Beer and Consumer Response Using Biometrics: Associations Assessment of Beer Compounds and Elicited Emotions
Abstract
1. Introduction
2. Materials and Methods
2.1. Physicochemical Characterization
2.2. Characterization of Simple Sugars by HPLC-Refractive Index
2.3. Determination of Bitterness
2.4. Characterization of Iso-α-Acids
2.5. Hordenine Determination by UPLC-MS/MS
2.6. Consumer Sensory Evaluation and Biometrics
 , (x) relaxed
, (x) relaxed  , (xi) winking face
, (xi) winking face  , (xii) stuck out tongue
, (xii) stuck out tongue  , (xiii) flushed
, (xiii) flushed  , (xiv) rage
, (xiv) rage  , (xv) smirk
, (xv) smirk  , and (xvi) disappointed
, and (xvi) disappointed  [39].
[39].2.7. Statistical Analysis
3. Results
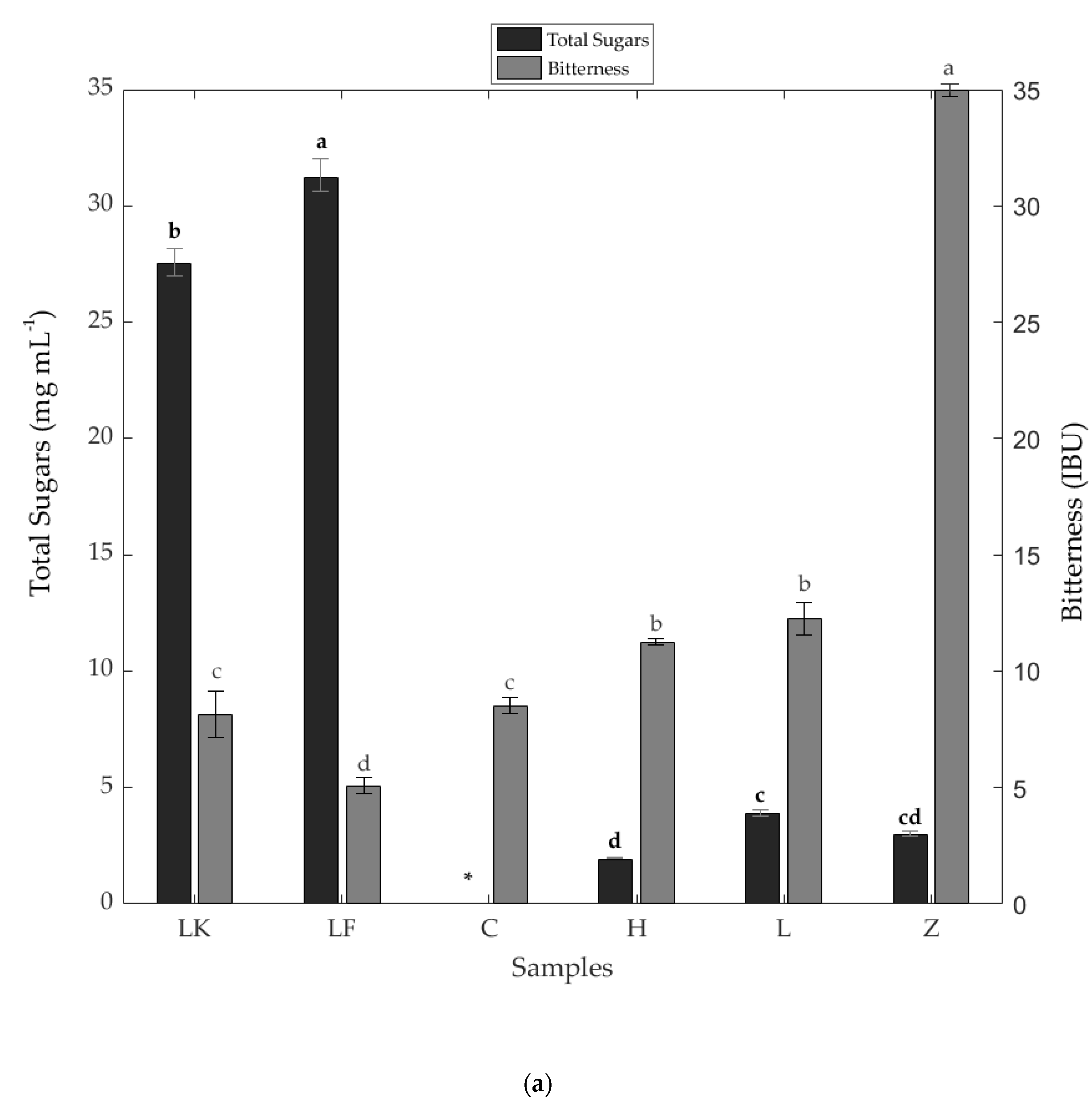
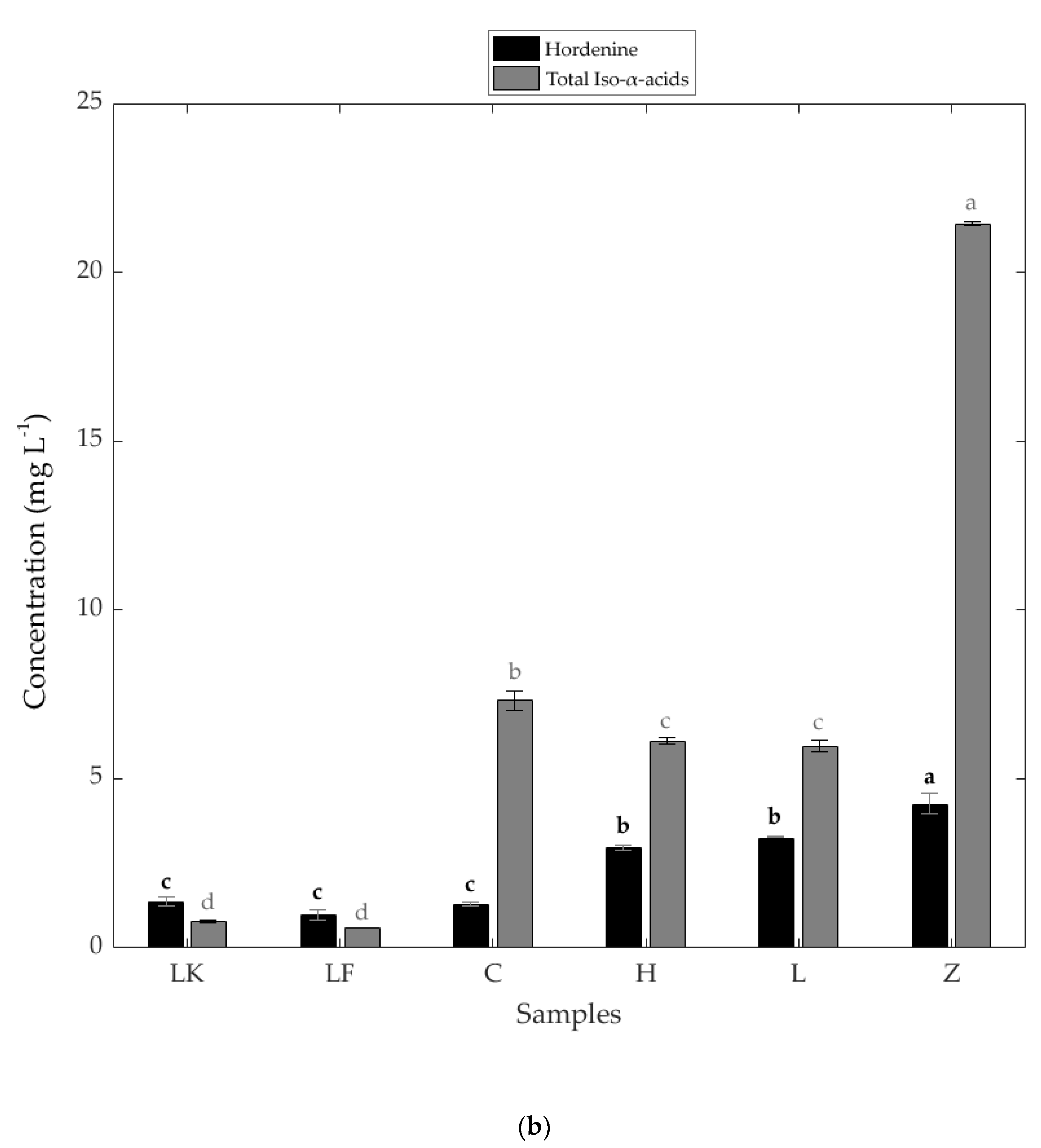
3.1. Physicochemical Results
3.2. Consumer Sensory Evaluation and Biometrics
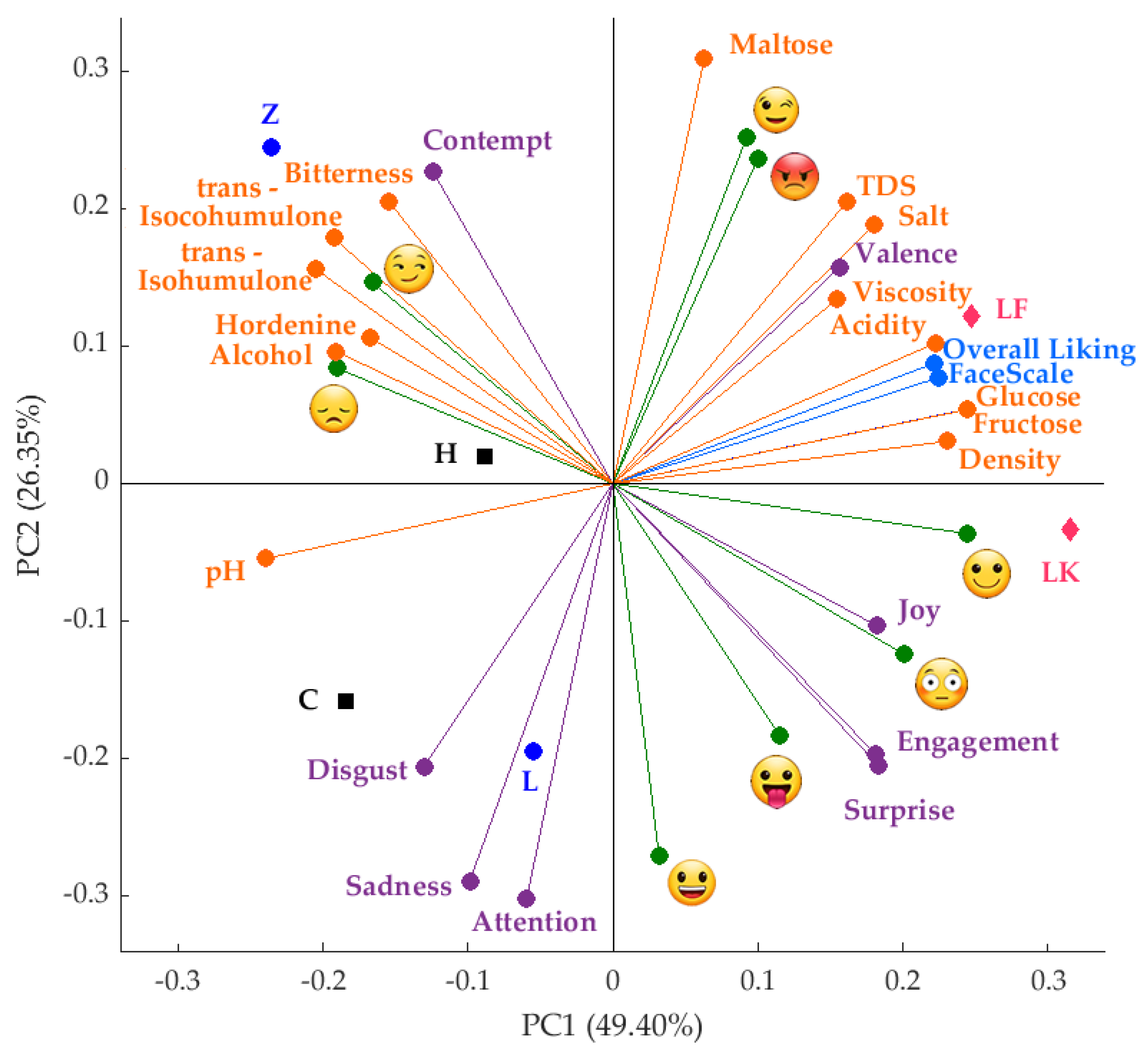
 (FL = 0.24), glucose (FL = 0.24), fructose (FL = 0.24) and density (FL = 0.23) represented PC1 on the positive side of the axis; while pH (FL = −0.24), trans-Isohumulone (FL = −0.20) and trans-Isocohumulone (FL = −0.19) characterized it on the negative side. On the other hand, PC2 was represented by maltose (FL = 0.31), winking face
(FL = 0.24), glucose (FL = 0.24), fructose (FL = 0.24) and density (FL = 0.23) represented PC1 on the positive side of the axis; while pH (FL = −0.24), trans-Isohumulone (FL = −0.20) and trans-Isocohumulone (FL = −0.19) characterized it on the negative side. On the other hand, PC2 was represented by maltose (FL = 0.31), winking face  (FL = 0.25) and rage
(FL = 0.25) and rage  (FL = 0.24) on the positive side; while attention (FL = −0.30), sadness (FL = −0.29), and smiley
(FL = 0.24) on the positive side; while attention (FL = −0.30), sadness (FL = −0.29), and smiley  (FL = −0.27) represented it on the negative side of the axis. Sugars such as fructose and glucose were positively related to overall liking, FaceScale and relaxed, with the spontaneous fermentation beers (LK and LF) associated with those components. On the contrary, hordenine presented a negative relationship with the latter descriptors and a positive relationship with alcohol content, iso-alpha acids, bitterness, smirk
(FL = −0.27) represented it on the negative side of the axis. Sugars such as fructose and glucose were positively related to overall liking, FaceScale and relaxed, with the spontaneous fermentation beers (LK and LF) associated with those components. On the contrary, hordenine presented a negative relationship with the latter descriptors and a positive relationship with alcohol content, iso-alpha acids, bitterness, smirk  , and disappointed
, and disappointed  , and beers such as H (bottom fermentation) and Z (top fermentation) were associated with these variables.
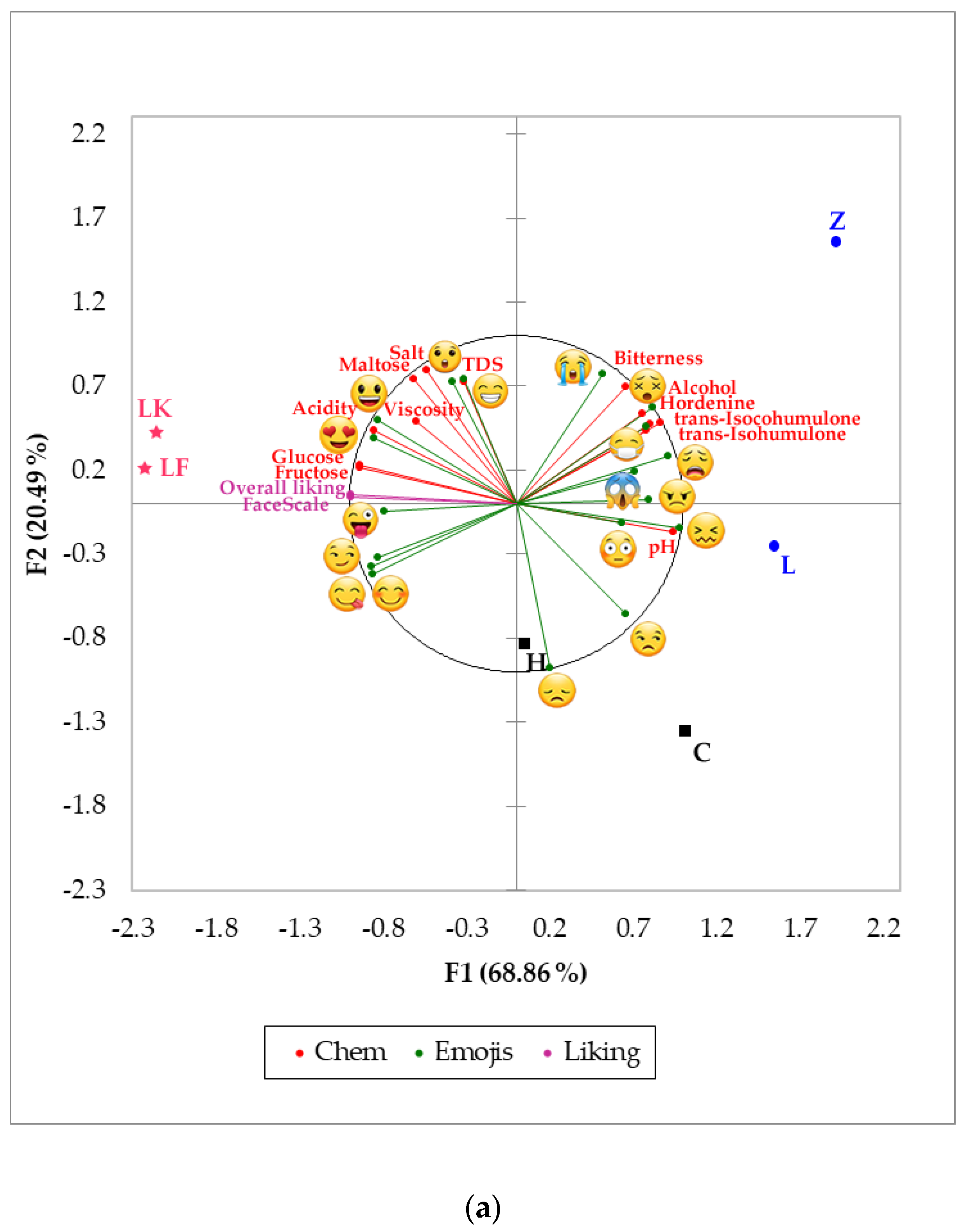
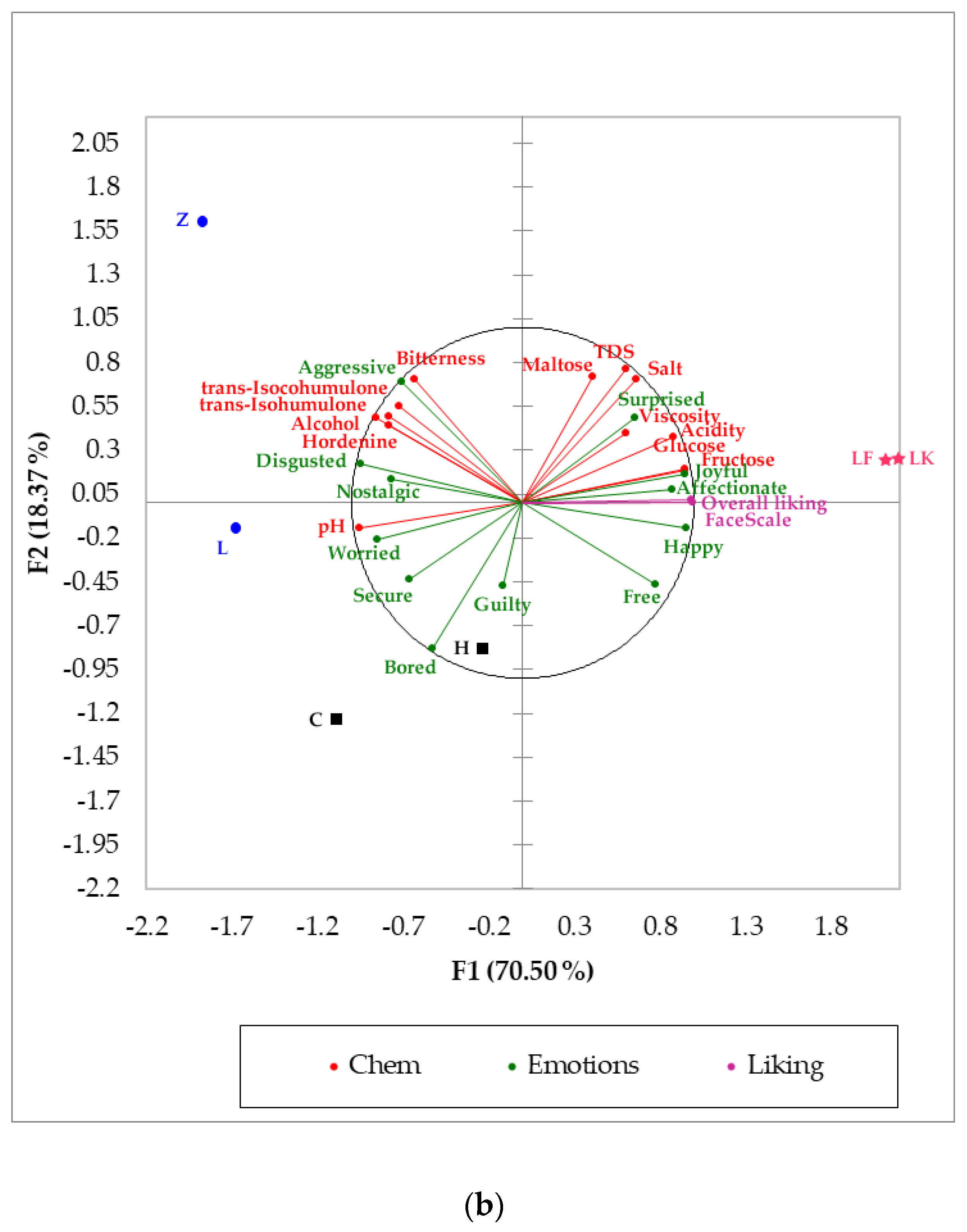
, and beers such as H (bottom fermentation) and Z (top fermentation) were associated with these variables. (FL = 1.13), pH (FL = 0.94), angry
(FL = 1.13), pH (FL = 0.94), angry  (FL = 0.88), and alcohol content (FL = 0.87) on the positive side of the axis, and by overall liking (FL = −0.99), FaceScale (FL = −0.99), glucose (FL = −0.94) and fructose (FL = −0.94) on the negative side. On the other hand, F2 was represented by crying
(FL = 0.88), and alcohol content (FL = 0.87) on the positive side of the axis, and by overall liking (FL = −0.99), FaceScale (FL = −0.99), glucose (FL = −0.94) and fructose (FL = −0.94) on the negative side. On the other hand, F2 was represented by crying  (FL = 1.67), TDS (FL = 0.80), salt (FL = 0.74) and maltose (FL = 0.73) on the positive side, and by disappointed
(FL = 1.67), TDS (FL = 0.80), salt (FL = 0.74) and maltose (FL = 0.73) on the positive side, and by disappointed  (FL = −0.50) and unamused
(FL = −0.50) and unamused  (FL = −0.29) on the negative side of the axis. Hordenine was positively related to alcohol content, iso-alpha acids, bitterness, and emojis such as sick
(FL = −0.29) on the negative side of the axis. Hordenine was positively related to alcohol content, iso-alpha acids, bitterness, and emojis such as sick  , dizzy
, dizzy  and weary
and weary  , with top fermentation beer samples such as Z and L associated with those variables. In contrast, overall liking and FaceScale had a positive relationship with glucose, fructose, winking face with tongue
, with top fermentation beer samples such as Z and L associated with those variables. In contrast, overall liking and FaceScale had a positive relationship with glucose, fructose, winking face with tongue  , and love
, and love  ; spontaneous fermentation samples LK and LF were most represented by these descriptors.
; spontaneous fermentation samples LK and LF were most represented by these descriptors.4. Discussion
 , love
, love  , winking face with tongue
, winking face with tongue  , affectionate, and FaceScale in both subconscious and conscious responses (emojis and emotion-terms). This coincides with findings by Kim et al. [46], who reported that samples of beverages and biscuits with the highest sugar content elicited positive emotions such as affectionate, pleased, joyful, glad, and happy. On the other hand, bitterness has been associated with rejection due to genetic factors and the innate relationship of bitter products with poisonous compounds [24,47,48]. Overall taste liking is the result of the intrinsic balance among the basic tastes that are sensed by the receptors located in the gustative system [49]. Individual taste compounds can elicit discrete sensations in consumers. However, different tastes can interact with each other, which can result in suppression or enhancement effects of certain perceptions [50,51]. For instance, minor concentrations of sugar can enhance the sourness of citric acid solutions; or slight concentrations of salt can enhance the sweetness of sugar solutions. The opposite can also occur as slight concentrations of quinine (a bitter compound) mixed with saccharides can suppress the sweetness of the solutions [52,53]. This can potentially explain the overall taste perception by the consumers in the present study. As the sugar content of the spontaneous fermentation beers was higher compared to the other samples, the bitterness perception of those beers was somewhat suppressed, which produced higher hedonic and emotional responses. This effect can be observed for both responses (conscious and subconscious) measured in this study, as the sweet taste was the main factor responsible for the overall satisfaction of consumers.
, affectionate, and FaceScale in both subconscious and conscious responses (emojis and emotion-terms). This coincides with findings by Kim et al. [46], who reported that samples of beverages and biscuits with the highest sugar content elicited positive emotions such as affectionate, pleased, joyful, glad, and happy. On the other hand, bitterness has been associated with rejection due to genetic factors and the innate relationship of bitter products with poisonous compounds [24,47,48]. Overall taste liking is the result of the intrinsic balance among the basic tastes that are sensed by the receptors located in the gustative system [49]. Individual taste compounds can elicit discrete sensations in consumers. However, different tastes can interact with each other, which can result in suppression or enhancement effects of certain perceptions [50,51]. For instance, minor concentrations of sugar can enhance the sourness of citric acid solutions; or slight concentrations of salt can enhance the sweetness of sugar solutions. The opposite can also occur as slight concentrations of quinine (a bitter compound) mixed with saccharides can suppress the sweetness of the solutions [52,53]. This can potentially explain the overall taste perception by the consumers in the present study. As the sugar content of the spontaneous fermentation beers was higher compared to the other samples, the bitterness perception of those beers was somewhat suppressed, which produced higher hedonic and emotional responses. This effect can be observed for both responses (conscious and subconscious) measured in this study, as the sweet taste was the main factor responsible for the overall satisfaction of consumers. (Figure 2), dizzy
(Figure 2), dizzy  , sick
, sick  , weary
, weary  (Figure 3a), disgusted, and aggressive (Figure 3b). This may be due to two main factors: i) the higher sugar concentration in beers LF and LK, which had a higher effect on consumers, and ii) the time of the sensory session, which may not have been long enough to increase hordenine concentration in the bloodstream significantly. Hence, since the effects of hordenine may be delayed, a sensory tasting session, including several sample beers, may not be appropriated to study the carry-over effects. This may be overcome by conducting further research allowing more time between beers for emotional assessments, so that there is enough hordenine level in the blood to more accurately assess the elicited emotional responses. Moreover, by comparing similar beer styles with alcoholic and non-alcoholic beers, it may render more information on the effects of hordenine and other compounds alone.
(Figure 3a), disgusted, and aggressive (Figure 3b). This may be due to two main factors: i) the higher sugar concentration in beers LF and LK, which had a higher effect on consumers, and ii) the time of the sensory session, which may not have been long enough to increase hordenine concentration in the bloodstream significantly. Hence, since the effects of hordenine may be delayed, a sensory tasting session, including several sample beers, may not be appropriated to study the carry-over effects. This may be overcome by conducting further research allowing more time between beers for emotional assessments, so that there is enough hordenine level in the blood to more accurately assess the elicited emotional responses. Moreover, by comparing similar beer styles with alcoholic and non-alcoholic beers, it may render more information on the effects of hordenine and other compounds alone.5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gonzalez Viejo, C.; Fuentes, S.; Li, G.; Collmann, R.; Condé, B.; Torrico, D. Development of a robotic pourer constructed with ubiquitous materials, open hardware and sensors to assess beer foam quality using computer vision and pattern recognition algorithms: RoboBEER. Food Res. Int. 2016, 89, 504–513. [Google Scholar] [CrossRef]
- Bamforth, C.; Russell, I.; Stewart, G. Beer: A Quality Perspective; Academic Press: Burlington, MA, USA, 2011. [Google Scholar]
- Gonzalez Viejo, C.; Fuentes, S.; Torrico, D.; Howell, K.; Dunshea, F. Assessment of beer quality based on foamability and chemical composition using computer vision algorithms, near infrared spectroscopy and machine learning algorithms. J. Sci. Food Agric. 2018, 98, 618–627. [Google Scholar] [CrossRef]
- De Keukeleire, D. Fundamentals of beer and hop chemistry. Quim. Nova 2000, 23, 108–112. [Google Scholar] [CrossRef]
- Bamforth, C. 125th Anniversary Review: The Non-Biological Instability of Beer. J. Inst. Brew. 2011, 117, 488–497. [Google Scholar] [CrossRef]
- Briggs, D.E.; Hough, J.S.; Stevens, R.; Young, T.W. Malting and Brewing Science: Hopped Wort and Beer; Kluwer Academic/Plenum Publishers: St Edmundsbury, UK, 1982; Volume 2. [Google Scholar]
- Pelletier, S.W. The nature and definition of an alkaloid. Alkaloids: Chem. Biol. Perspect. 1983, 1, 1–31. [Google Scholar]
- Bart, R. The Chemistry of Beer: The Science in the Suds; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar] [CrossRef]
- Wong, S.; Lua, P. Chocolate: Food for moods. Malays. J. Nutr. 2011, 17, 259–269. [Google Scholar] [PubMed]
- Mitchell, E.; Slettenaar, M.; Vd Meer, N.; Transler, C.; Jans, L.; Quadt, F.; Berry, M. Differential contributions of theobromine and caffeine on mood, psychomotor performance and blood pressure. Physiol. Behav. 2011, 104, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A. Xylitol: Sweet and healthy. Pacific J. Energy Med. 2008, 2, 58–64. [Google Scholar]
- Verna, R. The history and science of chocolate. Malays. J. Pathol. 2013, 35, 111. [Google Scholar]
- Pietraszek, M.; Urano, T.; Sumioshi, K.; Serizawa, K.; Takahashi, S.; Takada, Y.; Takada, A. Alcohol-induced depression: Involvement of serotonin. Alcohol Alcohol. 1991, 26, 155–159. [Google Scholar] [CrossRef]
- Duncan, M.; Smythe, G. Salsolinol and dopamine in alcoholic beverages. Lancet 1982, 319, 904–905. [Google Scholar] [CrossRef]
- Sommer, T.; Hübner, H.; El Kerdawy, A.; Gmeiner, P.; Pischetsrieder, M.; Clark, T. Identification of the beer component hordenine as food-derived dopamine D2 receptor agonist by virtual screening a 3D compound database. Sci. Rep. 2017, 7, 44201. [Google Scholar] [CrossRef] [PubMed]
- Brauers, G.; Steiner, I.; Daldrup, T. Quantification of the biogenic phenethylamine alkaloid hordenine by LC-MS/MS in beer. Toxichem Krimtech 2013, 80, 323–326. [Google Scholar]
- Steiner, I.; Brauers, G.; Temme, O.; Daldrup, T. A sensitive method for the determination of hordenine in human serum by ESI+ UPLC-MS/MS for forensic toxicological applications. Anal. Bioanal. Chem. 2016, 408, 2285–2292. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Perrine, D.M. Visions of the Night-Western Medicine Meets Peyote 1887–1899. Heffter Rev. Psychedelic Res. 2001, 2, 6–52. [Google Scholar]
- Baixauli, E. Happiness: Role of Dopamine and Serotonin on mood and negative emotions. Emerg. Med. 2017, 7, 350. [Google Scholar] [CrossRef]
- Oberlin, B.G.; Dzemidzic, M.; Tran, S.M.; Soeurt, C.M.; Albrecht, D.S.; Yoder, K.K.; Kareken, D.A. Beer flavor provokes striatal dopamine release in male drinkers: Mediation by family history of alcoholism. Neuropsychopharmacology 2013, 38, 1617–1624. [Google Scholar] [CrossRef]
- Gonzalez Viejo, C.; Fuentes, S.; Howell, K.; Torrico, D.; Dunshea, F. Robotics and computer vision techniques combined with non-invasive consumer biometrics to assess quality traits from beer foamability using machine learning: A potential for artificial intelligence applications. Food Control 2018. [Google Scholar] [CrossRef]
- Gonzalez Viejo, C.; Fuentes, S.; Torrico, D.; Howell, K.; Dunshea, F. Assessment of Beer Quality Based on a Robotic Pourer, Computer Vision, and Machine Learning Algorithms Using Commercial Beers. J. Food Sci. 2018, 83, 1381–1388. [Google Scholar] [CrossRef]
- Gonzalez Viejo, C.; Fuentes, S.; Howell, K.; Torrico, D.; Dunshea, F. Integration of non-invasive biometrics with sensory analysis techniques to assess acceptability of beer by consumers. Physiol. Behav. 2019, 200, 139–147. [Google Scholar] [CrossRef]
- Gonzalez Viejo, C.; Fuentes, S.; Torrico, D.; Godbole, A.; Dunshea, F. Chemical characterization of aromas in beer and their effect on consumers liking. Food Chem. 2019, 293, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Piggott, J. Alcoholic Beverages: Sensory Evaluation and Consumer Research; Elsevier Science: Philadelphia, PA, USA, 2011. [Google Scholar]
- Campbell-Platt, G. Food Science and Technology; Wiley: New York, NY, USA, 2017. [Google Scholar]
- Pielech-Przybylska, K.; Balcerek, M.; Dziekońska-Kubczak, U.; Pacholczyk-Sienicka, B.; Ciepielowski, G.; Albrecht, Ł.; Patelski, P. The role of Saccharomyces cerevisiae yeast and lactic acid bacteria in the formation of 2-propanol from acetone during fermentation of rye mashes obtained using thermal-pressure method of starch liberation. Molecules 2019, 24, 610. [Google Scholar] [CrossRef] [PubMed]
- Okafor, V.; Eboatu, A.; Anyalebechi, R.; Okafor, U. Comparative studies of the physicochemical properties of beers brewed with hop extracts and extracts from four selected tropical plants. J. Adv. Chem. Sci. 2016, 2, 382–386. [Google Scholar]
- Heredia-Olea, E.; Cortés-Ceballos, E.; Serna-Saldívar, S.O. Malting Sorghum with Aspergillus Oryzae Enhances Gluten-Free Wort Yield and Extract. J. Am. Soc. Brew. Chem. 2017, 75, 116–121. [Google Scholar] [CrossRef]
- Alonso-Gómez, L.A.; Heredia-Olea, E.; Serna-Saldivar, S.O.; Bello-Pérez, L.A. Whole unripe plantain (Musa paradisiaca L.) as raw material for bioethanol production. J. Sci. Food Agric. 2019, 99, 5784–5791. [Google Scholar]
- Martin, R.; Collazo, D.; Davis, J.; Hamp, F.; Huestis, S.; Jaskula-Goiris, B.; Jordan, J.; Juzeler, R.; Krug, S.; Lee, K. Reduced hazardous solvent use for determination of isohumulone bitterness units. J. Am. Soc. Brew. Chem. 2011, 69, 276–277. [Google Scholar]
- Vanhoenacker, G.; de Keukeleire, D.; Sandra, P. Analysis of iso-α-acids and reduced iso-α-acids in beer by direct injection and liquid chromatography with ultraviolet absorbance detection or with mass spectrometry. J. Chromatogr. A 2004, 1035, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Barber, L.; Arnberg, K.; Aron, P.; Mulqueen, S.; Raver, M.; Reffner, J.; Shellhammer, T.; Smith, B.; Porter, A. Determination of iso-α-acids in beer and wort by high-performance liquid chromatography. J. Am. Soc. Brew. Chem. 2011, 69, 288. [Google Scholar]
- Sommer, T.; Dlugash, G.; Hübner, H.; Gmeiner, P.; Pischetsrieder, M. Monitoring of the dopamine D2 receptor agonists hordenine and N-methyltyramine during the brewing process and in commercial beer samples. Food Chem. 2019, 276, 745–753. [Google Scholar] [CrossRef]
- Escobar, E.R.; Gamino, M.N.B.; Salazar, R.M.; Hernández, I.S.S.; Martínez, V.C.; Bautista, C.F.; López, M.d.L.G.; Buenabad, N.A.; Medina-Mora, M.E.; Velázquez, J.A.V. National trends in alcohol consumption in Mexico: Results of the National Survey on Drug, Alcohol and Tobacco Consumption 2016–2017. Salud Ment. 2018, 41, 7–15. [Google Scholar] [CrossRef]
- Garcia, D. En México, los regios son los que más consumen cerveza. Milenio 2019. [Google Scholar]
- King, S.C.; Meiselman, H.L. Development of a method to measure consumer emotions associated with foods. Food Qual. Prefer. 2010, 21, 168–177. [Google Scholar] [CrossRef]
- McDuff, D.; Mahmoud, A.; Mavadati, M.; Amr, M.; Turcot, J.; Kaliouby, R.E. AFFDEX SDK: A cross-platform real-time multi-face expression recognition toolkit. In Proceedings of the 2016 CHI Conference Extended Abstracts on Human Factors in Computing Systems, San Jose, CA, USA, 7–12 May 2016; pp. 3723–3726. [Google Scholar]
- De Keersmaecker, J. The mystery of lambic beer. Sci. Am. 1996, 275, 74–80. [Google Scholar] [CrossRef]
- Perozzi, C.; Beaune, H. The Naked Pint: An Unadulterated Guide to Craft Beer; Penguin Publishing Group: New York, NY, USA, 2012. [Google Scholar]
- Wiener, A.; Shudler, M.; Levit, A.; Niv, M.Y. BitterDB: A database of bitter compounds. Nucleic Acids Res. 2012, 40, D413–D419. [Google Scholar] [CrossRef] [PubMed]
- Oladokun, O.; Tarrega, A.; James, S.; Cowley, T.; Dehrmann, F.; Smart, K.; Cook, D.; Hort, J. Modification of perceived beer bitterness intensity, character and temporal profile by hop aroma extract. Food Res. Int. 2016, 86, 104–111. [Google Scholar] [CrossRef]
- Alonso-Esteban, J.I.; Pinela, J.; Barros, L.; Ćirić, A.; Soković, M.; Calhelha, R.C.; Torija-Isasa, E.; de Cortes Sánchez-Mata, M.; Ferreira, I.C. Phenolic composition and antioxidant, antimicrobial and cytotoxic properties of hop (Humulus lupulus L.) Seeds. Ind. Crops Prod. 2019, 134, 154–159. [Google Scholar]
- Haseleu, G.; Intelmann, D.; Hofmann, T. Structure determination and sensory evaluation of novel bitter compounds formed from β-acids of hop (Humulus lupulus L.) upon wort boiling. Food Chem. 2009, 116, 71–81. [Google Scholar] [CrossRef]
- Kim, J.-Y.; Prescott, J.; Kim, K.-O. Emotional responses to sweet foods according to sweet liker status. Food Qual. Prefer. 2017, 59, 1–7. [Google Scholar] [CrossRef]
- Dinehart, M.; Hayes, J.; Bartoshuk, L.; Lanier, S.; Duffy, V. Bitter taste markers explain variability in vegetable sweetness, bitterness, and intake. Physiol. Behav. 2006, 87, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Thomas, S.D.; Zhang, J. Relaxation of selective constraint and loss of function in the evolution of human bitter taste receptor genes. Hum. Mol. Genet. 2004, 13, 2671–2678. [Google Scholar] [CrossRef]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices; Springer Science & Business Media: Berlin, Germany, 2010. [Google Scholar]
- Keast, R.S.; Breslin, P.A. An overview of binary taste–taste interactions. Food Qual. Prefer. 2003, 14, 111–124. [Google Scholar] [CrossRef]
- Torrico, D.D.; Prinyawiwatkul, W. Psychophysical effects of increasing oil concentrations on saltiness and bitterness perception of oil-in-water emulsions. J. Food Sci. 2015, 80, S1885–S1892. [Google Scholar] [CrossRef] [PubMed]
- Kiefer, S.W.; Lawrence, G.J. The sweet–bitter taste of alcohol: Aversion generalization to various sweet–quinine mixtures in the rat. Chem. Senses 1988, 13, 633–641. [Google Scholar] [CrossRef]
- Bartoshuk, L.; Cleveland, C. Mixtures of substances with similar tastes. A test of a psychophysical model of taste mixture interactions. Sens. Process. 1977, 1, 177. [Google Scholar]
| Beer Style | Beer Fermentation | Country of Origin | Label |
|---|---|---|---|
| Lambic Kriek | Spontaneous | Belgium | LK |
| Lambic Framboise | Spontaneous | Belgium | LF |
| Pale Lager | Bottom | Mexico | C |
| Pale Lager | Bottom | Mexico | H |
| Blonde Ale | Top | Belgium | L |
| Porter | Top | Poland | Z |
| Question/Descriptor | Answers (Options) | Scale |
|---|---|---|
| Overall liking (rated at the start of the test) | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Foam stability | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Foam height | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Bitterness | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Sweetness | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Acidity | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Aroma | Dislike extremely—Like extremely | 15-cm non-structured scale |
| How do you feel when tasting this sample? |  | Face Scale (0–100) |
| Check all emojis that depict how you feel when tasting this sample |  | Check all that apply (CATA) |
| Check all emotions that depict how you feel when tasting this sample | Active/Joyful/Aggressive/Bored/Affectionate/Disgusted/Free/Friendly/Happy/Adventurous/Guilty/Nostalgic/Calm/Pleasant/Satisfied/Secure/Surprised/Worried * | Check all that apply (CATA) |
| Overall liking (rated at the end of the test) | Dislike extremely—Like extremely | 15-cm non-structured scale |
| Sample | Color | Density (g mL−1) | Viscosity (mPa s) | pH | Titratable Acidity | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| L* | a* | b* | Hue | Chroma | YI | |||||
| LK | 36.70 d† ± 0.12 | 23.68 b ± 0.08 | 17.76 c ± 0.05 | 0.64 b ± 0.001 | 29.60 c ± 0.09 | 69.15 c ± 0.39 | 1.02 a ± 0.001 | 2.16 a ± 0.11 | 3.17 d ± 0.01 | 0.41 a ± 0.01 |
| LF | 29.67 e ± 0.08 | 26.54 a ± 0.16 | 20.16 b ± 0.32 | 0.65 b ± 0.005 | 33.33 b ± 0.32 | 97.11 b ± 1.79 | 1.03 a ± 0.002 | 1.73 bc ± 0.06 | 2.94 e ± 0.01 | 0.32 b ± 0.03 |
| C | 59.36 a ± 0.07 | −1.27 d ± 0.01 | 6.52 f ± 0.04 | −1.38 c ± 0.003 | 6.64 f ± 0.03 | 15.69 f ± 0.07 | 1.00 c ± 0.003 | 1.48 d ± 0.09 | 4.29 b ± 0.00 | 0.11 d ± 0.00 |
| H | 58.72 b ± 0.25 | −1.21 d ± 0.03 | 8.99 e ± 0.09 | −1.44 d ± 0.002 | 9.07 e ± 0.09 | 21.87 e ± 0.21 | 1.00 c ± 0.002 | 1.80 b ± 0.07 | 4.31 b ± 0.01 | 0.10 d ± 0.01 |
| L | 56.68 c ± 0.27 | −1.02 d ± 0.01 | 16.25 d ± 0.08 | −1.51 e ± 0.001 | 16.28 d ± 0.08 | 40.96 d ± 0.09 | 1.01 b ± 0.002 | 1.54 cd ± 0.02 | 4.24 c ± 0.01 | 0.11 d ± 0.00 |
| Z | 26.58 f ± 0.16 | 16.82 c ± 0.14 | 37.28 a ± 0.52 | 1.14 a ± 0.003 | 40.90 a ± 0.53 | 200.40 a ± 1.59 | 1.00 c ±0.003 | 1.80 b ± 0.00 | 4.42 a ± 0.01 | 0.17 c ± 0.01 |
| Sample | Simple Sugars (mg mL−1) | Salt (%) | Total Dissolved Solids (ppm) | Alcohol Content (%) | Iso-α-Acids (mg L−1) | |||
|---|---|---|---|---|---|---|---|---|
| Glucose | Fructose | Maltose | Trans-Isocohumulone | Trans-Isohumulone | ||||
| LK | 13.91 a* ± 0.24 | 12.56 b ± 0.31 | 1.06 c ± 0.04 | 0.10 a ± 0.00 | 1148.00 b ± 11.00 | 3.53 e ± <0.001 | 0.33 e ± 0.01 | 0.45 d ± 0.01 |
| LF | 14.32 a ± 0.62 | 13.51 a ± 0.01 | 3.40 a ± 0.07 | 0.10 a ± 0.00 | 1226.00 a ± 7.00 | 2.53 f ± <0.001 | 0.22 e ± 0.01 | 0.38 d ± 0.01 |
| C | ND | ND | ND | 0.05 e ± 0.00 | 658.00 f ± 9.61 | 4.62 d ± <0.001 | 3.44 b ± 0.08 | 3.91 b ± 0.22 |
| H | 0.60 c ± 0.00 | 0.50 d ± 0.00 | 0.79 d ± 0.03 | 0.06 d ± 0.00 | 738.00 e ± 4.04 | 4.97 c ± <0.001 | 2.81 c ± 0.00 | 3.27 c ± 0.11 |
| L | 1.87 b ± 0.06 | 2.04 c ± 0.08 | 0.00 e ± 0.00 | 0.07 c ± 0.00 | 898.67 d ± 5.55 | 6.68 b ± <0.001 | 2.60 d ± 0.05 | 3.35 c ± 0.12 |
| Z | ND | ND | 2.97 b ± 0.12 | 0.09 b ± 0.00 | 1100.33 c ± 26.36 | 9.47 a ± <0.001 | 10.95 a ± 0.04 | 10.46 a ± 0.08 |
| Sample | Overall Liking-Start | Foam Stability | Foam Height | Bitter | Sweet | Acidity | Aroma | Overall Liking-End |
|---|---|---|---|---|---|---|---|---|
| LK | 10.35 a* ± 0.56 | 10.20 a ± 0.38 | 8.31 b ± 0.38 | 11.06 a ± 0.42 | 9.53 a ± 0.46 | 11.37 a ± 0.50 | 9.53 a ± 0.44 | 10.73 a ± 0.50 |
| LF | 10.04 ab ± 0.47 | 11.14 a ± 0.52 | 11.16 a ± 0.54 | 11.85 a ± 0.46 | 9.56 a ± 0.52 | 10.76 a ±0.51 | 9.50 a ± 0.50 | 10.79 a ± 0.50 |
| C | 7.57 cd ± 0.55 | 6.79 b ± 0.52 | 6.28 c ± 0.59 | 8.92 bc ± 0.45 | 8.62 a ± 0.53 | 7.07 b ±0.51 | 7.54 bc ± 0.53 | 7.74 bc ±0.54 |
| H | 8.72 bc ± 0.49 | 10.58 a ± 0.39 | 10.60 a ± 0.40 | 8.76 bc ± 0.51 | 9.51 a ± 0.51 | 7.35 b ±0.47 | 8.31 ab ± 0.51 | 9.07 b ± 0.49 |
| L | 6.69 d ± 0.58 | 10.63 a ± 0.43 | 10.32 a ± 0.41 | 7.78 c ± 0.57 | 6.61 b ± 0.58 | 6.91 b ±0.52 | 7.03 bc ± 0.55 | 6.90 c ± 0.63 |
| Z | 7.65 cd ± 0.63 | 10.46 a ± 0.51 | 10.83 a ± 0.44 | 9.48 b ± 0.60 | 6.83 b ± 0.61 | 7.59 b ±0.59 | 6.73 c ± 0.57 | 7.34 c ± 0.65 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez Viejo, C.; Villarreal-Lara, R.; Torrico, D.D.; Rodríguez-Velazco, Y.G.; Escobedo-Avellaneda, Z.; Ramos-Parra, P.A.; Mandal, R.; Pratap Singh, A.; Hernández-Brenes, C.; Fuentes, S. Beer and Consumer Response Using Biometrics: Associations Assessment of Beer Compounds and Elicited Emotions. Foods 2020, 9, 821. https://doi.org/10.3390/foods9060821
Gonzalez Viejo C, Villarreal-Lara R, Torrico DD, Rodríguez-Velazco YG, Escobedo-Avellaneda Z, Ramos-Parra PA, Mandal R, Pratap Singh A, Hernández-Brenes C, Fuentes S. Beer and Consumer Response Using Biometrics: Associations Assessment of Beer Compounds and Elicited Emotions. Foods. 2020; 9(6):821. https://doi.org/10.3390/foods9060821
Chicago/Turabian StyleGonzalez Viejo, Claudia, Raúl Villarreal-Lara, Damir D. Torrico, Yaressi G. Rodríguez-Velazco, Zamantha Escobedo-Avellaneda, Perla A. Ramos-Parra, Ronit Mandal, Anubhav Pratap Singh, Carmen Hernández-Brenes, and Sigfredo Fuentes. 2020. "Beer and Consumer Response Using Biometrics: Associations Assessment of Beer Compounds and Elicited Emotions" Foods 9, no. 6: 821. https://doi.org/10.3390/foods9060821
APA StyleGonzalez Viejo, C., Villarreal-Lara, R., Torrico, D. D., Rodríguez-Velazco, Y. G., Escobedo-Avellaneda, Z., Ramos-Parra, P. A., Mandal, R., Pratap Singh, A., Hernández-Brenes, C., & Fuentes, S. (2020). Beer and Consumer Response Using Biometrics: Associations Assessment of Beer Compounds and Elicited Emotions. Foods, 9(6), 821. https://doi.org/10.3390/foods9060821









