Variations in Amino Acid and Protein Profiles in White versus Brown Teff (Eragrostis Tef) Seeds, and Effect of Extraction Methods on Protein Yields
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Plant Materials
2.2. Chemicals and Reagents
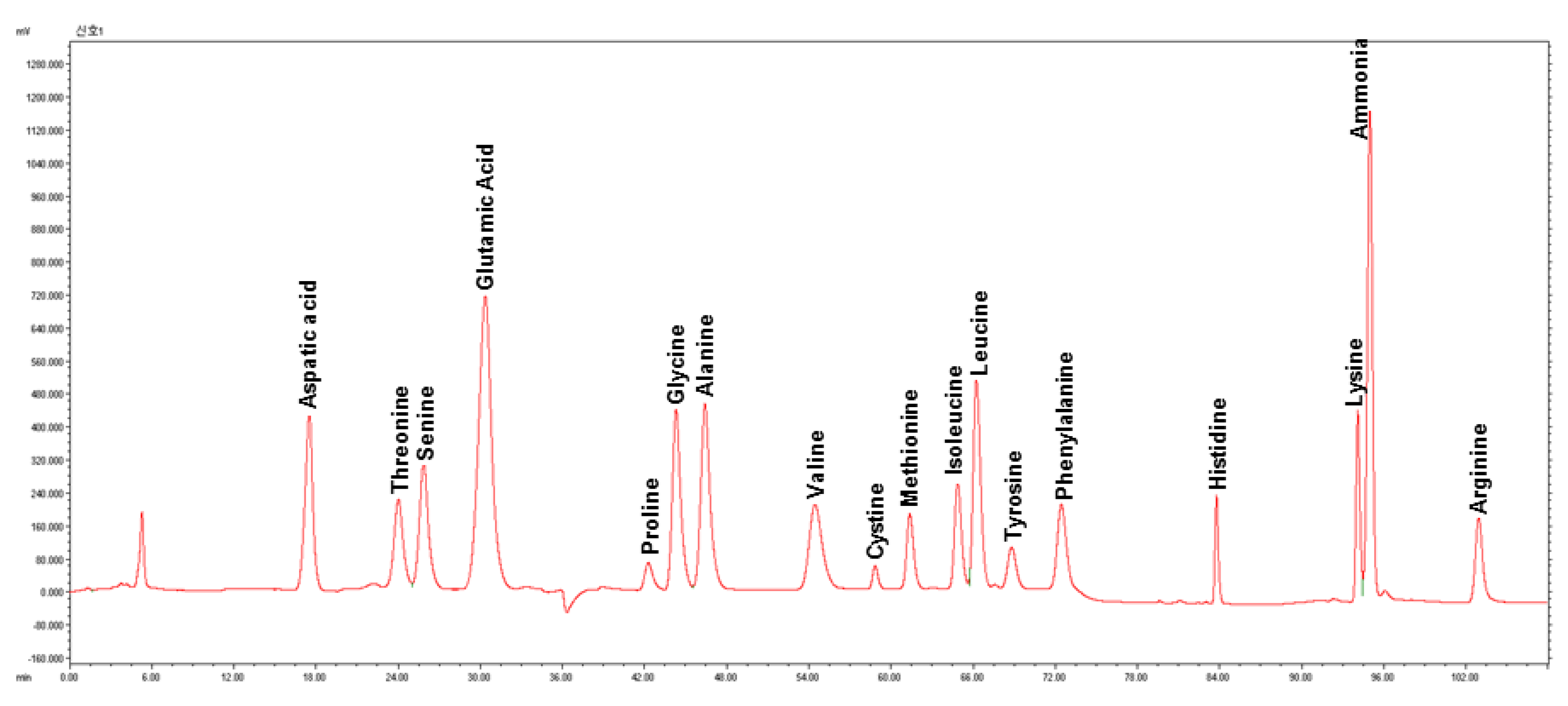
2.3. Amino Acid Analysis
2.4. Fractionation of Teff Proteins
2.5. Determination of Protein Concentrations
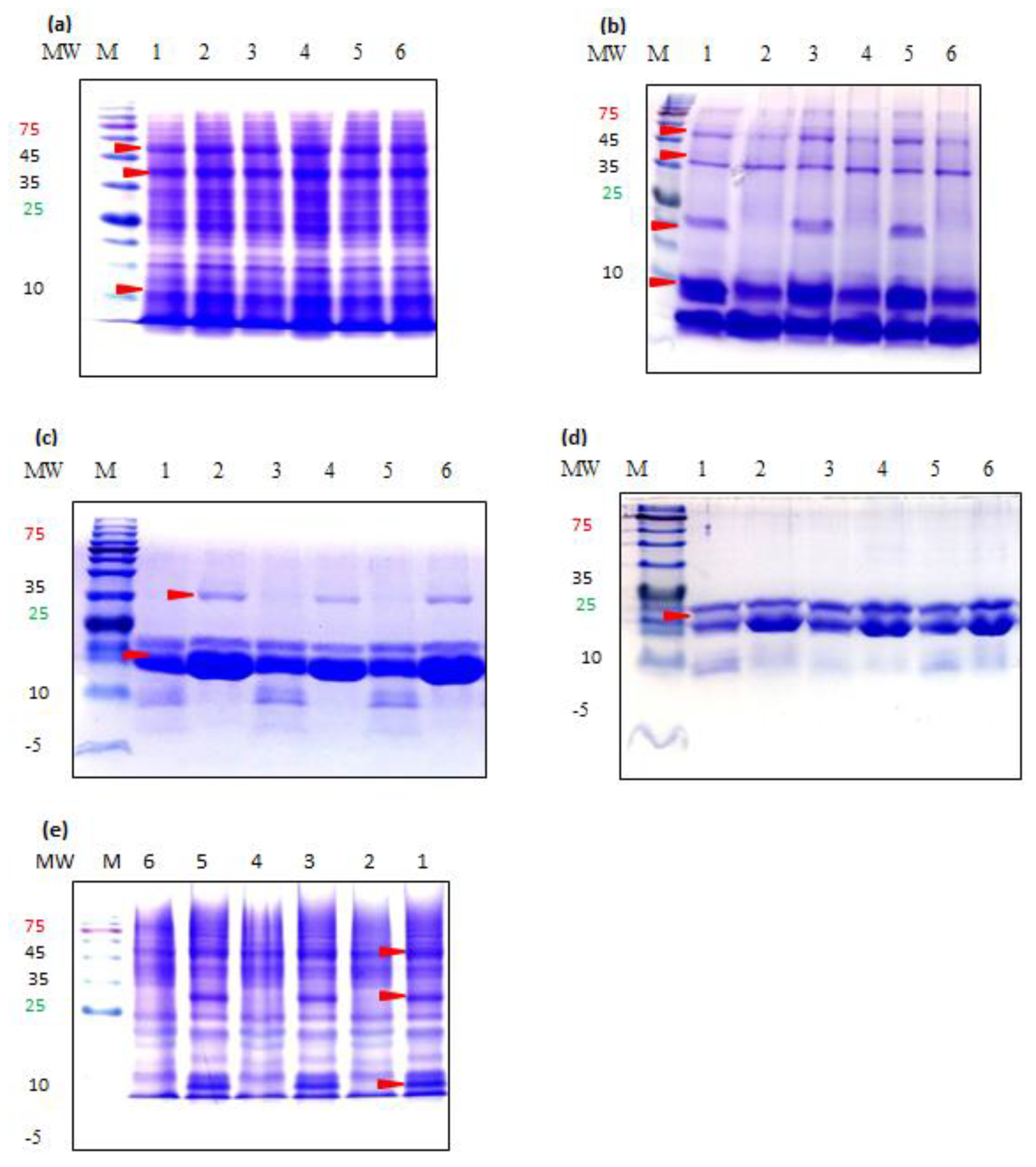
2.6. Sodium Dodecyl Sulfate Gel Electrophoresis (SDS-PAGE)
2.7. Statistical Analysis
3. Results
3.1. Physical Characteristics of Teff Seeds
3.2. Amino Acid Compositions
3.3. Effect of Extraction Methods on the Yield of TSSP
3.4. Molecular Weight Distribution of Teff Proteins
4. Discussions
4.1. Physical Characteristics
4.2. Amino Acid Compositions
4.3. Effect of Extraction Methods on the Yield of TSSP
4.4. SDS-PAGE
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Cannarozzi, G.; Plaza-Wüthrich, S.; Esfeld, K.; Larti, S.; Wilson, Y.; Girma, D.; Tadele, Z. Genome and transcriptome sequencing identifies breeding targets in the orphan crop tef (Eragrostis tef). BMC Genomics 2014, 15, 581. [Google Scholar] [CrossRef] [PubMed]
- Mezemir, S. Probiotic potential and nutritional importance of teff (eragrostis tef (zucc) trotter) enjerra-a review. Afr. J. Food Agric. Nutr. Dev. 2015, 15, 9964–9981. [Google Scholar]
- Shumoy, H.; Raes, K. Antioxidant Potentials and Phenolic Composition of Tef Varieties: An Indigenous Ethiopian Cereal. Cereal Chem. 2016, 93, 465–470. [Google Scholar] [CrossRef]
- Adebowale, A.-R.A.; Emmambux, M.N.; Beukes, M.; Taylor, J.R.N. Fractionation and characterization of teff proteins. J. Cereal Sci. 2011, 54, 380–386. [Google Scholar] [CrossRef]
- Baye, K. Teff: Nutrient Composition and Health Benefits. ESSP Working Paper 67. Available online: https://www.google.ch/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwjt7tfE09fiAhUiy4UKHZzTDtcQFjAAegQIAxAC&url=https%3A%2F%2Fwww.researchgate.net%2Fprofile%2FArvind_Singh56%2Fpost%2FWhat_does_the_global_production_and_consumption_of_teff_look_like%2Fattachment%2F59d64e4379197b80779a7a39%2FAS%253A492060228108288%25401494327708660%2Fdownload%2FBaye%2Bet%2Bal.%252C%2B2014.pdf&usg=AOvVaw0hh_EIUpZuCpZA_qq9qmDc (accessed on 05 January 2019).
- Zhang, W.; Xu, J.; Bennetzen, J.L.; Messing, J. Teff, an Orphan Cereal in the Chloridoideae, Provides Insights into the Evolution of Storage Proteins in Grasses. Genome Biol. Evol. 2016, 8, 1712–1721. [Google Scholar] [CrossRef] [PubMed]
- Gorinstein, S.; Pawelzik, E.; Delgado-Licon, E.; Haruenkit, R.; Weisz, M.; Trakhtenberg, S. Characterisation of pseudocereal and cereal proteins by protein and amino acid analyses. J. Sci. Food Agric. 2002, 82, 886–891. [Google Scholar] [CrossRef]
- Mota, C.; Santos, M.; Mauro, R.; Samman, N.; Matos, A.S.; Torres, D.; Castanheira, I. Protein content and amino acids profile of pseudocereals. Food Chem. 2016, 193, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Tovar-Pérez, E.G.; Guerrero-Legarreta, I.; Farrés-González, A.; Soriano-Santos, J. Angiotensin I-converting enzyme-inhibitory peptide fractions from albumin 1 and globulin as obtained of amaranth grain. Food Chem. 2009, 116, 437–444. [Google Scholar] [CrossRef]
- Leyva-Lopez, N.E.; Vasco, N.; Barba de la Rosa, A.P.; Paredes-Lopez, O. Amaranth seed proteins: Effect of defatting on extraction yield and on electrophoretic patterns. Plant Foods Hum. Nutr. 1995, 47, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Zheleznov, A.V.; Solonenko, L.P.; Zheleznova, N.B. Seed proteins of the wild and the cultivated Amaranthus species. Euphytica 1997, 97, 177–182. [Google Scholar] [CrossRef]
- Tömösközi, S.; Gyenge, L.; Pelcéder, Á.; Abonyi, T.; Schönlechner, R.; Lásztity, R. Effects of Flour and Protein Preparations from Amaranth and Quinoa Seeds on the Rheological Properties of Wheat-Flour Dough and Bread Crumb. Czech J. Food Sci. 2011, 29, 109–116. [Google Scholar] [CrossRef]
- El-Alfy, T.S.; Ezzat, S.M.; Sleem, A.A. Chemical and biological study of the seeds of Eragrostis tef (Zucc.) Trotter. Nat. Prod. Res. 2012, 26, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F. Chemical composition and food uses of teff (Eragrostis tef). Food Chem. 2018, 239, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Tatham, A.S.; Fido, R.J.; Moore, C.M.; Kasarda, D.D.; Kuzmicky, D.D.; Keen, J.N.; Shewry, P.R. Characterisation of the Major Prolamins of Tef (Eragrostis tef ) and Finger Millet (Eleusine coracana). J. Cereal Sci. 1996, 24, 65–71. [Google Scholar] [CrossRef]
- Spackman, D.H.; Stein, W.H.; Moore, S. Automatic Recording Apparatus for Use in Chromatography of Amino Acids. J. Anal. Chem. 1958, 30, 1190–1206. [Google Scholar] [CrossRef]
- Tella, A.F.; Ojehomon, O.O. An extraction method for evaluating the seed proteins of cowpea (Vigna unguiculata (L) Walp). J. Sci. Food Agric. 1980, 31, 1268–1274. [Google Scholar] [CrossRef]
- Wallace, J.C.; Lopes, M.A.; Paiva, E.; Larkins, B.A. New Methods for Extraction and Quantitation of Zeins Reveal a High Content of gamma-Zein in Modified opaque-2 Maize. Plant Physiol. 1990, 92, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Van der Walt, W.H.; Schussler, L.; Van der Walt, W.H. Fractionation of proteins from low-tannin sorghum grain. J. Agric. Food Chem. 1984, 32, 149–154. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. J. Anal. Chem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Ravisankar, S.; Abegaz, K.; Awika, J.M. Structural profile of soluble and bound phenolic compounds in teff (Eragrostis tef) reveals abundance of distinctly different flavones in white and brown varieties. Food Chem. 2018, 263, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Antwi-Boasiako, C.; Animapauh, S.O. Tannin extraction from the barks of three tropical hardwoods for the production of adhesives. J. Appl. Sci. Res. 2005, 8, 2959–2965. [Google Scholar]
- Chavan, J.K.; kadam, S.S.; Ghonsikar, C.P.; Salunkhe, D.K. Removal of tannins and improvement of in vitro protein digestibility of sorghum seeds by soaking in alkali. J. Food Sci. 1979, 44, 1319–1322. [Google Scholar] [CrossRef]
- Tessari, P.; Lante, A.; Mosca, G. Essential amino acids: Master regulators of nutrition and environmental footprint? Sci. Rep. 2016, 6, 26074. [Google Scholar] [CrossRef] [PubMed]
- Palombini, S.V.; Claus, T.; Maruyama, S.A.; Gohara, A.K.; Souza, A.H.P.; de Souza, N.E.; Visentainer, J.V.; Gomes, S.T.M.; Matsushita, M. Evaluation of nutritional compounds in new amaranth and quinoa cultivars. Food Sci. Technol. 2013, 33, 339–344. [Google Scholar] [CrossRef]
- Mcdermott, E.E.; Pace, J. The content of amino-acids in white flour and bread. Br. J. Nutr. 1957, 11, 446–452. [Google Scholar] [CrossRef]
- Mckevith, B. Nutritional aspects of cereals. Food Nutr. Bull. 2004, 29, 111–142. [Google Scholar] [CrossRef]
- Thomas, R.; Bhat, R.; Kuang, Y.T. Composition of amino Acids, fatty acids, minerals and dietary fiber in some of the local and import rice varieties of Malaysia. Int. Food Res. J. 2015, 22, 1148–1155. [Google Scholar]
- Ferreira, R.R.; Varisi, V.A.; Meinhardt, L.W.; Lea, P.J.; Azevedo, R.A. Are high-lysine cereal crops still a challenge? Braz. J. Med. Biol. Res. 2005, 38, 985–994. [Google Scholar] [CrossRef]
- Dennison, C.; Lizette, M.; Che, S.P. t-Butanol: Nature’s gift for protein isolation. S. Afr. J. Sci. 2000, 96, 159–160. [Google Scholar]
- Gonzalez, J.A.; Konishi, Y.; Bruno, M.; Valoy, M.; Prado, F.E. Interrelationships among seed yield, total protein and amino acid composition of ten quinoa (Chenopodium quinoa) cultivars from two different agroecological regions. J. Sci. Food Agric. 2012, 92, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Mainieri, D.; Marrano, C.A.; Prinsi, B.; Maffi, D.; Tschofen, M.; Espen, L.; Vitale, A. Maize 16-kD γ-zein forms very unusual disulfide-bonded polymers in the endoplasmic reticulum: Implications for prolamin evolution. J. Exp. Bot. 2018, 69, 5013–5027. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, H.C.; Paulis, J.W.; James, C.; Wall, J.S. Extraction and structure studies on corn glutelin proteins. Cereal Chem. 1970, 47, 501. [Google Scholar]
- Paulis, J.W.; Wall, J.S. Fractionation and properties of alkylated-reduced corn glutelin proteins. BBA 1971, 251, 57–69. [Google Scholar] [CrossRef]
- Getz, E.B.; Xiao, M.; Chakrabarty, T.; Cooke, R.; Selvin, P.R. A Comparison between the Sulfhydryl Reductants Tris(2-carboxyethyl)phosphine and Dithiothreitol for Use in Protein Biochemistry. Anal. Biochem. 1999, 273, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Singh Khatkar, B. Structure and functionality of wheat gluten proteins. In Proceedings of the 2014 International Conference of Food Properties (ICFP 2014), Kuala Lumpur, Malaysia, 24–26 January 2014. [Google Scholar]
- Coleman, J.M. Assessing the Potential Use of Teff as an Alternative Grain Crop in Virginia. MSc Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, April 2012. [Google Scholar]
- Žilić, S.; Barać, M.; Pešić, M.; Dodig, D.; Ignjatović-Micić, D. Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes. Int. J. Mol. Sci. 2011, 12, 5878–5894. [Google Scholar] [CrossRef]
- Vollmann, J.; Fritz, C.N.; Wagentristl, H.; Ruckenbauer, P. Environmental and genetic variation of soybean seed protein content under Central European growing conditions. J. Sci. Food Agric. 2000, 80, 1300–1306. [Google Scholar] [CrossRef]
- Javaid, A.; Ghafoor, A.; Anwar, R. Seed storage protein electrophoresis in groundnut for evaluating genetic diversity. Pak. J. Bot. 2004, 36, 25–29. [Google Scholar]
- Sadia, M.; Malik, S.A.; Rabbani, M.A.; Pearce, S.R. Electrophoretic Characterization and the Relationship between some Brassica Species. Electron. J. Bio. 2009, 5, 1–4. [Google Scholar]

| Amino Acid | Samples | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Addis-W | Ratio (%) | Addis-B | Ratio (%) | Mekel-W | Ratio (%) | Mekel-B | Ratio (%) | Debre-W | Ratio (%) | Debre-B | Ratio (%) | ||
| Essential Amino Acids | Thr | 6.73 ± 0.35 | 4.35 | 10.76 ± 0.66 | 4.48 | 7.53 ± 0.15 | 4.68 | 10.45 ± 0.07 | 4.78 | 6.90 ± 0.14 | 4.52 | 10.00 ± 0.00 | 4.46 |
| Val | 5.60 ± 0.20 | 3.62 | 8.63 ± 0.32 | 3.59 | 5.66 ± 0.32 | 3.52 | 8.35 ± 0.21 | 3.82 | 6.80 ± 0.28 | 4.46 | 9.20 ± 0.84 | 4.1 | |
| Met | 4.10 ± 0.17 | 2.65 | 7.16 ± 0.32 | 2.98 | 4.66 ± 0.49 | 2.9 | 6.15 ± 0.35 | 2.81 | 4.80 ± 0 | 3.15 | 6.80 ± 0.28 | 3.03 | |
| Ile | 9.23 ± 0.05 | 5.96 | 14.13 ± 0.11 | 5.88 | 9.20 ± 0.45 | 5.72 | 13.85 ± 0.63 | 6.33 | 9.60 ± 0.00 | 6.29 | 14.20 ± 0.28 | 6.33 | |
| Leu | 11.40 ± 0.26 | 7.36 | 17.53 ± 0.28 | 7.29 | 11.73 ± 0.64 | 7.29 | 16.70 ± 0.14 | 7.64 | 12.10 ± 0.42 | 7.93 | 18.30 ± 0.42 | 8.16 | |
| Phe | 8.23 ± 0.58 | 5.31 | 12.60 ± 0.36 | 5.24 | 8.43 ± 0.3 | 5.24 | 11.35 ± 0.49 | 5.19 | 8.90 ± 0.70 | 5.83 | 13.60 ± 0.84 | 6.07 | |
| His | 2.96 ± 0.05 | 1.91 | 4.43 ± 0.05 | 1.84 | 3.13 ± 0.11 | 1.94 | 4.15 ± 0.21 | 1.9 | 3.10 ± 0.14 | 2.03 | 15.60 ± 1.69 | 6.96 | |
| Lys | 12.00 ± 0.17 | 7.75 | 15.10 ± 0.40 | 6.28 | 12.16 ± 0.6 | 7.56 | 13.45 ± 0.49 | 6.15 | 14.30 ± 0.70 | 9.37 | 16.80 ± 0.00 | 7.49 | |
| Subtotal | 60.25 | 38.91 | 90.34 | 37.58 | 62.5 | 38.85 | 84.45 | 38.62 | 66.5 | 43.58 | 104.5 | 46.6 | |
| Other amino acids | Asp | 15.93 ± 0.55 | 10.29 | 19.23 ± 0.86 | 8 | 15.06 ± 0.75 | 9.36 | 18.30 ± 0.98 | 8.37 | 15.60 ± 0.84 | 10.22 | 19.60 ± 0.00 | 8.74 |
| Ser | 7.16 ± 0.55 | 4.62 | 11.06 ± 0.66 | 4.6 | 7.86 ± 0.25 | 4.88 | 10.90 ± 0.14 | 4.99 | 7.10 ± 0.42 | 4.65 | 11.20 ± 0.00 | 5 | |
| Glu | 38.90 ± 0.34 | 25.12 | 75.86 ± 8.25 | 31.56 | 43.86 ± 1.78 | 27.25 | 63.35 ± 9.26 | 28.97 | 33.50 ± 0.98 | 21.95 | 52.10 ± 2.68 | 23.24 | |
| Pro | 1.86 ± 0.11 | 1.2 | 2.96 ± 0.32 | 1.23 | 1.96 ± 0.32 | 1.22 | 2.95 ± 0.07 | 1.35 | 2.10 ± 0.42 | 1.38 | 3.40 ± 0.00 | 1.52 | |
| Gly | 5.76 ± 0.15 | 3.72 | 7.50 ± 0.26 | 3.12 | 6.00 ± 0.40 | 3.73 | 6.90 ± 0.28 | 3.16 | 5.90 ± 0.42 | 3.87 | 7.60 ± 0.28 | 3.39 | |
| Ala | 7.06 ± 0.15 | 4.56 | 10.40 ± 0.26 | 4.33 | 7.50 ± 0.51 | 4.66 | 12.15 ± 2.75 | 5.56 | 9.00 ± 0.28 | 5.9 | 10.40 ± 0.00 | 4.64 | |
| Cys | 0.63 ± 0.05 | 0.41 | 1.13 ± 0.05 | 0.47 | 0.63 ± 0.05 | 0.39 | 0.85 ± 0.07 | 0.39 | 0.4 ± 0.00 | 0.26 | ND | ND | |
| Tyr | 3.36 ± 0.05 | 2.17 | 5.46 ± 0.05 | 2.27 | 3.43 ± 0.15 | 2.13 | 5.05 ± 0.07 | 2.31 | ND | ND | 3.60 ± 0.00 | 1.61 | |
| Arg | 13.96 ± 1.22 | 9.01 | 16.43 ± 0.32 | 6.84 | 12.13 ± 0.75 | 7.54 | 13.75 ± 0.49 | 6.29 | 12.5 ± 0.98 | 8.19 | 11.80 ± 1.41 | 5.26 | |
| Subtotal | 94.62 | 61.1 | 150.03 | 62.42 | 98.43 | 61.16 | 134.2 | 61.39 | 86.1 | 56.42 | 119.7 | 53.4 | |
| Total | 154.87 ± 1.01 | 100 | 240.37 ± 4.17 | 100 | 160.93 ± 7.80 | 100 | 218.65 ± 8.27 | 100 | 152.6 ± 4.24 | 100 | 224.2 ± 14.56 | 100 | |
| Sample | TSSPF | Method 1 | Method 2 | Method 3 | |||
|---|---|---|---|---|---|---|---|
| Yield (g/100g flour) | Ratio (%) | Yield (g/100g flour) | Ratio (%) | Yield (g/100g flour) | Ratio (%) | ||
| Addis-W | Albumin | 2.13 ± 0.22 | 31.88 | 2.13 ± 0.16 | 29.7 | 2.06 ± 0.09 | 23.01 |
| Globulin | 0.38 ± 0.01 | 5.71 | 0.38 ± 0.01 | 5.31 | 0.36 ± 0.05 | 3.97 | |
| Prolamin | 0.10 ± 0.03 a | 1.46 | 0.43 ± 0.01 b | 5.96 | 1.62 ± 0.18 c | 18.09 | |
| Glutelin | 4.07 ± 0.30 a | 60.94 | 4.23 ± 0.25 a | 59.04 | 4.93 ± 0.19 b | 54.92 | |
| Total | 6.69 ± 0.55 a | 100 | 7.16 ± 0.41 a | 100 | 8.97 ± 0.27 b | 100 | |
| Addis-B | Albumin | 2.43 ± 0.29 | 38.37 | 2.50 ± 0.24 | 33.97 | 2.53 ± 0.15 | 26.98 |
| Globulin | 0.52 ± 0.12 | 8.28 | 0.55 ± 0.08 | 7.44 | 0.38 ± 0.29 | 4.08 | |
| Prolamin | 0.19 ± 0.03 a | 3.02 | 0.94 ± 0.02 b | 12.77 | 2.37 ± 0.32 c | 25.29 | |
| Glutelin | 3.19 ± 0.18 a | 50.33 | 3.37 ± 0.22 a | 45.81 | 4.09 ± 0.27 b | 43.64 | |
| Total | 6.34 ± 0.43 a | 100 | 7.35 ± 0.38 b | 100 | 9.37 ± 0.39 c | 100 | |
| Mekel-W | Albumin | 1.88 ± 0.14 | 41.81 | 2.01 ± 0.13 | 38.13 | 2.10 ± 0.16 | 31.37 |
| Globulin | 0.37 ± 0.01 | 8.26 | 0.26 ± 0.04 | 5.01 | 0.30 ± 0.01 | 4.46 | |
| Prolamin | 0.19 ± 0.01 a | 4.12 | 0.68 ± 0.05 b | 12.98 | 1.75 ± 0.11 c | 26.22 | |
| Glutelin | 2.06 ± 0.08 a | 45.81 | 2.31 ± 0.10 bc | 43.88 | 2.54 ± 0.10 c | 37.95 | |
| Total | 4.50 ± 0.23 a | 100 | 5.26 ± 0.19 b | 100 | 6.69 ± 0.36 c | 100 | |
| Mekel-B | Albumin | 1.53 ± 0.21 | 38.84 | 1.26 ± 0.17 | 25.36 | 1.71 ± 0.23 | 26.07 |
| Globulin | 0.17 ± 0.02 | 4.27 | 0.14 ± 0.02 | 2.88 | 0.13 ± 0.02 | 2.06 | |
| Prolamin | 0.24 ± 0.01 a | 6 | 1.23 ± 0.10 b | 24.73 | 2.24 ± 0.05 c | 34.16 | |
| Glutelin | 2.01 ± 0.08 a | 50.89 | 2.33 ± 0.07 bc | 47.03 | 2.47 ± 0.10 c | 37.71 | |
| Total | 3.94 ± 0.16 a | 100 | 4.96 ± 0.32 b | 100 | 6.55 ± 0.21 c | 100 | |
| Debre-W | Albumin | 1.82 ± 0.13 | 27.57 | 2.36 ± 0.14 | 30.8 | 2.42 ± 0.19 | 29.53 |
| Globulin | 0.29 ± 0.04 | 4.36 | 0.39 ± 0.01 | 5.13 | 0.38 ± 0.07 | 4.62 | |
| Prolamin | 0.23 ± 0.02 a | 3.5 | 0.46 ± 0.01 b | 5.96 | 1.55 ± 0.28 c | 18.87 | |
| Glutelin | 4.27 ± 0.20 | 64.57 | 4.44 ± 0.14 | 58.11 | 3.85 ± 0.18 | 46.98 | |
| Total | 6.61 ± 0.30 a | 100 | 7.65 ± 0.25 ab | 100 | 8.20 ± 0.62 b | 100 | |
| Debre-B | Albumin | 2.97 ± 0.27 | 42.14 | 2.82 ± 0.21 | 35.78 | 2.65 ± 0.16 | 30.29 |
| Globulin | 0.50 ± 0.10 | 7.03 | 0.61 ± 0.08 | 7.75 | 0.44 ± 0.07 | 5.05 | |
| Prolamin | 0.38 ± 0.05 a | 5.39 | 1.02 ± 0.03 b | 12.98 | 2.51 ± 0.22 c | 28.71 | |
| Glutelin | 3.20 ± 0.23 | 45.43 | 3.42 ± 0.19 | 43.48 | 3.15 ± 0.18 | 35.95 | |
| Total | 7.05 ± 0.53 a | 100 | 7.87 ± 0.35 ab | 100 | 8.75 ± 0.14 b | 100 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gebru, Y.A.; Hyun-II, J.; Young-Soo, K.; Myung-Kon, K.; Kwang-Pyo, K. Variations in Amino Acid and Protein Profiles in White versus Brown Teff (Eragrostis Tef) Seeds, and Effect of Extraction Methods on Protein Yields. Foods 2019, 8, 202. https://doi.org/10.3390/foods8060202
Gebru YA, Hyun-II J, Young-Soo K, Myung-Kon K, Kwang-Pyo K. Variations in Amino Acid and Protein Profiles in White versus Brown Teff (Eragrostis Tef) Seeds, and Effect of Extraction Methods on Protein Yields. Foods. 2019; 8(6):202. https://doi.org/10.3390/foods8060202
Chicago/Turabian StyleGebru, Yoseph Asmelash, Jun Hyun-II, Kim Young-Soo, Kim Myung-Kon, and Kim Kwang-Pyo. 2019. "Variations in Amino Acid and Protein Profiles in White versus Brown Teff (Eragrostis Tef) Seeds, and Effect of Extraction Methods on Protein Yields" Foods 8, no. 6: 202. https://doi.org/10.3390/foods8060202
APA StyleGebru, Y. A., Hyun-II, J., Young-Soo, K., Myung-Kon, K., & Kwang-Pyo, K. (2019). Variations in Amino Acid and Protein Profiles in White versus Brown Teff (Eragrostis Tef) Seeds, and Effect of Extraction Methods on Protein Yields. Foods, 8(6), 202. https://doi.org/10.3390/foods8060202





