A Chemometric Approach to Establish Underlying Connections between Lipid and Protein Oxidation and Instrumental Color and Texture Characteristics in Brazilian Dry-cured Loin
Abstract
1. Introduction
2. Materials and Methods
2.1. Brazilian Dry-Cured Loin Sample
2.2. Water Activity and Cross-Sectional Area
2.3. Lipid Oxidation
2.4. Protein Oxidation
2.5. Instrumental Color
2.6. Texture Profile Analysis (TPA)
2.7. Statistical Analysis
3. Results and Discussion
3.1. Influence of the Key Intrinsic Factors on the Physicochemical Characteristics of Brazilian Dry-Cured Loin
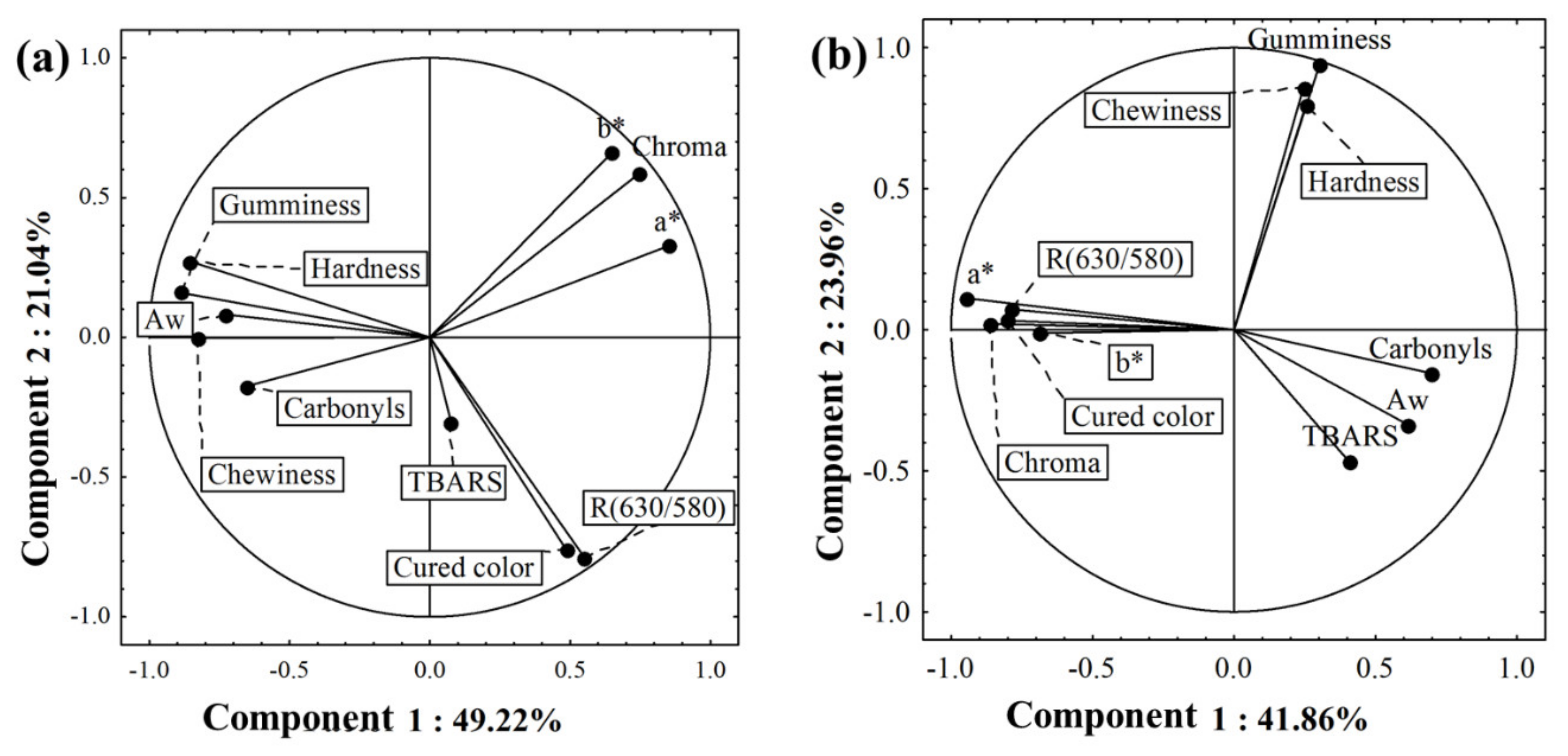
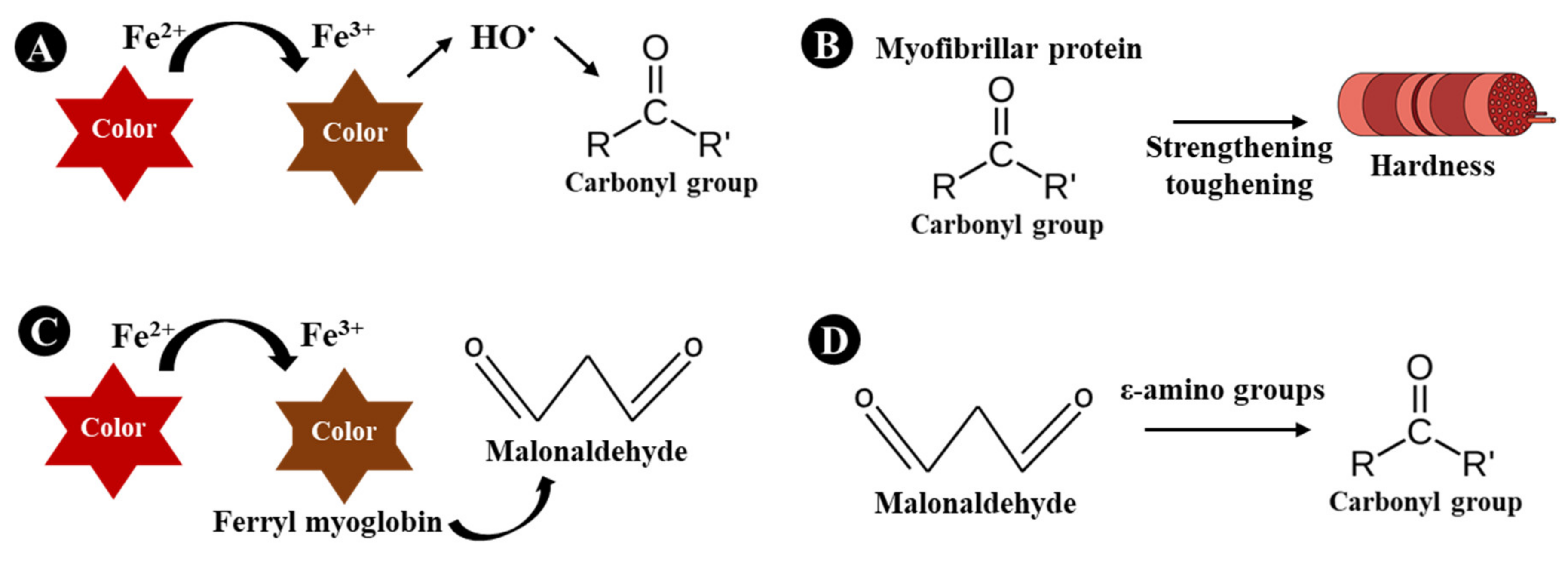
3.2. The Relationship among Physicochemical Parameters
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marušić, N.; Petrović, M.; Vidaček, S.; Petrak, T.; Medić, H. Characterization of traditional Istrian dry-cured ham by means of physical and chemical analyses and volatile compounds. Meat Sci. 2011, 88, 786–790. [Google Scholar] [CrossRef]
- Sebranek, J.G.; Bacus, J.N. Cured meat products without direct addition of nitrate or nitrite: What are the issues? Meat Sci. 2007, 77, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Gandemer, G. Lipids in muscles and adipose tissues, changes during processing and sensory properties of meat products. Meat Sci. 2002, 62, 309–321. [Google Scholar] [CrossRef]
- Ventanas, S.; Estevez, M.; Tejeda, J.F.; Ruiz, J. Protein and lipid oxidation in Longissimus dorsi and dry cured loin from Iberian pigs as affected by crossbreeding and diet. Meat Sci. 2006, 72, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Cunha, L.C.M.; Monteiro, M.L.G.; Costa-Lima, B.R.C.; Guedes-Oliveira, J.M.; Alves, V.H.M.; Almeida, A.L.; Tonon, R.V.; Rosenthal, A.; Conte-Junior, C.A. Effect of microencapsulated extract of pitaya (Hylocereus costaricensis) peel on color, texture and oxidative stability of refrigerated ground pork patties submitted to high pressure processing. Innov. Food Sci. Emerg. Technol. 2018, 49, 136–145. [Google Scholar] [CrossRef]
- Estévez, M. Protein carbonyls in meat systems: A review. Meat Sci. 2011, 89, 259–279. [Google Scholar] [CrossRef]
- Utrera, M.; Estévez, M. Oxidation of myofibrillar proteins and impaired functionality: Underlying mechanisms of the carbonylation pathway. J. Agric. Food Chem. 2012, 60, 8002–8011. [Google Scholar] [CrossRef] [PubMed]
- Estévez, M.; Luna, C. Dietary protein oxidation: A silent threat to human health? Crit. Rev. Food Sci. Nutr. 2017, 57, 3781–3793. [Google Scholar] [CrossRef]
- Soladoye, O.P.; Juárez, M.L.; Aalhus, J.L.; Shand, P.; Estévez, M. Protein oxidation in processed meat: Mechanisms and potential implications on human health. Compr. Rev. Food Sci. Food Saf. 2015, 14, 106–122. [Google Scholar] [CrossRef]
- Viana, F.M.; Canto, A.C.V.C.S.; Costa-Lima, B.R.C.; Salim, A.P.A.A.; Conte-Junior, C.A. Color stability and lipid oxidation of broiler breast meat from animals raised on organic versus non-organic production systems. Poult. Sci. 2017, 96, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H. Cytotoxicity and genotoxicity of lipid-oxidation products. Am. J. Clin. Nutr. 1993, 57, 779S–786S. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.A.P.; Estévez, M.; Ferreira, V.C.S.; Silva, S.A.; Lemos, L.T.M.; Ida, E.I.; Shimokomaki, M.; Madruga, M. Protein and lipid oxidations in jerky chicken and consequences on sensory quality. LWT 2018, 97, 341–348. [Google Scholar] [CrossRef]
- Maloney, J.F.; Labuza, T.P.; Wallace, D.H.; Karel, M. Autoxidation of methyl linoleate in freeze-dried model systems. I. Effect of water on the autocatalyzed oxidation. J. Food Sci. 1966, 31, 878–884. [Google Scholar] [CrossRef]
- Labuza, T.P. The effect of water activity on reaction kinetics of food deterioration. Food Technol. 1982, 34, 36–59. [Google Scholar]
- Estévez, M.; Padilla, P.; Carvalho, L.; Martín, L.; Carrapiso, A.; Delgado, J. Malondialdehyde interferes with the formation and detection of primary carbonyls in oxidized proteins. Redox Biol. 2019, 26, 101277. [Google Scholar] [CrossRef]
- Hernández, P.; Navarro, J.L.; Toldra, F. Lipolytic and oxidative changes in two Spanish pork loin products: Dry-cured loin and pickled-cured loin. Meat Sci. 1999, 51, 123–128. [Google Scholar] [CrossRef]
- Estévez, M.; Morcuende, D.; Cava, R. Oxidative and colour changes in meat from three lines of free-range reared Iberian pigs slaughtered at 90 kg live weight and from industrial pig during refrigerated storage. Meat Sci. 2003, 65, 1139–1146. [Google Scholar] [CrossRef]
- Faustman, C.; Sun, Q.; Mancini, R.; Suman, S.P. Myoglobin and lipid oxidation interactions: Mechanistic bases and control. Meat Sci. 2010, 86, 86–94. [Google Scholar] [CrossRef]
- Pateiro, M.; Franco, D.; Carril, J.A.; Lorenzo, J.M. Changes on physico-chemical properties, lipid oxidation and volatile compounds during the manufacture of celta dry-cured loin. J. Food Sci. Technol. 2015, 52, 4808–4818. [Google Scholar] [CrossRef]
- Shikha Ojha, K.; Granato, D.; Rajuria, G.; Barba, F.J.; Kerry, J.P.; Tiwari, B.K. Application of chemometrics to assess the influence of ultrasound frequency, Lactobacillus sakei culture and drying on beef jerky manufacture: Impact on amino acid profile, organic acids, texture and colour. Food Chem. 2018, 239, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Y.; Ren, H.; Zhan, Y. Comparison of bacterial diversity profiles and microbial safety assessment of salami, Chinese dry-cured sausage and Chinese smoked-cured sausage by high-throughput sequencing. LWT 2018, 90, 108–115. [Google Scholar] [CrossRef]
- Yang, Q.; Sun, D.W.; Cheng, W. Development of simplified models for nondestructive hyperspectral imaging monitoring of TVB-N contents in cured meat during drying process. J. Food Eng. 2017, 192, 53–60. [Google Scholar] [CrossRef]
- Granato, D.; Putnik, P.; Kovačević, D.B.; Santos, J.S.; Calado, V.; Rocha, R.S.; Cruz, A.G.; Jarvis, B.; Rodionova, O.Y.; Pomerantsev, A. Trends in chemometrics: Food authentication, microbiology, and effects of processing. Compr. Rev. Food Sci. Food Saf. 2018, 17, 663–677. [Google Scholar] [CrossRef]
- Nunes, C.A.; Alvarenga, V.O.; Souza Sant’Ana, A.; Santos, J.S.; Granato, D. The use of statistical software in food science and technology: Advantages, limitations and misuses. Food Res. Int. 2015, 75, 270–280. [Google Scholar] [CrossRef]
- Incaper, Instituto Capixaba de Pesquisa, Assistência Técnica e Extensão Rural. Monitoramento Agroclimático—Venda Nova do Imigrante/ES. Available online: https://meteorologia.incaper.es.gov.br/monitoramento-venda_nova_do_imigrante (accessed on 2 October 2017).
- Yin, M.C.; Faustman, C.; Riesen, J.W.; Williams, S.N. α-Tocopherol and ascorbate delay oxymyoglobin and phospholipid oxidation in vitro. J. Food Sci. 1993, 58, 1273–1276. [Google Scholar] [CrossRef]
- Joseph, P.; Suman, S.P.; Rentfrow, G.; Li, S.; Beach, C.M. Proteomics of muscle-specific beef color stability. J. Agric. Food Chem. 2012, 60, 3196–3203. [Google Scholar] [CrossRef]
- Oliver, C.N.; Ahn, B.W.; Moerman, E.J.; Goldstein, S.; Stadtman, E.R. Age-related changes in oxidized proteins. J. Biol. Chem. 1987, 262, 5488–5491. [Google Scholar]
- Armenteros, M.; Heinonen, M.; Ollilainen, V.; Toldrá, F.; Estévez, M. Analysis of protein carbonyls in meat products by using the DNPH-method, fluorescence spectroscopy and liquid chromatography–electrospray ionisation–mass spectrometry (LC–ESI–MS). Meat Sci. 2009, 83, 104–112. [Google Scholar] [CrossRef]
- Canto, A.C.V.C.S.; Costa-Lima, B.R.C.; Suman, S.P.; Monteiro, M.L.G.; Viana, F.M.; Salim, A.P.A.A.; Nair, M.N.; Silva, T.J.P.; Conte-Junior, C.A. Color attributes and oxidative stability of Longissimus lumborum and psoas major muscles from Nellore bulls. Meat Sci. 2016, 121, 19–26. [Google Scholar] [CrossRef]
- Mercier, Y.; Gatellier, P.; Viau, M.; Remignon, H.; Renerre, M. Effect of dietary fat and vitamin E on colour stability and on lipid and protein oxidation in Turkey meat during storage. Meat Sci. 1998, 48, 301–318. [Google Scholar] [CrossRef]
- AMSA. Meat Color Measurement Guidelines; American Meat Science Association: Champaign, IL, USA, 2012. [Google Scholar]
- Serra, X.; Ruiz-Ramírez, J.; Arnau, J.; Gou, P. Texture parameters of dry-cured ham m. biceps femoris samples dried at different levels as a function of water activity and water content. Meat Sci. 2005, 69, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Harkouss, R.; Astruc, T.; Lebert, A.; Gatellier, P.; Loison, O.; Safa, H.; Portanguen, S.; Parafita, E.; Mirade, P. Quantitative study of the relationships among proteolysis, lipid oxidation, structure and texture throughout the dry-cured ham process. Food Chem. 2015, 166, 522–530. [Google Scholar] [CrossRef]
- Ruiz-Ramírez, J.; Serra, X.; Arnau, J.; Gou, P. Profiles of water content, water activity and texture in crusted dry-cured loin and in non-crusted dry-cured loin. Meat Sci. 2005, 69, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Barden, L.; Decker, E.A. Lipid oxidation in low-moisture food: A review. Crit. Rev. Food Sci. Nutr. 2016, 56, 2467–2482. [Google Scholar] [CrossRef]
- Lund, M.N.; Heinonen, M.; Baron, C.P.; Estévez, M. Protein oxidation in muscle foods: A review. Mol. Nutr. Food Res. 2011, 55, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Utrera, M.; Estévez, M. Oxidative damage to poultry, pork, and beef during frozen storage through the analysis of novel protein oxidation markers. J. Agric. Food Chem. 2013, 61, 7987–7993. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.H.; Deng, Y.M.; Jia, N.; Li, R.R.; Cao, J.X.; Liu, D.Y.; Li, J.R. Low-field NMR determination of water distribution in meat batters with NaCl and polyphosphate addition. Food Chem. 2016, 200, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, C.S.; Mársico, E.T.; Ribeiro, R.O.R.; Conte-Júnior, C.A.; Mano, S.B.; Augusto, C.J.C.; Jesus, E.F.O. Low-Field Nuclear Magnetic Resonance (LF NMR 1H) to assess the mobility of water during storage of salted fish (Sardinella brasiliensis). J. Food Eng. 2016, 169, 321–325. [Google Scholar] [CrossRef]
- Böhner, N.; Hösl, F.; Rieblinger, K.; Danzl, W. Effect of retail display illumination and headspace oxygen concentration on cured boiled sausages. Food Packag. Shelf Life 2014, 1, 131–139. [Google Scholar] [CrossRef]
- Estévez, M.; Ventanas, S.; Cava, R. Protein oxidation in frankfurters with increasing levels of added rosemary essential oil: Effect on color and texture deterioration. J. Food Sci. 2005, 70, c427–c432. [Google Scholar] [CrossRef]
- Ganhão, R.; Morcuende, D.; Estévez, M. Protein oxidation in emulsified cooked burger patties with added fruit extracts: Influence on colour and texture deterioration during chill storage. Meat Sci. 2010, 85, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Lobo, F.; Ventanas, S.; Morcuende, D.; Estévez, M. Underlying chemical mechanisms of the contradictory effects of NaCl reduction on the redox-state of meat proteins in fermented sausages. LWT Food Sci. Technol. 2016, 69, 110–116. [Google Scholar] [CrossRef]
- Fuentes, V.; Ventanas, J.; Morcuende, D.; Estévez, M.; Ventanas, S. Lipid and protein oxidation and sensory properties of vacuum-packaged dry-cured ham subjected to high hydrostatic pressure. Meat Sci. 2010, 85, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Estévez, M.; Xiong, Y. Intake of oxidized proteins and amino acids and causative oxidative stress and disease. J. Food Sci. 2019, 84, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Feeney, R.E.; Blankenhorn, G.; Dixon, H.B. Carbonyl-amine reactions in protein chemistry. In Advances in Protein Chemistry; Academic Press: Cambridge, MA, USA, 1975; Volume 29, pp. 135–203. [Google Scholar]
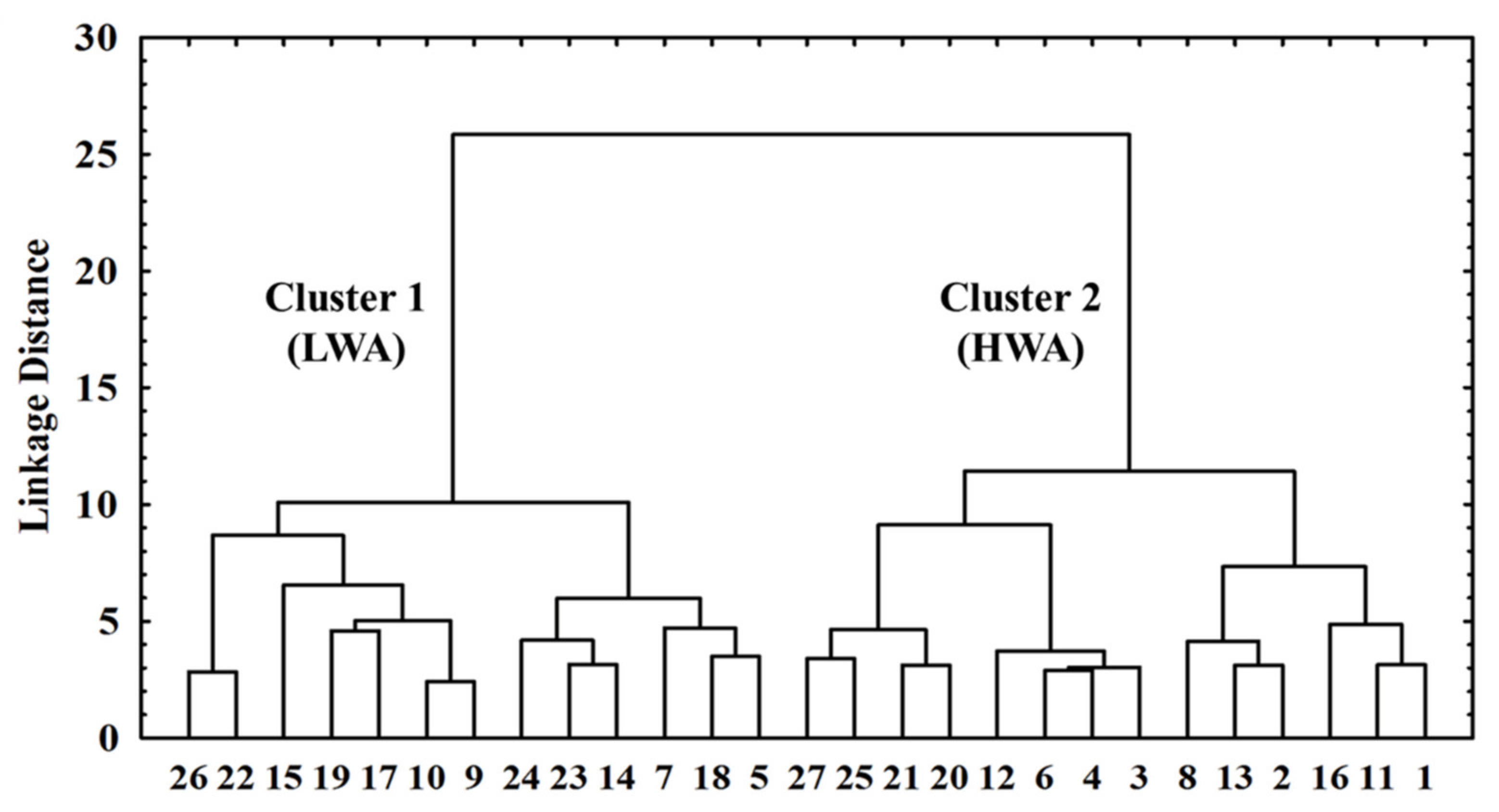
| Variables | Cluster 1 (LWA) | Cluster 2 (HWA) | p Values |
|---|---|---|---|
| Cross-sectional area (cm2) | 21.69 ± 2.11 | 23.30 ± 2.93 | 0.156 |
| Water activity | 0.79 ± 0.01 | 0.84 ± 0.01 | 0.001 |
| TBARS A | 0.16 ± 0.04 | 0.27 ± 0.07 | <0.001 |
| Carbonyls B | 1.93 ± 0.31 | 3.12 ± 0.91 | <0.001 |
| Hardness (N) | 6.45 ± 0.88 | 5.13 ± 0.84 | <0.001 |
| Adhesiveness (g·mm) | −9.19 ± 5.71 | −10.47 ± 4.60 | 0.543 |
| Springiness (mm) | 0.70 ± 0.05 | 0.71 ± 0.06 | 0.701 |
| Cohesiveness | 0.64 ± 0.03 | 0.64 ± 0.04 | 0.892 |
| Gumminess (N·mm) | 4099.5 ± 599.5 | 3286.0 ± 492.3 | 0.001 |
| Chewiness (g·mm) | 2898.1 ± 506.9 | 2335.8 ± 402.1 | 0.004 |
| Resilience | 0.22 ± 0.03 | 0.21 ± 0.02 | 0.304 |
| L* | 34.31 ± 2.24 | 33.09 ± 1.88 | 0.132 |
| a* | 6.62 ± 0.41 | 5.10 ± 0.46 | <0.001 |
| b* | 6.53 ± 0.83 | 5.43 ± 0.71 | 0.001 |
| Chroma | 9.33 ± 0.82 | 7.50 ± 0.77 | <0.001 |
| Hue | 44.68 ± 2.22 | 46.46 ± 2.82 | 0.078 |
| R(630/580) | 1.51 ± 0.02 | 1.41 ± 0.05 | <0.001 |
| Cured color | 1.58 ± 0.03 | 1.44 ± 0.06 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosario, D.K.A.; Furtado, M.R.; Mutz, Y.S.; Rodrigues, B.L.; Bernardo, Y.A.A.; Baltar, J.D.; Bernardes, P.C.; Estevez, M.; Conte-Junior, C.A. A Chemometric Approach to Establish Underlying Connections between Lipid and Protein Oxidation and Instrumental Color and Texture Characteristics in Brazilian Dry-cured Loin. Foods 2020, 9, 536. https://doi.org/10.3390/foods9040536
Rosario DKA, Furtado MR, Mutz YS, Rodrigues BL, Bernardo YAA, Baltar JD, Bernardes PC, Estevez M, Conte-Junior CA. A Chemometric Approach to Establish Underlying Connections between Lipid and Protein Oxidation and Instrumental Color and Texture Characteristics in Brazilian Dry-cured Loin. Foods. 2020; 9(4):536. https://doi.org/10.3390/foods9040536
Chicago/Turabian StyleRosario, Denes K. A., Maraysa R. Furtado, Yhan S. Mutz, Bruna L. Rodrigues, Yago A. A. Bernardo, Jéssica D. Baltar, Patricia C. Bernardes, Mario Estevez, and Carlos A. Conte-Junior. 2020. "A Chemometric Approach to Establish Underlying Connections between Lipid and Protein Oxidation and Instrumental Color and Texture Characteristics in Brazilian Dry-cured Loin" Foods 9, no. 4: 536. https://doi.org/10.3390/foods9040536
APA StyleRosario, D. K. A., Furtado, M. R., Mutz, Y. S., Rodrigues, B. L., Bernardo, Y. A. A., Baltar, J. D., Bernardes, P. C., Estevez, M., & Conte-Junior, C. A. (2020). A Chemometric Approach to Establish Underlying Connections between Lipid and Protein Oxidation and Instrumental Color and Texture Characteristics in Brazilian Dry-cured Loin. Foods, 9(4), 536. https://doi.org/10.3390/foods9040536








