High-Throughput 16S rRNA Gene Sequencing of Butter Microbiota Reveals a Variety of Opportunistic Pathogens
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Plating and Microbial Enrichment
2.3. DNA Isolation
2.4. DNA Barcoding
2.5. High-Throughput Sequencing
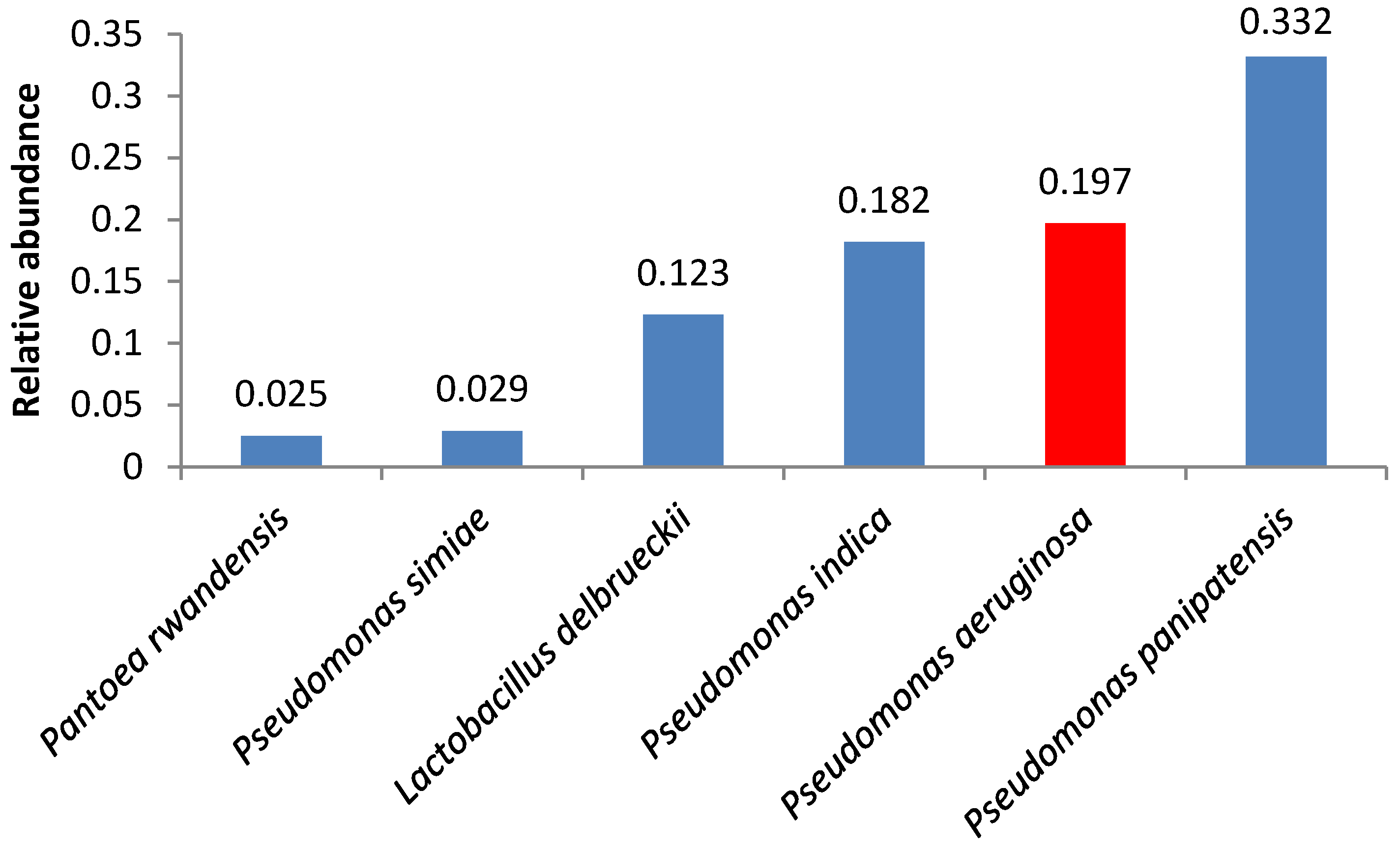
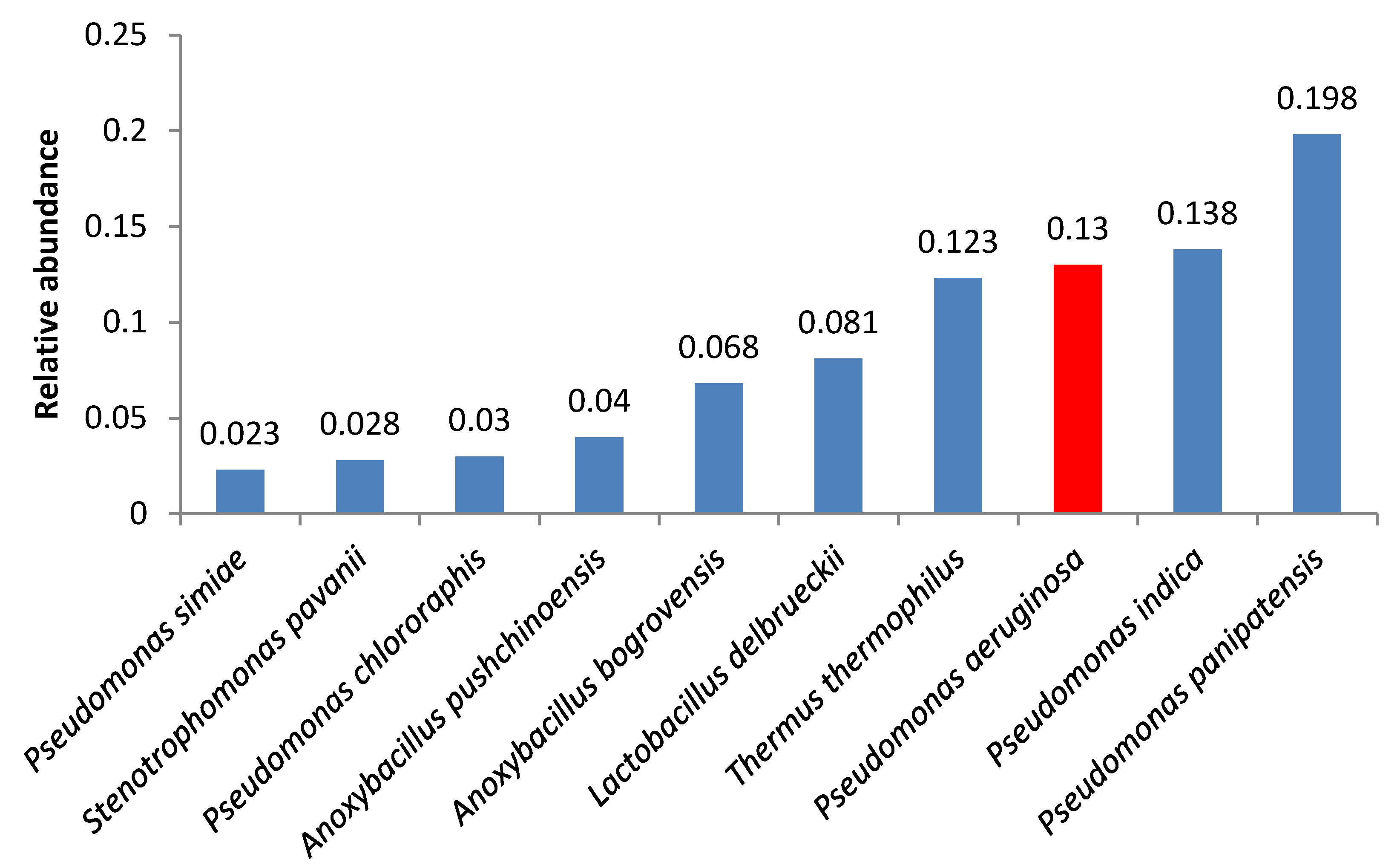
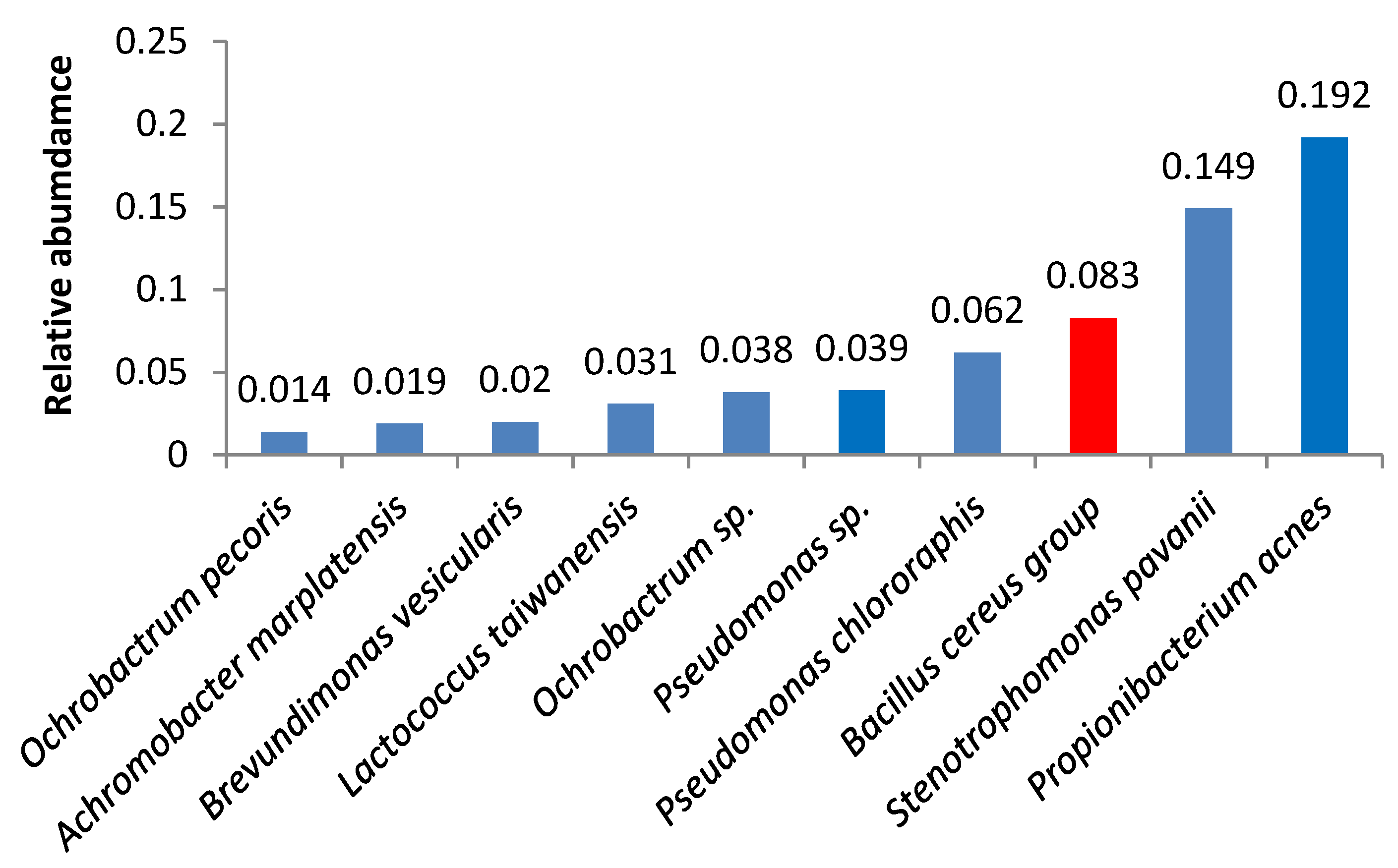
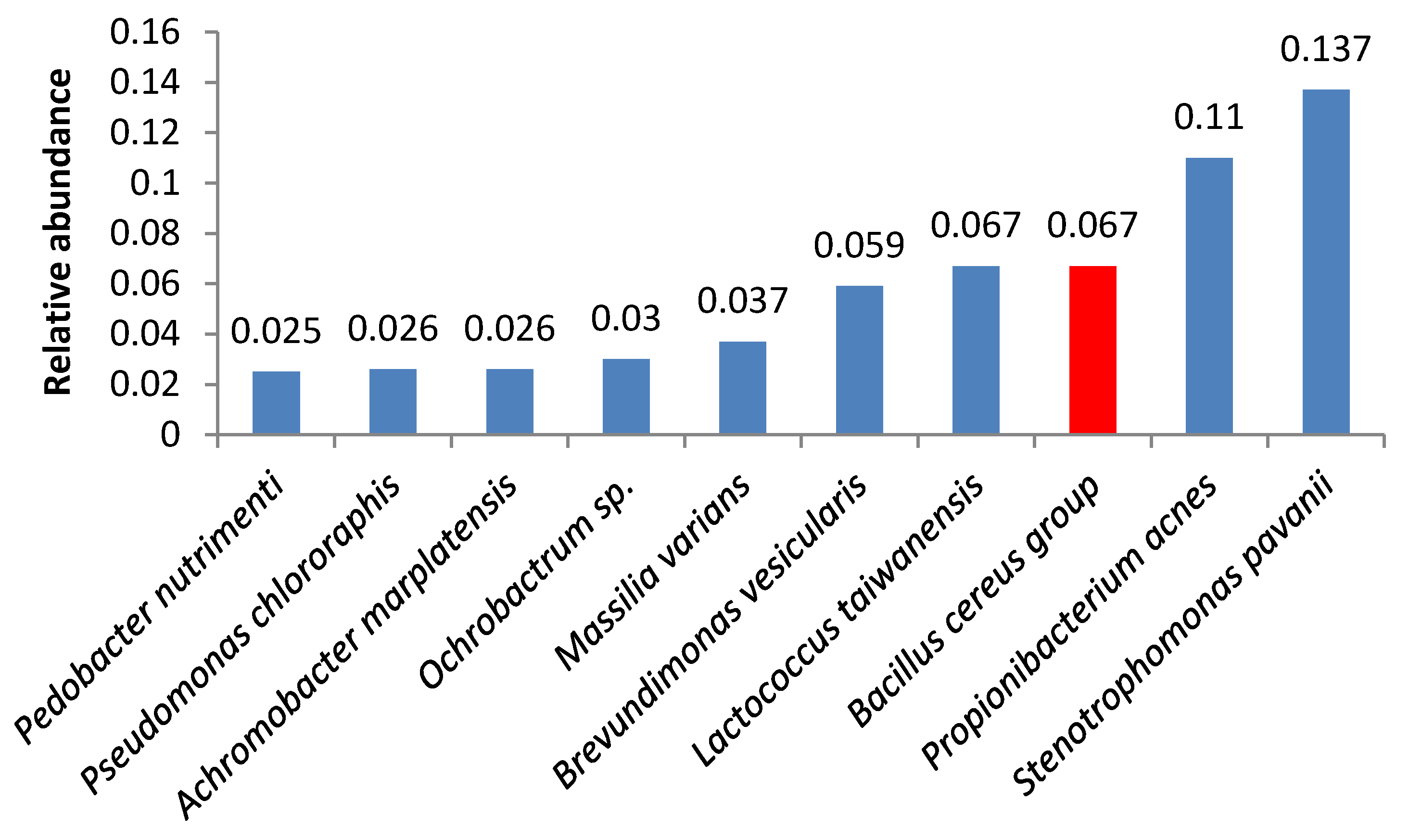
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Oliver, S.P.; Jayarao, B.M.; Almeida, R.A. Foodborne pathogens in milk and the dairy farm environment: Food safety and public health implications. Foodborne Pathog. Dis. 2005, 2, 115–129. [Google Scholar] [CrossRef]
- Quigley, L.; O’Sullivan, O.; Stanton, C.; Beresford, T.P.; Ross, R.P.; Fitzgerald, G.F.; Cotter, P.D. The complex microbiota of raw milk. FEMS Microbiol. Rev. 2013, 37, 664–698. [Google Scholar] [CrossRef]
- Headrick, M.L.; Korangy, S.; Bean, N.H.; Angulo, F.J.; Altekruze, S.F.; Potter, M.E.; Klontz, K.C. The epidemiology of raw milk-associated foodborne disease outbreaks reported in the United States, 1973 through 1992. Am. J. Public Health 1998, 88, 1219–1221. [Google Scholar] [CrossRef]
- Tibana, A.; Warnken, M.M.; Nunes, M.; Ricciardi, I.D.; Noleto, A.L.S. Occurrence of Yersinia species in raw and pasteurized milk in Rio de Janeiro, Brazil. Int. J. Food Microbiol. 1987, 50, 580–583. [Google Scholar] [CrossRef]
- Gitter, M.; Bradley, R.; Blampied, P.H. Listeria monocytogenes infection in bovine mastitis. Vet. Rec. 1980, 107, 390–393. [Google Scholar] [CrossRef]
- Syromyatnikov, M.Y.; Kokina, A.V.; Savinkova, O.V.; Panevina, A.V.; Solodskikh, S.A.; Orlova, M.V.; Grabovich, M.Y.; Starkov, A.A.; Popov, V.N. Study of the microbiological composition of dairy products and mayonaise using DNA barcoding and metabarcoding. Foods Raw Mater. 2018, 6, 144–153. [Google Scholar] [CrossRef]
- Cremonesi, P.; Ceccarani, C.; Curone, G.; Severgnini, M.; Pollera, C.; Bronzo, V.; Riva, F.; Addis, M.F.; Filipe, J.; Amadori, M.; et al. Milk microbiome diversity and bacterial group prevalence in a comparison between healthy Holstein Friesian and Rendena cows. PLoS ONE 2018, 13, e0205054. [Google Scholar] [CrossRef]
- Pang, M.; Xie, X.; Bao, H.; Sun, L.; He, T.; Zhao, H.; Zhou, Y.; Zhang, L.; Zhang, H.; Wei, R.; et al. Insights into the Bovine Milk Microbiota in Dairy Farms with Different Incidence Rates of Subclinical Mastitis. Front. Microbiol. 2018, 9, 2379. [Google Scholar] [CrossRef] [PubMed]
- Meshref, A.M. Microbiological quality and safety of cooking butter in Beni-Suef governorate-Egypt. Afr. Health Sci. 2010, 10, 193–198. [Google Scholar] [PubMed]
- Idoui, T.; Rechak, H.; Zabayou, N. Microbial quality, physicochemical characteristics and fatty acid composition of a traditional butter made from goat milk. Ann. Food Sci. Technol. 2013, 14, 108–114. [Google Scholar]
- Yoshida, N.; Yagi, K.; Sato, D.; Watanabe, N.; Kuroishi, T.; Nishimoto, K.; Yanagida, A.; Katsuragi, T.; Kanagawa, T.; Kurane, R.; et al. Bacterial communities in petroleum oil in stockpiles. J. Biosci. Bioeng. 2005, 99, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Ghasemloy Incheh, K.H.; Hassanzadazar, H.; Forouzan, S.H.; Banafshehchin, E.I.; Mozafarian, E.I.; Aminzare, M.; Hashemi, M. A survey on the quality of traditional butters produced in West Azerbaijan province, Iran. Int. Food Res. J. 2017, 24, 327–332. [Google Scholar]
- Yilmaz, Ö. Identification of microflora in butter samples from Turkey by using the microbial identification system S. Asian J. Chem. 2009, 21, 3257–3262. [Google Scholar]
- Syromyatnikov, M.Y.; Kiryanova, S.V.; Popov, V.N. Development and validation of a TaqMan RT-PCR method for identification of mayonnaise spoilage yeast Pichia kudriavzevii. AMB Express 2018, 8, 186. [Google Scholar] [CrossRef]
- Mayo, B. Impact of Next Generation Sequencing Techniques in Food Microbiology. Curr. Genom. 2014, 15, 293–309. [Google Scholar] [CrossRef]
- Ferrario, C.; Lugli, G.A.; Ossiprandi, M.C.; Turroni, F.; Milani, C.; Duranti, S.; Mancabelli, L.; Mangifesta, M.; Alessandri, G.; Sinderen, D.; et al. Next generation sequencing-based multigene panel for high throughput detection of food-borne pathogens. Int. J. Food. Microbiol. 2017, 256, 20–29. [Google Scholar] [CrossRef]
- Techo, S.; Shiwa, Y.; Tanaka, N.; Fujita, N.; Miyashita, M.; Shibata, C.; Booncharoen, A.; Tanasupawat, S. Enterococcus florum sp. nov., isolated from a cotton flower (Gossypium hirsutum L.). Int. J. Syst. Evol. Microbiol. 2019, 69, 2506–2513. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Edgar, R.C.; Flyvbjerg, H. Error filtering, pair assembly and error correction for next-generation sequencing reads. Bioinformatics 2015, 31, 3476–3482. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef]
- Yong, B.; Wang, Y.; Qian, P. Sensitivity and correlation of hypervariable regions in 16S rRNA genes in phylogenetic analysis. BMC Bioinform. 2016, 17, 135. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Holmes, S.P. Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME J. 2017, 11, 2639–2643. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; Proctor, D.; Relman, D.; Fukuyama, J.; Holmes, S. Reproducible research workflow in r for the analysis of personalized human microbiome data. Pac. Symp. Biocomput. 2016, 21, 183–194. [Google Scholar] [PubMed]
- Macheriotou, L.; Guilini, K.; Bezerra, T.N.; Tytgat, B.; Nguyen, D.T.; Nguyen, T.X.P.; Noppe, F.; Armenteros, M.; Boufahja, F.; Rigaux, A.; et al. Metabarcoding free-living marine nematodes using curated 18S and CO1 reference sequence databases for species-level taxonomic assignments. Ecol. Evol. 2019, 9, 1211–1226. [Google Scholar] [CrossRef]
- Caruso, V.; Song, X.; Asquith, M.; Karstens, L. Performance of microbiome sequence inference methods in environments with varying biomass. mSystems 2019, 4, e00163-18. [Google Scholar] [CrossRef]
- Edgar, R.C. Updating the 97% identity threshold for 16S ribosomal RNA OTUs. Bioinformatics 2018, 34, 2371–2375. [Google Scholar] [CrossRef]
- Rasko, D.A.; Altherr, M.R.; Han, C.S.; Ravel, J. Genomics of the Bacillus cereus group of organisms. FEMS Microbiol. Rev. 2005, 29, 303–329. [Google Scholar] [CrossRef]
- Liu, Y.; Lai, Q.; Göker, M.; Meier-Kolthoff, J.P.; Wang, M.; Sun, Y.; Wang, L.; Shao, Z. Genomic insights into the taxonomic status of the Bacillus cereus group. Sci. Rep. 2015, 5, 14082. [Google Scholar] [CrossRef]
- Guinebretière, M.H.; Velge, P.; Couvert, O.; Carlin, F.; Debuyser, M.L.; Nguyen-The, C. Ability of Bacillus cereus group strains to cause food poisoning varies according to phylogenetic affiliation (groups I to VII) rather than species affiliation. J. Clin. Microbiol. 2010, 48, 3388–33891. [Google Scholar] [CrossRef]
- Granum, P.E.; Lund, T. Bacillus cereus and its food poisoning toxins. FEMS Microbiol. Lett. 1994, 157, 223–228. [Google Scholar] [CrossRef]
- Agata, N.; Ohta, M.; Mori, M.; Isobe, M. A novel dodecadepsipeptide, cereulide, is an emetic toxin of Bacillus cereus. FEMS Microbiol. Lett. 1995, 129, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Gellatly, S.L.; Hancock, R.E. Pseudomonas aeruginosa: New insights into pathogenesis and host defenses. Pathog. Dis. 2013, 67, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Lambert, P.A. Mechanisms of antibiotic resistance in Pseudomonas aeruginosa. J. R. Soc. Med. 2002, 41, 22–26. [Google Scholar]
- Yan, Q.Q.; Condell, O.; Power, K.; Butler, F.; Tall, B.D.; Fanning, S. Cronobacter species (formerly known as Enterobacter sakazakii) in powdered infant formula: A review of our current understanding of the biology of this bacterium. J. Appl. Microbiol. 2012, 113, 1–15. [Google Scholar] [CrossRef]
- McMullan, R.; Menon, V.; Beukers, A.G.; Jensen, S.O.; van Hal, S.J.; Davis, R. Cronobacter sakazakii Infection from Expressed Breast Milk, Australia. Emerg. Infect. Dis. 2018, 24, 393–394. [Google Scholar] [CrossRef]
- Lang, E.; Guyot, S.; Peltier, C.; Alvarez-Martin, P.; Perrier-Cornet, J.M.; Gervais, P. Cellular injuries in Cronobacter sakazakii CIP 103183T and Salmonella enteric exposed to drying and subsequent heat treatment in milk powder. Front. Microbiol. 2018, 9, 475. [Google Scholar] [CrossRef]
- Osaili, T.M.; Shaker, R.R.; Al-Haddaq, M.S.; Al-Nabulsi, A.A.; Holley, R.A. Heat resistance of Cronobacter species (Enterobacter sakazakii) in milk and special feeding formula. J. Appl. Microbiol. 2009, 107, 928–935. [Google Scholar] [CrossRef]
- Ueda, S. Occurrence of Cronobacter spp. in Dried Foods, Fresh Vegetables and Soil. Biocontrol. Sci. 2017, 22, 55–59. [Google Scholar] [CrossRef]
- Akineden, Ö.; Murata, K.J.; Gross, M.; Usleber, E. Microbiological quality of raw dried pasta from the German market, with special emphasis on Cronobacter species. J. Food. Sci. 2015, 80, 2860–2867. [Google Scholar] [CrossRef]
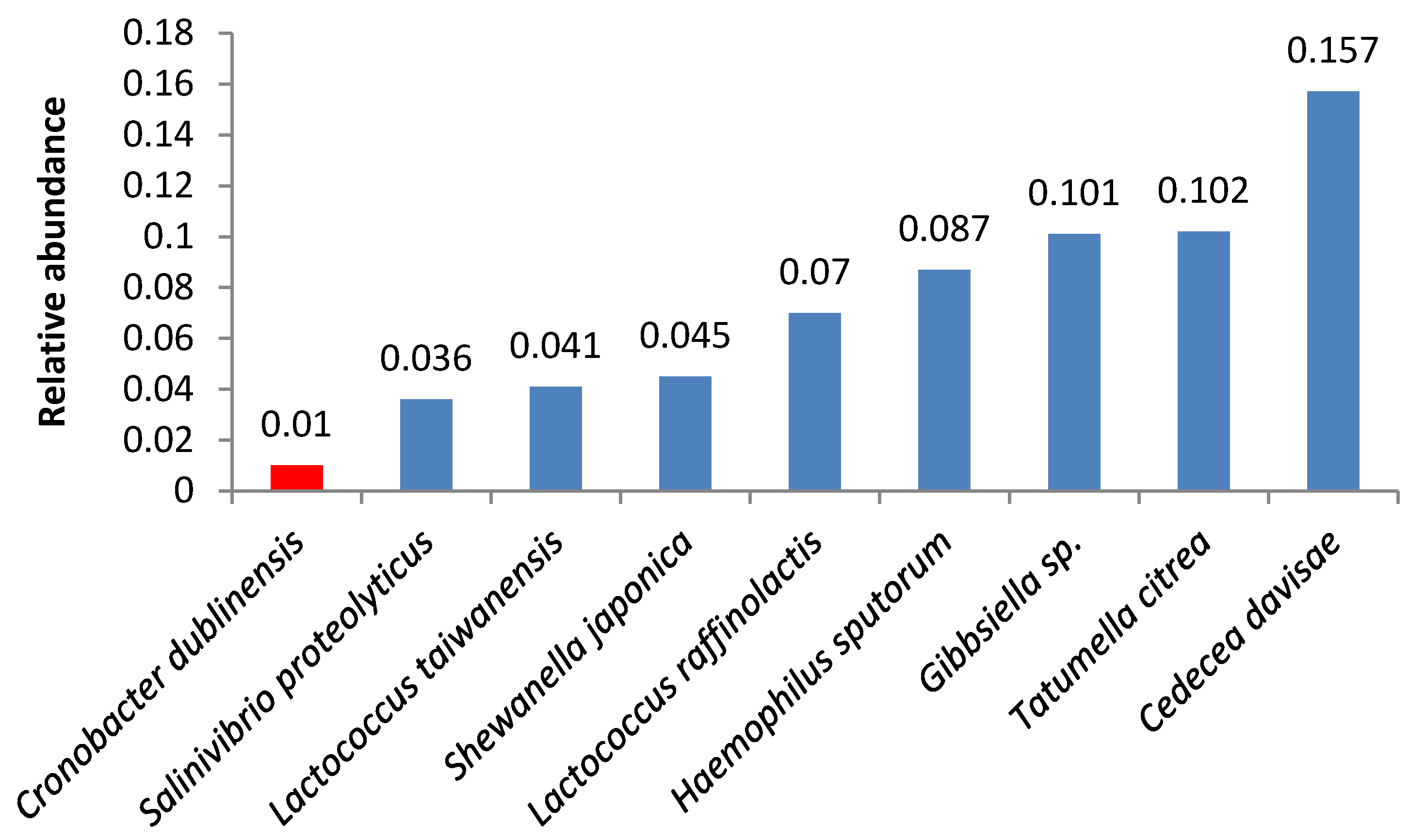
| Bacteria | Butter Sample | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | |
| Geobacillus spp. | |||||||||||||||||||||
| Stenotrophomonas spp. | |||||||||||||||||||||
| Thermus thermophilus | |||||||||||||||||||||
| Cedecea davisae | |||||||||||||||||||||
| Tatumella citrea | |||||||||||||||||||||
| Lactobacillus spp. | |||||||||||||||||||||
| Lactococcus spp. | |||||||||||||||||||||
| Streptococcus spp. | |||||||||||||||||||||
| Acinetobacter spp. | |||||||||||||||||||||
| Anoxybacillus spp. | |||||||||||||||||||||
| Xanthomonas bromi | |||||||||||||||||||||
| Pseudoxanthomonas spp. | |||||||||||||||||||||
| Propionibacterium acnes | |||||||||||||||||||||
| Leuconostoc spp. | |||||||||||||||||||||
| Pseudomonas spp. | |||||||||||||||||||||
| Massilia varians | |||||||||||||||||||||
| Ochrobactrum spp. | |||||||||||||||||||||
| Bacillus cereus group | |||||||||||||||||||||
| Chryseobacterium spp. | |||||||||||||||||||||
| Trabulsiella spp. | |||||||||||||||||||||
| Shewanella japonica | |||||||||||||||||||||
| Haemophilus sputorum | |||||||||||||||||||||
| Pantoea spp. | |||||||||||||||||||||
| Gibbsiella spp. | |||||||||||||||||||||
| Brevundimonas spp. | |||||||||||||||||||||
| <1% -no color | 1–10% | 21–30% | 21–30% | 31–40% | 41–50% | 51–60% | >61% | ||||||||||||||
| Opportunistic Pathogen | Found in Samples, % (10–100 Reads Per ASV) | Found in Samples, % (100–500 Reads Per ASV) | Found in Samples, % (>500 Reads Per ASV) |
|---|---|---|---|
| Bacillus cereus group | 100 | 66.7 | 52.4 |
| Pseudomonas aeruginosa | 38.1 | 23.8 | 9.6 |
| Cronobacter spp. | 57.2 | 42.9 | 14.3 |
| Escherichia coli | 61.9 | 28.6 | 14.3 |
| Listeria innocua | 38.1 | 14.3 | 4.8 |
| Citrobacter spp. | 14.3 | 9.5 | 4.8 |
| Enterococcus spp. | 90.5 | 66.7 | 14.3 |
| Klebsiella pneumoniae | 28.6 | 28.6 | 14.3 |
| Butter Sample | Amount of Bacteria, CFU/g | Bacteria Identified in the Sample |
|---|---|---|
| 2 | 3.0 × 105 | Bacillus pumilus, Acinetobacter guillouiae, Rothia sp., Rahnella aquatilis, Pseudomonas sp. |
| 3 | 3.0 × 104 | Bacillus thuringiensis, Neisseria sp., Moraxella osloensis |
| 4 | 2.0 × 104 | Enterococcus sp., Zimmermannella faecalis, Pseudomonas sp. |
| 6 | 5.0 × 104 | Lysinibacillus sp., Carnobacterium maltaromaticum, Pseudomonas sp. |
| 12 | 7.0 × 105 | Enterococcus italicus, Pseudomonas sp., Kocuria salsicia, Cronobacter sakazakii, |
| 18 | 1.0 × 106 | Enterobacter cloacae, Staphylococcus epidermidis, Lysinibacillus sp., Micrococcus sp. |
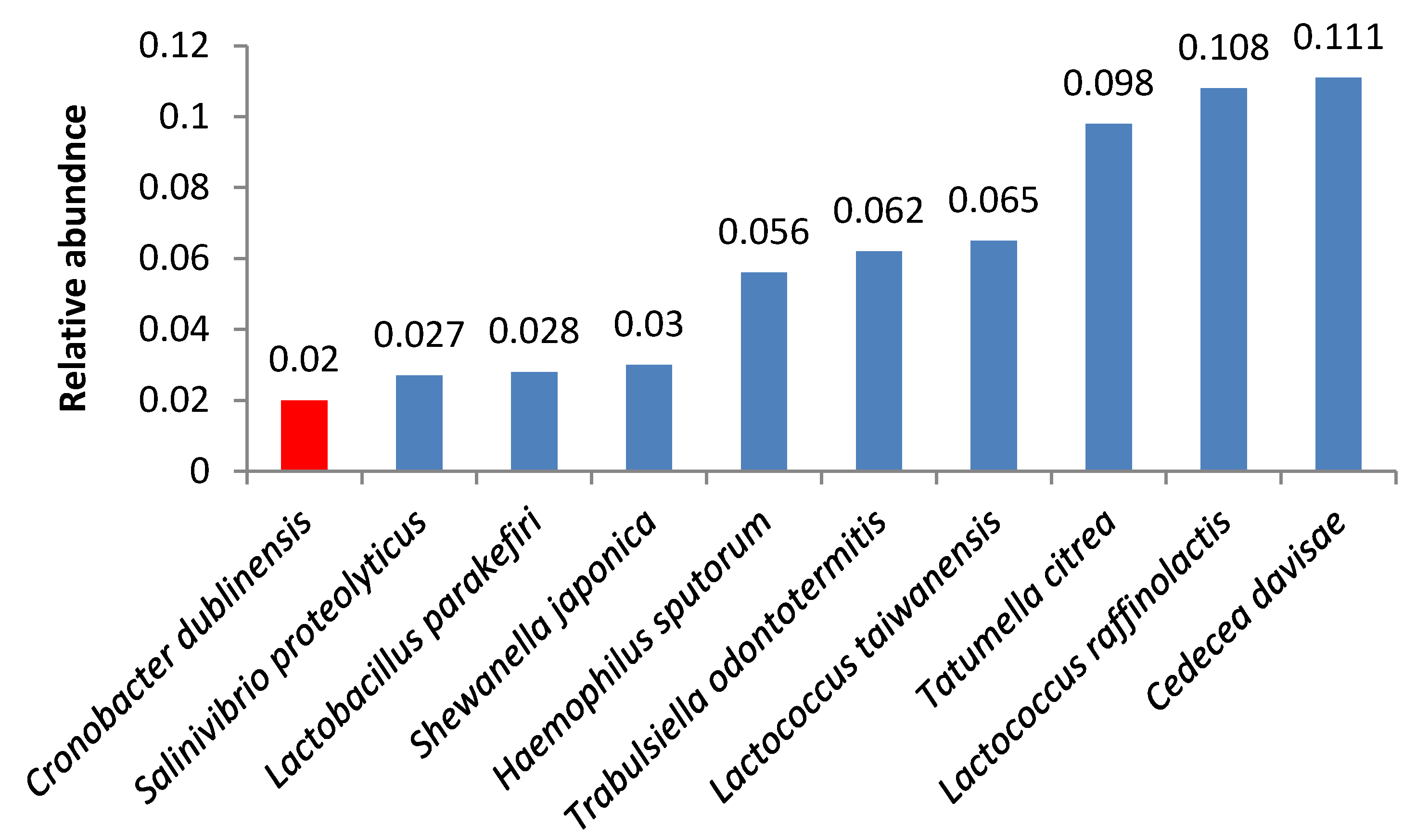
| Bacteria | Butter Sample | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | |
| Lactobacillus kisonensis | |||||||||||||||||||||
| Lactobacillus senioris | |||||||||||||||||||||
| Lactobacillus diolivorans | |||||||||||||||||||||
| Lactobacillus kefiri | |||||||||||||||||||||
| Lactobacillus parakefiri | |||||||||||||||||||||
| Lactobacillus delbrueckii | |||||||||||||||||||||
| Lactococcus taiwanensis | |||||||||||||||||||||
| Lactococcus chungangensis | |||||||||||||||||||||
| Lactococcus plantarum | |||||||||||||||||||||
| Lactococcus raffinolactis | |||||||||||||||||||||
| Streptococcus infantarius | |||||||||||||||||||||
| Streptococcus vestibularis | |||||||||||||||||||||
| Streptococcus porcorum | |||||||||||||||||||||
| Streptococcus hongkongensis | |||||||||||||||||||||
| Leuconostoc pseudomesenteroides | |||||||||||||||||||||
| 0% -no color | <0.1% | 0.1–1% | 2–15% | 16–30% | 31–45% | 46–60% | 61–75% | ||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Syromyatnikov, M.Y.; Kokina, A.V.; Solodskikh, S.A.; Panevina, A.V.; Popov, E.S.; Popov, V.N. High-Throughput 16S rRNA Gene Sequencing of Butter Microbiota Reveals a Variety of Opportunistic Pathogens. Foods 2020, 9, 608. https://doi.org/10.3390/foods9050608
Syromyatnikov MY, Kokina AV, Solodskikh SA, Panevina AV, Popov ES, Popov VN. High-Throughput 16S rRNA Gene Sequencing of Butter Microbiota Reveals a Variety of Opportunistic Pathogens. Foods. 2020; 9(5):608. https://doi.org/10.3390/foods9050608
Chicago/Turabian StyleSyromyatnikov, Mikhail Y., Anastasia V. Kokina, Sergey A. Solodskikh, Anna V. Panevina, Evgeny S. Popov, and Vasily N. Popov. 2020. "High-Throughput 16S rRNA Gene Sequencing of Butter Microbiota Reveals a Variety of Opportunistic Pathogens" Foods 9, no. 5: 608. https://doi.org/10.3390/foods9050608
APA StyleSyromyatnikov, M. Y., Kokina, A. V., Solodskikh, S. A., Panevina, A. V., Popov, E. S., & Popov, V. N. (2020). High-Throughput 16S rRNA Gene Sequencing of Butter Microbiota Reveals a Variety of Opportunistic Pathogens. Foods, 9(5), 608. https://doi.org/10.3390/foods9050608






