Influence of Homogenization Time and Speed on Rheological and Volatile Composition in Olive-Based Pâtés
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Rheological Analysis
2.3. Color Analysis
2.4. Volatile Compounds
2.5. Statistical Analysis
3. Results and Discussion
3.1. Rheological and Color Characteristics
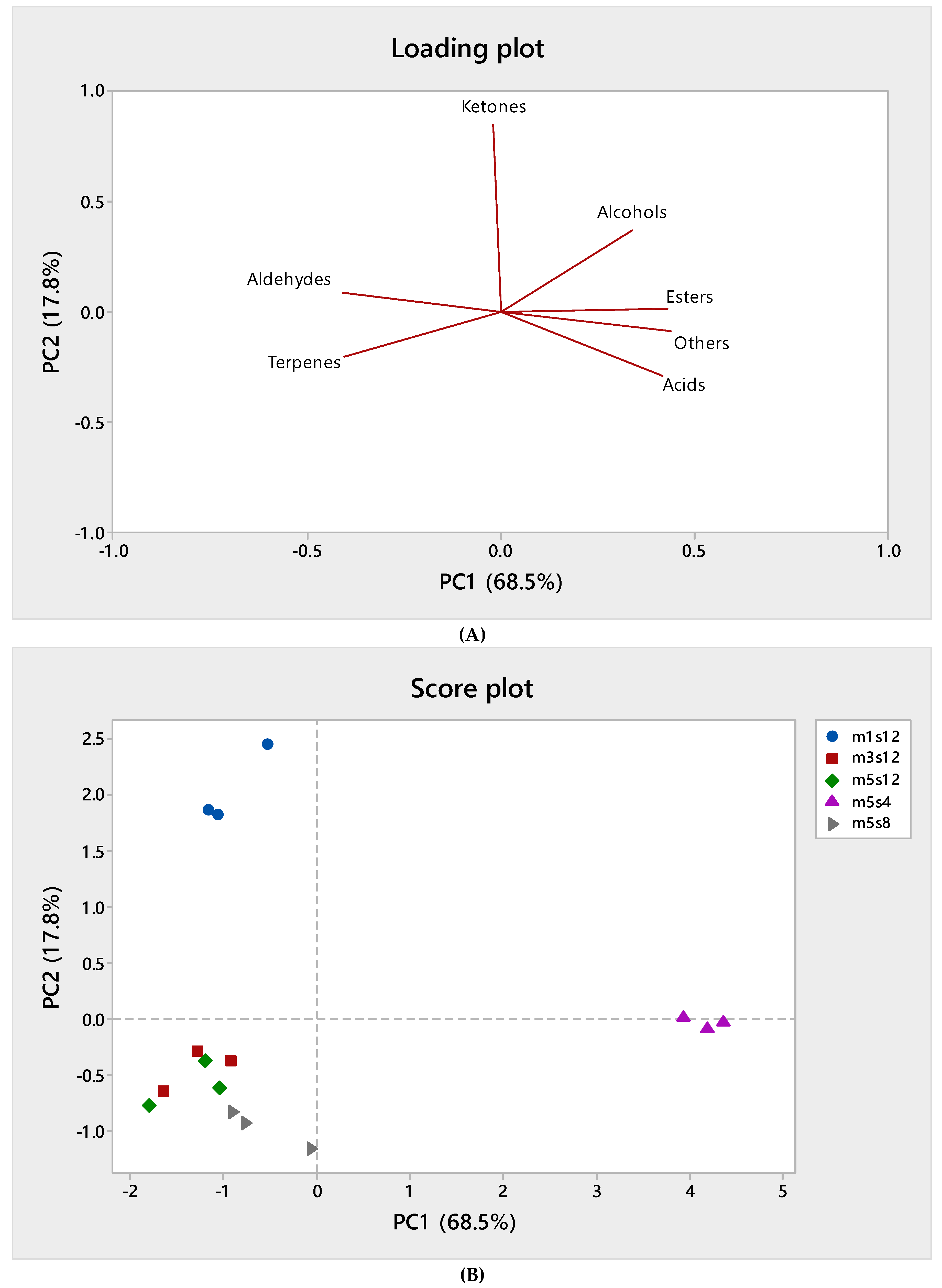
3.2. Volatile Compounds
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ünlüsayin, M.; Bilgin, S.; İzci, L.; Günlü, A. Chemical and sensory assessment of hot-smoked fish pâté. J. FisheriesSciences.com 2017, 1, 20–24. [Google Scholar] [CrossRef]
- Tamagnone, P.; Marchitelli, V.; Baiano, A.; Del Nobile, M.A. Quality decay kinetics of semi-preserved sauce as affected by packaging. J. Food Sci. 2005, 70, 92–97. [Google Scholar]
- Marudova, M.; Momchilova, M.; Antova, G.; Petkova, Z.; Yordanov, D.; Zsivanovits, G. Investigation of fatty acid thermal transitions and stability in poultry pates enriched with vegetable components. J. Therm. Anal. Calorim. 2017, 133, 539–547. [Google Scholar] [CrossRef]
- Guimarães, R.M.; Silva, T.E.; Lemes, A.C.; Boldrin, M.C.F.; Da Silva, M.A.P.; Silva, F.G.; Egea, M.B.; Silva, M.A.P. Okara: A soybean by-product as an alternative to enrich vegetable paste. LWT—Food Sci. Technol. 2018, 92, 593–599. [Google Scholar] [CrossRef]
- Cosmai, L.; Caponio, F.; Pasqualone, A.; Summo, C.; Paradiso, V.M. Evolution of the oxidative stability, bio-active compounds and color characteristics of non-thermally treated vegetable pâtés during frozen storage. J. Sci. Food Agric. 2017, 97, 4904–4911. [Google Scholar] [CrossRef] [PubMed]
- Cosmai, L.; Campanella, D.; Summo, C.; Paradiso, V.M.; Pasqualone, A.; De Angelis, M.; Caponio, F. Combined effects of a natural Allium spp. extract and modified atmospheres packaging on shelf life extension of olive-based paste. Int. J. Food Sci. Technol. 2017, 48, 1164–1175. [Google Scholar] [CrossRef]
- Cosmai, L.; Campanella, D.; Summo, C.; Paradiso, V.M.; Pasqualone, A.; De Angelis, M.; Caponio, F. New formulations of olive-based pâté: Development and quality. Ital. J. Food Sci. 2017, 29, 302–316. [Google Scholar]
- Gorlov, I.F.; Sitnikova, O.I.; Zlobina, E.Y.; Mikhailovn, T.; Slozhenkin, M.I.; Karpenko, E.V. New functional products with chickpeas: Reception, functional properties. Am. J. Food Technol. 2016, 11, 273–281. [Google Scholar] [CrossRef]
- Xiong, G.; Han, M.; Kang, Z.; Zhao, Y.; Xu, X.; Zhu, Y. Evaluation of protein structural changes and water mobility in chicken liver paste batters prepared with plant oil substituting pork back-fat combined with pre-emulsification. Food Chem. 2016, 196, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Ouden, F.; Vliet, T. Particle size distribution in tomato concentrate and effects on rheological properties. J. Food Sci. 1997, 62, 565–567. [Google Scholar] [CrossRef]
- Lopez-Sanchez, P.; Svelander, C.; Bialek, L.; Schumm, S.; Langton, M.; Lopez-Sanchez, P. Rheology and microstructure of carrot and tomato emulsions as a result of high-pressure homogenization conditions. J. Food Sci. 2010, 76, 130–140. [Google Scholar] [CrossRef]
- Sánchez, M.C.; Valencia, C.; Gallegos, C.; Ciruelos, A.; Latorre, A. Influence of processing on the rheological properties of tomato paste. J. Food Sci. Agric. 2002, 82, 990–997. [Google Scholar] [CrossRef]
- Tanglertpaibul, T.; Rao, M.A. Flow properties of tomato concentrates: Effect of serum viscosity and pulp content. J. Food Sci. 1987, 52, 318–321. [Google Scholar] [CrossRef]
- Yoo, B.; Rao, M. Effect of unimodal particle size and pulp content on rheological properties of tomato puree. J. Texture Stud. 1994, 25, 421–436. [Google Scholar] [CrossRef]
- Podsędek, A. Natural antioxidants and antioxidant capacity of Brassica vegetables: A review. LWT—Food Sci. Technol. 2007, 40, 1–11. [Google Scholar] [CrossRef]
- Nijsse, J.; Blonk, H.C.G.; Bialek, L.; Schumm, S.; Langton, M.; Lopez-Sanchez, P.; Lopez-Sanchez, P. Effect of mechanical and thermal treatments on the microstructure and rheological properties of carrot, broccoli and tomato dispersions. J. Food Sci. Agric. 2010, 91, 207–217. [Google Scholar]
- Colle, I.; Van Buggenhout, S.; Van Loey, A.; Hendrickx, M. High pressure homogenization followed by thermal processing of tomato pulp: Influence on microstructure and lycopene in vitro bioaccessibility. Food Res. Int. 2010, 43, 2193–2200. [Google Scholar] [CrossRef]
- Saija, A.; Uccella, N. Olive biophenols: Functional effects on human wellbeing. Food Sci. Technol. 2000, 11, 357–363. [Google Scholar] [CrossRef]
- Vercet, A.; Sánchez, C.; Burgos, J.; Montañes, L.; Buesa, P.L. The effects of manothermosonication on tomato pectic enzymes and tomato paste rheological properties. J. Food Eng. 2002, 53, 273–278. [Google Scholar] [CrossRef]
- Caponio, F.; Squeo, G.; Brunetti, L.; Pasqualone, A.; Summo, C.; Catalano, P.; Bianchi, B.; Paradiso, V.M. Influence of the feed pipe position of an industrial scale two-phase decanter on extraction efficiency and chemical-sensory characteristics of virgin olive oil. J. Food Sci. Agric. 2018, 98, 4279–4286. [Google Scholar] [CrossRef]
- Cosmai, L.; Summo, C.; Caponio, F.; Paradiso, V.M.; Gomes, T. Influence of the thermal stabilization process on the volatile profile of canned tomato-based food. J. Food Sci. 2013, 78, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Celeghin, A.G.; Minetti, F.; Contini, L.E.; Miccolo, M.E.; Rubiolo, A.C.; Olivares, M.L. Syneresis and sensory acceptability of desserts based on whey proteins concentrates. J. Food Nutr. Res. 2016, 4, 478–482. [Google Scholar]
- Fernández-López, J.; Pérez–Alvarez, J.A.; Aranda-Catalá, V. Effect of mincing degree on colour properties in pork meat. Color Res. Appl. 2000, 25, 376–380. [Google Scholar] [CrossRef]
- Flores, M.; Grimm, C.C.; Toldrá, F.; Spanier, A.M. Correlations of sensory and volatile compounds of Spanish “serrano” dry-cured ham as a function of two processing times. J. Agric. Food Chem. 1997, 45, 2178–2186. [Google Scholar] [CrossRef]
- Dirinck, P.; VandenDriessche, F.; Van Opstaele, F. Flavour differences between northern and southern European cured hams. Food Chem. 1997, 59, 511–521. [Google Scholar] [CrossRef]
- Caponio, F.; Difonzo, G.; Calasso, M.; Cosmai, L.; De Angelis, M. Effects of olive leaf extract addition on fermentative and oxidative processes of table olives and their nutritional properties. Food Res. Int. 2019, 116, 1306–1317. [Google Scholar] [CrossRef] [PubMed]
| Trial | Homogenization Conditions | |
|---|---|---|
| Time (min) | Speed (rpm) | |
| m1s12 | 1 | 12,000 |
| m3s12 | 3 | 12,000 |
| m5s12 | 5 | 12,000 |
| m5s8 | 5 | 8000 |
| m5s4 | 5 | 4000 |
| Parameter | m1s12 | m3s12 | m5s12 | m5s8 | m5s4 |
|---|---|---|---|---|---|
| Hardness (N) | 21.20 ± 0.40 a | 17.90 ± 0.50 b | 10.90 ± 0.40 c,C | 23.00 ± 1.50 B | 45.30 ± 3.70 A |
| Syneresis (% w/w) | 2.87 ± 0.21 a | 2.45 ± 0.08 a | 1.73 ± 0.20 b,C | 3.55 ± 0.36 B | 6.65 ± 0.20 A |
| L | 39.08 ± 0.01 b | 40.21 ± 0.04 a | 38.53 ± 0.03 c,B | 39.15 ± 0.01 A | 37.73 ± 0.02 C |
| a* | −4.48 ± 0.02 a | −4.98 ± 0.03 c | −4.61 ± 0.04 b,C | −4.06 ± 0.02 A | −4.30 ± 0.02 B |
| b* | 24.13 ± 0.01 c | 26.53 ± 0.01 b | 27.83 ± 0.04 a,A | 26.34 ± 0.03 C | 26.77 ± 0.01 B |
| 100 − L | 60.92 ± 0.01 b | 59.80 ± 0.04 c | 61.47 ± 0.03 a,B | 60.85 ± 0.01 C | 62.28 ± 0.02 A |
| Volatile Compounds | m1s12 | m3s12 | m5s12 | m5s8 | m5s4 |
|---|---|---|---|---|---|
| Terpenes | 18.00 ± 0.25 a | 19.73 ± 1.72 a | 19.01 ± 0.97 a,A | 21.20 ± 1.58 A | 11.62 ± 0.36 B |
| Aldehydes | 40.34 ± 0.86 b | 40.18 ± 1.15 b | 44.78 ± 1.93 a,A | 35.43 ± 2.31 B | 26.61 ± 3.33 C |
| Alcohols | 15.19 ± 1.03 a | 14.76 ± 0.79 a | 14.05 ± 0.53 a,B | 13.87 ± 0.23 B | 16.49 ± 0.71 A |
| Ketones | 5.73 ± 0.42 a | 1.96 ± 0.12 b | 2.08 ± 0.08 b,B | 2.36 ± 0.03 B | 2.73 ± 0.19 A |
| Esters | 4.39 ± 0.38 a | 4.29 ± 0.22 a | 4.34 ± 0.42 a,B | 4.46 ± 0.46 B | 6.19 ± 0.38 A |
| Acids | 0.55 ± 0.02 c | 0.72 ± 0.05 b | 0.80 ± 0.01 a,B | 1.22 ± 0.28 B | 2.02 ± 0.20 A |
| Others | 6.56 ± 0.27 a | 5.45 ± 0.10 b | 7.08 ± 0.25 a,C | 8.06 ± 0.15 B | 13.25 ± 0.40 A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caponio, F.; Difonzo, G.; Squeo, G.; Fortunato, S.; Silletti, R.; Summo, C.; Paradiso, V.M.; Pasqualone, A. Influence of Homogenization Time and Speed on Rheological and Volatile Composition in Olive-Based Pâtés. Foods 2019, 8, 115. https://doi.org/10.3390/foods8040115
Caponio F, Difonzo G, Squeo G, Fortunato S, Silletti R, Summo C, Paradiso VM, Pasqualone A. Influence of Homogenization Time and Speed on Rheological and Volatile Composition in Olive-Based Pâtés. Foods. 2019; 8(4):115. https://doi.org/10.3390/foods8040115
Chicago/Turabian StyleCaponio, Francesco, Graziana Difonzo, Giacomo Squeo, Stefania Fortunato, Roccangelo Silletti, Carmine Summo, Vito M. Paradiso, and Antonella Pasqualone. 2019. "Influence of Homogenization Time and Speed on Rheological and Volatile Composition in Olive-Based Pâtés" Foods 8, no. 4: 115. https://doi.org/10.3390/foods8040115
APA StyleCaponio, F., Difonzo, G., Squeo, G., Fortunato, S., Silletti, R., Summo, C., Paradiso, V. M., & Pasqualone, A. (2019). Influence of Homogenization Time and Speed on Rheological and Volatile Composition in Olive-Based Pâtés. Foods, 8(4), 115. https://doi.org/10.3390/foods8040115










