Robust Biofilm-Forming Bacillus Isolates from the Dairy Environment Demonstrate an Enhanced Resistance to Cleaning-in-Place Procedures
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Generation of Biofilm-Derived Spores
2.3. Staining Extracellular Matrix of Biofilm-Derived Spores
2.4. Preparation for Cleaning Tests and Enumeration of Biofilm-Derived Spores
2.5. Cleaning Solutions
2.6. Cleaning Test Installations
2.6.1. CIP Model System
2.6.2. Laboratory System
2.7. Evaluation of the Effect of the Cleaning Agents on the Viability of Bacillus Spores
2.8. Statistical Analysis
3. Results
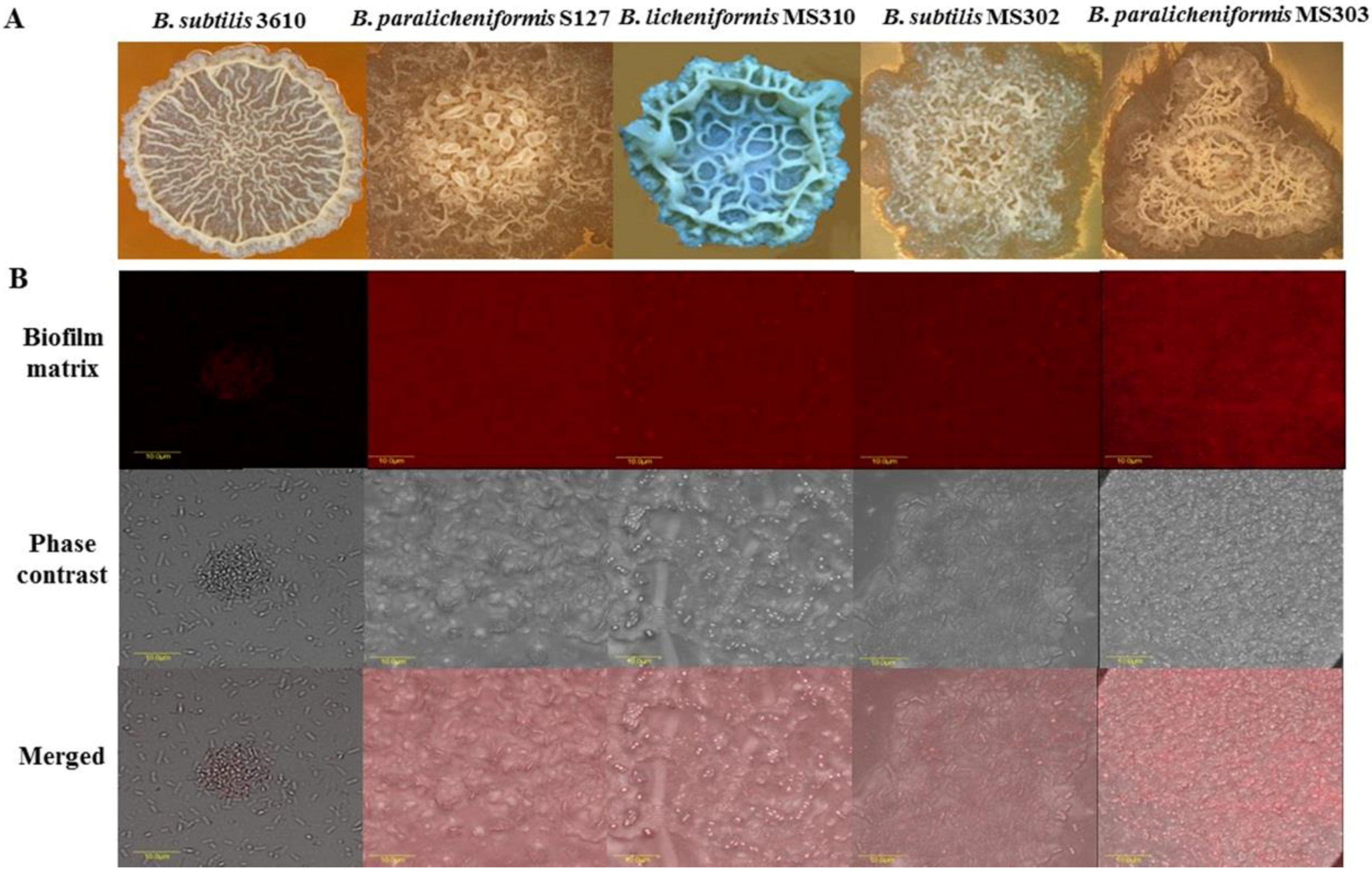
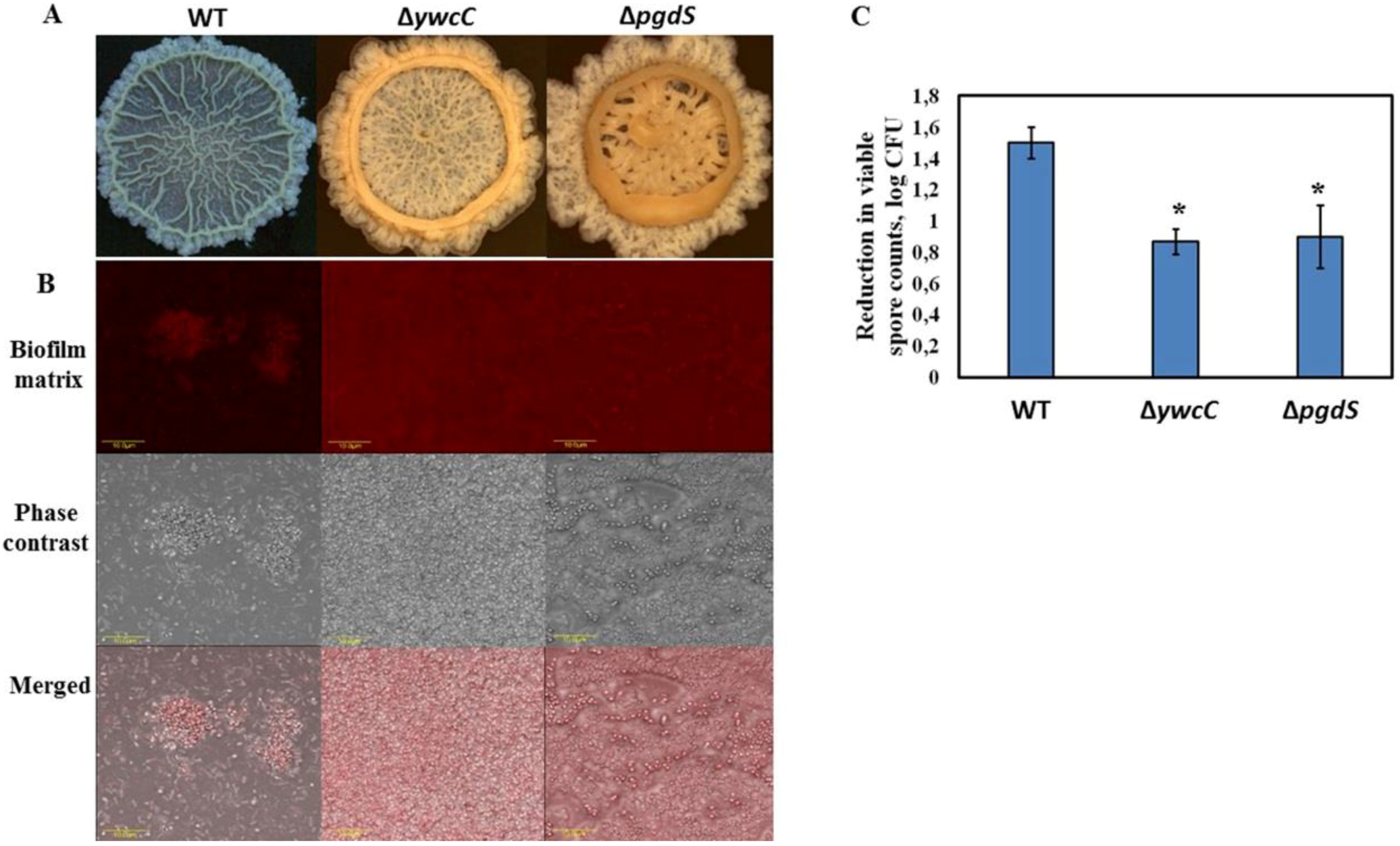
3.1. Dairy-Associated Bacillus Isolates Exhibit Robust Biofilm Phenotype Compared to B. subtilis 3610
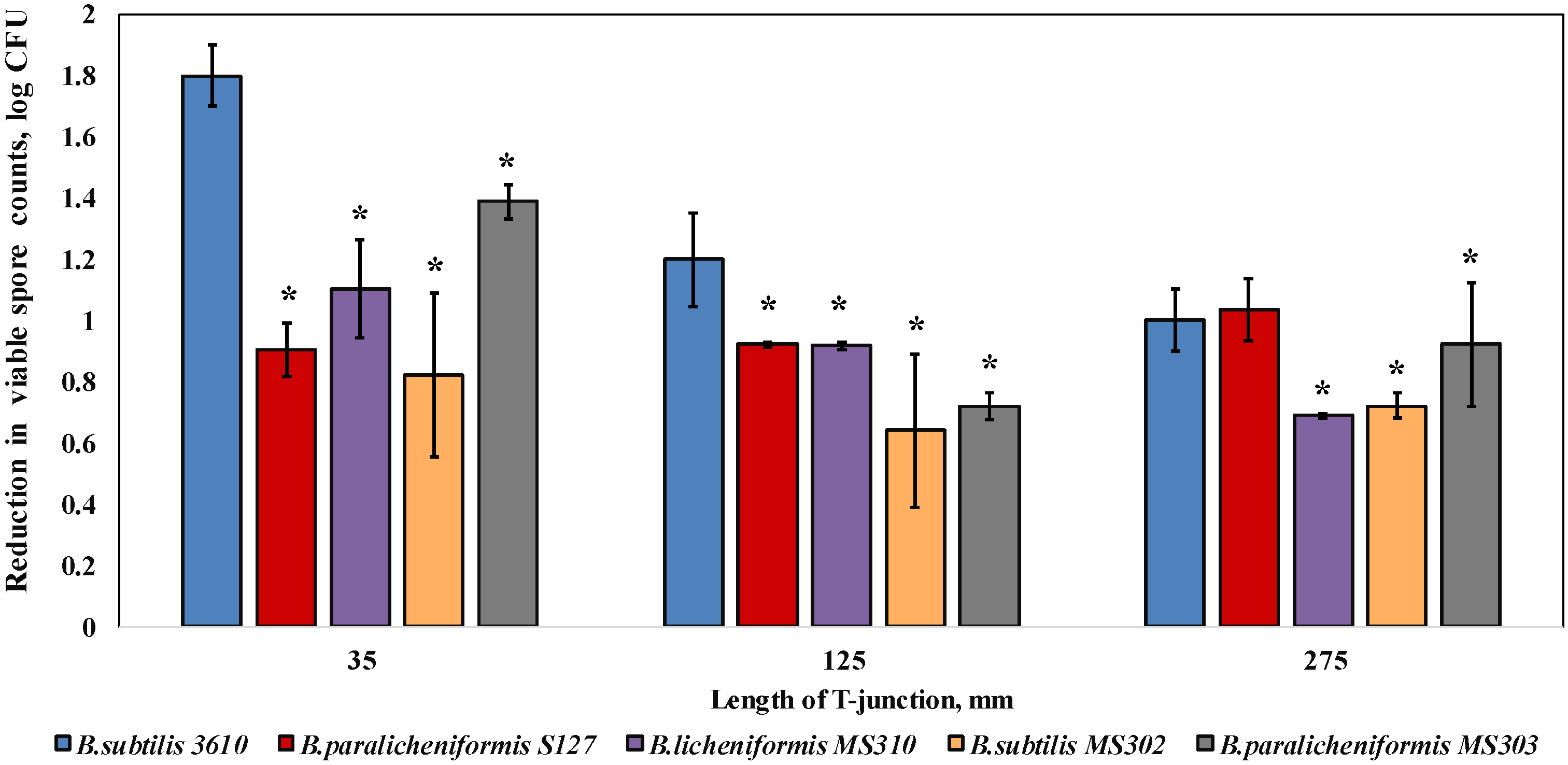
3.2. Dairy-Associated Bacillus Isolates Display an Enhanced Resistance to the Mechanical Effect of Water Circulation
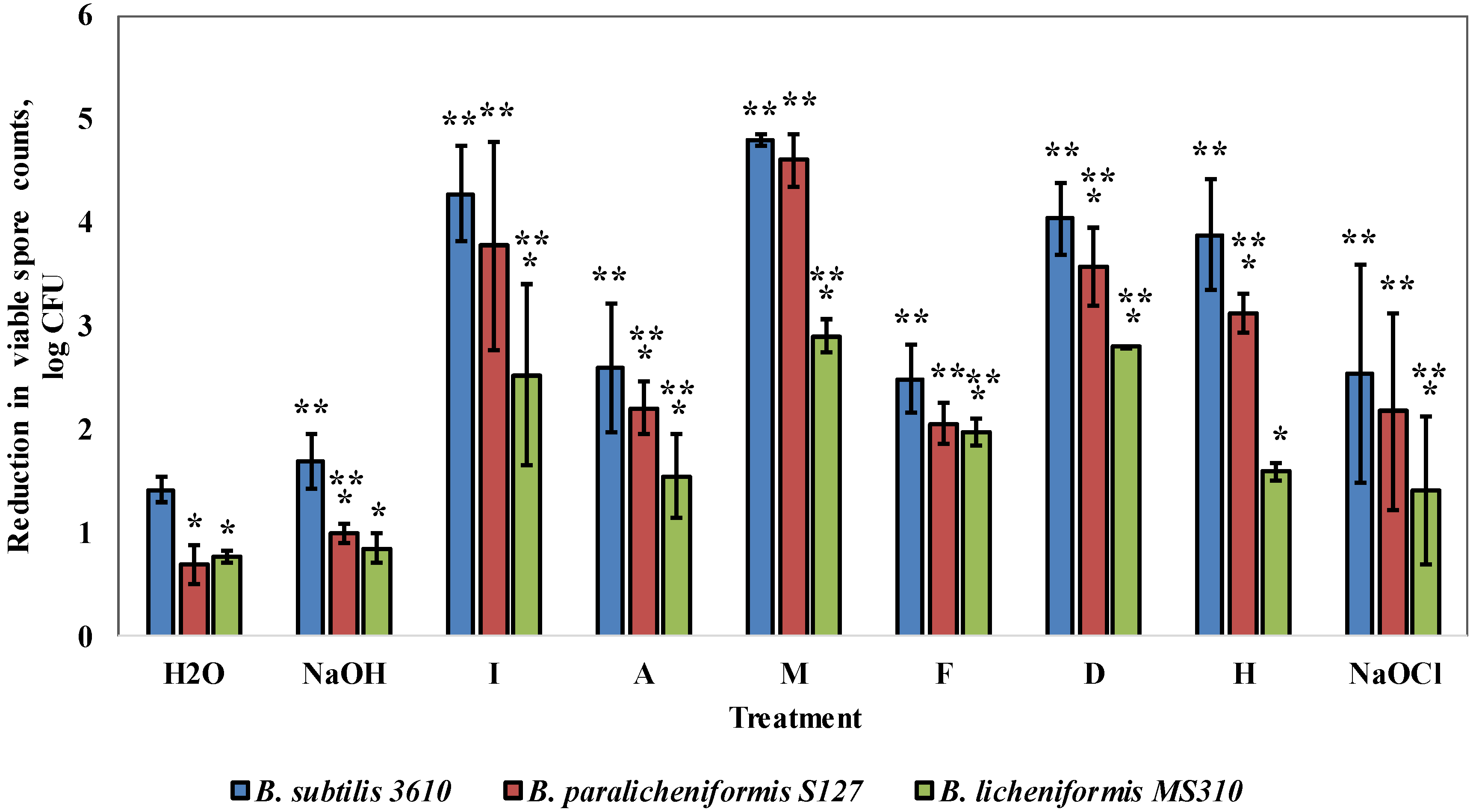
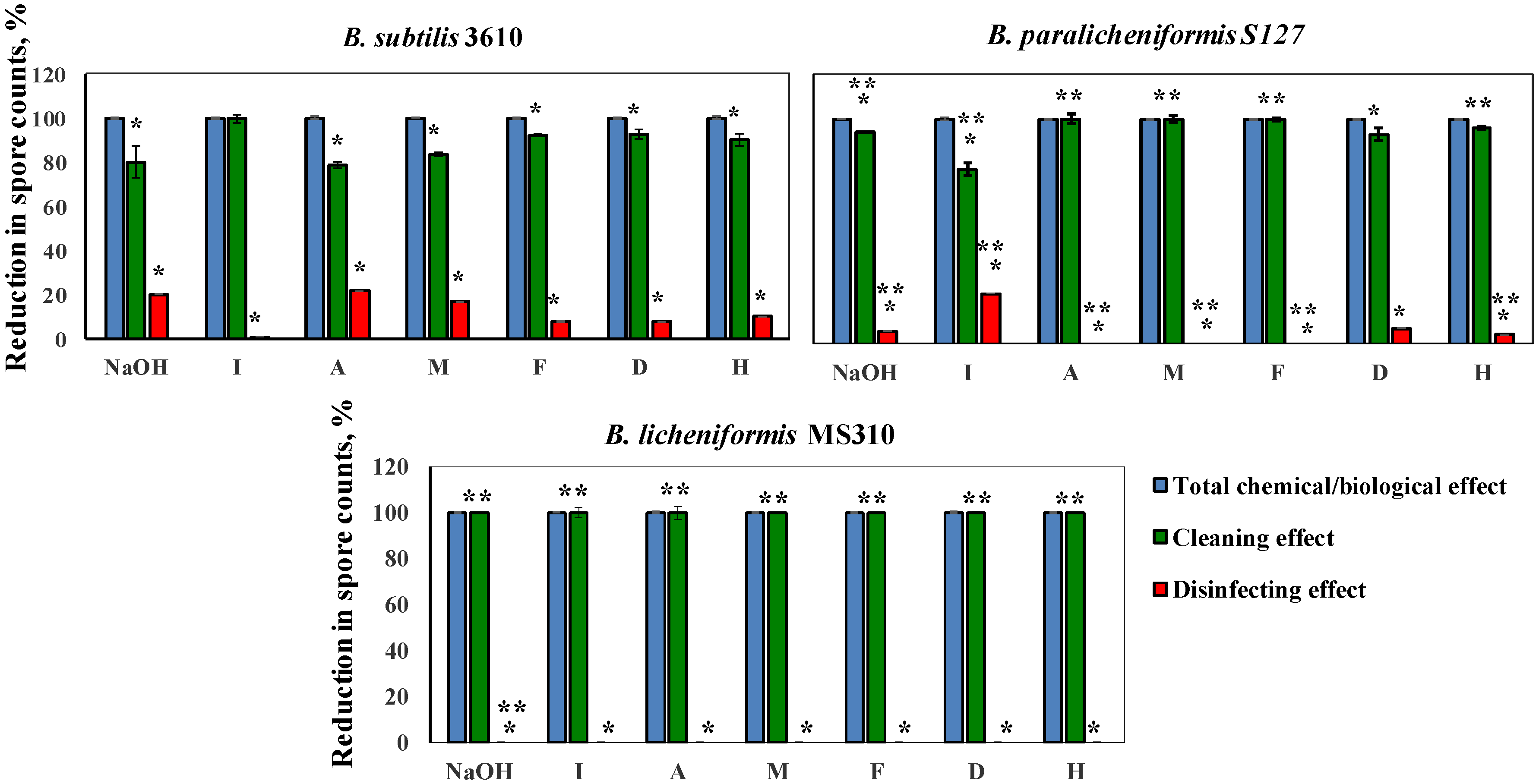
3.3. Dairy-Associated Bacillus Isolates Demonstrate an Enhanced Resistance to Commercial Cleaning Agents during CIP Procedures
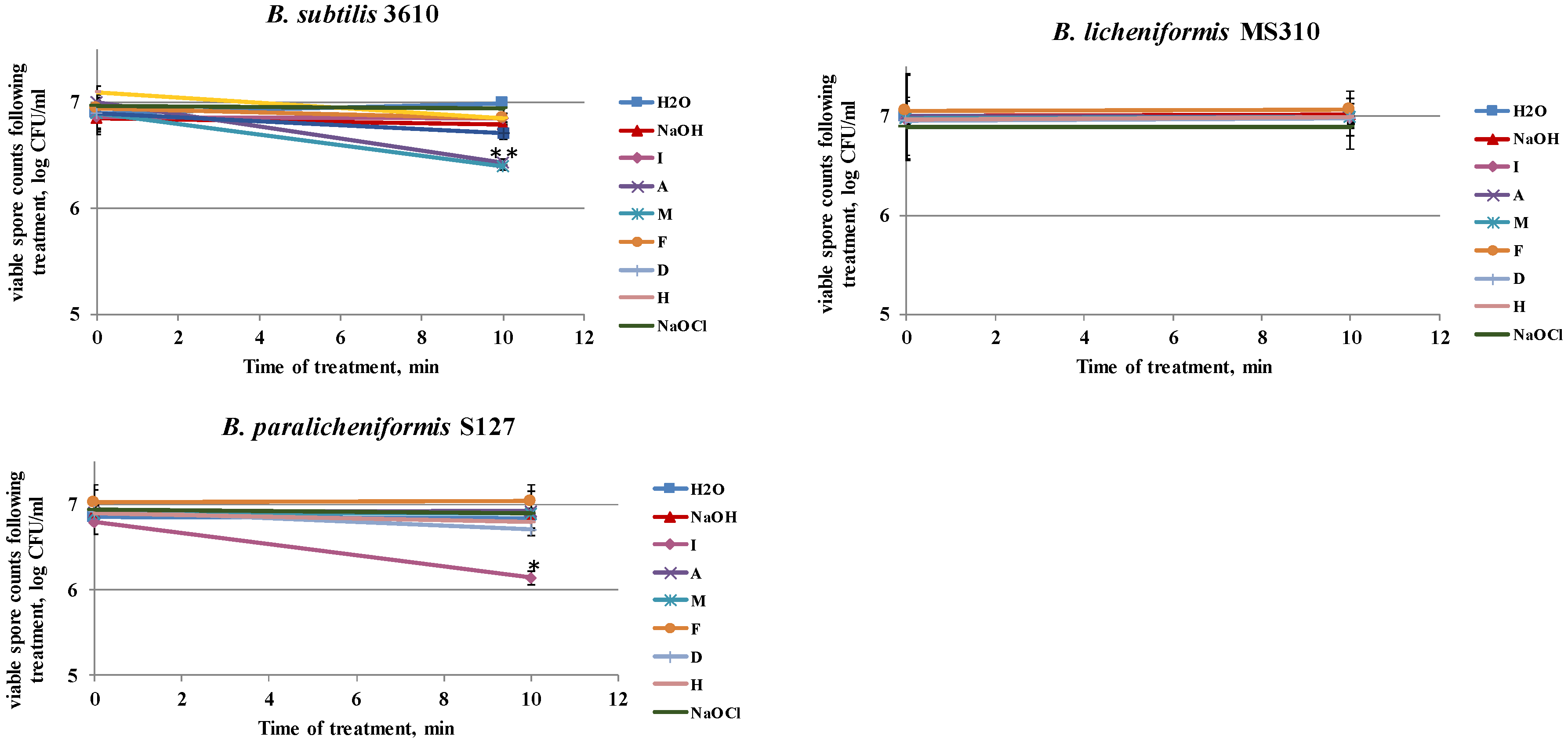
3.4. Dairy-Associated Bacillus Isolates Demonstrate an Enhanced Resistance to the Disinfecting Effect of the Tested Agents
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ivy, R.A.; Ranieri, M.L.; Martin, N.H.; den Bakker, H.C.; Xavier, B.M.; Wiedmann, M.; Boor, K.J. Identification and Characterization of Psychrotolerant Sporeformers Associated with Fluid Milk Production and Processing. Appl. Environ. Microbiol. 2012, 78, 1853–1864. [Google Scholar] [CrossRef]
- Ranieri, M.L.; Huck, J.R.; Sonnen, M.; Barbano, D.M.; Boor, K.J. High temperature, short time pasteurization temperatures inversely affect bacterial numbers during refrigerated storage of pasteurized fluid milk. J. Dairy Sci. 2009, 92, 4823–4832. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.G.; Anand, S.K. Significance of Microbial Biofilms in Food Industry: A Review. Int. J. Food Microbiol. 1998, 42, 9–27. [Google Scholar] [CrossRef]
- Bremer, P.J.; Fillery, S.; McQuillan, A.J. Laboratory Scale Clean-In-Place (CIP) Studies on the Effectiveness of Different Caustic and Acid Wash Steps on the Removal of Dairy Biofilms. Int. J. Food Microbiol. 2006, 106, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Flint, S.H.; Bremer, P.J.; Brooks, J.D. Biofilms in Dairy Manufacturing Plant-Description, Current Concerns and Methods of Control. Biofouling 1997, 11, 81–97. [Google Scholar] [CrossRef]
- Pasvolsky, R.; Zakin, V.; Ostrova, I.; Shemesh, M. Butyric Acid Released during Milk Lipolysis Triggers Biofilm Formation of Bacillus species. Int. J. Food Microbiol. 2014, 181, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Anand, S.K. Bacterial Biofilm on Food Contact Surfaces: A Review. J. Food Sci. Technol. 2002, 39, 573–593. [Google Scholar]
- Sharma, M.; Anand, S.K. Biofilms Evaluation as an Essential Component of HACCP for Food/Dairy Processing Industry—A Case. Food Control 2002, 13, 469–477. [Google Scholar] [CrossRef]
- Sharma, M.; Anand, S.K. Characterization of Constitutive Microflora of Biofilms in Dairy Processing Lines. Food Microbiol. 2002, 19, 627–636. [Google Scholar] [CrossRef]
- Burgess, S.A.; Lindsay, D.; Flint, S.H. Thermophilic Bacilli and their Importance in Dairy Processing. Int. J. Food Microbiol. 2010, 144, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, R.; Svensson, B.; Andersson, M.A.; Christiansson, A.; Salkinoja-Salonen, M. Persistence strategies of Bacillus cereus spores isolated from dairy silo tanks. Food Microbiol. 2010, 27, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.H.; Beuchat, L.R. Biofilm Formation and Sporulation by Bacillus cereus on a Stainless Steel Surface and Subsequent Resistance of Vegetative Cells and Spores to Chlorine, Chlorine Dioxide, and a Peroxyacetic Acid-Based Sanitizer. J. Food Prot. 2005, 68, 2614–2622. [Google Scholar] [CrossRef]
- Wijman, J.G.; de Leeuw, P.P.; Moezelaar, R.; Zwietering, M.H.; Abee, T. Air-liquid Interface Biofilms of Bacillus cereus: Formation, Sporulation, and Dispersion. Appl. Environ. Microbiol. 2007, 73, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Faille, C.; Benezech, T.; Blel, W.; Ronse, A.; Ronse, G.; Clarisse, M.; Slomianny, C. Role of Mechanical vs. Chemical Action in the Removal of Adherent Bacillus Spores during CIP Procedures. Food Microbiol. 2013, 33, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Branda, S.S.; Gonzalez-Pastor, J.E.; Ben-Yehuda, S.; Losick, R.; Kolter, R. Fruiting Body Formation by Bacillus subtilis. Proc. Natl. Acad. Sci. USA 2001, 98, 11621–11626. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, M.; Kagihiro, S.; Haruki, M.; Takano, K.; Branda, S.; Kolter, R.; Kanaya, S. Biofilm Formation by a Bacillus subtilis Strain that Produces Gamma-Polyglutamate. Microbiol. Sgm 2006, 152, 2801–2807. [Google Scholar] [CrossRef]
- Stanley, N.R.; Lazazzera, B.A. Defining the Genetic differences Between Wild and Domestic Strains of Bacillus subtilis that Affect Poly-gamma-dl-glutamic Acid Production and Biofilm Formation. Mol. Microbiol. 2005, 57, 1143–1158. [Google Scholar] [CrossRef]
- Yu, Y.Y.; Yan, F.; Chen, Y.; Jin, C.; Guo, J.H.; Chai, Y.R. Poly-gamma-Glutamic Acids Contribute to Biofilm Formation and Plant Root Colonization in Selected Environmental Isolates of Bacillus subtilis. Front. Microbiol. 2016, 7, 1811. [Google Scholar] [CrossRef]
- Midelet, G.; Carpentier, B. Impact of Cleaning and Disinfection Agents on Biofilm Structure and on Microbial Transfer to a Solid Model Food. J. Appl. Microbiol. 2004, 97, 262–270. [Google Scholar] [CrossRef]
- Simoes, M.; Simoes, L.C.; Vieira, M.J. A Review of Current and Emergent Biofilm Control Strategies. Lwt-Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef]
- Ostrov, I.; Harel, A.; Bernstein, S.; Steinberg, D.; Shemesh, M. Development of a Method to Determine the Effectiveness of Cleaning Agents in Removal of Biofilm Derived Spores in Milking System. Front. Microbiol. 2016, 7, 1498. [Google Scholar] [CrossRef] [PubMed]
- Zottola, E.A.; Sasahara, K.C. Microbial Biofilms in the Food-Processing Industry-Should They Be a Concern. Int. J. Food Microbiol. 1994, 23, 125–148. [Google Scholar] [CrossRef]
- Faille, C.; Fontaine, F.; Benezech, T. Potential Occurrence of Adhering Living Bacillus Spores in Milk Product Processing Lines. J. Appl. Microbiol. 2001, 90, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Ostrov, I.; Sela, N.; Belausov, E.; Steinberg, D.; Shemesh, M. Adaptation of Bacillus Species to Dairy Associated Environment Facilitates their Biofilm Forming Ability. Food Microbiol. 2019. [Google Scholar] [CrossRef]
- Ostrov, I.; Sela, N.; Freed, M.; Khateb, N.; Kott-Gutkowski, M.; Inbar, D.; Shemesh, M. Draft Genome Sequence of Bacillus licheniformis S127, Isolated from a Sheep Udder Clinical Infection. Genome Announc. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Shemesh, M.; Chai, Y.R. A Combination of Glycerol and Manganese Promotes Biofilm Formation in Bacillus subtilis via Histidine Kinase KinD Signaling. J. Bacteriol. 2013, 195, 2747–2754. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Tischenko, E.; Dubois-Brissonnet, F.; Herry, J.M.; Thomas, V.; Daddi-Oubekka, S.; Waharte, F.; Steenkeste, K.; Fontaine-Aupart, M.P.; Briandet, R. Deciphering Biofilm Structure and Reactivity by Multiscale Time-Resolved Fluorescence Analysis. Bact. Adhes. Chem. Biol. Phys. 2011, 715, 333–349. [Google Scholar] [CrossRef]
- Vlamakis, H.; Chai, Y.R.; Beauregard, P.; Losick, R.; Kolter, R. Sticking Together: Building a Biofilm the Bacillus subtilis Way. Nat. Rev. Microbiol. 2013, 11, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Sundberg, M.; Christiansson, A.; Lindahl, C.; Wahlund, L.; Birgersson, C. Cleaning Effectiveness of Chlorine-free Detergents for Use on Dairy Farms. J. Dairy Res. 2011, 78, 105–110. [Google Scholar] [CrossRef]
- Faille, C.; Benezech, T.; Midelet-Bourdin, G.; Lequette, Y.; Clarisse, M.; Ronse, G.; Ronse, A.; Slomianny, C. Sporulation of Bacillus spp. within Biofilms: A Potential Source of Contamination in Food Processing Environments. Food Microbiol. 2014, 40, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Sendamangalam, V.R.; Gruden, C.L.; Seo, Y. Multiple Roles of Extracellular Polymeric Substances on Resistance of Biofilm and Detached Clusters. Environ. Sci. Technol. 2012, 46, 13212–13219. [Google Scholar] [CrossRef]
- Leliévre, C.; Antonini, G.; Faille, C.; Bénézech, T. Cleaning-in-place, Modelling of Cleaning Kinetics of Pipes Soiled by Bacillus Spores Assuming a Process Combining Removal and Deposition. Food Bioprod. Process. 2002, 80, 305–311. [Google Scholar] [CrossRef]
- Leliévre, C.; Legentilhomme, P.; Legrand, J.; Faille, C.; Bénézech, T. Hygenic Design: Influence of the Local Wall Shear Stress Variations on the Cleanability of a Three-way Valve. Chem. Eng. Res. Des. 2003, 81, 1071–1076. [Google Scholar] [CrossRef]
- Wirtanen, G.; Husmark, U.; Mattila-Sandholm, T. Microbial Evaluation of the Biotransfer Potential from Surfaces with Bacillus Biofilms after Rinsing and Cleaning Procedures in Closed Food-Processing Systems. J. Food Prot. 1996, 59, 727–733. [Google Scholar] [CrossRef]
- Palmer, J.; Flint, S.; Brooks, J. Bacterial Cell attachment, the Beginning of a Biofilm. J. Ind. Microbiol. Biotechnol. 2007, 34, 577–588. [Google Scholar] [CrossRef]
- Mah, T.F.C.; O’Toole, G.A. Mechanisms of Biofilm Resistance to Antimicrobial Agents. Trends Microbiol. 2001, 9, 34–39. [Google Scholar] [CrossRef]
- Singh, R.; Ray, P.; Das, A.; Sharma, M. Penetration of Antibiotics through Staphylococcus aureus and Staphylococcus epidermidis Biofilms. J. Antimicrob. Chemother. 2010, 65, 1955–1958. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, A.; Langsrud, S.; Heir, E.; Mikkelsen, M.I.; Moretro, T. Biofilm Matrix Composition Affects the Susceptibility of Food Associated Staphylococci to Cleaning and Disinfection Agents. Front Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Lee, W.H.; Coburn, K.M.; Seo, Y. Selective Reactivity of Monochloramine with Extracellular Matrix Components Affects the Disinfection of Biofilm and Detached Cflusters. Environ. Sci. Technol. 2014, 48, 3832–3839. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Dubois-Brissonnet, F.; Greub, G.; Thomas, V.; Briandet, R. Dynamics of the Action of Biocides in Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 2011, 55, 2648–2654. [Google Scholar] [CrossRef]
- Ogunleye, A.; Bhat, A.; Irorere, V.U.; Hill, D.; Williams, C.; Radecka, I. Poly-gamma-glutamic Acid: Production, Properties and Applications. Microbiol. Sgm 2015, 161, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Shemesh, M.; Kolter, R.; Losick, R. The Biocide Chlorine Dioxide Stimulates Biofilm Formation in Bacillus subtilis by Activation of the Histidine Kinase KinC. J. Bacteriol. 2010, 192, 6352–6356. [Google Scholar] [CrossRef] [PubMed]
- Strempel, N.; Nusser, M.; Neidig, A.; Brenner-Weiss, G.; Overhage, J. The Oxidative Stress Agent Hypochlorite Stimulates c-di-GMP Synthesis and Biofilm Formation in Pseudomonas aeruginosa. Front. Microbiol. 2017, 8, 2311. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ostrov, I.; Paz, T.; Shemesh, M. Robust Biofilm-Forming Bacillus Isolates from the Dairy Environment Demonstrate an Enhanced Resistance to Cleaning-in-Place Procedures. Foods 2019, 8, 134. https://doi.org/10.3390/foods8040134
Ostrov I, Paz T, Shemesh M. Robust Biofilm-Forming Bacillus Isolates from the Dairy Environment Demonstrate an Enhanced Resistance to Cleaning-in-Place Procedures. Foods. 2019; 8(4):134. https://doi.org/10.3390/foods8040134
Chicago/Turabian StyleOstrov, Ievgeniia, Tali Paz, and Moshe Shemesh. 2019. "Robust Biofilm-Forming Bacillus Isolates from the Dairy Environment Demonstrate an Enhanced Resistance to Cleaning-in-Place Procedures" Foods 8, no. 4: 134. https://doi.org/10.3390/foods8040134
APA StyleOstrov, I., Paz, T., & Shemesh, M. (2019). Robust Biofilm-Forming Bacillus Isolates from the Dairy Environment Demonstrate an Enhanced Resistance to Cleaning-in-Place Procedures. Foods, 8(4), 134. https://doi.org/10.3390/foods8040134




