Effect of Different Reducing Agents on Aromatic Compounds, Antioxidant and Chromatic Properties of Sauvignon Blanc Wine
Abstract
1. Introduction
2. Materials and Methods
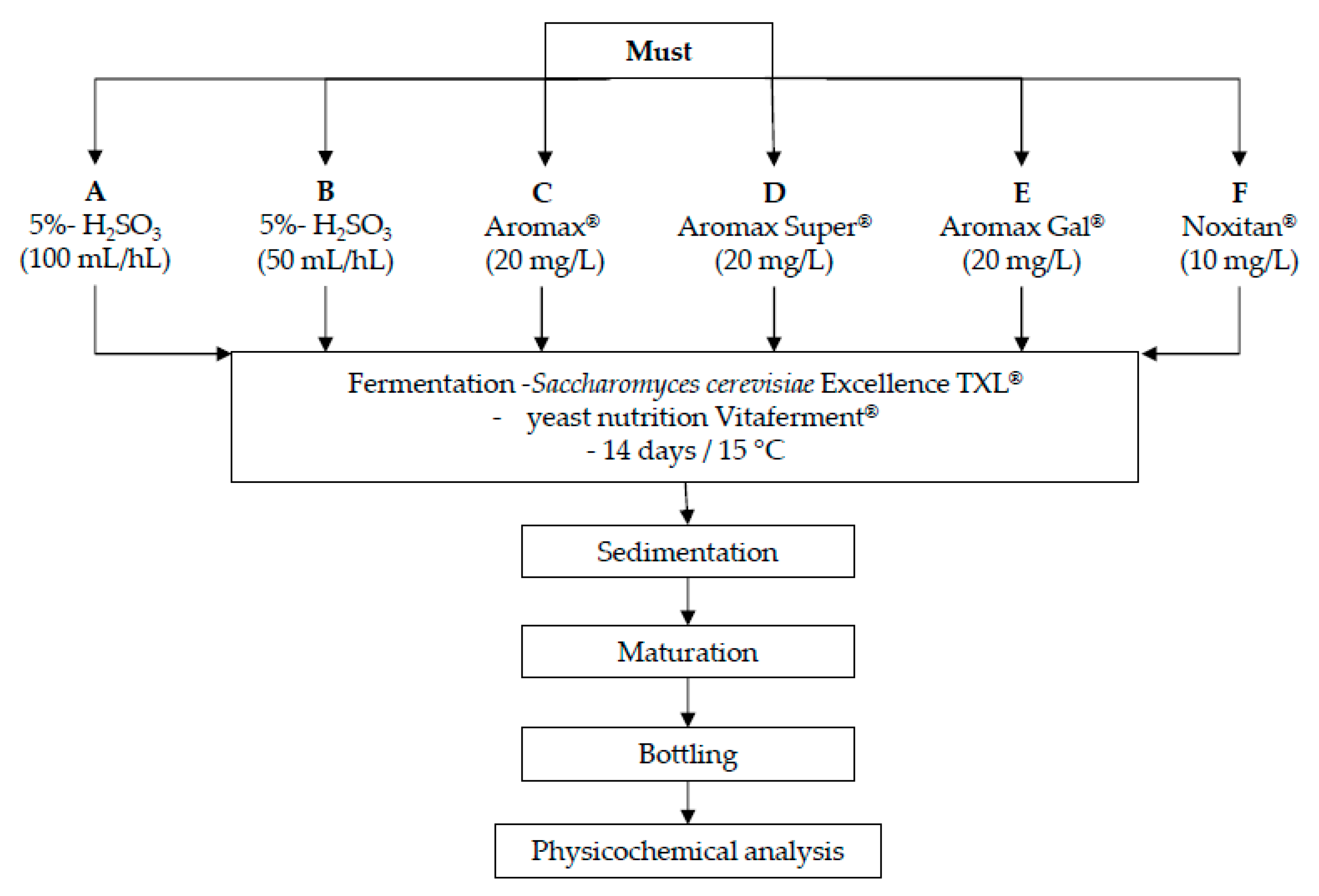
2.1. Wine Production
2.2. Must and Wine Analyses
2.2.1. Physicochemical Analysis
2.2.2. Free α-Amino Nitrogen
2.2.3. Total Phenols
2.2.4. Total Antioxidant Activity
2.2.5. Color Parameters
2.2.6. Volatile Compounds Analysis
2.2.7. Odor Activity Values and Relative Odor Contributions
2.3. Statistical Analysis
3. Results and Discussion
3.1. Must Composition
3.2. Physicochemical Properties, Total Phenols and Antioxidant Potential of Wines
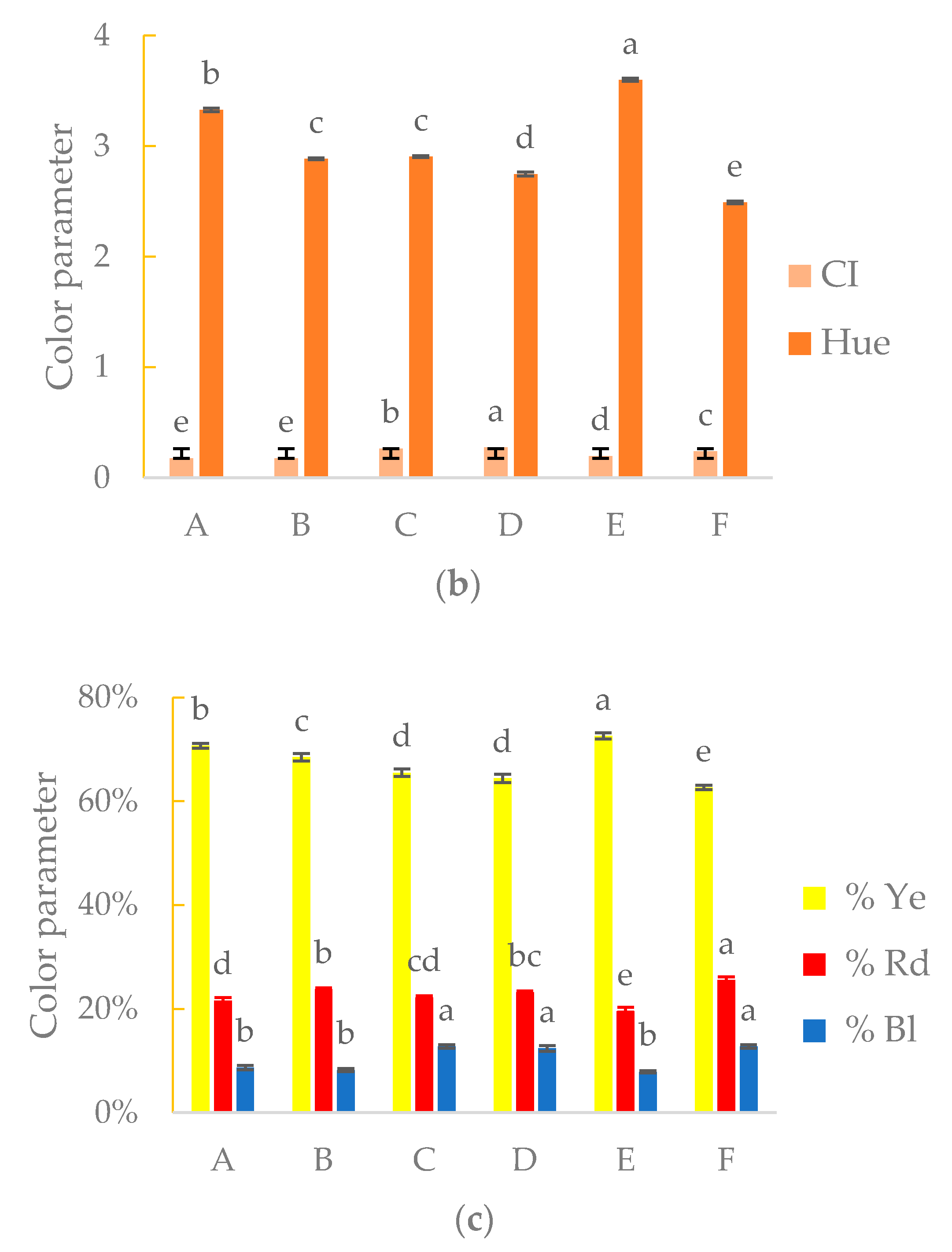
3.3. Chromatic Properties
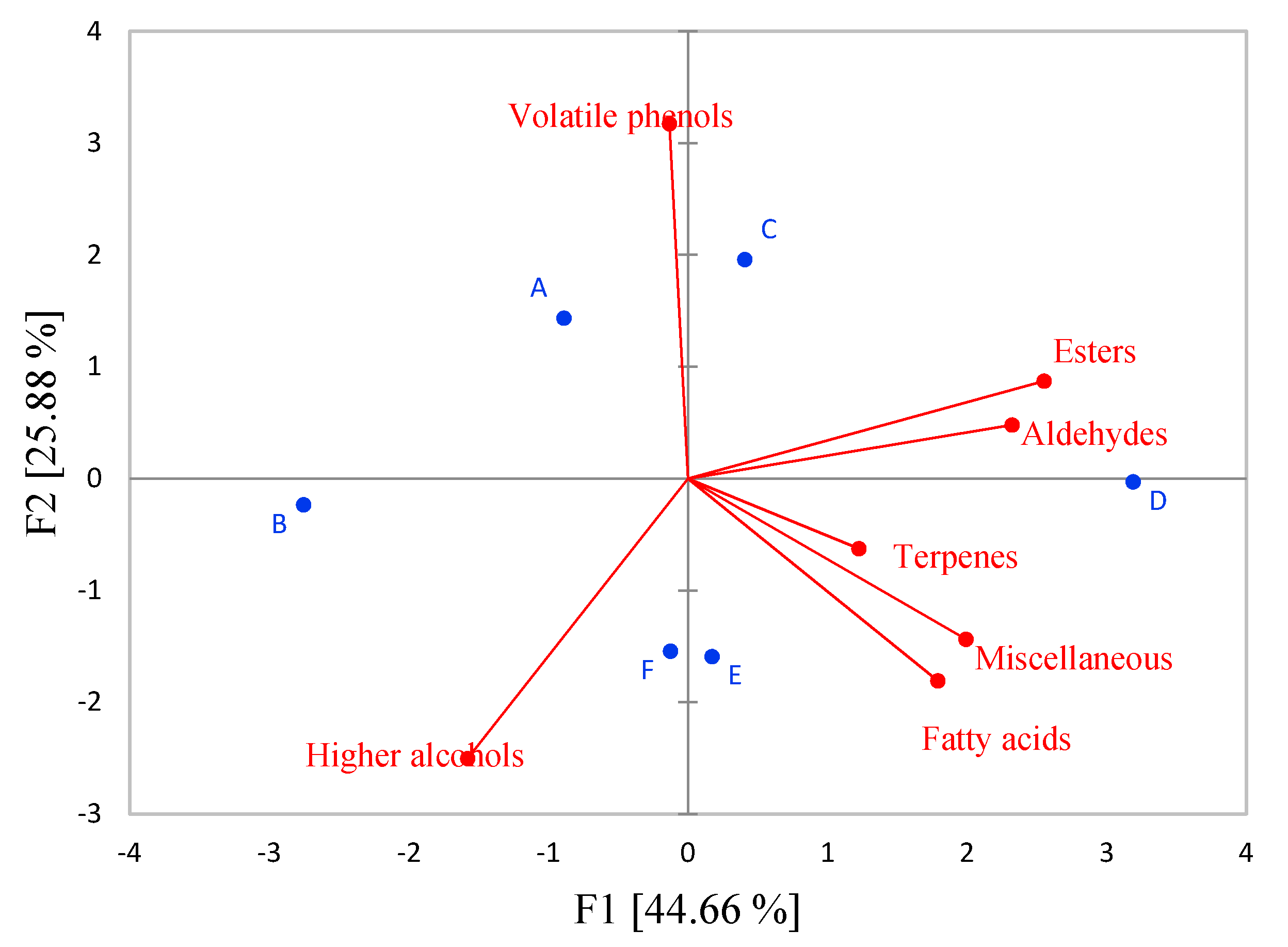
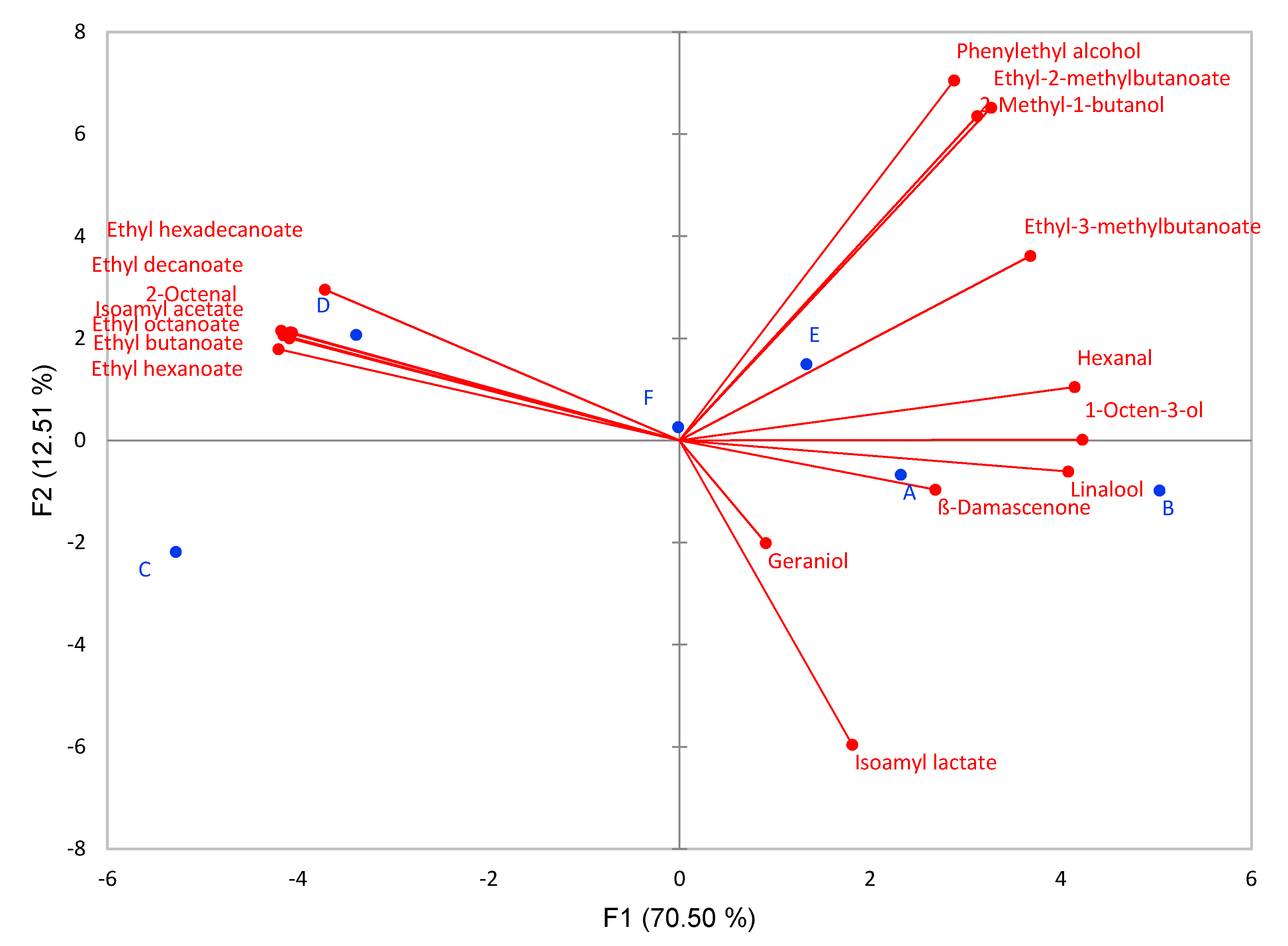
3.4. Aroma Compounds
3.5. Odor Activity Value (OAV) and Relative Odor Contribution (ROC)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chinnici, F.; Sonni, F.; Natali, N.; Riponi, C. Oxidative evolution of (+)-catechin in model white wine solutions containing sulfur dioxide, ascorbic acid or gallotannins. Food Res. Int. 2013, 51, 59–65. [Google Scholar] [CrossRef]
- Boulton, R.B.; Singleton, V.L.; Bisson, L.F.; Kunkee, R.E. Principles and Practices of Winemaking; Chapman and Hall: New York, NY, USA, 1996. [Google Scholar]
- Elias, R.J.; Waterhouse, A.L. Controlling the fenton reaction in wine. J. Agric. Food Chem. 2010, 58, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.L.; Sacks, G.L.; Jeffery, D.W. Understanding Wine Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar]
- Danilewicz, J.C.; Wallbridge, P.J. Further studies on the mechanism of interaction of polyphenols, oxygen, and sulfte in wine. Am. J. Enol. Vitic. 2010, 61, 166–175. [Google Scholar]
- Santos, M.C.; Nunes, C.; Saraiva, J.A.; Coimbra, M.A. Chemical and physical methodologies for the replacement/reduction of sulfur dioxide use during winemaking: Review of their potentialities and limitations. Eur. Food Res. Technol. 2012, 234, 1–12. [Google Scholar] [CrossRef]
- Falguera, V.; Forns, M.; Ibarz, A. UV-vis irradiation: An alternative to reduce SO2 in white wines? LWT Food Sci. Technol. 2013, 51, 59–64. [Google Scholar] [CrossRef]
- Guerrero, R.F.; Cantos-Villar, E. Demonstrating the efficiency of sulphur dioxide replacements in wine: A parameter review. Trends Food Sci. Technol. 2015, 42, 27–43. [Google Scholar] [CrossRef]
- Vally, H.; Misso, N.L.A.; Madan, V. Clinical effects of sulphite additives. Clin. Exp. Allergy 2009, 39, 1643–1651. [Google Scholar] [CrossRef]
- Ribereau-Gayon, P.; Dubourdieu, D.; Doneche, B.; Lonvaud, A. Handbook of Enology: The Microbiology of Wine and Vinifications: Second Edition; John Wiley & Sons, Ltd.: Chichester, UK, 2006; Volume 1, ISBN 9780470010365. [Google Scholar]
- International Organization of Vine and Wine, OIV. Available online: http://188.165.107.123/public/medias/2601/oiv-ma-c1-01.pdf (accessed on 22 May 2020).
- Lisanti, M.T.; Blaiotta, G.; Nioi, C.; Moio, L. Alternative Methods to SO2 for Microbiological Stabilization of Wine. Compr. Rev. Food Sci. Food Saf. 2019, 18, 455–479. [Google Scholar] [CrossRef]
- Morata, A.; Loira, I.; Vejarano, R.; González, C.; Callejo, M.J.; Suárez-Lepe, J.A. Emerging preservation technologies in grapes for winemaking. Trends Food Sci. Technol. 2017, 67, 36–43. [Google Scholar] [CrossRef]
- Yıldırım, H.K.; Darici, B. Alternative methods of sulfur dioxide used in wine production. J. Microbiol. Biotechnol. Food Sci. 2020, 9, 675–687. [Google Scholar] [CrossRef]
- Sonni, F.; Bastante, M.J.C.; Chinnici, F.; Natali, N.; Riponi, C. Replacement of sulfur dioxide by lysozyme and oenological tannins during fermentation: Influence on volatile composition of white wines. J. Sci. Food Agric. 2009, 89, 688–696. [Google Scholar] [CrossRef]
- Barril, C.; Clark, A.C.; Scollary, G.R. Chemistry of ascorbic acid and sulfur dioxide as an antioxidant system relevant to white wine. Anal. Chim. Acta 2012, 732, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Panero, L.; Motta, S.; Petrozziello, M.; Guaita, M.; Bosso, A. Effect of SO2, reduced glutathione and ellagitannins on the shelf life of bottled white wines. Eur. Food Res. Technol. 2015, 240, 345–356. [Google Scholar] [CrossRef]
- Bradshaw, M.P.; Barril, C.; Clark, A.C.; Prenzler, P.D.; Scollary, G.R. Ascorbic acid: A review of its chemistry and reactivity in relation to a wine environment. Crit. Rev. Food Sci. Nutr. 2011, 51, 479–498. [Google Scholar] [CrossRef]
- Versari, A.; Du Toit, W.; Parpinello, G.P. Oenological tannins: A review. Aust. J. Grape Wine Res. 2013, 19, 1–10. [Google Scholar] [CrossRef]
- Pascual, O.; Vignault, A.; Gombau, J.; Navarro, M.; Gómez-Alonso, S.; García-Romero, E.; Canals, J.M.; Hermosín-Gutíerrez, I.; Teissedre, P.L.; Zamora, F. Oxygen consumption rates by different oenological tannins in a model wine solution. Food Chem. 2017, 234, 26–32. [Google Scholar] [CrossRef]
- Morrot, G.; Brochet, F.; Dubourdieu, D. The color of odors. Brain Lang. 2001, 79, 309–320. [Google Scholar] [CrossRef]
- Olejar, K.J.; Fedrizzi, B.; Kilmartin, P.A. Influence of harvesting technique and maceration process on aroma and phenolic attributes of Sauvignon blanc wine. Food Chem. 2015, 183, 181–189. [Google Scholar] [CrossRef]
- Ferrer-Gallego, R.; Puxeu, M.; Nart, E.; Martín, L.; Andorrà, I. Evaluation of Tempranillo and Albariño SO2-free wines produced by different chemical alternatives and winemaking procedures. Food Res. Int. 2017, 102, 647–657. [Google Scholar] [CrossRef]
- Gabriele, M.; Gerardi, C.; Lucejko, J.J.; Longo, V.; Pucci, L.; Domenici, V. Effects of low sulfur dioxide concentrations on bioactive compounds and antioxidant properties of Aglianico red wine. Food Chem. 2018, 245, 1105–1112. [Google Scholar] [CrossRef]
- OIV. International Code of Oenological Practices. Part II Practice and Oenological Treatments; OIV: Paris, France, 2016. [Google Scholar]
- Dukes, B.C.; Butzke, C.E. Rapid determination of primary amino acids in grape juice using an o-phthaldialdehyde/N-acetyl-L-cysteine spectrophotometric assay. Am. J. Enol. Vitic. 1998, 49, 125–134. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Glories, Y. La couleur des vins rouges II, Connaissance de la vigne et du vin. Vigne Vin 1984, 18, 253–271. [Google Scholar]
- Maslov, L.; Tomaz, I.; Mihaljević Žulj, M.; Jeromel, A. Aroma characterization of predicate wines from Croatia. Eur. Food Res. Technol. 2017, 243, 263–274. [Google Scholar] [CrossRef]
- Falqué, E.; Fernández, E.; Dubourdieu, D. Differentiation of white wines by their aromatic index. Talanta 2001, 54, 271–281. [Google Scholar] [CrossRef]
- Allen, M.S.; Lacey, M.J.; Boyd, S. Determination of Methoxypyrazines in Red Wines by Stable Isotope Dilution Gas Chromatography–Mass Spectrometry. J. Agric. Food Chem. 1994, 42, 1734–1738. [Google Scholar] [CrossRef]
- Ohloff, G. The Fashion of Odors and Their Chemical Perspectives: Scent and Fragrances; Springer: Berlin/Heidelberg, Germany, 1994. [Google Scholar]
- Jackson, R. Wine Science, 4th ed.; Academic Press: San Diego, CA, USA, 2014. [Google Scholar]
- Ribéreau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Handbook of Enology, The Chemistry of Wine: Stabilization and Treatments, 2nd ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006; Volume 2, ISBN 9780470010396. [Google Scholar]
- Regulations of the Wine Production (2/2005). Available online: https://narodne-novine.nn.hr/clanci/sluzbeni/2005_01_2_17.html (accessed on 20 December 2019).
- Vinković Vrček, I.; Bojić, M.; Žuntar, I.; Mendaš, G.; Medić-Šarić, M. Phenol content, antioxidant activity and metal composition of Croatian wines deriving from organically and conventionally grown grapes. Food Chem. 2011, 124, 354–361. [Google Scholar] [CrossRef]
- Jakobović, S.; Jeromel, A.; Maslov, L.; Jakobović, M. Influence of grape ripeness of Rhine Riesling on the composition of polyphenolic compounds in must and wine. J. Food Agric. Environ. 2015, 13, 29–35. [Google Scholar]
- Jagatić Korenika, A.M. Utjecaj Hladne Maceracije na Polifenolni Sastav i Senzorna Svojstva Vina Kultivara Pošip, Maraština, Malvazija, Kraljevina i Škrlet (Vitis vinifera L.); University of Zagreb Faculty of Agriculture: Croatia, Zagreb, 2015. [Google Scholar]
- Ivanova, V.; Vojnoski, B.; Stefova, M. Effect of winemaking treatment and wine aging on phenolic content in Vranec wines. J. Food Sci. Technol. 2012, 49, 161–172. [Google Scholar] [CrossRef]
- Fairchild, M.D. The colors of wine. Int. J. Wine Res. 2018, 10, 13–31. [Google Scholar] [CrossRef]
- Rapp, A.; Versini, G. Influence of nitrogen compounds in grapes on aroma compounds of wines. In Developments in Food Science; Elsevier: Amsterdam, The Netherlands, 1995; Volume 37, pp. 1659–1694. [Google Scholar]
- Jackson, R.S. Wine Science Principles and Applications, 3rd ed.; Academic Press Elsevier: Burlington, MA, USA, 2008. [Google Scholar]
- Toci, A.T.; Tamborra, P.; Crupi, P.; Cantarini, L.; Antonacci, D. Comparison of winemaking techniques to produce wines with low sulphur dioxide. In Proceedings of the 35th World Congress of Vine and Wine, Izmir, Turkey, 18–22 June 2012. [Google Scholar]
- Patel, S.; Shibamoto, T. Flavor compounds in wines produced from chardonnay grapes fermented with fruit juices. Food Sci. Technol. Res. 2003, 9, 84–86. [Google Scholar] [CrossRef]
- Jackson, R.S. Wine Tasting, 2nd ed.; Academic Press Elsevier: Burlington, MA, USA, 2009. [Google Scholar]
- Pino, J.A.; Mesa, J. Contribution of volatile compounds to mango (Mangifera indica L.) aroma. Flavour Fragr. J. 2006, 21, 207–213. [Google Scholar] [CrossRef]
- Ferreira, V.; Ortín, N.; Escudero, A.; López, R.; Cacho, J. Chemical characterization of the aroma of Grenache rosé wines: Aroma extract dilution analysis, quantitative determination, and sensory reconstitution studies. J. Agric. Food Chem. 2002, 50, 4048–4054. [Google Scholar] [CrossRef] [PubMed]
- Herrero, P.; Sáenz-Navajas, P.; Culleré, L.; Ferreira, V.; Chatin, A.; Chaperon, V.; Litoux-Desrues, F.; Escudero, A. Chemosensory characterization of Chardonnay and Pinot Noir base wines of Champagne. Two very different varieties for a common product. Food Chem. 2016, 207, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, V.; López, R.; Cacho, J.F. Quantitative determination of the odorants of young red wines from different grape varieties. J. Sci. Food Agric. 2000, 80, 1659–1667. [Google Scholar] [CrossRef]
- Amoore, J.E.; Hautala, E. Odor as an ald to chemical safety: Odor thresholds compared with threshold limit values and volatilities for 214 industrial chemicals in air and water dilution. J. Appl. Toxicol. 1983, 3, 272–290. [Google Scholar] [CrossRef]
- Howard, K.L.; Mike, J.H.; Riesen, R. Validation of a solid-phase microextraction method for headspace analysis of wine aroma components. Am. J. Enol. Vitic. 2005, 56, 37–45. [Google Scholar]
- Guth, H. Quantitation and Sensory Studies of Character Impact Odorants of Different White Wine Varieties. J. Agric. Food Chem. 1997, 45, 3027–3032. [Google Scholar] [CrossRef]
- Yang, C.; Luo, L.; Zhang, H.; Yang, X.; Lv, Y.; Song, H. Common aroma-active components of propolis from 23 regions of China. J. Sci. Food Agric. 2010, 90, 1268–1282. [Google Scholar] [CrossRef]
- Buttery, R.G.; Teranishi, R.; Ling, L.C.; Turnbaugh, J.G. Quantitative and Sensory Studies on Tomato Paste Volatiles. J. Agric. Food Chem. 1990, 38, 336–340. [Google Scholar] [CrossRef]

| Parameter | A | B | C | D | E | F |
|---|---|---|---|---|---|---|
| Alcohol (%, v/v) | 13. 7± 0.0 | 13.6 ± 0.0 | 13.6 ± 0.0 | 13.5 ± 0.0 | 13.7 ± 0.0 | 13.6 ± 0.0 |
| Residual sugars (g/L) | 2.8 ± 0.1 e | 2.7 ± 0.1 e | 5.8 ± 0.1 b | 6.2 ± 0.1 a | 5.3 ± 0.1 d | 5.6 ± 0.1 c |
| Sugar-free extract (g/L) | 18.0 ± 0.1 d | 18.9 ± 0.1 a | 18.4 ± 0.1 b | 17.7 ± 0.1 f | 17.9 ± 0.1 e | 18.3 ± 0.1 c |
| Total acidity * (g/L) | 5.8 ± 0.1 e | 6.6 ± 0.1 a | 6.0 ± 0.1 d | 5.9 ± 0.0 e | 6.2 ± 0.1 c | 6.4 ± 0.0 b |
| Volatile acidity ** (g/L) | 0.60 ± 0.0 c | 0.54 ± 0.0 d | 0.65 ± 0.0 b | 0.70 ± 0.0 a | 0.65 ± 0.0 b | 0.59 ± 0.0 c |
| pH | 3.3 ± 0.0 | 3.3 ± 0.0 | 3.3 ± 0.0 | 3.3 ± 0.0 | 3.3 ± 0.0 | 3.3 ± 0.0 |
| SO2 bound (mg/L) | 37.0 ± 0.5 f | 40.0 ± 0.5 e | 59.0 ± 0.5 a | 55.0 ± 0.5 c | 57.0 ± 0.5 b | 45.0 ± 0.5 d |
| SO2 total (mg/L) | 37.0 ± 0.05 f | 40.0 ± 0.0 e | 59.0 ± 0.5 a | 55.0 ± 0.5 c | 57.0 ± 0.5 b | 45.0 ± 0.5 d |
| Ash (g/L) | 1.71 ± 0.0 d | 1.74 ± 0.0 b | 1.73 ± 0.0 c | 1.72 ± 0.0 c | 1.72 ± 0.0 c | 1.78 ± 0.0 a |
| Total phenols (mg/L GAE) | 241.69 ± 0.0 f | 250.75 ± 0.0 d | 275.63 ± 0.0 b | 267.8 ± 0.0 c | 278.02 ± 0.1 a | 247.77 ± 0.0 e |
| ABTS (µM/L TE) | 1.26 ± 0.0 d | 1.34 ± 0.0 c,d | 1.44 ± 0.0 a,b | 1.42 ± 0.0 a,b,c | 1.50 ± 0.0 a | 1.37 ± 0.0 b,c |
| Compounds | QIon | Rt | A | B | C | D | E | F | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aldehydes | ||||||||||||||||||||
| Hexanal | 44 | 21.93 | 460.00 | ± | 0.71 b | 466.86 | ± | 0.49 a | 333.28 | ± | 1.22 f | 381.17 | ± | 1.12 e | 427.69 | ± | 0.37 c | 397.71 | ± | 0.79 d |
| 2-Pentanal | 55 | 24.13 | 12.50 | ± | 0.26 e | 16.52 | ± | 0.02 b | 15.47 | ± | 0.27 c | 25.06 | ± | 0.15 a | 14.43 | ± | 0.01 d | 14.00 | ± | 0.11 d |
| 2-Octenal (g/L) | 41 | 44.71 | 1.15 | ± | 0.00 e | 1.18 | ± | 0.01 e | 2.72 | ± | 0.00 b | 2.83 | ± | 0.00 a | 1.79 | ± | 0.00 d | 1.94 | ± | 0.00 c |
| Decanal | 43 | 48.97 | 0.88 | ± | 0.01 f | 2.77 | ± | 0.08 d | 1.96 | ± | 0.03 e | 5.47 | ± | 0.06 a | 3.44 | ± | 0.02 c | 4.93 | ± | 0.03 b |
| Benzaldehyde | 106 | 51.01 | 17.60 | ± | 0.01 b | 21.04 | ± | 0.12 a | 13.24 | ± | 0.07 e | 17.06 | ± | 0.15 c | 11.89 | ± | 0.10 f | 14.06 | ± | 0.23 d |
| Esters | ||||||||||||||||||||
| Isobutyl acetate | 43 | 16.83 | 54.17 | ± | 0.02 d | 44.41 | ± | 0.06 e | 80.56 | ± | 0.16 b | 83.06 | ± | 0.12 a | 57.69 | ± | 0.38 c | 57.34 | ± | 0.23 c |
| Ethyl butanoate | 71 | 18.03 | 281.50 | ± | 0.57 c | 236.08 | ± | 1.29 d | 340.64 | ± | 4.94 b | 355.40 | ± | 13.30 a | 283.54 | ± | 0.70 c | 284.57 | ± | 0.78 c |
| Ethyl-2-methylbutanoate | 57 | 19.10 | 18.41 | ± | 0.03 e | 21.82 | ± | 0.05 b | 17.63 | ± | 0.01 f | 19.06 | ± | 0.01 d | 22.09 | ± | 0.04 a | 19.36 | ± | 0.06 c |
| Ethyl-3-methylbutanoate | 88 | 20.11 | 26.56 | ± | 0.01 b | 28.37 | ± | 0.05 a | 22.77 | ± | 0.01 d | 24.94 | ± | 0.13 c | 29.14 | ± | 0.22 a | 25.34 | ± | 0.68 b,c |
| Isoamyl acetate (g/L) | 43 | 23.51 | 1.69 | ± | 0.00 d | 1.06 | ± | 0.00 e | 2.23 | ± | 0.00 b | 2.28 | ± | 0.01 a | 1.70 | ± | 0.00 d | 1.78 | ± | 0.75 c |
| Ethyl hexanoate | 88 | 31.12 | 316.08 | ± | 0.67 e | 219.50 | ± | 0.69 f | 545.54 | ± | 0.86 b | 563.51 | ± | 2.32 a | 357.84 | ± | 2.16 d | 385.87 | ± | 0.41 c |
| Ethyl lactate | 45 | 38.67 | 18.75 | ± | 0.06 b | 19.76 | ± | 0.14 a | 13.95 | ± | 0.07 e | 13.45 | ± | 0.04 f | 15.95 | ± | 0.09 d | 18.40 | ± | 0.05 c |
| Ethyl-2-hydroxy-3-methylbutanoate | 73 | 44.52 | 417.86 | ± | 1.00 e | 293.81 | ± | 0.65 f | 672.94 | ± | 2.22 b | 697.27 | ± | 5.46 a | 440.19 | ± | 0.74 d | 476.98 | ± | 1.46 c |
| Ethyl octanoate | 88 | 44.69 | 585.83 | ± | 0.42 e | 409.07 | ± | 0.29 f | 943.26 | ± | 1.91 b | 979.04 | ± | 10.95 a | 620.44 | ± | 1.70 d | 671.91 | ± | 2.21 c |
| Isobutyl lactate | 45 | 53.70 | 87.93 | ± | 0.16 b | 114.87 | ± | 0.41 a | 72.30 | ± | 0.11 c | 71.72 | ± | 0.16 c | 68.57 | ± | 0.01 d | 57.84 | ± | 0.64 e |
| Isoamyl lactate | 45 | 53.70 | 31.66 | ± | 0.03 b | 42.64 | ± | 0.04 a | 31.50 | ± | 0.07 b | 27.28 | ± | 0.02 c | 23.08 | ± | 0.06 d | 22.35 | ± | 0.04 e |
| Ethyl furoate | 95 | 56.88 | 15.88 | ± | 0.01 e | 10.89 | ± | 0.03 f | 25.54 | ± | 0.01 b | 28.11 | ± | 0.37 a | 18.16 | ± | 0.03 d | 19.31 | ± | 0.01 c |
| Ethyl decanoate | 88 | 57.31 | 526.86 | ± | 0.01 e | 365.89 | ± | 0.04 f | 880.98 | ± | 2.46 b | 935.10 | ± | 0.78 a | 591.66 | ± | 1.42 d | 659.37 | ± | 1.50 c |
| Diethyl succinate (g/L) | 101 | 59.67 | 1.15 | ± | 0.04 b | 1.30 | ± | 0.02 a | 0.88 | ± | 0.00 d | 0.99 | ± | 0.02 c | 1.12 | ± | 0.01 b | 1.23 | ± | 0.70 a |
| Ethylmethyl succinate | 101 | 61.00 | 38.31 | ± | 0.01 e | 26.85 | ± | 0.04 f | 61.78 | ± | 0.02 b | 65.76 | ± | 0.02 a | 42.41 | ± | 0.01 d | 47.34 | ± | 0.08 c |
| Diethyl glutarate | 143 | 65.93 | 0.60 | ± | 0.01 d | 1.11 | ± | 0.01 a | 0.44 | ± | 0.01 e | 0.66 | ± | 0.01 c | 0.98 | ± | 0.01 b | 0.70 | ± | 0.01 c |
| Methyl-3-hydroxyoctanoate | 103 | 77.10 | 0.85 | ± | 0.04 b | 2.60 | ± | 0.11 a | 0.32 | ± | 0.01 d | 0.89 | ± | 0.02 b | 0.67 | ± | 0.02 c | 0.99 | ± | 0.01 b |
| Diethyl malonate | 117 | 79.86 | 0.36 | ± | 0.03 c | 0.73 | ± | 0.03 a | 0.47 | ± | 0.02 b | 0.31 | ± | 0.01 c | 0.35 | ± | 0.02 c | 0.32 | ± | 0.02 c |
| Ethyl hexadecanoate | 88 | 88.41 | 8.29 | ± | 0.19 d,e | 7.73 | ± | 0.09 e | 16.30 | ± | 0.07 b | 21.40 | ± | 0.60 a | 9.12 | ± | 0.23 d | 12.18 | ± | 0.21 c |
| Ethyl hydrogensuccinate (g/L) | 101 | 96.60 | 2.26 | ± | 0.00 b,c | 2.60 | ± | 0.00 a | 1.90 | ± | 0.00 e | 2.30 | ± | 0.00 b | 2.21 | ± | 0.02 cd | 2.18 | ± | 0.02 d |
| Ethyl linoleate | 67 | 101.16 | 2.17 | ± | 0.02 b | 3.81 | ± | 0.06 a | 1.79 | ± | 0.04 c | 1.54 | ± | 0.07 d | 1.27 | ± | 0.01 e | 1.24 | ± | 0.01 e |
| Alcohols | ||||||||||||||||||||
| 1-Butanol | 56 | 24.66 | 28.21 | ± | 0.04 e | 28.94 | ± | 0.10 e | 88.67 | ± | 0.49 b | 97.05 | ± | 0.04 a | 74.54 | ± | 0.75 d | 78.43 | ± | 0.15 c |
| 2-Methyl-1-butanol (g/L) | 55 | 28.96 | 31.40 | ± | 0.06 a | 32.18 | ± | 0.02 a | 26.45 | ± | 0.06 a | 30.92 | ± | 0.05 a | 31.01 | ± | 0.08 a | 30.49 | ± | 0.01 a |
| Isoamyl alcohol (g/L) | 41 | 29.03 | 33.53 | ± | 0.03 f | 60.75 | ± | 0.03 a | 49.10 | ± | 0.07 e | 58.55 | ± | 0.06 f | 59.25 | ± | 0.03 b | 57.01 | ± | 0.10 d |
| 1-Pentanol | 42 | 32.20 | 2.07 | ± | 0.01 a | 1.59 | ± | 0.02 b | 0.77 | ± | 0.06 d | 1.07 | ± | 0.03 c | 0.12 | ± | 0.02 e | 0.24 | ± | 0.01 e |
| 4-Methyl-1-pentanol | 56 | 36.70 | 101.77 | ± | 0.08 b | 79.66 | ± | 0.48 e | 86.77 | ± | 0.08 d | 101.36 | ± | 0.25 b | 117.60 | ± | 0.02 a | 97.76 | ± | 0.29 c |
| 2-Heptanol | 45 | 36.90 | 1.09 | ± | 0.01 b | 0.15 | ± | 0.01 e | 1.69 | ± | 0.02 a | 0.17 | ± | 0.01 e | 0.59 | ± | 0.02 c | 0.26 | ± | 0.01 d |
| 2-Pentene-1-ol | 57 | 37.10 | 58.20 | ± | 0.01 d | 44.48 | ± | 0.06 f | 54.27 | ± | 0.39 e | 64.24 | ± | 0.03 b | 69.45 | ± | 0.64 a | 59.94 | ± | 0.05 c |
| 3-Methyl-1-pentanol | 56 | 37.56 | 227.87 | ± | 0.16 d | 180.03 | ± | 0.30 f | 218.65 | ± | 0.62 e | 252.42 | ± | 0.37 b | 277.34 | ± | 0.70 a | 240.88 | ± | 0.25 c |
| 1-Hexanol (g/L) | 56 | 39.88 | 1.05 | ± | 0.01 b | 1.12 | ± | 0.01 a | 0.68 | ± | 0.00 f | 0.77 | ± | 0.01 e | 0.83 | ± | 0.00 d | 0.91 | ± | 0.00 c |
| 3-Hexene-1-ol, trans | 41 | 39.88 | 32.60 | ± | 0.14 d | 31.50 | ± | 0.11 e | 56.16 | ± | 0.04 b | 59.18 | ± | 0.21 a | 52.15 | ± | 0.29 c | 31.04 | ± | 0.39 e |
| 3-Hexene-1-ol, cis | 67 | 41.76 | 16.38 | ± | 0.04 e | 12.39 | ± | 0.12 f | 26.63 | ± | 0.28 b | 28.17 | ± | 0.21 a | 19.77 | ± | 0.07 d | 21.10 | ± | 0.06 c |
| Cyclohexanol | 57 | 41.86 | 1.50 | ± | 0.11 a | 1.29 | ± | 0.01 b | 0.92 | ± | 0.02 c | 0.80 | ± | 0.02 c,d | 1.17 | ± | 0.01 b | 0.73 | ± | 0.03 d |
| 2-Hexene-1-ol, trans | 57 | 42.72 | 9.21 | ± | 0.25 a | 8.06 | ± | 0.03 b | 4.81 | ± | 0.05 d | 5.46 | ± | 0.15 d | 6.24 | ± | 0.37 c | 7.55 | ± | 0.11 b |
| 1-Octen-3-ol | 57 | 45.53 | 39.36 | ± | 0.52 b | 44.70 | ± | 0.97 a | 25.01 | ± | 1.03 e | 30.37 | ± | 0.13 d | 35.43 | ± | 0.65 c | 33.77 | ± | 0.66 c |
| 1-Heptanol | 70 | 45.93 | 1.13 | ± | 0.01 d | 0.82 | ± | 0.04 e | 2.49 | ± | 0.05 a | 2.59 | ± | 0.06 a | 1.60 | ± | 0.01 b | 1.39 | ± | 0.04 c |
| 2-Ethyl-1-hexanol | 57 | 48.07 | 0.81 | ± | 0.02 a | 0.51 | ± | 0.03 c | 0.64 | ± | 0.04 b | 0.84 | ± | 0.03 a | 0.66 | ± | 0.01 b | 0.39 | ± | 0.03 d |
| 2,6-Dimethyl-4-heptanol | 69 | 51.86 | 5.27 | ± | 0.04 a,b | 3.86 | ± | 0.11 e | 4.42 | ± | 0.03 d | 4.89 | ± | 0.01 c | 5.03 | ± | 0.05 b,c | 5.29 | ± | 0.03 a |
| 1-Octanol | 56 | 52.32 | 5.43 | ± | 0.01 c | 5.14 | ± | 0.01 d | 5.59 | ± | 0.04 b | 5.76 | ± | 0.04 b | 5.63 | ± | 0.05 b | 6.04 | ± | 0.04 a |
| 2,3-Butanediol | 45 | 53.52 | 11.09 | ± | 0.02 c | 5.58 | ± | 0.01 e | 7.38 | ± | 0.05 d | 5.56 | ± | 0.01 e | 11.86 | ± | 0.08 b | 15.91 | ± | 0.13 a |
| 2-Octen-1-ol | 57 | 56.04 | 0.11 | ± | 0.01 c | 0.23 | ± | 0.02 b | 0.24 | ± | 0.00 b | 0.25 | ± | 0.01 b | 0.10 | ± | 0.01 c | 0.34 | ± | 0.01 a |
| 1-Decanol | 55 | 64.27 | 4.44 | ± | 0.01 b | 5.07 | ± | 0.02 a | 3.76 | ± | 0.04 e | 4.27 | ± | 0.02 c | 4.31 | ± | 0.02 c | 4.06 | ± | 0.01 d |
| Phenylethyl Alcohol (g/L) | 91 | 72.78 | 32.93 | ± | 0.44 b,c | 33.42 | ± | 0.10 a | 29.82 | ± | 0.02 d | 33.39 | ± | 0.07 ab | 33.99 | ± | 0.08 a | 31.97 | ± | 0.11 c |
| 2-Pentadecanol | 45 | 74.97 | 3.20 | ± | 0.01 b | 2.80 | ± | 0.13 c | 2.82 | ± | 0.01 c | 2.87 | ± | 0.01 c | 2.94 | ± | 0.03 c | 4.21 | ± | 0.02 a |
| 1-Octadecanol | 83 | 104.43 | 3.55 | ± | 0.10 a | 2.60 | ± | 0.07 b | 2.73 | ± | 0.01 b | 2.37 | ± | 0.13 b | 2.42 | ± | 0.04 b | 2.40 | ± | 0.06 b |
| Terpenes | ||||||||||||||||||||
| γ-Terpinene | 93 | 32.03 | 25.39 | ± | 0.06 b | 28.55 | ± | 0.35 a | 20.10 | ± | 0.16 d | 22.19 | ± | 0.08 c | 25.24 | ± | 0.38 b | 28.16 | ± | 0.02 a |
| Tetrahydrolinalool | 73 | 44.32 | 53.27 | ± | 0.09 b | 59.79 | ± | 0.24 a | 24.07 | ± | 0.23 f | 29.26 | ± | 0.09 e | 39.94 | ± | 0.12 c | 34.15 | ± | 0.23 d |
| Linalyl formate | 69 | 46.98 | 15.02 | ± | 0.09 b | 15.70 | ± | 0.09 a | 12.21 | ± | 0.14 c | 14.95 | ± | 0.28 b | 14.70 | ± | 0.33 b | 14.99 | ± | 0.11 b |
| Linalool | 71 | 51.72 | 46.63 | ± | 0.09 b | 52.94 | ± | 0.76 a | 35.01 | ± | 0.40 e | 39.40 | ± | 0.07 d | 42.84 | ± | 0.03 c | 40.47 | ± | 0.12 d |
| Terpinene-4-ol | 71 | 55.47 | 37.72 | ± | 0.35 b | 40.44 | ± | 0.40 a | 36.00 | ± | 0.86 b,c | 42.16 | ± | 0.42 a | 37.18 | ± | 0.28 b | 35.04 | ± | 0.04 c |
| Hotrienol | 71 | 55.74 | 51.16 | ± | 0.72 c | 55.72 | ± | 0.21 b | 51.14 | ± | 0.02 c | 58.02 | ± | 0.16 a | 51.36 | ± | 0.24 c | 48.23 | ± | 0.30 d |
| α-Terpineol | 59 | 60.92 | 4.93 | ± | 0.01 a | 4.95 | ± | 0.03 a | 4.02 | ± | 0.06 c | 4.58 | ± | 0.06 b | 4.99 | ± | 0.05 a | 4.53 | ± | 0.05 a |
| Linalool oxide pyran | 68 | 63.38 | 5.52 | ± | 0.01 b | 4.38 | ± | 0.06 c | 5.56 | ± | 0.04 a,b | 5.69 | ± | 0.01 a | 4.39 | ± | 0.01 c | 5.59 | ± | 0.02 a,b |
| Citronellol | 69 | 64.53 | 10.98 | ± | 0.06 b | 19.10 | ± | 0.11 a | 8.10 | ± | 0.04 d | 11.34 | ± | 0.08 b | 10.21 | ± | 0.13 c | 10.04 | ± | 0.11 c |
| Nerol | 69 | 66.54 | 159.66 | ± | 0.74 c | 171.77 | ± | 0.69 a | 150.52 | ± | 0.23 d | 174.11 | ± | 0.78 a | 164.61 | ± | 1.48 b | 157.98 | ± | 0.03 c |
| Geraniol | 69 | 68.99 | 49.29 | ± | 0.52 e | 40.18 | ± | 0.01 f | 81.83 | ± | 0.02 b | 83.83 | ± | 0.30 a | 55.35 | ± | 0.54 d | 57.61 | ± | 0.28 c |
| Terpendiol II | 67 | 74.09 | 39.80 | ± | 0.21 b | 22.34 | ± | 0.22 d | 41.87 | ± | 0.18 a | 41.05 | ± | 0.36 a | 34.72 | ± | 0.14 c | 41.99 | ± | 0.09 a |
| 6,7-Dihydro-7-hydroxylinalool | 71 | 75.47 | 3.39 | ± | 0.18 b | 2.68 | ± | 0.04 c | 4.24 | ± | 0.20 a | 3.99 | ± | 0.16 a | 2.31 | ± | 0.10 c | 3.24 | ± | 0.10 b |
| Neralidol | 69 | 78.55 | 18.49 | ± | 0.06 a | 12.01 | ± | 0.08 d | 17.22 | ± | 0.14 c | 17.94 | ± | 0.04 b | 12.28 | ± | 0.11 d | 18.68 | ± | 0.01 a |
| Geranyl acetate | 69 | 91.21 | 7.57 | ± | 0.02 c | 7.03 | ± | 0.06 c | 4.54 | ± | 0.18 d | 9.24 | ± | 0.08 b | 9.05 | ± | 0.13 b | 11.19 | ± | 0.26 a |
| 8-Hidroxylinalool | 43 | 91.41 | 4.51 | ± | 0.01 d | 1.81 | ± | 0.01 e | 7.50 | ± | 0.21 c | 13.22 | ± | 0.47 a | 4.62 | ± | 0.11 d | 12.02 | ± | 0.02 b |
| Volatile phenols | ||||||||||||||||||||
| Benzaacetaldehyde | 91 | 58.28 | 0.00 | ± | 0.00 | 0.00 | ± | 0.00 | 3.41 | ± | 0.01 a | 2.78 | ± | 0.02 c | 2.82 | ± | 0.01 b | 1.08 | ± | 0.01 d |
| Benzylalcohol | 78 | 70.92 | 4.52 | ± | 0.02 a | 4.79 | ± | 0.02 a | 3.57 | ± | 0.04 c | 3.99 | ± | 0.18 b | 3.38 | ± | 0.06 c | 3.74 | ± | 0.06 c |
| 4-Ethylguaiacol | 85 | 78.84 | 3.95 | ± | 0.04 a | 0.00 | ± | 0.00 | 3.16 | ± | 0.01 b | 0.00 | ± | 0.00 | 4.01 | ± | 0.04 a | 0.00 | ± | 0.00 |
| Tyrosol | 107 | 97.21 | 23.88 | ± | 0.08 a | 6.32 | ± | 0.01 e | 18.15 | ± | 0.01 b | 15.81 | ± | 0.02 f | 16.82 | ± | 0.40 c | 11.12 | ± | 0.31 d |
| Vanilin | 151 | 102.78 | 4.71 | ± | 0.01 a | 4.50 | ± | 0.01 b | 4.59 | ± | 0.02 b | 5.14 | ± | 0.06 b | 4.43 | ± | 0.12 b | 4.52 | ± | 0.04 b |
| Methyl vanillate | 151 | 104.17 | 2.75 | ± | 0.01 c | 3.21 | ± | 0.03 a | 2.61 | ± | 0.06 c | 2.94 | ± | 0.03 b | 2.47 | ± | 0.01 d | 2.28 | ± | 0.01 e |
| Ethyl vanillate | 151 | 106.62 | 3.06 | ± | 0.03 b | 5.89 | ± | 0.06 a | 0.45 | ± | 0.02 c | 0.00 | ± | 0.00 | 0.48 | ± | 0.02 c | 0.00 | ± | 0.00 |
| Homovanillyl alcohol | 137 | 115.17 | 1.40 | ± | 0.01 b | 0.37 | ± | 0.01 e | 1.55 | ± | 0.04 a | 1.27 | ± | 0.01 c | 0.38 | ± | 0.01 e | 0.77 | ± | 0.04 d |
| Fatty acids | ||||||||||||||||||||
| Propanoic acid | 74 | 51.97 | 29.95 | ± | 0.04 e | 20.47 | ± | 0.02 f | 33.91 | ± | 0.10 c | 32.20 | ± | 0.07 d | 38.42 | ± | 0.01 a | 36.09 | ± | 0.04 b |
| 2-Methylpropionic acid | 43 | 54.57 | 7.77 | ± | 0.01 b | 6.30 | ± | 0.02 c | 5.09 | ± | 0.02 e | 10.96 | ± | 0.13 a | 6.23 | ± | 0.03 c | 5.58 | ± | 0.03 d |
| Isovaleric acid | 60 | 60.48 | 4.41 | ± | 0.01 d | 5.30 | ± | 0.07 c | 5.53 | ± | 0.05 c | 8.24 | ± | 0.08 b | 11.02 | ± | 0.09 a | 5.46 | ± | 0.08 c |
| Heptanoic acid | 60 | 75.04 | 8.90 | ± | 0.08 d | 8.23 | ± | 0.06 e | 8.48 | ± | 0.02 e | 10.63 | ± | 0.05 b | 9.69 | ± | 0.06 c | 11.22 | ± | 0.16 a |
| Octanoic acid | 60 | 80.35 | 8.50 | ± | 0.01 a | 6.57 | ± | 0.02 c | 2.38 | ± | 0.05 e | 7.33 | ± | 0.04 b | 4.26 | ± | 0.17 d | 4.40 | ± | 0.01 d |
| Miscellaneous | ||||||||||||||||||||
| 4-Methyl--2-penten-2-one | 55 | 24.45 | 21.86 | ± | 0.02 e | 22.04 | ± | 0.04 e | 41.22 | ± | 0.24 b | 47.77 | ± | 0.10 a | 31.77 | ± | 0.62 d | 34.05 | ± | 0.11 c |
| Acetoin | 45 | 34.89 | 4.97 | ± | 0.08 b | 5.61 | ± | 0.02 a | 4.95 | ± | 0.04 b | 4.53 | ± | 0.03 c | 4.21 | ± | 0.03 d | 4.88 | ± | 0.01 b |
| 6-Methyl-5-hepten-2-on | 43 | 38.52 | 150.75 | ± | 0.13 e | 164.77 | ± | 0.71 d | 149.79 | ± | 0.05 e | 181.68 | ± | 0.64 c | 187.90 | ± | 0.11 b | 212.40 | ± | 0.44 a |
| N-Ethylacetamide | 43 | 57.77 | 3.87 | ± | 0.21 d | 5.05 | ± | 0.02 c | 2.51 | ± | 0.01 e | 7.38 | ± | 0.04 a | 5.77 | ± | 0.01 b | 2.48 | ± | 0.03 e |
| Dihydro-2-methyl-3(2H)furanone | 43 | 62.67 | 2.76 | ± | 0.00 a | 2.25 | ± | 0.02 b | 2.70 | ± | 0.01 a | 1.64 | ± | 0.03 c | 0.13 | ± | 0.01 e | 0.94 | ± | 0.05 d |
| ß-Damascenone | 69 | 68.11 | 6.64 | ± | 0.04 b | 6.80 | ± | 0.15 b | 5.70 | ± | 0.01 d | 5.77 | ± | 0.01 d | 6.10 | ± | 0.07 c | 7.44 | ± | 0.04 a |
| 4-(Methylthio)-1-butanol | 61 | 69.56 | 289.97 | ± | 0.33 e | 210.35 | ± | 1.75 f | 490.34 | ± | 6.85 b | 507.99 | ± | 0.32 a | 321.50 | ± | 0.74 d | 340.92 | ± | 1.08 c |
| γ-Carboethoxy-γ-butyrolactone | 85 | 85.57 | 109.90 | ± | 0.11 c | 15.04 | ± | 0.03 e | 29.13 | ± | 0.78 e | 66.23 | ± | 0.81 d | 390.76 | ± | 10.70 a | 169.20 | ± | 6.33 b |
| N-(2-phenylethyl) acetamide | 104 | 106.27 | 2.82 | ± | 0.01 b | 2.79 | ± | 0.04 bc | 3.08 | ± | 0.01 a | 2.84 | ± | 0.04 b | 2.59 | ± | 0.04 cd | 2.50 | ± | 0.06 d |
| Compounds | OTH (mg/L) | Odor Descriptor | OAV | ROC (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | A | B | C | D | E | F | |||
| 2-Octenal | 0.003 1 | Green, nut, fat | 384.00 | 394.64 | 905.60 | 942.40 | 597.70 | 647.29 | 46.82 | 49.72 | 69.61 | 69.13 | 60.37 | 60.87 |
| Hexanal | 0.045 1 | Green, grass | 102.22 | 103.75 | 74.06 | 84.70 | 95.04 | 88.38 | 12.46 | 13.07 | 5.69 | 6.21 | 9.60 | 8.31 |
| Isoamyl acetate | 0.030 2 | Banana | 56.42 | 35.24 | 74.45 | 76.01 | 56.76 | 59.47 | 6.88 | 4.40 | 5.72 | 5.57 | 5.73 | 5.59 |
| Ethyl octanoate | 0.580 3 | Sweet, floral, fruity, pear | 1.01 | <1 | 1.62 | 1.69 | 1.07 | 1.16 | 0.12 | - | 0.12 | 0.12 | 0.11 | 0.11 |
| Ethyl decanoate | 0.200 3 | Floral | 2.63 | 1.83 | 4.40 | 4.67 | 2.96 | 3.30 | 0.32 | 0.23 | 0.33 | 0.34 | 0.30 | 0.31 |
| Ethyl hexanoate | 0.014 2 | Fruity, green apple, banana | 22.56 | 15.68 | 38.97 | 40.25 | 25.56 | 27.56 | 2.75 | 1.98 | 2.99 | 2.95 | 2.58 | 2.59 |
| Ethyl butanoate | 0.020 4 | Pineapple, apple | 14.07 | 11.80 | 17.03 | 17.77 | 14.18 | 14.23 | 1.71 | 1.49 | 1.30 | 1.30 | 1.43 | 1.34 |
| Isoamyl lactate | 0.0016 5 | Fruit, apple, banana | 19.79 | 26.65 | 19.69 | 17.05 | 14.42 | 13.97 | 2.41 | 3.36 | 1.51 | 1.25 | 1.45 | 1.31 |
| Ethyl-3-methylbutanoate | 0.003 4 | Fruity, pineapple | 8.85 | 9.46 | 7.59 | 8.31 | 9.71 | 8.45 | 1.07 | 1.19 | 0.58 | 0.61 | 0.98 | 0.79 |
| Ethyl-2-methylbutanoate | 0.018 4 | Apple | 1.02 | 1.21 | <1 | 1.06 | 1.23 | 1.08 | 0.12 | 0.15 | - | 0.07 | 0.12 | 0.10 |
| Ethyl hexadecanoate | 0.0015 6 | Fruity, wax | 5.53 | 5.15 | 10.87 | 14.27 | 6.08 | 8.12 | 0.67 | 0.65 | 0.83 | 1.04 | 0.61 | 0.76 |
| Phenylethyl alcohol | 14 2 | Floral, rose, honey | 2.35 | 2.39 | 2.13 | 2.38 | 2.42 | 2.28 | 0.28 | 0.30 | 0.16 | 0.17 | 0.24 | 0.21 |
| 2-Methyl-1-butanol | 30 7 | Whiskey, burnt, nail polish | 1.05 | 1.07 | <1 | 1.03 | 1.03 | 1.02 | 0.13 | 0.13 | - | 0.07 | 0.10 | 0.09 |
| 1-Octen-3-ol | 0.001 8 | Mushroom | 39.36 | 44.70 | 25.01 | 30.37 | 35.43 | 33.77 | 4.80 | 5.63 | 1.92 | 2.22 | 3.58 | 3.17 |
| Geraniol | 0.020 2 | Citrus, citric fruit | 24.64 | 2.01 | 4.09 | 4.19 | 2.77 | 2.88 | 3.00 | 0.25 | 0.31 | 0.31 | 0.28 | 0.27 |
| Linalool | 0.025 2 | Citrus, floral, sweet | 1.86 | 2.12 | 1.40 | 1.58 | 1.71 | 1.62 | 0.23 | 0.27 | 0.11 | 0.11 | 0.17 | 0.15 |
| ß-Damascenone | 0.00005 9 | Sweet, fruity, floral, honey | 132.80 | 136.00 | 114.00 | 115.40 | 122.00 | 148.80 | 16.19 | 17.13 | 8.76 | 8.46 | 12.32 | 13.99 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jagatić Korenika, A.-M.; Biloš, J.; Kozina, B.; Tomaz, I.; Preiner, D.; Jeromel, A. Effect of Different Reducing Agents on Aromatic Compounds, Antioxidant and Chromatic Properties of Sauvignon Blanc Wine. Foods 2020, 9, 996. https://doi.org/10.3390/foods9080996
Jagatić Korenika A-M, Biloš J, Kozina B, Tomaz I, Preiner D, Jeromel A. Effect of Different Reducing Agents on Aromatic Compounds, Antioxidant and Chromatic Properties of Sauvignon Blanc Wine. Foods. 2020; 9(8):996. https://doi.org/10.3390/foods9080996
Chicago/Turabian StyleJagatić Korenika, Ana-Marija, Josipa Biloš, Bernard Kozina, Ivana Tomaz, Darko Preiner, and Ana Jeromel. 2020. "Effect of Different Reducing Agents on Aromatic Compounds, Antioxidant and Chromatic Properties of Sauvignon Blanc Wine" Foods 9, no. 8: 996. https://doi.org/10.3390/foods9080996
APA StyleJagatić Korenika, A.-M., Biloš, J., Kozina, B., Tomaz, I., Preiner, D., & Jeromel, A. (2020). Effect of Different Reducing Agents on Aromatic Compounds, Antioxidant and Chromatic Properties of Sauvignon Blanc Wine. Foods, 9(8), 996. https://doi.org/10.3390/foods9080996








