Quantitative Determination of Andrographolide and Related Compounds in Andrographis paniculata Extracts and Biological Evaluation of Their Anti-Inflammatory Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instrumentation and Conditions
2.3. Extraction Procedure
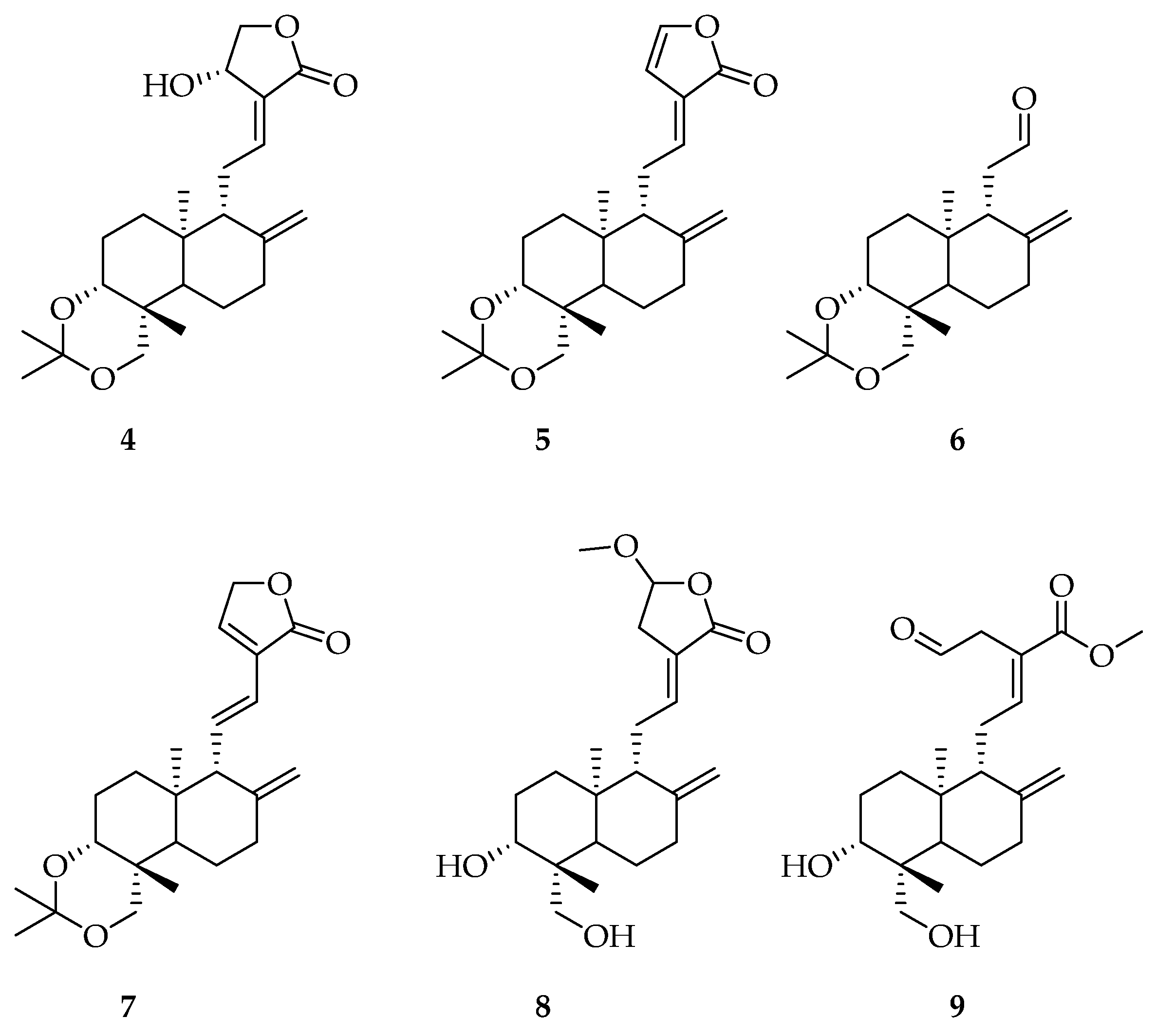
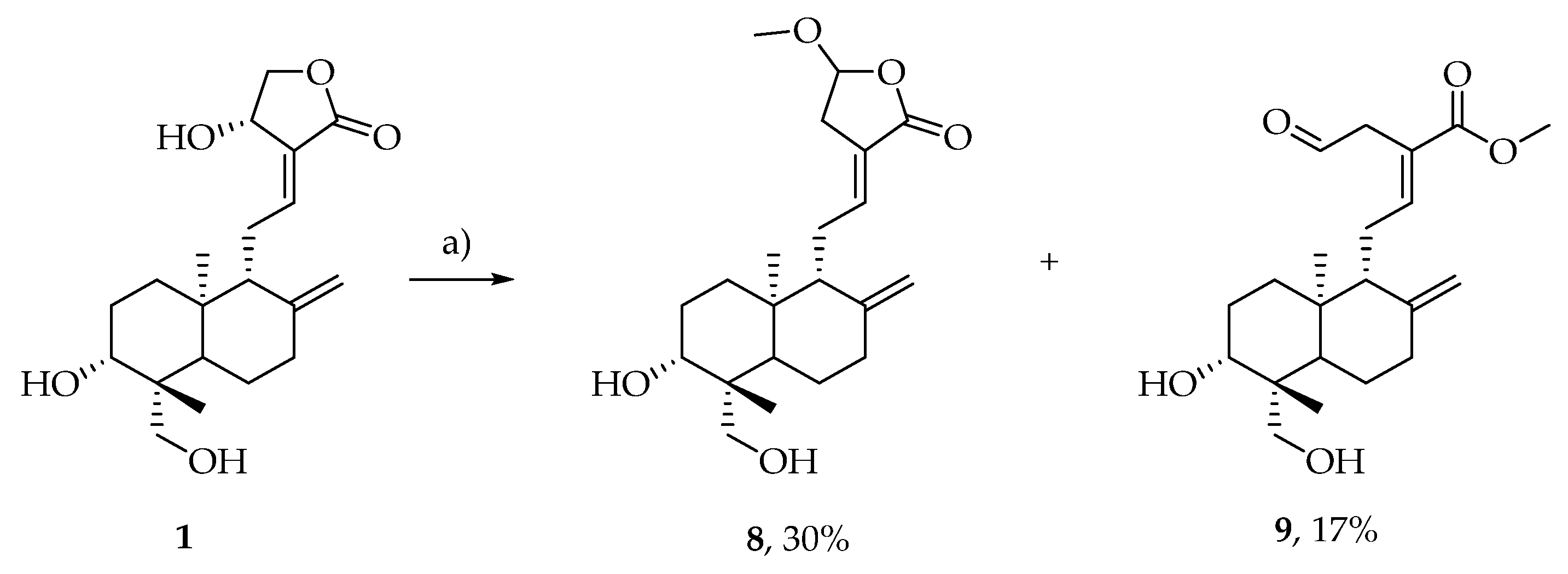
2.4. Preparation of Pure Compounds
2.5. Standard and Sample Preparation
2.6. NFκB-Dependent Luciferase Activity Assay
2.7. Assay Validation, Linearity of Calibration and Limit of Quantification (LOQ)
2.8. Assay Validation—Precision
2.9. Assay Validation—Accuracy
3. Results and Discussion
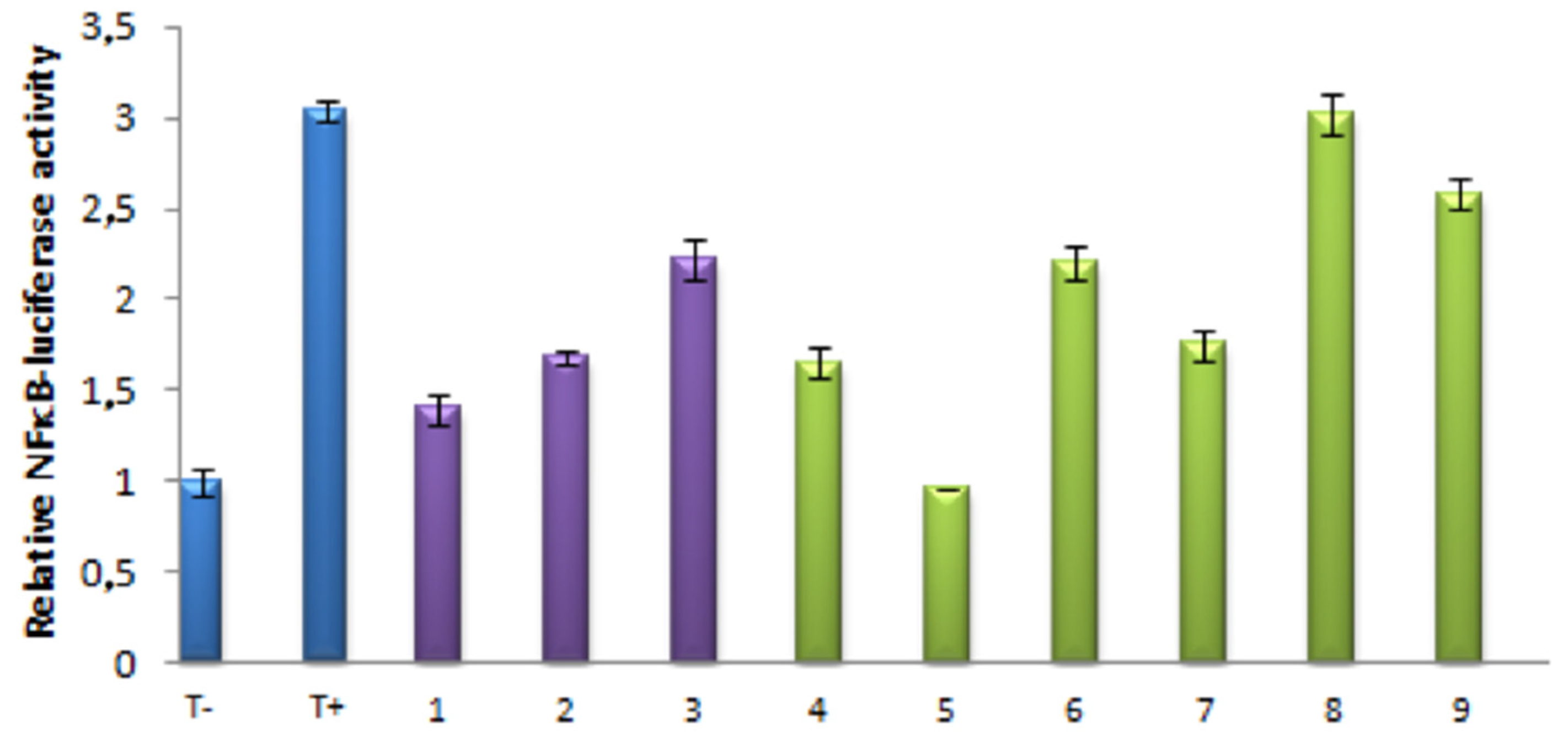
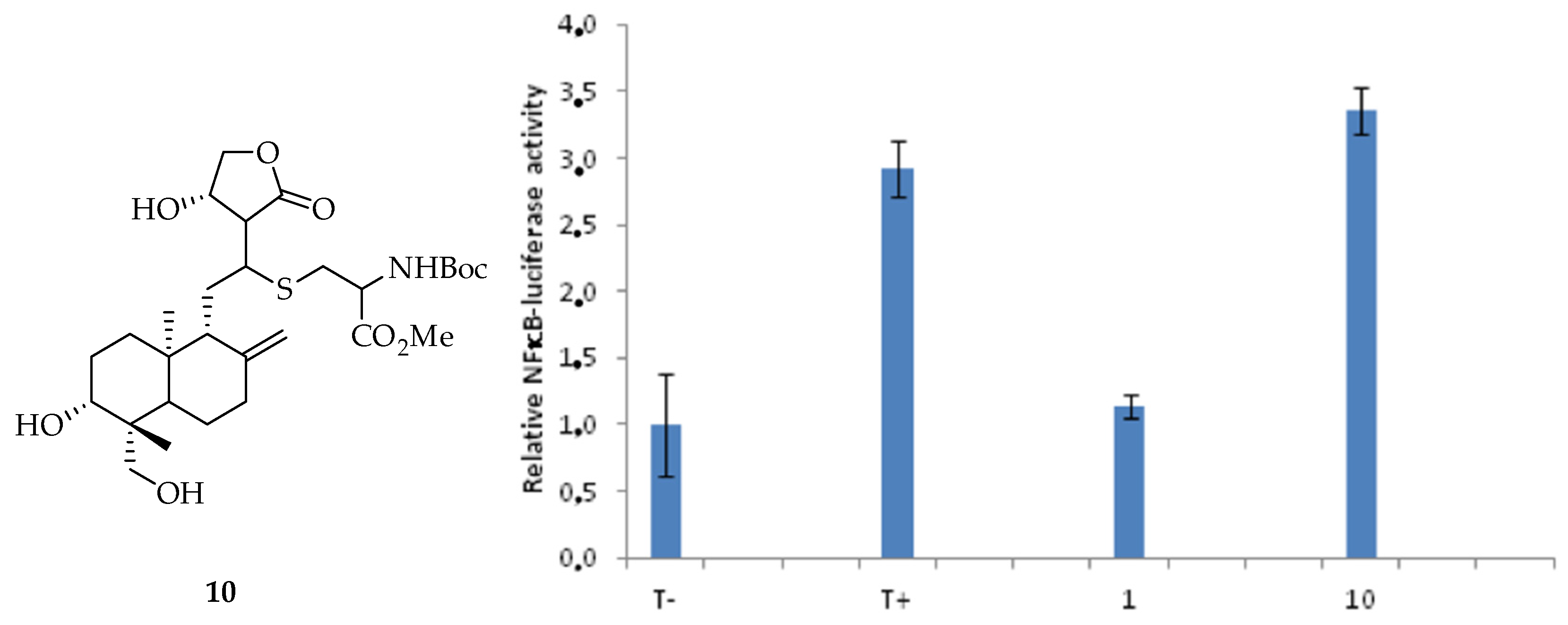
3.1. Effects of Isolated and Modified Compounds on TNFα Induced NFκB Transcriptional Activation
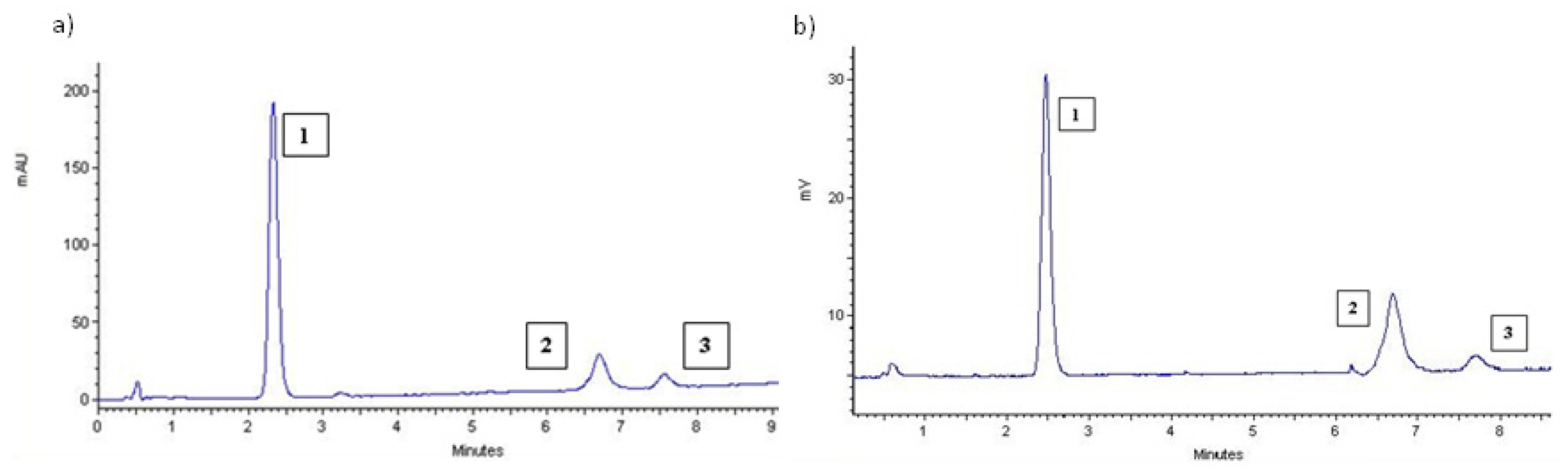
3.2. Quantitation of Commercial Preparations
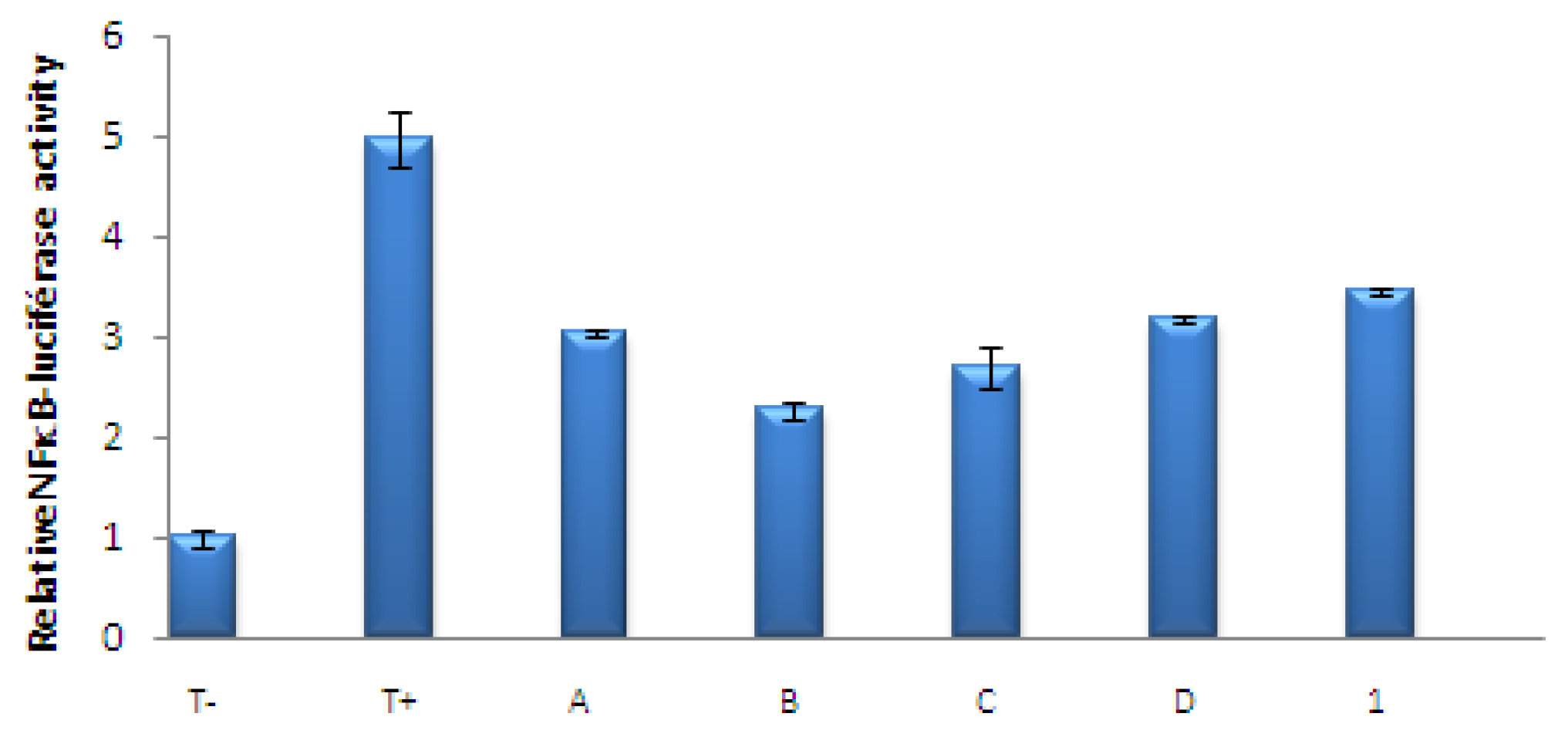
3.3. Effects of the Extracts of Known Concentrations on TNFα Induced NFκB Transcriptional Activation
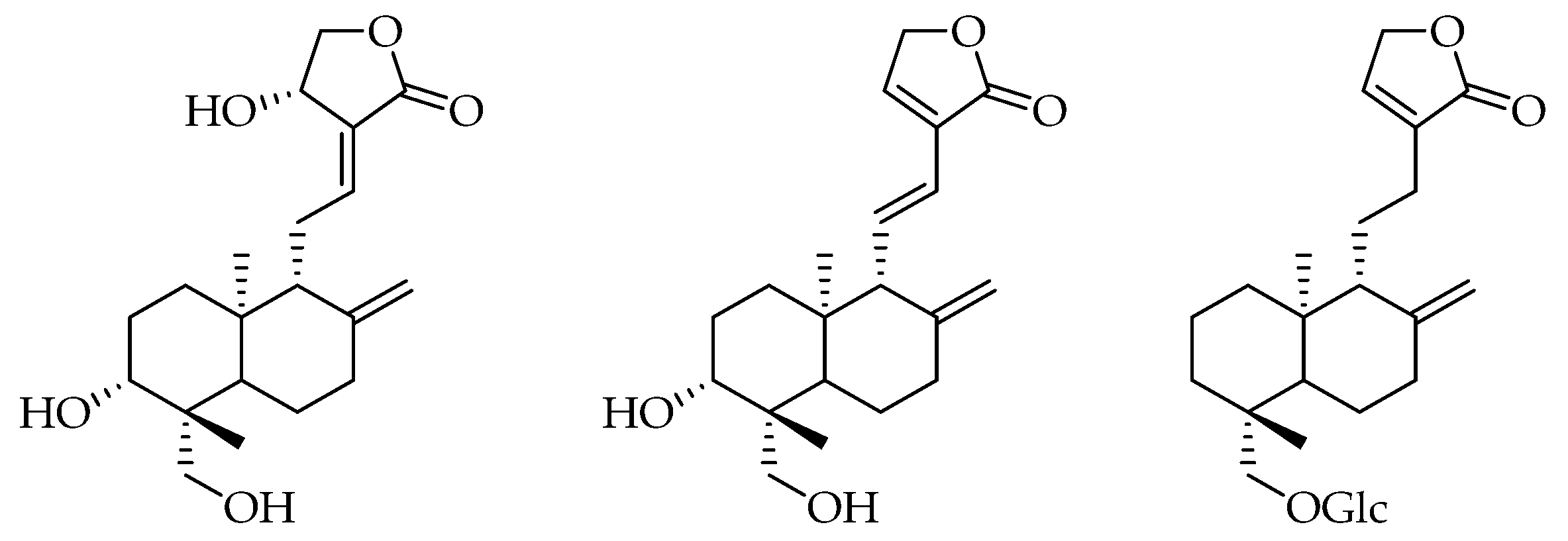
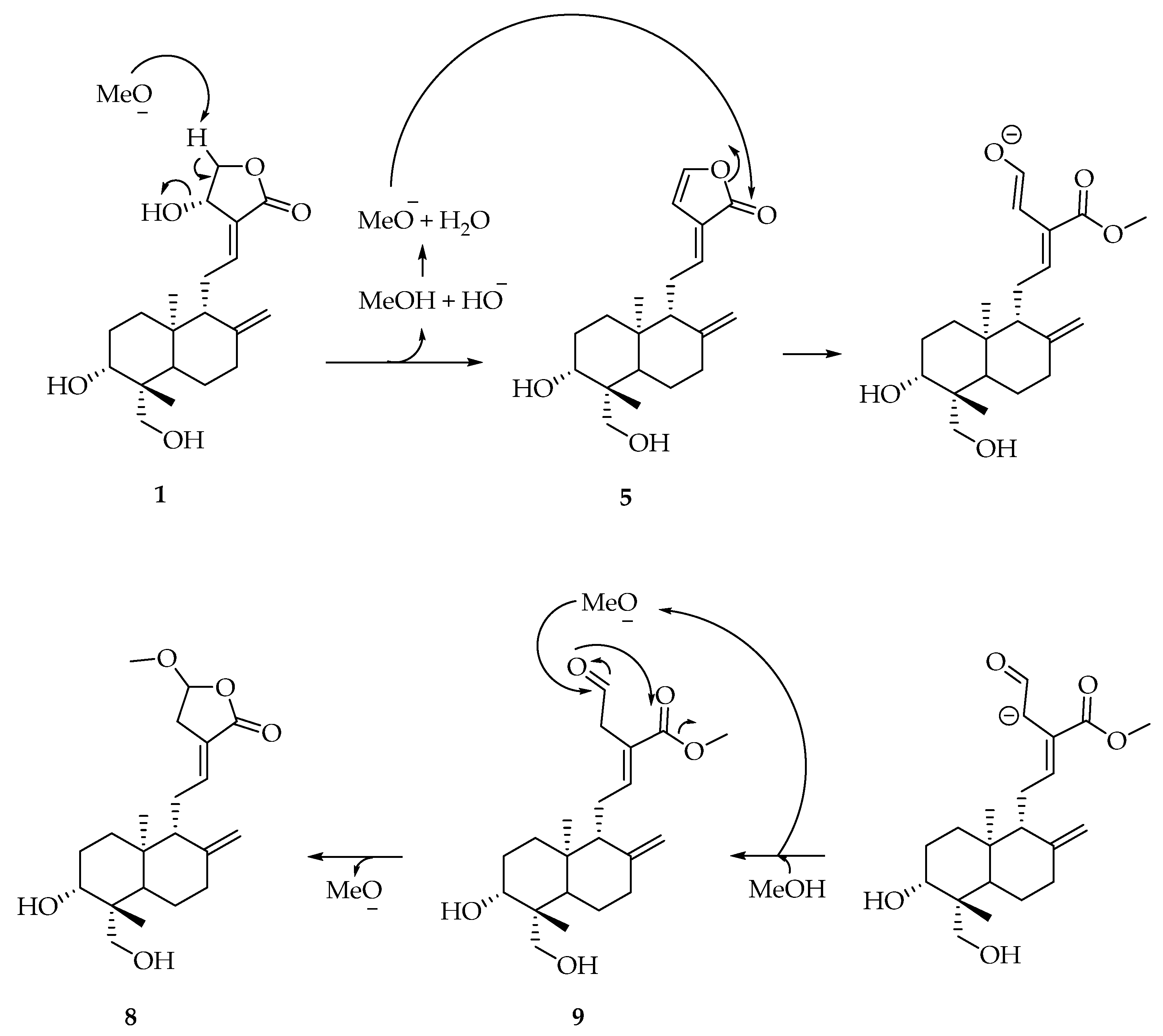
3.4. Characterization of Products 1, 2 and 3
- 1:
- 1H NMR (DMSO, 400 MHz) δ 6.62 (1H, dt, J11,12 = 7.0 Hz, J12,14 1.5 Hz, H12), 5.71 (1H, d, J14,OH 6.0 Hz, OH14), 5.05 (1H, d, J3,OH 5.0 Hz, OH-3), 4.91 (1H, t, J14,15 6.0 Hz, H14), 4.81 (1H, s, H17A), 4.62 (1H, s, 17B), 4.39 (1H, dd, J15A,15B 10Hz, J14,15A 6.0 Hz, H15A), 4.13 (1H, dd, J19B,OH 7.5 Hz, J19A,OH 2.5 Hz, OH-19), 4.03 (1H, dd, J15A,15B 10.0 Hz, J14,15B 2.0 Hz, H15B), 3.84 (1H, dd, J19A,19B 11.0 Hz, J19A,OH 2.5 Hz, H19A), 3.29–3.20 (2H, m, H3, H19B), 2.46 (1H, m, H11A), 2.32 (1H, br d, J7A,7B 13.0 Hz, H7A), 1.6–1.84 (2H, m, H7B, H11B), 1.75–1.61 (5H, m, H1A, H2, H6A, H9), 1.35 (1H, dq, J6A,6B J6B,7B 13.0 Hz, J6B,7A 4.0 Hz, H6B), 1.23–1.17 (2H, H1B, H5), 1.08 (3H, s, H18), 0.66 (3H, s, H20). 13C NMR (DMSO, 100 MHz) δ 170.8 (C16), 148.5 (C8), 147.2 (C12), 129.9 (C13), 109.2 (C17), 79.3 (C3), 75.2 (C15), 65.4 (C14), 63.6 (C19), 56.4 (C9), 55.3 (C5), 43.1 (C10), 39.5 (C4), 38.4 (C7), 38.1 (C11), 37.4 (C1), 28.8 (C2), 24.8 (C6), 24.0 (C18), 15.6 (C20). ESIMS (negative mode) m/z 331 [M-H2O]−, 349 [M-H]−, 395 [M-H+HCOOH]−, (positive mode) 351 [M+H]+, 373 [M+Na]+. HRMS: calcd for C20H31O5 [M+H]+ 351.2167, found 351.2166.
- 2:
- 1H NMR (DMSO, 400 MHz) δ 7.65 (1H, brs, H14), 6.74 (1H, dd, J11,12 15.5 Hz, J9,11 10.0 Hz, H11), 6.12 (1H, d, J11,12 15.5 Hz, H12), 5.03 (1H, d, J3,OH 5.0 Hz, OH-3), 4,89 (2H, brs, H15), 4.73 (1H, s, H17A), 4.42 (1H, s, H17B), 4.12 (1H, t, JOH,19B 6.5 Hz, OH-19), 3.84 (1H, dd, J19A,19B 13.0 Hz, J19A,OH 5.0 Hz, H19A), 3.28–3.16 (2H, m, H3, 19B), 2.36–2.32 (2H, m, H9, H7A), 1.97 (1H, m, H7B), 1.72 (1H, m, H2A), 1.58–1.54 (2H, m, H2, H6A), 1.40 (1H, dd, J6A,6B 13.0 Hz, J6B,7B 4.0 Hz, H6B), 1.28–1.12 (2H, m, H1), 1.07 (3H, s, H18), 0.76 (3H, s, H20). 13C NMR (DMSO, 100 MHz) δ 172.9 (C16), 149.5 (C8), 147.2 (C14), 134.7 (C11), 127.5 (C13), 121.6 (C12), 108.5 (C17), 79.1 (C3), 63.1 (C19), 61.0 (C9), 54.2 (C5), 42.8 (C4), 38.4 (C1), 36.7 (C7), 29.4 (C10), 28.1 (C2), 23.6 (C6), 23.4 (C18), 15.9 (C20). ESIMS (negative mode) m/z 331 [M-H]−, 377 [M-H+HCOOH]−, (positive mode) 355 [M+Na]+. HRMS: calcd for C20H28O4 [M+Na]+ 355.2035, found 355.2036.
- 3:
- 1H NMR (DMSO, 400 MHz) δ7.46 (1H, s, H14), 4.86–4.78 (6H, 3OH, H15, H17A), 4.59 (1H, s, H17B), 4.38 (1H, t, J6′,OH 6.0 Hz, OH-6’), 4.02 (1H, d, J1′,2′ 8.0 Hz, 3.88 (1H, d, J19A,19B 10.0 Hz, H19A), 3.63 (1H, dd, J6′A,6′B 10.5 Hz, J5′,6′A 6.0 Hz, H6A), 3.43 (1H, dd, J6′A,6′B 10.5 Hz, J5′,6′B 5.0 Hz, H6B), 3.17–3.04 (3H, m, H3’, H4’, H5’), 2.92 (1H, m, H2′), 2.36–0.90 (16H, m, H1, H2, H3, H5, H6, H7, H9, H11, H12), 0.95 (3H, s, H18), 0.61 (3H, s, H20). 13C NMR (DMSO, 100 MHz) δ 174.6 (C16), 148.1 (C8), 147.4 (C14), 132.6 (C13), 107.0 (C17), 104.0 (C1’), 77.3, 77.1, 70.6 (C3’, C4’, C5’), 74.0 (C2’), 71.2 (C15), 70.9 (C19), 61.5 (C6’), 56.1 (C9), 55.8 (C5), 38.8, 38.4, 36.1, 21.9, 19.0 (C1, C2, C3, C6, C7, C11, C12), 38.3 (C4), 28.0 (C18), 24.4 (C10), 15.6 (C20). ESIMS (negative mode) m/z 479 [M-H]−, 525 [M-H+HCOOH]−, (positive mode) 503 [M+Na]+. HRMS: calcd for C26H41O8 [M+H]+ 481.2795, found 481.2796.
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Acharya, R. A review of phytoconstituents and their pharmacological properties of Andrographis Paniculata (NEES). Int. J. Pharma Biol. Sci. 2017, 8, 77–83. [Google Scholar] [CrossRef]
- Chowdhury, A.; Biswas, S.K.; Raihan, S.Z.; Das, J.; Paul, S. Pharmacological Potentials of Andrographis paniculata: An Overview. Int. J. Pharmacol. 2012, 8, 6–9. [Google Scholar] [CrossRef]
- Hossain, M.S.; Urbi, Z.; Sule, A.; Rahman, K.M.H. Andrographis paniculata (Burm. f.) Wall. ex Nees: A Review of Ethnobotany, Phytochemistry, and Pharmacology. Sci. World J. 2014, 2014, 28. [Google Scholar] [CrossRef]
- Zhang, L.; Bao, M.; Liu, B.; Zhao, H.; Zhang, Y.; Ji, X.; Zhao, N.; Zhang, C.; He, X.; Yi, J.; et al. Effect of Andrographolide and Its Analogs on Bacterial Infection: A Review. Pharmacology 2019. [Google Scholar] [CrossRef]
- Grainne, H.; Franco, S.; Antonio, G.; Diego, C. Herbal medicinal products for inflammatory bowel disease: A focus on those assessed in double-blind randomised controlled trials. Phytother. Res. 2019. [Google Scholar] [CrossRef]
- Rahman, H.; Kim, M.; Leung, G.; Green, J.A.; Katz, S. Drug-Herb Interactions in the Elderly Patient with IBD: A Growing Concern. Curr. Treat. Options Gastroenterol. 2017, 15, 618–636. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Shahpiri, Z.; Bahramsoltani, R.; Moghaddamnia, M.; Najafi, F.; Rahimi, R. Efficacy and Tolerability of Phytomedicines in Multiple Sclerosis Patients: A Review. CNS Drugs 2017, 31, 867–889. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.S.D.; Liao, W.; Zhou, S.; Wong, W.S.F. Is there a future for andrographolide to be an anti-inflammatory drug? Deciphering its major mechanisms of action. Biochem. Pharmacol. 2017, 139, 71–81. [Google Scholar] [CrossRef]
- Majee, C.; Mazumder, R.; Choudhary, A.N. Medicinal plants with anti-ulcer and hepatoprotective activity: A review. Int. J. Pharm. Sci. Res. 2019, 1, 1–11. [Google Scholar] [CrossRef]
- Chua, L.S. Review on Liver Inflammation and Antiinflammatory Activity of Andrographis paniculata for Hepatoprotection. Phytother. Res. 2014, 28, 1589–1598. [Google Scholar] [CrossRef] [PubMed]
- Chao, W.-W.; Lin, B.-F. Hepatoprotective Diterpenoids Isolated from Andrographis paniculata. Chin. Med. 2012, 3, 136–143. [Google Scholar] [CrossRef][Green Version]
- Lu, J.; Ma, Y.; Wu, J.; Huang, H.; Wang, X.; Chen, Z.; Chen, J.; He, H.; Huang, C. A review for the neuroprotective effects of andrographolide in the central nervous system. Biomed. Pharmacother. 2019, 117, 109078. [Google Scholar] [CrossRef] [PubMed]
- Vijaya, N.R.; Abinaya, R. An effect of cardioprotective activity in various medicinal plants-a review. Int. J. Curr. Pharm. Res. 2019, 11, 1–6. [Google Scholar] [CrossRef]
- Rajendran, H.; Deepika, S.; Immanuel, S.C. An Overview of Medicinal plants for Potential Cardio-Protective Activity. Res. J. Biotechnol. 2017, 4, 104–113. [Google Scholar]
- Shourie, A.; Chandwani, A.; Singh, S.; Rawal, A.; Vijayalakshmi, U. Anticancerous potential of medicinal plants—A review. World J. Pharm. Res. 2018, 6, 462–483. [Google Scholar] [CrossRef][Green Version]
- Kumar, M.S.; Swati, T.; Archana, S.; Hyun, O.S.; Mook, K.H. Andrographolide and analogues in cancer prevention. Front. Biosci. (Elite Ed.) 2015, 7, 255–266. Available online: www.bioscience.org/2015/v7e/af/732/fulltext.htm (accessed on 9 November 2019).
- Gorter, M. The Bitter Constituent of Andrographis Paniculata Nees. Recueil des Travaux Chimiques des Pays-Bas 1911, 30, 151–160. [Google Scholar] [CrossRef]
- Jayakumar, T.; Hsieh, C.-Y.; Lee, J.-J.; Sheu, J.-R. Experimental and Clinical Pharmacology of Andrographis paniculata and Its Major Bioactive Phytoconstituent Andrographolide. Evid. Based Complementary Altern. Med. 2013, 2013, 16. [Google Scholar] [CrossRef]
- Cava, M.P.; Chan, W.R.; Stein, R.P.; Willis, C.R. Andrographolide: Further transformations and stereochemical evidence; the structure of isoandrographolide. Tetrahedron 1965, 21, 2617–2632. [Google Scholar] [CrossRef]
- Smith, A.B.; Toder, B.H.; Carroll, P.; Donohue, J. Andrographolide: An X-ray crystallographic analysis. J. Crystallogr. Spectrosc. Res. 1982, 12, 309–319. [Google Scholar] [CrossRef]
- Medforth, C.J.; Chang, R.S.; Chen, G.-Q.; Olmstead, M.M.; Smith, K.M. A conformational study of diterpenoid lactones isolated from the Chinese medicinal herb andrographis paniculata. J. Chem. Soc. Perkin. 1990, 2, 1011–1016. [Google Scholar] [CrossRef]
- Xia, Y.F.; Ye, B.Q.; Li, Y.D.; He, X.J.; Lin, X.; Yao, X.; Ma, D.; Slungaard, A.; Hebbel, R.P.; Key, N.S.; et al. Andrographolide attenuates inflammation by inhibition of NF-kappa B activation through covalent modification of reduced cysteine 62 of p50. J. Immunol. 2004, 173, 4207–4217. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Fitzloff, J. HPLC with Evaporative Light Scattering Detection as a Tool to Distinguish Asian Ginseng (Panax ginseng) and North American Ginseng (Panax quinquefolius). J. Liq. Chromatogr. Relat. Technol. 2002, 25, 1335–1343. [Google Scholar] [CrossRef]
- Xu, L.; Xiao, D.-W.; Lou, S.; Zou, J.-J.; Zhu, Y.-B.; Fan, H.-W.; Wang, G.-J. A simple and sensitive HPLC–ESI-MS/MS method for the determination of andrographolide in human plasma. J. Chromatogr. B 2009, 877, 502–506. [Google Scholar] [CrossRef]
- Srivastava, A.; Misra, H.; Verma, R.; Gupta, M. Chemical fingerprinting of Andrographis paniculata using HPLC, HPTLC and densitometry. Phytochem. Anal. 2004, 15, 280–285. [Google Scholar] [CrossRef]
- Gu, Y.; Ma, J.; Liu, Y.; Chen, B.; Yao, S. Determination of andrographolide in human plasma by high-performance liquid chromatography/mass spectrometry. J. Chromatogr. B 2007, 854, 328–331. [Google Scholar] [CrossRef]
- Chen, L.; Jin, H.; Ding, L.; Zhang, H.; Wang, X.; Wang, Z.; Li, J.; Qu, C.; Wang, Y.; Zhang, H. On-line coupling of dynamic microwave-assisted extraction with high-performance liquid chromatography for determination of andrographolide and dehydroandrographolide in Andrographis paniculata Nees. J. Chromatogr. A 2007, 1140, 71–77. [Google Scholar] [CrossRef]
- Ding, L.; Luo, X.B.; Tang, F.; Yuan, J.B.; Guo, M.; Yao, S.Z. Quality control of medicinal herbs Fructus gardeniae, Common Andrographis Herb and their preparations for their active constituents by high-performance liquid chromatography–photodiode array detection–electrospray mass spectrometry. Talanta 2008, 74, 1344–1349. [Google Scholar] [CrossRef]
- Du, Q.; Jerz, G.; Winterhalter, P. Separation of andrographolide and neoandrographolide from the leaves of Andrographis paniculata using high-speed counter-current chromatography. J. Chromatogr. A 2003, 984, 147–151. [Google Scholar] [CrossRef]
- Nanduri, S.; Nyavanandi, V.K.; Thunuguntla, S.S.R.; Velisoju, M.; Kasu, S.; Rajagopal, S.; Kumar, R.A.; Rajagopalan, R.; Iqbal, J. Novel routes for the generation of structurally diverse labdane diterpenes from andrographolide. Tetrahedron Lett. 2004, 45, 4883–4886. [Google Scholar] [CrossRef]
- Gomez-Bombarelli, R.; Calle, E.; Casado, J. Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions. J. Org. Chem. 2013, 78, 6868–6879. [Google Scholar] [CrossRef]
- Puri, A.; Saxena, R.; Saxena, R.P.; Saxena, K.C.; Srivastava, V.; Tandon, J.S. Immunostimulant agents from Andrographis Paniculata. J. Nat. Prod. 1993, 56, 995–999. [Google Scholar] [CrossRef]
- Villedieu-Percheron, E. Study about an Anti-Inflammatory Natural Diterpenes Family. Ph.D. Thesis, University of Orléans, Orléans, France, 23 September 2011. Available online: https://tel.archives-ouvertes.fr/tel-00687029 (accessed on 9 November 2019).


| Diterpene | A | B | C | D |
|---|---|---|---|---|
| 1 | 176.7 ± 12 | 138.8 ± 0.2 | 82.0 ± 0.1 | 127.3 ± 0.3 |
| 2 | 95.5 ± 0.7 | 136.9 ± 3.6 | 96.1 ± 2.1 | 51.3 ± 0.3 |
| 3 | 18.3 ± 0.7 | 13.4 ± 0.4 | 26.4 ± 0.4 | 9.8 ± 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villedieu-Percheron, E.; Ferreira, V.; Campos, J.F.; Destandau, E.; Pichon, C.; Berteina-Raboin, S. Quantitative Determination of Andrographolide and Related Compounds in Andrographis paniculata Extracts and Biological Evaluation of Their Anti-Inflammatory Activity. Foods 2019, 8, 683. https://doi.org/10.3390/foods8120683
Villedieu-Percheron E, Ferreira V, Campos JF, Destandau E, Pichon C, Berteina-Raboin S. Quantitative Determination of Andrographolide and Related Compounds in Andrographis paniculata Extracts and Biological Evaluation of Their Anti-Inflammatory Activity. Foods. 2019; 8(12):683. https://doi.org/10.3390/foods8120683
Chicago/Turabian StyleVilledieu-Percheron, Emmanuelle, Véronique Ferreira, Joana Filomena Campos, Emilie Destandau, Chantal Pichon, and Sabine Berteina-Raboin. 2019. "Quantitative Determination of Andrographolide and Related Compounds in Andrographis paniculata Extracts and Biological Evaluation of Their Anti-Inflammatory Activity" Foods 8, no. 12: 683. https://doi.org/10.3390/foods8120683
APA StyleVilledieu-Percheron, E., Ferreira, V., Campos, J. F., Destandau, E., Pichon, C., & Berteina-Raboin, S. (2019). Quantitative Determination of Andrographolide and Related Compounds in Andrographis paniculata Extracts and Biological Evaluation of Their Anti-Inflammatory Activity. Foods, 8(12), 683. https://doi.org/10.3390/foods8120683







