Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical Reagents and Standards
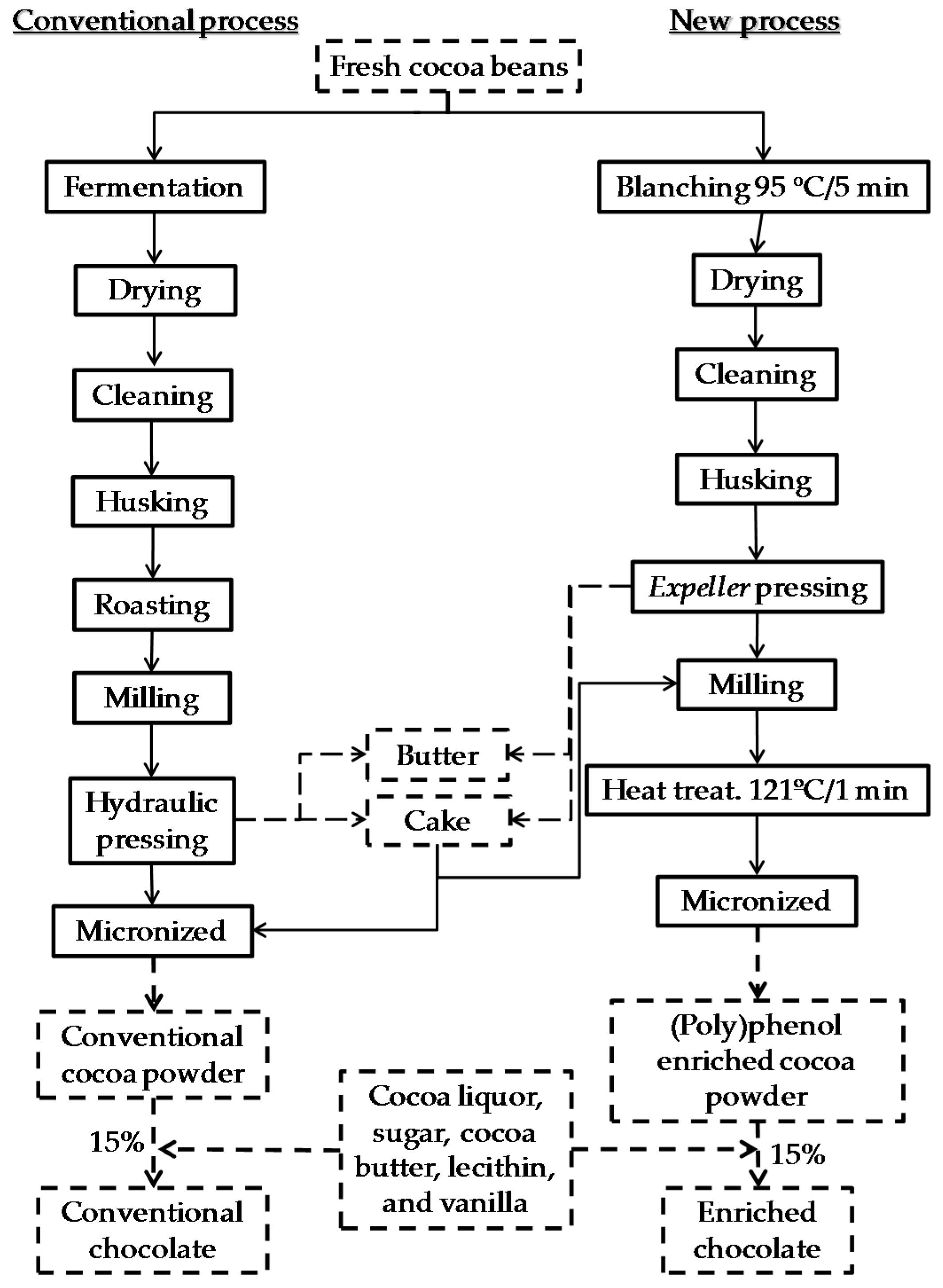
2.2. Development of (Poly)Phenol Enriched Dark Chocolate and Conventional Dark Chocolate
2.3. Determination of the Antioxidant Capacity
2.4. Analysis of Flavanols by High Performance Liquid Chromatography with UV-Vis Detection (HPLC-DAD)
2.5. Analysis of Flavan-3-ols and Procyanidins by High Performance Liquid Chromatography and Fluorescence Detection (HPLC-FL)
2.6. Statistical Analysis
3. Results
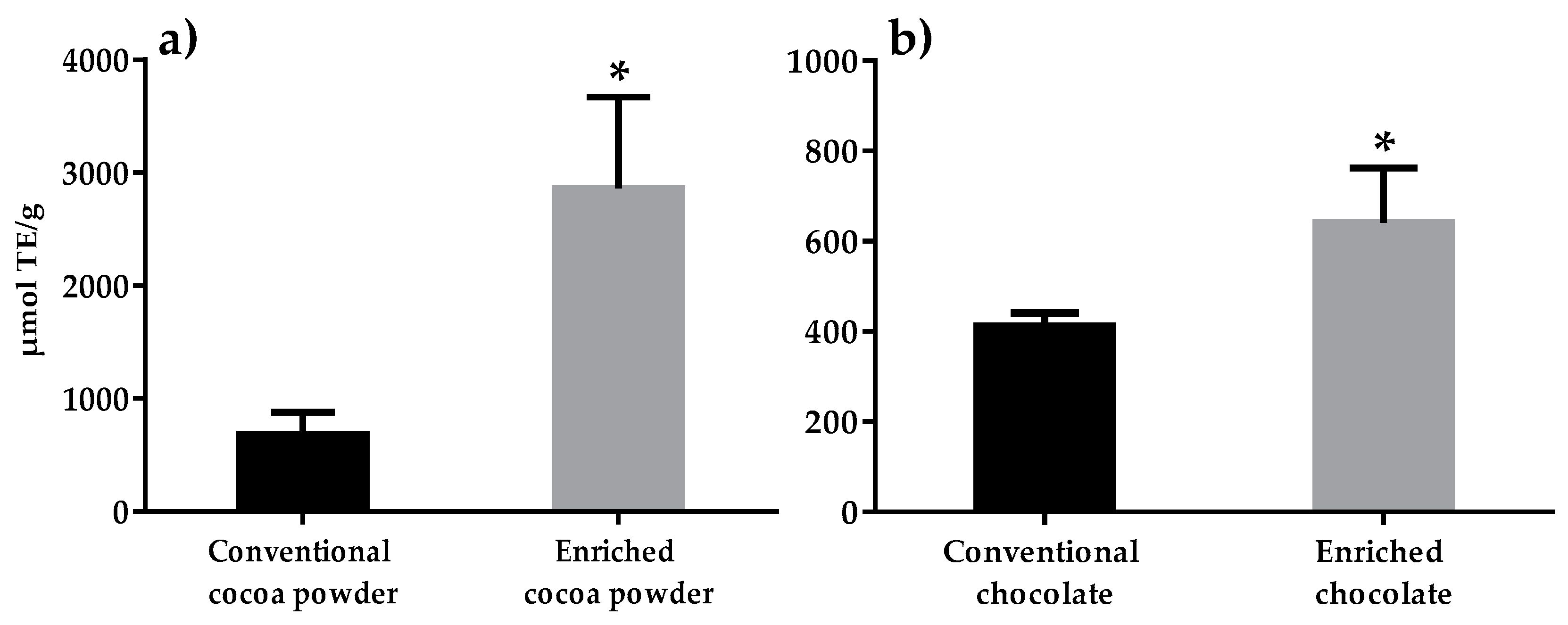
3.1. Antioxidant Capacity
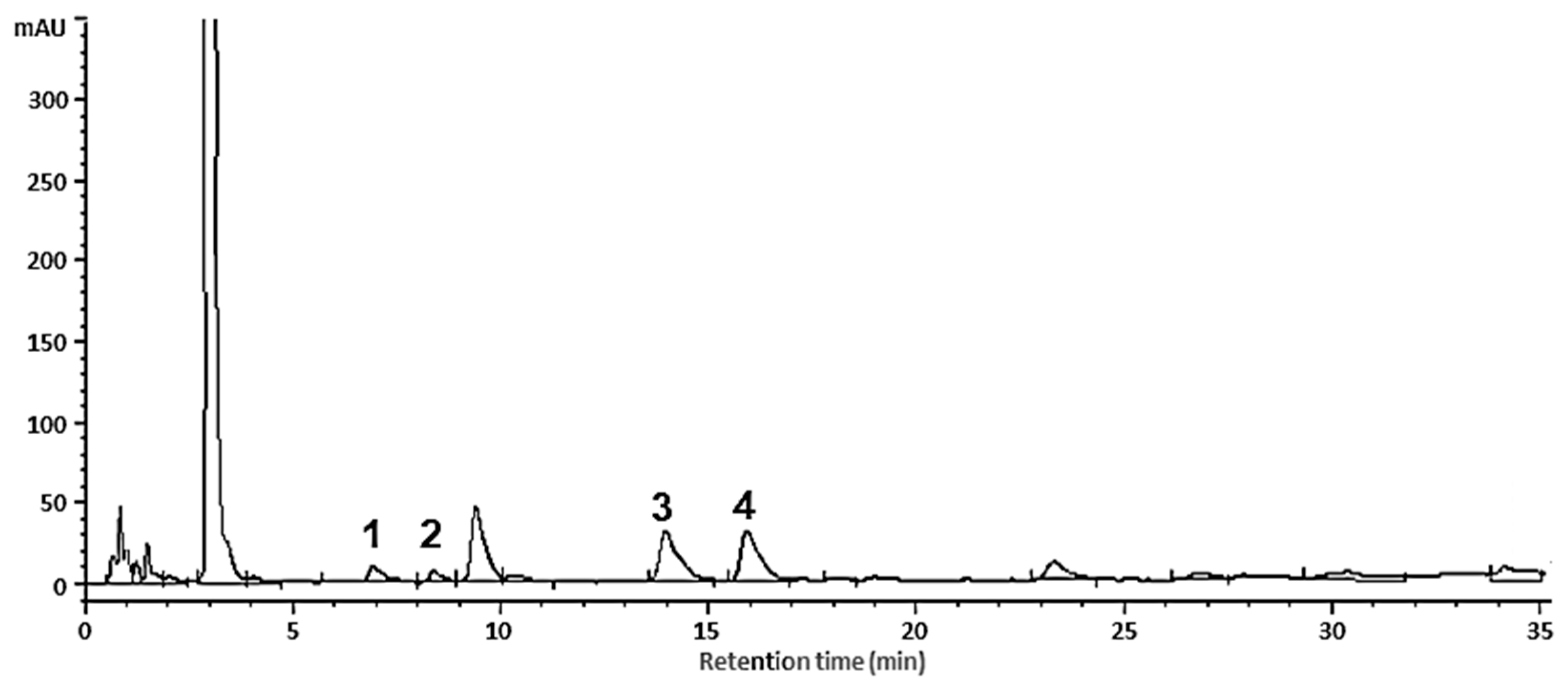
3.2. Flavanol Analysis by HPLC-DAD
3.3. Procyanidins Analysis by HPLC-FL
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martín, M.A.; Goya, L.; Ramos, S. Potential for preventive effects of cocoa and cocoa polyphenols in cancer. Food Chem. Toxicol. 2013, 56, 336–351. [Google Scholar] [CrossRef]
- Crews, W.D., Jr.; Harrison, D.W.; Wright, J.W. A double-blind, placebo controlled, randomized trial of the effects of dark chocolate and cocoa on variables associated with neuropsychological functioning and cardiovascular health: Clinical findings from a sample of healthy, cognitively intact older adults. Am. J. Clin. Nutr. 2008, 7, 872–880. [Google Scholar]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Cocoa polyphenols and their potential benefits for human health. Oxid. Med. Cell Longev. 2012, 2012, 1–23. [Google Scholar] [CrossRef]
- Roura, E.; Andrés-Lacueva, C.; Estruch, R.; Mata-Bilbao, M.L.; Izquierdo-Pulido, M.; Waterhouse, A.L.; Lamuela-Raventós, R.M. Milk does not affect the bioavailability of cocoa powder flavonoid in healthy human. Ann. Nutr. Metab. 2007, 51, 493–498. [Google Scholar] [CrossRef]
- Urbańska, B.; Derewiaka, D.; Lenart, A.; Kowalska, J. Changes in the composition and content of polyphenols in chocolate resulting from pre-treatment method of cocoa beans and technological process. Eur. Food Res. Technol. 2019, 245, 2101–2112. [Google Scholar] [CrossRef]
- Bordiga, M.; Locatelli, M.; Travaglia, F.; Coisson, J.D.; Mazza, G.; Arlorio, M. Evaluation of the effect of processing on cocoa polyphenols: Antiradical activity, anthocyanins and procyanidins profiling from raw beans to chocolate. Int. J. Food Sci. Tech. 2015, 50, 840–848. [Google Scholar] [CrossRef]
- Kelm, M.A.; Johnson, C.; Robbins, R.J.; Hammerstone, J.F.; Schmitz, H.H. High-performance liquid chromatography separation and purification of cacao (Theobroma cacao L.) procyanidins according to degree of polymerization using a diol stationary phase. J. Agric. Food Chem. 2006, 54, 1571–1576. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; House, S.E.; Wu, X.; Ou, B.; Prior, R.L. Procyanidin and catechin contents and antioxidant capacity of cocoa and chocolate products. J. Agric. Food Chem. 2006, 54, 4057–4061. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.; Jalil, A.M.; Weng, K.K.; Ismail, A.; Ghani, N.A.; Adenan, I. Epicatechin content and antioxidant capacity of cocoa beans from four different countries. Afr. J. Biotechnol. 2010, 9, 1052–1059. [Google Scholar]
- Afoakwa, E.O.; Quao, J.; Budu, A.S.; Takrama, J.F.; Saalia, F.K. Changes in total polyphenols, o-diphenols and anthocyanin concentrations during fermentation of pulp pre-conditioned cocoa (Theobroma cacao) beans. Int. Food Res. J. 2012, 19, 1071–1077. [Google Scholar]
- Andrés-Lacueva, C.; Monagas, M.; Khan, N.; Izquierdo-Pulido, M.; Urpi-Sarda, M.; Permanyer, J.; Lamuela-Raventos, R.M. Flavanol and Flavonol Contents of Cocoa Powder Products: Influence of the Manufacturing Process. J. Agric. Food Chem. 2008, 56, 3111–3117. [Google Scholar] [CrossRef] [PubMed]
- Kealey, K.S.; Snyder, R.M.; Romanczyk, L.J.; Geyer, H.M.; Myers, M.E.; Withcare, E.J.; Hammerstone, J.F.; Schmitz, H.H. Cocoa Components, Edible Products Having Enhanced Polyphenol Content, Methods of Making Same and Medical Uses. U.S. Patent 6,312,753; Mars Incorporated, USA, 1998. [Google Scholar]
- Zhong, J.L.; Muhammad, N.; Gu, Y.C.; Yan, W.D. A simple and efficient method for enrichment of cocoa polyphenols from cocoa bean husks with macroporus resins following a scale-up separation. J. Food Eng. 2019, 243, 82–88. [Google Scholar] [CrossRef]
- Schinella, G.; Mosca, S.; Cienfuegos-Jovellanos, E.; Pasamar, M.A.; Muguerza, B.; Ramón, D.; Ríos, J.L. Antioxidant properties of polyphenol-rich cocoa products industrially processed. Food Res. Int. 2010, 43, 1614–1623. [Google Scholar] [CrossRef]
- Cienfuegos-Jovellanos, E.; Quiñones, M.M.; Muguerza, B.; Moulay, L.; Miguel, M.; Aleixandre, A. Antihypertensive effect of a polyphenol-rich cocoa powder industrially processed to preserve the original flavonoids of the cocoa beans. J. Agric. Food Chem. 2009, 57, 57, 6156–6162, 9169–9180. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, L.M.; Kroon, P.A.; Wood, S.; Horgan, G.W.; Cienfuegos-Jovellanos, E.; Saha, S.; Duthie, G.G.; de Roos, B. Flavan-3-ol-enriched dark chocolate and white chocolate improve acute measures of platelet function in a gender-specific way—A randomized-controlled human intervention trial. Mol. Nutr. Food Res. 2013, 57, 191–202. [Google Scholar] [CrossRef]
- Crozier, S.J.; Preston, A.G.; Hurst, J.W.; Payne, M.J.; Mann, J.; Hainly, L.; Miller, D. Cacao seeds are a “Super Fruit”: A comparative analysis of various fruit powders and products. Chem. Cent. J. 2011, 5, 1–6. [Google Scholar] [CrossRef]
- Miller, K.B.; Stuart, D.A.; Smith, N.L.; Lee, C.Y.; Mc Hale, N.L.; Flanagan, J.A.; Ou, B.; Hurst, W.J. Antioxidant activity and polyphenol and procyanidin contents of selected commercially available cocoa-containing and chocolate products in the United States. J. Agric. Food Chem. 2006, 54, 4062–4068. [Google Scholar] [CrossRef]
- Lamuela-Raventós, R.M.; Romero-Pérez, A.I.; Andrés-Lacueva, C.; Tornero, A. Review: Health effects of cocoa flavonoids. Int. J. Food Sci. Tech. 2005, 11, 159–176. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Cienfuegos-Jovellanos, E.; Marín, A.; Muguerza, B.; Gil-Izquierdo, A.; Cerdá, B.; Zafrilla, P.; Morillas, J.; Mulero, J.; Ibarra, A.; et al. A new process to develop a cocoa powder with higher flavonoid monomer content and enhanced bioavailability in healthy humans. J. Agric. Food Chem. 2007, 55, 3926–3935. [Google Scholar] [CrossRef]
- Cienfuegos-Jovellanos, E.; Pasamar, M.A.; Fritz, J.; Ramón, D.; Castilla, Y. Method for Obtaining Polyphenol-Rich Cocoa Powder with a Low Fat Content and Cocoa thus Obtained; Patent Cooperation Treaty (PCT) WO 2007/096449A1; Natraceutical Industrial: Valencia, Spain, 2007. [Google Scholar]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radical. Biol. Med. 1993, 14, 301–311. [Google Scholar] [CrossRef]
- Andrés-Lacueva, C.; Lamuela-Raventós, R.M.; Jáuregui, O.; Casals, I.; Izquierdo-Pulido, M.; Permayer, J. An LC method for the analysis of cocoa phenolics. LC GC Eur. 2000, 13, 902–905. [Google Scholar]
- Gu, L.; Kelm, M.; Hammerstone, J.F.; Beecher, G.; Cunningham, D.; Vannozzi, S.; Prior, R.L. Fractionation of polymeric procyanidins from lowbush blueberry and quantification of procyanidins in selected foods with an optimized normal-phase HPLC-MS fluorescent detection method. J. Agr. Food Chem. 2002, 50, 4852–4860. [Google Scholar] [CrossRef]
- Prior, R.L.; Gu, L. Ocurrence and biological significance of proanthocyanidins in the American diet. Phytochemistry 2005, 66, 2264–2280. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Cao, G. Antioxidant phytochemicals in fruits and vegetables: Diet and health implications. Hortscience 2000, 35, 588–592. [Google Scholar] [CrossRef]
- Todorovic, V.; Radojcic, I.; Todorovic, Z.; Jankovic, G.; Dodevska, M.; Sobajic, S. Polyphenols, methylxanthines, and antioxidant capacity of chocolates produced in Serbia. J. Food Compos. Anal. 2015, 41, 137–143. [Google Scholar] [CrossRef]
- Rigaud, J.; Escribano-Bailón, M.; Prieur, C.; Souquet, J.M. Normal-phase high-performance liquid chromatographic separation of procyanidins from cacao beans and grape seeds. J. Chromatogr. A 1993, 654, 255–260. [Google Scholar] [CrossRef]
- Richelle, M.; Tavazzi, I.; Enslen, M.; Offord, E.A. Plasma kinetics in man of epicatechin from black chocolate. Eur. J. Clin. Nutr. 1999, 53, 22–26. [Google Scholar] [CrossRef]
- Rein, D.; Lotio, S.; Holt, R.R.; Keen, C.L.; Schmitz, H.H.; Fraga, C.G. Epicatechin in human plasma: In vivo determination and effect of chocolate consumption on a plasma oxidation status. J. Nutr. 2000, 130, 2109S–2114S. [Google Scholar] [CrossRef]
- Holt, R.R.; Lazarus, S.A.; Cameron Sullards, M.; Zhu, Q.Y.; Schramm, D.D.; Hammerstone, J.F.; Fraga, C.G.; Schmitz, H.H.; Keen, C.L. Procyanidin dimer B2 [epicatechin-(4b-8)-epicatechin] inhuman plasma after the consumption of a flavanol-richcocoa. Am. J. Clin. Nutr. 2002, 76, 798–804. [Google Scholar] [CrossRef]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Cienfuegos-Jovellanos, E.; Laghi, S.; Muguerza, B.; Ríos, J.L. Inhibition of ulcerative colitis in mice after oral administration of a polyphenol-enriched cocoa extract is mediated by the inhibition of STAT1 and STAT3 phosphorylation in colon cells. J. Agric. Food Chem. 2011, 59, 6474–6483. [Google Scholar] [CrossRef]
- Misnawi Jinap, S.; Jamilah, B.; Nazamid, S. Effects of incubation and polyphenol oxidase enrichment on colour, fermentation index, procyanidins and astringency of unfermented and partly fermented cocoa beans. Int. J. Food Sci. Tech. 2003, 38, 285–295. [Google Scholar] [CrossRef]
- Miller, K.B.; Hurst, W.J.; Flannigan, N.; Ou, B.; Lee, C.Y.; Smith, N.; Stuart, D. Survey of commercially available chocolate-and cocoa-containing products in the United States. 2. Comparison of flavan-3-ol content with nonfat cocoa solids, total polyphenols, and percent cacao. J. Agric. Food Chem. 2009, 57, 9169–9180. [Google Scholar] [CrossRef] [PubMed]
- Pedan, V.; Fischer, N.; Bernath, K.; Hühn, T.; Rohn, S. Determination of oligomeric proanthocyanidins and their antioxidant capacity from different chocolate manufacturing stages using the NP-HPLC-online-DPPH methodology. Food Chem. 2017, 214, 523–532. [Google Scholar] [CrossRef] [PubMed]
- European Union Law (EUR-Lex). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32015R0539&from=ES (accessed on 10 February 2020).

| Flavanols | Conventional Cocoa Powder | Enriched Cocoa Powder | Conventional Chocolate | Enriched Chocolate |
|---|---|---|---|---|
| (+)-catechin | 0.5 ± 0.1 | 3.4 ± 0.0 * | 0.3 ± 0.0 | 0.6 ± 0.0 *** |
| (−)-epicatechin | 1.9 ± 0.5 | 24.9 ± 0.0 *** | 1.1 ± 0.1 | 3.4 ± 0.2 *** |
| Procyanidin B1 | 0.1 ± 0.0 | 2.1 ± 0.1 *** | 1.2 ± 0.1 | 1.8 ± 0.1 ** |
| Procyanidin B2 | 1.0 ± 0.2 | 14.2 ± 0.3 *** | 1.3 ± 0.2 | 4.2 ± 0.1 *** |
| Total | 3.5 ± 0.8 | 44.6 ± 0.3 *** | 3.9 ± 0.1 | 10.0 ± 0.1 *** |
| Flavanols | Conventional Cocoa Powder | Enriched Cocoa Powder | Conventional Chocolate | Enriched Chocolate |
|---|---|---|---|---|
| Monomers | 2.70 ± 0.20 | 17.70 ± 0.10 *** | 3.20 ± 0.09 | 4.70 ± 0.10 ** |
| Dimers | 3.00 ± 0.20 | 16.70 ± 1.40 ** | 2.90 ± 0.20 | 4.20 ± 0.30 * |
| Trimers | 2.00 ± 0.20 | 13.00 ± 0.90 ** | 2.00 ± 0.01 | 3.20 ± 0.20 * |
| Tetramers | 1.30 ± 0.08 | 10.90 ± 0.07 *** | 1.50 ± 0.02 | 2.30 ± 0.10 ** |
| Pentamers | 0.77 ± 0.00 | 7.00 ± 0.06 *** | 0.97 ± 0.01 | 1.50 ± 0.00 *** |
| Hexamers | 0.51 ± 0.00 | 5.00 ± 0.40 ** | 0.66 ± 0.01 | 1.00 ± 0.02 ** |
| Heptamers | 0.13 ± 0.00 | 1.70 ± 0.30 * | 0.20 ± 0.01 | 0.30 ± 0.03 * |
| Octamers | 0.12 ± 0.02 | 0.90 ± 0.10 ** | 0.15 ± 0.01 | 0.20 ± 0.04 |
| Nonamers | 0.04 ± 0.01 | 0.47 ± 0.02 ** | 0.08 ± 0.02 | 0.14 ± 0.02 |
| Decamers | nd | 0.10 ± 0.10 | nd | 0.02 ± 0.01 |
| >Decamers | 6.20 ± 0.90 | 4.57 ± 0.19 | 3.30 ± 0.30 | 3.20 ± 1.10 |
| Total | 16.8 ± 1.6 | 78.0 ± 1.5 * | 15.0 ± 0.5 | 20.8 ± 0.9 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Barrio, R.; Nuñez-Gomez, V.; Cienfuegos-Jovellanos, E.; García-Alonso, F.J.; Periago-Castón, M.J. Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder. Foods 2020, 9, 189. https://doi.org/10.3390/foods9020189
González-Barrio R, Nuñez-Gomez V, Cienfuegos-Jovellanos E, García-Alonso FJ, Periago-Castón MJ. Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder. Foods. 2020; 9(2):189. https://doi.org/10.3390/foods9020189
Chicago/Turabian StyleGonzález-Barrio, Rocío, Vanesa Nuñez-Gomez, Elena Cienfuegos-Jovellanos, Francisco Javier García-Alonso, and Mª Jesús Periago-Castón. 2020. "Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder" Foods 9, no. 2: 189. https://doi.org/10.3390/foods9020189
APA StyleGonzález-Barrio, R., Nuñez-Gomez, V., Cienfuegos-Jovellanos, E., García-Alonso, F. J., & Periago-Castón, M. J. (2020). Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder. Foods, 9(2), 189. https://doi.org/10.3390/foods9020189





