aquatic environment impacting fish populations? Sci. Total Environ. 2005, 343, 1–34. [Google Scholar] [CrossRef]
- Yu, Q.; Geng, J.; Huo, H.; Xu, K.; Huang, H.; Hu, H.; Ren, H. Bioaugmentated activated sludge degradation of progesterone: Kinetics and mechanism. Chem. Eng. J. 2018, 352, 214–224. [Google Scholar] [CrossRef]
- Ward, S.; Back, D.J. Metabolism of gestodene in human liver cytosol and microsomes in vitro. J. Steroid Biochem. Mol. Biol. 1993, 46, 235–243. [Google Scholar] [CrossRef]
- National Library of Medicine. Available online: https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3594 (accessed on 27 December 2021).
- Kook, K.; Gabelnick, H.; Duncan, G. Pharmacokinetics of levonorgestrel 0.75 mg tablets. Contraception 2002, 66, 73–76. [Google Scholar] [CrossRef]
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB00367 (accessed on 27 December 2021).
- Liu, S.; Ying, G.-G.; Liu, Y.-S.; Peng, F.-Q.; He, L.-Y. Degradation of norgestrel by bacteria from activated sludge: Comparison to progesterone. Environ. Sci. Technol. 2013, 47, 10266–10276. [Google Scholar] [CrossRef] [PubMed]
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB00294 (accessed on 27 December 2021).
- Stanczyk, F.Z.; Roy, S. Metabolism of levonorgestrel, norethindrone, and structurally related contraceptive steroids. Contraception 1990, 42, 67–96. [Google Scholar] [CrossRef]
- Bick, A.J.; Louw-du Toit, R.; Skosana, S.B.; Africander, D.; Hapgood, J.P. Pharmacokinetics, metabolism and serum concentrations of progestins used in contraception. Pharmacol. Therapeut. 2021, 222, 107789. [Google Scholar] [CrossRef]
- Walker, C.J.; Cowan, D.A.; James, V.H.; Lau, J.C.; Kicman, A.T. Doping in sport: 3. Metabolic conversion of oral norethisterone to urinary 19-norandrosterone. Steroids 2009, 74, 341–349. [Google Scholar] [CrossRef]
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB09123 (accessed on 27 December 2021).
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB13981 (accessed on 27 December 2021).
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB00603 (accessed on 27 December 2021).
- National Library of Medicine. Compound Summary for CID 6279, Medroxyprogesterone Acetate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Medroxyprogesterone-acetate (accessed on 27 December 2021).
- National Library of Medicine. Compound Summary for CID 11683, Megestrol Acetate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Megestrol-acetate (accessed on 27 December 2021).
- DrugBank. Available online: https://www.drugbank.ca/drugs/DB01395 (accessed on 27 December 2021).
- Stout, E.P.; La Clair, J.J.; Snell, T.W.; Shearer, T.L.; Kubanek, J. Conservation of progesterone hormone function in invertebrate reproduction. Proc. Natl. Acad. Sci. USA 2010, 107, 11859–11864. [Google Scholar] [CrossRef]
- Ren, J.; Chung-Davidson, Y.-W.; Jia, L.; Li, W. Genomic sequence analyses of classical and non-classical lamprey progesterone receptor genes and the inference of homologous gene evolution in metazoans. BMC Evol. Biol. 2019, 19, 136. [Google Scholar] [CrossRef] [PubMed]
- Ziková, A.; Lorenz, C.; Hoffmann, F.; Kleiner, W.; Lutz, I.; Stöck, M.; Kloas, W. Endocrine disruption by environmental gestagens in amphibians—A short review supported by new in vitro data using gonads of Xenopus laevis. Chemosphere 2017, 181, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, M. Follicular Steroidogenesis in Fish, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; p. 632. [Google Scholar]
- Stacey, N.E.; Sorensen, P.W. Hormonally Derived Sex Pheromones in Fish, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; p. 632. [Google Scholar]
- Pankhurst, N.W. Gonadal Steroids: Functions and Patterns of Change, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; p. 632. [Google Scholar]
- Melcangi, R.C.; Giatti, S.; Calabrese, D.; Pesaresi, M.; Cermenati, G.; Mitro, N.; Viviani, B.; Garcia-Segura, L.M.; Caruso, D. Levels and actions of progesterone and its metabolites in the nervous system during physiological and pathological conditions. Prog. Neurobiol. 2014, 113, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Kime, D.E. A strategy for assessing the effects of xenobiotics on fish reproduction. Sci. Total Environ. 1999, 225, 3–11. [Google Scholar] [CrossRef]
- Nagahama, Y.; Yamashita, M. Regulation of oocyte maturation in fish. Dev. Growth Differ. 2008, 50, S195–S219. [Google Scholar] [CrossRef]
- NIEHS. Endocrine Disruptors. National Institute of Environmental Health Sciences. U.S. Department of Health and Human Sciences. Available online: www.niehs.nih.gov/ (accessed on 27 December 2021).
- EU. (EU) 2018/840 of 5 June 2018 establishing a watch list of substances for Union-wide monitoring in the field of water policy pursuant to Directive 2008/105/E.C. of the European Parliament and of the Council and repealing Commission Implementing Decision (EU) 2015/495. Off. J. Eur. Union 2018, 141, 9–12. [Google Scholar]
- Hoffmann, F.; Kloas, W. The synthetic progestogen, levonorgestrel, but not natural progesterone, affects male mate calling behavior of Xenopus laevis. Gen. Comp. Endocrinol. 2012, 176, 385–390. [Google Scholar] [CrossRef]
- Lorenz, C.; Contardo-Jara, V.; Pflugmacher, S.; Wiegand, C.; Nutzmann, G.; Lutz, I.; Kloas, W. The synthetic gestagen levonorgestrel impairs metamorphosis in Xenopus laevis by disruption of the thyroid system. Toxicol. Sci. 2011, 123, 94–102. [Google Scholar] [CrossRef]
- Contardo-Jara, V.; Lorenz, C.; Pflugmacher, S.; Nützmann, G.; Kloas, W.; Wiegand, C. Molecular effects and bioaccumulation of levonorgestrel in the non-target organism Dreissena polymorpha. Environ. Pollut. 2011, 159, 38–44. [Google Scholar] [CrossRef]
- Orlando, E.F.; Ellestad, L.E. Sources, concentrations, and exposure effects of environmental gestagens on fish and other aquatic wildlife, with an emphasis on reproduction. Gen. Comp. Endocrinol. 2014, 203, 241–249. [Google Scholar] [CrossRef]
- Chaves-Pozo, E.; Garcia-Ayala, A.; Cabas, I. Effects of Sex Steroids on Fish Leukocytes. Biology 2018, 7, 9. [Google Scholar] [CrossRef]
- Frankel, T.E.; Meyer, M.T.; Kolpin, D.W.; Gillis, A.B.; Alvarez, D.A.; Orlando, E.F. Exposure to the contraceptive progestin, gestodene, alters reproductive behavior, arrests egg deposition, and masculinises development in the fathead minnow (Pimephales promelas). Environ. Sci. Technol. 2016, 50, 5991–5999. [Google Scholar] [CrossRef]
- Liang, Y.Q.; Huang, G.Y.; Lin, Z.; Li, J.; Yang, J.W.; Zhong, L.Y.; Ying, G.G. Reproductive effects of synthetic progestin norgestrel in zebrafish (Danio rerio). Chemosphere 2018, 190, 17–24. [Google Scholar] [CrossRef]
- Šauer, P.; Tumová, J.; Steinbach, C.; Golovko, O.; Komen, H.; Maillot-Maréchal, E.; Máchová, J.; Grabic, R.; Aït-Aïssa, S.; Kocour Kroupová, H. Chronic simultaneous exposure of common carp (Cyprinus carpio) from embryonic to juvenile stage to drospirenone and gestodene at low ng/L level caused intersex. Ecotoxicol. Environ. Saf. 2020, 188, 109912. [Google Scholar] [CrossRef]
- Chen, S.; Lin, C.; Tan, J.; Wang, Y.; Wang, X.; Wang, X.; Liu, L.; Li, J.; Hou, L.; Liu, J.; et al. Reproductive potential of mosquitofish is reduced by the masculinising effect of a synthetic progesterone, gestodene: Evidence from morphology, courtship behaviour, ovary histology, sex hormones and gene expressions. Sci. Total Environ. 2021, 769, 144570. [Google Scholar] [CrossRef]
- Hua, J.; Han, J.; Guo, Y.; Zhou, B. The progestin levonorgestrel affects sex differentiation in zebrafish at environmentally relevant concentrations. Aquat. Toxicol. 2015, 166, 1–9. [Google Scholar] [CrossRef]
- Overturf, M.D.; Huggett, D.B. Responses to various exposure durations of levonorgestrel during early-life stages of fathead minnows (Pimephales promelas). Aquat. Toxicol. 2015, 161, 33–40. [Google Scholar] [CrossRef]
- Runnalls, T.J.; Beresford, N.; Kugathas, S.; Margiotta-Casaluci, L.; Scholze, M.; Scott, A.P.; Sumpter, J.P. From single chemicals to mixtures—Reproductive effects of levonorgestrel and ethinylestradiol on the fathead minnow. Aquat. Toxicol. 2015, 169, 152–167. [Google Scholar] [CrossRef]
- Svensson, J.; Mustafa, A.; Fick, J.; Schmitz, M.; Brunström, B. Developmental exposure to progestins causes male bias and precocious puberty in zebrafish (Danio rerio). Aquat. Toxicol. 2016, 177, 316–323. [Google Scholar] [CrossRef]
- Cardoso, P.G.; Rodrigues, D.; Madureira, T.V.; Oliveira, N.; Rocha, M.J.; Rocha, E. Warming modulates the effects of the endocrine disruptor progestin levonorgestrel on the zebrafish fitness, ovary maturation kinetics and reproduction success. Environ. Pollut. 2017, 229, 300–311. [Google Scholar] [CrossRef]
- Frankel, T.E.; Meyer, M.T.; Orlando, E.F. Aqueous exposure to the progestin, levonorgestrel, alters anal fin development and reproductive behavior in the eastern mosquitofish (Gambusia holbrooki). Gen. Comp. Endocrinol. 2016, 234, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Frankel, T.; Yonkos, L.; Frankel, J. Exposure effects of levonorgestrel on oogenesis in the fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2017, 36, 3299–3304. [Google Scholar] [CrossRef] [PubMed]
- Maasz, G.; Zrinyi, Z.; Takacs, P.; Lovas, S.; Fodor, I.; Kiss, T.; Pirger, Z. Complex molecular changes induced by chronic progestogens exposure in roach, Rutilus rutilus. Ecotox. Environ. Saf. 2017, 139, 9–17. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frankel, T.; Yonkos, L.; Ampy, F.; Frankel, J. Exposure to levonorgestrel increases nest acquisition success and decreases sperm motility in the male fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2018, 37, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, P.G.; Rodrigues, D.; Madureira, T.V.; Rocha, M.J.; Rocha, E. Histopathological evaluation of combined impacts of the synthetic progestin levonorgestrel and temperature on the female zebrafish maturation using a semi-quantitative grading analysis—is it enough? B Environ. Contam. Tox. 2018, 101, 417–422. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, K.; Fent, K. Regulation of zebrafish (Danio rerio) locomotor behavior and circadian rhythm network by environmental steroid hormones. Environ. Pollut. 2018, 232, 422–429. [Google Scholar] [CrossRef]
- Cardoso, P.G.; Resende-de-Oliveira, R.; Rocha, E. Combined effects of increased temperature and levonorgestrel exposure on zebrafish female liver, using stereology and immunohistochemistry against catalase, CYP1A, HSP90 and vitellogenin. Environ. Pollut. 2019, 252, 1059–1067. [Google Scholar] [CrossRef]
- De Courten, B.M.; Forbes, J.P.; Roark, H.K.; Burns, N.P.; Major, K.M.; White, J.W.; Li, J.; Mehinto, A.C.; Connon, R.E.; Brander, S.M. Multigenerational and transgenerational effects of environmentally relevant concentrations of endocrine disruptors in an estuarine fish model. Environ. Sci. Technol. 2020, 54, 13849–13860. [Google Scholar] [CrossRef]
- Teigeler, M.; Schaudien, D.; Böhmer, W.; Länge, R.; Schäfers, C. Effects of the gestagen levonorgestrel in a life cycle test with zebrafish (Danio rerio). Environ. Toxicol. Chem. 2021, 41, 580–591. [Google Scholar] [CrossRef]
- Pandelides, Z.; Ussery, E.J.; Overturf, M.D.; Guchardi, J.; Holdway, D.A. Inhibition of swim bladder inflation in Japanese medaka (Oryzias latipes) embryos following exposure to select pharmaceuticals alone and in combination. Aquat. Toxicol. 2021, 234, 105796. [Google Scholar] [CrossRef]
- Liang, Y.Q.; Huang, G.Y.; Zhao, J.L.; Shi, W.J.; Hu, L.X.; Tian, F.; Liu, S.S.; Jiang, Y.X.; Ying, G.G. Transcriptional alterations induced by binary mixtures of ethinylestradiol and norgestrel during the early development of zebrafish (Danio rerio). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2017, 195, 60–67. [Google Scholar] [CrossRef]
- Hou, L.P.; Chen, H.; Tian, C.E.; Liang, Y.; Wu, R.R.; Zhang, X.M.; Fang, X.W.; Zhang, C.P.; Hu, J.J.; Song, L.Y.; et al. Alterations of secondary sex characteristics, reproductive histology and behaviors by norgestrel in the western mosquitofish (Gambusia affinis). Aquat. Toxicol. 2018, 198, 224–230. [Google Scholar] [CrossRef]
- Hou, L.; Chen, S.; Liu, J.; Guo, J.; Chen, Z.; Zhu, Q.; Zhang, W.; Xu, G.; Liang, Y.; Wu, R.; et al. Transcriptomic and physiological changes in western mosquitofish (Gambusia affinis) after exposure to norgestrel. Ecotoxicol. Environ. Saf. 2019, 171, 579–586. [Google Scholar] [CrossRef]
- Steinbach, C.; Cisar, P.; Sauer, P.; Klicnarova, J.; Schmidt-Posthaus, H.; Golovko, O.; Kroupova, H.K. Synthetic progestin etonogestrel negatively affects mating behavior and reproduction in Endler’s guppies (Poecilia wingei). Sci. Total Environ. 2019, 663, 206–215. [Google Scholar] [CrossRef]
- Petersen, L.H.; Hala, D.; Carty, D.; Cantu, M.; Martinović, D.; Huggett, D.B. Effects of progesterone and norethindrone on female fathead minnow (Pimephales promelas) steroidogenesis. Environ. Toxicol. Chem. 2015, 34, 379–390. [Google Scholar] [CrossRef]
- Bridges, K.N.; Magnuson, J.T.; Curran, T.E.; Barker, A.; Roberts, A.P.; Venables, B.J. Alterations to the vision-associated transcriptome of zebrafish (Danio rerio) following developmental norethindrone exposure. Environ. Toxicol. Pharmacol. 2019, 69, 137–142. [Google Scholar] [CrossRef]
- Hou, L.; Chen, S.; Shi, W.; Chen, H.; Liang, Y.; Wang, X.; Tan, J.; Wang, Y.; Deng, X.; Zhan, M.; et al. Norethindrone alters mating behaviors, ovary histology, hormone production and transcriptional expression of steroidogenic genes in zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2020, 195, 110496. [Google Scholar] [CrossRef]
- Liang, Y.Q.; Jing, Z.; Pan, C.G.; Lin, Z.; Zhen, Z.; Hou, L.; Dong, Z. The progestin norethindrone alters growth, reproductive histology and gene expression in zebrafish (Danio rerio). Chemosphere 2020, 242, 125285. [Google Scholar] [CrossRef]
- Liang, Y.Q.; Xu, W.; Liang, X.; Jing, Z.; Pan, C.G.; Tian, F. The synthetic progestin norethindrone causes thyroid endocrine disruption in adult zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 236, 108819. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, H.; Li, D.; Zhan, M.; Hou, L.; Dong, W.; Luo, Y.; Xie, L. The effects of norethindrone on the ontogeny of gene expression along the hypothalamic-pituitary-adrenal and hypothalamic-pituitary-gonadal axes in zebrafish (Danio rerio). Sci. Total Environ. 2020, 747, 141554. [Google Scholar] [CrossRef]
- Vaillant, C.; Gueguen, M.-M.; Feat, J.; Charlier, T.D.; Coumailleau, P.; Kah, O.; Brion, F.; Pellegrini, E. Neurodevelopmental effects of natural and synthetic ligands of estrogen and progesterone receptors in zebrafish eleutheroembryos. Gen. Comp. Endocrinol. 2020, 288, 113345. [Google Scholar] [CrossRef]
- Wang, X.; Tan, Z.; Chen, S.; Gui, L.; Li, X.; Ke, D.; Hou, L.; Leung, J.Y.S. Norethindrone causes cellular and hepatic injury in zebrafish by compromising the metabolic processes associated with antioxidant defence: Insights from metabolomics. Chemosphere 2021, 275, 130049. [Google Scholar] [CrossRef]
- Brown, S.; Fraga, L.R.; Cameron, G.; Erskine, L.; Vargesson, N. The primodos components norethisterone acetate and ethinyl estradiol induce developmental abnormalities in zebrafish embryos. Sci. Rep. 2018, 8, 2917. [Google Scholar] [CrossRef]
- Schmid, S.; Willi, R.A.; Salgueiro-González, N.; Fent, K. Effects of new generation progestins, including as mixtures and in combination with other classes of steroid hormones, on zebrafish early life stages. Sci. Total Environ. 2020, 709, 136262. [Google Scholar] [CrossRef]
- Macikova, P.; Groh, K.J.; Ammann, A.A.; Schirmer, K.; Suter, M.J. Endocrine disrupting compounds affecting corticosteroid signaling pathways in Czech and Swiss waters: Potential impact on fish. Environ. Sci. Technol. 2014, 48, 12902–12911. [Google Scholar] [CrossRef]
- Zhao, Y.; Castiglioni, S.; Fent, K. Synthetic progestins medroxyprogesterone acetate and dydrogesterone and their binary mixtures adversely affect reproduction and lead to histological and transcriptional alterations in zebrafish (Danio rerio). Environ. Sci. Technol. 2015, 49, 4636–4645. [Google Scholar] [CrossRef]
- Shi, W.-J.; Ma, D.-D.; Jiang, Y.-X.; Xie, L.; Zhang, J.-N.; Huang, G.-Y.; Chen, H.-X.; Hou, L.-P.; Liu, Y.-S.; Ying, G.-G. Medroxyprogesterone acetate affects sex differentiation and spermatogenesis in zebrafish. Aquat. Toxicol. 2019, 212, 70–76. [Google Scholar] [CrossRef]
- Shi, W.J.; Huang, G.Y.; Jiang, Y.X.; Ma, D.D.; Chen, H.X.; Huang, M.Z.; Hou, L.P.; Xie, L.; Ying, G.G. Medroxyprogesterone acetate affects eye growth and the transcription of associated genes in zebrafish. Ecotoxicol. Environ. Saf. 2020, 193, 110371. [Google Scholar] [CrossRef]
- Han, J.; Wang, Q.; Wang, X.; Li, Y.; Wen, S.; Liu, S.; Ying, G.; Guo, Y.; Zhou, B. The synthetic progestin megestrol acetate adversely affects zebrafish reproduction. Aquat. Toxicol. 2014, 150, 66–72. [Google Scholar] [CrossRef]
- Hua, J.; Han, J.; Wang, X.; Guo, Y.; Zhou, B. The binary mixtures of megestrol acetate and 17α-ethynylestradiol adversely affect zebrafish reproduction. Environ. Pollut. 2016, 213, 776–784. [Google Scholar] [CrossRef]
- Hua, J.; Han, J.; Guo, Y.; Zhou, B. Endocrine disruption in Chinese rare minnow (Gobiocypris rarus) after long-term exposure to low environmental concentrations of progestin megestrol acetate. Ecotox. Environ. Saf. 2018, 163, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Blanco, M.; Fernandes, D.; Medina, P.; Blázquez, M.; Porte, C. Drospirenone intake alters plasmatic steroid levels and cyp17a1 expression in gonads of juvenile sea bass. Environ. Pollut. 2016, 213, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Rossier, N.M.; Chew, G.; Zhang, K.; Riva, F.; Fent, K. Activity of binary mixtures of drospirenone with progesterone and 17α-ethinylestradiol in vitro and in vivo. Aquat. Toxicol. 2016, 174, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Vračko, M. Chapter 10—Mathematical (Structural) Descriptors in QSAR: Applications in Drug Design and Environmental Toxicology. In Advances in Mathematical Chemistry and Applications; Basak, S.C., Restrepo, G., Villaveces, J.L., Eds.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2015; pp. 222–250. [Google Scholar]
- Kroupova, H.K.; Trubiroha, A.; Lorenz, C.; Contardo-Jara, V.; Lutz, I.; Grabic, R.; Kocour, M.; Kloas, W. The progestin levonorgestrel disrupts gonadotropin expression and sex steroid levels in pubertal roach (Rutilus rutilus). Aquat. Toxicol. 2014, 154, 154–162. [Google Scholar] [CrossRef]
- Nallani, G.C.; Paulos, P.M.; Venables, B.J.; Edziyie, R.E.; Constantine, L.A.; Huggett, D.B. Tissue-specific uptake and bioconcentration of the oral contraceptive norethindrone in two freshwater fishes. Arch. Environ. Con. Tox. 2012, 62, 306–313. [Google Scholar] [CrossRef]
- Gomez, C.F.; Constantine, L.; Huggett, D.B. The influence of gill and liver metabolism on the predicted bioconcentration of three pharmaceuticals in fish. Chemosphere 2010, 81, 1189–1195. [Google Scholar] [CrossRef]
- Steele, W.B.t.; Garcia, S.N.; Huggett, D.B.; Venables, B.J.; Barnes, S.E., 3rd; La Point, T.W. Tissue-specific bioconcentration of the synthetic steroid hormone medroxyprogesterone acetate in the common carp (Cyprinus carpio). Environ. Toxicol. Pharmacol. 2013, 36, 1120–1126. [Google Scholar] [CrossRef]
- Fick, J.; Lindberg, R.H.; Parkkonen, J.; Arvidsson, B.; Tysklind, M.; Larsson, D.G.J. Therapeutic levels of levonorgestrel detected in blood plasma of fish: Results from screening rainbow trout exposed to treated sewage effluents. Environ. Sci. Technol. 2010, 44, 2661–2666. [Google Scholar] [CrossRef]
- Huggett, D.B.; Cook, J.C.; Ericson, J.F.; Williams, R.T. A theoretical model for utilising mammalian pharmacology and safety data to prioritise potential impacts of human pharmaceuticals to fish. Hum. Ecol. Risk Assess. 2003, 9, 1789–1799. [Google Scholar] [CrossRef]
- Fitzsimmons, P.N.; Fernandez, J.D.; Hoffman, A.D.; Butterworth, B.C.; Nichols, J.W. Branchial elimination of superhydrophobic organic compounds by rainbow trout (Oncorhynchus mykiss). Aquat. Toxicol. 2001, 55, 23–34. [Google Scholar] [CrossRef]
- Shi, W.-J.; Zhao, J.-L.; Jiang, Y.-X.; Huang, G.-Y.; Liu, Y.-S.; Zhang, J.-N.; Ying, G.-G. Transcriptional and histological alterations in gonad of adult zebrafish after exposure to the synthetic progestin norgestrel. Environ. Toxicol. Chem. 2017, 36, 3267–3276. [Google Scholar] [CrossRef]
- Chang, H.; Wan, Y.; Wu, S.; Fan, Z.; Hu, J. Occurrence of androgens and progestogens in wastewater treatment plants and receiving river waters: Comparison to estrogens. Water Res. 2011, 42, 732–740. [Google Scholar] [CrossRef]
- Sanderson, H.; Johnson, D.J.; Wilson, C.J.; Brain, R.A.; Solomon, K.R. Probabilistic hazard assessment of environmentally occurring pharmaceuticals toxicity to fish, daphnids and algae by ECOSAR screening. Toxicol. Lett. 2003, 144, 383–395. [Google Scholar] [CrossRef]
- Von der Ohe, P.C.; Dulio, V.; Slobodnik, J.; De Deckere, E.; Kühne, R.; Ebert, R.-U.; Ginebreda, A.; De Cooman, W.; Schüürmann, G.; Brack, W. A new risk assessment approach for the prioritisation of 500 classical and emerging organic microcontaminants as potential river basin specific pollutants under the European Water Framework Directive. Sci. Total Environ. 2011, 409, 2064–2077. [Google Scholar] [CrossRef]
- Leung, H.W.; Minh, T.B.; Murphy, M.B.; Lam, J.C.W.; So, M.K.; Martin, M.; Lam, P.K.S.; Richardson, B.J. Distribution, fate and risk assessment of antibiotics in sewage treatment plants in Hong Kong, South China. Environ. Int. 2012, 42, 1–9. [Google Scholar] [CrossRef]
- ECHA. Guidance on Information Requirements and Chemical Safety Assessment: Characterisation of Dose [Concentration]-Response for Environment; European Chemicals Agency: Helsinki, Finland, 2008; Chapter R.10. [Google Scholar]
- Pickup, J.A.; Dewaele, J.; Furmanski, N.L.; Kowalczyk, A.; Luijkx, G.C.A.; Mathieu, S.; Stelter, N. A risk-based, product-level approach for assuring aquatic environmental safety of cleaning products in the context of sustainability: The Environmental Safety Check (ESC) scheme of the AISE Charter for Sustainable Cleaning. Integr. Environ. Assess. 2017, 13, 127–138. [Google Scholar] [CrossRef]
- Cano-Nicolau, J.; Garoche, C.; Hinfray, N.; Pellegrini, E.; Boujrad, N.; Pakdel, F.; Kah, O.; Brion, F. Several synthetic progestins disrupt the glial cell specific-brain aromatase expression in developing zebrafish. Toxicol. Appl. Pharm. 2016, 305, 12–21. [Google Scholar] [CrossRef]
- Liang, Y.-Q.; Huang, G.-Y.; Liu, S.-S.; Zhao, J.-L.; Yang, Y.-Y.; Chen, X.-W.; Tian, F.; Jiang, Y.-X.; Ying, G.-G. Long-term exposure to environmentally relevant concentrations of progesterone and norgestrel affects sex differentiation in zebrafish (Danio rerio). Aquat. Toxicol. 2015, 160, 172–179. [Google Scholar] [CrossRef]
- Bayer. Short-Term Reproduction Test with Dienogest (ZK 37659) on the Fathead Minnow (Pimephales promelas); Bayer: Leverkusen, Germany, 2009. [Google Scholar]
- FASS. Estradiol & Nomegestrolacetat. Available online: https://www.fass.se/LIF/product?-1.-documentTabPanel-tabs-panel-article~tools~bottom-articletools-printbiglink&userType=2&nplId=20100226000021&docType=78 (accessed on 27 December 2021).
- Zucchi, S.; Castiglioni, S.; Fent, K. Progestins and antiprogestins affect gene expression in early development in zebrafish (Danio rerio) at environmental concentrations. Environ. Sci. Technol. 2012, 46, 5183–5192. [Google Scholar] [CrossRef]

| Testosterone derivatives | Hormone | Family | Common Name (Acronym) & CAS | Structure & Molecular Formula | Generation & Activity |
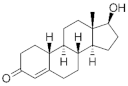 19-Nortestosterone | Gonanes (C17)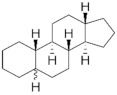 or LNG family | Gestodene (GES) 60282-87-4 |  C21H28O2 | 3rd Generation 1986 (+++) | |
| Levonorgestrel (LNG) 797-63-7 | 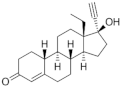 C21H28O2 | 2nd Generation 1966 (+++) | |||
| Norgestrel (NET) 6533-00-2 | 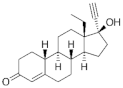 C21H28O2 | 2nd Generation 1966 (+++) | |||
| Etonogestrel (ENG) 54048-10-1 | 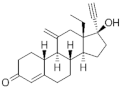 C22H28O2 | 3rd Generation 1998 (+) | |||
Estranes (C18) or NTD family | Norethisterone (NTD) 68-22-4 | 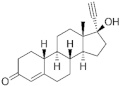 C20H26O2 | 1st Generation 1951 (++) | ||
| Norethisterone acetate (NTDA) 51-98-9 | 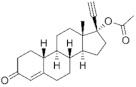 C22H28O3 | 1st Generation 1951 (++) | |||
| Dienogest (DIE) 65928-58-7 | 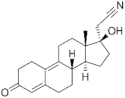 C22H28O2 | 4th Generation 1978 (-) | |||
Progesterone derivatives |  19-Norprogesterone | Norpregnanes (C20) | Nomegestrol acetate (NOMAC) 58652-20-3 | 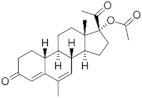 C23H30O4 | 4th Generation 1986 (-) |
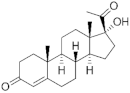 17α-Hydroxyprogesterone | Pregnanes (C21)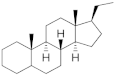 | Medroxyprogesterone (MEP) 520-85-4 |  C22H32O3 | 1st Generation 1957 (+) | |
| Medroxyprogesterone acetate (MPA) 71-58-9 | 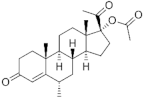 C24H34O4 | 1st Generation 1957 (+) | |||
| Megestrol acetate (MGA) 595-33-5 |  C22H30O4 | 1st Generation 1963 (-) | |||
| Spironolactone derivative | 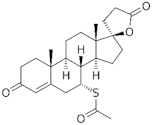 Spironolactone | Drospirenone (DSP) 67392-87-4 | 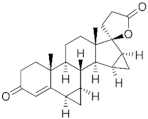 C24H30O3 | 4th Generation 1976 (-) | |
| Testosterone derivatives (Gonanes) | PGs | QM | WWTPi (ng/L) | WWTPe (ng/L) | % R | % RAv. | Local | References |
| GES | (1) | <3.0 | <1.0 | 66.7 | 82.9 | WWTPs (Switzerland). | [31] | |
| (2) | 6.6 | 0.5 | 92.8 | WWTPs (Czech Republic). | [32] | |||
| (2) | 4.3 | 0.77 | 95.5 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (3) | 3.0 | 1.0 | 66.7 | WWTPs (Switzerland). | [36] | |||
| (2) | <0.38 | <0.49 | −28.9 | −186 | WWTPs (Czech Republic). | [32] | ||
| (2) | <0.79 | <3.5 | −343 | WWTPs (Czech and Slovak Republics). | [35] | |||
| LNG | (1) | 652 | 35.5 | 94.4 | 62.4 | WWTPs (Malaysia). | [38] | |
| (2) | <1.8 | 0.5 | 72.4 | WWTPs (Czech Republic). | [32] | |||
| (2) | 0.43 | 0.20 | 54.3 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (1) | 58.6 | 26.0 | 53.7 | WWTPs (Canada). | [42] | |||
| (4) | Data from bibliography | 37.0 | WWTPs (China). | [34] | ||||
| (2) | <0.26 | <0.53 | −103.8 | −104 | WWTPs (Czech Republic). | [32] | ||
| NET | (4) | Data from bibliography | 96.0 | 96.0 | WWTPs (China). | [34] | ||
| ENG | (2) | 1.3 | 0.6 | 58.7 | 42.9 | WWTPs (Czech Republic). | [32] | |
| (2) | 0.52 | 0.38 | 27.1 | WWTPs (Czech and Slovak Republics). | [35] | |||
| NTD | (1) | 1093 | 242 | 78.0 | 75.7 | WWTPs (Malaysia). | [38] | |
| (6) | Data from bibliography | 98 | WWTPs (China). | [46] | ||||
| (1) | <3.0 | <0.6 | 80.0 | WWTPs (Switzerland). | [31] | |||
| (2) | 0.14 | 0.08 | 39 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (3) | <3.0 | <0.6 | 80.0 | WWTPs (Switzerland). | [36] | |||
| (1) | 4.8 | 2.0 | 58.3 | WWTPs (Canada). | [42] | |||
| (5) | 78.8 | 31.0 | 59.6 | WWTPs (Canada). | [43] | |||
| (4) | Data from bibliography | > 90 | WWTPs (China). | [34] | ||||
| (2) | 0.1 | 0.4 | −225 | −600 | WWTPs (Czech Republic). | [32] | ||
| (2) | 0.2 | 2.15 | −975 | WWTPs (Czech and Slovak Republics). | [35] | |||
| NTDA | (4) | Data from bibliography | >90.0 | >90.0 | WWTPs (China). | [34] | ||
| DIE | (1) | <0.8 | <0.3 | 62.5 | 83.0 | WWTPs (Switzerland). | [31] | |
| (3) | <0.8 | <0.3 | 62.5 | WWTPs (Switzerland). | [36] | |||
| (2) | 6.5 | 0.1 | 95.9 | WWTPs (Czech Republic). | [32] | |||
| (2) | 6.4 | 0.30 | 95.3 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (2) | 3.9 | 4 | −2.6 | −2.6 | WWTPs (Czech and Slovak Republics). | [35] | ||
| Progesterone derivatives | NOMAC | (2) | 1.3 | 0.1 | 72.0 | 72.0 | WWTPs (Czech Republic). | [32] |
| MEP | (1) | 6 | 3 | 50.0 | 52.6 | WWTPs (Switzerland). | [31] | |
| (3) | 6 | 3 | 50.0 | WWTPs (Switzerland). | [36] | |||
| (2) | 0.1 | 0.04 | 46.8 | WWTPs (Czech Republic). | [32] | |||
| (2) | 0.2 | 0.05 | 72.9 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (2) | <0.02 | 0.23 | −1050 | −725 | WWTPs (Czech Republic). | [32] | ||
| (2) | 0.19 | 0.95 | −400 | WWTPs (Czech and Slovak Republics). | [35] | |||
| MPA | (1) | Data from bibliography | 93.0 | 76.4 | WWTPs (Switzerland). | [31] | ||
| (2) | 2.4 | 0.3 | 71.0 | WWTPs (Czech Republic). | [32] | |||
| (2) | 2.2 | 0.22 | 90.1 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (3) | 3.1 | 0.2 | 85.6 | WWTPs (Switzerland). | [36] | |||
| (4) | Data from bibliography | 24.0 | WWTPs (China). | [34] | ||||
| MGA | (1) | Data from bibliography | 99.6 | 78.2 | WWTPs (Switzerland). | [31] | ||
| (2) | 6.4 | 0.3 | 95.3 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (3) | <0.03 | 0.4 | 93.7 | WWTPs (Czech Republic). | [32] | |||
| (4) | Data from bibliography | 24.0 | WWTPs (China). | [34] | ||||
| Spironolactone derivative | DSP | (1) | <4.0 | <1.0 | 75.0 | 61.5 | WWTPs (Switzerland). | [31] |
| (2) | 0.7 | 0.4 | 49.0 | WWTPs (Czech Republic). | [32] | |||
| (2) | 3.5 | 0.1 | 88.2 | WWTPs (Czech and Slovak Republics). | [35] | |||
| (3) | <4.0 | <1.0 | 75.0 | WWTPs (Switzerland). | [36] | |||
| (4) | Data from bibliography | 42.0 | WWTPs (China). | [34] | ||||
| Testosterone derivatives (Gonanes) | PGs | Main Metabolization(s) Route(s) | Elimination Route(s) | Active Metabolites | References |
| GES | Liver. Metabolisation occurs by CYP3A4 via partial or total reduction of the A-ring. | Urine and faeces at a ratio of about 6:4. | n.a. | [58,59] | |
| LNG | Liver. Metabolisation by CYP3A4 and CYP3A5. | Urine (45%). Faeces (32%). | In sludge, LNG metabolites generate four active molecules. | [60,61] | |
| NET | Liver. NET is converted to LNG. Then, it follows the same metabolisation paths of LNG. | Urine (46%). Faeces (32%). | In sludge, NET metabolites generate four active molecules. | [61,62] | |
| ENG | Liver Metabolization by CYP3A4. | n.a. | n.a. | [63] | |
| Testosterone derivatives Estranes) | NTD | Liver. Metabolisation occurs via partial or total reduction of the A-ring and oxidation by CYP3A4 and, to a much lesser extent, CYP2C19, CYP1A2, and CYP2A6. | Urine (50%). Faeces (20–40%). | The most known and active biologic metabolite is EE2. | [44,64,65,66] |
| NTDA | Liver. NTDA is converted to NTD. Then, it follows the same metabolisation paths of NTD. | Urine (50%). Faeces (20–40%). | The most known and active biologic metabolite is EE2. | [44,64,65,66] | |
| DIE | Liver. Metabolisation by P450 enzymes. | Urine and faeces at a ratio of about 3:1. | The metabolites are all inactive. | [67] | |
| Progesterone derivatives | NOMAC | n.a. | n.a. | n.a. | [68] |
| MEP | Liver. Metabolisation occurs via partial or total reduction of the A-ring. May happen side-chain reduction, loss of the acetyl group, hydroxylation in the 2-, 6-, and 21-positions or a combination of these positions. | Urine. | More than ten active metabolites. | [69] | |
| MPA | Liver. MPA is converted to MEP. Then, it follows the same metabolisation paths of MEP. | Urine. | More than ten active metabolites. | [44,69,70] | |
| MGA | Liver. | Urine. Respiratory excretion. Fat storage. | n.a. | [71] | |
| Spironolactone derivative | DSP | Liver. Metabolisation occurs by the opening of the lactone ring, known as M11, followed by the action of CYP3A4. | Urine (38–47%). Faeces (17–20%). | n.a. | [72] |
| Testosterone derivatives (Gonanes) | PGs | Structural or Functional Impact on Fish | References |
| GES | Induction of masculinisation in fathead minnow (Pimephales promelas). | [89] | |
| Reproductive disorders in zebrafish (Danio rerio). | [90] | ||
| Induction of intersex in common carp (Cyprinus carpio). | [91] | ||
| Masculinisation, potential reproduction reduction in mosquitofish (Gambusia affinis). | [92] | ||
| LNG | Interfere with sex differentiation in zebrafish (D. rerio). | [93] | |
| Decrease of larval growth and expression of 20β-HSD and CYP19A1, FSH and 3β-HSD in fathead minnows (P. promelas). | [94] | ||
| Inhibition of egg production in fathead minnows (P. promelas). | [95] | ||
| Induction of precocious puberty in zebrafish (D. rerio). | [96] | ||
| Alteration of fitness, ovary maturation kinetics and reproduction success in zebrafish (D. rerio). | [97] | ||
| Change of the anal fin development and reproductive behaviour in mosquitofish (Gambusia holbrooki). | [98] | ||
| Modification of oogenesis in fathead minnow (P. promelas). | [99] | ||
| Induction of metabolic disorders in roach (Rutilus rutilus). | [100] | ||
| Rise of nest acquisition success and loss of sperm motility in fathead minnow (P. promelas). | [101] | ||
| Decrease of mature oocytes in zebrafish (D. rerio). | [97,102] | ||
| Alteration of circadian gene regulation in zebrafish (D. rerio). | [103] | ||
| Alteration of liver function in zebrafish (D. rerio). | [104] | ||
| Transgenerational effects in inland silverside (Menidia beryllina). | [105] | ||
| Decrease of post-hatch survival in zebrafish (D. rerio). | [106] | ||
| Inhibition of swim bladder inflation in Japanese medaka (Oryzias latipes) embryos. | [107] | ||
| NET | Transcriptional alterations in early development in zebrafish (D. rerio). | [108] | |
| Alteration of secondary sex characteristics, reproductive histology, and behaviours in mosquitofish (G. affinis). | [109] | ||
| Transcriptomic and physiological changes in adult mosquitofish (G. affinis). | [110] | ||
| ENG | Change of mating behaviour and reproduction in Endler’s guppies (Poecilia wingei). | [111] | |
| Testosterone derivatives (Estranes) | NTD | Alteration of steroidogenesis in female fathead minnow (P. promelas). | [112] |
| Alteration of sex differentiation in zebrafish (D. rerio). | [93] | ||
| Alteration of circadian gene regulation in zebrafish (D. rerio). | [103] | ||
| Alter the development of visual function in zebrafish (D. rerio). | [113] | ||
| Induction of masculinisation and hepatopathological disorders in female mosquitofish (G. affinis). | [110] | ||
| Alteration of mating behaviours, ovary histology and hormone production in zebrafish (D. rerio). | [114] | ||
| Alters growth, reproductive histology, and gene expression in zebrafish (D. rerio). | [115] | ||
| Thyroid endocrine disruption in zebrafish (D. rerio). | [116] | ||
| Interfere with the HPG and the hypothalamic-pituitary-adrenal (HPA) axis in zebrafish (D. rerio). | [117] | ||
| Neurodevelopmental effects in zebrafish (D. rerio). | [118] | ||
| Hepatic injury in zebrafish (D. rerio). | [119] | ||
| NTDA | Induction of developmental abnormalities in zebrafish (D. rerio). | [120] | |
| DIE | Minor transcriptional alterations in zebrafish (D. rerio) early life stages. | [121] | |
| Progesterone derivatives | NOMAC | n.a. | n.a. |
| MEP | Potential endocrine disruptor in fish. | [122] | |
| MPA | Reproductive disorders (gonadal histology) in zebrafish (D. rerio). | [123] | |
| Affects sex differentiation and spermatogenesis in zebrafish (D. rerio). | [124] | ||
| Affects eye growth in zebrafish (D. rerio). | [125] | ||
| MGA | Reproductive disorders of zebrafish (D. rerio). | [126] | |
| Alters ovary histology of zebrafish (D. rerio). | [127] | ||
| Endocrine disruption in Chinese rare minnow (Gobiocypris rarus). | [128] | ||
| Spironolactone derivative | DSP | Alter plasma steroid levels and CYP17A1 expression in gonads of juvenile sea bass (Dicentrarchus labrax). | [129] |
| Ethinylestradiol antagonist in zebrafish (D. rerio) embryos. | [130] | ||
| Metabolic disorders in roach (R. rutilus). | [100] | ||
| Together with GES induces intersex of common carp (C. carpio). | [91] |
| PGs | Log Kow | BCFFP | CFP (ng/mL) | PECw (ng/L) | Sw (ng/L) | WWTPi (ng/L) | WWTPe (ng/L) |
|---|---|---|---|---|---|---|---|
| aGES | 3.26 | 32 | 1.0 | 31 | 10.8 | 10.9 | 10.8 |
| aLNG | 3.48 | 46 | 2.4 | 52 | 58.5 | 405 | 19.5 |
| bNET | 3.48 | 46 | - | 6.7 | - | 11.2 | 2.0 |
| aENG | 3.16 | 27 | 0.8 | 29 | 0.2 | 0.8 | 0.7 |
| aNTD | 2.97 | 19 | 9.8 | 516 | 115 | 568 | 132 |
| aNTDA | 3.99 | 108 | 9.8 | 91 | 0.3 | 10.5 | 0.62 |
| aDIE | 2.34 | 7 | 85.2 | 12,171 | 1.2 | 6.4 | 2.2 |
| aNOMAC | 3.55 | 52 | 7.2 | 138 | 0.1 | 1.8 | 0.1 |
| aMEP | 3.50 | 47 | 1 | 21 | 0.7 | 3.0 | 1.5 |
| aMPA | 4.09 | 128 | 1 | 8 | 0.2 | 4.1 | 0.3 |
| cMGA | 3.20 | 29 | - | - | 10.0 | 6.5 | 30.0 |
| aDSP | 4.02 | 113 | 30.8 | 273 | 2.2 | 3.5 | 0.5 |
| Available Data | Assessment Factor (AF) |
|---|---|
| One short-term E(L)C50 from each of the three trophic levels (fish, crustaceans, or algae). | 1000 |
| One long-term NOEC assay (either fish, crustaceans, or algae). | 100 |
| Two long-term NOEC assays considering species from two trophic levels (fish and/or crustaceans and/or algae). | 50 |
| Three long-term NOEC assays considering species from three trophic levels (fish, crustaceans and algae). | 10 |
| Species Sensitivity Distribution (SSD) method | 5–1 |
| Field data or model ecosystems. | Evaluated on a case-by-case basis. |
| PGs | Endpoint Value (ng/L) Fish | PNEC (ng/L) | MEC (ng/L) | RQs | Risk | References |
|---|---|---|---|---|---|---|
| GES | EC50 = 10; AF = 1000 | 0.01 | 10.8 | 1078 | High | [89] |
| LNG | NOEC = 0.42; AF = 50 | 0.01 | 59 | 6967 | High | [43,95] |
| NET | LOEC = 6.0; AF = 1000 | 0.01 | - | - | [90] | |
| ENG | EC50 = 12,654; AF = 1000 | 12.7 | 0.2 | 0 | Low | [146] |
| NTD | NOEC = 4; AF = 50 | 0.08 | 115 | 1438 | High | [43,147] |
| NTDA | NOEC = 816; AF = 1000 | 0.8 | 0.3 | 0 | Low | [43,147] |
| DIE | NOEC = 44; AF = 1000 | 0.04 | 1.2 | 36 | High | [148] |
| NOMAC | NOEC = 1300; AF = 10 | 130 | 0.1 | 0 | Low | [149] |
| MEP | - | - | 0.7 | - | - | - |
| MPA | NOEC = 342; AF = 50 | 6.8 | 0.2 | 0 | Low | [43,123] |
| MGA | NOEC = 33; AF = 50 | 0.7 | 10.0 | 15 | High | [43,126] |
| DSP | NOEC = 100; AF = 50 | 2.0 | 2.2 | 1.1 | High | [43,150] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocha, M.J.; Rocha, E. Synthetic Progestins in Waste and Surface Waters: Concentrations, Impacts and Ecological Risk. Toxics 2022, 10, 163. https://doi.org/10.3390/toxics10040163
Rocha MJ, Rocha E. Synthetic Progestins in Waste and Surface Waters: Concentrations, Impacts and Ecological Risk. Toxics. 2022; 10(4):163. https://doi.org/10.3390/toxics10040163
Chicago/Turabian StyleRocha, Maria João, and Eduardo Rocha. 2022. "Synthetic Progestins in Waste and Surface Waters: Concentrations, Impacts and Ecological Risk" Toxics 10, no. 4: 163. https://doi.org/10.3390/toxics10040163
APA StyleRocha, M. J., & Rocha, E. (2022). Synthetic Progestins in Waste and Surface Waters: Concentrations, Impacts and Ecological Risk. Toxics, 10(4), 163. https://doi.org/10.3390/toxics10040163